Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
|
|
|
|
|
|
|
|
Neurologic complications during cervical spine surgery remain a source of concern for patients when they are making a decision about a surgical procedure. Patients depend on surgeons to be both honest and informative about all the potential risks involved in surgery. One of the most common questions from patients when they are considering a surgical procedure is “Can I be paralyzed?” This question is often very difficult to answer because the actual incidence varies depending on the procedure and underlying disease; however, the simple answer is always yes. A risk of permanent neurologic injury always exists during surgical procedures, and patients absolutely need to be aware of this risk preoperatively. The purpose of this chapter is to provide an overview of the possible spinal cord injuries (SCIs) and individual nerve injuries, methods to avoid them, and management should they occur.
Iatrogenic SCIs are fortunately relatively rare occurrences in anterior and posterior cervical spine surgery, with a reported incidence less than 1%. An injury may occur during any phase of the perioperative period including intubation, head positioning, decompression, instrumentation, fracture reduction, and deformity correction, or it can be associated with hypotensive episodes resulting in decreased spinal cord perfusion. Additionally, postoperative SCIs can be directly associated with the surgical procedure (e.g., hematoma or seroma).
Besides the routine surgical precautions used to avoid direct trauma and damage to the spinal cord during decompression and instrumentation, several additional strategies can be employed to decrease the incidence of iatrogenic SCI. These include spinal cord monitoring, careful intubation, close monitoring of perioperative blood pressures, and maintaining a high clinical index of suspicion. Although the use of high-dose steroids remains controversial, it is briefly discussed here as well because it is an option, but it is no longer the standard of treatment for SCIs.
Although controversial, spinal cord monitoring during the surgical management of cervical radiculopathy is not routinely necessary. However, it is used more commonly during anterior or posterior procedures for myelopathy. The techniques employed for monitoring vary widely, and because this is the topic of a separate chapter (see Chapter 11 ), it is discussed only briefly here. Based on the anatomy of the spinal cord, direct trauma to the anterior spinal cord may be best monitored using motor-evoked potentials (MEPs) in procedures performed anteriorly or with evidence of ossification of the posterior longitudinal ligament. Conversely, direct trauma posteriorly may lead to a change in the dorsal sensory tracts and somatosensory-evoked potentials (SSEPs). The authors’ facility routinely uses MEPs and SSEPs for all cervical decompressions.
Because of reports of SCI occurring during intubation and neck extension, debate has centered on when to begin intraoperative spinal cord monitoring. The authors’ institution routinely obtains preintubation baseline monitoring. This monitoring should be individualized to the patient’s disorder and the experience of the surgical and anesthetic teams. In these cases, the surgical monitoring team must work closely with the anesthetic team to plan preintubation monitoring. Once preintubation baseline monitoring is obtained, subsequent monitoring after intubation, before positioning, and after positioning can be compared with these baseline values.
In addition to close coordination with the neuromonitoring team, the role of the anesthesia department is particularly crucial during intubation and for perioperative blood pressure control. In patients with any possibility of mechanical instability, with severe stenosis, or with an inability to tolerate neck extension, fiberoptic intubation, rather than direct laryngoscopy, should be considered. Ultimately, this decision should be made in conjunction with the anesthesiologist, but anesthesiologists rely on surgeons to inform them of patients who are at increased risk for neurologic compromise with neck extension. A review of all cases reported to the American Society of Anesthesiologists Closed Claims database found that SCIs caused by intubation were more likely to be associated with stenosis and spondylosis than they were with instability. Furthermore, this study also showed that 16% of the SCIs were associated with hypotensive episodes. In patients with an “at risk” spinal cord, the authors prefer to keep patients’ mean arterial pressure (MAP) higher than 85 mm Hg throughout the procedure and especially during the decompression.
Although every effort should be made to avoid injury to the spinal cord, management after SCI is recognized should be prompt and aggressive ( Fig. 51-1 ). Intraoperatively, SCIs are usually detected by monitoring, and when these injuries occur every member of the surgical team should determine the best course of action. As a rule, the last step in the procedure should be reversed if possible. For example, if the alert occurs during anterior graft placement, then the graft should be removed. Continued communication with the anesthesia team is also paramount to ensure that MAP is being maintained.
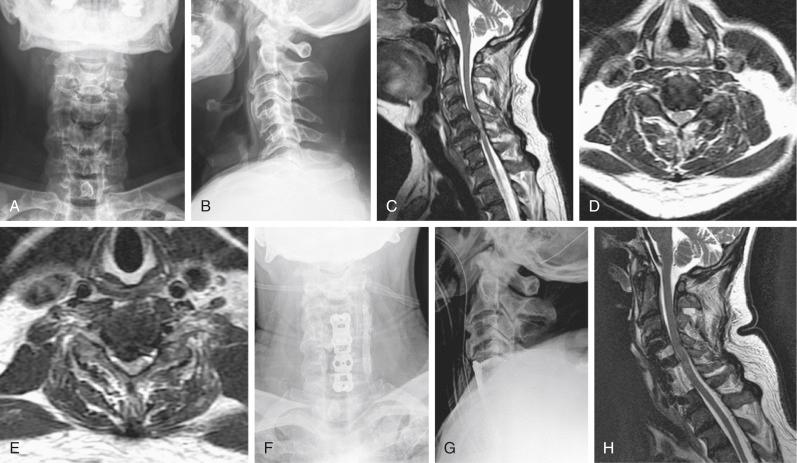
Although the data are controversial, the use of steroids could be considered an option. The authors’ institution generally administers steroids in accordance with the National Acute Spinal Cord Injury Study (NASCIS) II protocol. Additionally, every patient is managed postoperatively in a monitored setting, and MAP requirements are continued for 3 to 5 days. If evidence of spinal cord compression evolves postoperatively, epidural hematoma should be assumed, emergency magnetic resonance imaging (MRI) should be obtained, and emergency surgical intervention should be pursued if indicated ( Fig. 51-2 ).
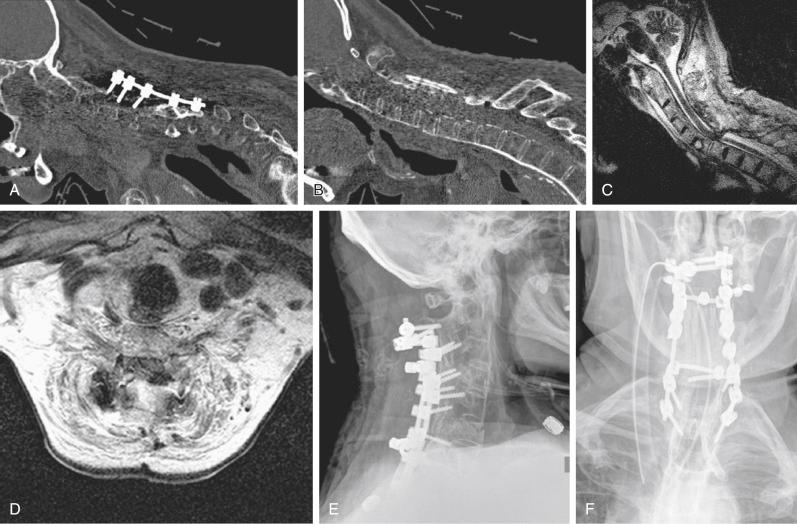
In conclusion, the keys to managing an intraoperative or postoperative SCI are early recognition, communication with the entire surgical team, and prompt and appropriate response. Unfortunately, despite careful planning and appropriate management, neurologic injury remains a known complication of cervical spine surgery, but the response to the event can affect patients’ outcomes.
One of the most common procedures performed by spine surgeons comprises anterior cervical decompression and fusion. Given the significant number of cases performed annually, multiple different complications are reported in the literature. Specifically, anterior cervical spine surgery can be complicated by individual nerve injuries, most occurring with the approach to the spine.
Injury to the hypoglossal nerve is a very rare complication, but this nerve is most at risk with anterior approaches to the upper cervical spine. Additionally, this injury has been a reported risk with transarticular screws penetrating the anterior cortex of C1 and bicortical C1 lateral mass screws. The hypoglossal nerve or the twelfth cranial nerve (CN XII) traverses the hypoglossal canal in the occiput and then courses with the carotid sheath until it emerges into the submandibular region innervating the muscles of the tongue. In a study by Haller and colleagues, the investigators found the hypoglossal nerve to be anatomically nearest the midline at C2 to C3 and not at risk when approaching levels caudal to C3 to C4. When the hypoglossal nerve is damaged, the patient presents with deviation of the tongue toward the side of the injured nerve. Unfortunately, very little can be done once the nerve is injured. The recovery rate of hypoglossal nerve injury is unknown, but of the reported cases, several have returned to function.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here