Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Spinal tumors occur predominantly in young or middle-aged adults and are rare in children. The most common presentation is vague pain, gait disturbance, change in spinal curvature, motor weakness, and bowel and bladder dysfunction. In this chapter, general concepts regarding spinal neoplasms are discussed, followed by anatomic categories of lesions. Categories include neoplasms located in the intramedullary, intradural-extramedullary, or extradural compartments.
The imaging features of spinal neoplasms are often nonspecific and findings overlap. Familiarity with age at diagnosis, key imaging findings, and associations can narrow the differential diagnosis, as summarized in Table 45.1 .
| Location | Neoplasm | Key Imaging Features | Other Comments |
|---|---|---|---|
| IM | Astrocytoma | Cervical most common Tend to be eccentric in cord ± syrinx Cystic with enhancing nodule |
10 years of age Treatment: surgical debulking ± chemotherapy and radiation |
| Ependymoma | Tend to be central in cord Cervical most common Hemosiderin cap on T2 sequences Drop metastases |
13–14 years of age If multiple lesions, consider NF2 Treatment: surgery ± radiation and chemotherapy |
|
| Ganglioglioma | Similar imaging to ependymoma | 7 years; associated with NF2 Treatment: surgical resection |
|
| Hemangioblastoma | Cystic with an enhancing mural nodule Flow voids |
Associated with von Hippel–Lindau disease Treatment: surgery |
|
| EM | Leptomeningeal metastasis | Lumbosacral involvement Variable size and number of lesions |
Common with ependymomas, medulloblastomas, higher-grade astrocytomas Treatment: depends on primary lesion |
| Nerve sheath tumor (schwannoma, neurofibroma) | Target sign Expansion of intervertebral foramina Posterior vertebral body scalloping |
Associated with NF1 and NF2 Treatment: conservative; rarely, debulking |
|
| Meningioma | Isointense on noncontrast imaging Homogeneous, vigorous enhancement |
Teenagers; multiple lesions in setting of NF2 Treatment: surgical resection |
|
| ED | Osteoblastoma | Posterior element, solid, expansile, enhancing mass | 10–30 years; 40% occur in spine Treatment: surgical resection |
| Aneurysmal bone cyst | Posterior element, expansile, multicystic, fluid-fluid levels | 10–30 years; 20% occur in spine Treatment: embolization, surgical curettage, and packing |
|
| Osteoid osteoma | Posterior element lucent nidus with marked surrounding enhancement, ± central calcification | 90% <25 years of age; 10% occur in spine Treatment: radiofrequency ablation |
|
| Lymphoma | Vertebral body lesion with little or no loss of height; single > multiple | Spinal involvement with systemic disease; rarely primary lesion Treatment: chemotherapy |
|
| Leukemia | Vertebral body lesion with slight loss of height; multiple > single | Spinal involvement with systemic disease Treatment: chemotherapy |
|
| Langerhans cell histiocytosis | Vertebral body lesion(s) with severe loss of height (vertebra plana) and soft tissue mass; single > multiple | Ranges from single osseous lesion (histiocytosis X) to systemic disease Treatment: usually chemotherapy |
|
| Neuroblastoma | Paraspinous (adrenal is most common) mass encasing vessels with calcifications | 1–5 years of age Treatment: chemotherapy and surgery |
|
| Sacrococcygeal teratoma | Heterogeneous signal intensity; enhancement of solid tumor portions; ± osseous erosions; mature types are mostly cystic | 80% <6 months, found in utero; female > male; 4 types based on intrapelvic or extrapelvic location |
Although osseous erosion can occur as a late finding with pediatric spinal cord tumors, plain radiographs are of limited utility. Computed tomography (CT) is helpful in imaging primary osseous lesions, but is less useful in cord evaluation. Instead, magnetic resonance imaging (MRI) is the study of choice to characterize and define spinal cord lesions. Differentiating intramedullary (within the cord) versus intradural-extramedullary (outside the cord, but inside the dura) versus extradural (outside the thecal sac) location is important to narrow the differential diagnosis. Intramedullary lesions show cord expansion, whereas extramedullary lesions will be separate from the cord in at least one plane. Baseline postoperative MRI is deferred for at least 12 weeks because surgical changes make early postoperative scans difficult to interpret.
Treatment differs depending on the grade and type of neoplasm (see Table 45.1 ). The goal with lower-grade, noninfiltrative spinal neoplasms is complete excision of both the solid tumor and associated syrinx cavities. Although tumors may recur or have drop metastasis with suboptimal resection, they are generally amenable to repeat resection. In subtotal resection, adjuvant radiotherapy is useful in adults, but is controversial in children due to increased morbidity from damage to an immature cord. Infiltrative or higher-grade neoplasms may preclude total surgical resection.
Intraoperative ultrasound is often used to determine tumor margins during surgical planning and resection. Electrophysiologic monitoring techniques can be helpful as the periphery of the tumor is approached to prevent damage to the uninvolved portions of the spinal cord.
Prognosis varies depending on tumor grade with high-grade tumors having the worst prognosis, presence or absence of cerebrospinal fluid (CSF) dissemination, and degree of resection. Preoperative neurologic deficits may persist postoperatively, but most tumors can be removed without causing new morbidity. With most cord neoplasms, contrast enhanced MRI of the entire cerebrospinal axis is warranted.
Pediatric intramedullary tumors occur most commonly in the cervicothoracic cord. Nonspecific symptoms often lead to a delay in diagnosis. Younger children may present with spinal pain (dull and aching) or root pain, rigidity, persistent torticollis, and muscle spasm. Older children may present with gait disturbance and/or progressive scoliosis. Extremity weakness and paresthesias are common. A subset of patients may present with symptoms of increased intracranial pressure (ICP) and hydrocephalus.
Up to 60% of intramedullary tumors in children are astrocytomas and the cervical cord is most commonly involved. Spinal astrocytomas usually occur in children around 10 years of age, with an equal sex predilection. Spinal astrocytomas are rarely seen in neonates and infants and may present with irritability, torticollis, and loss or absence of developmental milestones.
Astrocytomas arise from astrocytes and range from benign (grade I) to malignant (grade IV). Spongioblastomas and pilocytic astrocytomas are at the benign end of the spectrum, and high-grade astrocytomas and glioblastoma multiforme tumors are at the malignant end of the spectrum.
Spinal astrocytomas may be cystic, mixed cystic and solid, solid, or contain necrotic components. Malignant astrocytomas can mimic spinal vascular malformations as a result of hypervascularity with possible intratumoral hemorrhage. Lesion size varies from focal to involvement of the entire spinal cord (holocord), which usually is seen during the first year of life.
Key features of astrocytomas include cord expansion, eccentric location, hypointense to isointense T1 signal, heterogeneous hyperintense T2 signal, and variable enhancement, sometimes of a mural nodule, on postcontrast images ( Figs. 45.1 and 45.2 ). T2 heterogeneity depends on the presence of solid, cystic, and necrotic components. Tumors may be small with associated syringohydromyelia and subarachnoid seeding.
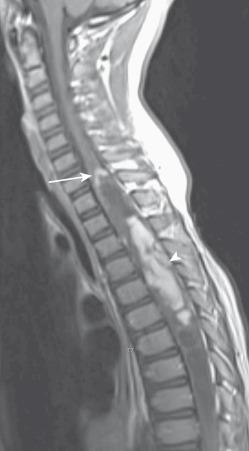
Up to 30% of intramedullary tumors in children are ependymomas and the thoracic cord is most commonly involved. Ependymomas present in an older age group than do astrocytomas with a slight female predilection. Ependymomas originate from ependymal cells within the central cord and frequently span multiple vertebral segments. A variety of histologic subtypes exist, with cellular ependymoma being the most common.
Myxopapillary ependymoma occurs exclusively in the lower cord and filum terminale. When located in the cauda equina, this subtype may be associated with subarachnoid hemorrhage, back pain, lower extremity weakness, numbness, pain, and bowel and bladder incontinence.
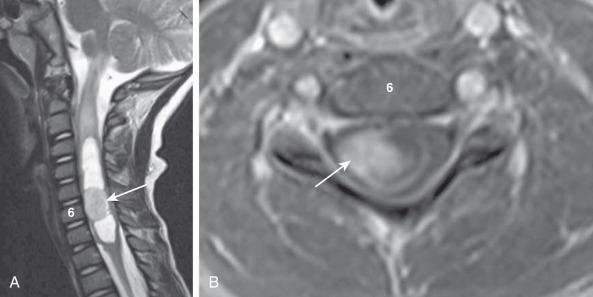
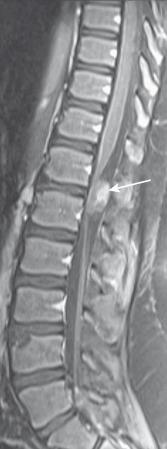
Ependymomas are distinguished by the findings of central cord location, cord expansion, heterogeneous signal intensity on all sequences, and a T2-hypointense hemosiderin cap along the cranial or caudal aspects of the tumor ( Figs. 45.3 and 45.4 ). Contrast-enhanced spinal MRI is useful in the evaluation of drop metastasis, a common feature of ependymomas. Multiple ependymomas should prompt consideration of neurofibromatosis type 2 (NF2).
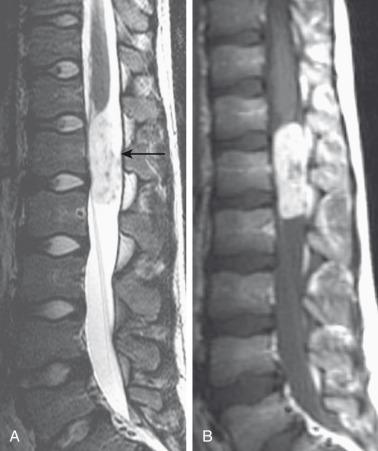
Gangliogliomas contain both neoplastic glial and neuronal components. They present in the first three decades with an average presentation age of 12 years. Gangliogliomas are usually low grade with low malignant potential, but have a propensity for local recurrence.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here