Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Systemic lupus erythematosus (SLE) is the prototypic systemic autoimmune disease. It is characterized by multiple autoantibodies associated with a multisystem illness where any organ can be targeted. Autoantibodies most commonly associated with SLE are antinuclear antibodies (ANAs) and anti–double-stranded (native) DNA (anti-dsDNA). The presentation, disease course, and outcome of SLE are unpredictable. Patients often experience disease flares and more rarely remissions. Childhood-onset SLE (cSLE) patients typically have more severe disease than adults, and if untreated, the 5-year mortality rate reaches 95.3%. Treatment requires the clinician to balance immunosuppression, reducing organ damage, and autoimmune dysfunction with morbidity from current therapies.
To improve the clinician’s ability to identify the disease, as well as the researcher’s ability to study the disease, the American Rheumatism Association, now known as the American College of Rheumatology (ACR), developed a classification system in 1971, modified in 1982 and 1997 ( Table 23.1 ). , The Systemic Lupus International Collaborating Clinics (SLICC) reviewed the classification system and published a new set of criteria in 2012 ( Box 23.1 ).
| Criterion | Definition |
|---|---|
| Malar rash | Fixed erythema, flat or raised, over the malar eminences, tending to spare the nasolabial folds |
| Discoid rash | Erythematosus raised patches with adherent keratotic scaling and follicular plugging: atrophic scarring may occur in older lesions |
| Photosensitivity | Skin rash as a result of unusual reaction to sunlight, by patient history or physician observation |
| Oral ulcers | Oral or nasopharyngeal ulceration, usually painless, observed by physician |
| Arthritis | Nonerosive arthritis involving two or more peripheral joints, characterized by tenderness, swelling, or effusion |
| Serositis | Pleuritis—convincing history of pleuritic pain or rubbing heard by a physician or evidence of pleural effusion or pericarditis—documented by ECG or rub or evidence of pericardial effusion |
| Renal disorder | Persistent proteinuria greater than 0.5 g/day (or >3+ if quantitation nor performed) or cellular casts—may be red cell, hemoglobin, granular, tubular, or mixed |
| Neurological disorder | Seizures in the absence of offending drugs or known metabolic derangements (e.g., uremia, ketoacidosis, or electrolyte imbalance) or psychosis in the absence of offending drugs or known metabolic derangements (e.g., uremia, ketoacidosis, or electrolyte imbalance) |
| Hematological disorder | Hemolytic anemia with reticulocytosis or leukopenia less than 4000/mm 3 total on two or more occasions, or lymphopenia less than 1500/mm 3 on two or more occasions, or thrombocytopenia less than 100,000/mm 3 in the absence of offending drugs |
| Immunological disorder | 1. Positive anti-DNA antibody to native DNA in abnormal titer, or 2. Presence of anti-Sm nuclear antigen, or 3. Positive finding of antiphospholipid antibodies based on: a. an abnormal serum level of IgG or IgM anticardiolipin antibodies b. a positive test result for lupus anticoagulant using a standard method, or c. a false-positive serological test and confirmed by Treponema pallidum immobilization or fluorescent treponemal antibody absorption test. |
| Antinuclear antibody | An abnormal titer of antinuclear antibody by immunofluorescence or an equivalent assay at any point in time and in the absence of drugs known to be associated with drug-induced lupus syndrome |
Acute cutaneous lupus, including:
Lupus malar rash (do not count if malar discoid)
Bullous lupus
Toxic epidermal necrolysis variant of SLE
Maculopapular lupus rash
Photosensitive lupus rash
in the absence of dermatomyositis
OR subacute cutaneous lupus (nonindurated psoriasiform and/or annular polycyclic lesions that resolve without scarring, although occasionally with postinflammatory dyspigmentation or telangiectasias)
Chronic cutaneous lupus, including:
Classic discoid rash
Localized (above the neck)
Generalized (above and below the neck)
Hypertrophic (verrucous) lupus
Lupus panniculitis (profundus)
Mucosal lupus
Lupus erythematosus tumidus
Chilblain lupus
Discoid lupus/lichen planus overlap
Oral ulcers
Palate
Buccal
Tongue
OR nasal ulcers
In the absence of other causes, such as vasculitis, Behçet disease, infection (herpesvirus), inflammatory bowel disease, reactive arthritis, and acidic foods
Nonscarring alopecia (diffuse thinning or hair fragility with visible broken hairs)
In the absence of other causes such as alopecia areata, drugs, iron deficiency, and androgenic alopecia
Synovitis involving two or more joints, characterized by swelling or effusion
OR tenderness in two or more joints and at least 30 minutes of morning stiffness
Serositis
Typical pleurisy for more than 1 day
OR pleural effusions
OR pleural rub
Typical pericardial pain (pain with recumbency improved by sitting forward) for more than 1 day
OR pericardial effusion
OR pericardial rub
OR pericarditis by electrocardiography
OR pericardial effusion
OR pericardial rub
OR pericarditis by electrocardiography
In the absence of other causes, such as infection, uremia, and Dressler pericarditis
Renal
Urine protein-to-creatinine ratio (or 24-hour urine protein) representing 500 mg protein/24 hours
OR red blood cell casts
Neurological
Seizures
Psychosis
Mononeuritis multiplex
In the absence of other known causes such as primary vasculitis
Myelitis
Peripheral or cranial neuropathy
In the absence of other known causes such as primary vasculitis, infection, and diabetes mellitus
Acute confusional state
In the absence of other causes, including toxic/metabolic, uremia, drugs
Hemolytic anemia
Leukopenia (<4,000/mm 3 at least once)
In the absence of other known causes such as Felty syndrome, drugs, and portal hypertension
OR
Lymphopenia (<1,000/mm 3 at least once)
In the absence of other known causes such as corticosteroids, drugs, and infection
Thrombocytopenia (<100,000/mm 3 ) at least once
In the absence of other known causes such as drugs, portal hypertension, and thrombotic thrombocytopenic purpura
Immunological Criteria
ANA level above laboratory reference range
anti-dsDNA antibody level above laboratory reference range (or 2-fold the reference range if tested by ELISA)
Anti-Sm: the presence of antibody to Sm nuclear antigen
Antiphospholipid antibody positivity as determined by
positive test for lupus anticoagulant
false positive test result for rapid plasma regain
medium or high titer anticardiolipin antibody level (IgM, IgA or IgG)
positive test result for anti-2-glycoprotein I (IgM, IgA or IgG)
low complement (C3, C4 or CH50)
Direct Coombs’ test (in the absence of hemolytic anemia)
According to the ACR system, four of 11 ACR criteria must be found in a patient to fulfill the classification of SLE (definite lupus). When applied to a small cohort of children, the criteria were 96% sensitive and 100% specific. The SLICC criteria include 11 clinical and six immunological items, requiring four items with at least one clinical and one immunological item or biopsy proven nephritis compatible with lupus in the presence of an ANA or anti-dsDNA. Several studies have sought to determine the specificity and sensitivity of the SLICC classification criteria in pediatric lupus. , SLICC criteria have slightly improved sensitivity with slightly decreased specificity from the ACR criteria. The European League Against Rheumatism (EULAR)/ACR classification criteria have been recently published and require the presence of ANAs at a titer of 1:80 or greater as an “entry criterion.” Clinical and laboratory criteria are weighted, and a total score of 10 or more, if the entry criterion is fulfilled, meets the requirements for a classification of SLE ( Table 23.2 ). Definitions of the criteria are provided in the reference. Further study of larger populations and children are needed to validate this classification. Although the classification criteria are excellent guides, clinicians should use clinical judgment to determine the diagnosis of individual patients.
| Entry Criterion | Antinuclear Antibody Positivity at Titer of 1:80 or Greater | |
|---|---|---|
|
Weight | |
|
|
2 points |
|
|
3 points |
|
4 points | |
|
4 points | |
|
|
2 points |
|
3 points | |
|
5 points | |
|
|
2 points |
|
2 points | |
|
4 points | |
|
6 points | |
|
|
5 points |
|
6 points | |
|
|
6 points |
|
|
4 points |
|
8 points | |
|
10 points | |
|
||
|
|
2 points |
|
||
|
3 points | |
|
4 points | |
|
||
| Anti-dsDNA OR anti-Smith | 6 points |
Reports of the incidence and prevalence of cSLE are variable depending on factors such as access to care, referral patterns, ethnic diversity, and the ability to ascertain cases for calculation. The overall incidence of lupus varies from 0.36 to 2.5 per 100,000. Differences are likely the result of data collection methods and ethnic diversity. The low prevalence of cSLE makes clinical research challenging.
cSLE has been estimated to account for 10% to 20% of all cases of SLE. The average age of onset is 12 years, with rare cases occurring at 5 years of age or under, more often in non-Caucasian populations.
The gender distribution of cSLE is 4.5:1 to 5:1 (female:male) compared with 9:1 to 10:1 in the adult population. The lower ratio has been attributed to differences in hormone status between children and adults.
Although SLE is found worldwide, several studies on incidence and prevalence demonstrate higher rates of lupus in non-Caucasian populations including in Aboriginal, Latino, Asian, and Afro-Caribbean populations. ,
Lupus is a very complex disease of immune dysregulation with alterations in both innate and adaptive immunity. Initial observations about the immune mechanisms of lupus come from studies of spontaneous and induced immune dysregulation in mice leading to lupus-like syndromes. Advances in technology have led to many seminal observations including identification of monogenic causes of lupus, such as complement deficiencies, genetic overproduction of interferon-alpha (INF-α), and apoptosis defects.
The current hypotheses associated with the generation of lupus and loss of tolerance start with a host defense response gone awry. Abnormal regulation of type I IFNs (IFN-α), which stimulate production of innate immune cells and the inflammatory response, is well described. Apoptosis or cell death requires rapid clearance of intracellular debris. However, in lupus, defects in apoptotic clearance and/or regulation of clearance allow presentation of intracellular materials, including autoantigens, RNA, and DNA. Defects in complement, complement receptors, immunoglobulin receptors, Bcl2, Fas/Fas ligand, and programmed cell death 1 (PD1) can all contribute to different rates of clearance of apoptotic materials. Phagocytic immune cells respond to specific RNA and DNA motifs that have been endocytosed through Toll-like receptor (TLR) activation of TLR7 and TLR9. TLR signaling leads to persistent overproduction of proinflammatory cytokines including type I INFs.
Complement plays a dual and seemingly paradoxical role in the pathogenesis of cSLE lupus nephritis (LN). Hereditary complement deficiencies, such as homozygous C1, C2, or C4 deficiencies, are associated with a high incidence of LN because of impaired clearance of apoptotic cells and immune complexes (ICs). Anti-C1q antibodies have been identified that interfere with the interaction between C1q, immunoglobulin (Ig)G, and C-reactive protein (CRP), likely resulting in a functional C1q deficiency and impaired clearance of apoptotic cells and ICs. Therefore, certain complement components might be protective against SLE and LN by clearing apoptotic fragments and preventing the accumulation of chromatin. In contrast, complement activation also plays a role in glomerular tissue damage.
In lupus patients, neutrophil apoptosis is increased and dysregulated, leading to increased autoantigen exposure, especially in the presence of autoantibodies, particularly anti-ribonucleoprotein (RNP) antibodies releasing neutrophil extracellular traps (NETs). This material is rich in DNA, LL37, and high-mobility group protein B1 (HMGB1). The latter proteins are associated with activation of plasmacytoid dendritic cells (pDCs) by mammalian DNA. pDCs have been demonstrated to produce high levels of IFN and are associated with active lupus. ,
Lupus patients have abnormalities in B cells, T cells, and antigen-presenting cells. , The hallmark of lupus is the production of autoantibodies and hypergammaglobulinemia. Several different mechanisms can account for this, including antigen stimulation, polyclonal B-cell proliferation, T-cell dysregulation, and intrinsic B-cell defects. The result is antibody-mediated disease by direct cellular injury, seen as cytopenias, as well as by ICs resulting from an immune system that has an ineffective or overwhelmed clearance mechanism, as seen in LN and vasculitis. IC formation leads to activation of complement, leading to inflammation and tissue damage. Autoantibodies such as anti-dsDNA are thought to arise from somatic mutation induced by environmental stimuli or from autoantigens such as nucleosomes or chromatin. T-cell dysregulation is marked by T-cell cytopenia, particularly of natural killer (NK) cells and regulatory T cells, leading to cytokine profiles that support B-cell activation. Furthermore, overexpression of CD40/CD40 ligand leads to B-cell activation.
Dendritic cells have a central role in the pathophysiology of lupus. pDCs, activated by ICs, produce INF-α. FcγRIIa on the pDC membrane delivers ICs to lysosomal TLRs (TLR7 for RNA containing ICs and TLR9 for DNA containing ICs). TLR-induced production of INF-α leads to differentiation of monocytes into dendritic cells (DCs) and activation of immature myeloid DCs (mDCs). These subsequently activate autoreactive T and B cells, leading to increased autoantibody production. Activated mDCs also cause CD8 + T cells to differentiate into cytotoxic T cells. This leads to furthering tissue damage, generation of nucleosomes, formation of ICs, and complement activation, which reloads dendritic activity. Nuclear particles also can directly activate B cells via the B-cell receptor without the presence of antigen-presenting cells.
Family studies reveal an important genetic contribution to the occurrence of SLE. About 10% of first-degree relatives of patients with SLE also have the disease, compared with 1% in control families. There is a 20-fold increased risk of siblings having SLE compared with the general population. , Among twins, 24% of monozygous compared with 2% of dizygous twins are concordant for SLE. One in 10 family members of patients with SLE have another connective tissue disease. , ,
Single gene mutations associated with SLE are rare but give important clues regarding the genetic mechanisms underlying SLE. SLE has been reported in over 90% of patients with homozygous C1q deficiency. Approximately one-third of patients with homozygous C2 deficiency develop SLE, and there is a higher association of C2 deficiency and anti-Ro antibodies. C4 allele deficiencies are also associated with increased risk of SLE.
Early studies in SLE susceptibility using candidate gene association and linkage analysis implicated a region on chromosome 6 (6p11-21) containing the human leukocyte antigen (HLA) region in susceptibility to SLE. The advent of genome-wide association studies (GWAS) and further candidate gene studies have expanded to list of genes linked to SLE to over 40. Most of these genes can be grouped into common physiological pathways, including the following: (1) HLA regulation; (2) DNA degradation, apoptosis, and clearance of cellular debris; (3) defective clearance of ICs containing nuclear antigens; (4) TLR and type I IFN pathway activation, such as seen in Aicardi–Goutières syndrome; (5) nuclear factor-κB (NF-κB) signaling; (6) B-cell function and signaling; (7) T-cell function and signaling; and (8) monocyte and neutrophil function and signaling.
For a few patients with cSLE, single gene mutations have been identified and referred to as monogenic or mendelian forms , such as Aicardi–Goutières syndrome and complement defects. These include defects in C1q, C1r, C1s, C2, C4, DNASE1, DNASE1L3, Fas ligand, protein kinase C delta (PRKCD), SAM domain and HD domain-containing protein 1 (SAMHD1), and TREX1, resulting in a clinical cSLE phenotype. Although these rare mutations in single genes do not contribute significantly to the overall population heritability, the effects can be devastating in the affected individual and have provided important insights into disease pathophysiology of “classical” SLE.
In addition to individual genes, it is clear that SLE is a complex disease influenced by the interactive effects of multiple genes. Recent studies have shown that epistasis, or gene–gene interaction, plays an important role in SLE susceptibility. For example, cytotoxic T-lymphocyte antigen 4 (CTLA4) interacts with a single nucleotide polymorphism (SNP) of the MutS protein homolog 5 (MSH5) gene, located in the HLA class III region, to increase SLE susceptibility with an odds ratio of 1.19. CTLA4 also interacts with an SNP of complement factor B (CFB2) in the HLA class III region, increasing the risk of SLE by 20%. CTLA4, a receptor on the surface of T cells, negatively regulates T cells after activation by antigen-presenting cells. This negative regulation is important for induction of self-tolerance through interactions with both effector and regulatory T cells. Thus CTLA4 and HLA epistasis illustrates the role of inappropriate antigen presentation, T-cell activation, and loss of self-tolerance SLE.
Since monozygous twins do not have a 100% concordance rate for SLE, it is clear that factors in addition to genetics play a role in the disease. It is thought that SLE susceptibility is determined by a complex interaction of environment, genetic predisposition, and differential gene expression through epigenetic modifications ( Fig. 23.1 ). Differences in DNA hypomethylation contribute to our understanding of discordance in lupus presentation between monozygotic twins. Processes leading to epigenetic modification of gene expression include cytosine methylation, histone modifications, and microRNA profile. DNA hypomethylation may play a role in the development of SLE, as evidenced by globally decreased DNA methylation in patients with the disease. More specifically, hypomethylation has been demonstrated in immune genes such as ITGAL (integrin, alpha L, or CD11a), CD70, perforin, and CD40 ligand (CD40L) in the T cells of lupus patients. Hypomethylation of these genes has been linked to overactivity of B cells and increased autoreactivity of monocytes and macrophages. CD40L is overexpressed in women with SLE and may contribute to the higher incidence of SLE in women, in addition to hormonal effects. Genome-wide DNA methylation analysis has led to the identification of hypomethylation of IFN genes, which poises the genes for increased IFN gene expression.
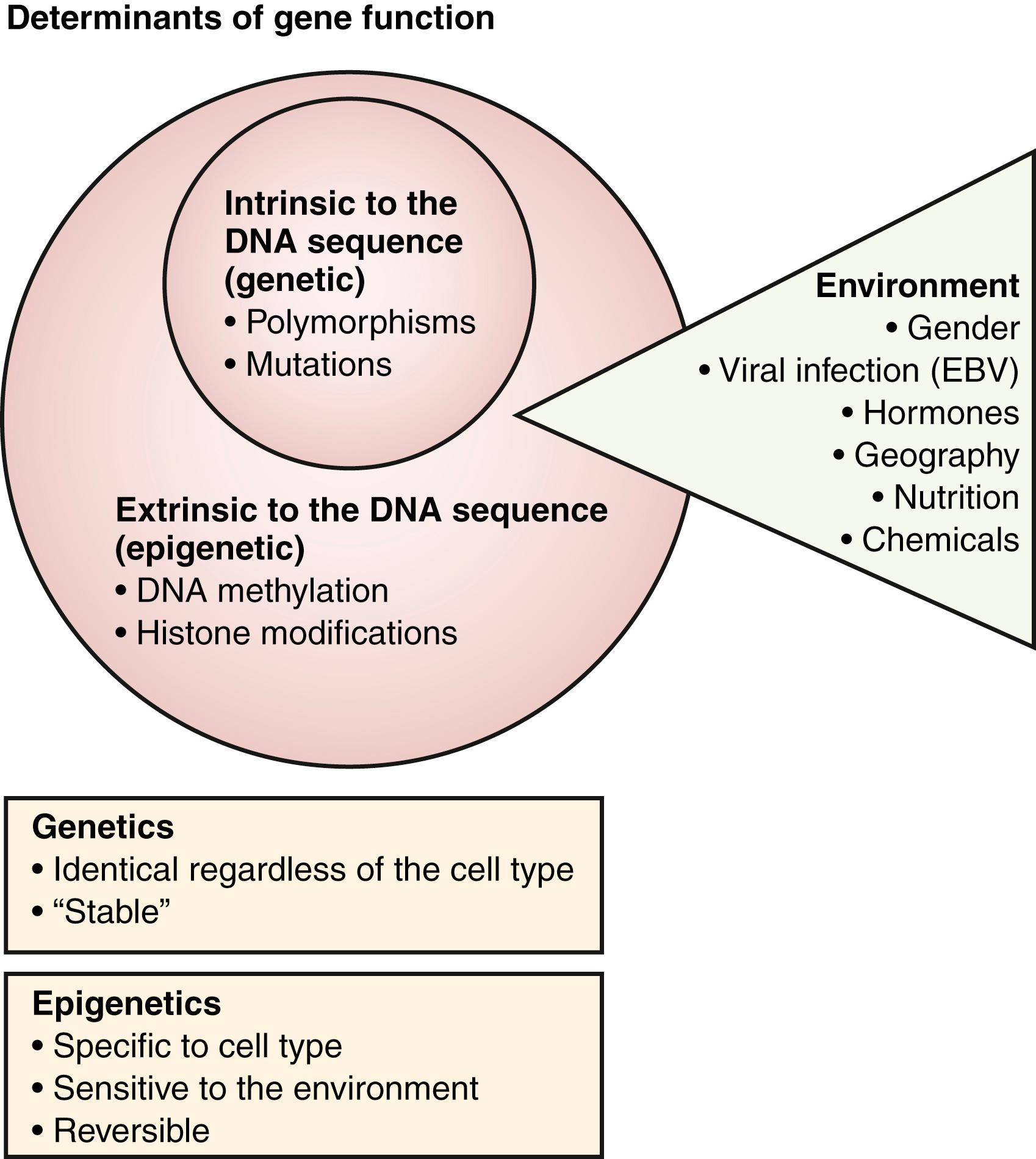
MicroRNAs (miRNAs or miR) are small, noncoding RNA molecules that modify gene expression by interacting with their target mRNAs. Upregulation of miR-21, miR-148a, miR-126, miR-155, miR-31, miR-182, and miRNA-96-183 and downregulation of miR-146a and miR-125a might contribute to DNA hypomethylation, T-cell activation, loss of self-tolerance, and autoantibody expression in SLE. MicroRNAs may be a useful as biomarkers of disease as well as potential targets for future therapeutics in the treatment of lupus.
Lupus primarily occurs in female patients between menarche and menopause. Thus the presence of estrogens and low levels of androgens play a role in disease risk. Estrogen has a variety of immune effects, including modulation of the innate and adaptive immune responses, alteration of the number of Ig-secreting cells, increased antigen presentation by DCs and macrophages, and modulation of the T helper (Th)1 and Th2 responses. Of note, there is an increased risk of lupus in patients with Klinefelter syndrome. Patients with Klinefelter syndrome, however, generally have mild disease, unlike XY adult men with SLE in which there is a higher risk of nephritis and renal failure. Disease frequency does not predict disease.
Some of the environmental factors implicated in SLE include ultraviolet radiation, viral infections, drugs, hormones, and chemicals.
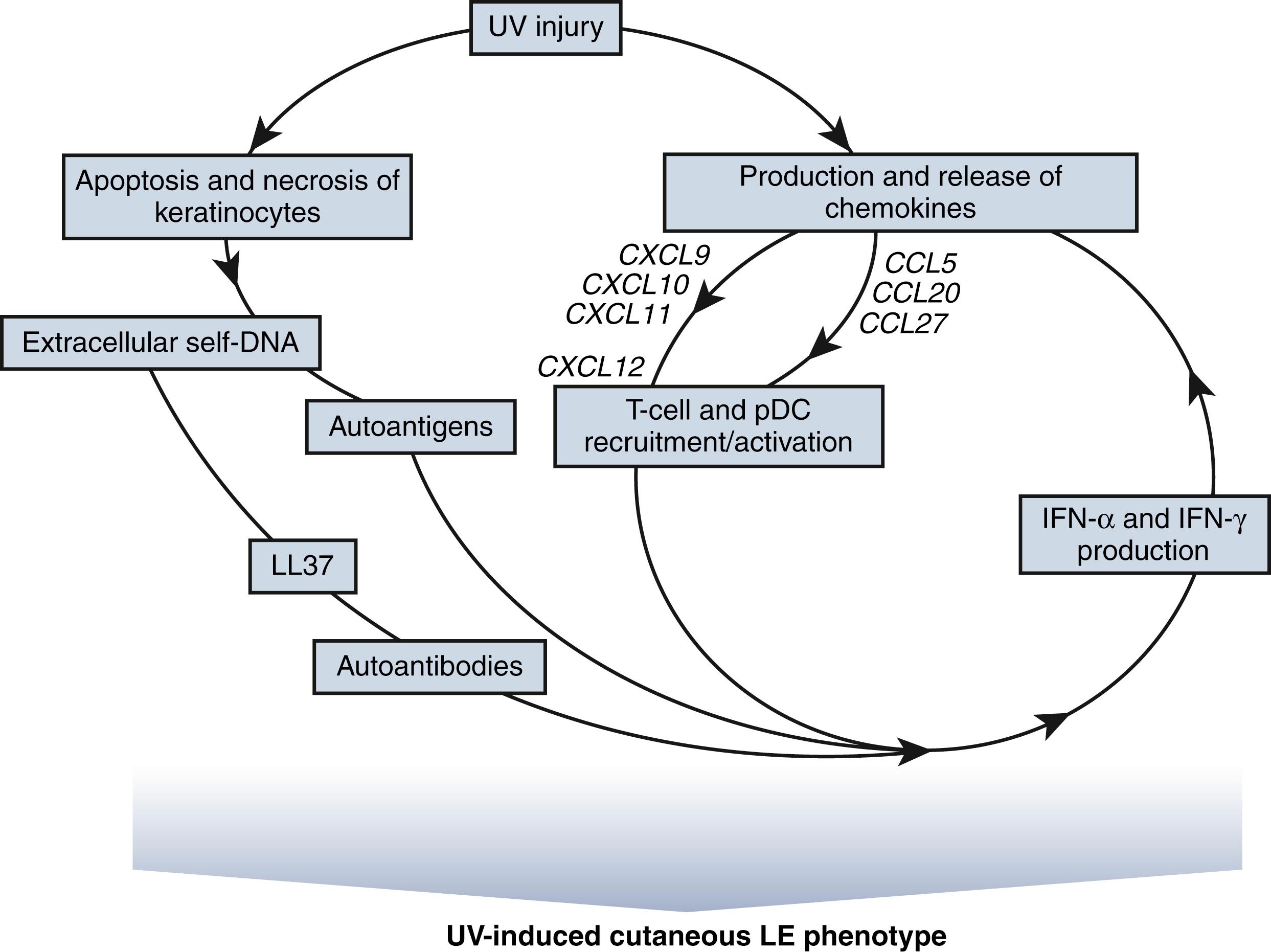
Ultraviolet radiation (UV), in particular UVB, is a trigger of cutaneous and systemic lupus. UVB directly induces keratinocytes to release chemokines and cytokines, which recruit memory T cells and pDCs into the skin. UVB also induces apoptosis and necrosis of keratinocytes. Nucleic acids, in combination with anti-DNA antibodies or nucleoproteins, activate pDCs through TLRs, leading to production of INF-α and activation of auto-reactive T cells. INF-α subsequently can induce production of more chemokines and cytokines, leading to more T-cell and pDC recruitment, furthering the cycle of inflammation( Fig. 23.2 ). Additionally, UVB can decrease DNA methylation in peripheral blood mononuclear cells, especially in patients with active lupus, which might alter gene expression and play a role in the pathogenesis of SLE.
Viruses can induce autoimmunity through a variety of mechanisms, including molecular mimicry, activation of T cells by superantigens, and expansion of previously activated T cells at a site of inflammation (bystander activation). A higher prevalence of Epstein–Barr virus (EBV) infection has been described in SLE patients compared with the general population. Cytomegalovirus (CMV), another member of the herpes virus family, has also been associated with triggering and exacerbating SLE. Higher rates of infection and seropositivity have been found in SLE patients compared with controls. ,
Over 80 medications have been associated with the drug-induced lupus (DIL). These drugs fall into 10 categories, including antiarrhythmics, medications for hypertension, antipsychotics, antibiotics, anticonvulsants, thyroid medications, antiinflammatory medications, diuretics, statins, and biologics ( Table 23.3 ). Drugs most commonly associated with DIL include hydralazine, procainamide, isoniazid, methyldopa, quinidine, minocycline, and chlorpromazine. In children, antiepileptic medications are most commonly implicated.
| Agent | Risk |
|---|---|
| Antiarrhythmics | |
| Procainamide | High |
| Quinidine | Moderate |
| Disopyramide | Very low |
| Propafenone | Very low |
| Antihypertensives | |
| Hydralazine | High |
| Methyldopa | Low |
| Captopril | Low |
| Acebutolol | Low |
| Enalapril | Very low |
| Clonidine | Very low |
| Atenolol | Very low |
| Labetalol | Very low |
| Pindolol | Very low |
| Minoxidil | Very low |
| Prazosin | Very low |
| Antipsychotics | |
| Chlorpromazine | Low |
| Phenelzine | Very low |
| Chlorprothixene | Very low |
| Lithium carbonate | Very low |
| Antibiotics | |
| Isoniazid | Low |
| Minocycline | Low |
| Nitrofurantoin | Very low |
| Anticonvulsants | |
| Carbamazepine | Low |
| Phenytoin | Very Low |
| Trimethadione | Very low |
| Primidone | Very low |
| Ethosuximide | Very low |
| Antithyroidals | |
| Propylthiouracil | Low |
| Antiinflammatories | |
| D -Penicillamine | Low |
| Sulfasalazine | Low |
| Phenylbutazone | Very low |
| Diuretics | |
| Chlorthalidone | Very low |
| Hydrochlorothiazide | Very low |
| Miscellaneous | |
| anti-tumor necrosis factor-α | Very low |
| Lovastatin and other statins | Very low |
| Levodopa | Very low |
| Aminoglutethimide | Very low |
| Interferon α and other cytokines | Very low |
| Timolol eye drops | Very low |
Patients with DIL tend to have milder disease manifestations than those with idiopathic SLE, though severe and even fatal cases of DIL have been described. DIL usually occurs after several months of treatment and might be associated with higher and cumulative medication doses. DIL typically presents with constitutional symptoms, and major organ involvement is rare. Antihistone antibodies are elevated in more than 90% of cases and are suggestive but not diagnostic of DIL. DIL is not the result of a typical drug hypersensitivity reaction. Proposed pathogenic mechanisms of DIL include activation of B and T cells, interference with T-cell maturation, cytokine effects, DNA methylation, and formation of reactive metabolites. , , DIL usually resolves within weeks to months after discontinuation of the offending drug. Lack of reversibility might indicate that the drug unmasked underlying idiopathic SLE. Rarely, corticosteroids or other immunosuppressive therapies are needed to treat patients with DIL Autoantibody titers decline gradually and can persist for months to years after resolution of clinical symptoms. Reexposure to the causative drug typically causes return of DIL manifestations with 1 to 2 days, and, with each exposure, the latency to onset of symptoms may decrease and severity of symptoms increase.
A variety of other environmental, chemical, hormonal and occupational exposures have been associated with SLE with varying degrees of evidence, including smoking, pesticides, solvents, silica, heavy metals, and hair dyes. The chemicals in cigarette smoke, including tars, nicotine, carbon monoxide, and polycyclic aromatic hydrocarbons, have multiple effects on the immune system, including tissue hypoxia and damage, toxin-induced cellular necrosis, release of intracellular antigens, and augmentation of autoreactive B cells. The evidence regarding exogenous estrogens and estrogen-like compounds is mixed and further investigation is required.
cSLE can present with subtle persisting or intermittent symptoms; however, most cSLE patients have more severe disease than their adult counterparts , with more organ involvement and laboratory abnormalities ( Table 23.4 ). Most lupus patients have constitutional symptoms including fever, weight loss, fatigue, and loss of appetite. Generalized signs of immune activation are seen, such as lymphadenopathy and hepatosplenomegaly. cSLE may initially manifest in a single organ system but will ultimately become a multiorgan disease ( eTable 23.5 ). Fulfillment of classification criteria is not required to make a diagnosis and treat severe single organ manifestations.
| Clinical Features ∗ | Within First Year of Diagnosis | Any Time |
|---|---|---|
| Fever | 35%–90% | 37%–100% |
| Lymphadenopathy | 11%–45% | 13%–45% |
| Hepatosplenomegaly | 16%–42% | 19%–43% |
| Weight loss | 20%–30% | 21%–32% |
| Organ Involvement | ||
| Musculoskeletal | ||
| Arthritis | 60%–88% | 60%–90% |
| Myositis | <5% | <5% |
| Any skin involvement | 60%–80% | 60%–90% |
| Malar rash | 22%–68% | 30%–80% |
| Discoid rash | <5% | <5% |
| Photosensitivity | 12%–45% | 17%–58% |
| Mucosal ulceration | 25%–32% | 30%–40% |
| Alopecia | 10%–30% | 15%–35% |
| Other rashes | 40%–52% | 42%–55% |
| Nephritis | 20%–80% | 48%–100% |
| Neuropsychiatric disease | 5%–30% † | 15%–95% ‡ |
| Psychosis | 5%–12% | 8%–18% |
| Seizures | 5%–15% | 5%–47% |
| Headache | 5%–22% | 10%–95% |
| Cognitive dysfunction | 6%–15% | 12%–55% |
| Acute confusional state | 5%–15% | 8%–35% |
| Peripheral nerve involvement | <5% | <5% |
| Cardiovascular disease | 5%–30% | 25%–60% |
| Pericarditis | 12%–20% | 20%–30% |
| Myocarditis | <5% | <5% |
| Pulmonary disease | 18%–40% | 18%–81% |
| Pleuritis | 12%–20% | 20%–30% |
| Pulmonary hemorrhage | <5% | <5% |
| Pneumonitis | <5% | <5% |
| Gastrointestinal disease | 14%–30% | 24%–40% |
| Peritonitis (sterile) | 10%–15% | 12%–18% |
| Abnormal Liver Function | 20%–40% | 25%–45% |
| Pancreatitis | <5% | <5% |
∗ Not all reports commented on all features or incidence in first year.
† Had highest prevalence of central nervous system disease but did not describe incidence in first year.
FLOAT NOT FOUND
| Constitutional | Fever, malaise, weight loss, anorexia |
| Cutaneous | Butterfly rash, photosensitivity, mucosal ulcerations, periungual erythema, alopecia |
| Musculoskeletal | Polyarthralgia/arthritis, morning stiffness, tenosynovitis, myositis |
| Renal | Glomerulonephritis, nephrotic syndrome, hypertension |
| Neurological | Acute confusional state, seizures, psychosis, cognitive deficits chorea, cerebrovascular accident, pseudotumor cerebri |
| Cardiac | Pericarditis and effusion, chest pain, Libman–Sacks endocarditis |
| Pulmonary | Pleuritis, abnormal pulmonary function tests, pneumonitis, pulmonary hemorrhage, infection |
| Gastrointestinal | Ascites, abdominal pain, peritonitis, colitis, abnormal liver function tests |
| Reticuloendothelial | Hepatomegaly, splenomegaly, diffuse lymphadenopathy, Kikuchi lymphadenitis |
| Vascular | Raynaud phenomenon, thrombosis, livedo reticularis |
| Ocular | Exudates, papilledema, retinopathy |
Kidney disease is a significant cause of morbidity and mortality in both adult and pediatric patients with SLE. About 20% to 75% of children with SLE will develop nephritis, with 18% to 50% progressing to end-stage kidney disease (ESKD). In general, the prevalence of LN is higher in children (50% to 67%) than in adults (34% to 48%), with 80% to 90% of childhood LN presenting within the first year of diagnosis. , ,
Murine models suggest the LN is initiated by the formation of ICs, leading to activation of complement and Fcγ receptors ( Fig. 23.3 ). Indeed, the histological hallmark of LN, the “full-house” pattern of IgG, IgM, IgA, C3, C1q, and kappa and lambda light chain deposits in the kidney, support the role of ICs and complement activation in its pathogenesis. ICs form directly in the glomeruli by binding of autoantibodies to components of chromatin likely derived from the nuclear debris of apoptotic renal cells. , As in extrarenal SLE, genetic variants leading to increased apoptosis and decreased clearance of apoptotic debris lead to accumulation of chromatin in apoptotic nucleosomes within the kidney. IC formation within the kidney leads to activation of complement, leading to inflammation and tissue damage.
![Fig. 23.3, The role of extracellular chromatin fragments and anti-dsDNA antibodies in progressive lupus nephritis. Retention of chromatin is assumed to start with reduced clearance of apoptotic cells (station 1). Secondary to this, chromatin may be exposed in tissues (station 2) and is assumed to activate dendritic cells (station 3). These cells present chromatin-derived peptides in the context of major histocompatibility complex (MHC) class II molecules to peptide-specific CD4+ T cells (station 4). When primed, the peptide-specific T cells recirculate and bind the same chromatin-derived peptides presented in the context of MHC class II by chromatin-specific B cells (here recognizing double-stranded DNA [dsDNA] in chromatin) (station 5). As a consequence of cognate interaction of dsDNA-specific B cells and peptide-specific CD4+ T cells, the B cells transform into plasma cells that secrete immunoglobulin (Ig)G anti-dsDNA antibodies (station 6), which bind chromatin fragments (station 7). Immune complexes that consist of IgG antibodies and chromatin fragments bind in the glomerular mesangial matrix and initiate mesangial lupus nephritis (station 8). This early inflammation is followed by silencing of renal DNAse I in tubular and glomerular cells (station 9) and accumulation of undigested chromatin fragments (station 10). These fragments promote upregulation of Toll-like receptors (TLRs), proinflammatory cytokines, and matrix metalloproteases (station 11). Finally, IgG autoantibodies recognize and bind the chromatin fragments, and these immune complexes deposit in the glomerular basement membranes (GBMs) and aggravate renal inflammation (station 12). In this sense, chromatin fragments and antichromatin (here: anti-dsDNA) antibodies are the partners that impose the classic murine and human lupus nephritis. Thus antichromatin antibodies are pathogenic only in the context of exposed chromatin structures. DC, Dendritic cell; GBM, glomerular basement membrane; IC, immune complex; TLR, Toll-like receptor. Fig. 23.3, The role of extracellular chromatin fragments and anti-dsDNA antibodies in progressive lupus nephritis. Retention of chromatin is assumed to start with reduced clearance of apoptotic cells (station 1). Secondary to this, chromatin may be exposed in tissues (station 2) and is assumed to activate dendritic cells (station 3). These cells present chromatin-derived peptides in the context of major histocompatibility complex (MHC) class II molecules to peptide-specific CD4+ T cells (station 4). When primed, the peptide-specific T cells recirculate and bind the same chromatin-derived peptides presented in the context of MHC class II by chromatin-specific B cells (here recognizing double-stranded DNA [dsDNA] in chromatin) (station 5). As a consequence of cognate interaction of dsDNA-specific B cells and peptide-specific CD4+ T cells, the B cells transform into plasma cells that secrete immunoglobulin (Ig)G anti-dsDNA antibodies (station 6), which bind chromatin fragments (station 7). Immune complexes that consist of IgG antibodies and chromatin fragments bind in the glomerular mesangial matrix and initiate mesangial lupus nephritis (station 8). This early inflammation is followed by silencing of renal DNAse I in tubular and glomerular cells (station 9) and accumulation of undigested chromatin fragments (station 10). These fragments promote upregulation of Toll-like receptors (TLRs), proinflammatory cytokines, and matrix metalloproteases (station 11). Finally, IgG autoantibodies recognize and bind the chromatin fragments, and these immune complexes deposit in the glomerular basement membranes (GBMs) and aggravate renal inflammation (station 12). In this sense, chromatin fragments and antichromatin (here: anti-dsDNA) antibodies are the partners that impose the classic murine and human lupus nephritis. Thus antichromatin antibodies are pathogenic only in the context of exposed chromatin structures. DC, Dendritic cell; GBM, glomerular basement membrane; IC, immune complex; TLR, Toll-like receptor.](https://storage.googleapis.com/dl.dentistrykey.com/clinical/SystemicLupusErythematosusMixedConnectiveTissueDiseaseandUndifferentiatedConnectiveTissueDisease/2_3s20B9780323636520000239.jpg)
Intrarenal ICs also activate pDCs, leading to production of type 1 INF-α and induction of a variety of immune responses. Chemokines play a role in recruiting different leukocyte subsets to different parts of the kidney, such as the glomeruli or the interstitium. , T and B cells form lymphoid aggregates in the renal tubulointerstitium, in some cases forming germinal centers around follicular DCs. These germinal centers might play a role in persistence of renal inflammation and local generation of autoantibodies. , Once deposited in the mesangial matrix, ICs appear to downregulate the production of DNase I, the major renal nuclease, through incompletely understood mechanisms that might involve transcriptional interference. Decreased DNase I activity impairs clearance of apoptotic cells and leads to further and large-scale exposure of chromatin fragments. These chromatin fragments can bind directly to glomerular basement membrane and form more ICs, thereby extending the disease from the mesangial space to the glomerular capillaries. The chromatin fragments are also taken up by DCs and macrophages, where they bind to TLRs and upregulate production of metalloproteinases, which contribute to tissue injury ( Fig. 23.3 ). ,
Once LN has been established, tissue damage can progress to chronic and irreversible stages through processes similar in many different forms of kidney disease. Focal necrosis of glomerular capillaries leads to migration of parietal epithelial cells into the glomerular tuft and production of extracellular matrix, contributing to the process of glomerulosclerosis. Breaks in the glomerular basement membrane allow leakage of plasma filtrate into the Bowman space and formation of cellular crescents that evolve into fibrocellular and fibrous crescents with glomerulosclerosis, characteristic of end-stage (class VI) LN. , ,
Classification of LN follows the 2003 criteria of the International Society of Nephrology/Renal Pathology Society (ISN/RPS) ( Table 23.6 ). Kidney biopsy tissue is examined by light microscopy to assess general features, cellular findings, glomerular and tubular lesions, and extent of fibrosis; immunofluorescent microscopy to determine the nature and extent of immune deposits; and electron microscopy to determine ultrastructural features and location of immune deposits within the basement membrane. LN is defined by the presence of “full-house” immune deposits. Lack of this full-house deposition calls into question the diagnosis of LN. Illustrations of class II to VI can be seen in Figs. 23.4 to 23.7 . Of note, mixed classifications occur, with features of both membranous and proliferative nephritis identified. Although not universally adopted, many pathologists report an activity index (AI) (range, 0 to 24) and chronicity index (CI) (range, 0 to 12) to quantify the degree of acute and chronic changes in a kidney biopsy for LN , ( eTable 23.7 ). Because the degree of active lesions (such as endocapillary proliferation, necrosis, and cellular crescents) versus chronic lesions (sclerosis, fibrous crescents, interstitial fibrosis) can play a role in treatment decisions, the ISN/RPS classification advises, but does not require, the inclusion of AI and CI in assessment of LN.
| Class I |
|
| Class II |
|
| Class III |
|
| Class III (A) |
|
| Class III (A/C) |
|
| Class III (C) |
|
| Class IV |
|
| Class IV-S (A) |
|
| Class IV-G (A) |
|
| Class IV-S (A/C) |
|
| Class IV-G (A/C) |
|
| Class IV-S (C) |
|
| Class IV-G (C) |
|
| Class V |
|
| Class VI | Advanced sclerotic lupus nephritis ≥90% of glomeruli globally sclerosed without residual activity |
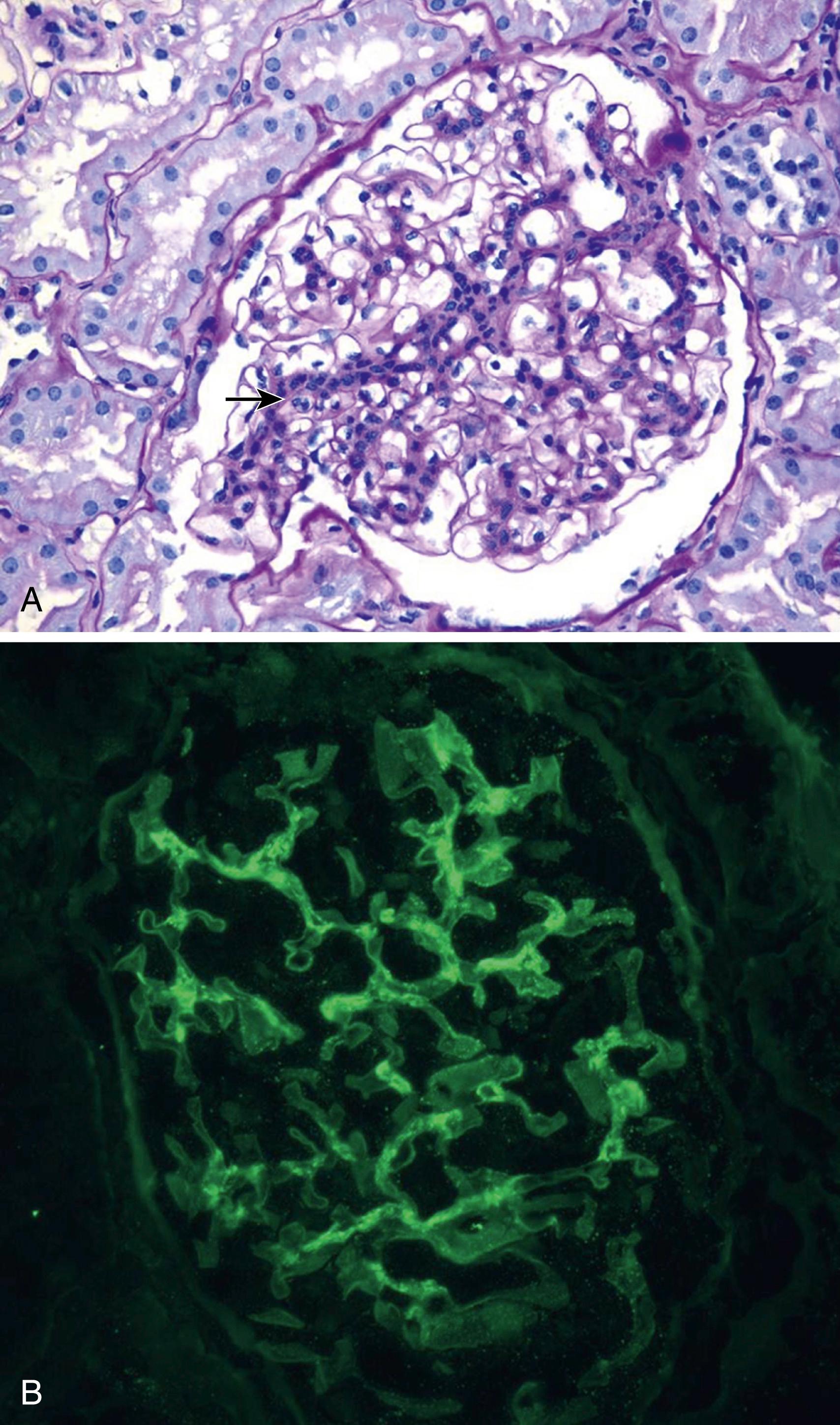
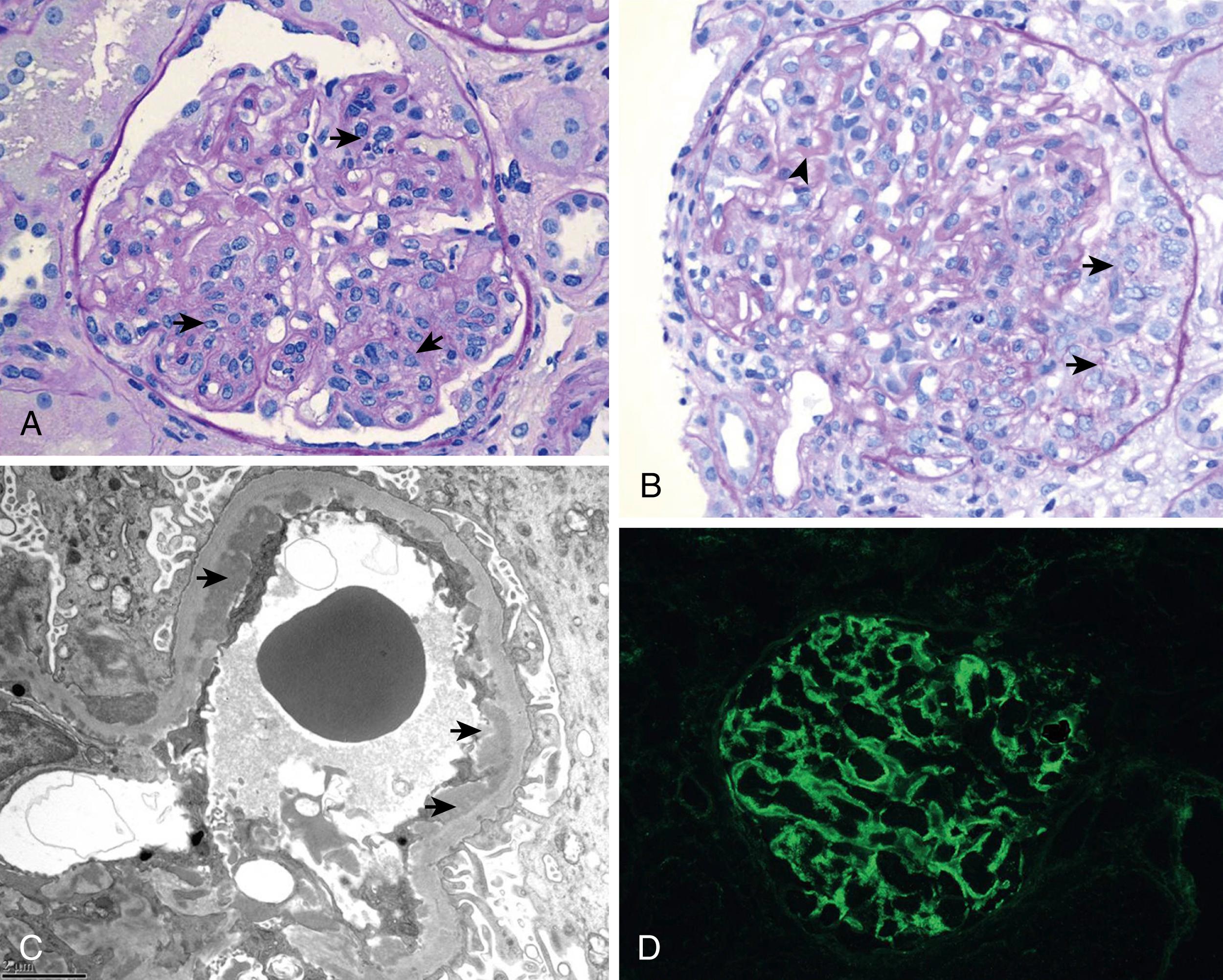
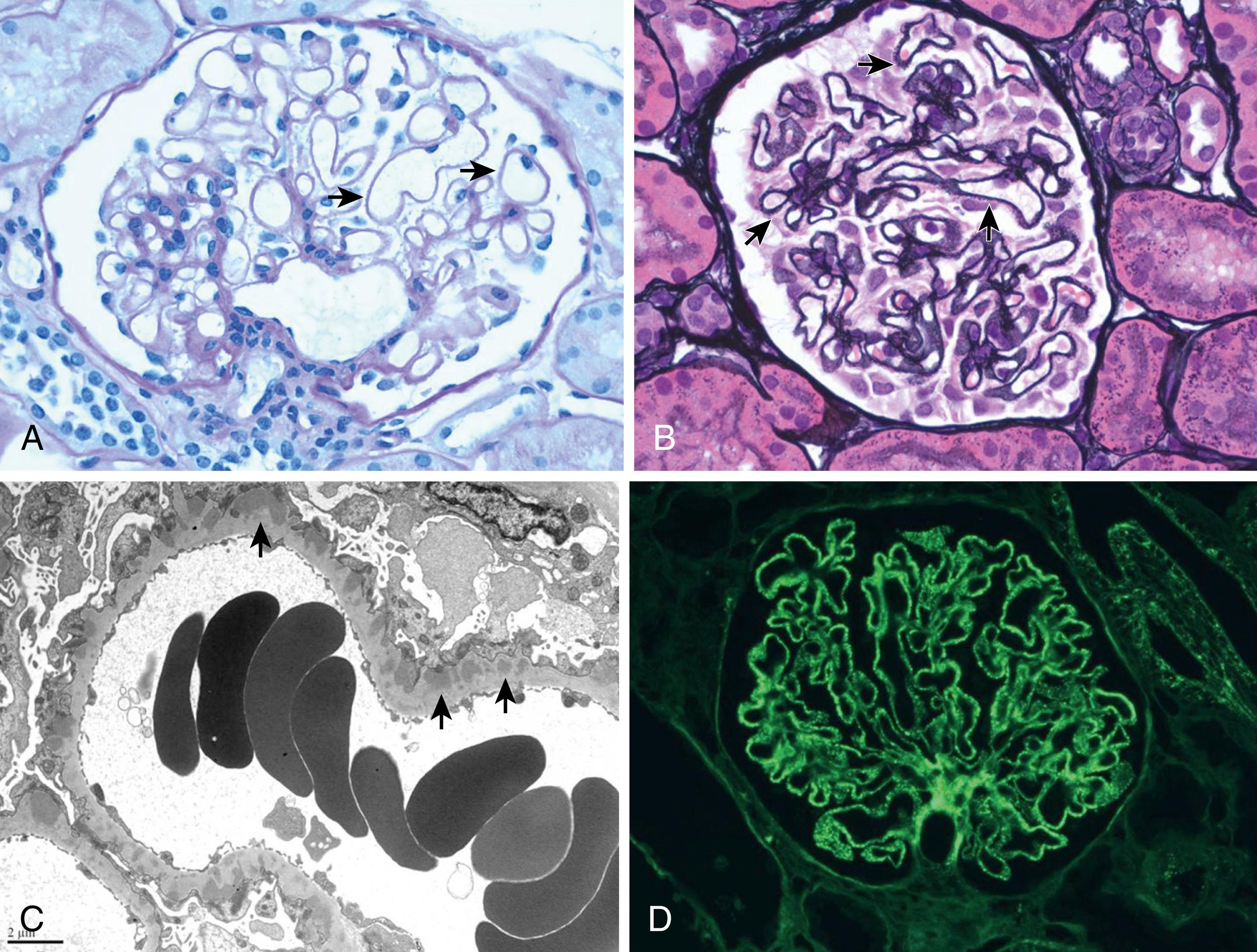
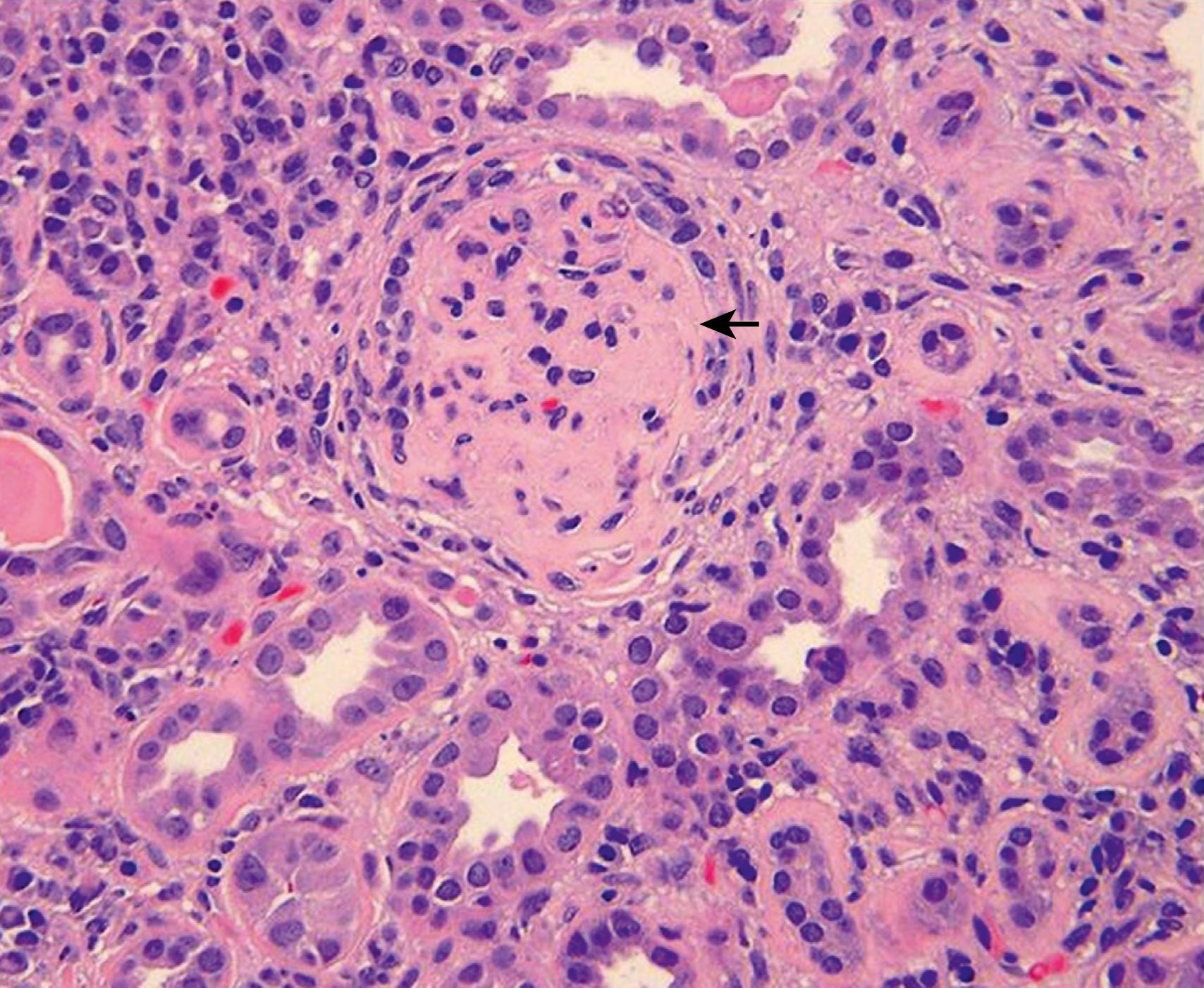
FLOAT NOT FOUND
| Activity Index | Chronicity Index |
|---|---|
| Glomerular Abnormalities | |
|
|
| Tubulointerstitial Abnormalities | |
|
|
Although correlations often can be made between clinical presentation and histological findings in LN, the presentation of the various classes of LN can overlap, and sometimes patients with the same clinical and laboratory manifestations can have different histological patterns of glomerular involvement. Additionally, severe LN can be “silent” or present with minimal clinical and laboratory findings more characteristic of milder forms of the disease.
In general, the presentation of LN can be grouped into the following broad clinical syndromes:
Isolated asymptomatic hematuria and/or nonnephrotic proteinuria: generally seen in class II LN, but sometimes seen in other classes, especially if biopsy was performed after at least several months of immunosuppressive treatment.
Acute nephritic syndrome: defined by hematuria (microscopic or occasionally macroscopic), usually accompanied by hypertension and proteinuria ranging from mild to nephrotic. Presence of hypertension at diagnosis is suggestive of more severe disease (such as proliferative LN or advanced chronic kidney disease and scarring). Acute nephritic syndrome is commonly a manifestation of class III and IV LN. Gross (macroscopic) hematuria may be a manifestation of the acute nephritic syndrome, but it is not commonly seen in patients with SLE. When gross hematuria is present in a patient with SLE, it should raise suspicion for other causes such as renal vein thrombosis (as a complication of nephrotic syndrome or antiphospholipid antibodies), thrombotic microangiopathy, or a clotting factor deficiency (such as prothrombin deficiency). ,
Nephrotic syndrome: defined by nephrotic range proteinuria, hyperlipidemia, hypoalbuminemia, and edema and may occur in isolation or associated with nephritic features. This is the most common presentation of class V LN, although class III and IV often present with nephrotic syndrome.
Chronic kidney disease (CKD): defined by persistent elevation of serum creatinine or reduced glomerular filtration rate (GFR). Advancing degrees of CKD can be accompanied by sequelae such as hypertension, anemia, azotemia, and metabolic bone disease. LN can occasionally present with CKD or ESKD, usually as a result of proliferative nephritis but occasionally as a result of class V LN.
Given the cumbersome nature of 24-hour urine collection, and the possible unreliability in young or noncontinent children, spot urine protein/creatinine (or urine albumin/creatinine) determination greater than 0.2 mg/mg creatinine in a first-morning urine collection is a convenient and reliable method for evaluating and following LN in children. It also shows good correlation with 24-hour urine collection.
The early diagnosis of LN is critical in minimizing the risk of disease progression and ESKD. The ACR has suggested the following indications for biopsy in the initial evaluation of LN: increasing serum creatinine without compelling alternative causes (such as sepsis, hypovolemia, or medication); proteinuria of 1 g or greater per 24 hours (24-hour urine or random urine protein/creatinine is acceptable); proteinuria of 0.5 g or greater per 24 hours with hematuria, 5 red blood cell (RBC) count or greater per high power field (HPF); or proteinuria of 0.5 g or greater per 24 hours with cellular casts in urine. The evidence for biopsy in children is scant; however, it is generally agreed that the clinical syndromes mentioned in the previous section are indications for biopsy in the initial evaluation of children with suspected LN. Therefore clear indications for biopsy include nephrotic syndrome, acute nephritic syndrome, and persistent elevation in creatinine. More controversial are indications for biopsy in mild, asymptomatic hematuria and/or proteinuria. These recommendations are reflected in the Single Hub and Access point for pediatric Rheumatology in Europe (SHARE) initiative where biopsy is indicated for persistent proteinuria or impaired GFR.
Given the consequences of missing and undertreating LN, and given the possibility of clinically silent LN with minimal laboratory findings and severe histology, it is the authors’ practice to have a low threshold for biopsy, which is consistent with the SHARE initiative. We recommend biopsy with any urinary abnormalities, such as urine casts, persistent proteinuria (urine protein/creatinine > 0.2 mg/mg), and/or persistent microhematuria (≥5 RBC/HPF) in addition to the more clinically overt presentations. Additionally, we recommend kidney biopsy when urine testing has not been performed prior to initiation of treatment. In a new patient with cSLE, no prior corticosteroid or other immunosuppressive therapy, normal serum creatinine, and normal urinalysis, a kidney biopsy is not indicated.
In general, a repeat biopsy should be considered in a patient with persistent or increasing proteinuria, worsening kidney function, or new development of active urine sediment. , A repeat biopsy also should be considered with flare of renal disease after successful treatment or if the patient fails to respond as anticipated. As defined by the Childhood Arthritis and Rheumatology Research Alliance (CARRA), a proteinuric/nephrotic renal flare is a persistent increase in the urine protein to creatinine ratio of 0.5 on spot urine after complete remission of proteinuria, or a doubling of urine protein/creatinine with a value great than 1.0 after achieving a partial response. A nonproteinuric/nephritic flare is defined as an increase or recurrence of active urine sediment (increased hematuria with or without casts), with or without an increase in proteinuria. In general, a repeat biopsy can help assess response to treatment and guide subsequent treatment. For example, persistence of active lesions might warrant a change or intensification of immunosuppression, whereas a high degree of chronic, irreversible changes might portend inevitable decline of kidney function and suggest a less aggressive approach. A repeat biopsy might also be useful in determining whether and when immunosuppression can be weaned after LN has gone into remission, though the optimal duration of treatment for LN has yet to be defined. A number of studies of urine biomarkers as an alternative to kidney biopsy in pediatric LN are ongoing.
Since untreated proliferative (class III and IV) LN is associated with a high rate of mortality and ESKD, drug trials have primarily focused on treatment of proliferative LN. Early drug trials by the National Institutes of Health (NIH) established cyclophosphamide (CYC) with corticosteroids as the treatment of choice, despite higher rates of infections and amenorrhea with CYC compared with azathioprine (AZA). , More recently the Euro-Lupus Nephritis Trial (ELNT) demonstrated that low-dose intravenous (IV) CYC (500 mg every 2 weeks for six doses with corticosteroids followed by AZA for maintenance therapy) compared favorably with the earlier higher-dose, longer-duration regimen. ,
Over the past decade, mycophenolate mofetil (MMF) has found increasing favor over CYC in the treatment of LN. In 2004 Contreras et al. demonstrated that, after a 6-month induction period with monthly IV CYC and steroids, maintenance therapy with either MMF or AZA was superior to long-term CYC in patient and kidney survival, with lower rates of relapse with MMF and lower rates of complications (such as amenorrhea and infections) with MMF and AZA. This study led to the abandonment of long-term CYC maintenance treatment by most practitioners. In 2005, Dooley et al. demonstrated that MMF was as effective as and safer than CYC for inducing initial remission. The MAINTAIN Nephritis Trial demonstrated that, after the ELNT regimen CYC induction, MMF and AZA were similar in efficacy for long-term maintenance in rates of relapse, disease activity, and kidney function, with lower rates of anemia and leukopenia in the MMF group. The largest randomized controlled drug trial in LN to date, the Aspreva Lupus Management Study (ALMS) study found that, in the 6-month early induction period, patients treated with corticosteroids and either MMF or IV CYC overall had similar rates of remission, although African American and Hispanic patients had a better response to MMF. In the long-term ALMS study, patients were randomized, after achieving remission, to AZA or MMF. Over 3 years of follow-up, the ALMS study demonstrated that MMF was superior to AZA in maintaining response to induction treatment and preventing relapse. A comprehensive Cochrane meta-analysis concurred that MMF was as effective as CYC for inducing remission with decreased risk of ovarian failure, and that, for maintenance therapy, the rate of relapse was lower for MMF than AZA.
In recent years there has been increasing interest in the use of biological therapies for SLE and LN. Given the role of B cells in the pathophysiology of LN, rituximab (RTX), a chimeric anti-CD20 monoclonal antibody that depletes circulating B cells, would seem to be a good candidate as alternative or adjunctive therapy in LN. The Lupus Nephritis Assessment with Rituximab (LUNAR) trial, which randomized patients with proliferative LN to receive corticosteroids and MMF plus either placebo or intermittent RTX, demonstrated no benefit in renal outcomes with the addition of RTX. However, it is possible that the use of high-dose corticosteroids and high-dose MMF left little room for improvement with the addition of RTX. The subgroup analysis of this study demonstrated that African American patients, who have a poorer prognosis, demonstrated higher partial response to RTX. Additionally, a number of observational studies report favorable results for RTX in the induction period in patients with relapsing disease or LN refractory to MMF and CYC. A recent review of 26 reports and 300 patients demonstrated combined complete and partial remission in 87% (class III), 76% (class IV), and 67% (class V) LN. Patients received one of two RTX regimens across these 26 studies, namely four weekly doses of RTX at 375 mg/m 2 or two fortnightly doses of 750 mg/m 2 with a maximum of 1000 mg. Patients also received a variety of concomitant treatments including CTX, MMF, and AZA. Further study is needed to determine the efficacy and optimal use of RTX.
Belimumab, a human monoclonal antibody that prevents B-cell activation (rather than depleting B cells) by blocking B lymphocyte stimulator (BLyS), was recently approved for treatment of nonrenal SLE and demonstrated improvement in disease scores when added to standard of care SLE treatment. , Although the belimumab trials (BLISS-52, BLISS-76) excluded patients with severe LN, a post hoc analysis of patients in the study with mild to moderate LN demonstrated greater renal improvement with belimumab added to standard treatment compared with placebo. , Preliminary results of a trial of belimumab in cSLE indicate that the benefit to risk profile of IV belimumab plus standard of care is generally consistent with its use in adult SLE, although final results are awaited ( https://clinicaltrials.gove/ct2/show/NCT01649765 ).
Abatacept, a fusion protein of CTLA4-Ig, blocks CD80 and CD86 on antigen-presenting cells from interacting with CD28 on T cells, thereby preventing T-cell activation. A clinical trial failed to show a benefit of abatacept over placebo when added to background immunosuppression.
Treatment of patients with mixed membranous and proliferative LN (class III/V or IV/V) generally follows the recommended treatments for proliferative disease. Few studies have examined treatment of pure class V disease because there is a lower risk of ESKD (20% at 10 years). Class V LN with low-grade proteinuria and well-preserved kidney function might only require treatment with angiotensin-converting enzyme inhibitors (ACEI) to lower proteinuria and preserve renal function. However, pure class V LN with nephrotic syndrome has a higher rate of complications, including thrombosis and cardiovascular events. Therefore immunosuppressive treatment has been recommended for class V disease with nephrotic-range proteinuria and/or decreased kidney function, although there are few randomized controlled trials defining optimal treatment for this condition. Various small studies have combined corticosteroids with another agent, such as MMF, tacrolimus (TAC), cyclosporine A (CSA), AZA, or IV CYC with beneficial results. , ,
There are few pediatric trials, mostly small observational studies, that examine the various adult treatment regimens mentioned previously in the context of pediatric LN. These studies have demonstrated beneficial effects in treatment of pediatric LN with different combinations of corticosteroids and MMF, CYC, TAC, and CSA. An important step in the standardization of treatment and future study of pediatric LN was taken with the 2012 CARRA publication of consensus guidelines for induction therapy of proliferative pediatric LN ( Fig. 23.8 ). These guidelines include three corticosteroid strategies to reflect practitioner preferences. The SHARE initiative used a nominal group method and had similar recommendations although the use of IV pulse corticosteroids was recommended only for severe disease. Maintenance therapy with MMF for pediatric proliferative LN is recommended by the SHARE initiative. For pediatric patients with pure class V disease, treatment can be adapted from the ACR adult guidelines (also endorsed by SHARE) consisting of corticosteroids and MMF or AZA, with change of treatment to IV pulse steroids and CYC for disease unresponsive to MMF or AZA. ,
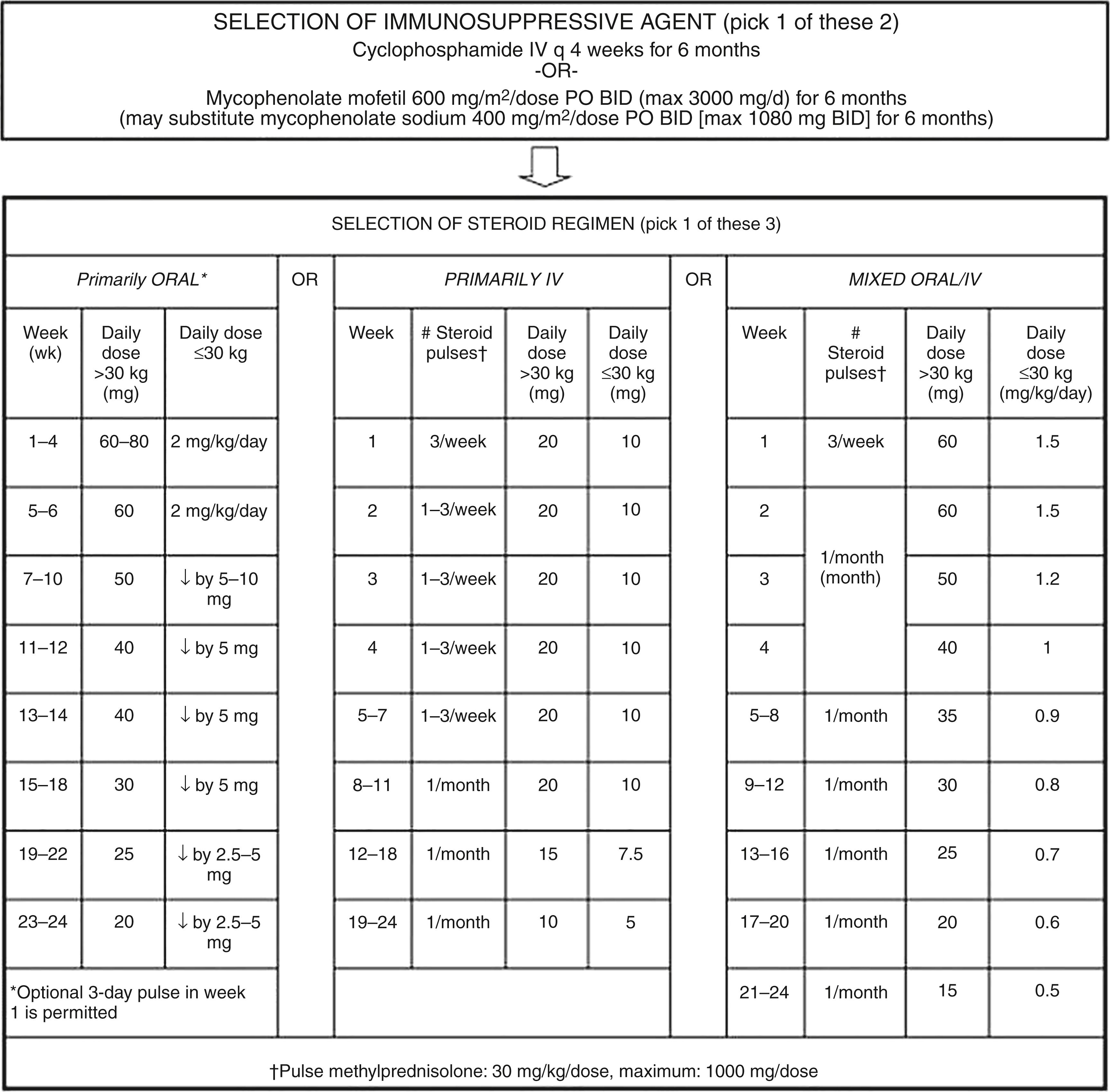
Hydroxychloroquine reduces rates of flare in SLE and incidence of renal disease and improves renal outcomes in adult studies and should be strongly recommended for use in pediatric LN. Similarly, adult studies have demonstrated the benefit of adding ACEI in reducing proteinuria and preserving kidney function, independent of the effect on hypertension.
Various definitions of complete and partial response and of renal flares have been proposed by the ACR, the EULAR, and CARRA ( Table 23.8 ). Adult clinical trials indicate complete or partial treatment responses in approximately 50% of patients after 6 months of induction treatment and 60% to 90% of patients during the maintenance phase of care. Rates of response are similar in proliferative and membranous LN. Approximately 40% of patients with LN have a relapse within a median time of 32 to 79 months. Because of the scarcity of studies in pediatric LN, it is difficult to define rates of response to treatment.
| ACR | EULAR | CARRA in juvenile LN | |
|---|---|---|---|
| Mild response | 30–50% improvement in 2 core renal parameters (proteinuria, renal function as creatinine clearance or serum creatinine, or urine sediment) without clinically relevant worsening of the remaining renal core parameter | ||
| Improved/partial response/moderate response | 25% increase in estimated abnormal baseline GFR ≥ 50% reduction in the urinary protein:creatinine ratio to a value of 0.2–2.0 Change from active urinary sediment (> 5 RBC/hpf and > 5 WBC/hpf and/or ≥ 1 cellular casts) to inactive sediment (≤ 5 RBC/hpf and ≤ 5 WBC/hpf and no cellular casts) |
Proteinuria ≤ 0.5 g/day a Normal GFR (> 90 ml/min) or, if previously abnormal, improved to a stable level within 10% of normal GFR (≥ 81 ml/min); Inactive urinary sediment (≤ 5 RBC or WBC/hpf [or < 10/mm 3 or < 10/μl]) and no cellular casts |
At least 50% improvement in 2 core renal parameters (with maximum spot protein/creatinine ratio ≤ 1.0) without clinically relevant worsening of the remaining renal core parameter |
| Complete response | Estimated GFR of ≥ 90 ml/min/1.73 m 2 and ≥ 50% reduction in the urinary protein:creatinine ratio to a value of < 0.2 and inactive urinary sediment | Proteinuria ≤ 0.2 g/day a Normal GFR (> 90 ml/min) or, if previously abnormal, improved to a stable level within 10% of normal GFR (≥ 81 ml/min); Inactive urinary sediment (≤ 5 RBC or WBC/hpf [or < 10/mm 3 or < 10/μl] and no cellular casts) |
Normalization of renal function Inactive urine sediment (≤ 5 WBC/hpf, < 5 RBC/hpf, and no casts) Spot protein/creatinine ratio < 0.2 or age appropriate |
| Remission | A sustained response of at least 3–6 months, but it cannot be judged to be a complete remission in the absence of a biopsy |
a The expert group did not reach an absolute consensus on the values for proteinuria that constituted partial or complete responses, but the values given here reflect the majority opinion.
Outcomes in pediatric LN have improved. A retrospective study reported improvement in 5-year pediatric patient survival from 83% to 91% and renal survival from 52% to 88% over the three decades (1980 to 2010). Predictors of poor renal outcome include African American race, low GFR (<60 mL/min/1.73 m 2 ), and nephrotic-range proteinuria at presentation. Other factors associated with increased risk of ESKD in adult studies include treatment resistance, lack of response in the first 6 months of induction therapy, renal, hypertension, delay in diagnosis of LN, higher chronicity score on biopsy, and greater degree of chronic tubulointerstitial disease.
Once a patient approaches ESKD, options include transplantation or renal replacement therapy (peritoneal dialysis or hemodialysis). There is evidence that preemptive transplantation, prior to the need for dialysis, improves graft and patient survival, as well as extrarenal SLE activity. In both children and adults with LN, there is an increased risk of death on dialysis (more than twofold in children) compared with patients with non-LN related ESKD. Many transplant centers require SLE to be in remission for 6 to 12 months prior to transplantation. Recurrence of SLE after transplantation occurs in approximately 2% to 30% of patients. Recurrence is generally mild and is not associated with decreased patient survival but is associated with a greater rate of graft loss.
Mortality in African American children with ESKD is double the rate in Caucasian children. In both pediatric and adult LN patients, the leading causes of death, before and after ESKD, are infection and cardiovascular complications.
Tubulointerstitial (TI) changes, such as tubular epithelial cell alterations, tubular atrophy, interstitial inflammatory infiltrate, and interstitial fibrosis, are found in approximately two-thirds of all cases of LN and strongly correlate with progression of renal disease. However, isolated tubulointerstitial nephritis (TIN) is rare. A variety of renal vascular lesions (RVLs) also are seen in SLE. RVLs can be seen in SLE associated with higher rates of active proliferative nephritis, hypertension, and renal insufficiency. They have poorer survival than patients without RVLs and often progress to ESKD.
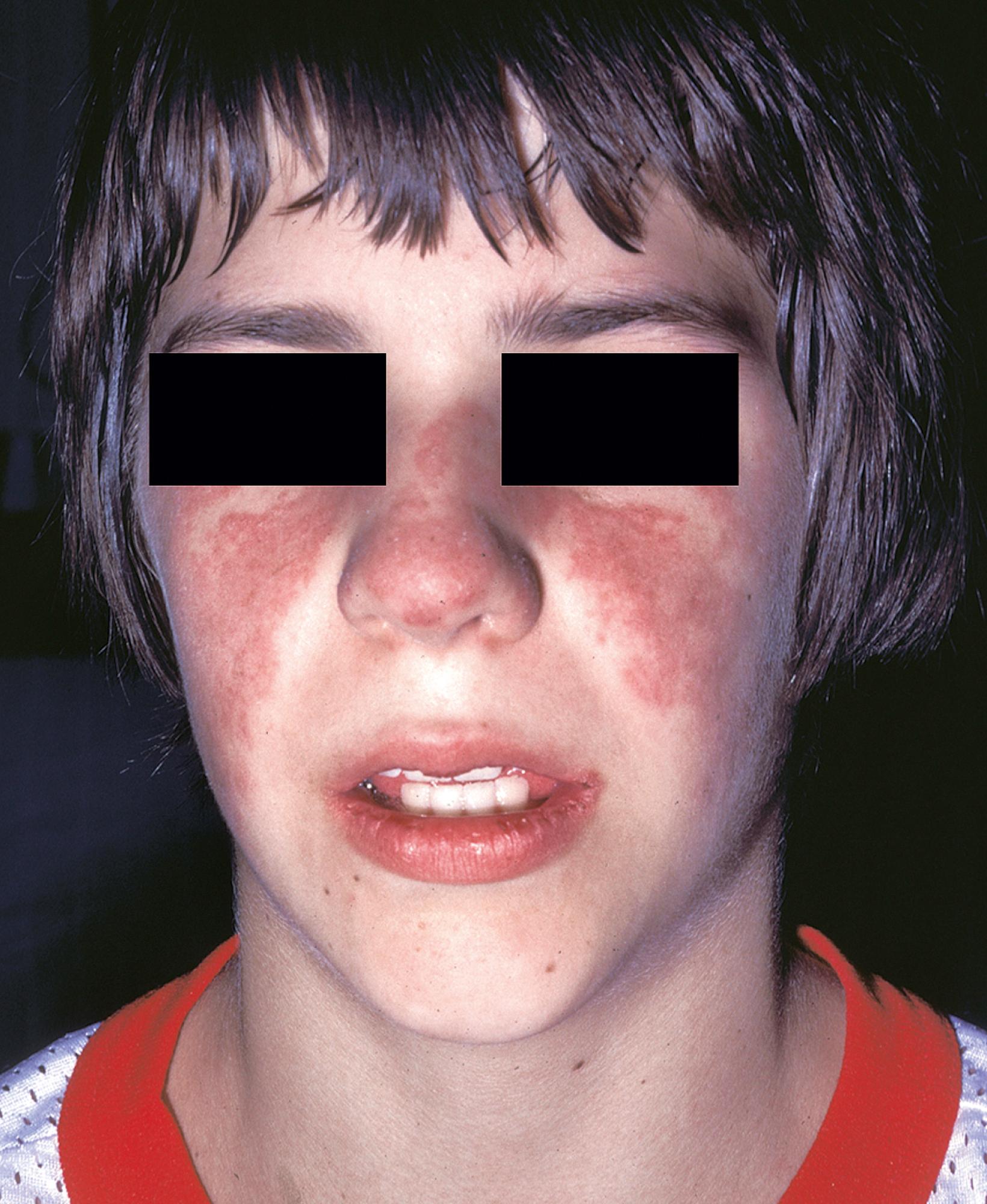
Most patients develop typical rashes over the course of disease (up to 85%). The archetypical malar or butterfly rash, a maculopapular rash that includes the nose and cheeks but spares the nasolabial folds ( Figs. 23.9 and 23.10 ). It is variable in intensity from a mild blush appearance to an intense erythema with a follicular or scaly appearance ( Fig. 23.11 ). It may be a diffuse lesion or have multiple satellite lesions, including the forehead and chin. This rash usually resolves without scarring. It is usually, but not necessarily, photosensitive and may be sensitive to fluorescent light as well ( Fig. 23.12 ). The lupus patient may have photosensitive annular or diffuse rashes on the limbs and chest, as well as the face. Other syndromes, including dermatomyositis, may have a photosensitive rash in the malar area ( eBox 23.2 ). However, lupus patients may have a wide range of mucocutaneous manifestations, and their presentations may differ between pediatric and adult patients.
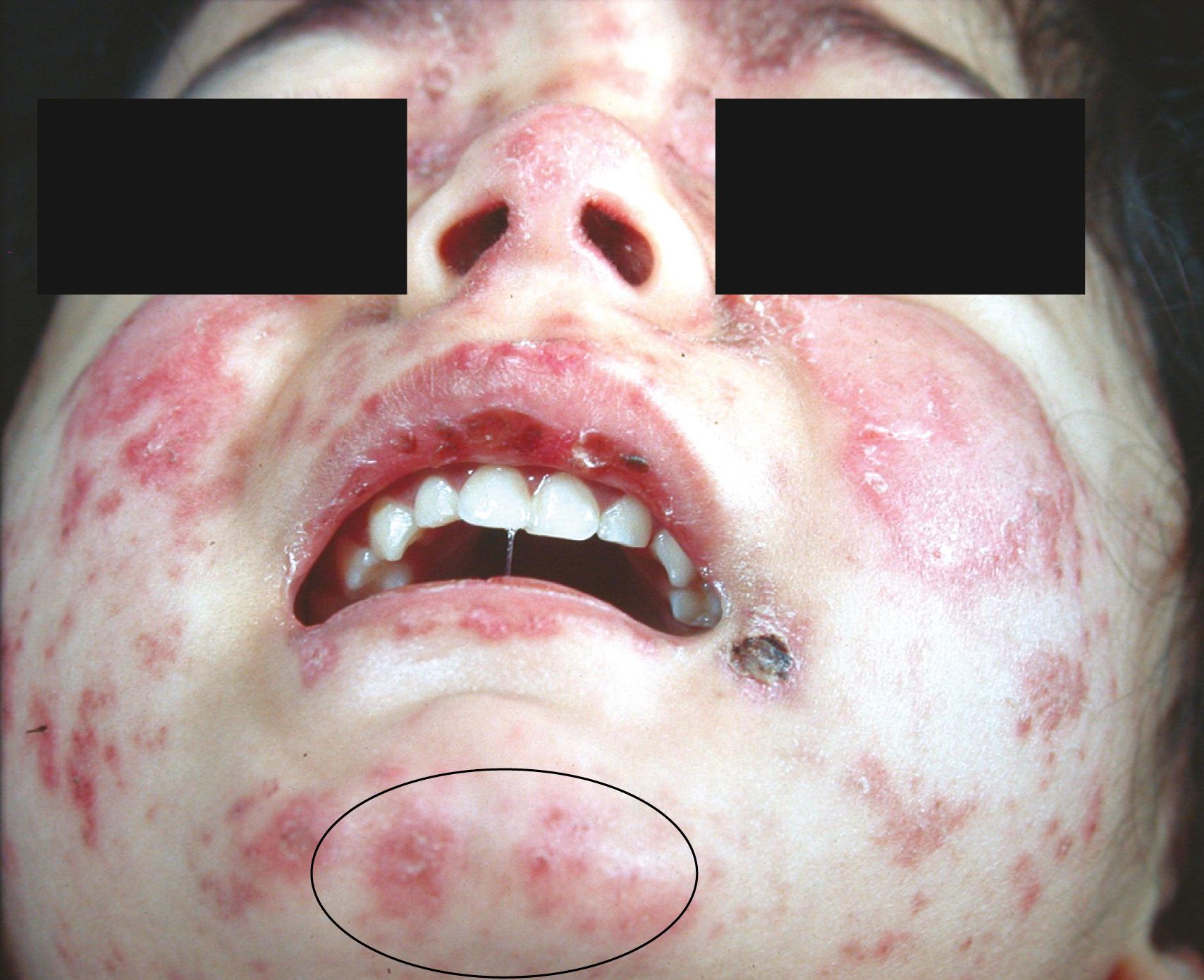
FLOAT NOT FOUND
Parvovirus ,
Erythropoietic protoporphyria ,
Bloom syndrome ,
Cockayne syndrome ,
Rothmund Thomson syndrome ,
Syndrome of telangiectasia and membranoproliferative glomerulonephritis
Prolidase deficiency ,
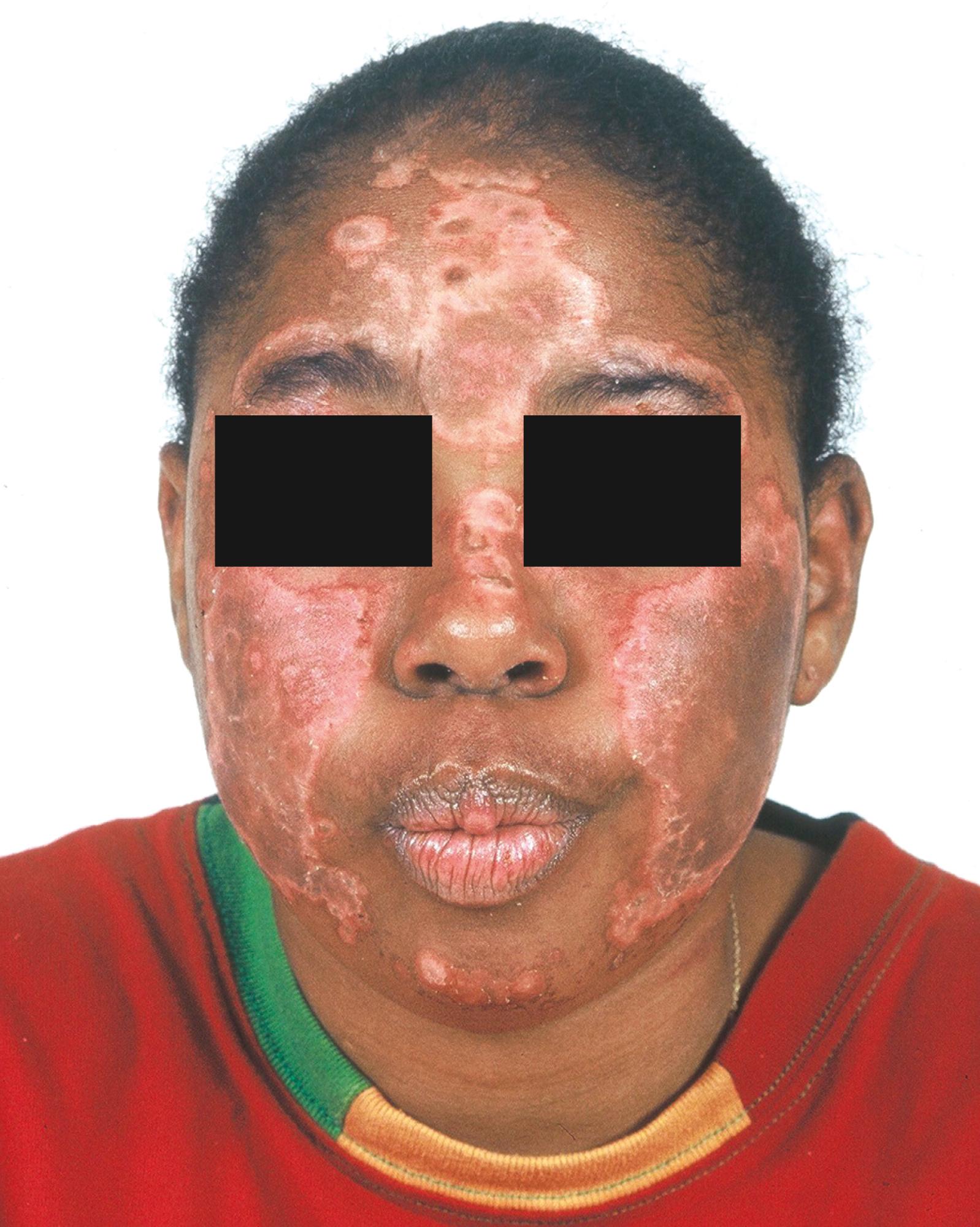
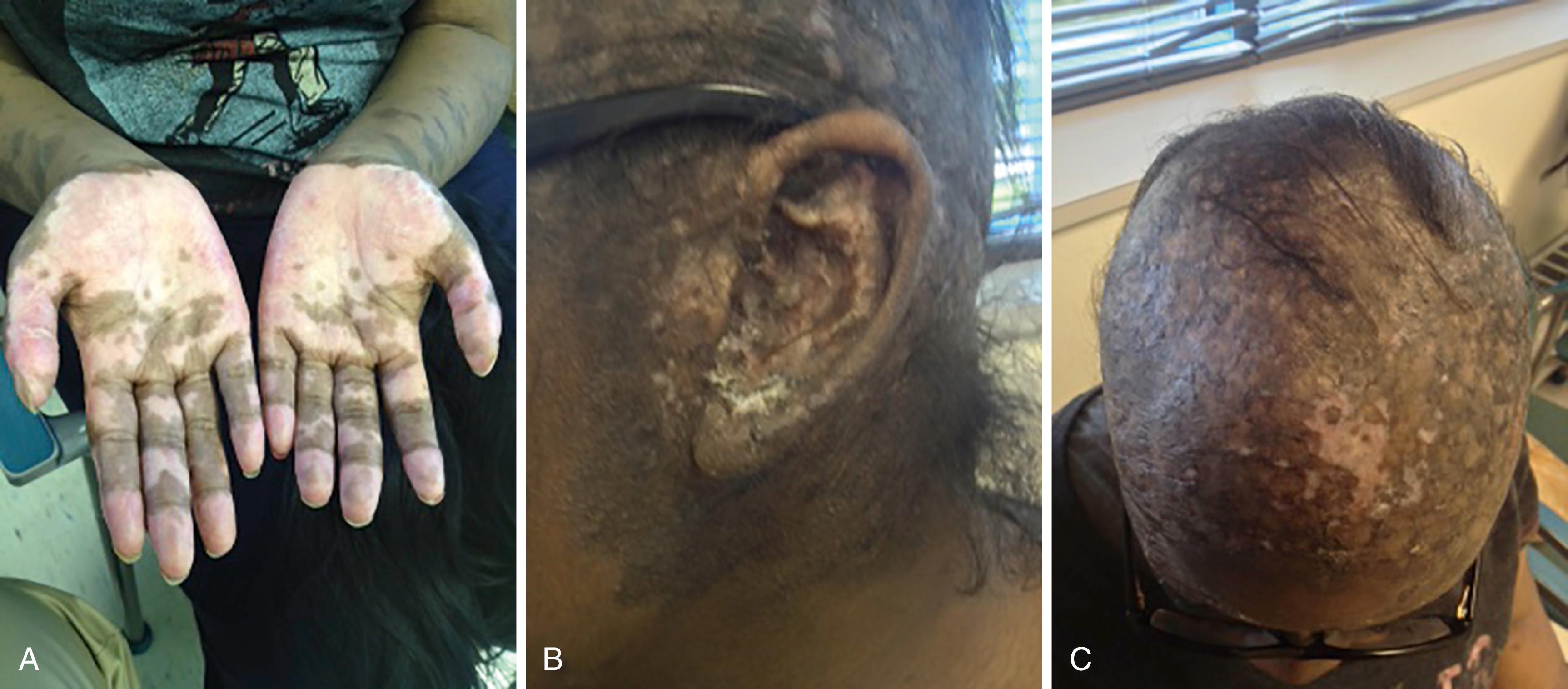
Discoid lupus is seen more frequently in adults either as an isolated skin disease or as part of SLE. Discoid lesions tend to occur around the eyes and ears of patients but can be distributed over the face, scalp, and body as a generalized rash ( Fig. 23.12 A to C ) . The rash tends to be hyperpigmented; however, some patients may develop hypopigmentation as well. The lesion is notable for a thick adherent scale and follicular involvement leading to hair loss at the area of the lesion. The lesions can become confluent and disfiguring and can lead to chronic alopecia in those who have scalp involvement from atrophic changes and scarring.

Subacute cutaneous lupus presents most frequently as an annular lesion associated with anti-Ro/SSA and anti-La/SSB antibodies ( Fig. 23.13 ) . Some patients have postinflammatory hyperpigmentation, which can lead to chronic scarring. This is more often seen in patients with darker pigmentation. , , Isolated subacute cutaneous lupus erythematosus (SCLE) is also more common in adult SLE than in cSLE.
Bullous lupus erythematosus is a rare manifestation of lupus; however, it is a potentially severe lesion where extensive disease causes a diffuse skin disruption requiring care similar to patients with large burns. Bullae can also occur on mucosal areas, including the oropharynx, which can compromise the airway. Another common cutaneous phenomenon in SLE is Raynaud phenomenon (see Chapter 30 ).
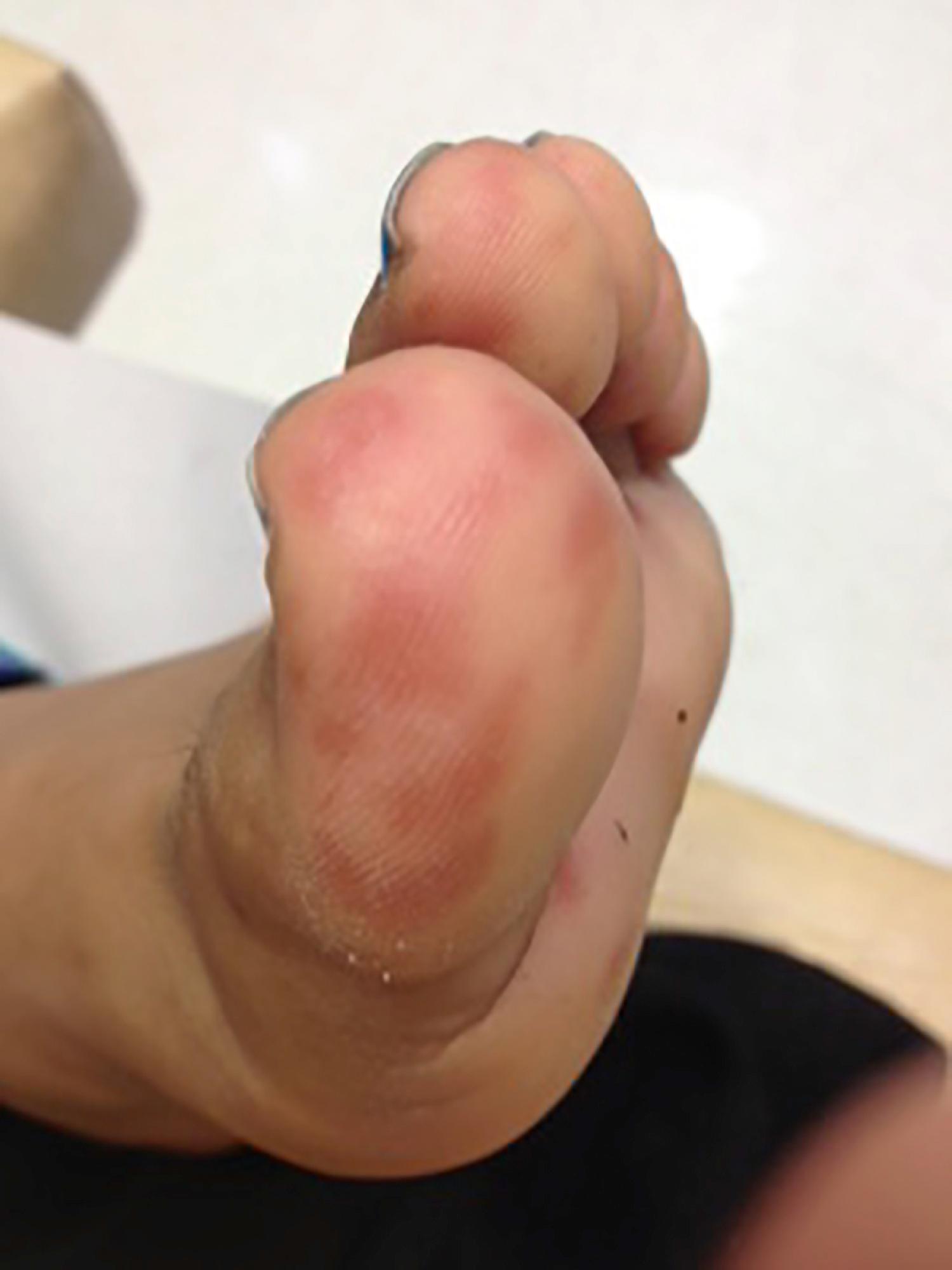
Patients with cSLE may have cutaneous manifestations of vasculopathy and vasculitis. Nailfold capillaroscopy may reveal dilated vessels and loss of capillary loops similar to that seen in dermatomyositis. Livedo reticularis may be associated with the presence of antiphospholipid antibodies. Patients will also have erythema, palpable purpura ( Figs. 23.14 and Figs. 23.15 ), and ulcers or urticaria associated with lupus vasculitis or antiphospholipid syndrome (APS). Poorly controlled cutaneous vasculitis lesions can be associated with complement defects. Patients can present with petechiae and purpura because of thrombocytopenia, similar to that seen in IgA vasculitis and acute lymphocytic leukemia.
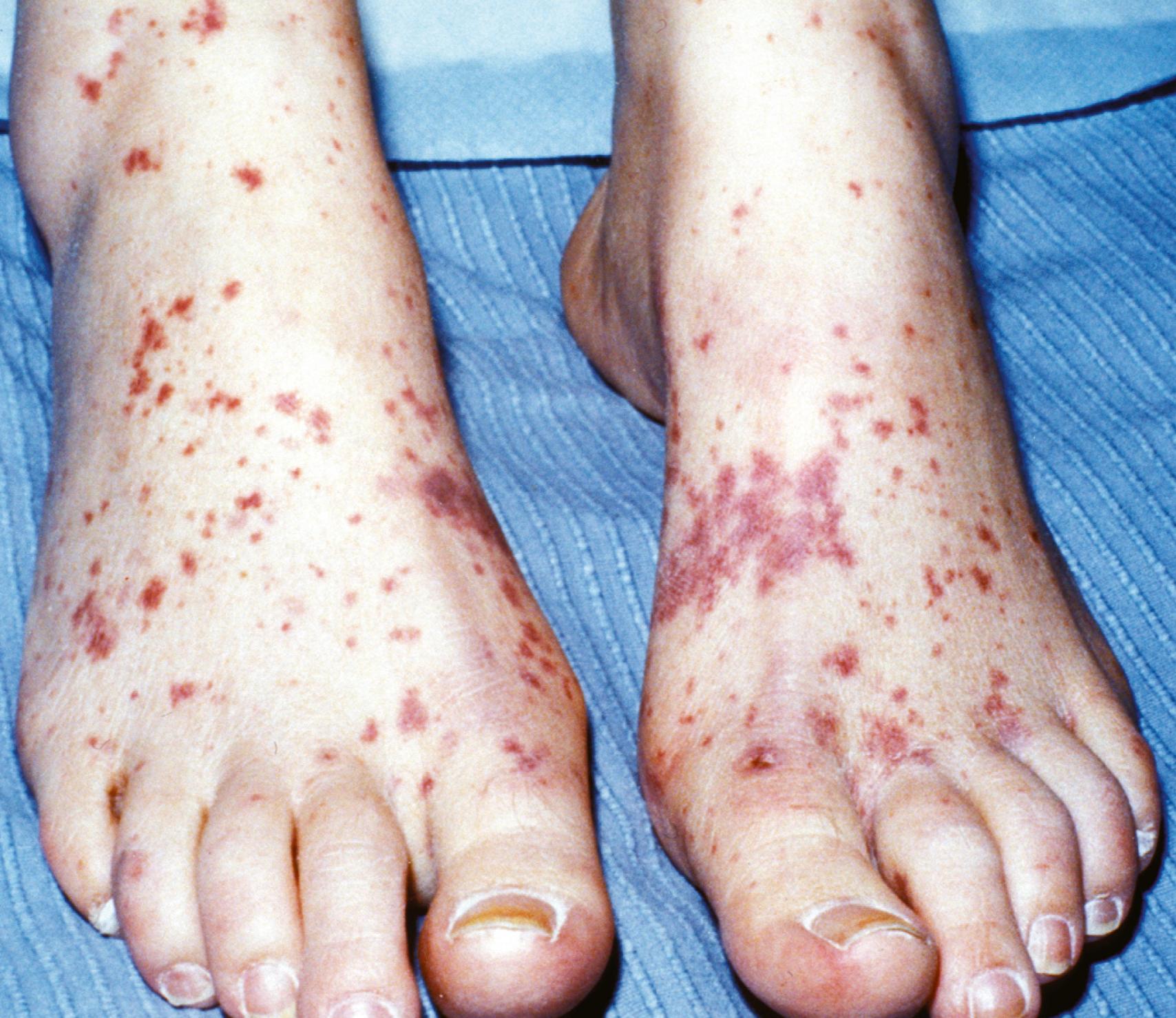
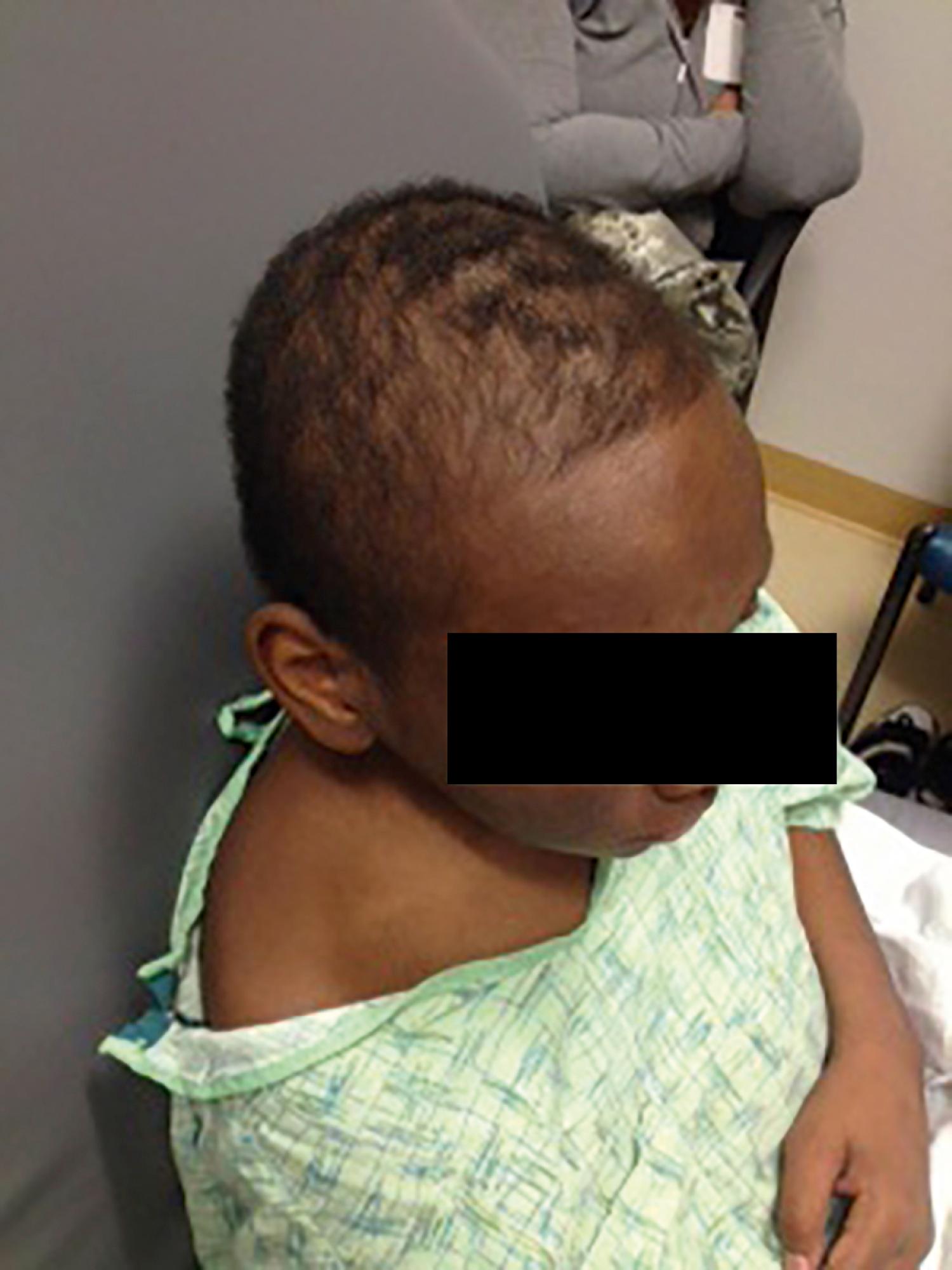
Alopecia associated with discoid lupus is rare and causes scarring. Alopecia without scarring, however, is a common finding in systemic lupus. It tends to occur at the frontotemporal line but can be more diffuse ( Fig. 23.16 ). It is a frequent symptom at disease onset and can also herald disease flares.
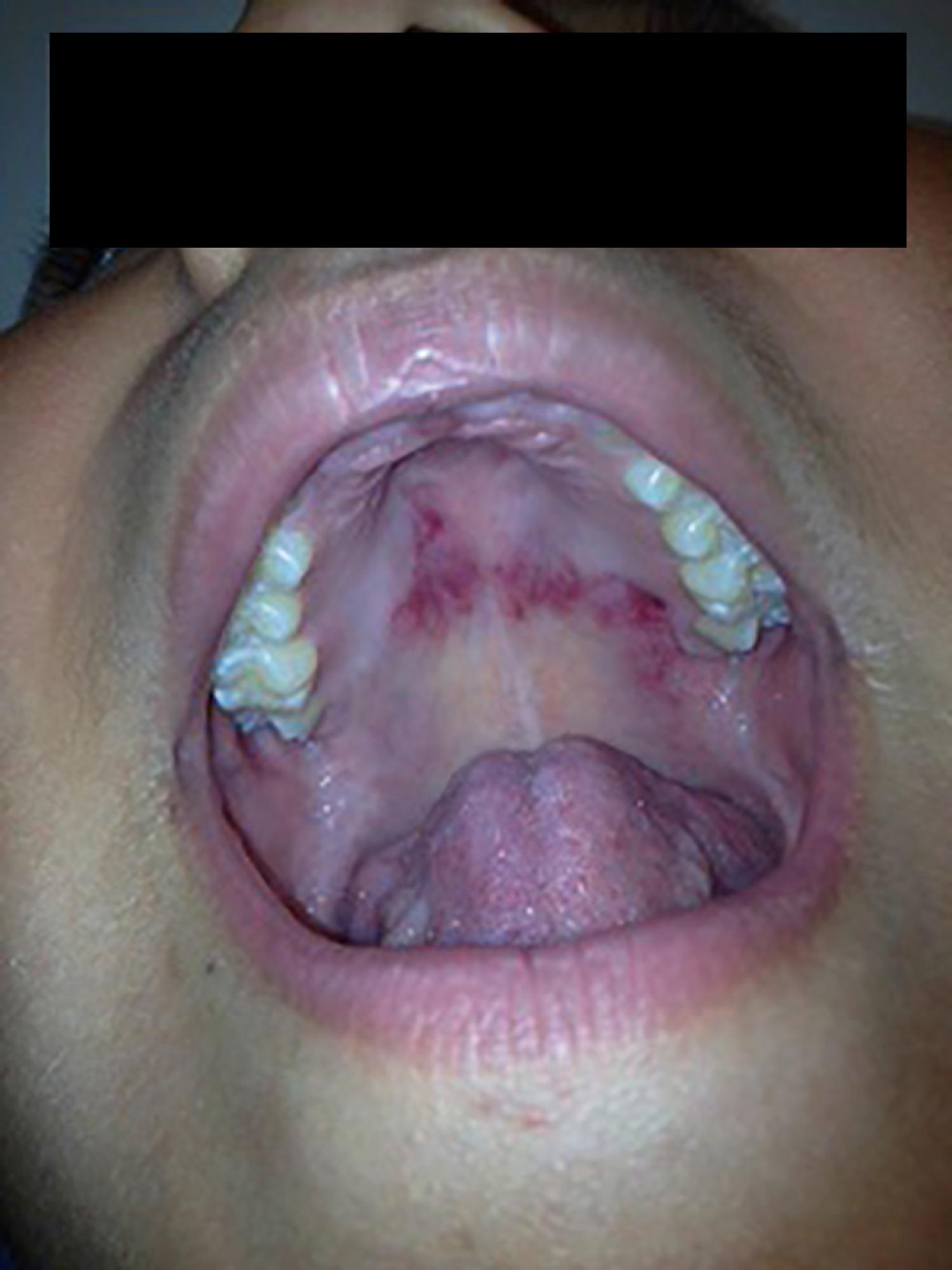
The mucosal surfaces of the oropharynx are frequently altered in systemic lupus. True nasal ulcers, sometimes with septal perforation, are seen rarely. Ulcers of the tongue and hard palate are common. The lesions are typically painless although some patients do report discomfort. Hard palate erythema, however, is quite common ( Fig. 23.17 ).
Acute lupus skin lesions are notable for disruption of the dermal–epidermal junction, with infiltration of T lymphocytes and fibrinoid degeneration. Chronic cutaneous lesions have thickening of the keratotic layer, epidermal atrophy, follicular plugging, and fibrosis of the elastic tissues. The presence of ICs by fluorescent staining is seen in the dermal–epidermal junction of normal and lesional skin.
Sun avoidance and sun protection are the cornerstones to care. Application of broad-spectrum (UVA and UVB) 30 SPF or more sunscreens at least 30 minutes prior to sun exposure allows the chemicals to bind to the skin. Patients may choose to use a physical barrier sunscreen (titanium dioxide or zinc oxide), which is preferred over a chemical sunscreen. Wide-brimmed hats, dark colors, long sleeves, and tightly woven clothes are helpful. Sunscreen additives such as Rit Sun Guard can be washed into clothes. Pretreated clothes can be purchased as well.
Treatment of rashes may require the use of topical or oral corticosteroids (for severe skin lesions). Topical corticosteroids are commonly used in cSLE patients and are very effective. High-potency corticosteroids may be necessary to induce remission of lesions but then gradually tapered to discontinuation. Facial skin is very sensitive and thin, requiring the use of mild corticosteroids. TAC can also be used to treat lesions topically. Hydroxychloroquine is the standard oral medication used to treat topical lesions and nonscarring alopecia. Some patients will require use of second-line therapies such as dapsone or AZA ( Table 23.9 ). , ,
| Treatment | Dosage |
|---|---|
| Antimalarials | |
| Hydroxychloroquine | 3–5 mg/kg/day (maximum 400 mg/day) |
| Chloroquine | 250 mg/day |
| Topical (in order of suggested use) | |
| Corticosteroids | 1.0% hydrocortisone or 0.1% Betnovate |
| Vitamin D analogs (e.g., calcipotriene) | |
| Calcineurin inhibitors (e.g., pimecrolimus or tacrolimus) | |
| Immunosuppressive Agents | |
| Systemic corticosteroids | Usually <0.5 mg/kg/day orally |
| Methotrexate | Up to 15 mg/m 2 /week either orally or subcutaneously (maximum 25 mg/week) |
| Azathioprine | 2–3 mg/kg/day (maximum 150 mg/day) orally |
| Mycophenolate mofetil | 1 g/m 2 /day divided BID orally |
| Calcineurin inhibitors | |
| Cyclosporine | Up to 5mg/kg/day divided BID (measure levels)—use lowest effective dose |
| Tacrolimus | 0.10–0.20 mg/kg/day maximum 10 mg/day orally divided BID (measure levels)—use lowest effective dose |
| Intravenous immunoglobulin | 1–2 g/kg/dose (maximum 70 g) |
Patients with cSLE often present with arthralgia and myalgia that resolve with initial treatment. True arthritis of the small joints of the hands and other peripheral joints is frequent. The arthritis tends to be more painful than proliferative. A smaller group of patients with cSLE have arthritis as a prominent feature with typical joint swelling, range loss, tenderness, and pain on motion (see Table 23.4 and eTable 23.5 ). Patients with persisting arthritis require arthritis-specific therapies. Patients with cSLE may also present with or develop an inflammatory myositis, which may manifest as either just weakness or at times pain. Muscle enzyme elevation can be remarkable and is responsive to appropriate therapy.
Although musculoskeletal symptoms are typical and common in lupus patients, it is important to also consider other causes of musculoskeletal complaints. Steroid myopathy can present at any time after corticosteroid therapy is started and is more severe when fluorinated steroids are used or the patient is bedridden (intensive care unit [ICU] myopathy). , creatine phosphokinase (CPK) elevations can also be drug induced (statins, hydroxychloroquine). A newly swollen or painful joint, particularly if it is a single joint, can represent infection of the joint (or bone), osteonecrosis, or infarction of bone. Patients may have complex regional pain syndromes or pain amplification syndromes both at presentation or after treatment.
The reported prevalence of neuropsychiatric SLE (NP-SLE) in lupus is widely variable (20% to 95%) in a number of pediatric studies that have defined NP-SLE differently, have different patient populations, and have small sample sizes. , The ACR published a guideline for describing NP-SLE in adults that includes 19 specific diagnoses (see eBox 23.3 ). , It is notable that NP-SLE is more frequent in the pediatric population and typically presents at disease onset, although it may develop at any point in the disease course. The approach to treatment of patients with NP-SLE requires a team approach, which may include neurologists, psychologists, and psychiatrists. eTable 23.10 reviews the frequency of the individual NP manifestations in cSLE. FLOAT NOT FOUND FLOAT NOT FOUND
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here