Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The growth and development of the human testis can be divided into three major phases: (1) static, from birth to age 4 years; (2) growth, from age 4 to 10 years; and (3) developmental (maturation), from age 10 years to puberty. At birth, the seminiferous tubules are compactly filled with small undifferentiated cuboidal cells. Leydig cells are seen in the newborn because of the influence of maternal hormones, but then disappear to reappear only later. Increases in tubule and cell size are slow and gradual, with barely perceptible growth from age 4 to 10 years, increasing tortuosity, and lumen formation. At age 10 years, a definite spurt is noted, coinciding with the appearance of gonadotropins and 17-ketosteroids in urine. Mitotic figures appear in the tubular cells, and Leydig cells again become recognizable in the interstitium. At age 11 years, mitotic activity is pronounced, and primary and secondary spermatocytes appear. At age 12 years, spermatids are numerous. Finally, spermatozoa appear. The number of maturing tubules with active spermatogenesis increases gradually until the adult stage is reached. Because of the great variation in the age at which puberty normally occurs, the age of the individual cannot be determined by histologic study of the testis after the 12th year.
The normal adult testis is a paired organ that lies within the scrotum suspended by the spermatic cord. The average weight of each testis is 15–19 g, the right usually being 10% heavier than the left. The organ is covered by a capsule composed of three layers: the outer serosa or tunica vaginalis (covered by a flattened layer of mesothelial cells), the tunica albuginea , and the inner tunica vasculosa . The posterior portion of the capsule, called the mediastinum, contains blood and lymph vessels, nerves, and the mediastinal portion of the rete testis. The parenchyma is divided into approximately 250 lobules, each lobule containing up to four seminiferous tubules. The usual testicular biopsy is composed of three to five lobules together with portions of the septa (which should not be misinterpreted as areas of fibrosis). The seminiferous tubules are bound by a limiting membrane made up of basal lamina and alternating layers of myoid cells and collagen fibers. These tubules contain germ cells in various stages of development and Sertoli cells.
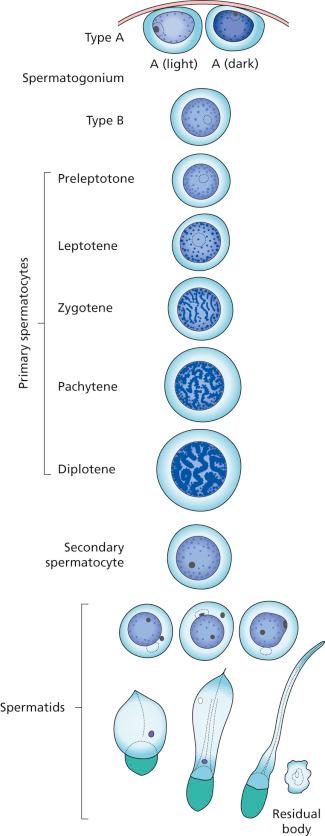
The maturation of germ cells in man covers a period of about 70 days and follows these steps: spermatogonium (of which two types have been described, known respectively as A and B), primary spermatocyte (further subdivided depending on the stage of meiosis into preleptotene, leptotene, zygotene, pachytene, and diplotene), secondary spermatocyte, spermatid, and (mature) spermatozoon ( Fig. 27.1 ). Until spermatozoa are formed, all progeny of a spermatogonium are held together by a narrow cytoplasmic bridge. This maturation proceeds in an orderly overlapping helical pattern along the length of the tubule ; as a consequence, not all stages of differentiation of germ cells are seen in any one cross-sectional view of a seminiferous tubule. This normal pattern should not be confused with a maturation arrest .
Sertoli cells have a columnar shape, lie on the basement membrane of the tubule, and send cytoplasmic prolongations around the germ cell elements. Their nuclei are irregularly shaped, are highly folded, and have a prominent nucleolus. The cytoplasm may contain Charcot–Bottcher crystalloids, which represent bundles of filaments. Immunohistochemically, the intermediate filament of Sertoli cells is vimentin, but coexpression of keratin and desmin is present in the fetal testis and can reappear in pathologic conditions.
The interstitium contains stromal elements (including the already mentioned myoid cells) and Leydig cells. The latter occur either singly or in clusters and are often associated with nerve fibers. Their cytoplasm contains lipid droplets, lipochrome pigment, and sometimes Reinke crystalloids, which appear ultrastructurally as hexagonal prisms. Immunohistochemically, there is reactivity for inhibin and Melan-A (Mart-1). Leydig cells gradually decrease in number with aging.
In 1 out of every 10 males, the testis has not descended into the scrotum at the time of birth, but has remained in the inguinal region or abdomen, the former situation being about four times as common as the latter. Most of these “retained” or “retractile” testes descend into the scrotum during the first year of life. In only 1 in 100 individuals will a permanent retention of the testis outside the scrotum occur, a condition known as cryptorchidism. The exact pathogenesis is unknown, but most evidence favors a role for testosterone under the influence of the hypothalamic–pituitary axis. If by the age of 2 or 3 years the testis has not spontaneously descended into the scrotum (or sooner, if the abnormality is bilateral), an open or laparoscopic orchiopexy should be carried out; otherwise permanent anatomic alterations will occur.
Cryptorchidism is unilateral in 80% of the cases. Of patients with bilateral cryptorchid testes in whom repair is performed before the age of 5 years, 50% are fertile and 31% are normospermic. The microscopic appearance of the prepubertal testicle as seen in a small biopsy at the time of orchiopexy is a good predictor of the degree of post-pubertal spermatogenesis and, therefore, of fertility.
Grossly, cryptorchid testes in adults are small and brown. The testicular tubules are atrophic, and their basement membrane is greatly thickened. The Leydig cells are prominent; some of them may be present inside the tubules. Not infrequently, foci of hyperplastic Sertoli cells occur. They are usually multiple and may appear grossly as minute white nodules. In some instances, atypical germ cells are seen at the base of the tubules; these are indicative of germ cell neoplasia in situ (GCNIS, intratubular germ cell neoplasia [IGCN]) and are the forerunner of malignancy. Traditionally, the atrophy and infertility associated with cryptorchidism have been attributed to the higher intrascrotal temperature. However, the facts that boys with cryptorchidism have histologically abnormal seminiferous tubules also in the contralateral descended testis and that men who had unilateral cryptorchidism corrected in childhood have lower sperm counts and higher serum concentrations of follicle-stimulating hormone as adults than do normal men suggest that even unilateral cryptorchidism is associated with a bilateral testicular abnormality.
A cryptorchid testis is more likely to develop a germ cell tumor than is a normally placed organ. Of 7000 cases of testicular germ cell tumors reviewed in a classic study by Gilbert and Hamilton, 10.9% occurred in cryptorchid organs. The incidence of malignancy is greater in the abdominal than in the inguinal testis. Seminoma is the most common type, but other germ cell tumors also occur. The contralateral testis in patients with unilateral cryptorchidism also has an increased incidence of malignancy. If a cryptorchid testis is surgically placed in the scrotum, it may still develop a germ cell neoplasm, especially if the operation has been done at a late age. Dow and Mostofi reported 14 cases of such tumors occurring in patients who had had orchiopexy between the ages of 11 and 36 years. In a large Swedish study, the relative risk of testicular cancer among cryptorchidic patients who underwent orchiopexy before reaching 13 years of age was 2.23 as compared to the Swedish general population, whereas for those treated at 13 years of age or older, the relative risk was 5.40. Because of these findings, it has been recommended that high-positioned testes that have not been surgically placed into the scrotum before mid-adolescence be removed.
Atrophy of the testis may result from a large variety of causes: the already mentioned cryptorchidism; the orchitis of mumps, especially when the infection occurs at or after puberty ; liver cirrhosis, as a result of increased circulating endogenous estrogens not metabolized by the diseased liver ; administration of estrogens or gonadotropin-releasing hormone analogue in the treatment of prostatic carcinoma ; radiation exposure; chemotherapy, particularly cyclophosphamide ; and exposure to an environmental toxin, the nematocide dibromochloropropane.
In advanced testicular atrophy of any of these etiologies, the tubules are small, with thick basement membranes and few or no germ cells. The interstitial tissue shows varying degrees of fibrosis, and there may be an increased number of Leydig cells.
An extreme degree of testicular atrophy occurs in the testicular regression (“vanishing testis”) syndrome, a condition characterized by a rudimentary epididymis and spermatic cord with no identifiable testicular tissue. There is instead in this location a dense fibrovascular tissue (“nubbin”) with foci of calcification and hemosiderin deposition. The suggestion has been made that this condition results from testicular infarct, presumably caused by torsion occurring in utero. It affects approximately 5% of patients with cryptorchidism.
Various degrees of testicular atrophy occur frequently in AIDS-affected patients, the precise pathogenesis being unclear.
The effect of vasectomy on testicular morphology is minimal but detectable; there is thickening of the tubular wall, reduction in the number of spermatids and Sertoli cells, and sometimes foci of interstitial fibrosis.
The male factor is the primary or contributing cause of infertility in 40%–60% of the cases. The causes of male infertility fall into one of three categories: pretesticular, testicular, and post-testicular. The pretesticular causes are extragonadal endocrine disorders, usually originating in the pituitary or adrenal gland. The testicular causes are primary diseases of the testes, and little treatment is available at the present time. The post-testicular causes consist mainly of obstructions of the ducts leading away from the testes. These obstructions may be congenital, postinflammatory, or postsurgical and include Young syndrome, in which the obstructive azoospermia is combined with chronic sinopulmonary infections. These obstructions have generally little or no adverse effect on spermatogenesis. Treatment consists of epididymovasostomy or vasovasostomy to bypass the obstruction. Impaired sperm mobility, presumably because of faulty maturation or storage of spermatozoa in the epididymis, and immunologic factors are included among the post-testicular causes of infertility.
The evaluation of the infertile male includes a thorough clinical history and examination, semen analysis, quantitation of leukocytes in semen, and search for antisperm antibodies. Tests of sperm function include evaluation of cervical mucus interaction, ova penetration, and the hemizona assay. Additional tests, performed in selected cases, are transrectal ultrasonography, venography, and testicular biopsy. The latter is particularly useful in the individual with azoospermia and normal endocrine findings. Punch biopsy is not nearly as satisfactory as an open surgical biopsy. The material should be handled with extreme care. Zenker and Bouin fixatives preserve cytologic features better than formalin; however, some hospitals no longer allow their use. Fine-needle aspiration cytology has been proposed as an alternative to testicular biopsy, some studies showing a high correlation between the two methods.
Biopsy specimens from infertile men with total lack of spermatozoa (azoospermia) usually show one of the following conditions :
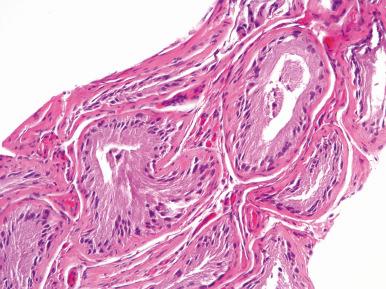
Germ cell aplasia (Sertoli cell-only syndrome) (29%), in which the tubules are populated by only Sertoli cells ( Fig. 27.2 ), measure 100–150 µm in diameter, and may show some thickening of the tubular basement membrane; germ cells are completely absent. Leydig cells are usually normal but on occasion are found to be reduced in size and number.
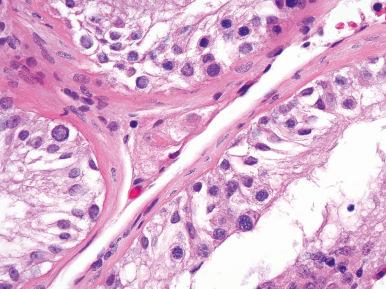
Spermatocytic arrest (26%), characterized by a halt of the maturation sequence ( Fig. 27.3 ), usually at the stage of the primary spermatocyte (presumably at the end of the meiotic prophase at late pachytene) ; no spermatids or spermatozoa are present despite the presence of abundant cells in division; Leydig cells are normal.
Generalized fibrosis (18%).
Normal spermatogenesis (27%).
The last finding suggests the diagnosis of obstructive azoospermia and implies the bilateral obstruction or absence of some part of the duct system. In the series of Nistal et al., of patients with obstructive azoospermia of known etiology, over half had a normal testicular biopsy, whereas most of the others exhibited subtle alterations of the adluminal compartment of the seminiferous tubules (early or late sloughing of primary spermatocytes), possibly induced by the increase in hydrostatic pressure.
In patients with a reduction in the number of spermatozoa (oligospermia), the following microscopic patterns are recognizable, often combined: (1) incomplete spermatocytic arrest, characterized by arrest of spermatogenesis in some of the tubules; (2) regional or incomplete fibrosis; (3) spermatogenic hypoplasia (hypospermatogenesis), characterized by tubules with a reduced population of germ cells and a poor order of spermatogenesis; (4) tubular hyalinization, a probably heterogeneous group that includes Klinefelter syndrome, in which the tubules have a small diameter and a markedly thickened basement membrane, and Leydig cells are often increased in number; (5) mixed atrophy, that is, the synchronous occurrence of seminiferous tubules containing germ cells and tubules with Sertoli cells only; and (6) normal or essentially normal spermatogenesis, usually implying a partial occlusion of some part of the duct system. Another pattern sometimes described is that of sloughing and disorganization, in which spermatogenesis has a disorderly appearance and the tubular lumina are filled with desquamated immature cells. We have found that this pattern is rather nonspecific, since it can be seen in spermatogenic hypoplasia as well as with testicular blockage; it also can be closely simulated by artifactual detachment of germ cells resulting from rough handling of the biopsy. The degree of spermatogenic hypoplasia, a very subjective evaluation, can be estimated with quantitative techniques.
Multinucleated giant stromal cells of reactive nature have been observed in cases of testicular atrophy after estrogen therapy.
In the series of over 800 cases of azoospermia studied via testicular biopsy by Girgis et al., the etiology was obstructive in about 55%. In more than half of these, epididymovasostomy proved beneficial. Patients with varicocele are often oligospermic and infertile. The most common patterns at biopsy are those of spermatogenic hypoplasia plus sloughing and disorganization. The azoospermia commonly seen in patients with cystic fibrosis is obstructive, secondary to structural abnormalities of the epididymis and vas deferens.
Biopsies are rarely done in patients with testicular failure involving primarily the endocrine function, since the diagnosis is usually established by hormonal determinations. Failure of pubertal maturation is the cardinal symptom if the disease is congenital or begins in childhood. Three different patterns can be seen in testicular biopsies: (1) hypogonadotrophic eunuchoidism (60%), accompanied by low gonadotropin levels and characterized by small, infantile tubules with scattered spermatogonia and Sertoli cells but few or no Leydig cells; (2) Klinefelter syndrome (30%), with fibrosis of tubules, prominent thickening of the basement membrane, and Leydig cell hyperplasia ; and (3) testicular aplasia (10%), characterized by the absence of testicular tissue and elevation of urinary gonadotropin levels. Patients with Klinefelter syndrome have an increased incidence of breast carcinoma ; cases of Leydig cell tumor and testicular and extragonadal (mainly mediastinal) germ cell tumors also have been reported in this population.
Infarct of the testis is usually the result of torsion of the spermatic cord, but it may also be due to venous thrombosis secondary to pyogenic epididymo-orchitis (see later), a sequela of inguinal hernia repair, or, rarely, vasculitis of various causes, such as that accompanying Crohn disease. Intermittent torsion may cause histologic changes that mimic other etiologies.
Viral-type orchitis can simulate testicular neoplasia at the microscopic level. The architecture is preserved, but there is hemorrhage, edema, and a patchy lymphohistiocytic infiltrate within and between the seminiferous tubules.
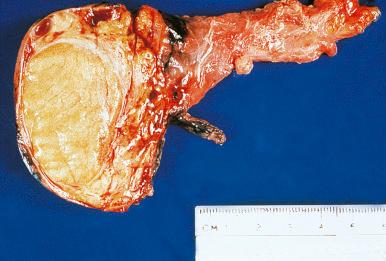
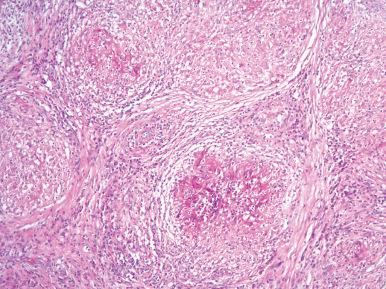
Granulomatous orchitis is characterized grossly by a solid nodular enlargement of the testis ( Fig. 27.4 ) and microscopically by granulomatous lesions centered in the seminiferous tubules, as described in the classic paper by Spjut and Thorpe. Epithelioid cells, multinucleated giant cells, lymphocytes, and plasma cells are present. The granulomatous response is at least in part secondary to the products of disintegrated sperm. A history of trauma to the region is often obtained. The clinical evolution is benign, but this should obviously be distinguished from infectious etiologies.
Malakoplakia can affect the testis, either alone or accompanied by epididymal involvement. It is associated with abscess formation, atrophy of tubules, and the characteristic Michaelis–Gutmann bodies. Thrombosed blood vessels are often found. Bacteria (mainly coliform organisms) may be found on culture. Three other proliferative histiocytic disorders that occasionally involve the testis are juvenile xanthogranuloma, Rosai–Dorfman disease (sinus histiocytosis with massive lymphadenopathy), and xanthogranulomatous orchitis.
Pyogenic epididymo-orchitis may be complicated by venous thrombosis and septic infarct of the testis. Escherichia coli is the organism usually recovered. Microscopically, many similarities exist between this condition and granulomatous orchitis, suggesting a common ischemic background.
Tuberculosis, atypical mycobacteriosis, leprosy, sarcoidosis, syphilis, and Crohn disease can involve the testis ( Fig. 27.5 ). Clinically, a syphilitic gumma can resemble a neoplasm.
Other infectious agents responsible for orchitis are Toxoplasma (in immunosuppressed patients), fungi, parasites, and Brucella . These are rarely seen as surgical specimens.
Multinucleated stromal giant cells are a relatively common and clinically insignificant finding in the testis. They seem to be age related and are said to be particularly common in cases of testicular atrophy due to estrogen therapy.
Necrotizing vasculitis can involve the testis, either as a manifestation of polyarteritis nodosa or other systemic disease or as an isolated finding.
Ectopic spleen in the scrotum (splenogonadal fusion syndrome) results from a congenital malformation occurring at a stage when the splenic and gonadal anlages are topographically close. In the continuous variant, there is a fibrous or splenic tissue-containing cord that connects the spleen and the scrotal ectopic splenic tissue. In the discontinuous variant, the connection is lost, and the ectopic splenic tissue appears as a scrotal accessory spleen, which may clinically simulate a tumor. All of the reported cases have been on the left side.
Cystic dysplasia of the testis is a rare congenital defect that results in the formation of numerous irregularly shaped cystic spaces within the mediastinum testis. The cysts are lined by flattened cuboidal epithelium that morphologically and immunohistochemically resembles the epithelium of rete testis. Thus this condition merges with those described in the section on rete testis .
Cysts located within the testicular parenchyma away from the rete testis are lined by a similar flattened cuboidal (non-ciliated) epithelium and may have a similar pathogenesis.
Testicular implants (silicone gel-filled) can elicit a tissue reaction similar to that seen in breast implants. Microscopically, it is characterized by a fibrous capsule infiltrated by lymphocytes and foamy macrophages, and occasionally featuring a synovial-like cleft at the interphase.
Testicular tumors can be divided into five general categories: germ cell tumors (90%) arising from the germinal epithelium of the seminiferous tubules; sex cord–stromal tumors; mixed germ cell–sex cord–stromal tumors; primary tumors not specific to the testis; and metastatic tumors.
This current classification of testicular neoplasia is based on the recent restructuring of terminology recommended by the WHO 2016 classification. In this system, germ cell tumors are divided into categories based on their relationship with germ cell neoplasia in situ (GCNIS), previously termed intratubular germ cell neoplasia, unclassified (ITGCN). In addition, minor changes to the nomenclature of sex cord–stromal tumors are based on more recent molecular genetic data.
Although somewhat confusing when approached from a histologic perspective, urologists and oncologists divide testicular germ cell tumors into two categories: “seminoma” (pure classic seminoma) and “non-seminoma” (any other pure or mixed subtype, including those with an admixed seminoma component). The latter is often described as “non-seminomatous germ cell tumor” (NSGCT).
Testicular germ cell tumors make up only a small percentage of all malignant neoplasms in men (approximately 1%), but are the most common malignant tumors in young men between 25 and 29 years of age. The worldwide incidence is 1.5 cases per 100,000, with rates highest in Norway and Switzerland. Their incidence seems to be increasing, at least for seminomas.
The main factors thought to predispose to testicular germ cell tumors are cryptorchidism, disorders of sex development, family history, testicular microliths in subfertility, and prior germ cell tumor. Hereditary forms are described, but almost all are related to low penetrance autosomal recessive susceptibility genes. Specifically, the AZFc region (gr/gr microdeletion) of chromosome Y, 12q22 (KITLG), and 5q31 (RAS-ERK-MAPK pathway) are proposed as susceptibility loci, in some cases possibly due to interactions with c-KIT. It is also suggested that PTEN hamartoma tumor syndrome, Li–Fraumeni syndrome, and neurofibromatosis type 1 may rarely be associated with germ cell tumors.
There is a good correlation between age and the incidence of the different types of testicular tumor. The median age at presentation is 35 years for seminoma, 25 years for non-seminoma, and 30 years from non-seminomas that include a seminomatous component. In individuals over 60 years of age, germ cell tumors are rare. Seminomas (and spermatocytic tumor described below with germ cell tumors unrelated to GCNIS) are the most common germ cell tumors in this age group, outnumbered only by malignant lymphomas.
Most testicular germ cell tumors present with progressive, painless enlargement of the testis. A small tumor may be found in a testis by palpation or ultrasonography. They may grow slowly or with appalling speed. Sometimes, the initial presentation is in the form of a metastatic deposit in the retroperitoneum, lung, or mediastinum. Presentation with significant clinical symptomatology is uncommon in seminoma, but relatively common for choriocarcinoma, in which the patient may have gynecomastia; large mediastinal, pulmonary, liver and/or brain metastases; and markedly increased levels of serum human chorionic gonadotropin (hCG).
Bilateral testicular involvement by germ cell tumors is seen in 1.0%–2.7% of the cases according to different series. The risk of bilaterality rises to 15% if both testes are undescended. It can be seen as a synchronous or, more commonly, as a metachronous event. The tumors may have a different histology. In the presence of bilateral testicular tumors in an elderly patient, the most likely diagnosis is malignant lymphoma.
Germ cell tumors of adults and adolescents (almost always of the post-pubertal GCNIS-associated type) show one or more copies of an isochromosome of chromosome 12p (i(12p)) or other forms of 12p amplification. Since this genetic aberration is absent in isolated GCNIS, the gain of genes on 12p appears to be crucial for the development of invasive germ cell tumor. 12p harbors multiple genes that promote cell growth. Other common non-random genetic changes include loss of chromosome 1p, 11, 13, 18, or Y and gain of chromosome 21, 7, 8, or X. KIT mutation and/or amplification (most commonly in seminoma) and KRAS mutation are also described in a small subset of cases. In cases of NSGCT with an admixed seminoma component, in which the two components were evaluated separately from a genetic standpoint, some showed similar clonal abnormalities (suggesting an origin from the same stem cell), whereas others did not.
Spermatocytic tumor shows completely different genetic features (with an absence of 12p abnormality), and are discussed separately.
It has been generally assumed that the first manifestation of local spread of testicular germ cell tumors is in the tunica albuginea, but invasion of the testicular hilum is actually a much more common event. Since this may be grossly inapparent, microscopic sampling of this region is mandatory, as discussed in staging.
In terms of lymph-borne metastases, testicular tumors spread first to periaortic and iliac lymph nodes and later to mediastinal and left supraclavicular nodes. Retroperitoneal lymph node metastases are on the side of the tumor in about 80%–86% of cases and bilateral in about 13%–20%. The presence of contralateral metastases in the absence of ipsilateral metastases is rare. Characteristically, the first retroperitoneal metastases are highly placed, immediately below the renal vessels. In general, involvement of inguinal lymph nodes does not occur unless there is invasion of the skin of the scrotum, tumor recurrence in a cutaneous scar, or a history of previous operation in the area (such as correction of cryptorchidism, herniorrhaphy, or scrotal orchiectomy). Blood-borne metastases occur most frequently in lungs, liver, brain, and bone. There is a high incidence of brain metastases in choriocarcinoma and of bone metastases in seminoma. Embryonal carcinoma frequently metastasizes early, and choriocarcinoma almost always has widespread metastases by the time the tumor is diagnosed. Teratoma occurring in the post-pubertal testis can be accompanied by metastases no matter how well-differentiated the primary tumor appears, which is why the mature/immature designation is no longer recommended.
The microscopic appearance of the metastases may differ from that of the primary tumor. In general, classic seminoma metastasizes as classic seminoma, but it may also spread as other types. In combined mixed germ cell tumors with admixed seminoma, the non-seminomatous component is the one most likely to metastasize. Late metastases of testicular germ cell tumors often have a teratomatous appearance. Mixed germ cell tumors with focal choriocarcinoma tend to metastasize as pure choriocarcinomas. Surgical removal of the postchemotherapy residual tumors is recommended. In some instances, total spontaneous regression of the primary tumor or of the metastatic foci has been observed. Sometimes, tumor foci with a somatic-type malignancy (rather than germ cell) appearance develop in these metastases.
The microscopic appearance of the lung metastases after chemotherapy correlates well with prognosis. The presence of total necrosis or fully mature elements is indicative of a good prognosis ; viable non-teratomatous components are associated with a decreased survival, and the presence of sarcomatous elements is a particularly ominous sign.
The initial treatment for all testicular germ cell tumors is inguinal orchiectomy with high ligation of the spermatic cord (“radical orchiectomy”). The subsequent treatment of confirmed low-stage testicular germ cell tumors is evolving. Depending on the institution, orchiectomy may be followed by retroperitoneal lymphadenectomy, chemotherapy, or surveillance. Radiation therapy is used less commonly. Many groups have recommended treating stage I seminoma or even NSGCT with orchiectomy alone, with further therapy for only those cases that show relapse on close monitoring. Along similar lines, it has been suggested that stage II NSGCT could be treated with orchiectomy and retroperitoneal lymphadenectomy, with chemotherapy reserved for those patients who develop relapse. The “surveillance only” approach is more controversial in patients with a predominance of the embryonal carcinoma or in tumors exhibiting blood vessel invasion because of the high probability of distant metastases under these circumstances; however, high-risk pathology is not an absolute contraindication in certain settings, given that therapies still have high cure rates for relapses.
Controversy exists as to whether patients undergoing orchiectomy for germ cell tumors should also have a biopsy of the contralateral testis searching for IGCN. Most authors do not favor this approach, but the practice varies significantly by country. We never receive such biopsies.
With current treatment protocols, testicular germ cell tumors represent the most curable solid cancer in men, even when metastatic spread is present. For stage I tumors, whether seminoma or non-seminoma, cure rates are close to 100%. With metastatic spread, cure rates remain high, but vary with prognostic groups as defined by the International Germ Cell Cancer Collaborative Group classification. Even in this metastatic group, overall cure rates are over 80%, and salvage therapy may cure over half of the patients with initial treatment failures. Later relapses (defined as >2 years after initial treatment with complete remission) are less common, but may occur and portend an adverse prognosis, particularly when associated with secondary somatic malignancy.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here