Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Structural heart disease intervention is a rapidly growing field that has expanded beyond transcatheter aortic valve implantation to include procedures that address each cardiac valve, as well as paravalvular leak, intracardiac shunts, and left atrial appendage occlusion for stroke and systemic embolism prevention. New devices and new indications for existing devices continue to be investigated.
Transcatheter aortic valve implantation is one of the most widely adopted structural heart disease procedures and is commonly performed using a balloon-expandable or self-expanding transcatheter heart valve.
Transcatheter edge-to-edge repair of the mitral valve recently received approval for commercial use in the United States to treat functional mitral regurgitation, in addition to the previously approved indication of degenerative mitral regurgitation.
It is important for the cardiac anesthesiologist to be familiar with the various structural heart disease procedures, their indications and contraindications, general procedural steps, and anesthetic considerations.
The cardiac anesthesiologist should be aware of the potential complications of each procedure and be prepared to diagnose and manage them.
Intraprocedural imaging guidance with echocardiography is a critical part of many structural heart disease procedures and is often performed by the cardiac anesthesiologist.
Structural heart disease refers to noncoronary heart disease for which some therapy, surgical or percutaneous, exists. This broadly includes all anatomic cardiac pathology involving the valves or walls of the heart. While the term is not new, it has recently become a common way to group together cardiac conditions that may be treated with newly described transcatheter procedures.
Due to the rapid development of catheter-based therapies, the contemporary evaluation of patients with structural heart disease requires a new paradigm. Previously when patients were diagnosed with a valve condition, they would be referred to a cardiac surgeon who would decide whether or not to perform traditional open-heart surgery. With the proliferation of transcatheter procedures as alternative nonsurgical options, patients are now evaluated by a multidisciplinary panel of specialists termed the heart team , and this model has been adopted by both US and European cardiac societies as standard practice in 2021. Consisting of cardiac surgeons, interventional cardiologists, cardiovascular imaging experts, cardiac anesthesiologists, and others, this diverse group of specialists is best suited to formulate the most appropriate course of action after discussion with the primary care team and patient. This approach is known as shared decision-making. Due to their role on the heart team and involvement intraprocedurally, it is essential that cardiac anesthesiologists understand the full breadth of structural heart disease procedures.
No structural heart disease procedure has become more universally accepted and widely adopted than transcatheter aortic valve implantation (TAVI, also known as transcatheter aortic valve replacement [TAVR]), a definitive treatment for aortic stenosis (AS). The first human TAVI was performed in April 2002, and after years of investigation, the United States Food and Drug Administration (FDA) approved the device for commercial use in November 2011. , By 2016 TAVI volume first exceeded surgical aortic valve replacement (SAVR) volume and in 2019, 72,991 commercial implants were performed in the United States with a 30-day mortality rate of 2.5% ( Fig. 23.1 ).
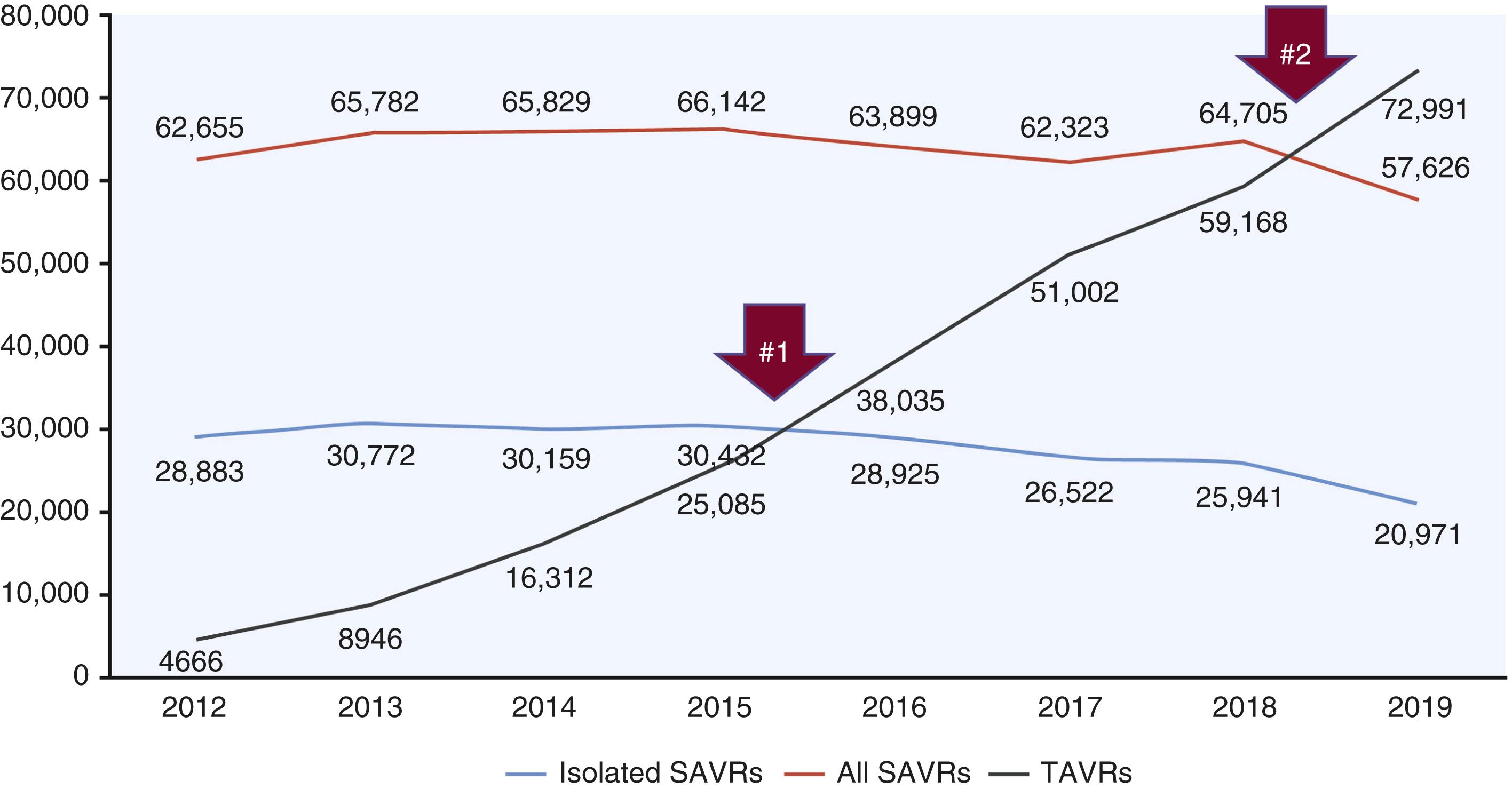
While improvements in device engineering and operator experience largely account for the success of TAVI, challenges persist, and despite expanded indications, not all individuals with AS are candidates. Because TAVI is a relatively new therapy and performed primarily in patients with advanced age, there are limited long-term data on device durability in comparison to SAVR.
While numerous TAVI systems have been used experimentally and commercially over the past 20 years, nearly all TAVI is currently performed using one of two devices. A third system has been recently approved for select patient populations ( Fig. 23.2 ). Each system has a unique mechanism of deployment and carries advantages and disadvantages. Because most centers are capable of using either major system, selection should be made based on unique patient considerations and anatomic features.
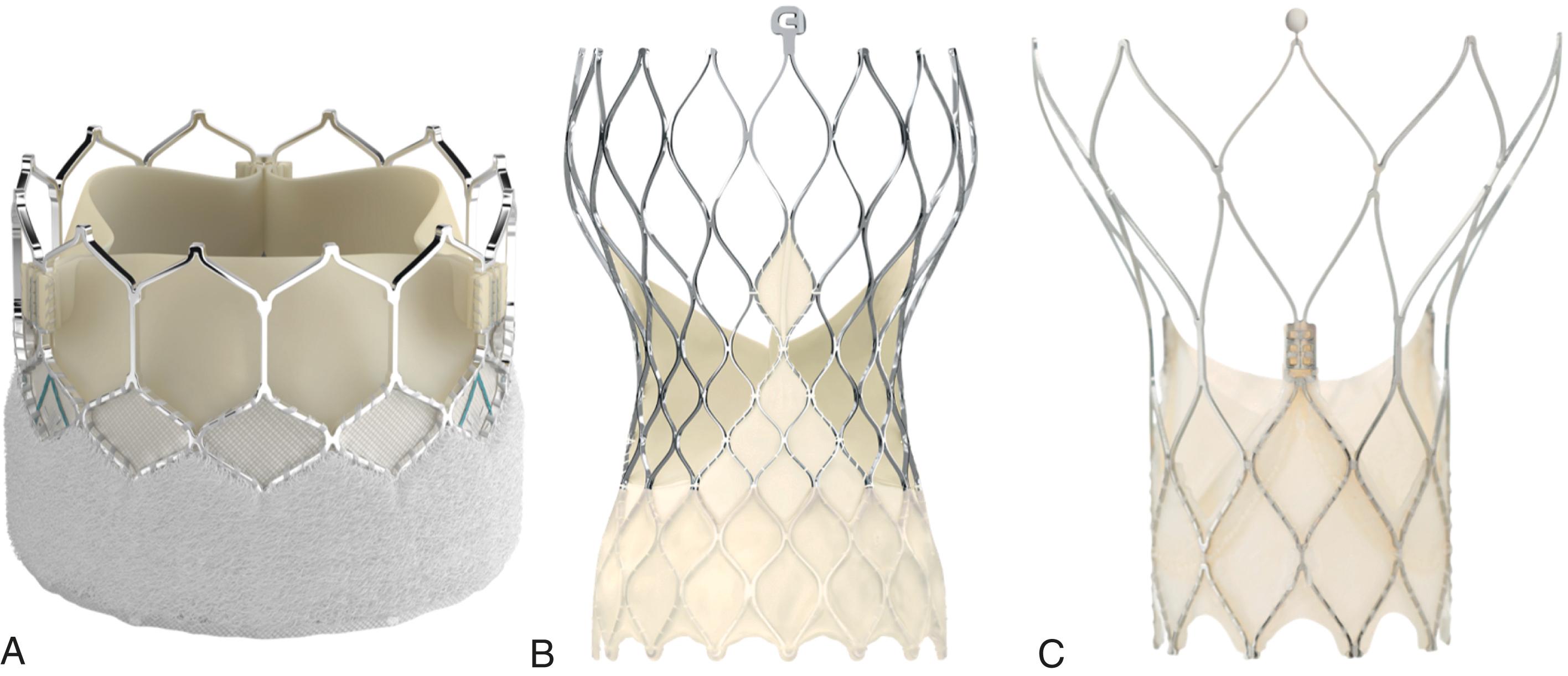
The SAPIEN 3 transcatheter heart valve (THV) (Edwards Lifesciences LLC, Irvine, CA) is a trileaflet, bovine pericardium intraannular valve mounted into a radiopaque, cobalt-chromium alloy frame. The 29-mm valve is not currently available in the latest model, the SAPIEN 3 Ultra. An internal polyethylene terephthalate skirt at the inflow portion of the valve is larger than previous versions. A second external skirt wrapped around the stent frame is also taller than previous iterations and helps to seal the valve in order to reduce paravalvular regurgitation (PVR). The overall stent frame profile is low, which allows for less complicated coronary access after TAVI.
The SAPIEN 3 Ultra THV is mounted to the delivery system, which consists of a balloon catheter and a separate flexible catheter to aid in positioning. The valve is then prepared with a crimper that reduces the overall diameter of the system, allowing insertion into a 14- or 16-Fr expandable vascular access sheath, depending on valve size. This sheath expands transiently to allow passage of the valve and can be used in patients with a vessel diameter ≥5.5 or 6 mm. This represents a significant improvement from previous generations and enables more transfemoral and percutaneous access. Valve expansion occurs by balloon inflation. Following valve expansion, the balloon is deflated and removed from within the stent frame. The SAPIEN 3 THV is not repositionable once expanded; however, deployment is generally straightforward.
The CoreValve Evolut R System (Medtronic, Minneapolis, MN) is a trileaflet porcine pericardium valve sutured on a radiopaque nitinol alloy frame made of nickel and titanium. Improving on a previous design, the Evolut Pro and Evolut Pro+ systems have an additional external tissue wrap to reduce PVR. The valve is engineered to be supraannular to achieve a higher effective orifice area and lower pressure gradients. An increased frame height compared to the balloon-expandable valve may make future coronary access more challenging.
A proprietary loading system compresses the bioprosthesis into a flexible, nonsteerable external delivery catheter. This catheter system uses a true 14-, 16-, or 18-Fr inline sheath, depending on valve size. Because the sheath retains its outer diameter and does not expand, it can be used with access vessel diameters as small as 5.0, 5.5, or 6.0 mm, respectively. The catheter maintains the valve in a compressed position until it is positioned across the native annulus. The catheter is then retracted allowing the valve to self-expand and conform to the native annulus with a consistent radial force. If the position is suboptimal, the delivery catheter can recapture and reposition the valve after partial deployment of up to 80%.
The Portico TAVI System (Abbott Structural Heart, Santa Clara, CA) is a fully resheathable, repositionable, and retrievable self-expanding valve. Receiving FDA approval in September 2021, it is currently used in a minority of cases.
TAVI has been well studied and is the only therapy for severe AS shown to prolong survival in a randomized clinical trial. Data from industry-sponsored pivotal clinical trials for balloon-expandable and self-expanding TAVI systems are available comparing TAVI to SAVR.
The Placement of AoRTic TraNscathetER Valve (PARTNER) trial investigators randomly assigned 699 high-risk patients from 25 centers as part of the PARTNER 1 trial and found balloon-expandable TAVI to be noninferior to SAVR in 1 year survival (rate of death 24.2% vs 26.8%; P = .001 for noninferiority). Concurrently, a high-risk trial evaluating self-expanding valve systems in 795 patients at 45 centers found TAVI to be superior to SAVR in 1 year survival (rate of death 14.2% vs 19.1%; P < .001 for noninferiority, P = .04 for superiority).
The follow-up PARTNER 2 trial randomized 2032 intermediate-risk patients at 57 centers to either balloon-expandable TAVI or SAVR. Results showed TAVI was similar to SAVR using the primary endpoint of death or disabling stroke at 2 years (19.3% vs 21.1%; P = .001 for noninferiority). The Surgical Replacement and Transcatheter Aortic Valve Implantation (SURTAVI) trial similarly evaluated the self-expanding prosthesis by randomizing 1746 intermediate-risk patients at 87 centers to either TAVI or SAVR. In this population, TAVI was again found to be noninferior to SAVR with respect to the primary endpoint of death or disabling stroke at 2 years using Bayesian analysis to calculate a 2-year probability based on 1 year data (12.6% vs 14.0%; probability of noninferiority > .999).
To evaluate the low surgical–risk population, the PARTNER 3 trial again randomized 1000 patients at 71 centers to either balloon-expandable TAVI or SAVR. The composite rate of death, stroke, or rehospitalization at 1 year was significantly lower with TAVI compared to SAVR (8.5% vs 15.1%; P < .001 for noninferiority, P = .001 for superiority). A comparable low-risk randomized study of 1403 patients at 86 centers using a self-expanding prosthesis found TAVI to be similar to SAVR with respect to the composite endpoint of death or disabling stroke at 24 months using Bayesian methods (5.3% vs 6.7%; probability of noninferiority > .999).
The CHOICE trial randomized 241 high-risk patients to either balloon-expandable or self-expanding TAVI systems. Device success was greater in the balloon-expandable group (95.9% vs 77.5%; P < .001), attributable to a less frequent need for more than one valve, and a lower rate of residual aortic regurgitation that was more than mild. The combined safety endpoint was similar between valve systems; however, permanent pacemaker implantation was more frequent in the self-expanding group. After 5 years, the balloon-expandable group had a smaller prosthetic valve area and a greater mean gradient. The rates of death, stroke, and repeat hospitalization for heart failure were similar at 1 and 5 years between groups. ,
Due to the recency of TAVI development and the actuarial life expectancy of the early cohort of TAVI patients, limited long-term data exist. Although not designed to assess long-term outcomes, 8-year data from the Nordic Aortic Valve Intervention (NOTION) trial showed no difference between self-expanding TAVI or SAVR in the primary composite endpoint of all-cause mortality, stroke, or myocardial infarction (54.5% vs 54.8%; P = .94), and no difference in any component. Randomized patients were low risk for surgery with a mean age of 79.1 years. Structural valve deterioration was lower with TAVI (13.9% vs 28.3%; P = .002); however, risk of bioprosthetic valve failure was similar (8.7% vs 10.5%; P = .61).
Outside of clinical trials, numerous multicenter registries allow for the study of TAVI in a real-world, nonrandomized population. The Society of Thoracic Surgeons–American College of Cardiology Transcatheter Valve Therapy (STS/ACC TVT) Registry is a Centers for Medicare & Medicaid Services mandated registry of commercial TAVI at all US centers. Registry data show TAVI is now performed in younger and healthier patients. In 2019, the median age of patients undergoing TAVI was 80 years, with a median 30-day STS predicted risk of mortality of 4.4%, compared to 84 years and 6.9% in 2013 ( Table 23.1 ). Registry results also showed an in-hospital stroke rate of 1.6%, a permanent pacemaker implantation rate of 8.3%, and a 1-year all-cause mortality rate of 10.2%, all significantly lower over time.
| Level | Overall ( n = 276,316) | ≤2013 ( n = 13,723) | 2014 ( n = 16,312) | 2015 ( n = 25,085) | 2016 ( n = 38,035) | 2017 ( n = 51,002) | 2018 ( n = 59,168) | 2019 ( n = 72,991) | P Value | |
|---|---|---|---|---|---|---|---|---|---|---|
| Demographics | ||||||||||
| Age, yr a | n [median] | 276,316 [81.00] | 13,723 [84.00] | 16,312 [83.00] | 25,085 [83.00] | 38,035 [82.00] | 51,002 [81.00] | 59,168 [81.00] | 72,991 [80.00] | <.0001 |
| Sex | Male | 149,657 (54.16) | 6704 (48.85) | 8587 (52.64) | 13,250 (52.82) | 20,533 (53.98) | 27,701 (54.31) | 32,171 (54.37) | 40,711 (55.78) | <.0001 |
| History and Risk Factors | ||||||||||
| % Predicted mortality (STS SAVR model) | n [median] | 276,282 [5.22] | 13,720 [6.91] | 16,309 [6.65] | 25,079 [6.26] | 38,031 [5.73] | 50,994 [5.12] | 59,165 [4.89] | 72,984 [4.38] | <.0001 |
| Procedure Information | ||||||||||
| Valve sheath access site | Femoral | 248,985 (90.11) | 7833 (57.08) | 11,335 (69.49) | 21,733 (86.64) | 35,028 (92.09) | 47,780 (93.68) | 55,743 (94.21) | 69,533 (95.26) | <.0001 |
| Heart team reason for procedure | <0.0001 | |||||||||
| Inoperable or extreme/high risk | 179,397 (64.92) | 13,164 (95.93) | 15,552 (95.34) | 23,712 (94.53) | 33,284 (87.51) | 31,049 (60.88) | 31,038 (52.46) | 31,598 (43.29) | ||
| Intermediate risk | 84,108 (30.44) | 11 (0.08) | 573 (3.51) | 1078 (4.30) | 4318 (11.35) | 18,863 (36.98) | 26,568 (44.90) | 32,697 (44.80) | ||
| Low risk | 11,534 (4.17) | 0 (0.00) | 85 (0.52) | 253 (1.01) | 371 (0.98) | 983 (1.93) | 1447 (2.45) | 8395 (11.50) | ||
| Conversion to open-heart surgery | Yes | 1611 (0.58) | 192 (1.40) | 199 (1.22) | 208 (0.83) | 195 (0.51) | 242 (0.47) | 278 (0.47) | 297 (0.41) | <0.0001 |
| Mortality | ||||||||||
| Discharge mortality status | Deceased | 5559 (2.01) | 736 (5.36) | 662 (4.06) | 720 (2.87) | 757 (1.99) | 849 (1.66) | 880 (1.49) | 955 (1.31) | <.0001 |
| 30-Day death (30 days) | Deceased | 7980 (3.12) | 904 (6.59) | 908 (5.57) | 1032 (4.11) | 1132 (2.98) | 1380 (2.71) | 1445 (2.44) | 1179 (2.26) | <.0001 |
| 1-yr Death (1 yr) | Deceased | 22,979 (12.21) | 2478 (18.06) | 2642 (16.20) | 3406 (13.58) | 4387 (11.53) | 5590 (10.96) | 4476 (10.15) | — | <.0001 |
| Nonfatal Endpoints, In-Hospital | ||||||||||
| Any stroke | Yes | 5009 (1.81) | 288 (2.10) | 360 (2.21) | 502 (2.00) | 702 (1.85) | 944 (1.85) | 1056 (1.78) | 1157 (1.59) | <.0001 |
| Pacemaker (v1.3) | Yes | 22,911 (9.98) | 170 (9.10) | 1765 (12.95) | 2798 (13.23) | 3594 (11.02) | 4386 (9.88) | 4833 (9.38) | 5365 (8.34) | <.0001 |
| Major Vascular access site complications (v1.3) | Yes | 3210 (1.21) | 35 (1.57) | 196 (1.20) | 334 (1.33) | 457 (1.20) | 603 (1.18) | 663 (1.12) | 922 (1.26) | .7639 |
| Echocardiogram Outcomes | ||||||||||
| In-hospital/30-day aortic insufficiency (30 days) | None/trace/mild | 235,208 (92.05) | 10,758 (78.39) | 13,779 (84.47) | 21,948 (87.49) | 34,820 (91.55) | 48,108 (94.33) | 56,119 (94.85) | 49,676 (95.16) | <.0001 |
| Length of Stay | ||||||||||
| Length of stay | n [median] | 276,316 [3.00] | 13,723 [7.00] | 16,312 [6.00] | 25,085 [4.00] | 38,035 [3.00] | 51,002 [2.00] | 59,168 [2.00] | 72,991 [2.00] | <.0001 |
a P values are based on chi-square 1 degree of freedom rank correlation statistics for all continuous/ordinal row variables. All tests treat the column variable as an ordinal.
The FRANCE-2 (French Aortic National CoreValve and Edwards) registry enrolled all 4201 patients who underwent TAVI in France during a 2-year period. In this high-risk population, 5-year mortality was 60.8%, comparable to the 5-year results from the TAVI group of the PARTNER 1 trial (67.8%). A national registry from all centers in Finland performing TAVI was used to retrospectively compare TAVI to SAVR in a low-risk population. Patients were propensity matched producing 304 pairs with similar characteristics, and no difference in 30-day or 3-year survival was reported. TAVI was associated with shorter hospital length of stay and a lower rate of major perioperative bleeding. However, SAVR was associated with less PVR and permanent pacemaker implantation.
Not all studies have been favorable to TAVI. The OBSERVANT (Observational Study of Effectiveness of SAVR-TAVI Procedures for Severe Aortic Stenosis Treatment) Study is an observational, prospective, multicenter cohort study that enrolled consecutive patients with AS undergoing TAVI or SAVR at 93 Italian centers. In an analysis of 650 propensity-matched groups at low and intermediate risk, the rates of death and major cardiac and cerebrovascular events were similar at 1 year. However, at 5 years, these outcomes were more frequent in the TAVI group. Furthermore, in a sample of 355 pairs of low-risk patients, 3-year survival and freedom from major cardiac and cerebrovascular events was also higher in the SAVR group. Further comparative long-term data using current generation technology from randomized controlled trials and clinical registries are necessary to determine appropriate use in all populations.
As of August 2019, both leading balloon-expandable and self-expanding TAVI systems are FDA approved for use and indicated for relief of AS in patients at all levels of surgical risk with symptomatic heart disease due to severe native calcific AS who are judged by a heart team to be appropriate. For each valve system, the FDA took a stepwise approach to approval based on available evidence of safety and efficacy, first approving its use in nonoperable and high-surgical-risk patients. As data from intermediate- and low-risk studies became available, the indications for use were expanded to include these patient populations. Both leading TAVI systems are also indicated for use in patients with symptomatic heart disease due to failure (stenosed, insufficient, or combined) of a surgical bioprosthetic aortic valve who are judged by a heart team to be at high or greater risk for SAVR. The Portico TAVI system is restricted to use in patients at high or extreme risk for surgery.
In addition to the FDA indications, society guidelines recommend aortic valve intervention (TAVI or SAVR) in a subset of patients with asymptomatic disease, including those with a left ventricular (LV) ejection fraction <50% or <55%. , US guidelines state TAVI is recommended over SAVR for patients >80 years of age or with a life expectancy of <10 years. European guidelines recommend TAVI in patients ≥75 years or in those who are high risk for SAVR. In all cases, the mode of intervention should be determined by an experienced heart team after weighing risks and benefits of each approach.
TAVI is contraindicated in patients who cannot tolerate an anticoagulation or antiplatelet regimen, or who have active bacterial endocarditis or other active infections. Use of the self-expanding TAVI system is also contraindicated in patients with hypersensitivity to titanium or nickel. Relative contraindications include a life expectancy less than 12 months due to a noncardiac cause, a low coronary ostium height, or atypical cardiac anatomy including a bicuspid or noncalcified aortic valve.
All patients with known or suspected AS should be evaluated by a heart team capable of performing both TAVI and SAVR. A thorough history and physical examination should be performed to determine symptoms and comorbid disease. An assessment of functional status and frailty should be completed. A baseline electrocardiogram should be reviewed to evaluate the rhythm and identify conduction disturbances. A comprehensive echocardiogram with Doppler interrogation should be performed to determine severity of disease, baseline biventricular function, and to detect concurrent valve disease. Transthoracic echocardiography (TTE) is most frequently used.
Once severe AS is confirmed, multidetector computed tomography (MDCT) should be performed and serves multiple purposes. First, it provides necessary information about arterial caliber, tortuosity, and calcium burden that determines suitability for femoral or alternative access sites. Second, MDCT may be able to rule out concurrent coronary artery disease in some patients and thus eliminate the need for cardiac catheterization. Third, MDCT best defines the anatomy of the aortic root including the coronary ostia, aortic annulus, and LV outflow tract. MDCT is the gold standard for annular measurements used for valve sizing. While three-dimensional (3D) transesophageal echocardiography (TEE) may provide similar measurements when MDCT data are not available, it cannot assess coronary artery or access site anatomy. Whatever studies are to be performed, they should be completed prior to the procedure date to allow proper planning.
When all information is available the heart team should formulate a plan for intervention based on disease, risk assessment, comorbidities, and frailty. Options include balloon aortic valvuloplasty (BAV), TAVI, or SAVR. When TAVI is considered, both type and size of valve should be determined as well as the need for necessary concurrent procedures including those for cerebral embolic or coronary protection. The heart team recommendation should then be discussed with the patient and their primary physician before a final decision is made, as part of a shared decision-making approach.
In 2019, 95.3% of TAVI performed in the United States was done with femoral arterial access, although transapical, transaortic, subclavian, transcarotid, and transcaval approaches were also used. For the transfemoral approach, bilateral access is required. First, a 7-Fr introducer sheath is placed on the nontreatment side. Through this, a 5-Fr pigtail catheter is guided into the noncoronary cusp of the aortic root under fluoroscopy. Blood pressure can be monitored by transducing the sheath or catheter. Next, treatment side access is gained with a 5-Fr sheath. Heparin is administered to a target activated clotting time of 250 seconds and vascular site closure devices are prepositioned to prepare for the delivery sheath.
Rapid ventricular pacing is required for balloon-expandable systems but may also assist BAV and deployment of self-expanding systems. Pacing may also be necessary in the setting of new-onset conduction disturbances and therefore should be obtained for all TAVI. Temporary right ventricular (RV) pacing can be achieved via the right internal jugular vein or femoral vein and maintained postoperatively. Temporary LV pacing has also been described.
Once access is achieved, the aortic valve is crossed with an exchange-length wire via the treatment side sheath. Ventricular ectopy may occur. Predeployment BAV may be performed when indicated. The TAVI delivery system is then placed over the wire and across the valve. During deployment, diminished cardiac output should be anticipated transiently; however, more serious complications such as coronary obstruction, ventricular wire perforation, or vascular injury must be ruled out if hypotension persists. Appropriate valve position and function after deployment is confirmed with fluoroscopy and echocardiography. Post-deployment BAV may be performed to correct PVR. The wire and delivery system are withdrawn, and protamine is administered. The arteriotomy is then closed by tightening the previously placed closure devices. Peripheral angiography can be used to rule out vascular injury.
Contemporary TAVI can be performed under monitored anesthesia care (MAC) or general anesthesia (GA). As of 2019, 64.1% of percutaneous transfemoral TAVI in the United States was performed under sedation ( Fig. 23.3 ). The use of less invasive access, as well as experience with the procedure have led to an increasing use of MAC and an overall less invasive anesthetic management, including fewer invasive lines and monitors and faster extubation when an endotracheal tube is used. Continuous blood pressure monitoring can be obtained by placement of a radial arterial catheter or by transducing the procedural access site. Central venous access for fluid administration may also be shared from the procedural field. Pulmonary artery (PA) catheters are no longer frequently used.
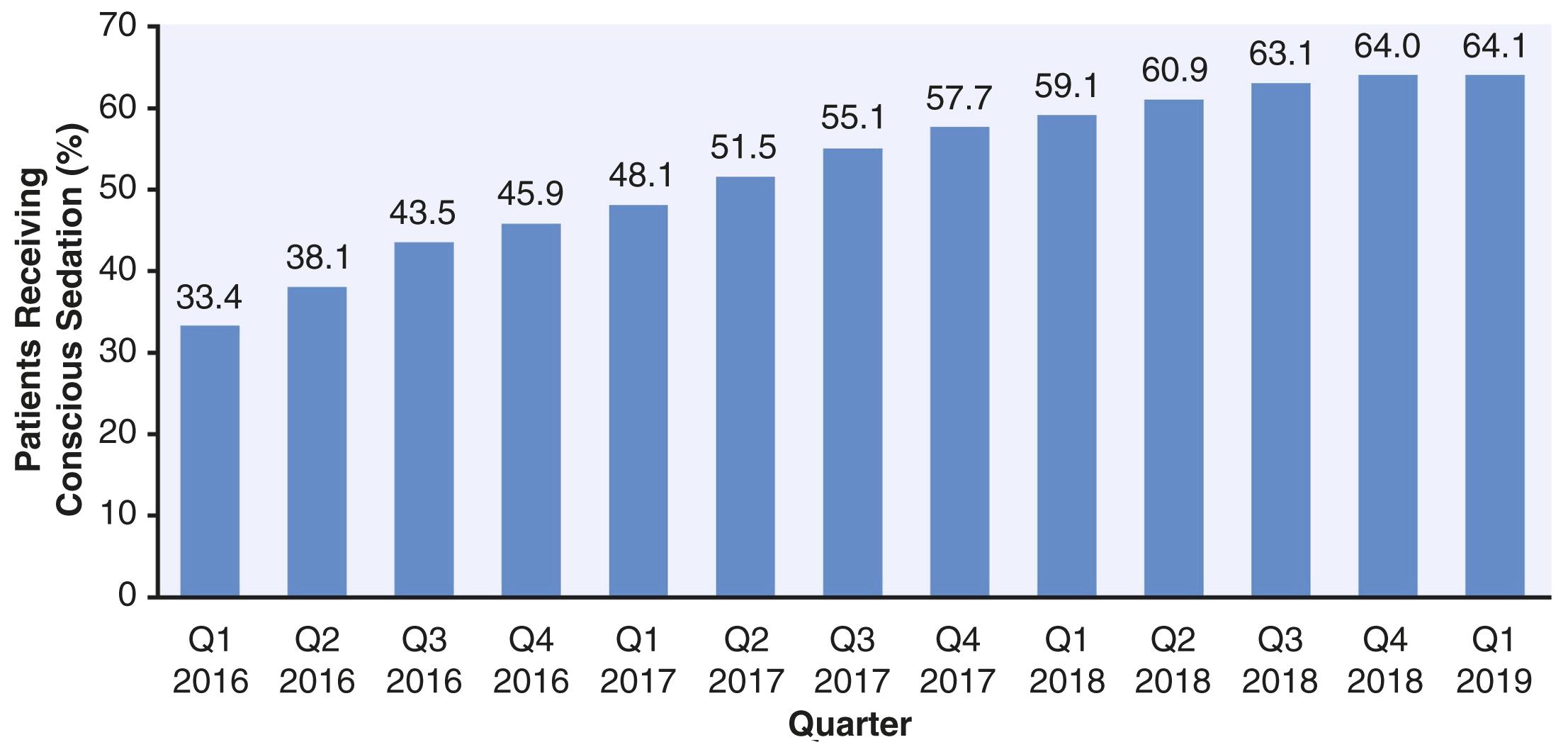
In TAVI, indications for GA are similar to other procedures and include a high risk of aspiration, obstructive sleep apnea, and patient preference. The incidence of conversion to GA in current practice is between 0.6% and 5.9% and may occur due to procedural complications or poor tolerance of sedation. , Procedure-specific indications for GA in TAVI include the use of alternative access sites or institutional practice. The use of TEE may also encourage but does not require the use of GA. When MAC is selected, the use of various short-acting infusions has been described, including dexmedetomidine, propofol, and remifentanil. These medications and volatile anesthetics have also been used for the maintenance of GA. Inotropic and vasoconstrictive medications should be available but are not frequently required. Sedation under the direction of the proceduralist and without an anesthesiologist has been described.
While improved procedural outcomes have facilitated a transition in anesthetic technique, there is evidence suggesting the choice of anesthesia care itself may be associated with better outcomes. A study comparing conscious sedation and GA in 10,997 patients undergoing transfemoral TAVI found a decrease in in-hospital mortality, 30-day mortality, a composite endpoint of stroke and mortality, and hospital length of stay associated with avoidance of GA.
TAVI is typically performed using either TTE or TEE, although it is also performed with fluoroscopy as the sole intraprocedural imaging modality. As the more invasive approach, TEE should generally be reserved for when GA is used, or when a clinical question remains after other imaging studies. Interventional echocardiography for TAVI can be performed by an anesthesiologist, a cardiologist, or an echocardiography technician when reviewed by the procedural team.
A baseline echocardiogram with spectral Doppler should be performed to confirm the presence of AS, quantify biventricular function, and detect any pericardial effusion. When using TTE, it is particularly important to identify the windows that best visualize cardiac structures preprocedurally in order to expedite subsequent imaging. The goals of post-deployment echocardiography are to evaluate valve seal, peak velocity, shape, and to identify any complications. A well-functioning valve should be without significant transvalvular or PVR with color-Doppler interrogation on short and long axis. Spectral Doppler measured peak systolic velocity should be <2.0 m/s with an early peaking morphology. The valve should appear circular on short axis and only a few millimeters below the native annulus on long axis. Self-expanding valves should be evaluated after partial deployment and findings may indicate the need to recapture or reposition before final release. Both valve types should be expeditiously evaluated post deployment in the presence of hemodynamic instability. Valve migration, new wall motion abnormalities, PVR, aortic injury, and pericardial effusion can all be diagnosed with echocardiography. Identification of these findings requires a discussion with the procedural team and may necessitate balloon post dilation, placement of a second valve, pericardiocentesis or sternotomy.
Routine TAVI completed without complication no longer requires postoperative management in the intensive care unit (ICU). Conduction disturbances remain a common complication, with 8.3% of TAVI requiring permanent pacemaker implantation during index hospitalization. Conduction disturbances most frequently occur intraprocedurally; however, delayed-onset complete heart block may manifest hours or days after TAVI. Telemetry monitoring is required for all patients and an electrocardiogram should be evaluated when changes from baseline are suspected. Access site bleeding or hematoma can occur, and patients should remain supine and limit hip flexion for a minimum of 2 to 4 hours. After this period, early ambulation is encouraged as enhanced recovery protocols allow for early discharge as soon as postoperative day 1. In 2019, the median US length of stay was 2 days. While anticoagulation protocols vary, evidence from a randomized controlled trial found that compared to aspirin and clopidogrel, patients receiving aspirin monotherapy had lower rates of bleeding without an increase in thromboembolic events.
Further expansion of use in patients with bicuspid or other atypical valve morphologies will likely continue. However before widespread use of TAVI can proceed in those with longer life expectancies, there needs to be a better understanding of valve durability, defined as the absence of bioprosthetic valve dysfunction. There is current ongoing research into understanding how subclinical changes over time may progress to outright valve failure that results in mortality or the need for valve reintervention. The finding of hypoattenuated leaflet thickening, a diagnosis made by MDCT, may be an important marker. While this process frequently self-resolves, the FDA has mandated surveillance and 10-year follow-up of the pivotal clinical trials to determine whether progression may result in clinically significant events. Finally, further study using the Portico TAVI System will evaluate use in an expanded patient population.
BAV, also known as percutaneous aortic balloon dilation, was first performed in September 1985 as an alternative to SAVR for AS. Due to high procedural morbidity and restenosis, enthusiasm was limited. The recent development of TAVI has led to greater experience with catheter-based techniques, which in turn has resulted in refinements in technology and practices that have improved outcomes in BAV.
A retrospective review of 1874 BAV procedures from a national US registry between 2010 and 2013 found an in-procedure mortality rate of 1.5% and an in-hospital mortality rate of 8.1% with low institutional volume, a significant predictor of mortality. Permanent pacemaker implantation occurred in 3.3% and stroke in 1.8%.
An early randomized controlled trial of nonsurgical candidates compared TAVI to a standard therapy group, of which 84.8% received BAV. The standard therapy group had a significantly greater 1-year mortality rate (50.7% vs 30.7%; P < .001). Separate covariate analyses were performed in the standard therapy group comparing those that did and did not undergo BAV, and no significant difference in mortality was found. Compared to TAVI, standard therapy was associated with a lower incidence of stroke (5.0% vs 1.1%; P = .06) and major vascular complications (16.2% vs 1.1%; P < .001). Repeat BAV was performed in 26.3% within 1 year.
Society guidelines state BAV is indicated for patients with severe symptomatic AS as a bridge to decision regarding definitive treatment with SAVR or TAVI. , There is a role for BAV in noncalcified, congenital AS in pediatric patients. BAV should be considered when feasible during pregnancy if severe symptoms are present despite medical therapy. In patients with symptomatic disease and a life expectancy of less than 12 months, BAV should be considered as a palliative measure. There is inconsistency between guidelines regarding the use of BAV before urgent high-risk noncardiac surgery. , BAV has a role during TAVI for predilation and as a treatment for incomplete deployment or PVR. Contraindications include moderate or severe aortic regurgitation, endocarditis, or when vascular access is prohibitive. BAV should not be considered a long-term alternative to SAVR or TAVI.
Quantification of AS should be performed before BAV and repeated post procedure using invasive pressure monitoring or TTE. Percutaneous femoral arterial access is established with an 8 to 12 Fr catheter, and a wire is placed retrograde into the LV. A balloon catheter, sized based on annular dimensions, is threaded over the wire across the annulus. The balloon is then inflated up to 6 atm for approximately 5 seconds, then deflated. Repeat inflations may be required. This results in fracture of calcium deposits within valve leaflets, separation of commissural fusion, and annular stretching. Rapid ventricular pacing may be used to prevent systolic ejection of the inflated balloon. Percutaneous closure devices may be used at the access site.
MAC may be used in routine cases and skin infiltration with local anesthetic should provide sufficient analgesia. The anesthesiologist should be prepared to manage transient hemodynamic instability related to diminished cardiac output and coronary perfusion during balloon inflation. Transient or permanent conduction disturbances may occur and can be managed with transcutaneous or transvenous pacing. Access site vascular complications may result in bleeding or the need for surgical cutdown. Other complications include new-onset aortic regurgitation, aortic dissection, and annular rupture.
Although outcomes may continue to improve with expanded worldwide experience in transcatheter procedures, large contemporary studies are lacking. Further investigation should evaluate the role of BAV as a bridge to facilitate noncardiac surgery, as this remains controversial.
Based on the surgical Alfieri stitch technique first described in 1991, clinical use of mitral valve transcatheter edge-to-edge repair (TEER) for treatment of mitral regurgitation (MR) has expanded rapidly since the first human case in 2003. , TEER is the most successful of the transcatheter mitral valve procedures and is a proven alternative to surgical mitral valve intervention in carefully selected patients.
There are two devices currently approved for mitral valve TEER worldwide: MitraClip (Abbott Structural Heart) and the PASCAL repair system (Edwards Lifesciences, Irvine, CA) ( Fig. 23.4 ). Both devices use a leaflet repair method that apposes the anterior and posterior leaflets at the site of the regurgitant jet, improving coaptation while creating a double-orifice valve ( Fig. 23.5 ).
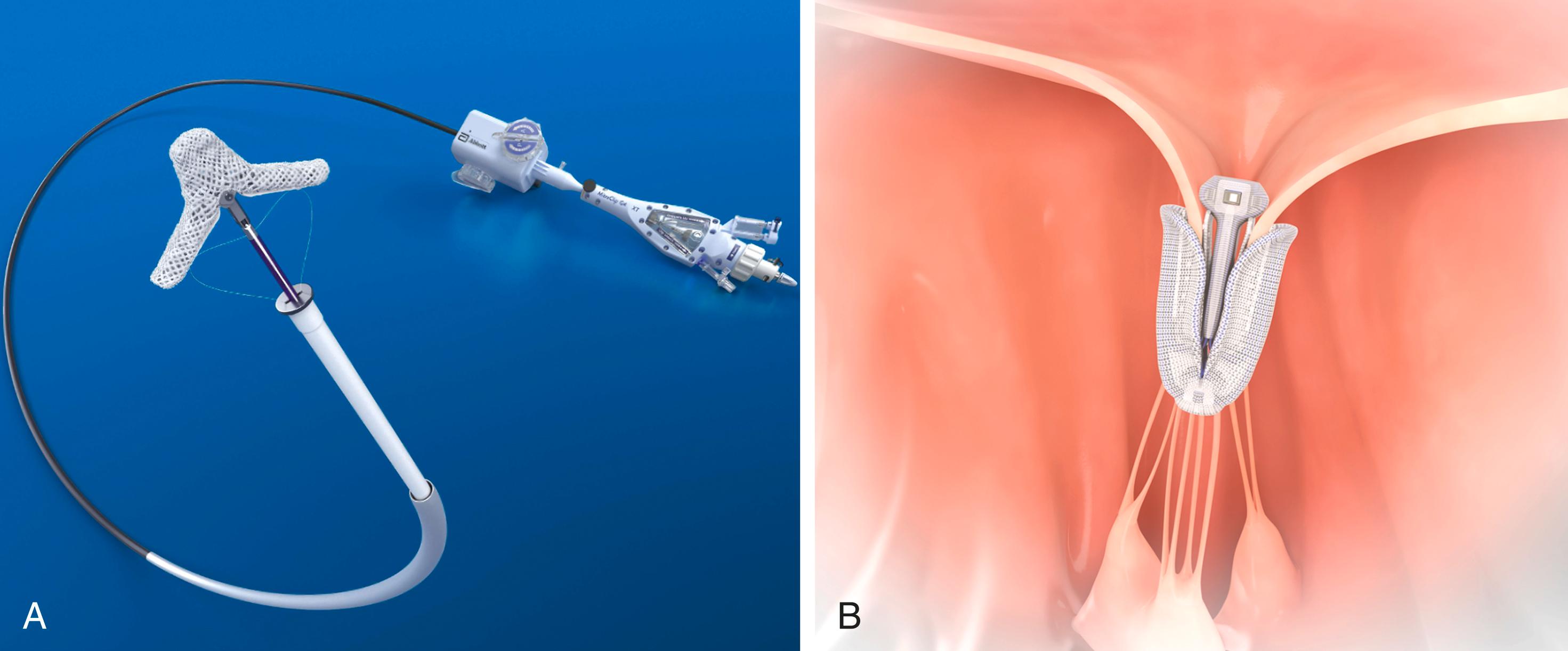
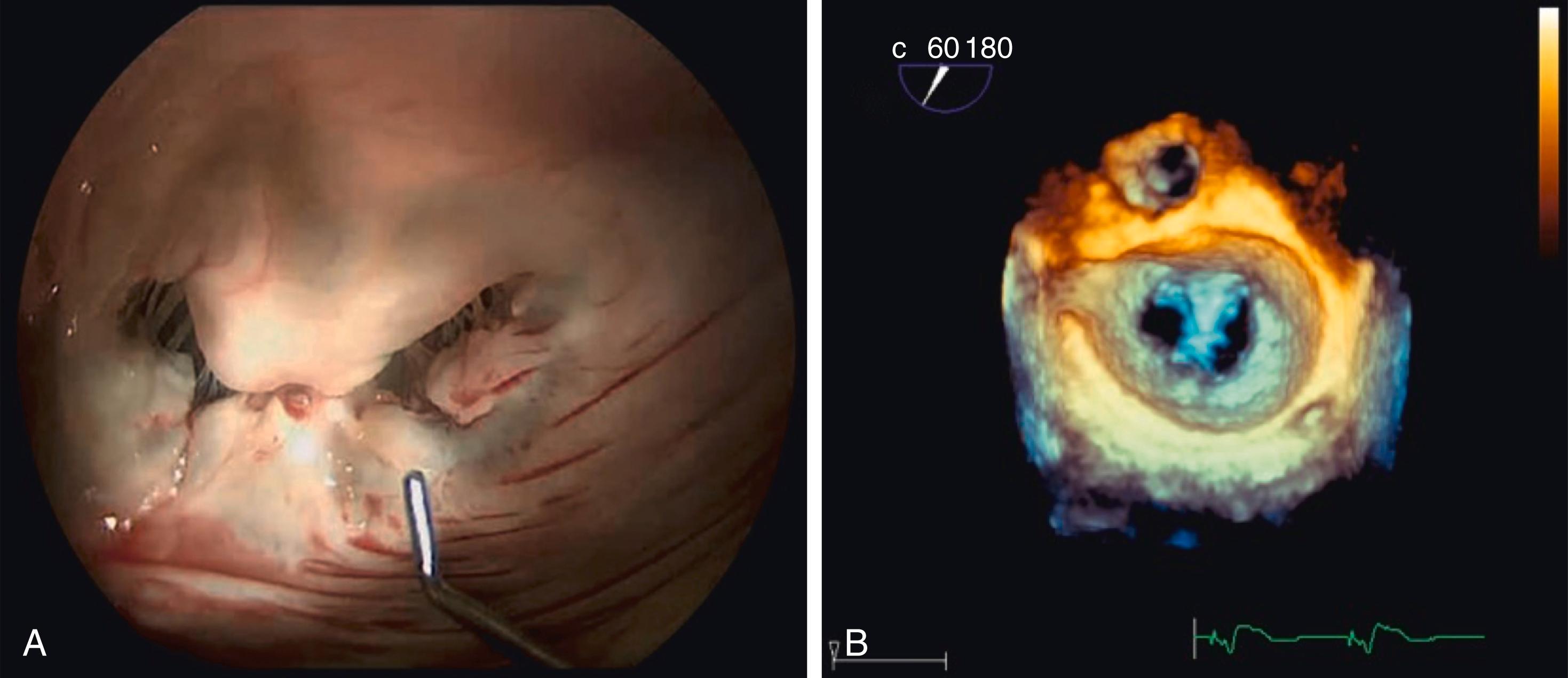
MitraClip was the first TEER device and remains the most widely used. It consists of a 24-Fr steerable guide catheter and a clip delivery system, which includes a 16-Fr steerable sleeve, a 10-Fr delivery catheter, and the implantable clip. Access is transseptal via the femoral vein. The implant is a polypropylene fabric–covered cobalt-chromium device with two grasping arms that bring together the valve leaflets. The current fourth-generation system has been modified to increase coaptation area and comes in four sizes that vary in width and clip arm length, allowing for easier leaflet grasping and independent gripper function. Other modifications allow for improved navigation and clip positioning in the left atrium (LA) by the delivery system and live hemodynamic monitoring.
The PASCAL repair system was designed in part to address several perceived limitations of the MitraClip. Using the same transseptal approach, the PASCAL system consists of a 22-Fr guide sheath, a steerable catheter devised for improved navigation within the LA, and an implant catheter that allows for simultaneous or independent leaflet grasping. The implant itself is affixed to the distal end of the catheter and includes a 10-mm central spacer designed to occupy the regurgitant orifice of the mitral valve. The implant also utilizes two paddles and clasps to grasp the leaflet and is designed to distribute tension across the leaflet edge.
The Endovascular Valve Edge-to-Edge Repair Study (EVEREST) phase 1 clinical trial described the early clinical experience using MitraClip in 27 patients with 3 to 4+ MR. An expansion of this early cohort including 107 patients from 31 sites found 74% of treated patients had a reduction to ≤2+ MR, and 64% were discharged with ≤1+ MR. A 12-month follow-up study found 66% of patients were alive and free from surgery and significant (>2+) MR. The EVEREST II pivotal trial randomized 279 patients with moderate-to-severe MR to either surgery or TEER using the MitraClip system. Inclusion criteria limited mitral valve pathology to a primary regurgitant jet originating from the central portion of the anterior and posterior leaflets. Compared to TEER, surgery was associated with a better 12-month composite endpoint of freedom from death, surgery for mitral valve dysfunction, or greater than moderately severe (>3+) MR (55% vs 73%; P = .007). However, surgery was also associated with a greater rate of major adverse events within 30 days (15% vs 48%; P < .001). At 5 years, surgery continued to have a better composite endpoint, mostly due to improved rates of >3+ MR and less need for surgical valve intervention, which generally presented within the first year.
Two landmark randomized clinical trials evaluating the utility of TEER for functional MR using MitraClip reported conflicting results. In the Multicenter Study of Percutaneous Mitral Valve Repair with the MitraClip Device in Patients with Severe Secondary Mitral Regurgitation (MITRA-FR) study, 307 patients were randomized to receive either TEER and medical therapy or medical therapy alone. At 12 months, where was no difference in the primary outcome of death or hospitalization for heart failure. The Cardiovascular Outcomes Assessment of the MitraClip Percutaneous Therapy for Heart Failure Patients With Functional Mitral Regurgitation (COAPT) trial performed a similar randomization in 614 patients from 78 sites and found TEER resulted in a reduction of hospitalizations for heart failure at 24 months (35.8% per patient-year vs 67.9%; P < .001). Differences in patient selection and the definition of goal-directed medical therapy in the two studies may account for these differences.
The PASCAL repair system has been less studied to date, with the first human use occurring in 2016. The initial compassionate use trial involving 23 patients with ≥3+ MR at high or inoperable surgical risk established feasibility with 96% achieving a reduction to ≤2+ MR. Procedural complications occurred in 9% of patients. A study of the first 309 patients treated in Europe post approval at 10 centers reported similar results, with a reduction to ≤2+ MR in 93.5% after TEER and a 30-day major adverse events rate of 4.1%.
The single-arm Edwards PASCAL Transcatheter Mitral Valve Repair System (CLASP) study enrolled 62 patients with 3 to 4+ degenerative or functional MR at 14 sites. At 30 days, the rate of major adverse events and mortality was 6.5% and 1.6%, respectively. An expanded cohort of 109 patients confirmed previously promising 30-day outcomes, with a reduction to ≤2+ MR in 96% and ≤1+ MR in 80%. At 1 year, Kaplan-Meier survival was 92%, and 88% achieved freedom from heart failure hospitalization. Data at 2 years showed a sustained reduction in MR, reverse remodeling of the LV and improvement in symptoms and quality of life in patients with functional and degenerative MR.
In October 2013, the MitraClip was FDA approved for percutaneous reduction of significant symptomatic MR ≥3+ due to a degenerative etiology in patients deemed to be at prohibitive risk for mitral valve surgery by a heart team, and in whom existing comorbidities would not preclude an expected benefit from reduction in MR. In March 2019, FDA approval was expanded to include functional MR. The PASCAL system gained Conformité Européenne (CE) marking in February 2019 but is not currently FDA approved for commercial use. Society guidelines state TEER is a class IIA recommendation in patients with primary severe MR and high surgical risk or greater, or with chronic severe secondary MR related to LV systolic dysfunction with persistent symptoms.
TEER is contraindicated in patients who cannot tolerate procedural anticoagulation, a post procedural antiplatelet regimen, in patients with evidence of intracardiac, inferior vena cava or femoral venous thrombus, and in patients with rheumatic mitral valve disease or active endocarditis. Relative contraindications include patients with a mitral valve area ≤4 cm 2 , unfavorable mitral valve anatomy including leaflet calcium, and patients with a life expectancy less than 12 months due to a noncardiac cause.
Before proceeding with TEER, a complete history and physical examination must be performed to evaluate suitability for cardiac surgery. Next, an anatomic assessment of the mitral valve is necessary to determine whether TEER is feasible. Preprocedural TTE can determine the severity of MR and identify concomitant valvular disease that may influence the decision perform surgery. Treatment of severe AS with TAVI first should also be considered, as this may contribute to both symptoms and severity of MR.
Preoperative 3D TEE should be performed for a more detailed understanding of valve pathology. Favorable anatomy for degenerative MR consists of an A2/P2 prolapse or flail with a gap <10 mm and width <15 mm ( Table 23.2 ). , For functional MR, there should be normal leaflet thickness and a coaptation depth <11 mm. With either pathology, the mean gradient should be ≤4 mm Hg with a grasping zone >10 mm. Unfavorable valvular features include multiple jets, the presence of clefts near leaflet prolapse, leaflet restriction, previous annuloplasty ring, Barlow’s disease or severe mitral annular calcification. Other unfavorable anatomic findings include the presence of a thickened interatrial septum and hypertrophic cardiomyopathy with systolic anterior motion of the mitral valve. Up to one-third of patients screened are declined due to unfavorable anatomy.
| Favorable Anatomy | Difficult Anatomy | Contraindications | |
|---|---|---|---|
| Location of pathology | Central, segment 2, single jet | Segment 2, 3, multiple jets | Defect in leaflet body/perforation |
| Degenerative MR | A2/P2 prolapse Flail A2/P2 prolapse and flail gap/width <10 and 15 mm, respectively |
Multiple prolapse or flail segments Anterior leaflet prolapse with ruptured chords Cleft near leaflet grasp Barlow’s disease |
|
| Functional MR | Normal leaflet motion, leaflet length > 10 mm Central MR location Single Jet |
Restricted leaflet motion Tethering height or coaptation depth > 11 mm Multiple jets, or not centrally located |
Rheumatic mitral valve |
| Calcification | None | Severe MV annular calcification, <5 mm of leaflet for grasp | Calcification of leaflet at site of grasp |
| MV area/gradient | MV area > 5 cm 2 MG < 3 mm Hg |
MV area < 4 cm 2 MG = 4–5 mm Hg |
MV area < 3 cm 2 MG > 5 mm Hg |
| Grasping zone length | >10 mm | 7–10 mm | <7 mm |
| Septum considerations | Thickened interatrial septum Patent foramen ovale/ASD Previous septal puncture/closure |
LA appendage thrombus | |
| MV and LV anatomic considerations | Normal-sized MV annulus Normal sided or mildly dilated LV |
3D MV annular area > 140 cm 2 Dilated LV > 65 mm Previous MV annuloplasty ring HOCM with SAM |
A 5-Fr femoral sheath is first placed, preferably in the right femoral vein. This sheath is then exchanged for a device-specific dilator, which is positioned into the right atrium (RA). Through this, a needle is then advanced to the atrial septum and used for transseptal puncture under fluoroscopic and TEE guidance. The site of the puncture should be superior and posterior in the fossa ovalis to optimize maneuverability of the delivery system. This puncture should be 4 cm above the level of the anterior leaflet, with more height needed for medial lesions. Hemodynamic tracings together with TEE confirm successful puncture into the LA. The device-specific sheath is then advanced into the LA and heparin is administered to achieve a target activated clotting time of 250 seconds. An exchange-length guidewire is inserted, the device sheath is removed, and the device catheter and delivery system is advanced into the LA and oriented perpendicular to the coaptation line at the desired valve scallops. The implant arms are then opened and the delivery system is advanced across the mitral valve. The catheter is retracted slowly and the leaflets are visualized entering the arms of the implant, which are then closed. TEE is used to confirm leaflet capture, adequate tissue bridge, and to assess the degree of residual MR. The arms can be reopened and repositioned prior to definitive release if the grasp is unsatisfactory. A second implant may be placed after assessment of mitral valve gradient. The delivery system is then retracted, and hemostasis of the femoral vein is achieved.
Anesthetic management for patients undergoing TEER routinely involves GA with an endotracheal tube to facilitate a comprehensive TEE examination. The use of MAC is less common but has been described. It should be recognized that the use of GA may lead to the underestimation of MR. While large hemodynamic changes are not expected during a routine uneventful case, invasive arterial monitoring is usually the standard of care. Patients who are New York Heart Association (NYHA) class 3 and above with heart failure symptomatology will need careful preoperative optimization, as well as the use of inotropes and vasopressors administered either through PA catheters placed in the jugular vein or via femoral access placed by the proceduralist. The anesthesiologist should be prepared for potential complications including intracardiac perforation, cardiac arrhythmia, and air embolism via the delivery catheter. Significant reduction of MR will acutely increase LV afterload. Peripheral venous injury is typically easier to manage in venous structures compared to arterial injury. Fluid restriction is preferred in patients with decompensated heart failure. Patients are typically extubated at the end of the procedure.
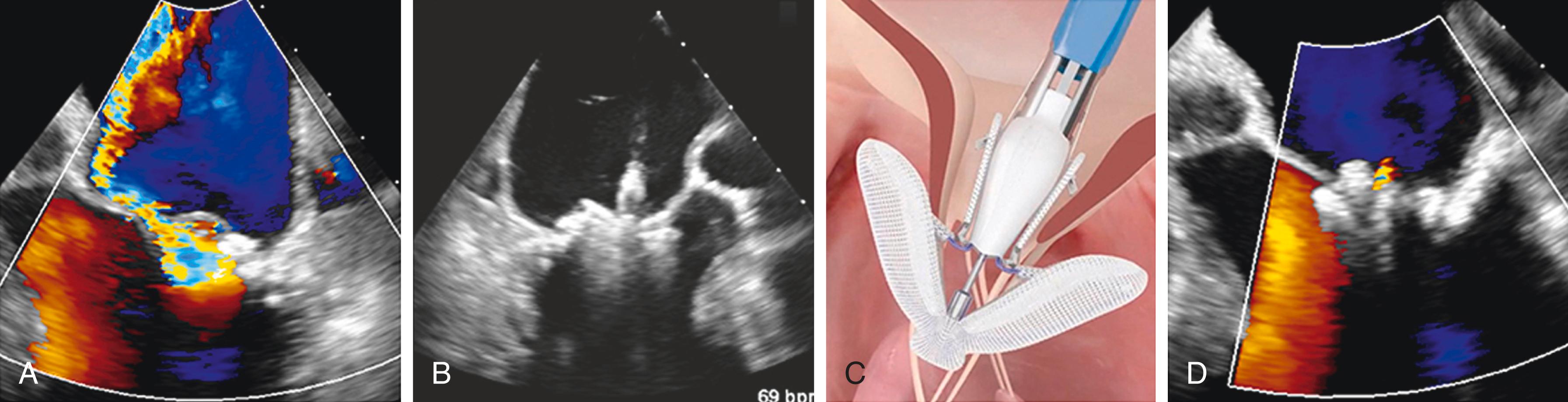
Table 23.3 follows the key aspects of TEE guidance for TEER with MitraClip. Successful procedural guidance begins a comprehensive baseline intraoperative TEE to confirm previous defined anatomic features including the location and number of regurgitant jets. Transseptal puncture is guided using a modified midesophageal aortic valve short-axis view and midesophageal bicaval view, allowing for determination of the anterior-posterior and superior-inferior orientation. Simultaneous biplane imaging can be used. Tenting of the septum should be visualized prior to puncture. Entry of the guide catheter should be visualized. Three-dimensional en face visualization of the mitral valve, known as the surgeon’s view, should be used to position the guide catheter at the location of maximal MR before the LV is entered. The midesophageal mitral commissural and midesophageal long-axis view should be used to confirm proper implant arm orientation. A simultaneous biplane image of these views can be used to confirm grasp of the leaflets. Assessment of mitral valve gradient and residual MR should then occur. Fig. 23.6 shows an illustrative case of severe secondary MR successfully treated with implantation of a PASCAL implant.
| Procedural Step | Imaging Objectives | Typical TEE Views | Intraprocedural Complications | Anesthesia Pearls |
|---|---|---|---|---|
|
Guide the TSP through the midposterior IAS approximately 4–4.5 cm above the level of the annulus in systole | ME AV SAX (anterior-posterior orientation), ME bicaval (superior-inferior orientation), ME 4C (height above systolic MV leaflet position) | Aortic or atrial injury, pericardial effusion/tamponade, transient ST-segment elevation | Heparinization with an ACT goal > 250 s |
|
Continuous visualization of the tip of the SGC and CDS as the clip exits the SGC in the LA to prevent injury to nearby structures | 2D and 3D ME views, typically ME bicaval, ME 4C, or ME 2C | Vascular injury from 24 Fr SGC, air embolism, injury to nearby structures | |
|
Guide the positioning of the clip to be above the grasping plane | ME mitral commissural and ME LAX with color Doppler (simultaneous biplane imaging) | Injury to structures with clip movement, for example, chordae | |
| Clip orientation so that the arms are perpendicular to the line of coaptation | ||||
| 3D surgical view of the MV | ||||
|
Ensure that the clip maintains the appropriate trajectory and orientation as it is advanced into the LV | Simultaneous biplane imaging of ME mitral commissural (medial-lateral trajectory, clip arms should not be visible) and ME LAX (anterior-posterior trajectory, clip arms should be visible) | Clip entanglement and/or injury to subvalvular apparatus; worsened MR not amenable to clip repair | Apnea, administration of adenosine, rapid pacing can assist with leaflet grasping |
| Reconfirm clip orientation and position beneath the regurgitant orifice | 3D surgical view of MV with imaging gain decreased until clip arms are seen; simultaneous biplane imaging of ME mitral commissural and ME LAX with color Doppler | |||
| Visualize grasping of both leaflets and confirm adequate leaflet capture | ME LAX–grasping view (or biplane) to image length of leaflet within clip arms and appropriate restriction of leaflet motion | |||
|
Evaluate severity of residual MR qualitatively | ME views with color Doppler to assess VC, 3D planimetry of the VCA | MS, possible clip detachment, decreased ejection fraction | Lighten anesthesia and simulate awake vital signs for accurate determination of the relevant transmitral PG and amount of residual MR |
| Assess new double-orifice MV, new MVA, and tissue bridge Quantitatively |
||||
| 3D surgical view of the MV-pulsed-wave Doppler of pulmonary vein flow continuous-wave Doppler to assess peak and mean transmitral pressure gradients, 3D planimetry of both orifice areas | ||||
|
Image CDS and SGC to ensure safety | ME 4C, ME 2C, or ME mitral commissural | Iatrogenic residual ASD with the potential to cause hypoxemia, gastrointestinal bleeding | Reversal of heparin with protamine |
| ME bicaval or 4C | ||||
| Evaluate residual ASD to assess if needs percutaneous closure |
TEER patients should be monitored in a telemetry unit for immediate postoperative complications relating to vascular injury, arrhythmia, and effusion. Patients are at high risk for postoperative delirium due to advanced age. However, compared to TAVI, TEER postoperative recovery may be more straightforward owing fewer conduction disturbances, and the use of venous access. Postoperative ICU admission is rare but is seen in heart failure patients and those with associated comorbidities that preclude rapid extubation. Research using 2016 data from a national US database found 61% were discharged in 2 days or less. The readmission rate in this cohort was 10.1%, compared to 13.5% overall. Fifty-six percent of readmissions were of a cardiovascular etiology. Same-day discharge has been described but is uncommon. There are no formal imaging recommendations; however, TTE may be performed at 30 days or sooner if a complication is suspected.
Long-term clinical outcomes in TEER remain unknown. Further randomized trials comparing TEER to open surgical repair for both degenerative and functional MR are required before expansion of use into a lower-risk population. Due to the conflicting results of studies using TEER in functional MR, further research is necessary to guide optimal patient selection. , The CLASP investigators are currently enrolling patients in CLASP IID and IIF pivotal trials, which will compare PASCAL to MitraClip in patients with degenerative and functional MR. In the future, TEER will also need head-to-head comparison to other transcatheter therapies.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here