Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Systemic lupus erythematosus (SLE) is a multisystem autoimmune disease that results from immune system–mediated tissue damage. The molecular triggers of the disease are not known, but the pathogenesis involves the production of autoantibodies specific for nucleic acids and nucleic acid–binding proteins. Immune complexes, along with immune system cells and soluble mediators, generate inflammation and tissue damage. Manifestations of SLE can involve the skin, joints, kidney, central nervous system (CNS), cardiovascular system, serosal membranes, and hematologic and immune systems. The disease is highly heterogeneous, with individual patients manifesting variable combinations of clinical features. In most patients with SLE, the disease is characterized by a waxing and waning clinical course, although some patients demonstrate a pattern of chronic activity. Therapeutic approaches generally involve immunosuppression, although promising biologic agents targeting specific molecular mechanisms are in development.
A notable feature of SLE is that it occurs much more frequently in females than in males. As in Hashimoto thyroiditis and Sjögren syndrome, the female-to-male ratio is approximately 9 : 1 in adults, and most cases are diagnosed between the ages of 15 and 44 years. In children and patients older than age 55 years, the ratio is closer to 2 : 1. The prevalence of SLE in the United States is estimated to be approximately 72.8 per 100,000, and the incidence of new cases is about 5.1 per 100,000 per year. , The prevalence, severity, and characteristics of disease differ in different ethnic groups, with SLE being three- to four-fold more prevalent in Black, American Indian, and Native Alaskan women than in White women. Asians also have a higher incidence of disease than Whites. The severity of disease, based on standardized mortality ratios, is greatest among Asians and Hispanics, although the highest percent mortality is among Blacks. Studies of lupus in minority populations indicate that socioeconomic factors are major contributors to the increased prevalence and severity of disease in Blacks and Hispanic Americans.
Current understanding of lupus pathogenesis incorporates roles for genetic susceptibility based on a cumulative hit model involving multiple genes, environmental triggers (e.g., the endogenous microbiome or virome, microbial infection, sunlight), certain drugs, and altered immune system function. Studies of immunology, genetics, and epigenetics have focused attention on the mechanisms of nucleic acid sensing and the type I interferon pathway. Impaired inactivation of the X-chromosome may partially explain the striking female sex predominance. Induction of cellular stress responses, including oxidative modification of cell proteins, may link environmental triggers to altered immune function. Together, these mechanisms result in the generation and deposition of immune complexes in tissue, the recruitment of lymphocytes, macrophages, and neutrophils, the activation of the complement system, and tissue damage.
An important role for a genetic contribution to lupus susceptibility is suggested by the high concordance of disease in monozygotic twins (24%), with an estimated 44% of risk for SLE attributable to heritable factors. Rare mutations in genes encoding components of the complement pathway, including C1q, C2, and C4A, contribute to increased lupus susceptibility or severity. Impaired production of these early complement components may decrease the clearance of apoptotic or necrotic debris, thereby augmenting the pool of available autoantigens, or decrease the solubility of immune complexes. Rare mutations in genes encoding DNase enzymes can contribute to impaired degradation of endogenous DNA and induction of type I interferon. The association of SLE with the major histocompatibility complex (MHC) class II alleles human leukocyte antigen (HLA)-DR3 (DRB1∗03:01-DQA1∗05:01-DQB1∗02:01) and HLA-DR15 (DRB1∗15:01/03-DQA1∗01:02-DQB1∗06:01) shows the strongest risk, and these class II alleles are associated with the production of particular autoantibodies. A large transancestral genome-wide association study (GWAS) of subjects of European, African, and Hispanic Amerindian ancestry identified 80 non-HLA single-nucleotide polymorphisms associated with SLE. Some associations at a high level of significance were related to ancestry ( E-Table 245-1 ). Polymorphic variants in components of the toll-like receptor (TLR) pathways that regulate type I interferon production, including interferon regulatory factor 5 ( IRF5 ) and IRF7 , are associated with a diagnosis of SLE and increased plasma interferon activity in some populations. Polymorphisms in the Fc receptor genes FCGR2A and FCGR3A have been associated with SLE nephritis, possibly based on altered clearance of immune complexes. Variants of the PTPN22 gene, which encodes a phosphatase that regulates T-cell activation, are also associated with SLE. GWASs have identified variations in regulators of innate immune system activation (e.g., TNFAIP3, ITGAM, IFIH1 ) and signaling molecules important in lymphocyte activation (e.g., STAT4, BANK1 , and BLK ). Rare mutations in genes encoding proteins that regulate nucleic acid integrity and degradation, including TREX1 , encoding a DNase; SAMHD1 , a triphosphohydrolase; RNASEH2A , B , and C ; and ADAR , an RNA-specific adenosine deaminase, have been documented in some patients with a lupus-like disorder called Aicardi-Goutières syndrome, characterized by skin lesions, CNS disease, autoantibodies, and high levels of interferon. Mutations in these genes have also been documented in rare patients with SLE and highlight the likely contribution of endogenous nucleic acids to activation of the innate immune system and the pathogenesis of lupus. Composite genetic risk scores are associated with early onset of disease, tissue damage, and decreased survival. These genetic loci that are associated with lupus confer either increased activation or impaired regulation of innate or adaptive immune responses ( Chapter 35 ), with increased type I interferon often observed in association with the risk genotype.
| GENE ∗ | PROTEIN |
|---|---|
| EUROPEAN ATG5 BANK1 BLK CLEC16A DEF6 DGKQ ETS1 FCGR2A GRB2 GTF2I IFIH1 IKZF1 IKZF3 IL10 IL12A IRF5 IRF7 IRF8 ITGAM JAZF1 MSRA NMNAT2 OLIG3 PKIA PTPN22 PTTG1-MIR146A PXK SLC15A4 SLC17A4 STAT4 TMEM39A TIMMDC1 |
Autophagy related 5 B-cell scaffold protein with ankyrin repeats 1 B-lymphocyte–specific tyrosine kinase C-type lectin domain containing 16 A DEF6, guanine nucleotide exchange factor Diacylglycerol kinase theta ETS proto-oncogene 1 Fc fragment of IgG receptor IIa Growth factor receptor bound protein 2 General transcription factor IIi Interferon induced with helicase C domain 1 IKAROS family zinc finger 1 IKAROS family zinc finger 3 Interleukin 10 Interleukin 12A Interferon regulatory factor 5 Interferon regulatory factor 7 Interferon regulatory factor 8 Integrin subunit alpha M JAZF zinc finger 1 Methionine sulfoxide reductase A Nicotinamide nucleotide adenylyltransferase 2 Oligodendrocyte transcription factor 3 cAMP-dependent protein kinase inhibitor α Protein tyrosine phosphatase nonreceptor type 22 Pituitary tumor–transforming 1 PX domain containing serine/threonine kinase–like Solute carrier family 15 member 4 Solute carrier family 17 member 4 Signal transducer and activator of transcription 4 Transmembrane protein 39A-translocase of inner mitochondrial membrane Mitochondrial membrane domain containing 1 TNF α-induced protein 3/A20 TNF superfamily member 4/Ox40 ligand TNFAIP3 interacting protein 1 Tyrosine kinase 2 Ubiquitin conjugating enzyme E2 L3 WDFY family member 4 |
| AFRICAN BLK IRF5 ITGAM PLAT PTTG1 |
B-lymphocyte–specific tyrosine kinase Interferon regulatory factor 5 Integrin subunit α M Plasminogen activator, tissue type Pituitary tumor–transforming 1 |
| HISPANIC CLEC16A GALC IRF5 ITGAM NCF2 TNIP1 STAT4 |
C-type lectin domain containing 16A Galactosylceramidase Interferon regulatory factor 5 Integrin subunit alpha M Neutrophil cytosolic factor 2 TNFAIP3 interacting protein 1 Signal transducer and activator of transcription 4 |
∗ Genes listed have P value <5 × 10 −8 . cAMP = cyclic adenosine monophosphate; ETS = erythroblast transformation specific; HLA = human leukocyte antigen; IgG = immunoglobulin G; JAZF = juxtaposed with another zinc finger protein; PX = phox homology; TNF = tumor necrosis factor; TNFAIP3 = tumor necrosis factor, alpha-induced protein 3.
Epidemiologic studies have identified several classes of potential environmental triggers for lupus, including exposure to silica, smoking, and use of oral contraceptive agents or postmenopausal hormones. A role for microbial triggers—particularly virus infection—has been postulated, consistent with the constitutional symptoms that often characterize the earliest stage of the disease. Prior infection with Epstein-Barr virus ( Chapter 348 ) is significantly higher in SLE patients than in the general population (99 vs. 94%). Orthologs of human Ro60, a common target of lupus autoantibodies, are seen in commensal bacteria. Ultraviolet light exposure is a well-described trigger of lupus flares. Possible mechanisms include DNA damage, induction of cellular stress responses, and induction of apoptosis of skin cells, which result in concentration of nucleic acids and associated proteins in cell membrane blebs that can be processed by antigen-presenting cells. Certain drugs, including procainamide and hydralazine, can induce a lupus-like syndrome, but the symptoms usually abate after discontinuing use of the drug. These agents may promote demethylation of DNA, thereby altering gene expression and potentially increasing the availability of immunostimulatory DNA. Sulfa antibiotics have induced lupus flares in some patients. Administration of recombinant interferon-α to patients with hematologic malignancies or hepatitis C infection has been associated with induction of a lupus-like syndrome. In addition, anti–tumor necrosis factor agents have induced lupus autoantibodies and occasionally clinical lupus in patients with rheumatoid arthritis.
Genetic and environmental factors that increase the probability of development of SLE act on the immune system to induce autoimmunity and consequent tissue inflammation and damage. In addition to mechanisms that increase the availability of self-antigens (such as ultraviolet light), altered expression of gene products that mediate or regulate apoptosis or impaired clearance of apoptotic debris results in generalized activation of the immune system and contributes to autoimmunity in lupus. In parallel with the events that account for effective immune responses directed at exogenous microbes, the autoimmunity that occurs in SLE patients requires activation of both innate and adaptive immune responses ( Chapter 35 ). Characterization of the TLR family of pattern recognition receptors has provided new understanding of the mechanisms through which the innate immune system is activated by exogenous and endogenous stimuli, including nucleic acid–containing immune complexes, and promotes induction of a self-directed adaptive immune response ( Chapter 35 ). Impaired inactivation of the X-chromosome may augment the differentiation of B cells and the production of autoantibodies by increasing the expression of TLR7 or other X-chromosome–encoded immune response genes.
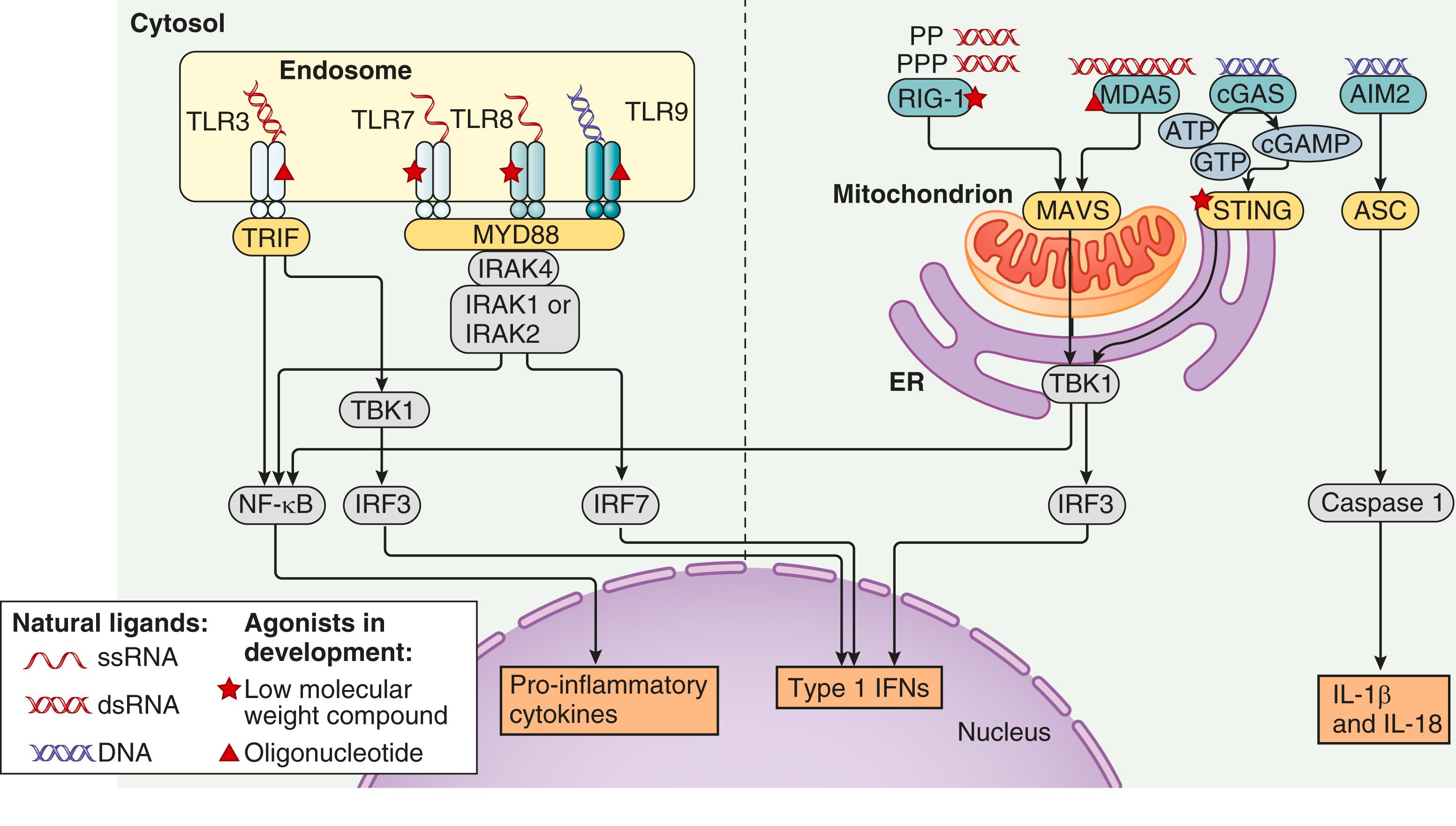
Studies of gene expression in peripheral blood mononuclear cells of SLE patients using microarray and RNA sequencing technology have demonstrated a sustained and broad “signature” of type I interferon–induced gene transcripts that reflect activation of the innate immune system. Interferon-α (IFN-α), along with other type I IFNs (e.g., IFN-β, IFN-ω, and IFN-κ in skin) may be responsible for many of the immunologic alterations observed in SLE. Immune complexes containing DNA or RNA, nuclear debris from neutrophils in the form of neutrophil extracellular traps (NETs), or extracellular mitochondrial DNA are among the stimuli postulated to induce the production of type I interferon in SLE. Demethylated CpG-rich DNA or RNA associated with nucleic acid–binding proteins can activate plasmacytoid dendritic cells and other immune system cells through TLRs and thereby result in the production of type I interferon and other proinflammatory cytokines ( E-Fig. 245-1 ). Induction of an immunostimulatory microenvironment by IFN-α may support the development of a humoral immune response directed at self-antigens, particularly intracellular particles that contain nucleic acids and nucleic acid–binding proteins. In addition to its effects on immune system function, type I interferon has been associated with altered endothelial cell function and microglial function in the brain and may contribute to the development of atherosclerotic vascular pathology and CNS disease in patients with lupus.
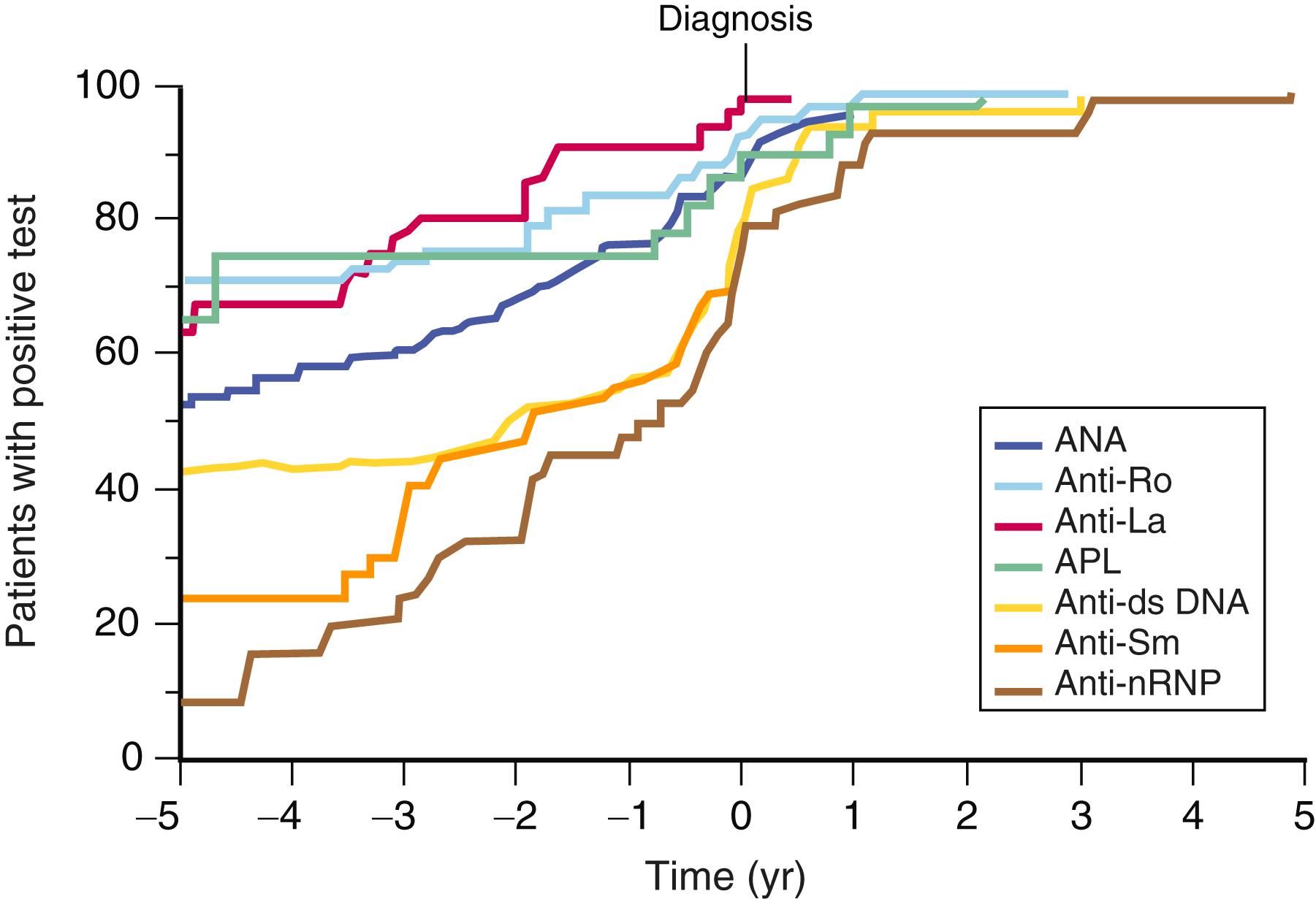
The most characteristic lupus autoantibodies target intracellular particles that contain both nucleic acid and nucleic acid–binding proteins. Analysis of the spectrum of autoantibodies present in the sera of individuals in whom SLE is later diagnosed suggests that autoantibodies reactive with certain RNA-binding proteins, including the Ro protein, occur early in the preclinical stage of the disease, along with a positive antinuclear antibody (ANA) test. These abnormal test results are often followed by anti-double-stranded DNA antibodies and, finally, by the development of antibodies specific for the spliceosomal proteins Smith (Sm) and ribonucleoprotein (RNP) at approximately the time of diagnosis ( E-Fig. 245-2 ). These observations suggest that individuals who demonstrate progression from antibodies targeting RNA to antibodies targeting DNA and spliceosomal proteins are those in whom sufficient autoimmunity develops to manifest clinical symptoms. Approximately one third of SLE patients have autoantibodies that are reactive with phospholipids or the proteins associated with them, particularly β 2 -glycoprotein I. These autoantibody specificities can also be present independently of SLE in primary antiphospholipid antibody syndrome ( Chapter 157 ).
Tissue and organ damage in SLE is mediated by the deposition or in situ formation of immune complexes, the activation of complement, and inflammation. The complement system ( Chapter 37 ), which is composed of more than 30 proteins that act in concert to protect the host against invading organisms, initiates inflammation and tissue injury. Activation of complement promotes chemotaxis of inflammatory cells and generates proteolytic fragments that enhance phagocytosis by neutrophils and monocytes. The classical complement pathway is activated when antibodies bind to antigen and generate potent effectors. Activation of the alternative complement pathway is initiated by the binding of spontaneously activated complement components to the surfaces of pathogens or self-tissues. C3a, which is an anaphylatoxin that binds to receptors on leukocytes and other cells, causes activation and release of inflammatory mediators. C5a is a potent soluble inflammatory, anaphylatoxic, and chemotactic molecule that promotes recruitment and activation of neutrophils and monocytes and mediates endothelial cell activation through its receptor. The release of reactive oxygen and nitrogen intermediates is an additional mechanism that contributes to tissue damage.
Tissues targeted by immune system activity in lupus include the skin (where immune complexes and complement are deposited in a linear pattern), the glomeruli, and heart valves. Antibodies reactive with hippocampal neurons in the brain can mediate excitotoxic death. Microvascular damage is observed in splenic arteries and is characterized by the typical onion-skin pattern of concentric connective tissue deposition. In addition to inflammation, thrombosis (including microthrombi) contributes to ischemia and cell necrosis in the brain and other organs.
In general, the pattern of renal disease reflects the site of deposition of immunoglobulins and the quality of the effector mechanisms they induce. Mesangial deposition of immunoglobulin induces mesangial cell proliferation and is associated with microscopic hematuria and mild proteinuria. Subendothelial deposition of immune complexes results in proliferative and exudative inflammation, together with hematuria, mild to moderate proteinuria, and reduced glomerular filtration rate. Subepithelial deposition of immune complexes adjacent to podocytes and along the glomerular basement membrane can result in membranous nephritis with nephrotic-range proteinuria. In addition, antiphospholipid antibodies may support the development of thrombotic or inflammatory vascular lesions within or external to glomeruli.
The pathophysiologic mechanisms that account for the neuropsychiatric manifestations of SLE are diverse and complex. Autoantibodies cross-reactive with neuronal cell surface glutamate receptors and DNA may mediate excitotoxic death of neurons and are proposed to contribute to cognitive dysfunction. Antibodies directed against ribosomal P protein have also been associated with neuropsychiatric lupus, and antiphospholipid antibodies can contribute to a procoagulant state, vascular thrombosis, and cerebral ischemia. Noninflammatory small vessel vasculopathy is the most common lesion and can be associated with microinfarcts. Inflammatory mediators, including the cytokines interleukin-6 (IL-6) and IFN-α, and matrix metalloproteinases may also contribute to the neuropsychiatric manifestations of SLE. IFN-α mediates microglial activation and excessive synaptic pruning.
SLE involves virtually all components of the immune system and can be accompanied by constitutional symptoms similar to those seen in the setting of microbial infection. Fatigue, headaches, weight loss, and fevers are common, along with generalized arthralgias, myalgias, and lymphadenopathy. The level of activity of lupus typically follows a pattern of flares and remissions, although some patients sustain active disease for prolonged periods.
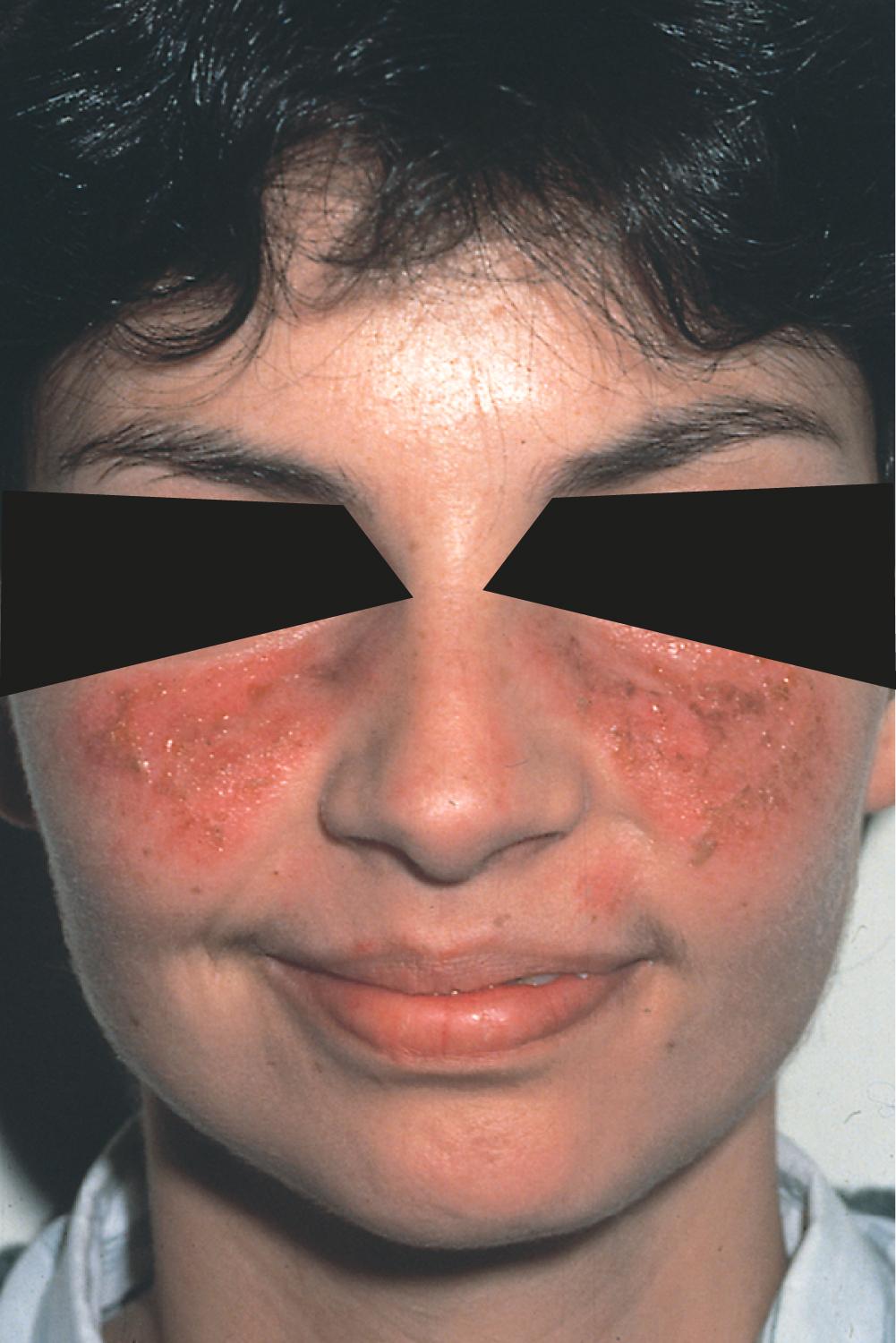
The skin and mucous membranes are affected in most lupus patients ( Table 245-1 ). The classic erythematous facial rash, with a butterfly distribution across the malar and nasal prominences and sparing of the nasolabial folds, is seen in 30 to 60% of patients ( Fig. 245-1 ). The butterfly rash is often triggered by sun exposure, but photosensitivity can also occur diffusely in other areas of the body.
| MANIFESTATION | APPROXIMATE FREQUENCY (%) |
|---|---|
| Cutaneous | 88 |
| Arthritis/arthralgias | 76 |
| Neuropsychiatric | 66 |
| Pleurisy/pericarditis | 63 |
| Anemia | 57 |
| Raynaud phenomenon | 44 |
| Vasculitis | 43 |
| Atherosclerosis | 37 |
| Nephritis | 31 |
| Thrombocytopenia | 30 |
| Sensorimotor neuropathy | 28 |
| Cardiac valvar disease | 18 |
| Pulmonary alveolar hemorrhage | 12 |
| Pancreatitis | 10 |
| Myositis | 5 |
| Myocarditis | 5 |
Discoid skin lesions, which are seen in about 25% of patients, are erythematous plaques with central scarring, sometimes covered with scale and involving the scalp, face, and ears. Patients may develop alopecia ( Chapter 409 ). Discoid lesions can be present in the absence of systemic manifestations of SLE (discoid lupus). In addition to the scarring alopecia of discoid lupus, transient alopecia may be a clinical sign of increased disease activity.
Inflammation of the deep dermis and subcutaneous fat can result in lupus panniculitis, with firm, painful nodules that sometimes adhere to the epidermis, thereby causing irregularities in the superficial skin. Subacute cutaneous lupus erythematosus is seen in sun-exposed areas and can involve erythematous plaques or psoriasiform lesions. It is associated with autoantibodies to the Ro (SSA) RNA-binding protein. Mucosal ulcerations, especially of the buccal mucosa and upper palate, result from mucositis and are typical of SLE. Manifestations of vasculopathy include arteriolar spasm or infarcts in the nail folds, a diffuse lacey pattern over the skin described as livedo reticularis, and petechial-purpuric or urticarial lesions on the extremities. Vasculopathy in SLE is often associated with the presence of antiphospholipid antibodies.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here