Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Schwannoma, also known as neurilemmoma and neurinoma, is a benign nerve sheath tumor that occurs primarily in soft tissue but can also occur primarily within bone or secondarily involve bone.
Intraosseous schwannomas are rare, with a reported incidence of less than 1%. They typically affect young adults in the second to fifth decades of life; however, they can occur at any age. Their rarity in bone is thought to be due to the low density of sensory nerves within bone from which schwannomas are belived to arise.
The most common intraosseous sites include the mandible and the sacrum. Schwannomas have also been reported in the humerus, tibia, femur, ribs, scapula, and maxilla, as well as in the small bones of the hands and feet and vertebral bodies. These tumors are usually solitary and asymptomatic, but they can give rise to pain and swelling, especially in those cases that impinge on nerves or arise within the spinal canal.
Schwannomas present as well-defined lucencies with sclerotic borders. Cortical expansion, thinning and perforation may also occur with secondary soft tissue extension. These tumors can thus mimic giant cell tumor of bone or aneurysmal bone cyst radiologically. On magnetic resonance imaging (MRI), they are isointense on T1-weighted images and hyperintense on T2-weighted images. A low-signal intensity rim is sometimes noted and reflects the encapsulated nature of this neoplasm. Intraosseous schwannomas are rarely suspected radiologically, given their rarity and nonspecific findings. A case of subperiosteal schwannoma has been described. Schwannomas arising from the sensory roots of the spinal cord may secondarily involve bone and often have a dumbbell appearance.
Grossly, schwannomas appear as circumscribed white or yellow masses. Larger lesions may have cystic and hemorrhagic areas.
Schwannomas in bone are similar to their soft tissue counterparts. They show the classic Antoni-A and Antoni-B areas; the former is characterized by increased cellularity with prominent or inconspicuous nuclear palisading and the latter by less cellular areas with a myxoid background. A fibrous capsule may not be present in schwannomas of bone; however, scattered thick hyalinized vessels are typically seen. Cystic degeneration, histiocyte infiltration, and hemosiderin deposition are more common in larger tumors. The lesional cells are usually bland. However, significant degenerative-type atypia (i.e., “ancient change”) is occasionally seen, especially in large, long-standing tumors. Melanin pigment deposition, psammomatous calcifications, and adult adipose cell metaplasia are expected in the melanotic variant.
Although any form of schwannoma can occur in bone, the majority of tumors occurring in bone display classic morphologic features. Less common variants include cellular and epithelioid subtypes. Myers et al. reported a case of melanotic schwannoma of the ilium. In this rare variant of schwannoma, the cells express melanin and the tumor shows scattered psammomatous calcifications.
In its classic form, a diagnosis of schwannoma is usually straightforward, especially when adequate tissue is obtained. Histologic mimics in bone include desmoplastic fibroma and fibrous histiocytoma. Although neurofibromas and schwannomas are histologically and nosologically related, the former rarely, if ever, occur in bone. In the cellular form of schwannoma, one must exclude a smooth muscle tumor as well as a monophasic synovial sarcoma. Spindle cell/desmoplastic melanoma may also be a consideration, particularly in the head and neck region; however, this usually presents more aggressively and shows more uniform atypia. Radiologically, the differential diagnosis includes giant cell tumor of bone and aneurysmal bone cyst. In the mandible, odontogenic cysts and tumors should also be considered.
Schwannomas diffusely express S-100 protein. Glial fibrillary acidic protein and CD57 have also been reported but are less sensitive and specific. Electron microscopy shows that schwannomas are composed almost exclusively of Schwann cells, which are characterized by complex processes that emanate from the cell body and are coated with basal lamina. Melanotic schwannomas show patchy positivity for melanocytic markers by immunohistochemistry and, ultrastructurally, feature melanosomes in various stages of development.
The majority of intraosseous schwannomas are sporadic. In the setting of neurofibromatosis, schwannomas have an inactivating germline mutation of the tumor suppressor gene NF2 located on chromosome 22. Psammomatous melanotic schwannomas are associated with Carney complex, an autosomal dominant condition linked to the 17q22 chromosomal region. This condition features myxomas, lentiginous pigmentation, blue nevi, and endocrine hyperactivity.
Schwannomas are benign tumors and are best treated with conservative local excision. Recurrence is rare if adequately excised. Ang and colleagues reported a recurrent schwannoma of the tibia; however, the tumor in this case was large (14 cm) with cortical destruction and soft tissue extension.
Lipomas are benign tumors composed of mature adipocytes. They may arise in intramedullary, intracortical, or parosteal sites. Parosteal lipoma is characterized by an intimate attachment to the periosteum of bone. Although they have been considered by some to represent reactive lesions, the result of hyperlipidemia, the end-stage of an infectious/inflammatory process, or part of the spectrum of bone infarct, the presence of cytogenetic alterations argues to the contrary.
Intraosseous lipoma was first described by Seerig in the German literature in 1836. Parosteal lipoma was suggested by Power in 1888.
Although lipoma is the most common mesenchymal neoplasm in soft tissue, it is decidedly rare in bone. The reported incidence is less than 0.1%; however, this is believed to be an underestimate, since many of these lesions are not biopsied or excised because of their benign appearance on imaging. They can also be misclassified as other benign bone lesions, particularly bone infarcts. Lipomas span a wide age range but are most common in adults. Intraosseous lipomas tend to present in persons who are a decade younger (median age, 37 years) than parosteal lipomas.
Lipomas can affect any bone but are most common in the proximal femur (intertrochanteric and subtrochanteric regions) and calcaneus. Other reported sites include the pelvis, tibia, fibula, humerus, vertebrae, sacrum, skull, mandible, maxilla, and ribs. In long bones, they involve the metaphysis and sometimes the diaphysis. The small bones of the hands and feet are typically spared. Lipomas can be intramedullary, parosteal, and rarely cortical. Parosteal lipomas are most common in the femur and humerus but have also been reported in other sites, including the clavicle, pelvis, and ribs. Lipomas may also involve joints and tendon sheaths. Within the joint they can form a discrete mass or be diffuse, often referred to as lipoma arborescens (see Chapter 21 ). Finally, lipomas have been reported within the spinal canal, where they can lead to medullary compression and neurologic symptoms.
Lipomas tend to be asymptomatic and solitary, and they are usually discovered incidentally. Pain has been described in 50% to 70% of cases in some series; however, it may not be related to the lipoma itself but rather what draws the patient's attention to the lipoma. Pathologic fractures are rare. Motor or sensory deficits from nerve compression caused by parosteal lipomas are common, and the nerve most commonly involved is the posterior interosseous nerve. Jebson and coworkers described a case of intraosseous lipoma with extraosseous extension and associated nerve palsy.
The radiologic characteristics of lipoma depend on its composition and the presence of cystic degeneration and/or osseous metaplasia (see subsequent discussion). In its classic form, it appears as a well-defined lucency, which is often associated with cortical expansion and a sclerotic rim. A calcified nidus within a radiolucent lesion in the neutral triangle of the calcaneus is pathognomonic (target-like or cockade image). Trabeculation may be evident. Computed tomography (CT) and/or MR help confirm the fatty nature of the lesion, which has a low attenuation value on the former modality and high signal intensity on the latter. Cyst formation may also be noted. Parosteal lipoma appears as a juxtacortical radiolucent lipomatous mass with varying degrees of septation associated with surface bone reactive changes such as cortical thickening, cortical erosion, ossific protuberances and, occasionally, bowing of the bone ( Fig. 16-1A ).
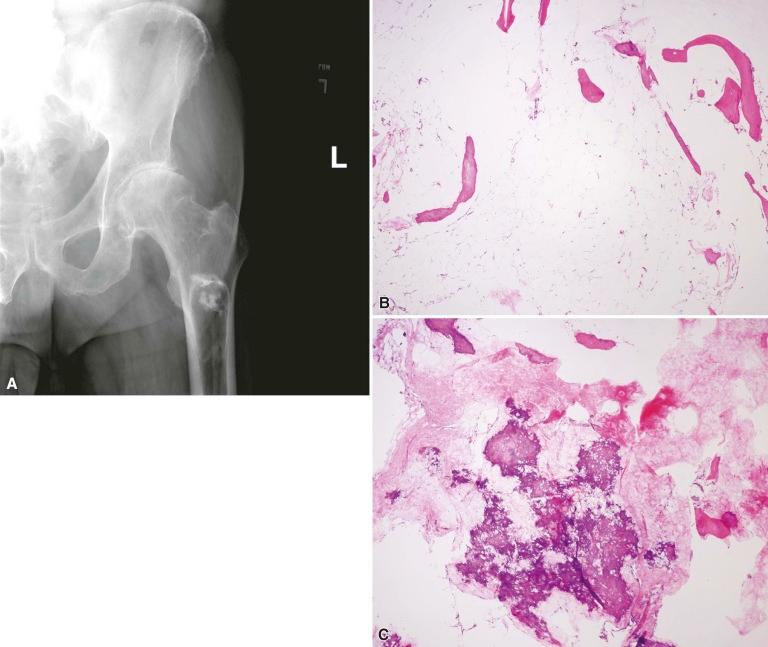
Macroscopically, both intramedullary and parosteal lipomas appear as well-circumscribed yellow masses. Firm, cystic, and calcified areas may be noted. Parosteal lipomas are usually firmly attached to the underlying bone, and they may have portions of the periosteum and a rim of bone attached to them on resection. In addition, they tend to grow larger than intraosseous ones and can measure 4–10 cm as compared to 3–5 cm for the latter.
The histopathologic features of a lipoma are only meaningful in the context of the radiologic imaging studies. Similar to their soft tissue counterparts, bone lipomas are composed of lobules of mature fat. Entrapped bone trabeculae may or may not be present. Varying degrees of fibrosis, myxoid change, fat necrosis, and calcification are expected. Osseous and cartilaginous metaplasia and cyst formation have been described. The involuted lipoma as described by Milgram may not contain any areas of typical lipoma but have cystic spaces, necrotic pink acellular tissue, calcified fat, and reactive woven bone that stains dark purple and lacks a lamellar pattern. This type of reactive ossification related to infarcted fat is quite distinctive (see Fig. 16-1B and C ).
Milgram divided intraosseous lipomas into three different stages based on what he considered their degree of involution. In stage 1, they are composed of viable fat cells; in stage 2, they show focal fat necrosis and calcification, and in stage 3, they have undergone extensive fat necrosis, cyst formation, and new bone formation. The presence of these changes accounts for the variability in the radiographic features of lipomas as described previously.
On conventional radiography, lipomas may be confused with a large number of benign bone lesions, including bone infarct, Brodie abscess, fibrous dysplasia, nonossifying fibroma, giant cell tumor, simple bone cyst, aneurysmal bone cyst, osteoblastoma, plasmacytoma, and benign chondroid tumors. However, CT and/or MR help confirm the fatty nature of the lesion. Histologically, lipomas appear distinct from most of the abovementioned lesions. They can be distinguished from normal marrow fat by the presence of a mass lesion. Bone infarcts can look quite similar to lipomas; however, lipomas show resorption of normal bone within the lesion, which is an important defining characteristic.
No ancillary studies are necessary to make a diagnosis of lipoma other than correlation with radiologic features. Adipocytic nuclei stain positive for S-100 protein. If well-differentiated liposarcoma of bone is a consideration, assessment for MDM2 amplification is a possibility; however, there is no report of MDM2 evaluation of these tumors in the literature.
A (3;12) translocation has been described in parosteal lipomas. Kubo et al. suggested that the presence of this translocation can be used to confirm the benign nature of a lipoma in cases where the radiologic features are worrisome for well-differentiated liposarcoma. This translocation results in an HMGIC/LPP fusion transcript, which is also a common finding in lipomas of soft tissue. Hsieh and colleagues have shown that loss of PTEN leads to lipoma formation along the spines of mice.
If asymptomatic, conservative clinical and radiologic follow-up for a lipoma is sufficient. In symptomatic cases, simple curettage with or without bone grafting is the treatment of choice, and the prognosis is excellent. Indications for surgery include suspicion of malignancy, nerve entrapment, cosmetic deformity, and pain. Radl and coworkers had no recurrence after a mean follow-up period of 32 months for 27 lipomas treated by curettage. Because parosteal lipomas tend to be strongly adherent to the underlying periosteum, they require subperiosteal dissection. Milgram described malignant transformation in four cases of intraosseous lipomas. Whether these cases represent true malignant transformations or a sarcoma occurring in association with a benign lipoma is difficult to tell, especially given that the malignant tumor is often described as a malignant fibrous histiocytoma and a pathologist was not part of the reporting team. In any case, biopsy and/or excision are warranted in cases with worrisome clinical and/or radiologic features.
Infantile hamartoma of the chest wall is a benign, non-neoplastic lesion that presents as an expansile rib lesion. It is characterized by nodules of cartilage and mesenchymal tissue with secondary aneurysmal bone cyst–like changes. The term chondromesenchymal hamartoma has been proposed because it more accurately describes the pathologic features and is less likely to cause confusion with the entity fibrous hamartoma of infancy , a distinct benign soft tissue neoplasm. Other names used for this entity include chest wall hamartoma of infancy , mesenchymoma , mesenchymal hamartoma of chest wall , and vascular hamartoma of infancy . It is generally accepted that this lesion represents a locally disordered overgrowth of normal skeletal elements and is not neoplastic.
In 1962, Le Ber and Stout used the general rubric benign mesenchymoma to describe a group of pediatric mesenchymal tumors composed of various elements that occurred in different areas of the body. The term hamartoma was first used by Mcleod and Dahlin in their report of nine cases in 1979 to reflect their belief that these are not true neoplasms. The common term mesenchymal hamartoma was then used by Odell and Benjamin in 1986.
More than 60 cases of infantile hamartoma of the chest wall have been recorded in the literature. The reported incidence is 0.03%. Most tumors are diagnosed at or shortly after birth; Dounies and colleagues found that 55% of cases were discovered at birth and 97% were documented by 1 year of age. Some cases have been diagnosed in utero by CT scan or ultrasound. The male-to-female ratio is 2 : 1.
Infantile hamartoma of the chest wall is thought to develop antenatally with a variable rate of growth in early childhood. They arise almost exclusively from the posterior or lateral portions of the ribs, although a similar lesion has been described in the nasal passages. They are often multiple and are bilateral in 9% of cases. Most present as an asymptomatic mass; however, they may cause difficulties during childbirth and often result in respiratory distress. Groom and colleagues reported respiratory symptoms in 58% of cases. One infant died of respiratory insufficiency immediately after delivery. Rapid growth of the lesion has also been described.
Radiographs show an expansile mass, often with cartilage-type calcifications, involving one or more ribs. The degree of bony destruction may be suggestive of malignancy. Ultrasound shows a heterogeneous mixed echoic mass with solid and cystic portions, as well as highly echogenic areas with posterior shadowing suggesting calcification or ossification. Hemorrhagic cavities with detectable fluid–fluid levels can be appreciated on CT scan. MR shows alternating low and high signals on T1-weighted and T2-weighted sequences, reflecting both solid and cystic components. Some degree of parenchymal compression and mediastinal shift is often present ( Fig. 16-2A ).
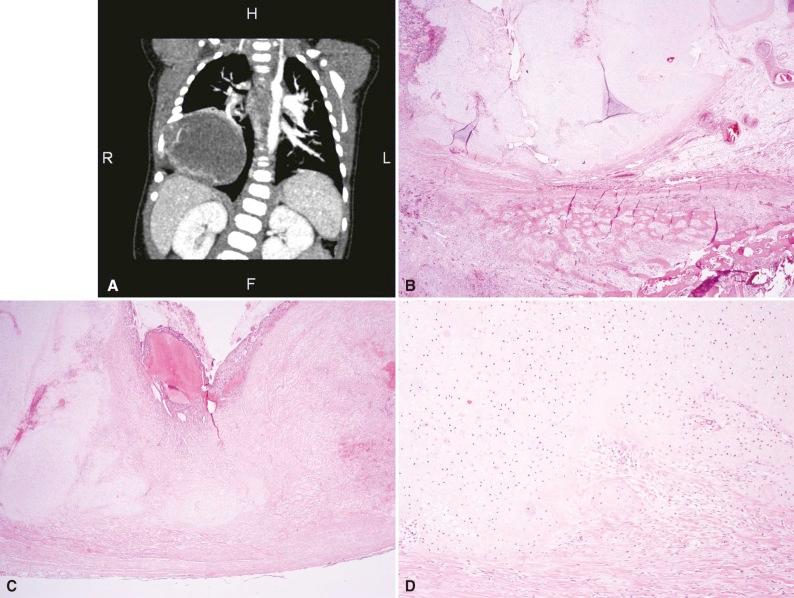
Grossly, this is a well-circumscribed, lobulated mass composed of cystic hemorrhagic areas admixed with meaty red-brown soft tissue and gray to white solid nodules of cartilage. The main mass ranges in size from 2 to 15 cm (average, 6.3 cm).
If biopsy is attempted, it should be done under controlled settings because these lesions may bleed profusely. The tumor is composed of solid and cystic areas; the latter are indistinguishable from an aneurysmal bone cyst and are composed of hemorrhagic spaces lined by spindled stromal cells without significant atypia or abnormal mitotic figures admixed with numerous giant cells. Peripherally, cellular cartilaginous nodules are present that may show enchondral ossification. The cartilage shows variable maturation and may be quite cellular and appear primitive, leading to a misdiagnosis of chondrosarcoma. Areas of chondroblastoma-like proliferation, woven bone, and osteoid have also been described. Histologic maturation of multiple lesions resected sequentially is thought to support the hamartomatous nature of these lesions. Fine needle aspiration cytology has been used with success, especially when correlated with clinical and imaging findings (see Fig. 16-2B to D ).
The presence of a chest wall mass with both solid and cystic components in an infant is suggestive of a chest wall hamartoma. Awareness of this entity with its unique clinical and imaging features is the most important factor in the diagnostic process. Aneurysmal bone cyst and cartilaginous tumors such as osteochondroma, chondromyxoid fibroma, and chondroblastoma are considerations in the differential diagnosis. Other lesions to be considered include fibrous dysplasia, Langerhans cell histiocytosis, and hemangioma. The combination of an aggressive radiologic appearance, rapid growth, and primitive histologic appearance has resulted in misdiagnosis as chondrosarcoma, osteosarcoma, and ostoblastoma. Ewing sarcoma may be considered in the radiologic differential diagnosis, but the histologic features are not those of a small, round, blue cell tumor.
The cartilaginous nodules are positive for S-100 protein. Baretton and coworkers showed a heterogeneous distribution of various types of interstitial collagens in accordance with their presumed hamartomatous nature.
Cytogenetic studies performed on one case showed a normal karyotype. A case of chest wall hamartoma in association with Wiedemann-Beckwith syndrome has been reported, suggesting a possible link to the 11p15.5 gene locus associated with this syndrome.
The natural course is unpredictable, and although some of these lesions regress spontaneously, others progressively enlarge, especially early in the first year of life. It has been noted that symptomatic lesions tend to progress, whereas incidental ones remain stable. Most cases are surgically resected because of symptoms, suspicion of malignancy, or chest wall deformity. If surgical removal is attempted, a conservative approach is recommended to avoid scoliosis, and multistage resection may be necessary; however, recurrence is common if there is no en bloc resection. If observation is elected, close clinical follow-up is necessary to monitor tumor enlargement and the degree of chest wall deformity. Radiofrequency thermoablation has been reported to be successful in one case. There is one report of malignant transformation in the literature; however, it was described by the authors themselves as a fibrous hamartoma . No metastases have been reported to date.
Malignant peripheral nerve sheath tumor encompasses a number of entities and synonyms, including neurofibrosarcoma, malignant schwannoma, malignant neurilemmoma, malignant perineural fibroblastoma, and malignant neurofibroma. However, malignant peripheral nerve sheath tumor (MPNST) is now the most widely accepted term. There is controversy regarding exactly what criteria are necessary for this diagnosis; some authors require evidence that a tumor arises from a nerve or a neurofibroma or, otherwise, occurs in the setting of neurofibromatosis, whereas other authors require only that the tumor show classic histologic features of MPNST. The most specific way to define MPNST would be tumors that arise from, are composed of, or show differentiation toward nerve sheath tissue. In this context, it is important to recall that the “nerve sheath” is a heterogeneous structure composed of Schwann cells, perineurial cells, and fibroblasts.
These tumors are exceedingly rare as primary tumors of bone. Terry et al. found only 18 cases in their review, 15 of which occurred in the mandible or maxilla. The age of presentation is variable (4–76 years), with the majority of cases occurring in young to middle-aged adults. Three patients have been reported with a history of neurofibromatosis. Another well-known association is in the setting of previous irradiation, particularly in tumors affecting the spine.
The majority of the reported cases are in the mandible, especially in pediatric cases, and only a handful of cases have been reported in the long bones. Pain followed by swelling is the most common presenting symptom and/or sign. Spinal tumors are relatively common; they arise from the nerve roots and cause neurologic symptoms.
Radiology typically shows a lytic lesion with extensive cortical destruction. MR often demonstrates bone marrow infiltration and soft tissue extension. Grossly, these tumors are composed of fleshy tan-gray myxoid tissue. Necrosis and hemorrhage have been reported in high-grade tumors ( Fig. 16-3A ).
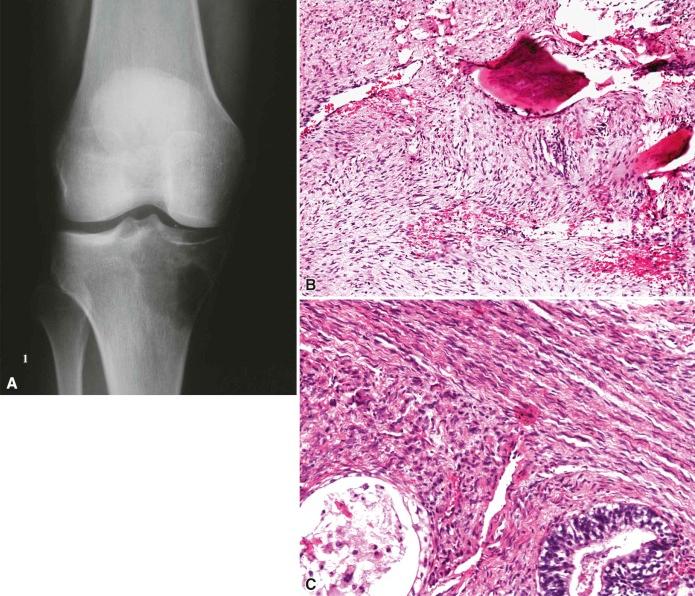
The tumor is composed of interlacing fascicles of spindle or polygonal cells with a clear to eosinophilic cytoplasm and a variably myxoid stroma. Irregular wavy nuclei, focal nuclear palisading, and subendothelial proliferation are features suggestive of the diagnosis. Cellular areas may alternate with myxoid areas, and foci of undifferentiated primitive cells have also been described. Tactoid bodies reminiscent of the tactile organs of skin have been described. Increased cellularity, moderate nuclear pleomorphism, increased mitotic activity, and necrosis are useful in differentiating benign from malignant nerve sheath tumors. Heterotopic mesenchymal elements in the form of osteoid, cartilage, and skeletal muscle (traditionally reported as a Triton tumor) have been reported. Glandular differentiation and epithelioid morphology have also been described (see Fig. 16-3B and C ).
The differential diagnosis includes other spindle cell sarcomas, including synovial sarcoma, fibrosarcoma, and clear cell sarcoma. Metastatic tumors should also be considered, including melanoma and clear cell sarcoma of kidney. MPNST with significant glandular differentiation may mimic metastatic carcinoma.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here