Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Soft-tissue tumors of the head and neck are very rare, as compared to the far more common epithelial tumors that arise in these anatomic locations. Although benign lesions doubtless account for the great majority of head and neck soft-tissue tumors, it is essentially impossible to give any estimate as to their relative frequency. Malignant soft-tissue tumors of the head and neck comprise approximately 1% of head and neck malignancies and between 5% and 15% of all soft-tissue sarcomas in adults. In children, however, approximately 35% of sarcomas involve the head and neck region, reflecting the preponderance of rhabdomyosarcoma in this age group.
This chapter covers the clinical features, pathologic features, and differential diagnosis of a large number of common and rare soft-tissue tumors of the head and neck. Whenever possible, specific issues related to the diagnosis of these tumors in the head and neck are emphasized. It is not possible to cover all aspects of the pathology of soft-tissue tumors in this relatively brief chapter, and readers are referred to the primary literature and to larger, soft-tissue–specific textbooks.
The staging of soft-tissue sarcomas generally has changed significantly in the eighth edition of the American Joint Commission on Cancer (AJCC) Staging Manual . Whereas previously all soft-tissue sarcomas in any anatomical location were staged similarly. The eighth edition recognizes that the clinical and pathological parameters that best allow for the stratification of patients with soft-tissue sarcomas vary significantly, depending upon anatomical location. Thus, sarcomas of the head and neck now have their own staging system. The reader is referred to pages 499 to 505 for extension discussion of the staging of head and neck sarcomas, and the changes from the previous edition. Briefly, the most important changes to the staging of head and neck sarcomas involve T stage, with T1 tumors being those of <2 cm, T2 tumors being those of >2 to <4 cm, T3 tumors being those of >4 cm, and T4 tumors now defined as those showing invasion of adjoining structures. N and M stages are unchanged, with N1 and M1 tumors representing those with lymph node and distant metastases, respectively.
Another important change to the eighth edition of the AJCC Cancer Staging Manual for soft-tissue sarcomas is the explicit endorsement of the French Federation of Cancer Centers (FNCLCC) grading system for soft-tissue sarcomas. Briefly, this is a three-tier grading system that evaluates a “differentiation score,” a “mitotic count score,” and a “necrosis” score, and sums these scores to provide the final grade. Detailed information on the application of this grading system is available in tabular form in the eighth edition of the Cancer Staging Manual, in the most recent World Health Organization (WHO) Classification of Tumors of Soft Tissue and Bone and in the current edition of Enzinger and Weiss’ Soft Tissue Tumors.
In previous editions of the AJCC Cancer Staging Manual, final stage grouping required synthesis of the tumor grade and tumor node metastasis (TNM) stages. However, stage groupings are not provided for sarcomas of the head and neck in the eighth edition, as insufficient data is currently available for these tumors.
Hypertrophic scars and keloids represent an abnormal fibroblastic reaction to connective tissue injury, with subsequent exuberant scar formation. Both occur most commonly in children and young adults, most often in darkly pigmented patients, particularly African-Americans. Keloids very frequently involve the head and neck, often following piercing, and present as raised, well-circumscribed plaques. Keloids usually extend beyond the original site of injury and may attain very large size. Hypertrophic scars tend to remain confined to the site of original injury and may regress.
Both hypertrophic scars and keloids are composed of cellular bundles of proliferating, cytologically bland fibroblasts and myofibroblasts, arranged in parallel arrays. A sparse chronic inflammatory cell infiltrate is often present, as are small blood vessels, arranged perpendicular to the skin surface. A distinctive feature of keloids is the presence of bundles of refractile, glassy collagen ( Fig. 9.1A and B ). By immunohistochemistry (IHC), both hypertrophic scars and keloids are variably positive for smooth muscle actins, in a “tram-track” pattern, indicative of myofibroblastic differentiation. Hypertrophic scars may also contain scattered S100 protein-positive cells, representing Langerhans cells and proliferating nerve twigs. Desmin is usually negative. Β-catenin expression is not a feature of keloids and hypertrophic scars.
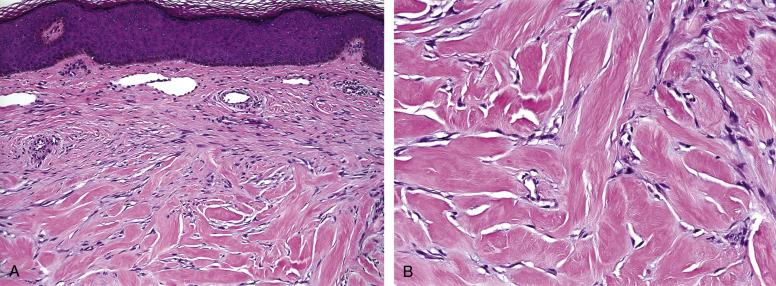
The glassy collagen of keloids is very distinctive and is the key to their correct diagnosis. Although fibromatoses may on occasion show similar collagen, they are much larger, more deeply seated lesions, which only secondarily involve the skin and subcutis. Fibromatoses also tend to show much longer fascicles, as well as a well-developed, thin-walled, dilated vasculature. Benign fibrous histiocytomas lack glassy collagen, show peripheral collagen trapping, and usually show a more prominent storiform growth pattern, with admixed siderophages and foamy macrophages. The distinction of hypertrophic scar from desmoplastic melanoma may at times be difficult; important clues to the latter diagnosis include the presence of very large, hyperchromatic spindled cells arranged in “packets” and the presence of deep, nodular lymphoid aggregates. Desmoplastic melanomas are also strongly positive for S100 protein and SOX10 in the lesional cells, unlike the scattered, bland-appearing S100 protein–positive cells seen in scars.
Juvenile hyaline fibromatosis is an extremely rare disease of uncertain pathogenesis that affects young children, often with an autosomal recessive pattern of inheritance. Juvenile hyaline fibromatosis may be associated with systemic abnormalities, including gingival hypertrophy, contractures, bone lesions, and mental retardation, and it has been suggested that it represents an inherited disorder of collagen metabolism rather than a true neoplasm. Mutations in capillary morphogenesis gene-2 have been reported in both patients with juvenile hyaline fibromatosis and the related disorder, infantile systemic hyalinosis. Some cases have also been associated with mutations in the ANTXR2 gene. Juvenile hyaline fibromatosis commonly involves the head and neck, as well as the upper trunk and extremities, and usually presents before 5 years of age, as multiple, small, slowly growing, exophytic masses. Over time, however, these lesions may coalesce into large, disfiguring, fungating masses, requiring multiple surgical procedures. The lesions of juvenile hyaline fibromatosis have no malignant potential.
Juvenile hyaline fibromatosis consists of abundant, hyalinized, glassy-appearing collagen, arranged in sheets and strands, between which are found small, bland fibroblastic cells ( Fig. 9.2A and B ). The fibroblasts may have an epithelioid appearance, and are often surrounded by a clear halo, separating them from the surrounding matrix.
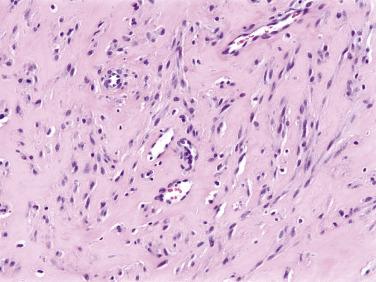
Juvenile hyaline fibromatosis is usually a rather straightforward diagnosis, provided one is aware of this extremely rare tumor. A history of similar-appearing lesions in close relatives should certainly suggest this diagnosis. The differential diagnosis principally involves other lesions that may show glassy-appearing collagen, such as keloids, fibromatoses, and older examples of nodular fasciitis. Juvenile hyaline fibromatosis lacks the cellular bundles of myofibroblasts seen in both keloid and fibromatosis, and does not show the cellular, somewhat “random-appearing” proliferation of myofibroblasts with associated microcystic change, seen at least focally in all examples of nodular fasciitis.
Nuchal fibroma is a relatively rare, benign fibroblastic tumor that typically occurs in the neck and lower back of adults between approximately 20 and 60 years of age. Males are affected far more often than females, and a significant percentage of patients with nuchal fibroma also have diabetes mellitus, although it is unclear whether this represents a direct association or an epiphenomenon. Identical lesions, often but not always occurring in nonnuchal locations, may be seen in infants and very young children, and show a strong association with Gardner syndrome. Children with nuchal-type fibromas in extranuchal locations may go on to develop desmoid-type fibromatoses, either at the site of the fibroma, or in other locations, and the identification of these fibromas in children may rarely result in the subsequent identification of parents with Gardner syndrome-associated polyposis coli and colorectal adenocarcinomas. Ordinary nuchal fibromas do not appear to have a risk of progression to fibromatoses, and are entirely benign lesions that may be treated with simple excision.
Nuchal fibromas consist of a well-circumscribed but unencapsulated proliferation of disorderly bundles of mature collagen, with interspersed bland fibroblasts ( Fig. 9.3 ). This proliferation may entrap nerves and native collagen bundles. The presence of fascicular growth should raise the possibility of a fibromatosis arising within a Gardner-associated fibroma, and the possibility of Gardner syndrome should be excluded in the patient and his/ her family. Most cases of Gardner-associated fibroma demonstrate nuclear immunoreactivity for β-catenin, presumably as the result of APC mutation or activating β-catenin mutations.
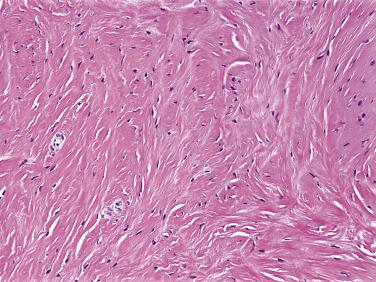
Nuchal fibromas occurring in the upper back should be distinguished from elastofibromas, which typically arise in a subscapular location, and are associated with a history of manual labor. Bundles of degenerating elastic fiber, often forming elastin bodies, are diagnostic of elastofibroma and are not seen in nuchal fibroma. Neurofibromas contain a more prominent population of spindled cells with wavy nuclei and are S100 protein positive and SOX10 positive. Fibromatoses consist of long fascicles of bland fibroblastic cells, arranged about numerous small, often dilated blood vessels.
Immunoglobulin (Ig)G4-related fibrosclerosing lesions are rare, idiopathic, histologically benign appearing, tumor-forming fibroinflammatory lesions that clinically simulate a malignant neoplasm. The spectrum of IgG4-related fibrosclerosing lesions includes Riedel thyroiditis, retroperitoneal fibrosis, sclerosing mediastinal fibrosis, and lesions previously referred to as tumefactive fibroinflammatory lesions . IgG4-related diseases have been reported to involve almost every anatomic area in the head and neck region, including parotid and submandibular glands, maxillary sinus, nasal area, orbit, tongue base, face, nasopharynx, mandible, infratemporal fossa, skull base, and neck. Coexisting IgG4-related fibrosclerotic processes have been also seen in approximately 20% of cases, including retroperitoneal fibrosis, sclerosing cholangitis, autoimmune pancreatitis, sclerosing mediastinal fibrosis and orbital pseudotumor. IgG4-related disease can affect a wide age range, with the majority presenting in the fifth or sixth decades; there is a slight male predominance. IgG4-related fibrosclerosing disease typically presents as an enlarging mass, which frequently is painful. Radiographic studies often show a destructive process, and the initial clinical impression is one of a malignant process. Affected patients are often (but not always) found to have elevated serum IgG4 levels and/or levels of circulating plasmablasts. IgG4-related disease classically responds dramatically to immunosuppressive therapy, especially corticosteroids. However, there may be an incomplete response or regrowth following stopping of the steroid therapy. Surgical excision is frequently successful if the lesion is in a surgically accessible area. Radiation is usually reserved for patients that are unresponsive to steroid therapy and when the tumor’s extent precludes safe surgical excision. Rarely, tamoxifen may also be used. Patients with IgG4-related disease require long follow-up, as recurrence and disease persistence are common. IgG4-related lesions do not metastasize, although patients may develop multifocal disease in other body sites, and are only rarely fatal.
Grossly, these lesions are firm and tannish-white to gray-white in color and vary from being somewhat circumscribed to locally invasive. The classic morphologic appearance of IgG4-related lesions is similar regardless of the involved site: cellular fibrosis often arranged in a storiform pattern, chronic inflammatory cells with lymphocytes, plasma cells, and eosinophils, and obliterative phlebitis ( Fig. 9.4A–C ). It frequently extends into adjacent soft tissue and may encase blood vessels and/or nerves. It is infiltrative but lacks the interlacing bundles, cellularity, atypia and mitotic activity of a sarcoma. While IgG4-related disease invariably demonstrates elevated numbers of IgG4-positive plasma cells ( Fig. 9.4D ), the minimum number required for a diagnosis varies based on location.
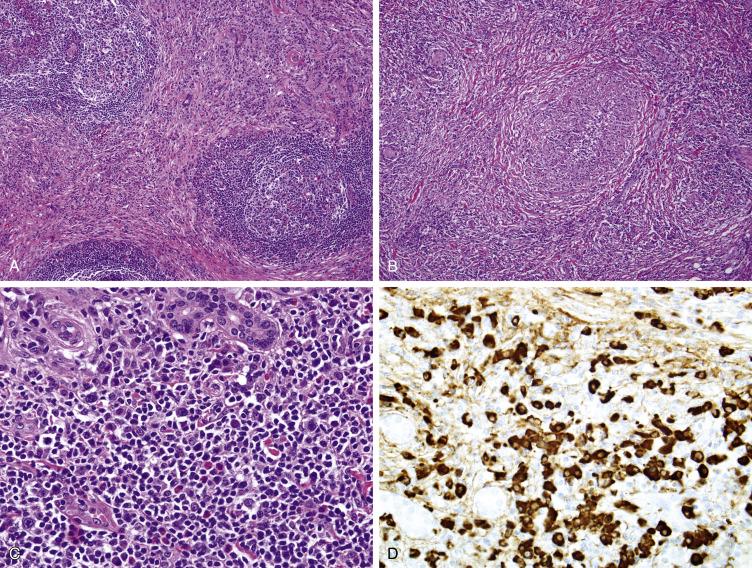
Inflammatory myofibroblastic tumors, especially the hyalinizing variant, should be considered in the differential diagnosis of IgG4-related fibrosclerosing disease. However, unlike IgG4-related disease, they are usually well circumscribed and do not invade adjacent tissues. Nodular fasciitis is more cellular, shows microcystic change, often contains abundant mitoses and usually lacks a prominent inflammatory infiltrate. Fibromatoses consists of long, sweeping, cellular fascicles of fibroblastic cells, with only patchy chronic inflammatory cell infiltrates. Plasma cells are not typically a feature of fibromatoses.
Infantile myofibromatosis is a disease of infants and young children with a predilection for males. Lesion are most often unifocal (solitary myofibroma), but may be multiple, involving visceral organs and bone (myofibromatosis). Identical lesions occur in adults, but are almost always unifocal. The head and neck and trunk are the most common sites of skin and soft-tissue involvement; the tumors typically present as small, soft subcutaneous or soft-tissue nodules. Solitary myofibromas may spontaneously regress if incompletely excised, but may also on occasion recur, and conservative, complete excision is recommended. Patients with multiple lesions involving viscera may have a fatal outcome, depending on the extent of disease.
Myofibromas are usually well circumscribed but unencapsulated. The tumors show distinct zonation, with a fascicular, myoid to hyalinized or even vaguely chondroid-appearing peripheral zone and a more cellular central zone, composed of round, pericyte-like cells, often arrayed about a hemangiopericytoma-like vasculature ( Fig. 9.5A and B ). Necrosis, vascular invasion and mitotic activity may be present, simulating a malignant neoplasm. Some tumors in children consist nearly entirely of the cellular component, probably accounting for most tumors previously diagnosed as so-called infantile hemangiopericytoma. In adults, the pattern of zonation may be reversed, with myoid zones in the center of the lesion, and cellular zones at the periphery. By IHC, both solitary and multicentric myofibromas show a myofibroblastic phenotype, with expression of smooth muscle actins, in the absence of desmin expression. The more primitive-appearing central zones may lack actin expression. Recent evidence has shown that a variant of myofibromas that frequently harbor SRF-RELA fusions demonstrate atypical features in the form of increased cellularity, an elevated mitotic rate, infiltrative borders, and inconspicuous to absent myoid nodules, yet appear to behave in a benign fashion ( Fig. 9.5C ). Gain of function platelet–derived growth factor receptor β (PDGFRB) mutations have recently been shown to be a common feature in tumors from patients with sporadic multicentric myofibromatosis, but are much less common in isolated lesions.
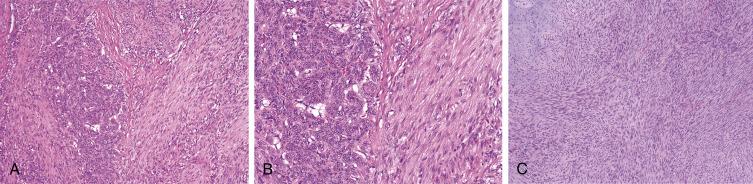
Cases in which the primitive component predominates may be confused with a variety of pediatric round cell sarcomas; identification of small foci of myoid differentiation should allow this important distinction. In cases where the myoid component predominates, and particularly in cellular myofibromas of the type showing SRF-RELA fusions, the differential diagnosis includes benign and malignant smooth muscle tumors, as well as fibromatosis. Again, identification of the diagnostic biphasic pattern of myofibroma is critical in making this distinction. At times, the myoid zones may resemble cartilage, raising such possibilities as a dermal mixed tumor or a chondroma of soft parts. The absence of true epithelial or chondroblastic differentiation and the characteristic zonated pattern of myofibromas should help to establish the correct diagnosis.
Nodular fasciitis is a benign connective tissue tumor that classically presents as a painless, rapidly growing subcutaneous mass. The head and neck are relatively common locations for nodular fasciitis, particularly in children. Cranial fasciitis, a clinicopathologically distinctive fasciitis-like myofibroblastic/fibroblastic proliferation seen in infants, involves the soft tissue of the scalp and may erode into underlying bone. Intravascular nodular fasciitis is also particularly common in the head and neck region. It is important to recognize that the classic clinical history of rapid growth may not be present in all cases, and that some cases of nodular fasciitis may have been present for some time before biopsy. Nodular fasciitis may occur in suprafascial, intrafascial, and subfascial locations. Nodular fasciitis is entirely benign and may in fact spontaneously regress. Rare cases do recur, most often when incompletely excised. For unknown reasons, nodular fasciitis occurring in periauricular locations appears to be particularly prone to local recurrences. Before the diagnosis of “recurrent nodular fasciitis,” however, one should very carefully reevaluate the primary and recurrent lesions for any subtle evidence of malignancy.
The growth pattern of nodular fasciitis depends to a degree on where it occurs. Subfascial and suprafascial examples usually grow as circumscribed but nonencapsulated masses. In contrast, intrafascial examples (so-called fascial variant) are poorly circumscribed and may appear quite infiltrative, as they extend along the fascial planes. Most cases are 2 cm or less in greatest dimension, but occasional cases may be up to 10 cm in dimension.
Histologically, classic lesions are composed of short, randomly arranged fascicles of cytologically bland myofibroblastic cells, arranged in a so-called tissue culture pattern. Unlike true sarcomas, they lack a well-developed vasculature and typically undergo microcystic change, with extravasation of red blood cells ( Fig. 9.6A and B ). Mitotic figures are easily identified. Older lesions may show prominent stromal hyalinization. Occasional cases of nodular fasciitis, more often in children, show marked hypercellularity, and even occasionally small foci of coagulative tumor cell necrosis. Nodular fasciitis may also show quite prominent intravascular growth (so-called intravascular fasciitis). Intravascular fasciitis almost always occurs in children and may have a multinodular growth pattern, owing to involvement of multiple branches of a blood vessel. Osteoclastic giant cells are frequently seen in intravascular fasciitis, and may be an important clue to this diagnosis in an otherwise solid and cellular-appearing tumor ( Fig. 9.6C ). Proliferative fasciitis is a variant of nodular fasciitis distinguished by the presence of large, ganglion-like cells with abundant amphophilic or basophilic cytoplasm ( Fig. 9.6D ). These distinctive cells are superimposed on a background of typical nodular fasciitis, often with central hyalinization. Proliferative fasciitis in children may display alarming cellularity and necrosis, mimicking various sarcomas. Intramuscular tumors with the same histology have been designated proliferative myositis. By IHC, nodular fasciitis shows myofibroblastic differentiation, with peripheral cytoplasmic (“tram-track”) smooth muscle actin expression and variable desmin expression. Nodular fasciitis may express Factor XIIIa, potentially resulting in confusion with benign fibrous histiocytoma/dermatofibroma. Once believed to be a purely reactive lesion, it has been shown that most cases of nodular fasciitis harbor MYH9-USP6 fusions, and therefore may represent a form of “transient neoplasia.” Proliferative fasciitis has not been reported to show MYH9-USP6 fusions. Cranial fasciitis may show strong, aberrant nuclear immunoreactivity for β-catenin protein and recur showing morphological features of desmoid-type fibromatosis ( Fig. 9.6E and F ) ; it is currently unclear whether cranial fasciitis represents an unusual form of pediatric fibromatosis, rather than a lesion related to other forms of nodular fasciitis.
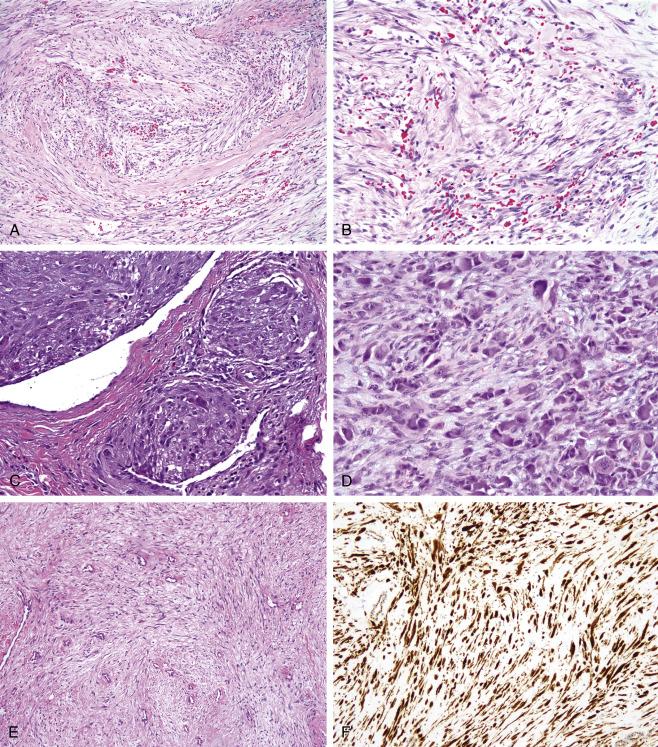
Nodular fasciitis is frequently mistaken for a malignant neoplasm, owing to its rapid growth, high mitotic activity, infiltrative pattern, and occasional hypercellularity. It is critical to always consider the possibility of nodular fasciitis, when dealing with any small and superficially located tumor in the head and neck, particularly in young patients. The hypercellularity, elevated mitotic rates, and necrosis seen in cases of pediatric cellular proliferative fasciitis are particularly treacherous. Awareness of this distinctive form of proliferative fasciitis, immunohistochemical study showing absent keratin immunoreactivity and retained expression of SMARCB1 (INI1), and identification of peripheral areas of more typical proliferative fasciitis, with stromal collagenization, are the keys to the distinction of this lesion from its most important mimic, proximal-type epithelioid sarcoma/malignant extrarenal rhabdoid tumor. Nodular fasciitis with a prominent storiform growth pattern may closely simulate benign fibrous histiocytoma (dermatofibroma) but lacks the peripheral collagen entrapment and hemosiderin-laden and foamy macrophages commonly seen in fibrous histiocytoma. Desmoid-type fibromatosis is more uniformly cellular lesion, which grows in long, sweeping fascicles, contains a uniformly distributed, thin-walled, dilated vasculature, and lacks microcystic change. Myxoid zones within fibromatoses, however, may closely mimic nodular fasciitis, and identification of more typical, nonmyxoid areas is required for definitive diagnosis. Leiomyosarcomas (LMSs) show intersecting fascicles of hyperchromatic spindled cells with distinctly eosinophilic cytoplasm, unlike the bland spindled cells with lightly eosinophilic cytoplasm and tapered nuclei seen in nodular fasciitis. Low-grade myofibroblastic sarcoma, which frequently involves the head and neck, may be extremely difficult to distinguish from nodular fasciitis. A diffusely infiltrative growth pattern and the presence of definite hyperchromatism are the most useful features favoring the diagnosis of low-grade myofibroblastic sarcoma. Identification of USP6 rearrangement, present in nodular fasciitis but not low-grade myofibroblastic sarcoma, may be very useful in this setting. Intravascular forms of nodular fasciitis may closely mimic soft-tissue giant cell tumors but lack the bone shell and rounded, mononuclear cell component of the latter tumor. The ganglion-like cells of proliferative fasciitis and myositis may occasionally be confused with rhabdomyoblasts; lack of cross-striations, cytoplasmic basophilia rather than eosinophilia, and absent immunoreactivity for desmin, MyoD1, and myogenin should readily allow for this distinction. Nodular fasciitis not uncommonly affects the parotid gland, where it is often confused with a myoepithelioma; however, keratin expression is lacking.
Epithelioid fibrous histiocytoma (EFH; epithelioid cell histiocytoma) was previously believed to be simply a variant of cutaneous benign fibrous histiocytoma but recently has become recognized as a distinct entity. EFH typically occurs in young to middle-aged adults (mean age approximately 40 years) and presents as a small, flesh-colored papule on the skin of the extremities. Head and neck involvement is rare. EFH is a benign neoplasm that only rarely recurs following excision.
EFH is a well-circumscribed dermal-based exophytic nodule surrounded by a collar of epidermis at the lesional base ( Fig. 9.7A ). EFH is defined by having >50% polygonal epithelioid cells, with abundant eosinophilic cytoplasm, and nuclei with vesicular chromatin and a prominent nucleolus ( Fig. 9.7B ). EFH may have areas of more conventional-appearing benign fibrous histiocytoma, though an inflammatory component, giant cells, and collagen trapping is less commonly encountered in EFH. The stroma is typically highly vascular. Rarely, EFH shows an unusual pattern of pericellular calcification, reminiscent of that seen in chondroblastoma of bone. By IHC, EFH may show limited expression of Factor XIII ( Fig. 9.7C ) and epithelial membrane antigen (EMA), and is occasionally CD34 and smooth muscle actin positive as well. Desmin, keratin, and S100 protein are negative. Recent studies have shown that EFH consistently harbors ALK rearrangements with associated expression of ALK1 protein ( Fig. 9.7D ), a finding that solidifies EFH as a distinct entity.
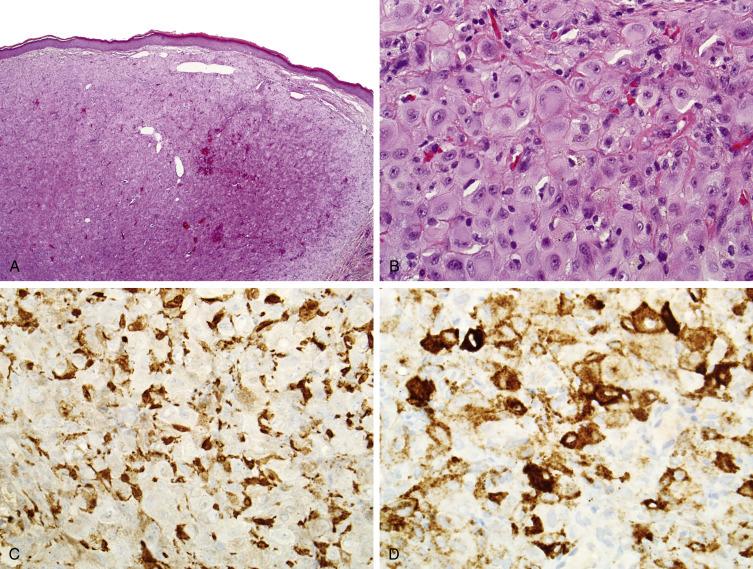
Other epithelioid cutaneous tumors in the morphologic differential diagnosis of EFH include cutaneous syncytial myoepithelioma and epithelioid sarcoma, tumors that also present in young patients and express EMA. Cutaneous syncytial myoepithelioma, however, is also typically positive for S100 protein and negative for ALK1. Epithelioid sarcomas exhibit more cytologic atypia and a much more infiltrative growth pattern than does EFH, are negative for ALK1, and characteristically demonstrate loss of SMARCB1 expression.
Benign fibrous histiocytomas (BFH) are extremely common lesions in the legs, arms, and trunk. However, conventional BFH are extremely unusual in the head and neck, with less than 1% of cases occurring in this location. For unknown reasons, however, up to 30% of the cellular variant of benign fibrous histiocytoma (CFH) may involve the head and neck. CFH present as asymptomatic nodules ranging in size from <1 to 2.5 cm, and may be clinically mistaken for a variety of entities, including epidermoid cyst, pyogenic granuloma, and basal cell carcinoma. In contrast to ordinary dermatofibrosarcoma (DF), which recur in <5% of cases, CFH have been reported to have a high rate of local recurrence (approximately 20%–30%), and should be excised with a cuff of normal tissue. In our experience, the true local recurrence rate of CFH is currently considerably less than has been historically reported, possibly owing to more aggressive surgical treatment of these tumors. Extraordinarily rare bona fide CFH may metastasize to regional lymph nodes or the lungs; such tumors appear to pursue a relatively indolent course, with long survival following resection of metastases.
Histologically, CFH have a number of low-power architectural similarities to ordinary BFH, including overall circumscription, epidermal hyperplasia, and peripheral collagen trapping. A small percentage may be polypoid with a surrounding collarette of epidermis. CFH may show focal extension into the subcutis in up to a third of cases, either in a lacelike pattern, involving the edge of the fat lobules, or a fascicular pattern, following the subcutaneous septae ( Fig. 9.8A and B ). Trapping of individual fat cells, as seen in dermatofibrosarcoma protuberans (DFSP) is not seen, however. CFH arising in areas with relatively little subcutaneous fat, such as the face, occasionally show infiltration of deeper structures, such as skeletal muscle and even parotid gland. CFH are more cellular than ordinary BFH, and often grow in a predominantly fascicular, or occasionally storiform, pattern. In contrast to ordinary BFH, which are usually heterogeneous lesions containing an admixture of spindled to oval tumor cells, siderophages, multinucleated giant cells and inflammatory cells, CFH tend to be composed almost exclusively of spindled tumor cells. Mitotic figures are usually easily identified in CFH, and the mitotic rate may rarely be as high as 10 mitotic figures/10 high-powered fields. Atypical mitotic figures, however, are not seen. Central necrosis may be seen in approximately 10% of otherwise unremarkable CFH.
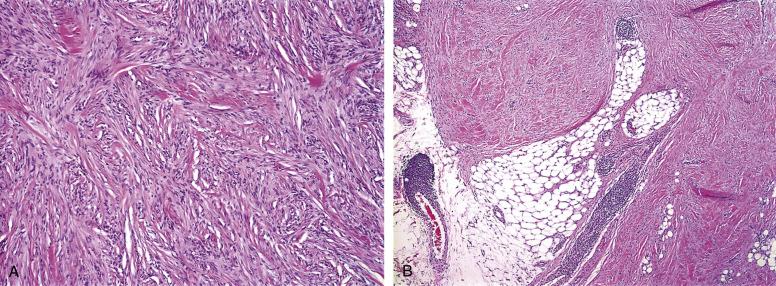
IHC plays only a limited role in the diagnosis of CFH, principally in the exclusion of other entities. Unlike conventional BFH, most CFH are Factor XIIIa negative. CFH may show some smooth muscle actin expression in a “tram-track” pattern, reflective of myofibroblastic differentiation, and sometimes expresses desmin. Most importantly, the lesional cells of CFH are negative for CD34 and S100 protein. It is important to recognize that CFH, as well as ordinary DF, may contain entrapped CD34 and/or S100 protein-positive cells, and may also, on occasion, show a peripheral or deeply located cuff of CD34-positive reactive cells. These reactive cell populations should be rigorously distinguished from the CD34 or S100-negative tumor cells.
The differential diagnosis of CFH includes principally DFSP and LMS, as well as spindled variants of ES. CFH with a pronounced storiform growth pattern can easily be mistaken for DFSP, especially on small biopsies. However, DFSP are typically arranged in a more uniform storiform pattern, have more uniform, slender spindle cells, and show extensive infiltration into the subcutaneous fat, with entrapment of individual adipocytes in a “honeycomb” pattern. This is in contrast to the less extensive involvement of the subcutis that may be seen in CFH. Unlike CFH, DFSP do not show peripheral collagen trapping. In addition, DFSP tend to be much more monomorphic than CFH, lacking secondary cell types, lipidization, or aneurysmal change. IHC for CD34, strongly positive in almost all DFSP, is also valuable in this differential diagnosis. DFSP do not express Factor XIIIa, although occasional cases may show entrapped or peripheral Factor XIIIa-positive cells, analogous to CD34-positive cells in CFH. In particularly difficult cases, demonstration of absent PDGFRB amplification/rearrangement may be helpful in excluding DFSP. CFH with a more principally fascicular growth may be confused with cutaneous (pilar) LMS. LMS differs from CFH by virtue of its more orderly fascicular arrangement, with intersection of fascicles at right angles, and by its more prominent cytoplasmic eosinophilia, with perinuclear vacuoles, and elongated, straight-sided nuclei (“cigar shaped”). LMS also lacks peripheral collagen trapping and secondary elements. By IHC, LMS shows uniform expression of smooth muscle actin, with filling of the entire cell cytoplasm, and is much more often desmin-positive, as compared with CFH. CFH may closely simulate a predominantly spindled epithelioid sarcoma (ES), and vice versa. Exclusively or largely spindled ES are often deceptively bland appearing and may contain hyalinized collagen, reminiscent of CFH. ES do not contain secondary elements, such as foamy macrophages and siderophages, and generally show at least focal hyperchromatism, which may be subtle. IHC for keratins, EMA, and SMARCB1 may be crucial in this important distinction, and one should have a very low threshold for performing these studies, particularly for lesions in children. It should be noted that ES may be Factor XIIIa positive.
Fibromatosis is a nonmetastasizing but locally aggressive tumor, which is currently classified by the WHO as a mesenchymal tumor of intermediate (borderline) malignancy. Approximately 10% of fibromatoses occur in the head and neck region, where they may involve essentially any location. A subset of fibromatoses occur in association with Gardner syndrome, where they are associated with mutations in the adenomatous polyposis coli (APC) gene and subsequent downstream activation of β-catenin; sporadic fibromatoses show mutations in the CTNNB1 gene, with accumulation of β-catenin protein. Fibromatoses are more common in women, and may be associated with prior surgery or trauma, and with hormonal stimulation, as during pregnancy. Clinically, fibromatoses present as slowly growing, deeply seated lesions that may appear fixed to adjacent structures. Although fibromatoses are nonmetastasizing tumors, those located in the head and neck may recur in up to 70% of cases, because of difficulties in achieving complete resection with histologically negative margins in these locations, and a small minority of patients may die from this disease. Adjuvant radiotherapy and hormonal manipulation may be of value in some patients with head and neck fibromatoses.
Fibromatosis colli is a unique variant of fibromatosis that occurs in the sternocleidomastoid muscle of newborns. It typically is discovered within the first 2 weeks of life and involves the right sternocleidomastoid muscle in 75% of cases. Most cases regress within 8 months of diagnosis.
Desmoid-type fibromatoses show identical histologic features in the head and neck as in other soft-tissue locations, and consist of an infiltrative proliferation of cytologically bland, fibroblastic to myofibroblastic-appearing spindled cells, arranged in very long, sweeping fascicles ( Fig. 9.9A and B ). The neoplastic cells are arrayed about numerous small, dilated, nonarborizing blood vessels. A patchy chronic inflammatory cell infiltrate is often present at the periphery of the lesion. A minority of cases may show myxoid change or keloidal collagen; myxoid zones within fibromatoses may closely resemble nodular fasciitis. The tumor nuclei are characteristically hypochromatic, particularly compared to the endothelial cells of nearby intratumoral blood vessels. Fibromatoses may contain a variable number of mitotic figures, but should not contain abnormal mitoses. Necrosis is not seen. Fibromatoses have a myofibroblastic immunophenotype with frequent smooth muscle actin but rare desmin positivity. Most fibromatoses exhibit nuclear immunostaining with β-catenin in keeping with their frequent mutations in the APC/β-catenin pathway ( Fig. 9.9C ). Molecular genetic techniques for the identification of CTNNB1 mutations, such as pyrosequencing, may be more sensitive for the diagnosis of fibromatosis, but are not as yet widely available.
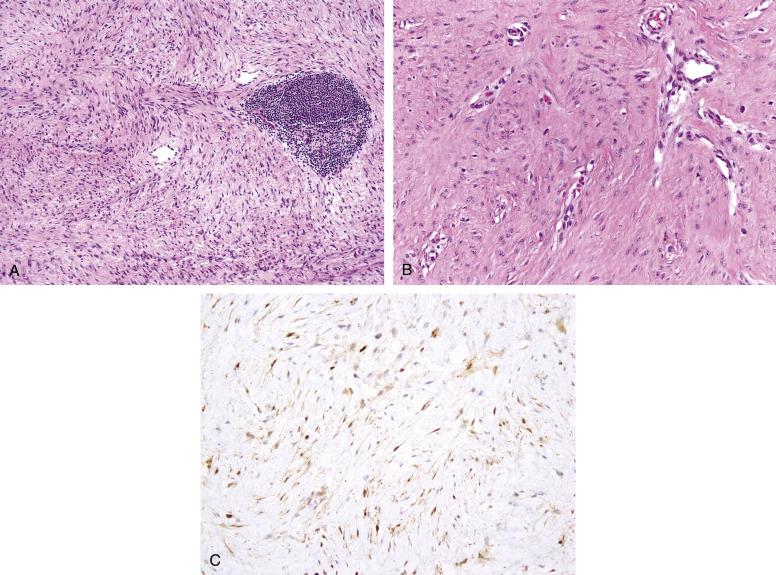
Adult-type fibrosarcomas are much more cellular tumors, which grow in a herringbone pattern, and show considerable nuclear hyperchromatism, in association with higher mitotic activity and often necrosis. It should be emphasized that there is no “cut off” in mitotic activity that distinguishes fibromatoses from fibrosarcomas, and this distinction rests principally on cytoarchitectural grounds. Most “fibrosarcomas” can be shown to represent other specific tumor types, in particular monophasic synovial sarcoma, malignant solitary fibrous tumor and fibrosarcomatous DFSP, and identification of features pointing toward one of these diagnoses should obviously point away from the diagnosis of fibromatosis. Fibromatoses with myxoid change may closely resemble nodular fasciitis, within these myxoid areas. In general, nodular fasciitis is a smaller, often more superficially located process, which lacks long, sweeping fascicles, and shows characteristic microcystic change within hypovascular areas. Fibromatoses with keloidal collagen may be distinguished from keloids by virtue of their deeper location, and by the presence of areas of more typical fibromatosis. Low-grade fibromyxoid sarcomas are extremely unusual in the head and neck, and are characterized by extremely bland, heavily collagenized zones, with abrupt transition into myxoid nodules, displaying curvilinear blood vessels and a whorled arrangement of hyperchromatic spindled cells. Low-grade fibromyxoid sarcomas show MUC4 expression by IHC and FUS gene rearrangements by fluorescent in situ hybridization (FISH), features that may be very helpful in their distinction from fibromatosis.
Although dermatofibrosarcoma protuberans (DFSP) most commonly arises in the trunk or proximal extremities, a significant percentage involves the head and neck, accounting for 6% of head and neck sarcomas in a series from the MD Anderson Cancer Center. DFSP most commonly presents in early to mid-adult life, but may occur in children and even infants. The initial presentation is as a plaque that may subsequently develop into a nodular or multinodular mass. DFSP are highly infiltrative tumors that often grow in a highly asymmetric fashion, with small tentacles of tumor extending for some distance beyond the main mass. For this reason, DFSP are associated with a very high rate of local recurrences, as high as 20%, even with wide excision (3 cm margins). Mohs micrographic surgery appears to offer a better approach with local recurrence rates between 2% to 7%. Distant metastases are extremely uncommon in DFSP without fibrosarcomatous change. Fibrosarcomatous change occurs in between 5% to 15% of DFSP, most often in recurrent tumors, and is associated with a significant risk for distant metastases. The precise metastatic risk associated with fibrosarcomatous change has been controversial, with some studies showing metastases in up to 30% of fibrosarcomatous DFSP, and others showing no metastatic risk in correctly diagnosed and widely excised tumors. A more recent study, from the Mayo Clinic, showed a metastatic risk for widely excised fibrosarcomatous DFSP of approximately 15%.
In its classic form, DFSP consists of a uniformly storiform proliferation of monotonous, slender spindled cells with dark nuclei and lightly staining cytoplasm, with diffuse infiltration of the subcutaneous adipose tissue in the so-called honeycomb pattern ( Fig. 9.10A and B ). DFSP do not contain siderophages and foamy macrophages, helping to distinguish them from CFH. Unusual features seen in small numbers of DFSP include myxoid change, giant cell fibroblastoma-like areas, and myoid nodule formation. Pigmented DFSP (Bednar tumor) are morphologically and clinically identical to ordinary DFSP, with the exception of the presence of melanin-laden dendritic cells.
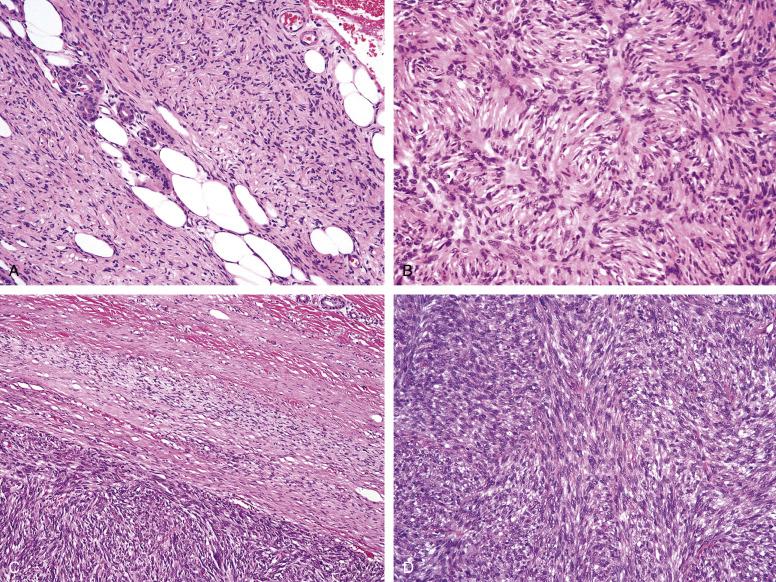
Fibrosarcomatous change in DFSP usually presents as an area of rapid, sudden growth within a preexisting or recurrent DFSP. Microscopically, such lesions show an abrupt transition between areas of typical DFSP and hypercellular, mitotically active zones, which grow in long, fibrosarcoma-like fascicles, rather than storiform arrays ( Fig. 9.10C and D ). As compared with typical DFSP, fibrosarcomatous DFSP display considerable nuclear enlargement and hyperchromatism, although frank pleomorphism is seen in only a small number of cases. Rarely, sarcomatous DFSP may mimic malignant fibrous histiocytoma or myxofibrosarcoma.
By IHC, DFSP are uniformly and intensely positive for CD34, and negative for essentially all other markers. Foci of myoid change may express smooth muscle actin. CD34 expression is often lost in fibrosarcomatous foci.
By cytogenetics, most cases of DFSP contain supernumerary ring chromosomes, which have been shown to contain an occult translocation, t (17;22), resulting in fusion of the platelet derived growth factor β (PDGFB) gene and the collagen type 1 α1 (COL1A1) gene. An identical translocation has been identified in giant cell fibroblastoma and in pigmented DFSP. This fusion gene product may promote autocrine stimulation of tumor growth in DFSP, and there has been clinical success in the treatment of metastatic and unresectable DFSP with imatinib mesylate, a selective tyrosine kinase inhibitor which blocks the platelet-derived growth factor-β receptor, among others. This molecular genetic event may be identified by FISH studies for PDGFB amplification/rearrangement on formalin-fixed, paraffin embedded tissue sections.
The differential diagnosis of DFSP primarily centers on CFH and diffuse neurofibroma. DFSP is a monotonous tumor, composed of more lightly staining, monotonous spindled cells, which lacks siderophages, foamy macrophages and peripheral collagen trapping, and shows permeative involvement of the subcutaneous fat. In contrast, the cells of cellular fibrous histiocytoma are more eosinophilic and plumper, siderophages, foamy macrophages and collagen trapping are frequently present, and involvement of the subcutaneous fat is much more limited, when present at all. By IHC, DFSP shows uniform, strong expression of CD34 in essentially all of the lesional cells, occasionally with a reactive cuff of Factor XIIIa-positive cells, whereas cellular fibrous histiocytoma typically shows a more mixed population of CD34 and Factor XIIIa-positive cells, admixed with S100 protein–positive Langerhans cells and CD68-positive histiocytes. Diffuse neurofibroma consists of bland, round cells embedded in a distinctly fibrillar-appearing background. Although diffuse neurofibroma may infiltrate fat in an identical fashion as DFSP, the presence of Wagner-Meissner bodies, as well as areas resembling ordinary or plexiform neurofibroma help to separate these two entities. By IHC, diffuse neurofibroma is intensely S100 protein positive and SOX10 positive, whereas DFSP is not.
Giant cell fibroblastoma is now felt to represent the juvenile form of DFSP, a concept supported by its histologic and clinical overlap with DFSP, the existence of tumors with composite features of DFSP and giant cell fibroblastoma, their identical immunophenotype, and by cases of giant cell fibroblastoma that have recurred as DFSP and vice versa. Most importantly, giant cell fibroblastoma also harbors the same cytogenetic abnormality as DFSP. As compared with DFSP, giant cell fibroblastoma usually occurs in a much younger patient population, with the majority of patients in the first decade of life. Like DFSP, most giant cell fibroblastomas involve the trunk, thigh or inguinal region, with only rare cases reported in the head and neck. The clinical behavior of giant cell fibroblastoma is essentially the same as that of DFSP, with frequent local recurrences, but only very rare metastases. Fibrosarcomatous change in giant cell fibroblastoma is extraordinarily rare.
Giant cell fibroblastoma is composed of mildly pleomorphic spindled to stellate cells loosely arranged in an abundant myxoid to hyalinized stroma. The cells of giant cell fibroblastoma infiltrate around adnexal structures and though fat in an identical fashion to dermatofibrosarcoma protuberans ( Fig. 9.11A ). A very characteristic feature is the presence of prominent cracking artifact within the stroma, leading to the formation of pseudovascular spaces lined by a discontinuous layer of enlarged, multinucleated tumor giant cells ( Fig. 9.11B ). Like DFSP, giant cell fibroblastomas are uniformly CD34 positive.
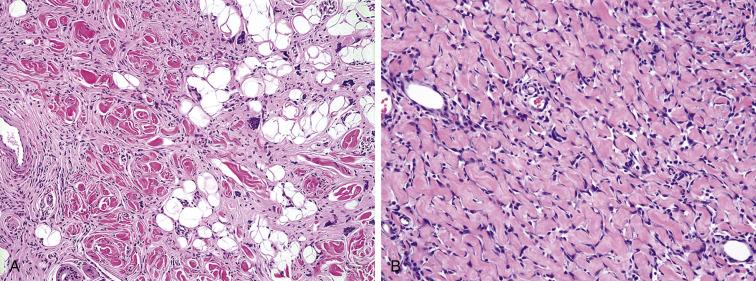
The most important entity in the differential diagnosis of giant cell fibroblastoma is the fibrous hamartoma of infancy. Like giant cell fibroblastoma, fibrous hamartomas occurs in infants, contains infiltrative monomorphic spindled cells embedded in a myxohyaline matrix, and may be CD34 positive. Fibrous hamartomas lack multinucleated giant cells, however, and are triphasic tumors, containing distinctive nodules of primitive-appearing mesenchymal cells and mature fat. Fibrous hamartomas of infancy also lack PDGFBB amplification/rearrangement by FISH. Giant cell fibroblastomas may also, on occasion, be mistaken for a vascular tumor, particularly a lymphangioma, because of the presence of pseudovascular spaces. Spindle cell foci and multinucleated giant cells are, however, not found in lymphangiomas.
Angiomatoid fibrous histiocytoma, previously known as angiomatoid malignant fibrous histiocytoma , was originally regarded as a variant of malignant fibrous histiocytoma (undifferentiated pleomorphic sarcoma), based on the presence of metastatic disease in five of the first 22 reported patients. Subsequent investigation has proven the generally indolent behavior of this tumor, with local recurrences in approximately 12% of cases, and metastases in fewer than 5% of cases. The WHO currently considers angiomatoid fibrous histiocytoma a mesenchymal tumor of intermediate (borderline) malignancy, rather than a fully malignant sarcoma.
Most angiomatoid fibrous histiocytoma occur in children and young adults, where they may be associated with a variety of systemic manifestations including fevers, weight loss, anemia, polyclonal gammopathy, renal disease and rarely a Castleman disease–like lymphadenopathy. Most cases occur in the subcutis or deep dermis of the extremities, although a significant minority may involve the head and neck. Clinically, they may resemble hematoma, hemangioma, or a benign cyst. As noted earlier, angiomatoid fibrous histiocytomas frequently recur locally, but only rarely metastasize, often to regional lymph nodes. Extended survival and even cure are possible with resection of metastases. Tumors located in the head and neck appear to have a somewhat worse prognosis than do tumors of the extremities, presumably related to difficulties in achieving complete resection.
Angiomatoid fibrous histiocytoma is a distinctive tumor characterized by the presence of a dense fibrous capsule and a surrounding lymphocytic infiltrate with germinal center formation. This histologic appearance may mimic metastatic disease involving a lymph node. Long-standing lesions often show very striking fibrosis with associated hemosiderin pigment deposition, amongst which are found small nests and island of neoplastic cells. Pseudovascular, blood-filled spaces are present in most but not all cases, and are lined by flattened tumor cells rather than true endothelium. The neoplastic cells vary from distinctly histiocytoid in appearance to spindled and are arranged in sheets, meningioma-like whorls, and short-fascicles ( Fig. 9.12A and B ). The cells of angiomatoid fibrous histiocytoma are typically bland, but may on rare occasions show striking pleomorphism, which does not appear to impact the behavior of the tumor. The mitotic rate is typically low. Cases without angiomatoid spaces show at least some evidence of hemorrhage, such as intracytoplasmic hemosiderin.
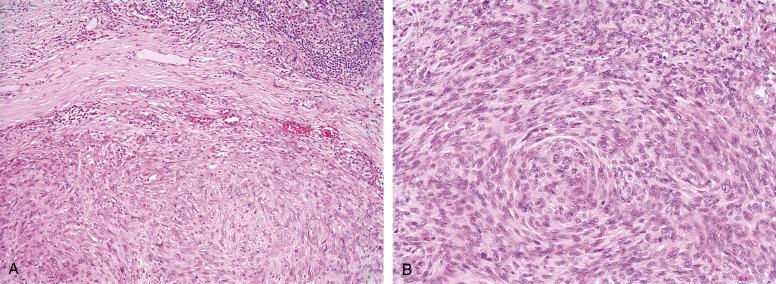
Angiomatoid fibrous histiocytoma has a very unique immunophenotype, with coexpression of desmin, epithelial membrane antigen and CD68 in over 60% of cases. Although they may show muscle actin expression, they are never myogenin or MyoD1 positive. Angiomatoid fibrous histiocytomas do not express endothelial markers. Occasional cases show limited expression of synaptophysin and/or CD99.
Angiomatoid fibrous histiocytoma most often harbor EWSR1 fusions, with CREB1 and ATF1 as the most common partner genes. The variant fusion FUS-ATF1 has also been described.
The differential diagnosis of angiomatoid fibrous histiocytoma includes BFH with aneurysmal change, metastatic tumor in a lymph node, various vascular neoplasms, and rhabdomyosarcoma. Unlike angiomatoid fibrous histiocytomas, aneurysmal BFH show features of ordinary fibrous histiocytoma, with collagen trapping, a storiform growth pattern, and admixed siderophages and foamy macrophages. Angiomatoid fibrous histiocytoma may be distinguished from a lymph node metastasis with relative ease, by noting the absence of structures found in normal lymph nodes, such as the subcapsular sinus or afferent lymphatics. Unlike true vascular neoplasms, angiomatoid fibrous histiocytomas do not form endothelial-lined channels, and lack expression of vascular markers, such as CD31. Perhaps most importantly, desmin-positive angiomatoid fibrous histiocytomas must be rigorously distinguished from rhabdomyosarcomas. This is particularly true in children, where there is a tendency to “rule out” rhabdomyosarcoma with desmin immunostaining on any unusual-appearing lesion. Unlike rhabdomyosarcomas, angiomatoid fibrous histiocytomas are superficially located tumors composed of bland, histiocytoid cells, rather than clearly malignant-appearing round cells with strap cells and eosinophilic giant cells. Angiomatoid fibrous histiocytomas do not express myogenin or MyoD1, which are expressed in nearly all rhabdomyosarcomas.
Plexiform fibrohistiocytic tumor is an unusual fibrohistiocytic tumor of intermediate malignancy that most commonly presents as a dermal or subcutaneous mass of the extremities in children or young adults. Any anatomic site may be involved, although location in the head or neck is very rare. Plexiform fibrohistiocytic tumors recur in between 12% and 40% of cases but have a very low risk of metastatic disease. Lymph node metastases have been reported in two cases, and three patients have suffered pulmonary metastases. There do not appear to be any histologic features that predict which plexiform fibrohistiocytic tumors will metastasize.
Plexiform fibrohistiocytic tumors grow as a poorly circumscribed, plexiform proliferation of multiple small nodules, surrounded by short fascicles of fibroblastic and/or myofibroblastic-appearing spindled cells ( Fig. 9.13A and B ). The individual nodules are composed of an unusual admixture of rounded mononuclear cells and osteoclast-like giant cells. The mononuclear cells typically resemble cytologically bland histiocytes. Rarely, plexiform fibrohistiocytic tumors may contain pleomorphic cells. Mitotic activity is low. Immunohistochemically, the spindled component may express smooth muscle actin in a myofibroblastic pattern, and the rounded cells and osteoclasts are usually CD68 positive. Approximately 40% of cases show a bimorphic appearance, 40% consist primarily of nodules of histiocytic cells without a spindled component, and 20% show principally of the spindled component.
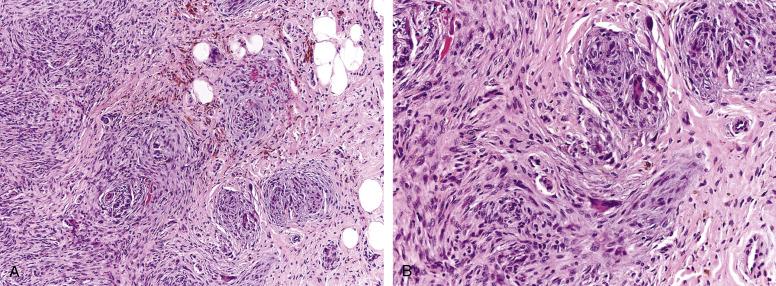
The differential diagnosis of plexiform fibrohistiocytic tumor is fairly narrow, owing to its distinctive growth pattern and appearance. The nodules of rounded cells and osteoclasts seen in plexiform fibrohistiocytic tumor are essentially indistinguishable from those seen in soft-tissue giant cell tumor (see later), and in cases lacking a spindled component, this distinction may be difficult. Features that favor plexiform fibrohistiocytic tumor include younger patient age and the presence of a distinctly plexiform growth pattern with minute nodules. In general, soft-tissue giant cell tumors grow as larger nodules, often with foci of metaplastic bone, aneurysmal bone cyst–like changes and fibrohistiocytic zones. Granulomatous disease may be excluded by the absence of Langhans-type giant cells and central necrosis, and by the presence of a spindled component. Plexiform fibrohistiocytic tumors, composed principally of spindled cells, may closely resemble a fibromatosis. However, fibromatoses are usually deeply seated lesions that lack a plexiform growth pattern and grow in longer, sweeping fascicles. So-called cellular neurothekeoma may also mimic plexiform fibrohistiocytic tumor, by virtue of its distinctly nodular growth, with small nests of generally bland epithelioid cells, separated by thin fibrous septae. Cellular neurothekeomas tend to express some combination of CD10, SMA, and MiTF, markers generally absent in plexiform fibrohistiocytic tumor.
Soft-tissue giant cell tumors are rare, relatively recently described fibrohistiocytic tumors of intermediate (borderline) malignancy. They have historically been thought to represent the soft-tissue counterpart of the giant cell tumor of bone, but appear to lack the point mutations of the H3F3A gene that characterize osseous giant cell tumors. Soft-tissue giant cell tumors usually present as multinodular masses in the skin or subcutis of young to middle aged adults, and may involve any location, including the head and neck. Local recurrences are very common, most likely reflecting the multinodular growth pattern exhibited by most cases, but metastases are extremely uncommon. The behavior of superficially and deeply located soft-tissue giant cell tumors appears to be similar.
Soft-tissue giant cell tumors typically grow as multinodular masses in the dermis and subcutis, frequently with prominent surrounding hemorrhage and fibrosis. A metaplastic woven bone shell is frequently present, similar to that seen surrounding soft-tissue extensions of giant cell tumors of bone. The individual tumor nodules are identical to giant cell tumors of bone, with innumerable osteoclast-like giant cells, bland mononuclear cells, and short fascicles of bland spindled cells ( Fig. 9.14A and B ). Mitotic figures are numerous, but atypical forms are not present. Intravascular extension is commonly present and does not appear to worsen prognosis. Other changes that may be seen include spindled “fibrohistiocytic” proliferations of the type seen in giant cell tumors of bone, aneurysmal bone cyst–like changes, and very rarely cartilage. Necrosis, atypical mitotic figures and severe cytologic atypia are not seen in soft-tissue giant cell tumor, and should prompt consideration of other diagnoses. Although histologically similar, giant cell tumors of soft tissue appear to be molecularly distinct from giant cell tumors of bone, with >90% of bone tumors harboring H3FA mutations, which are consistently absent in the soft-tissue form.
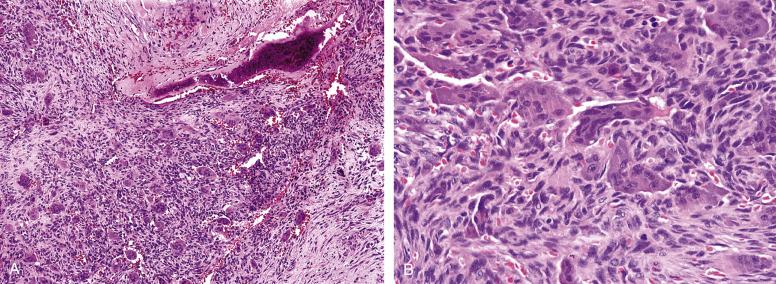
The differential diagnosis of soft-tissue giant cell tumor includes both benign and malignant entities. Numerous osteoclast-like giant cells may be seen in a wide variety of malignant neoplasms, including sarcomas, such as the giant cell-rich variant of undifferentiated pleomorphic sarcoma (malignant giant cell tumor of soft parts), extraskeletal osteosarcoma, and osteoclast-rich LMSs, malignant melanomas, and various carcinomas. Recognition of these entities depends on identification of more typical areas, as well as ancillary IHC studies for markers, such as smooth muscle actin, desmin, cytokeratin and S100 protein. Among benign tumors, soft-tissue giant cell tumors are most often confused with tenosynovial giant cell tumors or giant cell-rich forms of nodular fasciitis (intravascular fasciitis). Unlike soft-tissue giant cell tumors, tenosynovial giant cell tumors are uninodular masses that occur near joint spaces or bursae, and show greater cellular heterogeneity, with small synoviocyte-like cells, siderophages, foamy histiocytes, and lymphocytes. Intravascular forms of nodular fasciitis may contain numerous osteoclasts, closely simulating soft-tissue giant cell tumor. However, careful inspection shows these tumors to be nearly entirely within blood vessels, and to at least contain focal areas of more typical, spindled nodular fasciitis. Metaplastic bone production is not seen in tenosynovial giant cell tumor or intravascular fasciitis.
Myofibroblastic sarcoma is a rare low-grade soft-tissue sarcoma that has a predilection for the head and neck, especially the tongue. Myofibroblastic sarcoma can occur in a wide age range, with a mean of approximately 40 years. Patients with low-grade myofibroblastic sarcoma typically present with a slow-growing, painless mass. Following resection, local recurrences are common but distant metastases and tumor-related deaths are rare.
Low-grade myofibroblastic sarcoma is a highly cellular tumor composed of loosely arranged fascicles of spindled cells showing an infiltrative growth pattern into nearby skeletal muscle, resembling fibromatosis ( Fig. 9.15A ). The fusiform tumor nuclei demonstrate mild pleomorphism and hyperchromatism, with a variably high mitotic rate ( Fig. 9.15B ). The background is often collagenized. By IHC, the tumors are variably positive for myofibroblastic markers smooth muscle actin, desmin, and calponin, but negative for caldesmon and myogenin.
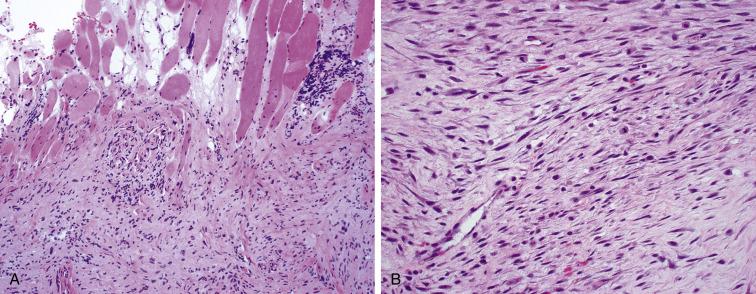
Fibromatosis has a similar infiltrative pattern and myofibroblastic appearance to myofibroblastic sarcoma, but the nuclei of fibromatosis are more hypochromatic and lack atypia. Myofibroma may show a focally infiltrative border, but also lacks nuclear atypia and demonstrates a biphasic pattern of fascicular and more cellular areas with slit-like or staghorn vessels. The cells of inflammatory myofibroblastic tumor may resemble those of low-grade myofibroblastic sarcoma, but are also accompanied by an infiltrate of chronic inflammatory cells. LMS has a better developed architecture of fascicles arranged in right angles, and tends to demonstrate a more pushing, less infiltrative border when compared to myofibroblastic sarcoma. Myofibroblastic sarcomas are not yet known to harbor a specific genetic event, unlike many of the tumors that enter their differential diagnosis (e.g., CTNNB1 mutations in fibromatosis, PDGFRB mutations and SRF-RELA fusions in myofibroma, ALK rearrangements in inflammatory myofibroblastic tumors), and testing for these genetic events may be helpful in selected cases.
Formerly considered one of the most common subtypes of adult soft-tissue sarcoma, fibrosarcoma is now a rather rare diagnosis, owing to advances in the histologic, immunohistochemical and genetic classification of soft-tissue tumors. Fibrosarcomas are now considered to represent no more than 1% to 3% of adult sarcomas. Although it has been estimated that 5% to 10% of fibrosarcomas occur in the head and neck, it is unclear how many of these tumors would still be considered fibrosarcoma today. Many previously described fibrosarcomas of the head and neck likely represent misdiagnosed monophasic synovial sarcomas, malignant peripheral nerve sheath tumors, biphenotypic sinonasal sarcomas, DFSP with fibrosarcomatous change, malignant solitary fibrous tumors, dedifferentiated liposarcomas, and mitotically active fibromatoses. As described, fibrosarcoma of the head and neck occurs most often in middle aged adults, and presents as a deeply seated, nonspecific soft-tissue mass. While previously the soft tissues of the neck and the paranasal sinuses have been reported to be the most common locations for head and neck fibrosarcomas, it is likely that most of these tumors, in fact, represented unrecognized biphenotypic sinonasal sarcomas. A history of prior radiation or thermal injury is present in up to 25% of patients. The great majority of head and neck fibrosarcomas are high-grade (National Cancer Institute [NCI] or FNCLCC grade 2–3) tumors, which require multimodality therapy. The overall 5-year survival for head and neck fibrosarcoma has been reported to be 55% to 70%.
Fibrosarcomas are diffusely infiltrative, highly cellular tumors, consisting of hyperchromatic, mitotically active fibroblastic cells, arranged in long, interlacing fascicles, creating a so-called herringbone pattern ( Fig. 9.16A and B ). In general, cases diagnosed as “fibrosarcoma” should show relatively limited pleomorphism; strikingly pleomorphic tumors are better classified as “undifferentiated pleomorphic sarcomas” (so-called malignant fibrous histiocytoma), although this distinction is subjective and of unclear clinical significance. Fibrosarcomas with myxoid change are usually considered myxofibrosarcomas (see later). Necrosis is often present, connoting a categorically high-grade (grade III) fibrosarcoma. By IHC, fibrosarcomas should lack expression of essentially all markers, except vimentin. Limited smooth muscle actin expression, indicative of myofibroblastic differentiation may be present, and the distinction of fibrosarcomas with limited actin expression from myofibroblastic sarcomas is arbitrary and not known to be of clinical significance.
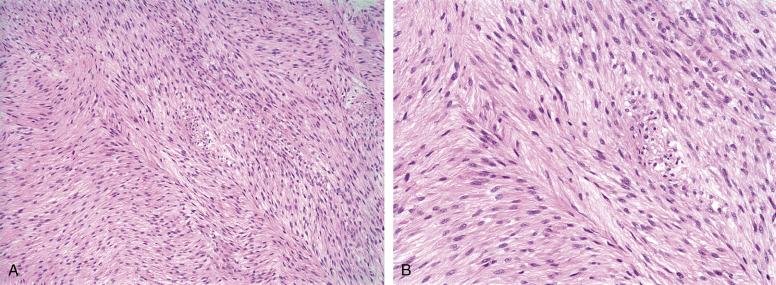
As can be inferred from the aforementioned, fibrosarcoma is a diagnosis of exclusion. The presence of wiry collagen, a hemangiopericytoma-like growth pattern, and alternating hypo- and hypercellular growth should suggest monophasic synovial sarcoma or malignant peripheral nerve sheath tumor. IHC for keratins, S100 protein, SOX10, H3K27me3, a clinical history of neurofibromatosis type 1 (NF1), and genetic study looking for the synovial sarcoma-associated t(X;18) may be essential in distinguishing fibrosarcoma from synovial sarcoma and malignant peripheral nerve sheath tumor (MPNST). Any fibrosarcoma arising in the superficial soft tissues should be carefully examined for evidence of a preexisting DFSP. Malignant solitary fibrous tumors typically show residual foci of typical solitary fibrous tumor, with broad areas of collagenization showing cracking artifact, bland spindled cells arranged in a “patternless pattern,” a hemangiopericytoma-like vasculature, and CD34 and STAT6 immunoreactivity.
Infantile fibrosarcoma is a distinctive tumor of very young children and infants, which occurs either congenitally or within the first year of life in essentially all patients. Approximately 15% of cases occur in the head and neck, where they present as rapidly growing soft-tissues tumors, sometimes with erosion of underlying bone. Infantile fibrosarcoma is locally aggressive and may recur in up to 50% of cases, but only very infrequently metastasizes. Chemotherapy may be dramatically effective in cases with proven ETV6/NTRK3 fusion genes, emphasizing the need for genetic characterization of any suspected infantile fibrosarcoma. Some infantile fibrosarcomas also show fusions involving the NTRK1 gene, and may respond to NTRK targeted therapies.
In general, infantile fibrosarcoma is histologically identical to adult fibrosarcoma, although a hemangiopericytoma-like growth pattern may be more prominent. Intratumoral lymphocytes and foci of extramedullary hematopoiesis may be potential clues to this diagnosis ( Fig. 9.17A and B ). Mitotic figures and necrosis are frequently present. By IHC, infantile fibrosarcomas express vimentin and may occasionally express muscle actins. IHC for NTRK3 does not appear to be a specific marker of infantile fibrosarcoma, as it may also be positive in a wide variety of other tumors in this differential diagnosis.
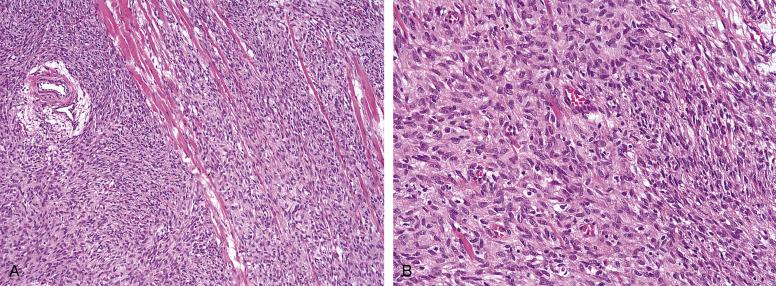
At the genetic level, almost all infantile fibrosarcomas are characterized by the translocation t(12;15) (p13; q26), which results in fusion of the ETV6 gene on chromosome 12p to the NTRK3 gene located on chromosome 15, producing a ETV6/NTRK3 fusion gene and protein. ETV6 rearrangements may be detected by FISH in paraffin sections.
Given its excellent prognosis and chemosensitivity, infantile fibrosarcoma must be distinguished from other pediatric sarcomas, such as embryonal or spindle cell rhabdomyosarcoma, as well as from adult fibrosarcoma. Embryonal rhabdomyosarcomas usually show at least focal rhabdomyoblastic differentiation at the light microscopic level, and express desmin, myogenin, and MyoD1, which are absent in infantile fibrosarcoma. Adult fibrosarcomas occur in much older patients and lack ETV6/NTRK3 fusion genes. Fibromatoses in children lack cytologic atypia and necrosis. Infantile myofibromas often display a very prominent hemangiopericytoma-like growth pattern and should at least focally show better differentiated, myoid-appearing spindled zones.
Tumors now classified as undifferentiated sarcoma were previously termed malignant fibrous histiocytoma , and occasionally pleomorphic fibrosar coma. It is now clear that these lesions are unrelated to histiocytes, and that they represent instead pleomorphic sarcomas lacking evidence of a specific line of differentiation (using currently available technologies), or the end stage in the histologic progression of various pleomorphic sarcomas (e.g., dedifferentiated liposarcoma). It is anticipated that advances in molecular genetics will allow for more precise classification of many of these tumors over the coming years. Myxofibrosarcoma (myxoid “malignant fibrous histiocytoma”) represents a clinicopathologically distinct myxoid sarcoma, lacking evidence of specific differentiation, and typically occurring in the superficial soft tissues of the extremities of older adults.
Although undifferentiated pleomorphic sarcoma (UPS) is generally considered to be the most common adult sarcoma, only 2% to 3% of cases occur in the head and neck. Head and neck UPS occur most often in the soft tissues of the neck, but may occur in essentially any location, including the oral cavity and upper aerodigestive tract. UPS usually occur in older adults, where they present as enlarging, deeply seated, large, soft-tissue masses. Myxofibrosarcoma is often more superficially located and presents as an ill-defined mass that may extend for some distance beyond the clinical impression of its extent, by virtue of diffuse infiltration along preexisting fibrous septae of the subcutis and muscle. The prognosis of UPS is closely related to their grade and size, with larger, high-grade tumors having a distinctly worse prognosis.
Most UPS consist of a storiform and fascicular proliferation of bizarre-appearing, pleomorphic, hyperchromatic spindled to epithelioid cells ( Fig. 9.18A and B ). Multinucleated tumor giant cell, mitotic figures, including atypical forms, and necrosis are frequently present. Some UPS may contain large numbers of osteoclast-like giant cells, and have been referred to as the “ giant cell variant ” (malignant giant cell tumor of soft parts). Some UPS may show a lesser degree of pleomorphism, and the distinction of these tumors from fibrosarcomas may be extremely difficult, if not arbitrary. Stromal hyalinization may be prominent, and the neoplastic cells in areas of abundant hyalinized collagen may appear relatively low grade.
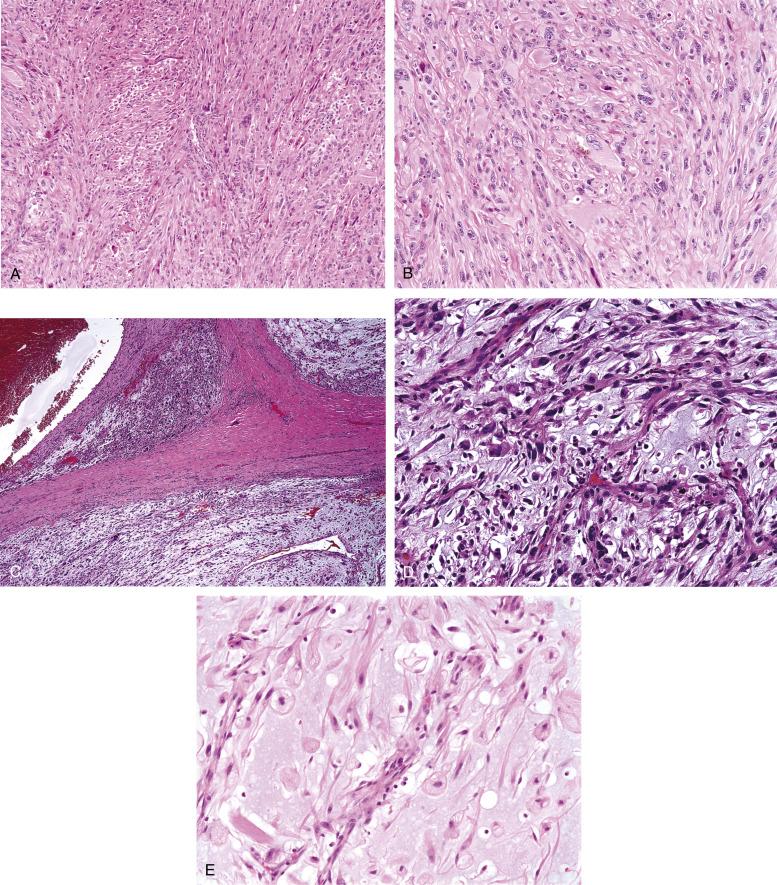
Myxoid change is very frequently encountered in UPS, and may be focal or very extensive. It has been shown that the prognosis of tumors with >50% myxoid areas is significantly better than that of those with less myxoid change, and the diagnosis of “myxoid UPS” or “myxofibrosarcoma” should be restricted to such tumors. Myxofibrosarcomas typically display a multinodular growth pattern, often with diffuse infiltration along preexisting fibrous septae and extension for some distance beyond their grossly apparent borders ( Fig. 9.18C and D ). Within the myxoid areas, characteristic arcuate or curvilinear, thick-walled vessels are seen, with the neoplastic cells often appearing to radiate from these blood vessels in a “Christmas tree” pattern. The cells within the myxoid areas are spindled and stellate and have atypical hyperchromatic nuclei. “Pseudolipoblasts,” representing fibroblastic cells with distended endoplasmic reticulum containing myxoid substance are frequently are present, and should be distinguished from true lipoblasts, which contain optically clear, lipid-filled vacuoles ( Fig. 9.18E ).
UPS is a diagnosis of exclusion. Careful histologic evaluation, looking for evidence of specific differentiation (e.g., smooth muscle, rhabdomyoblasts, osteoid, cartilage, preexisting areas of well-differentiated liposarcoma (WDL) or pleomorphic lipoblasts) is the key to distinguishing other pleomorphic sarcomas from UPS. Similarly, evidence of a preexisting in situ or invasive epithelial or melanocytic lesion should point strongly toward the diagnoses of sarcomatoid carcinoma or melanoma, respectively. This is particularly true in the head and neck, where sarcomatoid carcinomas and melanomas far outnumber true sarcomas. IHC for epithelial, melanocytic, myoid, and hematolymphoid markers also plays a critical role in the distinction of UPS from its various mimics. UPS may show limited smooth muscle actin expression, indicating myofibroblastic differentiation (so-called pleomorphic myofibrosarcoma), which does not appear to be of clinical significance. Low-grade myxofibrosarcoma must be distinguished from myxomas and nodular fasciitis, which lack hyperchromatic cells, pseudolipoblasts and a well-developed, thick-walled, arborizing vasculature. Myxoid liposarcomas do not show the degree of pleomorphism shown by myxofibrosarcoma, and has a characteristic “chicken-wire” vascular pattern, different from the thick-walled vasculature of myxofibrosarcoma. Focal myxoid change may also be seen in essentially any pleomorphic sarcoma. Osteoclast-like giant cells may be seen in carcinomas and melanomas, as well as essentially any other sarcoma, and these must be excluded before diagnosing “giant cell” UPS.
The term atypical fibroxanthoma (AFX) should be reserved for small (<1.5 cm) lesions that are confined to the dermis and at most superficial subcutis, and which occur in sun-damaged skin. This diagnosis should be reserved for lesions which have been completely excised and visualized; incompletely excised lesions, for example in shave biopsy specimens, are best diagnosed as “atypical fibroxanthoma/superficial undifferentiated pleomorphic sarcoma, depending on final depth and size.” AFX occur almost exclusively in sun-damaged skin in older adults, most often in the head and neck. The clinical appearance of AFX is nonspecific, and may mimic squamous cell carcinoma, basal cell carcinoma, or melanoma. Although AFX are histologically identical to much more ominous undifferentiated pleomorphic sarcoma, most have a benign clinical course when they strictly meet the criteria listed earlier, and require only complete excision. Rarely, however, correctly diagnosed cases of AFX, particularly those occurring in the scalp and periauricular region, may metastasize to regional lymph nodes. The prognosis for larger and more deeply invasive lesions, with identical histology, is considerably worse, and these may require more extensive surgery and possibly adjuvant therapy. Such tumors are better classified as pleomorphic dermal sarcomas .
AFX may ulcerate the epidermis or may exhibit a grenz zone between the epidermis and the neoplasm. A polypoid growth pattern, frequently with an epidermal collarette is often present. AFX are identical to more deeply situated UPS, with a storiform to fascicular growth pattern, hyperchromatic, pleomorphic cells, and frequent mitotic activity. Some AFX may consist of relatively monomorphic, but still malignant-appearing spindled cells. Granular cell change is seen in rare cases. Inflammatory cells are commonly present. Necrosis and vascular invasion should not be present and should prompt consideration of other diagnoses.
As with UPS, AFX is a diagnosis of exclusion. All potential AFX should be carefully evaluated for any features that might suggest sarcomatoid squamous cell carcinoma or melanoma, such as in situ carcinoma or pagetoid intraepidermal proliferation of malignant cells. Immunostains for keratins, S100 protein, and melanocytic markers, such as SOX10, HMB45, and Melan A, are also mandatory. It should be noted that scattered S100 protein-positive Langerhans cells are often present within AFX and should not be confused with immunoreactivity in tumor cells. Angiosarcomas, which also typically arise in sun-damaged skin, may on occasion show an almost exclusively spindled growth pattern, and may be extremely difficult to distinguish from AFX. In most cases, careful evaluation of the periphery of the lesion will disclose areas of more typical angiosarcoma, with malignant cells lining dissecting vascular channels. Immunostains for CD31, FLI1, and ERG may be extremely helpful, although it should be remembered that AFX may contain CD31-positive intratumoral macrophages.
Traumatic neuroma is a nonneoplastic lesion that occurs in response to injury, including previous surgery. Occasionally, traumatic neuromas arise after relatively minor injury, such as injections for dental surgery. In the head and neck, the most common locations for traumatic neuroma include the lips, tongue, and gingiva. Typically, traumatic neuromas present as small, firm, often painful nodules. Complete excision is curative in most cases.
Traumatic neuromas are composed of a haphazard arrangement of small nerve fascicules, containing axons, Schwann cells, and perineurial cells, with surrounding fibrosis ( Fig. 9.19 ). Other reactive changes, such as capillary and myofibroblastic proliferation, are often present.
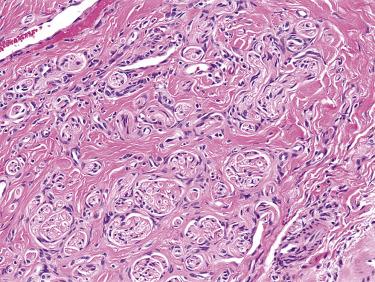
Traumatic neuroma should be distinguished from neurofibroma, which does not grow in distinct nerve twigs, and consists of wavy spindled cells dispersed among randomly dispersed collagen bundles, and from mucosal neuromas, which are submucosal, nodular lesions without surrounding fibrosis, occurring in patients with multiple endocrine neoplasia (MEN) IIb.
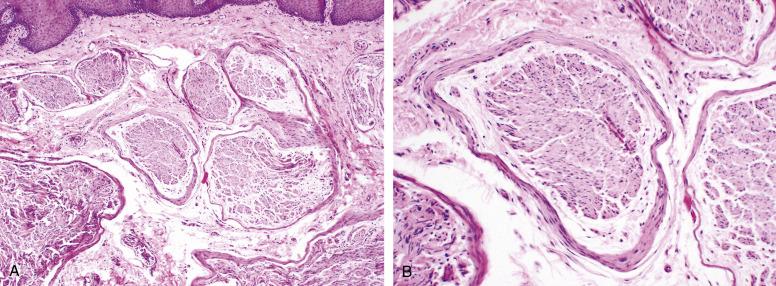
Mucosal neuromas occur on the mucosal surfaces of the oral cavity, eyelids and intestinal tract in patients with MEN IIb syndrome (bilateral pheochromocytoma, C-cell hyperplasia of the thyroid or medullary thyroid carcinoma, and parathyroid hyperplasia). They occur as multiple mucosal nodules, and may be the first manifestations of MEN IIb syndrome. Mucosal neuromas are entirely benign.
Mucosal neuromas consist of irregular submucosal nerve bundles surrounded by prominent perineurium. Myxoid change may be prominent ( Fig. 9.20 ).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here