Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Burns of the foot pose unique and difficult problems in initial management, reconstruction, and attainment of long-term functional results. The primary reconstructive goals for this region are unimpeded ambulation and pain-free weight bearing. These objectives can be achieved by strict adherence to established principles of wound management, a clear delineation of the reconstructive requirements of the foot, and a team approach toward achieving these goals.
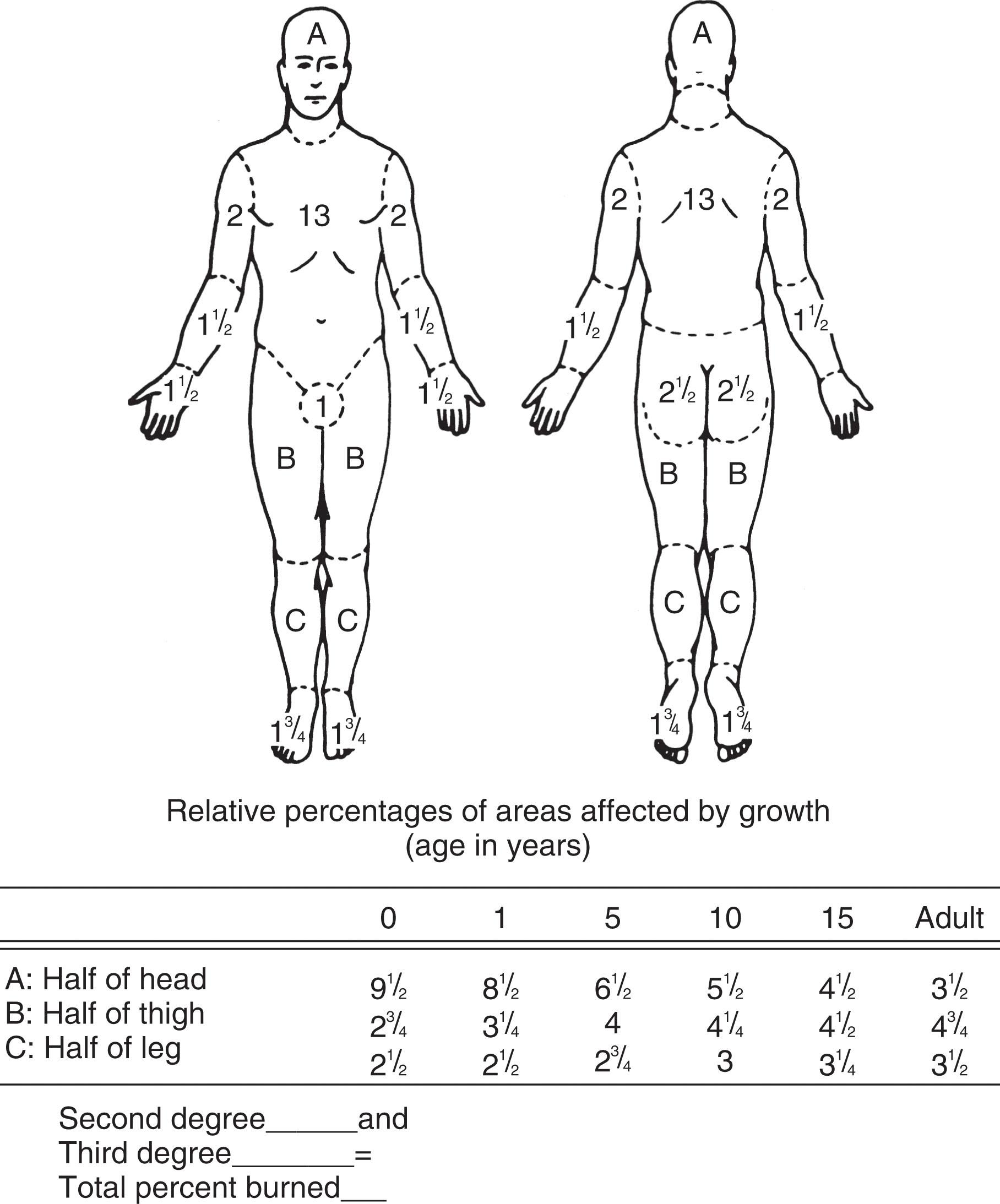
Burns have classically been described according to depth ( Table 15-1 ) and percentage of total body surface area (TBSA). In 1944, Lund and Browder produced a method for calculating percentage TBSA in which each foot accounts for 3.5% TBSA (for both children and adults) ( Fig. 15-1 ). The relatively small contribution each foot makes to TBSA can erroneously lead to the impression that burns to the foot are minor. Because of tremendous functional risks associated with these injuries, however, all but very superficial burns of the feet should be referred to a specialized burn center for appropriate management ( Box 15-1 ).
| Degree | Description |
|---|---|
| First | Erythema without blistering |
| Second | Erythema with blistering |
| Third | Destruction of full thickness of skin and often deeper tissues |
The following types of burn injuries usually require referral to a burn center:
Partial-thickness burns greater than 10% of the total body surface area (TBSA)
Burns involving face, hands, feet, genitalia, perineum, or major joints
Third-degree burns in any age group
Electrical burns, including lightning injury (significant volumes of tissue beneath the surface may be injured and result in acute renal failure and other complications)
Significant chemical burns
Inhalation injury
Burn injury in patients with pre-existing illness that could complicate management, prolong recovery, or affect mortality
Any burn patient in whom concomitant trauma, such as fracture, poses an increased risk for morbidity or mortality (patient may initially be stabilized in a trauma center prior to transfer)
Children with burns in hospitals without qualified personnel or equipment for their care
Burn injury in patients who will require special social, emotional, or long-term rehabilitative support, including cases involving suspected child abuse and neglect
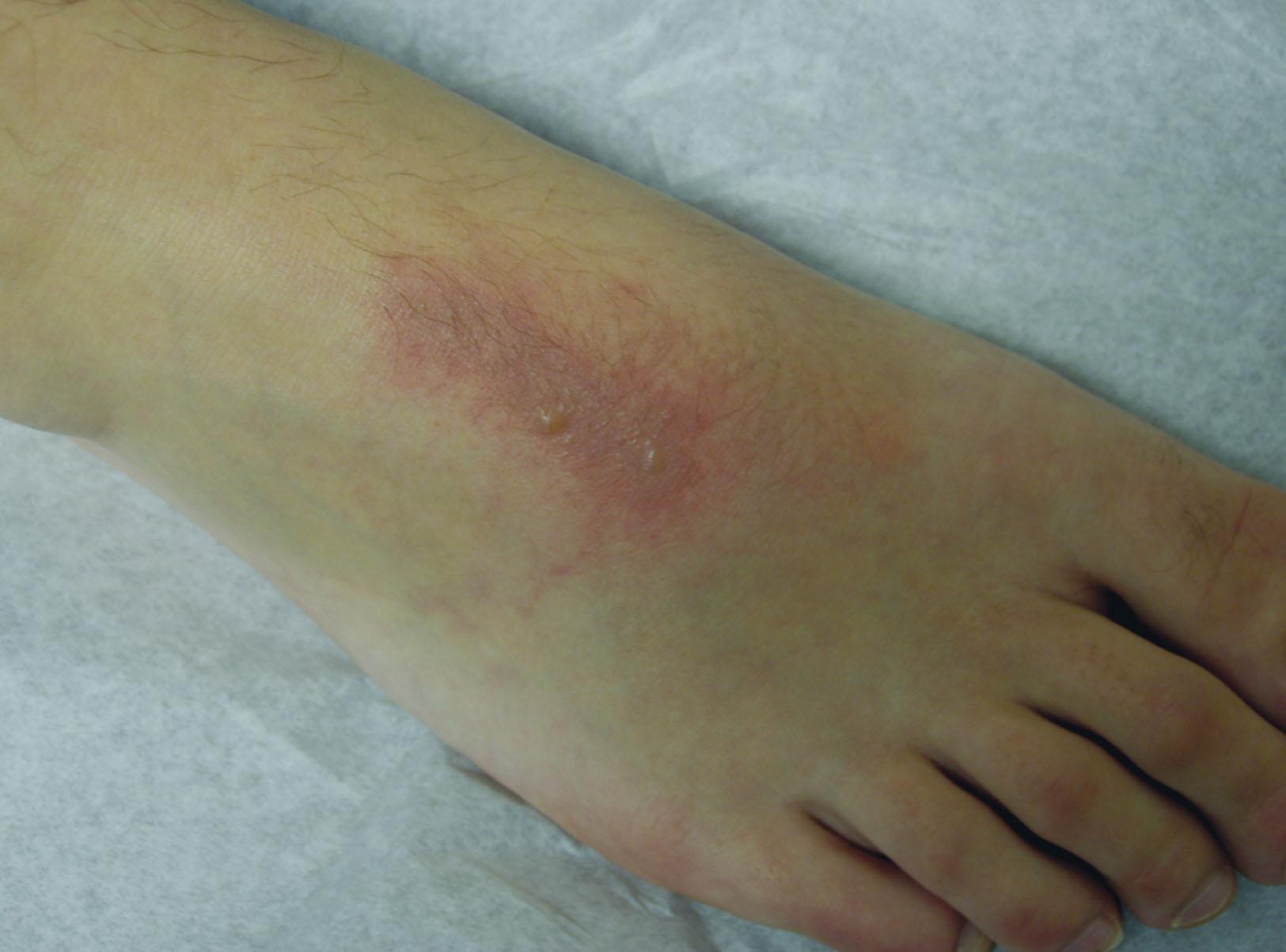
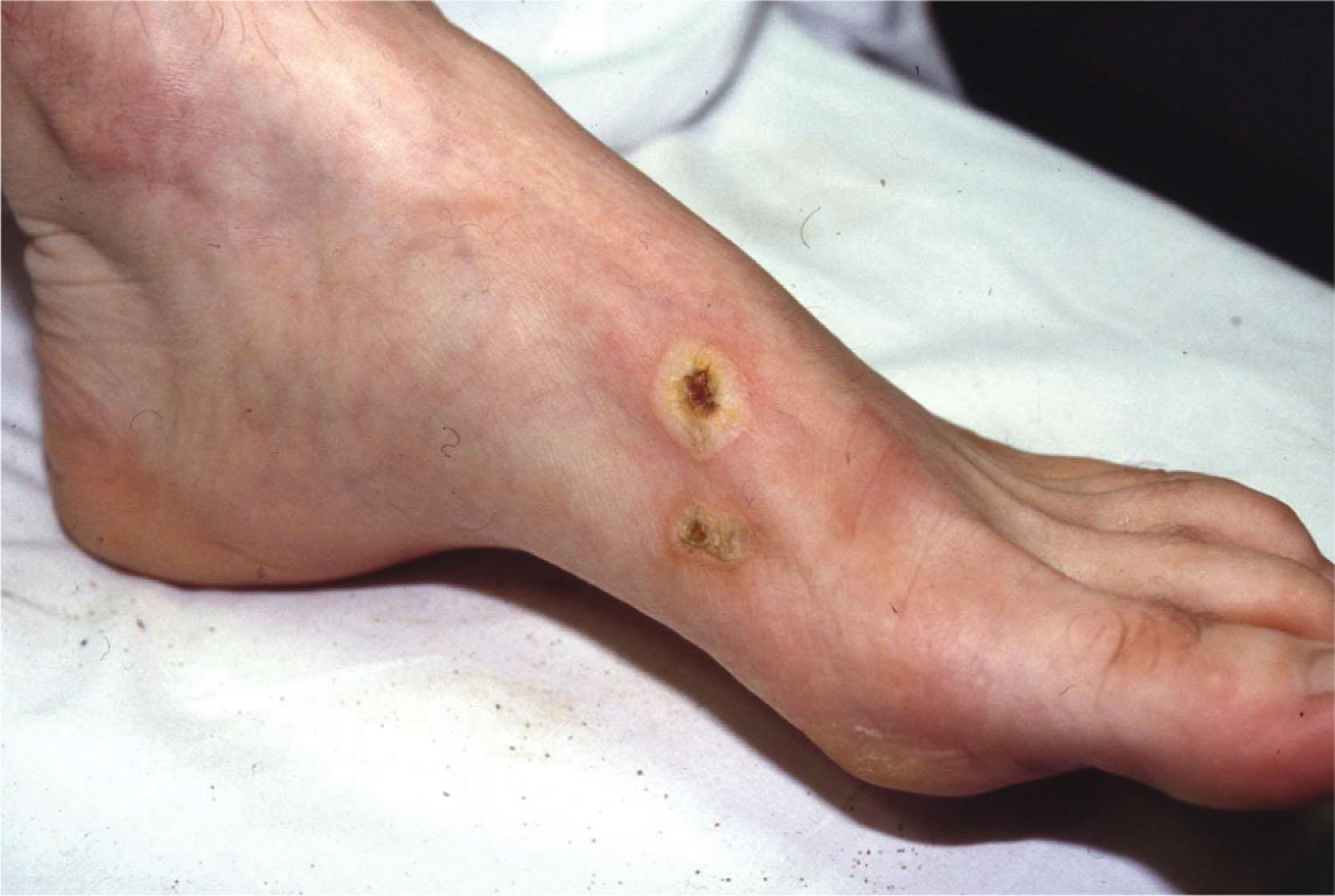
Isolated pedal burns are most often sustained at home, indoors, and in the kitchen. Even such minor burns of the lower extremities can cause severe disability, chronic pain, and loss of work. These injuries can be especially troubling in patients with decreased sensory feedback resulting from peripheral neuropathy, because inadequate treatment can lead to increased morbidity. For example, a typical scenario might involve a patient who sustains a minor burn on the dorsum of the foot ( Fig. 15-2 ). The local outpatient center treats this patient with an emollient and analgesics, and the patient is permitted to return to work. With the resultant dependency of the extremity and footwear abrading the dorsum of the foot, the patient may develop secondary lymphedema and cellulitis, leading to hospitalization, potential need for surgical treatment and prolonged foot elevation. Burns to the feet must therefore be recognized early and treated expeditiously and appropriately to minimize functional impairment.
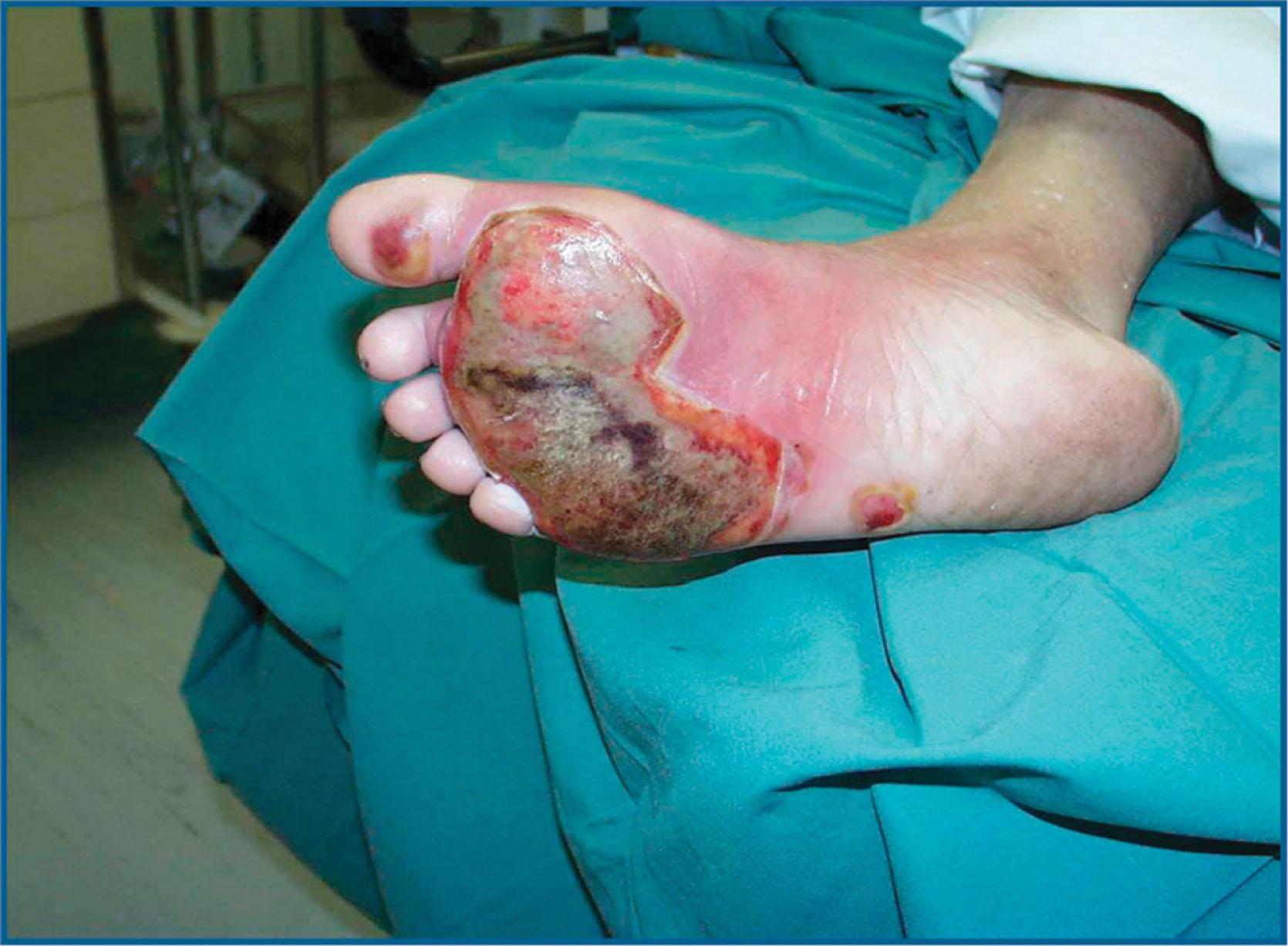
It has been estimated that more than 90% of burns can be attributed to carelessness and over 50% of burns are related to smoking or alcohol. Diabetic neuropathy is a risk factor for foot burns due to the lack of protective sensation, with nearly three times the risk of sustaining a burn compared to nondiabetic age-matched controls ( Fig. 15-3 ). As expected, diabetic patients with burns also are at increased risk of infections, repeat wound debridements, ICU requirement, complications, and longer length of hospital stay. This underscores the importance of educating this at-risk population to avoid heating packs and warming foot baths and to test water temperature prior to soaking (less than 100º F is considered safe).
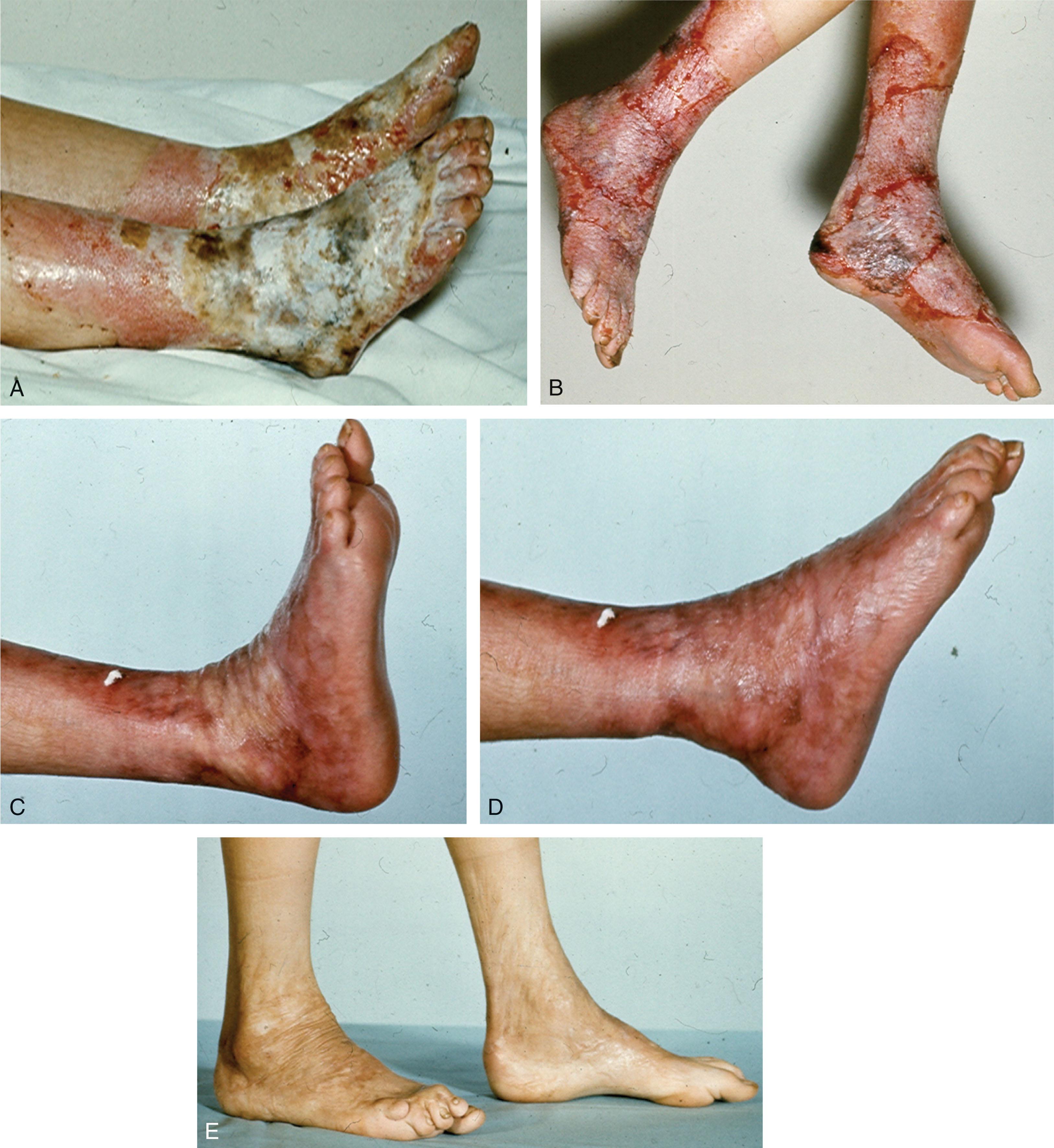
Minor burns are common and require early and appropriate care to prevent infection and scarring. Damage can be minimized by immersing the foot in cold water as soon as possible, halting the burning process. The goals of care are then to remove dead tissue and protect viable tissue during healing. Very superficial burns require only an emollient (over-the-counter burn cream) to limit inflammation and prevent desiccation.
More severe burns can be effectively excised and grafted by general or plastic surgeons in hospitals that have the proper facilities, as long as the involved area is relatively small. However, burns of functionally important areas (hands and feet) should be excised by a burn specialist. Burns in excess of 10% TBSA should be excised in a burn treatment facility with specially trained support personnel, including experienced anesthesiologists, nurses, and critical care teams. Circumferential burns of the lower extremities may require urgent escharotomy in order to prevent compartment syndrome. Compartment measurements should be followed in patients with significant burns because pressures can increase in time and pulses have no predictive value for ischemia. Even in noncircumferential burns, inadequate fasciotomies or escharotomy can result in foot drop, muscle necrosis, or even amputation. In massively burned patients, burns involving the feet are a negative predictor of survival.
Definitive burn excision and coverage should be undertaken when the patient is stable and medically optimized, ideally within 72 hours. Studies have shown a higher incidence of graft failure and sepsis when definitive management is delayed beyond 7 days.
Small (less than 20% TBSA) full-thickness burns and indeterminate (deep partial-thickness vs. full-thickness) burns, if treated by an experienced surgeon, can be excised safely and grafted ( Fig. 15-4 ). Converting such burns to a closed wound results in fewer painful debridements, decreased hospital stay, decreased cost to the patient, and less time away from school or work, with healing occurring in less than 3 weeks. Early coverage also facilitates more rapid ambulation and rehabilitation and minimizes scarring. Split-thickness skin grafting, as opposed to secondary-intention healing with granulation tissue, is more cosmetically acceptable ( Fig. 15-5 ).
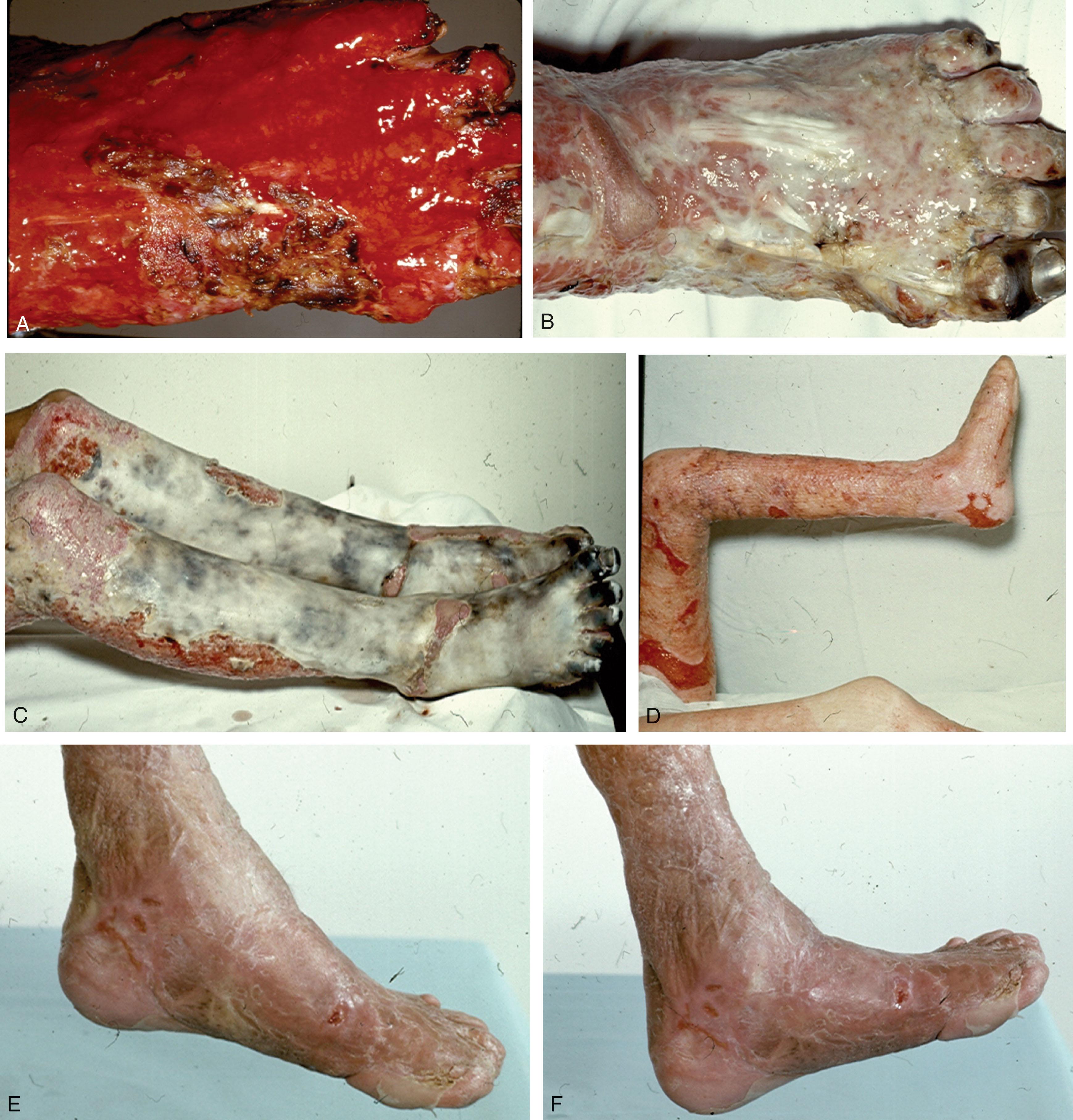
If not excised, even a small burn can take 3 to 6 weeks to separate and another 2 to 3 weeks for grafting and final healing. Early excision and grafting also decrease stress, hypermetabolism, and the overall bacterial load, thus enabling the patient to resist and fight other burn complications more effectively. Because of subsequent graft contracture and extremity growth, however, early excision and grafting of foot burns in children and adolescents might not have the same advantages as seen in adults.
If the skin excision is to the level of the fat domes, the hair follicles and sweat glands are preserved, and graft healing can be expected in 14 to 18 days. If the bulk of the deep epithelial elements remain, healing will take 2 to 3 weeks. With deep partial-thickness burns, only meager epithelial elements remain, and healing can take 3 to 6 weeks, with poor scar formation.
Traditional treatment of foot and ankle burns after excision and grafting was bed rest, limb elevation, and gradual ambulation after 5 to 10 days. Newer protocols advocate immediate application of an Unna boot in the operating room or the next day, followed by normal ambulation 4 hours later and hospital discharge. With use of sheets of narrowly meshed grafts, results were good to excellent in 95 of 100 patients. This technique avoids frequent dressing changes, permits brief or no hospital stay, and provides excellent graft take with prompt return to work. Plaster casts or posterior splints may also sufficiently support the graft site to permit early weightbearing postoperatively.
There are many coverage options including dermal autograft, dermal allograft, or synthetic materials. Partial-thickness burns that are clean and superficial can benefit from a biosynthetic bilaminar membrane dressing (cultured epidermal autograft), which forms a skin substitute while protecting the wound. These materials may be a useful adjunct to burn care but are expensive ($6000–$10,000 per 1% TBSA).
Fasciocutaneous flaps, adipofascial reverse dorsal metatarsal artery flaps, and distally based island flaps of the dorsalis pedis artery are used to treat great toe defects. Distally based reverse island flaps of the extensor digitorum brevis muscle are harvested to cover tissue defects. Cross-leg free flaps are used to cover extensive burn wounds of the lower extremity. Full-thickness burns of the sole’s plantar surface can be treated with radial forearm flaps, which are supple and adapt to the contours of the foot, preserving normal shape and making orthoses unnecessary. For full-thickness burns in the distal one third of lower leg, successful outcomes have been shown with the neurocutaneous sural flap ( Fig. 15-6 ).
Reconstruction of bilateral distal lower limb defects presents a major challenge. The problem is compounded in a large burn because of the limited availability of normal unscarred donor sites and recipient vessels. To close the wound temporarily after escharectomy, human cadaver skin or biosynthetic skin may be used. With massive burns, the patient’s and family’s desires must be combined with the burn team’s realistic prognosis to determine the most judicious use of the available donor tissue to meet reconstructive needs.
Contractures are common after hospitalization due to burn injury, with incidence of approximately 33% despite preventative measures like splinting and early mobilization. After healing, the initial appearance of the burn wound may appear satisfactory, but the true prognosis is uncertain until at least 4 months have elapsed, when the healing area becomes elevated and hard. Wound healing in burns is marked by increases in vascularity, fibroblasts, myofibroblasts, collagen deposition, interstitial material, and edema. These changes are conducive to the formation of hypertrophic scars and contractures.
During the hypervascular phase, surgery is not indicated because of the potential for massive postoperative scarring. Constant-pressure dressings, splinting, and local injection of steroids best treat this fused mass of collagen. After wound re-epithelization, gel or gel sheets (with or without silicone) may be applied until the scar has matured. Although anecdotally useful, massage therapy has not been shown to be effective within the first 1 year after the burn incident. Once the scar has matured, local tissue response is less, and improved surgical results can be expected.
Scar formation is most common on the dorsum of the foot, often resulting in hyperextension of the toes and metatarsophalangeal (MTP) joint subluxation. Prevention includes splinting and wearing shoes to maintain the toes in the straight position.
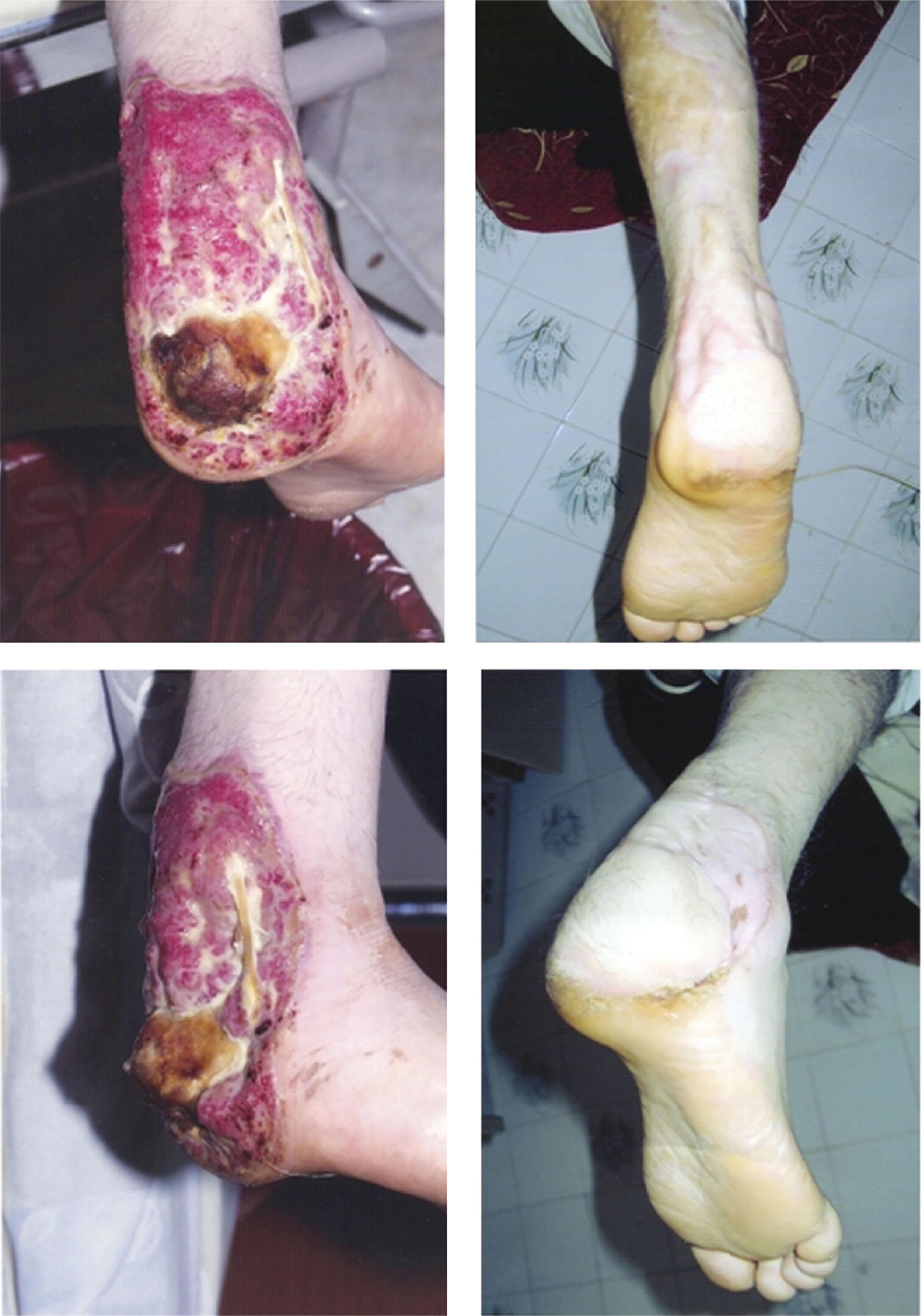
Fixed contractures in adults respond to excision of the scar over the MTP joints and application of a skin graft ( Fig. 15-7 ). The toe flexors alone are often enough to maintain correction after scar excision, but if not, toe traction is used during healing. High-top shoes with padded tongues are worn constantly in the postoperative period to maintain correction. Severe equinus contractures are treated with excision of scarred tissue and z-lengthening of the Achilles tendon. The subsequent soft tissue defect is typically repaired with free tissue transfer ( Fig. 15-8 ).
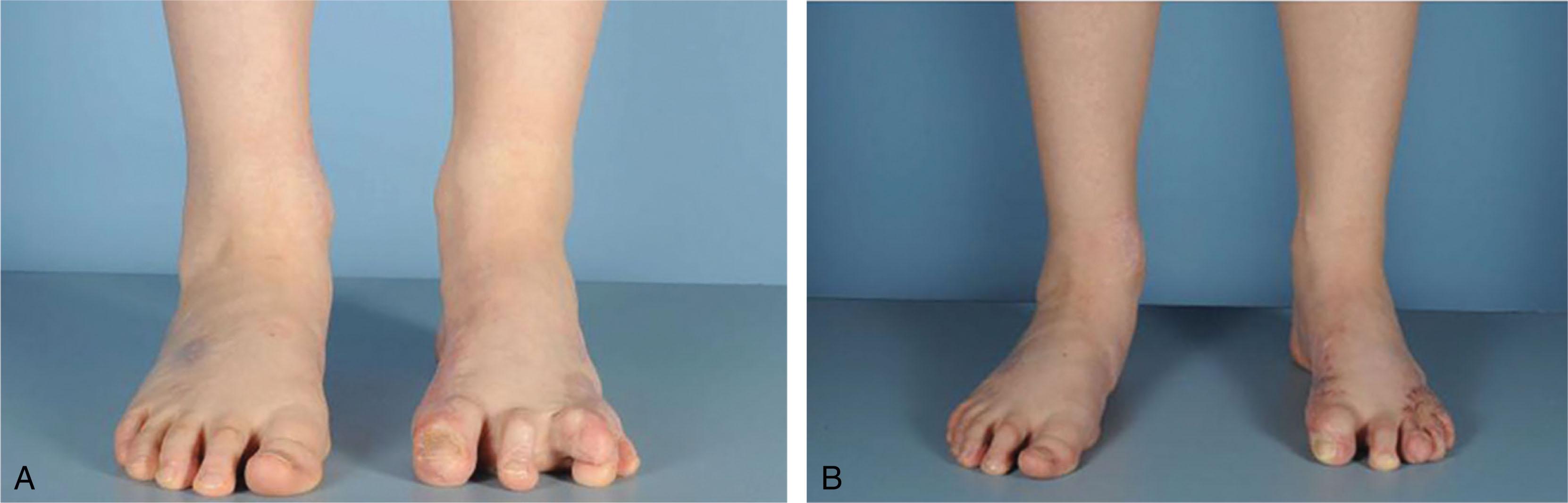
In children, burn scar contractures of the feet are often treated by release of the contracture band and placement of a skin graft in the resulting defect, with an expected 85% success rate. A delay from the thermal injury to the reconstructive procedure does not affect the outcome. Immobilization postoperatively in a dynamic splint is important to prevent contracture recurrence. Initial positioning and splinting plus the use of serial casting and supportive tone-reducing orthoses are important. Equinus contractures can be corrected from 45 degrees of equinus to neutral dorsiflexion over 2 months with biweekly cast changes.
However, contractures present for a long period cannot be brought into anatomic position easily because of ankylosed joints and shortened tendons, nerves, and vessels. Although tendons can be lengthened, neural and vascular structures cannot. Complete excision of the scarred portions of involved muscles in fixed postburn equinovarus contractures is advocated. External fixators (e.g., Ilizarov) associated with soft tissue procedures can be used to correct major foot deformities gradually. With an equinus contracture, patella tendon-bearing (PTB) orthoses are used to allow weight bearing until definitive reconstruction can be carried out.
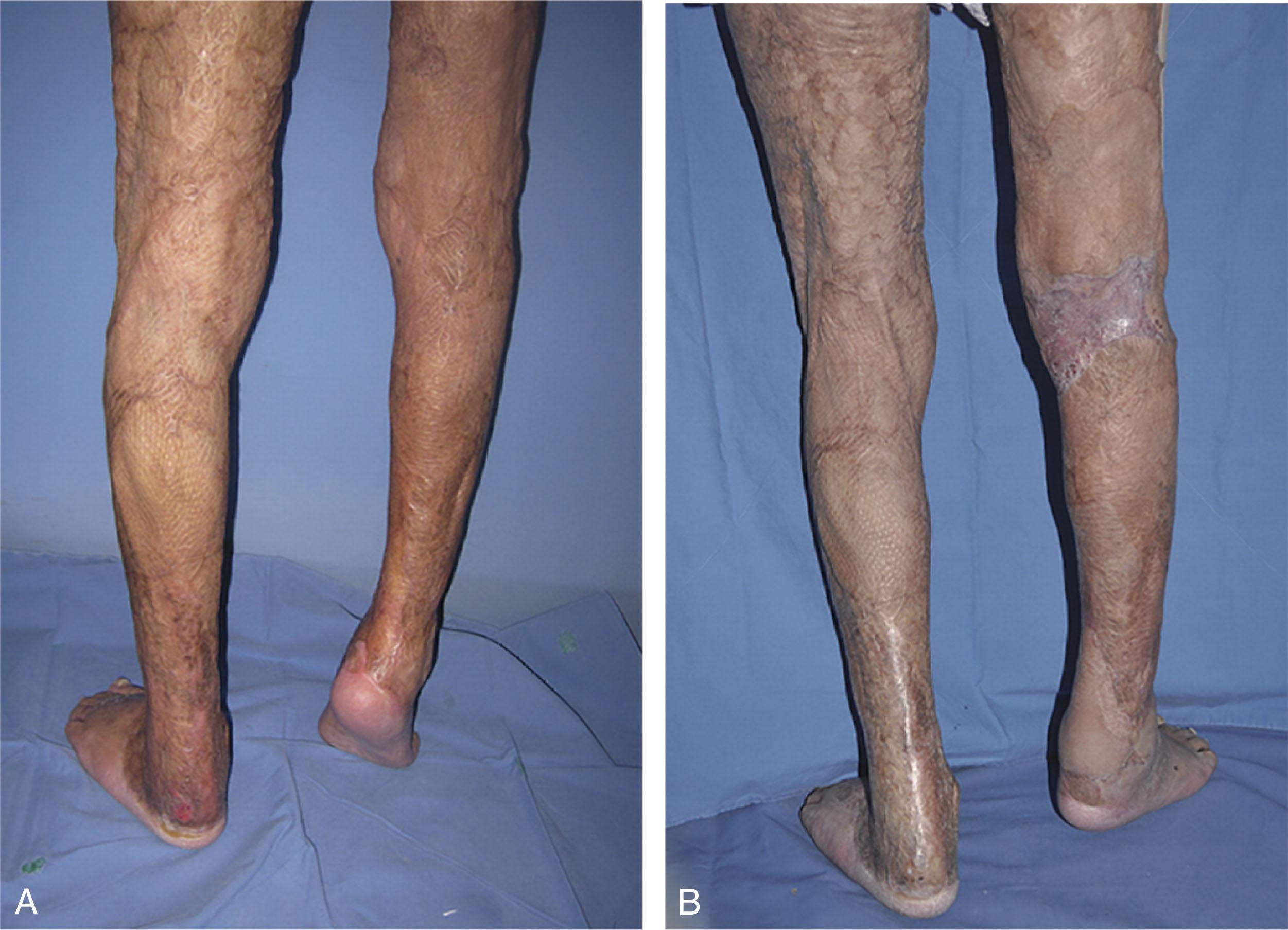
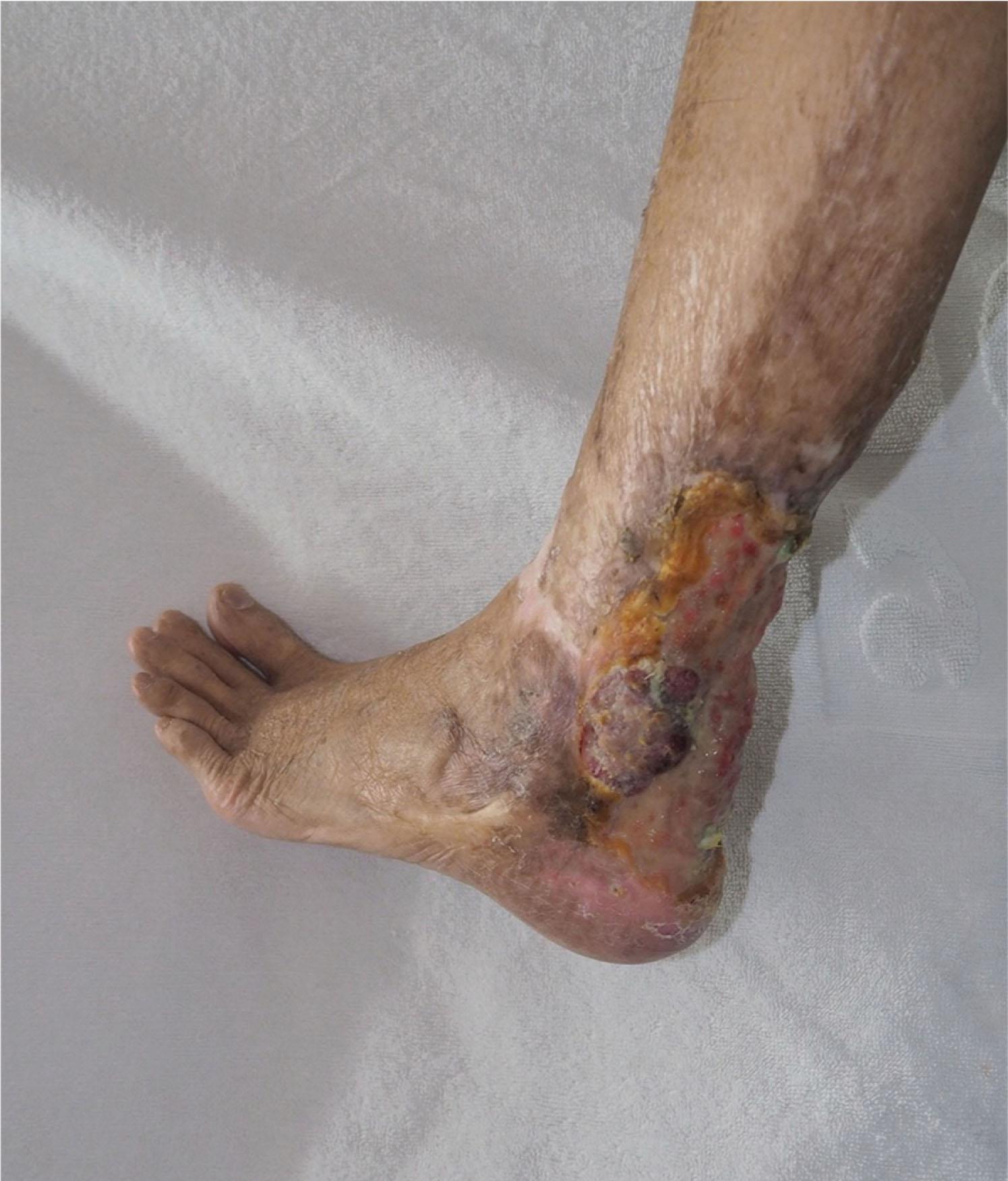
Malignant changes in burn scars have been reported in the literature. Most cases are long-standing ulcers (Marjolin ulcers), which evolve into squamous cell or some other epithelial cell carcinoma ( Fig. 15-9 ). Malignant ulcers are more common after burns to the lower extremities than upper and manifest after a mean of 27 years. Because malignant changes appear decades after the initial injury, residual burn scars should be routinely inspected and patients forewarned about the importance of expeditious treatment for breakdown of a previously healed burn wound.
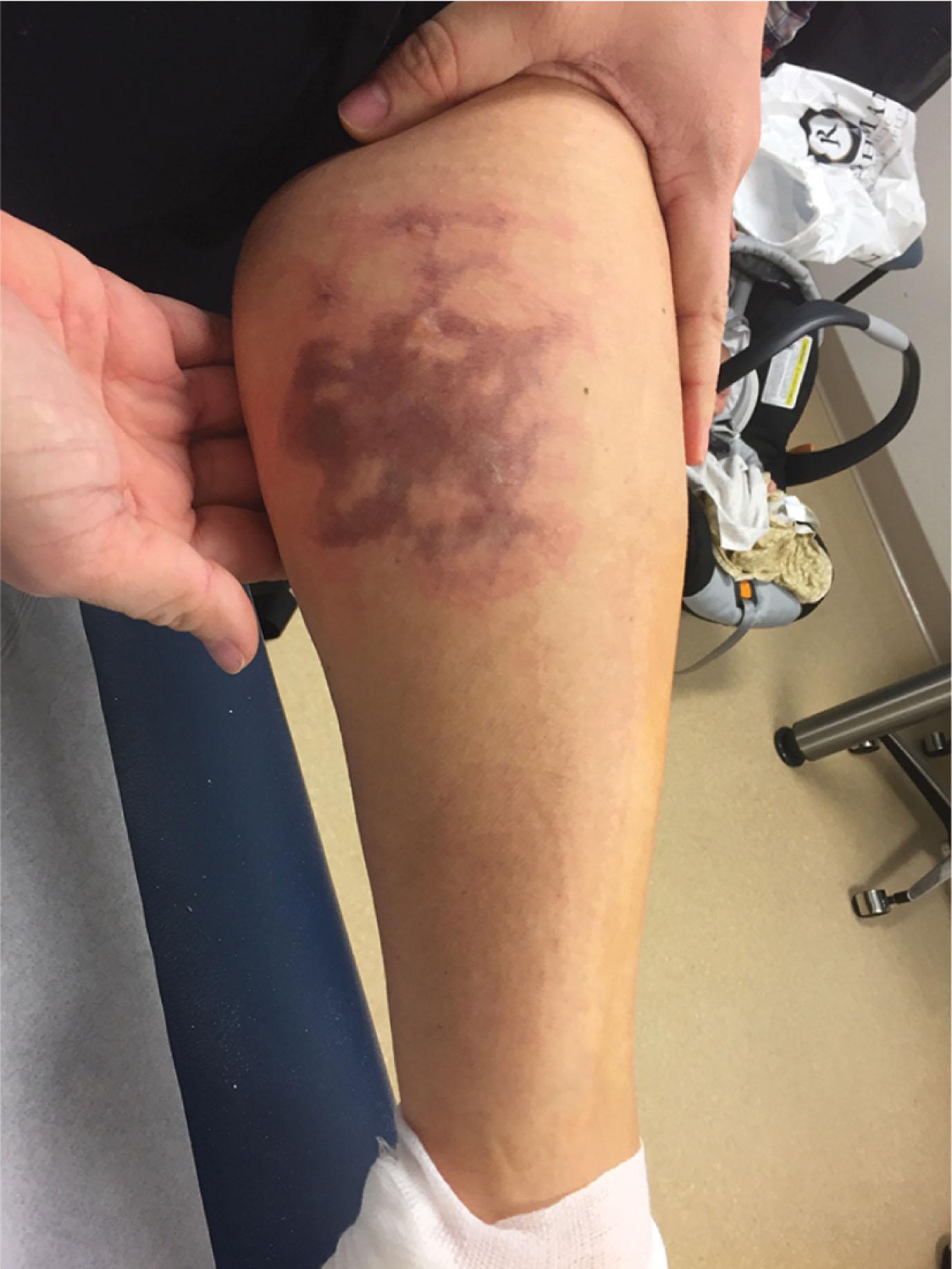
Of particular relevance to the orthopedic surgeon is thermal injury related to the application and removal of casts. Fortunately, these are rare. Plaster and fiberglass undergo an exothermic process when combined with water. In fact, first-degree burns have been inflicted when using 30 layers of plaster dipped into water at 42°C ( Fig. 15-10 ). In general, however, when eight or more layers of plaster are dipped into water warmer than 24°C, high enough temperatures can be reached to damage skin. Therefore, judicious use of padding including at least two layers of Webril, lukewarm water (lower than 24°C), and adequate material are recommended. Pressure application during casting is associated with increased temperature over the molded area, especially when plaster is overwrapped by fiberglass. When molding a plaster splint overwrapped with fiberglass, the provider should wait until the plaster has fully cooled.
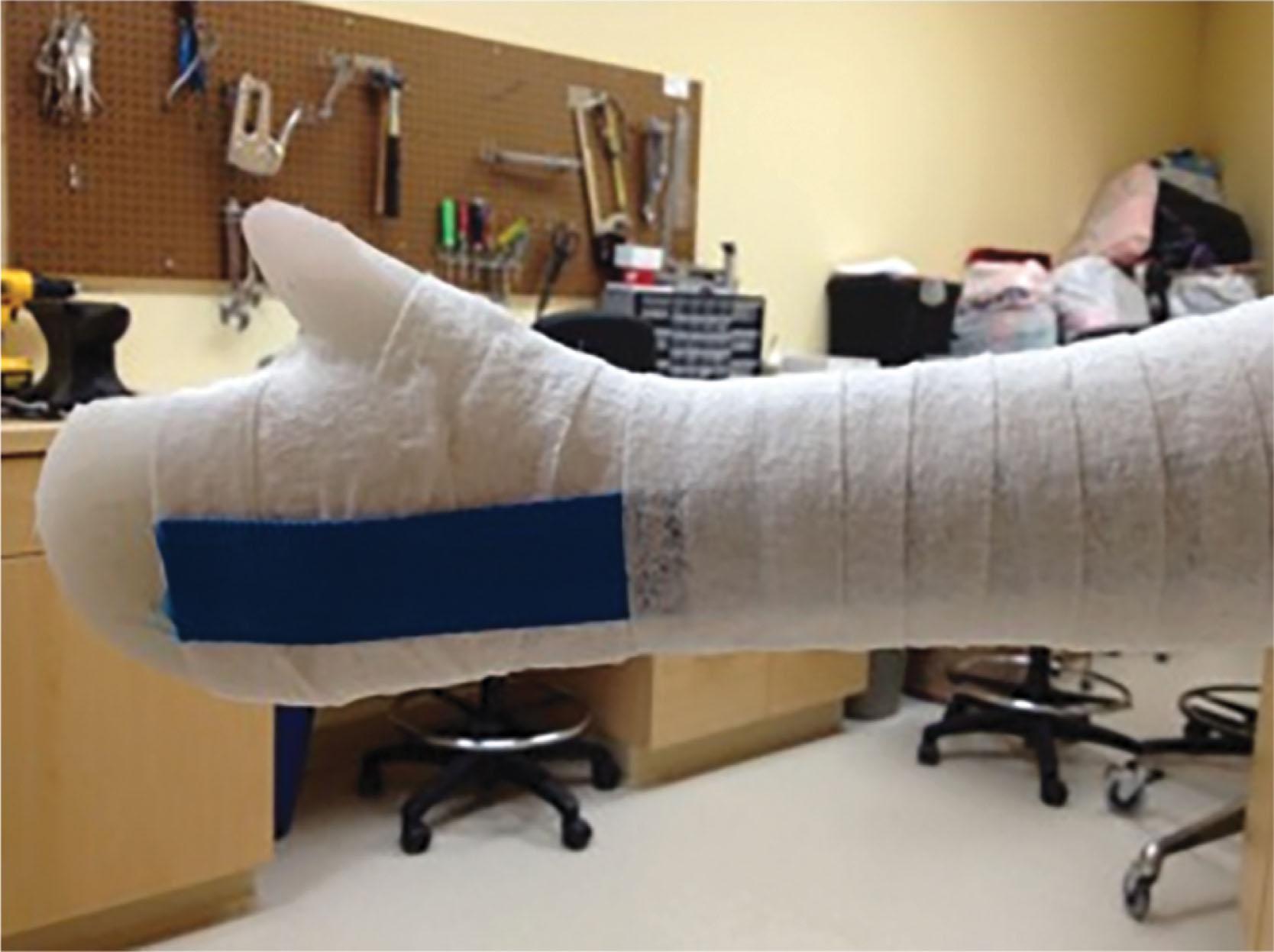
Oscillating circular saw blades have also been known to cause minor burns during cast removal with an incidence of 1.3 to 7.2 burns per 1000 casts removed. Risk factors for cast saw burns include lower level of training and cast location near the ankle and foot ( Box 15-2 ). Dull blades, and specifically stainless-steel blades, are more likely to consistently generate blade temperatures of 120°F to 130°F. To minimize the potential morbidity of this routine in-office procedure, operators should be well-trained in the proper technique and in blade maintenance. In a cadaver model, Shuler and Grisafi found that a poor removal technique (the cast saw blade never leaving the cast material during cutting), fiberglass casting material, and thinner cast padding resulted in significantly higher skin temperatures. The poor technique increased skin temperatures by an average of 5.0°C ( P < 0.05). Fiberglass casting materials increased skin temperatures by an average of 7.4°C ( P < 0.05). When compared to two layers of cast padding, four layers significantly reduced skin temperatures during cast removal. Another protective measure includes safety strips incorporated into casts that have been shown to reduce blade-to-skin contact and decrease heat transfer in a clinical model ( Fig. 15-11 ).
Situational
Lower level of training
Cast location near wrist, elbow, ankle, foot
Emergency department procedure
Overnight procedure
Equipment
Dull blade
Synthetic saw blade (metal is preferred)
Lack of vacuum
Casting Technique
> 12 layers of plaster
Fiberglass generates more heat than plaster during removal
< or = 2 layers of padding
Saw Technique *⁎ *
* space between the inner cast border and patient’s skin.
Lack of pauses to assess blade temperature and cool with water
Sliding blade down the cast (in-and-out technique is safer)
Reduced safety distance (can increase safety distance by cutting cast parallel to force of gravity to allow the limb to fall as far away from inner cast perimeter as possible)
The passage of electrical current through the body generates intense heat and causes tissue injury. The greater the voltage and electrical resistance of the tissue, the greater the temperature. Alternating current (AC), which causes tetany, is more dangerous than direct current (DC), and high-tension injuries (e.g., high-voltage power lines) involve contact with greater than 1000 V.
Electrical injuries do not usually involve foot contact, but the lower extremities conduct the current to the ground, with the current exiting through the foot. Although the superficial manifestation of an exit wound on the lower extremity may be small, it can overlie extensive damage to deeper structures ( Fig. 15-12 ). Additional trauma is caused by arc burn across flexion areas in the popliteal fossa and ankle, which generates heat up to 4000º F. AC-induced tetany often results in the victim’s inability to release the current source, thus intensifying the destruction. The likelihood of associated injury is high. Victims often fall from high-tension wires or electrical poles, and the clothing can ignite, causing additional surface burning. There are unique bone and soft tissue changes that are associated with electrical burns ( Box 15-3 ).
Fractures can result from muscle cramps, mainly in the midthoracic vertebral bodies and humeral, femoral, and scapular necks. Longitudinal fractures, often after a zigzag contour (osteoschisis), can result from a direct mechanical splitting effect of the passing current.
Melting of bone from intense heat can result in small rounded osseous fragments resembling drips of wax (bone pearls) lying along the surface of the bone.
Osteonecrosis can result in osteolytic medullary lucent cystic areas in both the diaphysis and the metaphysis. Cortical sequestration, collapse of subchondral bone, epiphyseal collapse, and fragmentation have all been described.
Growth disturbances can result from premature fusion of the epiphysis in children.
Laceration of the skin can allow access of microorganisms with resulting osteomyelitis and suppurative arthritis. Septic arthritis can result in joint ankylosis.
Scars and contractures of soft tissues and periarticular calcification and ossification can cause limited joint movement.
In one electrical burn case, a 25-year-old man was digging a trench with a shovel and hit a high-voltage line with the shovel ( Fig. 15-13 ). Initial injury was on his finger, and the large exit wound on his back required grafting. As he fell, his foot kicked the high-voltage line and the electricity produced a second entrance wound on his foot; the current traveled through his leg and exited through the knee. After a prolonged hospital stay, the patient survived and the burn wounds went on to heal. The patient has a residual foot drop from the nerve injury.
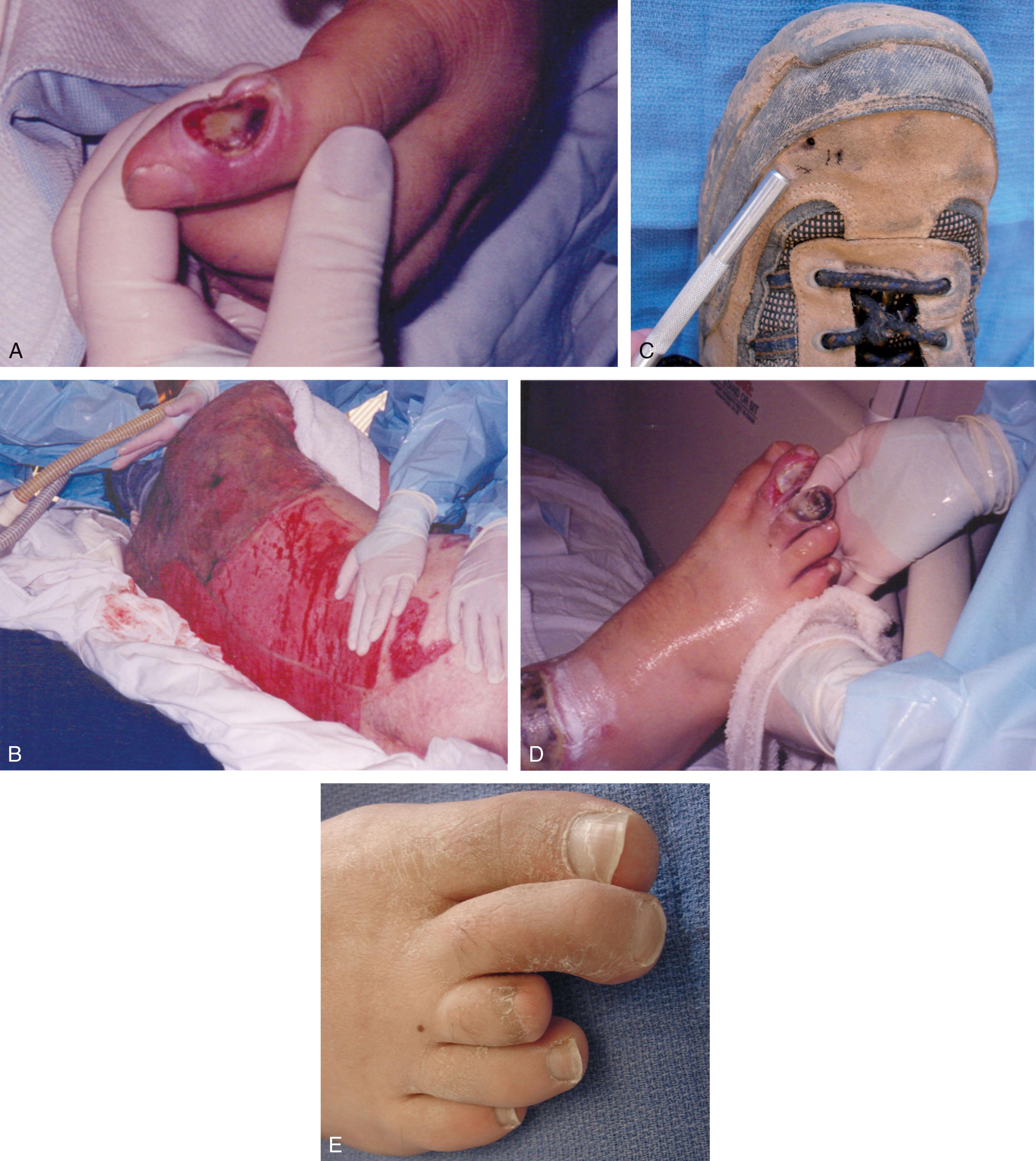
Management guidelines for electrical injuries are similar to those for other thermal burns, but the physician needs to assess the effects of electricity on vital systems. After ensuring adequate resuscitation, the involved lower extremity should be assessed for neurovascular compromise. The incidence of deep tissue necrosis and compartment syndrome is significant; pressures and pulses need careful monitoring. Muscle necrosis can cause myoglobinuria, requiring large fluid volumes for alkalization of the urine. High-voltage burns demand an immediate fasciotomy and escharectomy ( Fig. 15-14 ). Survival of the extremity depends on extent of the injury, viability of the blood vessels, and management of the impending ischemia.
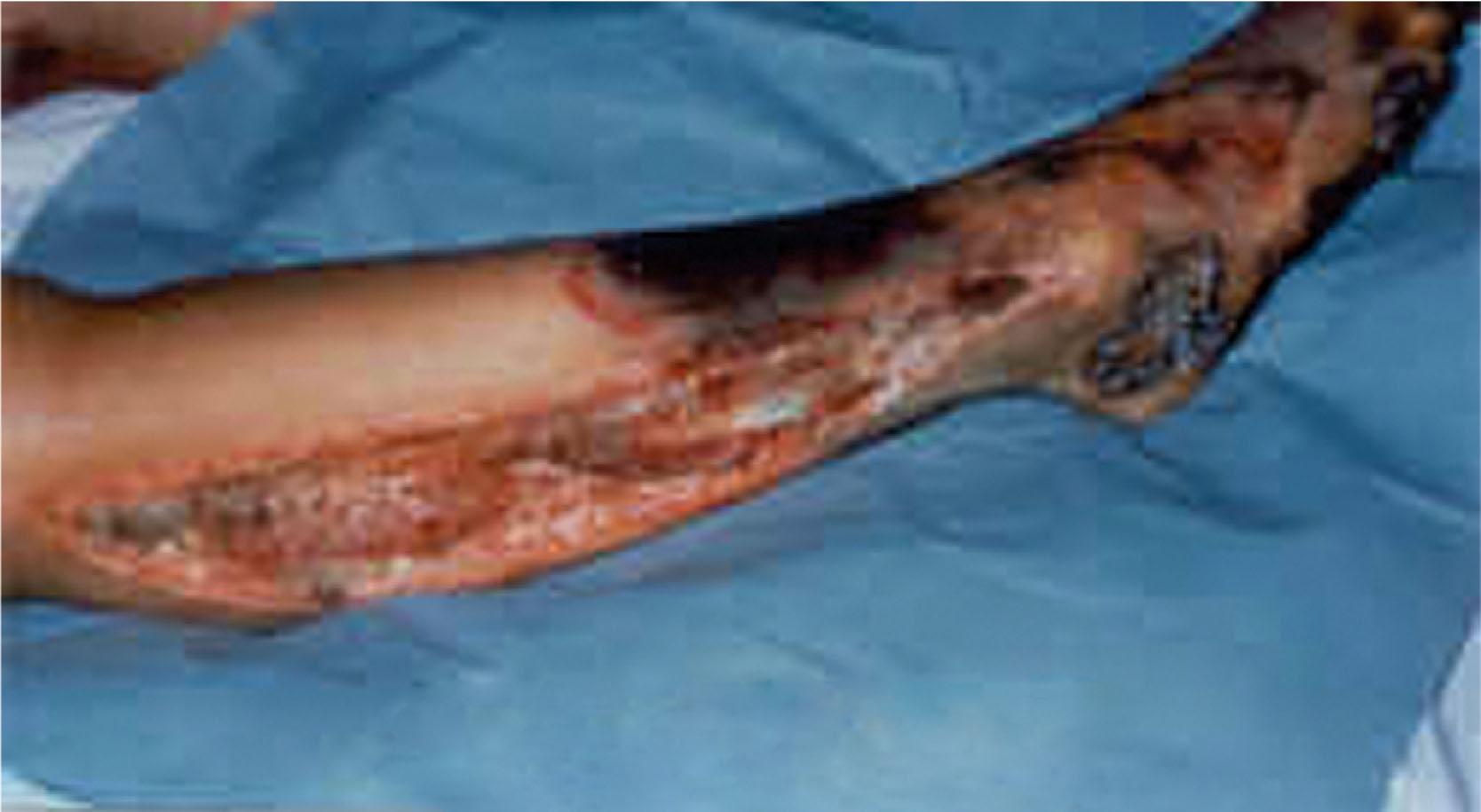
Tissue loss is often progressive. Muscle that is initially contractile may be nonviable on subsequent evaluation because of ongoing microvascular thrombosis; this is unique to electrical burns. Delayed gangrene is not unusual. Staged sequential exploration with conservative meticulous debridement is the best way to assess and manage the extent of injury. Blowout lesions can occur at the current exit wounds in the extremity and can require amputation. Closure of the amputation site is delayed until the surgeon is certain that no further progressive deep necrosis exists. Significant new bone formation at long-bone amputation sites (tibia, fibula) is common after electrical injury.
Newer literature suggests improved outcomes after high-voltage electrical burn with early and aggressive debridement within 2 to 3 days followed by flap coverage within 10 days. For electrical burns to the foot and ankle, distally based neurofasciocutaneous flaps (sural or saphenous) have a high success rate, even when the flap pedicle is in close proximity to the zone of injury ( Fig. 15-15 ).
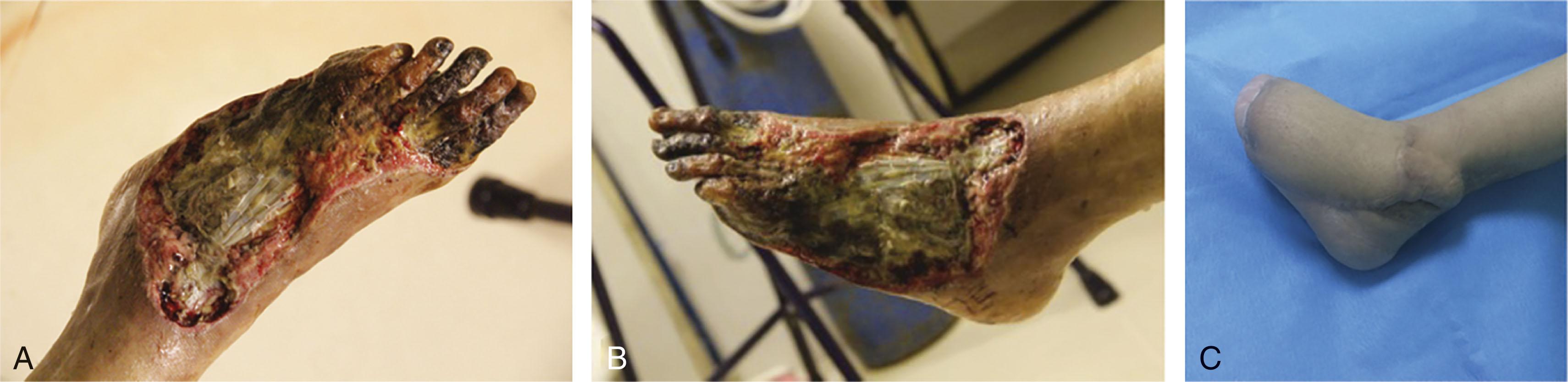
Chemicals burn the foot through direct contact, acutely destroying tissue by a multitude of reactions that devitalize living cells ( Fig. 15-16 ). The burn can also be insidious and develop over a long time. For example, in a chemical plant worker who wears heavy socks with work boots, the socks can absorb toxic materials and burn slowly without the worker being aware.
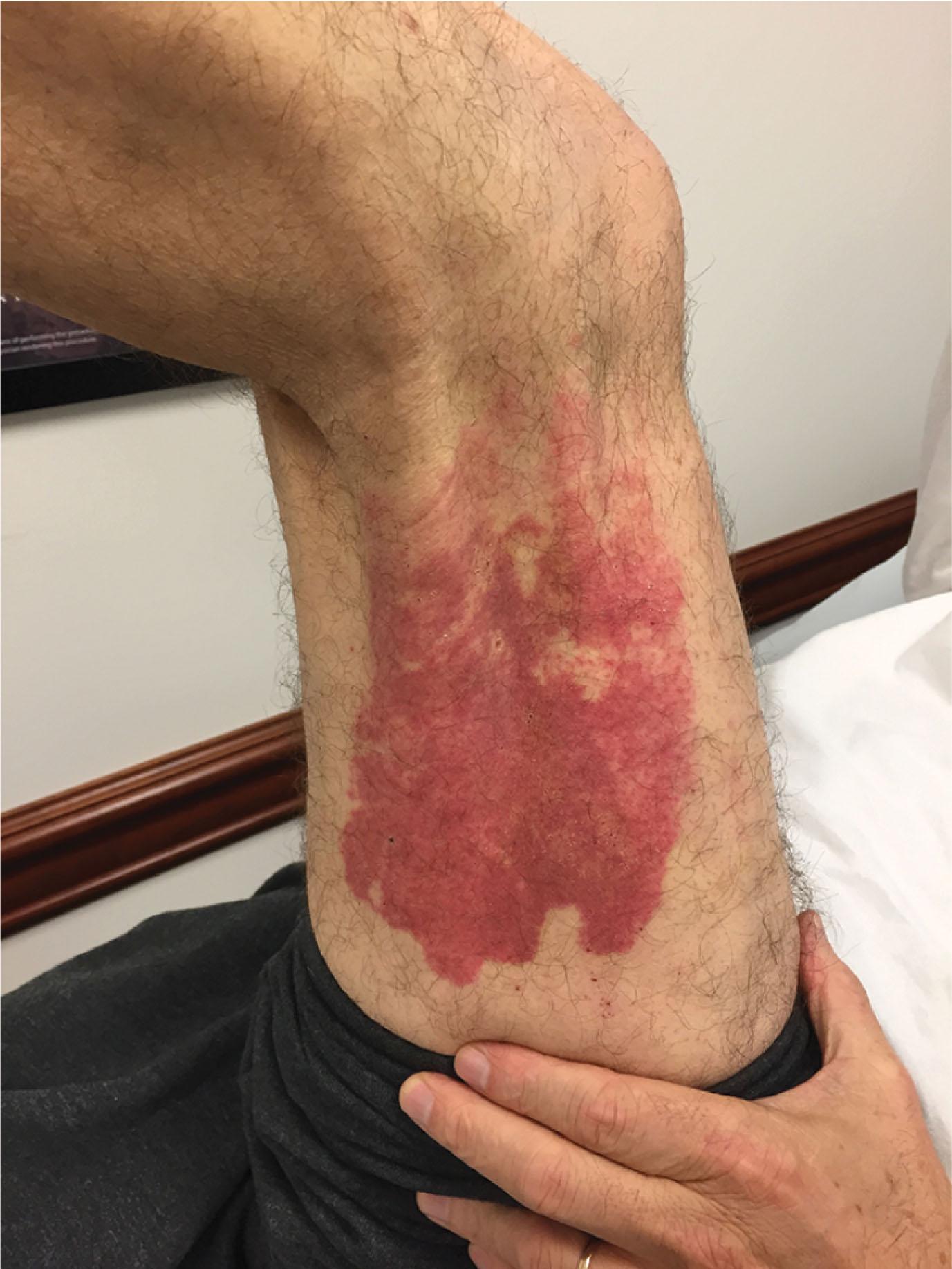
The burns themselves do not provide any long-term management problems not previously discussed under thermal or electrical burns. The same careful attention needs to be given to prophylactic antibiotics, tetanus protection, foot elevation, pulse measurements, and compartment pressures.
The key issue, however, is the acute management of the burn and obtaining the proper neutralization solutions. Lists of the solutions needed are in most emergency rooms, but the solutions themselves are often not immediately available. In general, contaminated clothing and shoes should be removed, particulate matter debrided, and vigorous water lavage commenced at once to dilute the chemical and decrease the intensity of the exposure. However, there are some notable exceptions to the use of water ( Table 15-2 ). In some cases, water lavage can result in a chemical reaction that exacerbates the tissue damage.
| Chemical Burn | Neutralizing Agent | Water Lavage Result |
|---|---|---|
|
|
Water causes ionization and increased heat production |
|
2% copper sulfate solution to visualize particles | Water lavage is insufficient; particles must be manually debrided |
| Hydrofluoric acid |
|
Water lavage is insufficient because the acid is absorbed into exposed tissues |
| Phenol (carbolic acid) |
|
Water lavage is insufficient because the acid has limited solubility in water |
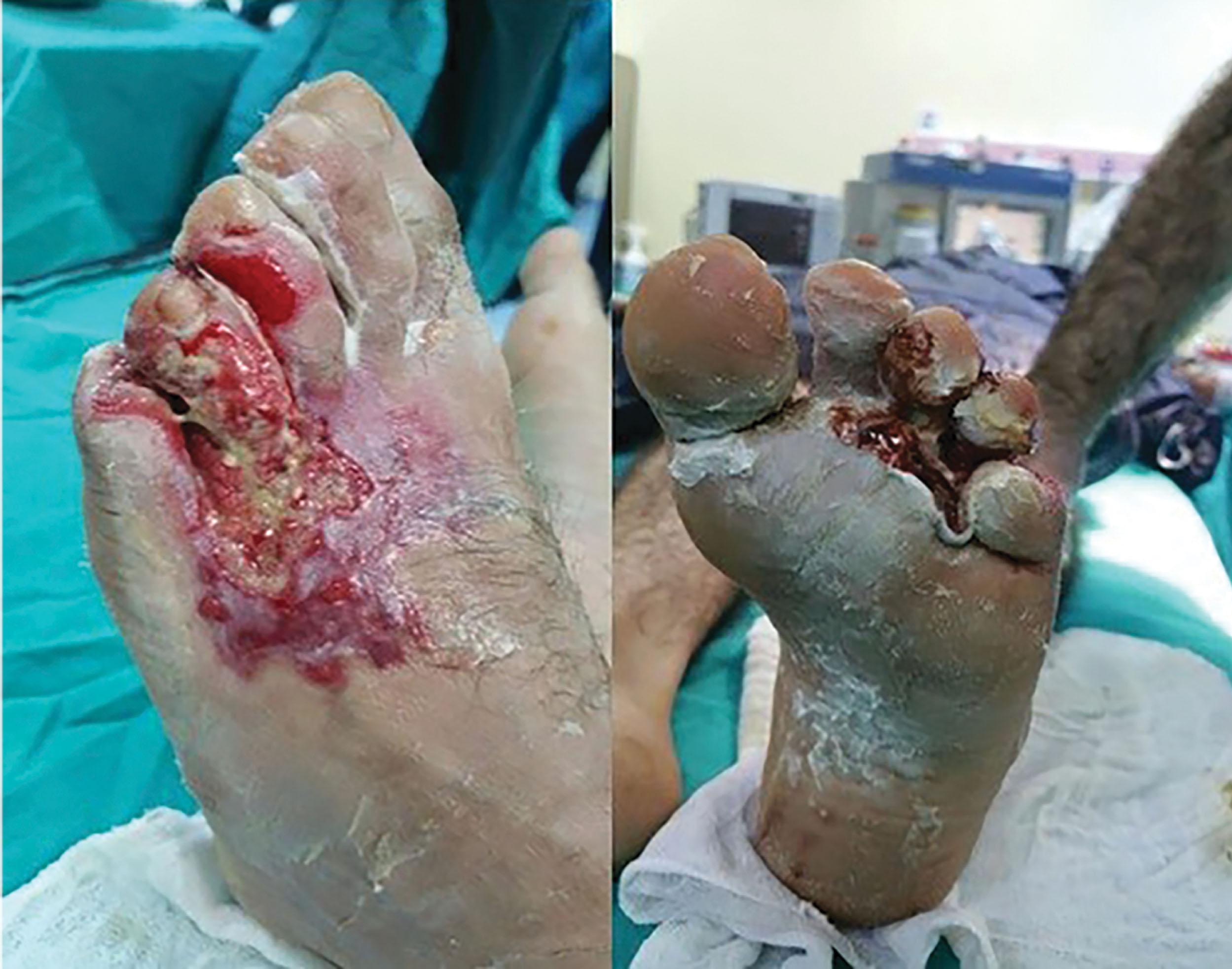
Another special considering is wet cement, a seemingly benign common material, that can cause significant caustic burns of the foot and ankle in home improvement enthusiasts and the children who play in it. Professional masons are well aware of cement’s toxicity. Cement contains a high percentage of calcium oxide (lime). When mixed with water, lime creates a highly alkaline solution capable of burning unprotected skin. With an exposure time of 1 to 6 hours, the alkaline contact dissolves tissues resulting in deep penetration. Early recognition and lavage are critical, but often the patient does not present until later in the day when shoes and socks are removed. Cement burns can range from superficial blistering to full-thickness necrosis ( Fig. 15-17 ).
Radiation burns result from exposure of tissues to ionizing radiation. Electromagnetic radiation consists of a pulse of electrical and magnetic energy of very short wavelength in the form of x-rays or gamma rays. These high-energy photons are produced in linear accelerators (x-rays) or emitted from the unstable nuclei of radioactive substances (e.g., radium, cobalt, cesium). Particulate radiation is produced by particles (e.g., electrons, protons, alpha particles, neutrons, mesons) that are capable of ionizing any tissues through which they pass.
In radiation injury, deoxyribonucleic acid (DNA) is the key recipient. The extent of the injury depends on the type of radiation, the radiation absorbed dose (rad), and the duration of exposure.
Acute changes include erythema and subsequent ulceration of varying depth that requires a surprisingly long time to heal ( Fig. 15-18 ). The late changes, however, make this a difficult injury to reconstruct. After exposure and a prolonged latency period, parenchymal fibroblasts are gradually depleted, endarteritis obliterans and excessive tissue fibrosis develop, and the potential for cellular replication decreases. This results in radiodermatitis, radioosteonecrosis, and the increased potential for local malignant transformation.
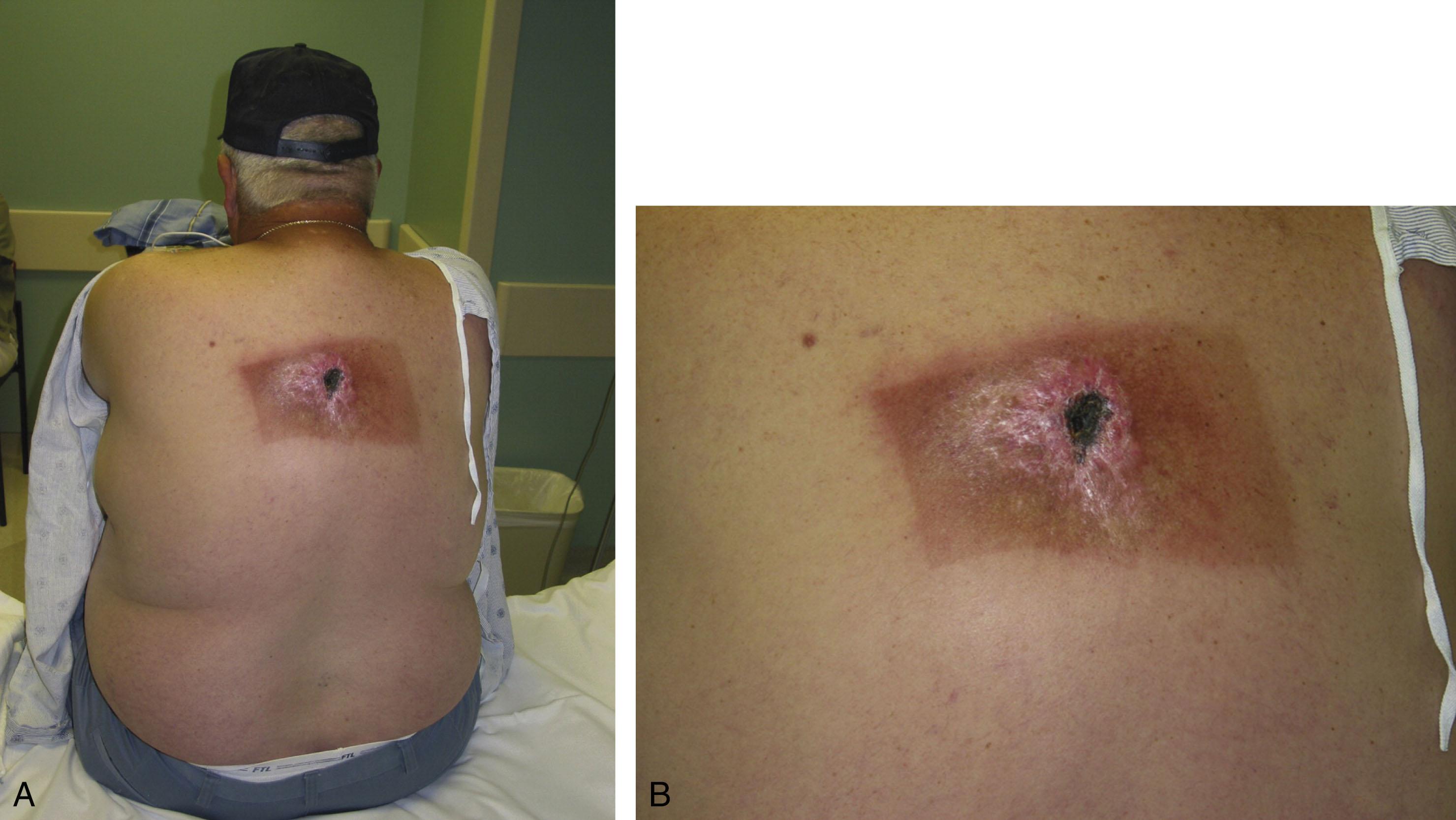
Chronic superficial ulceration secondary to radiation injury is best managed by skin grafts if the bed is well vascularized. Deeper lesions require flap coverage. Because of the high incidence of malignancy, selective biopsy should be done before surgical intervention.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here