Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Liver resection plays an increasingly important role in the management of benign as well as primary and metastatic liver tumors (see Chapters 50 , 51B , 88 , 89 , and Chapter 90, Chapter 91, Chapter 92, Chapter 93 ). Historically, the high morbidity and mortality of liver resections limited their use. However, over time, the morbidity and mortality of liver resections have decreased due to advances allowing for reduced intraoperative blood loss and preservation of liver parenchyma with the adoption of segment-oriented liver resection. Resection of individual segments or sectors of the liver rather than an entire lobe allows for maximal conservation of normal liver parenchyma while optimizing oncologic outcomes (see Chapters 102A and 102B ). Parenchymal preservation is especially important in patients with underlying hepatocellular dysfunction or limited future residual volume. Advances in understanding the liver anatomy (see Chapters 2 and 102B ) and improvements in radiographic technology (see Chapters 2 , Chapter 13, Chapter 14, Chapter 15 , and 19 ) are the basis of segmental liver surgery.
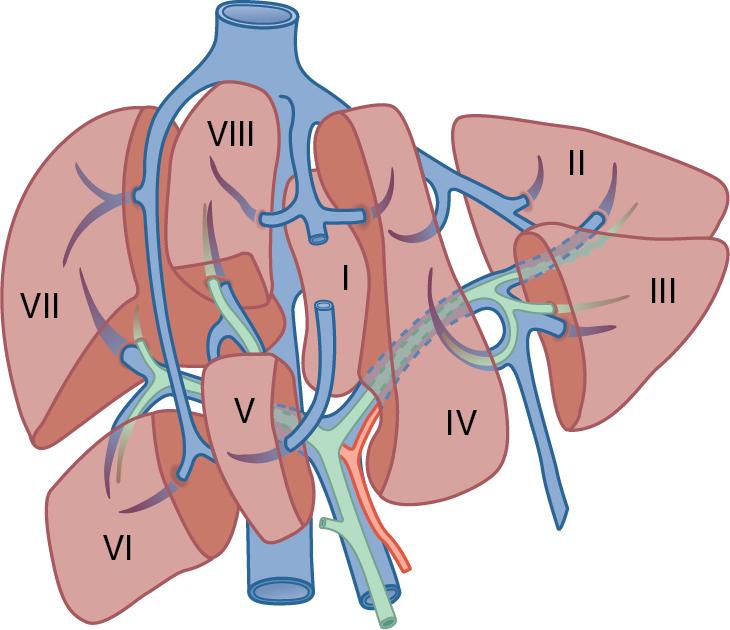
Lortat-Jacob et al. reported one of the first anatomic liver resections for cancer in 1952. Following this, Claude Couinaud solidified the current understanding of the segmental anatomy with his comprehensive descriptions of the segmental anatomy of the liver. Using vascular and biliary casts of the liver, Couinaud determined that it comprised eight segments, each with its independent vascular inflow and outflow, as well as biliary drainage ( Fig. 118B.1 ). His work supported the idea that it is possible to remove individual segments without disrupting the remaining liver. This idea that each segment could be resected independently was further confirmed by Bismuth in 1982. , These segments have evolved to become the standard for hepatic nomenclature; however, there has been significant variation in the terminology of liver anatomy between America, Europe, and Japan. The Brisbane 2000 Terminology of Hepatic Anatomy and Resection aimed to eliminate this variation and standardize terminology. This consensus proposed three orders of division: hemiliver (first-order division), section (second-order division), and segment (third-order division), which are not interchangeable, thus providing universal terminology for better communication among liver surgeons. Operations described in this chapter are those at the second- and third-order divisions ( Table 118B.1 ).
| ANATOMIC TERM | ALTERNATIVE | TERM FOR SURGICAL RESECTION | SEGMENTS |
|---|---|---|---|
| Second-Order Divisional Resections | |||
| Right anterior section | Right anterior sector | Right anterior sectionectomy | 5, 8 |
| Right posterior section | Right posterior sector | Right posterior sectionectomy | 6, 7 |
| Left medial section | Segment 4 | Segmentectomy 4 | 4 |
| Left lateral section | Left lateral segment | Left lateral sectionectomy | 2, 3 |
| Third-Order Divisional Resections | |||
| Segments 1-8 | Segmentectomy 1-8 | ||
| Atypical Resections | |||
| Bisegmentectomy 5, 6; 7, 8 | Bisegmentectomy | 5, 6; 7, 8 | |
| Segments 4, 5, 6 | Central hepatectomy | Mesohepatectomy | 4, 5, 8 |
Precise knowledge of the surgical anatomy of the liver is imperative to perform segmental liver resections (see Chapter 2 ). The first-order divisions are right liver (segments V through VIII) and left liver (segments I through IV), or hemiliver, the boundary of which lies along Cantlie’s line marked by the path of the middle hepatic vein (MHV), from the middle of the gallbladder fossa to its termination in the inferior vena cava (IVC) ( Fig. 118B.2 ). The second-order division into liver sections is based on hepatic arterial supply and biliary drainage.
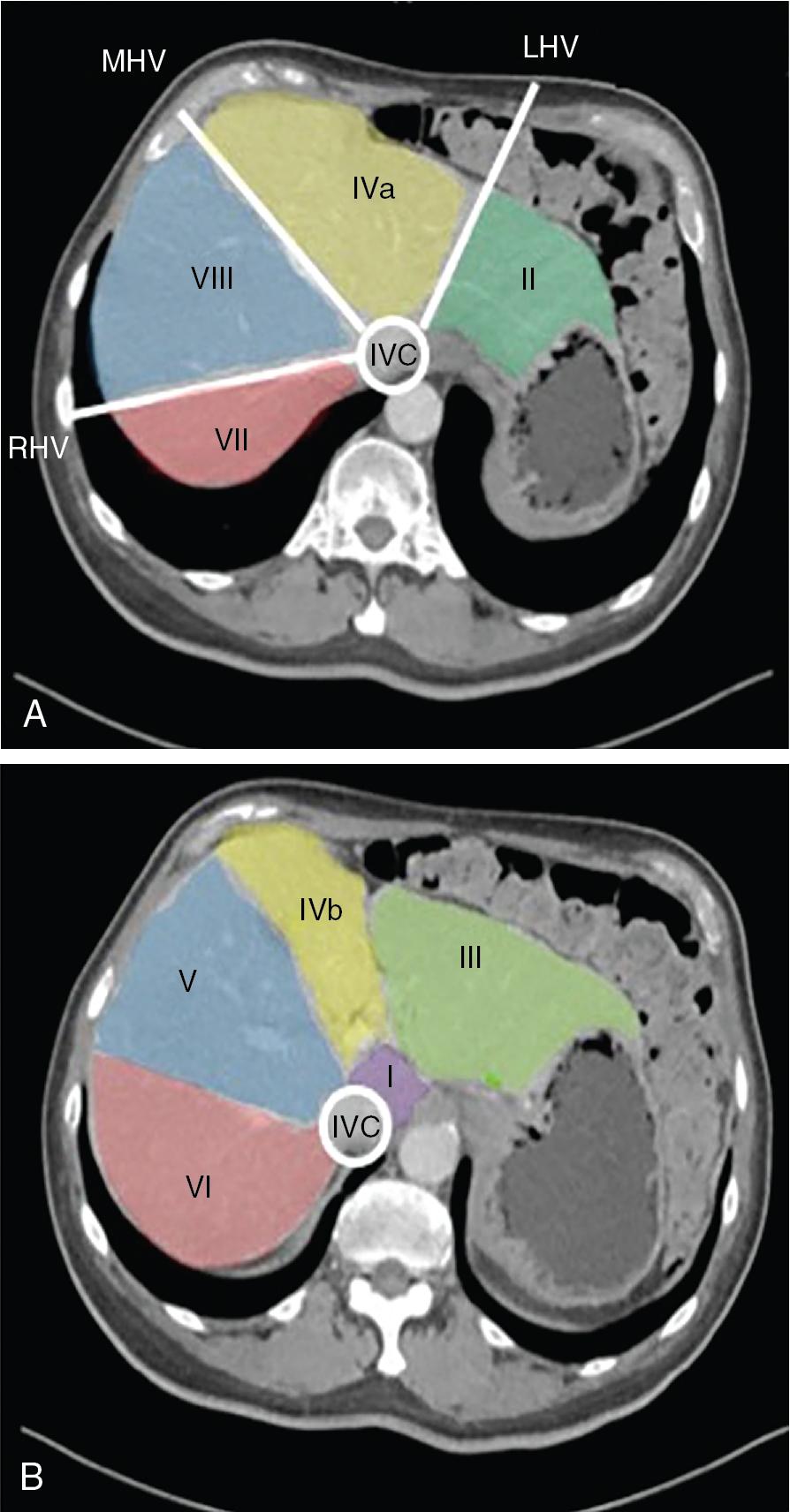
The sections are derived from the primary divisions of the major right and left portal triads. The right hemiliver is divided into sections known as the right anterior (segments V and VIII) and right posterior (segments VI and VII), separated by the right hepatic vein (RHV). The left hemiliver is divided into left lateral (segments II and III) and left medial (segments IVa and IVb) sections by the umbilical fissure and falciform ligament. The third-order division, segments I through VIII, is defined by hepatic arterial supply and biliary drainage. The axial plane is at the level of the intersection of the hepatic veins and the axial plane of the bifurcation of the portal vein. When considering a segmental resection, it is important to identify common biliary, arterial, and portal venous anomalies on preoperative imaging that may affect the technical approach ( Fig. 118B.3 ) (see Chapters 2 , 13 , and 102B ).
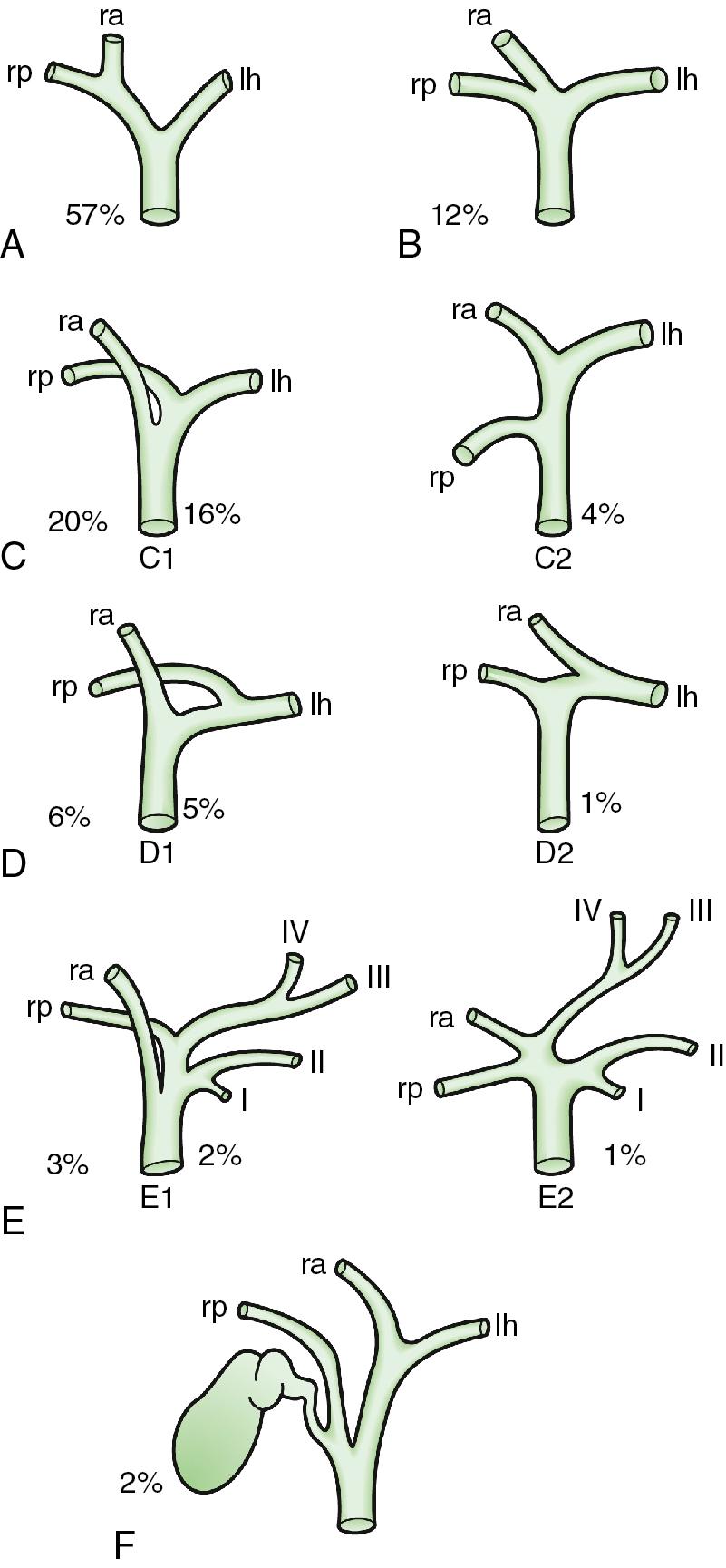
When planning a segmental resection, the surgeon must choose to follow either hepatic venous outflow or portal pedicle inflow to guide a segmental resection. It turns out the two are largely overlapping but not the same.
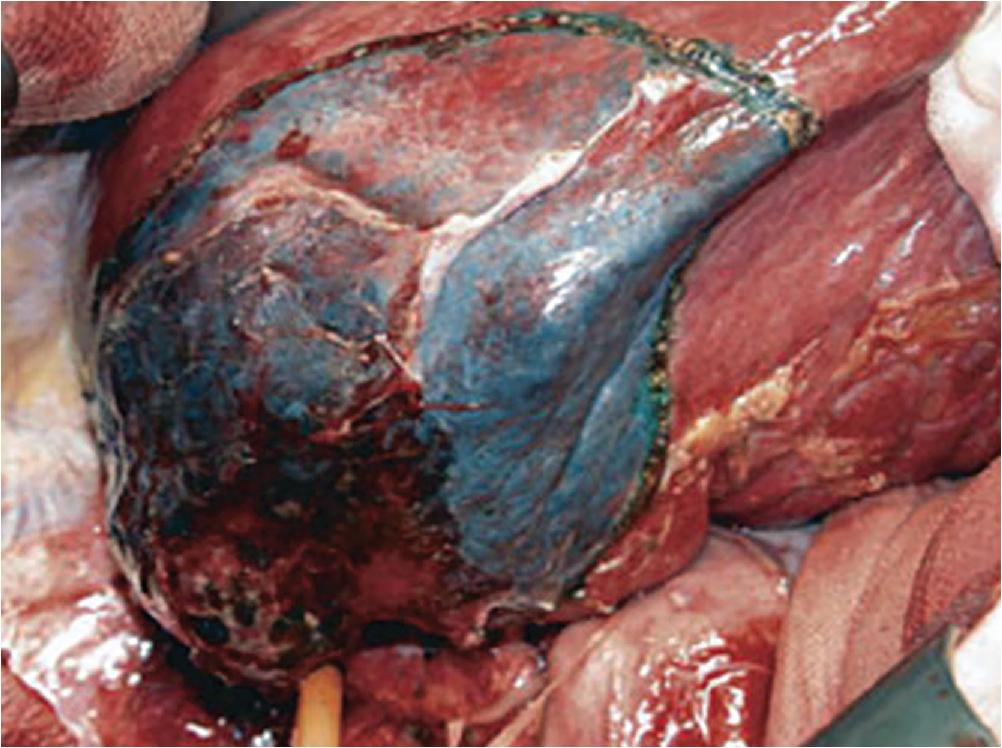
The anatomy used to guide segmental resection varies based on tumor histology and geographic region of the surgeon’s practice (see Chapters 2 and 102B ). Within Asia, hepatocellular carcinoma is the more common tumor resected by the liver surgeon, largely due to higher rates of hepatitis B infection. Hepatocellular carcinomas tend to metastasize via tumor seeding along the portal veins (see Chapter 89 ). , As such, the segments as defined by the portal inflow are resected. This may be performed with a so-called “Glissonian pedicle approach” in which the portal inflow is identified by dissecting it out and ligating it, and then parenchymal transection proceeds by following the intersegmental plane marked by the vascular color change. Portal venous dye has also been used to demarcate liver segments intended for resection ( Fig. 118B.4 ). This technique is performed by identifying the portal veins in question using intraoperative ultrasound (IOUS) (see Chapters 24 and 103 ), and then injecting them with 5 mL of 1% methylene blue or 0.8% indigo carmine. This segmental injection of dye is most easily performed when the tumor is fed by a single portal vein branch. Once the liver parenchyma is stained, it is marked off using electrocautery, and a “systematic segmentectomy” may proceed, during which the hepatic veins are exposed and preserved. This technique may be especially helpful for segments VII and VIII, which can be challenging due to their deep vascular pedicles and a relative absence of surface anatomic landmarks.
An alternative methodology for performing a segmental liver resection is to follow the major hepatic veins during transection ( Fig. 118B.5 ), ensuring complete resection of entire segments using hepatic veins as boundaries. The tumor type most commonly resected in the Western world is metastatic colorectal cancer (see Chapter 90 ). Traditionally, anatomic resections were advocated for metastatic colorectal cancer because of the high likelihood of a positive margin for wedge resections. In a classic series from Johannes Scheele, wedge resections were associated with a positive margin rate of 19%. This is likely due to the difficulty in judging margins of the tumor deep in the parenchyma. Wedge resections are also complicated by the fact that the transection line tends to fracture at the interface of the hard colorectal tumor and soft normal liver. Following the hepatic veins during a segmental resection evolved to assist the surgeon in achieving a negative margin. A recent meta-analysis of more than 2500 patients undergoing resection for colorectal liver metastases concluded that both margin status and overall survival were similar between the wedge and anatomic resections. This is likely due to expertise of surgeons at present for choosing to use either wedge or segmental resections to achieve a low rate of margin positivity.
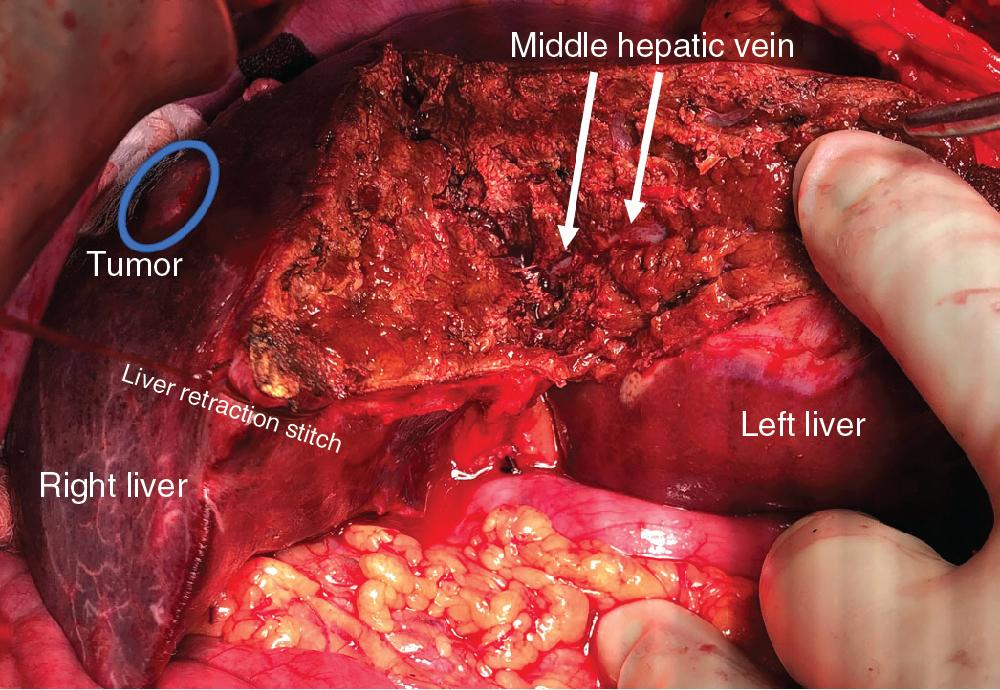
Another reason for choosing a hepatic venous based resection is the failure of IOUS to identify tumors. Many patients who have had a lot of neoadjuvant chemotherapy develop fatty liver or chemotherapy-associated steatohepatitis (CASH) (see Chapters 69 and 98 ). The liver becomes hyperechoic and the hyperechoic tumors become indistinguishable from the surrounding liver. Following the hepatic veins (see Fig. 118B.5 ) allows the liver surgeon to best avoid a positive margin or leaving cancer behind.
Symptomatic benign liver cysts or tumors of low malignant potential should be surgically addressed with liver-sparing techniques including enucleation or nonanatomic parenchymal sparing resection (see Chapters 88 and 91 ). Enucleation can be used for most benign liver lesions, including symptomatic liver cysts, hemangiomas, adenomas, cystadenomas, and focal nodular hyperplasia, or for hydatid cysts of the liver.
Well-differentiated neuroendocrine tumors separate well from the surrounding liver (see Chapter 91 ). They require almost no margin and have low local recurrence even when enucleated.
Imaging is performed both to stage patients to determine the appropriateness of resection and to allow anatomic technical planning for the resection (see Chapter 12, Chapter 13, Chapter 14, Chapter 15, Chapter 16 , 24 , and 102B ).
The first consideration when patients are being referred for segmental liver resection is the oncologic appropriateness. For most primary and metastatic disease to be resected from the liver, the best staging is afforded by computed tomography (CT) scan of the chest in conjunction with either a triphasic CT or magnetic resonance imaging (MRI) of the abdomen and pelvis. A CT of the chest even without contrast offers the most sensitive detection of pulmonary metastases. An MRI offers the best detection and characterization of liver lesions regardless of parenchymal liver disease, including fatty liver and cirrhosis. Of note, subcentimeter pulmonary nodules found on chest imaging should not necessarily preclude hepatic resection, especially in the setting of colorectal liver metastases. Depending on the primary tumor, a positron emission tomography (PET) scan may also be considered.
Diagnostic laparoscopy (see Chapter 24 ) may be performed in certain select cases where a laparotomy is planned to identify patients with subradiographic metastatic disease to avoid unnecessary incision and prolong recovery after nontherapeutic laparotomy. The tumors with the highest diagnostic yield from laparoscopy include cholangiocarcinoma and gallbladder cancer.
Cross-section scanning by CT or MRI allows direct visualization of the arterial, portal, and hepatic venous anatomy for surgical planning (see Chapter 13, Chapter 14, Chapter 15, Chapter 16 ). There are many intrahepatic portal anatomic variations (see Chapter 2 ). Typical portal anatomy is present in approximately two-thirds of patients, with variations present in one-third (see Fig. 118B.3 ). Anatomic understanding is crucial and is greatly facilitated by studying the patient’s imaging with either MRI or CT that includes portal venous and venous contrast phases.
Interpretation of imaging can be further facilitated by three-dimensional (3D) reconstructions, which can be generated from two-dimensional (2D) imaging including CT and MRI (see Chapter 13 ). Surgeons are better able to localize liver tumors on models after viewing 3D reconstructions, as compared with 2D scans. These 3D reconstructions may also enable faster surgical planning with trainees and greater concordance with staff on surgical planning. Products such as 3D Slicer exist as free open-source software. Companies and commercially available software also exist that create 3D graphical liver models, including TeraRecon (Foster City, California, USA), Materialise Mimics (Leuven, Belgium), and Visible Patient (IRCAD, Strasbourg, France).
Software providing 3D CT reconstructions of the liver allows the surgeon to view the anatomy dynamically and identify the relationship of the tumor with the segmental hepatic venous outflow and portal venous inflow. One technique for preoperative planning is to first determine which hepatic veins require resection and at what level for an adequate margin, and then to consider the portal triads that need to be included as a basis for the segmental resection.
More sophisticated software programs allow virtual surgical resection of the liver, which can be used to determine the appropriate plane of transection, taking into account the minimum resection margin and residual liver volume. Some of the available systems include the MeVIS imaging system (MeVisLab, Bremen, Germany), Myrian XP-Liver (Intrasense, Paris), Synapse 3D (Fujifilm, Tokyo), IQQA-Liver (EDDA Technology, Princeton, NJ), and Hitachi Image Processing System (Hitachi Medical Corporation, Nishinomiya, Japan). Furthermore, some of these systems have been extended to facilitate real-time, image-guided resection by coupling the preoperative images of the virtual liver to real-time liver and instrument tracking systems. These advances offer significant potential for increased precision of preoperative planning for segment-oriented liver resections and intraoperative application to facilitate image-guided surgery. However, limited data currently exist on their use in liver surgery and whether they are an improvement over current best practice.
Segmental liver resections are currently performed using open as well as minimally invasive techniques, including laparoscopy and robotics. Minimally invasive techniques are increasingly being adopted, which can lead to decreased length of hospitalization. When performing minimally invasive liver surgery, this author’s preference is to use a robotic surgical system to overcome some of the limitations of laparoscopy. ,
For patients with disease concentrated in a segment and an additional deep central lesion, a minimally invasive segmental resection with additional ablation of the deep lesion can be considered to avoid a major or extended hepatectomy in select cases. The following technical considerations, however, will focus on an open operative approach, which still often is necessary. Indications and technical considerations for minimally invasive liver surgery are discussed in further detail elsewhere (see Chapter 127 ).
For open segmental liver resections, the patient should be placed in the supine position. The authors prefer to have arms tucked. Arms are tucked for two reasons. The first is that this gives the greatest flexibility for table-mounted retractor placement and assistant positioning. The second is that these operations may take several hours to complete, and leaving the arms out may produce perioperative upper extremity nerve traction injuries through arm hyperabduction. In general, a large-bore IV is placed in each arm, and the arterial line is also placed when necessary before tucking the arms. This author rarely uses central lines and will monitor central venous pressure (CVP) for low CVP anesthesia by liver ultrasound inspection of hepatic veins (see Chapters 24 and 25 ). The patient is placed in a Trendelenburg position to allow for maximum venous return to the heart in the setting of low venous vascular volume to optimize cardiac output while keeping CVP low.
The abdomen is prepped from nipples to pubis, and laterally down to the surgical table. Drapes should be placed to enable maximum exposure to the abdomen. Lights are positioned so that one is directly over the operative field, and a second is positioned angled over the right shoulder of the primary surgeon and between the surgeon and the assistant. Two suctions should be available so that one is always available to both the surgeon and the assistant.
For an open segment-oriented liver resection, operative exposure can be achieved through a variety of incisions, which should take into account the patient’s body habitus and tumor location. The choice of incision is almost always determined in order of importance by (1) body habitus, (2) location of the liver, (3) location of the lesion, and (4) history of prior incisions. In a thin, tall patient, almost any resection can be performed through a long midline incision. In a patient with a high body mass index or a liver high in a barreled chest, a transverse extension of the incision is almost always needed.
A midline incision is usually adequate for left-sided lesions. This incision affords the option to extend the incision laterally at the level of the umbilicus to a point midway between the costal margin and anterior superior iliac spine (L-incision). Other common incisions include the right subcostal with midline extension cephalad (“hockey stick”) or a bilateral subcostal incision with midline extension cephalad (“Mercedes” incision) or without (“Chevron” incision). Occasionally for posterior lesions, a thoracoabdominal incision may be required, although it can often be avoided using a transverse extension of a midline incision with appropriate costal margin retraction. Of note, trifurcated incisions such as the chevron incision are associated with higher ascitic leak and hernia formation.
Excellent retraction cephalad is essential, thus the best retractors for liver surgery generally have a crossbar across the patient’s chest. The authors’ preference is a Thompson retractor (Thompson Surgical Instruments, Traverse City, MI). A bilateral crossbar (hinged or unhinged) is positioned approximately 15 cm above the skin and 10 cm superior to the apex of the incision. A single crossbar is then positioned on the patient’s right, with the angle of the bar toward the patient’s body and the short end of the bar toward the patient’s head. Subcostal retractors connected to a micro-adjustable clip-on ratchet is used on the right and left costal margins. Retractors positioned on the left and right inferior aspects of the wound provide lateral retraction. If a long midline incision is chosen, a Balfour retractor may assist in even-tensioned lateral retraction.
Other suitable retractors include the Omni-Tract (Omni-Tract Surgical, St. Paul, MN) or the Iron Intern (Automated Medical Products, Edison, NJ). The Bookwalter (Codman, Raynham, MA) retractor may be adequate, especially for left-sided segment-oriented resection, but we find that it provides inadequate exposure for right posterior lesions.
If diagnostic laparoscopy is not performed (see Chapter 24 ), a minimal incision should be made first and systematic inspection of the entire abdominal cavity performed to check for metastatic disease in (1) peritoneal lining, (2) diaphragms, (3) lesser omentum, (4) portal-caval lymph nodes, (5) hepatic artery lymph nodes, and (6) celiac axis lymph nodes. If possible, the lower abdomen should also be inspected, but this may be difficult in patients who have had previous surgery. Extensive lysis of adhesions to inspect the pelvis is not necessary and reliance on adequate preoperative imaging is acceptable.
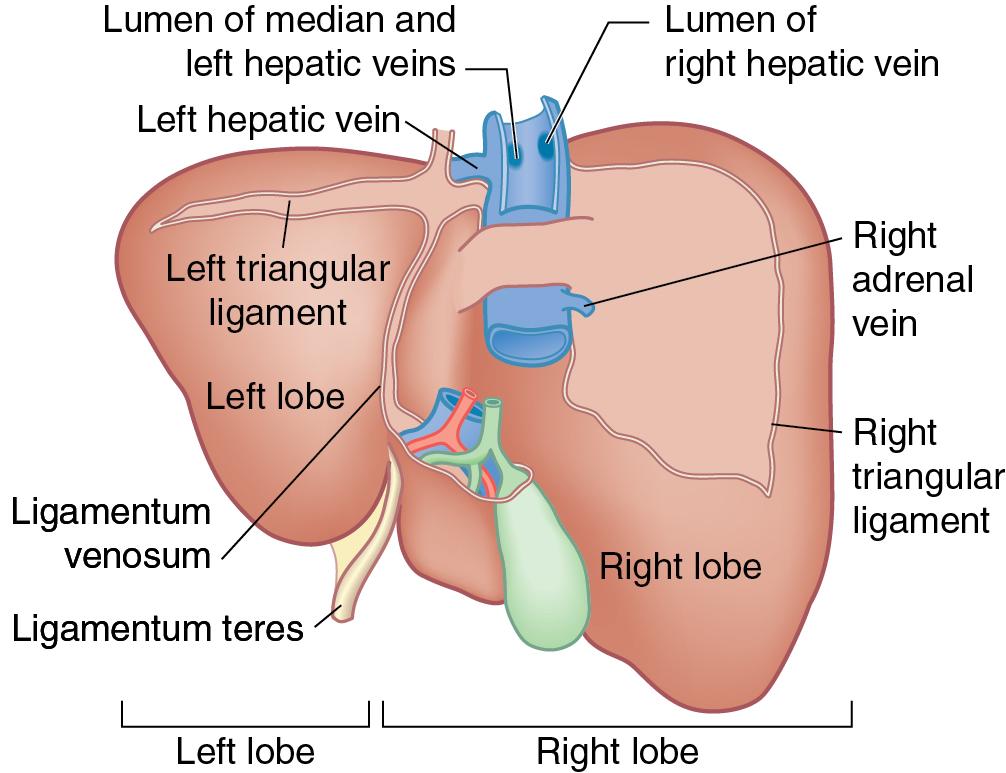
In open hepatectomy, after the laparotomy and general inspection of the abdomen, the liver is mobilized by transection of the ligamentum teres and division of the falciform ligament cephalad to the subdiaphragmatic inferior vena cava (IVC) (see Chapter 2 ). A long silk tie should be left on the ligamentum teres to use as a handle for manipulation of the liver or fixed cephalad to the retractor to provide additional exposure to the undersurface of the liver and hepatic hilum. The falciform ligament is transected using Bovie electrocautery to identify the origins of the hepatic veins. At this point, visual and manual palpation inspection of the liver should be performed. The bare area in the lesser omentum should be divided to expose the caudate lobe (segment I) for inspection and palpation. Care should be taken during this step to identify and avoid an anomalous left hepatic artery, arising from the left gastric artery. IOUS may be used to identify any lesions that are not seen on preoperative cross-sectional imaging that would prevent proceeding with resection or change the operative plan. See Chapter 103 for more details on IOUS.
Mobilization of the left and right lobes may be performed either before or after pedicular dissection, which is at the discretion of the surgeon. Mobilization of the left liver requires division of the left triangular ligament. Laparotomy pads should be placed posterior to segment II to protect the esophagus, cardia of the stomach, and spleen during division of the triangular ligament. The superior vena cava (SVC) and left hepatic vein should become visible. Care should be taken to identify the path of the left phrenic vein to protect it.
Mobilization of the right liver requires division of the falciform ligament to expose the extrahepatic right hepatic vein. Early identification of the vein allows the surgeon to avoid injury to it during lateral mobilization. Next, division of the right triangular ligament is performed ( Fig. 118B.6 ). The peritoneal reflection at the edge of the triangular ligament is incised laterally and division of the anterior layer of the coronary ligament, as it reflects from the diaphragm onto segment VIII, and the posterior layer of the coronary ligament that reflects onto segment VI is developed. The liver is mobilized off the right hemidiaphragm, thereby exposing the bare area of segments VI and VII. The right adrenal gland is separated from segment VI, and the retrohepatic IVC is identified. This allows the liver to be rotated to the left and access the posterior segments.
If the tumor is noted to be adherent to the diaphragm, it can be separated if there is an adequate transection plane or the area resected, and diaphragm repaired primarily. It is particularly important to be careful during dissection of the tendinous portion of the diaphragm. Here, the diaphragm is only one layer thick, and errant dissection especially with the Bovie will produce a pneumothorax. Pneumothorax may lead to contamination of the chest with tumor in cancer cases and bacteria in contaminated cases. The diaphragm could also billow downwards obscuring the operative field.
Thus all defects in the diaphragm should be repaired immediately if possible. Diaphragmatic repair is performed using nonabsorbable running sutures. Typically, the author sutures each corner and runs the two sutures to the middle where they are tied. Before tying the suture, a red rubber catheter is threaded through the defect, and the anesthesiologist is asked to perform a Valsalva maneuver. The rubber catheter is then quickly pulled out of the defect as the suture is being tied down.
If the segments to be resected contain a tumor in proximity to the vena cava, the dissection must proceed posteriorly and medially until the IVC is visualized and ultimately completely exposed. A pair of closed Metzenbaum scissors can be used to gently push toward the vena cava to create this plane. The small venous branches draining from the back of the liver to the vena cava must then be divided ( Fig. 118B.7 ). Failure to divide all small venous attachments to the IVC may result in future tearing of these veins and hemorrhage that is difficult to control. Ligation of these vessels can be performed using metal clips, silk ties, or a bipolar vessel sealer. The authors prefer ties over clips in the region of the major hepatic veins since clips may interfere with stapled transections of the hepatic veins and may lead to misfiring of the stapler ( Fig. 118B.8 ).
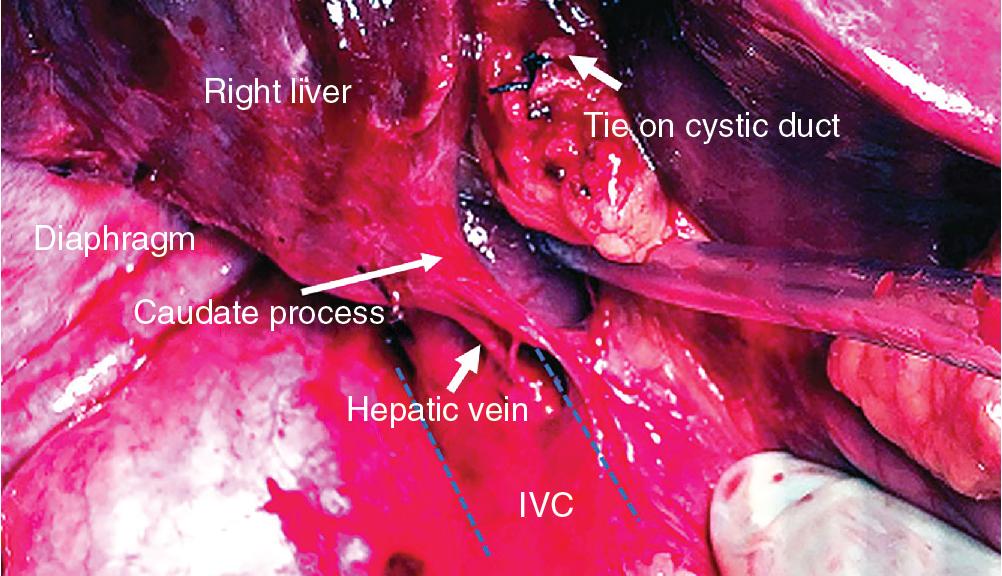
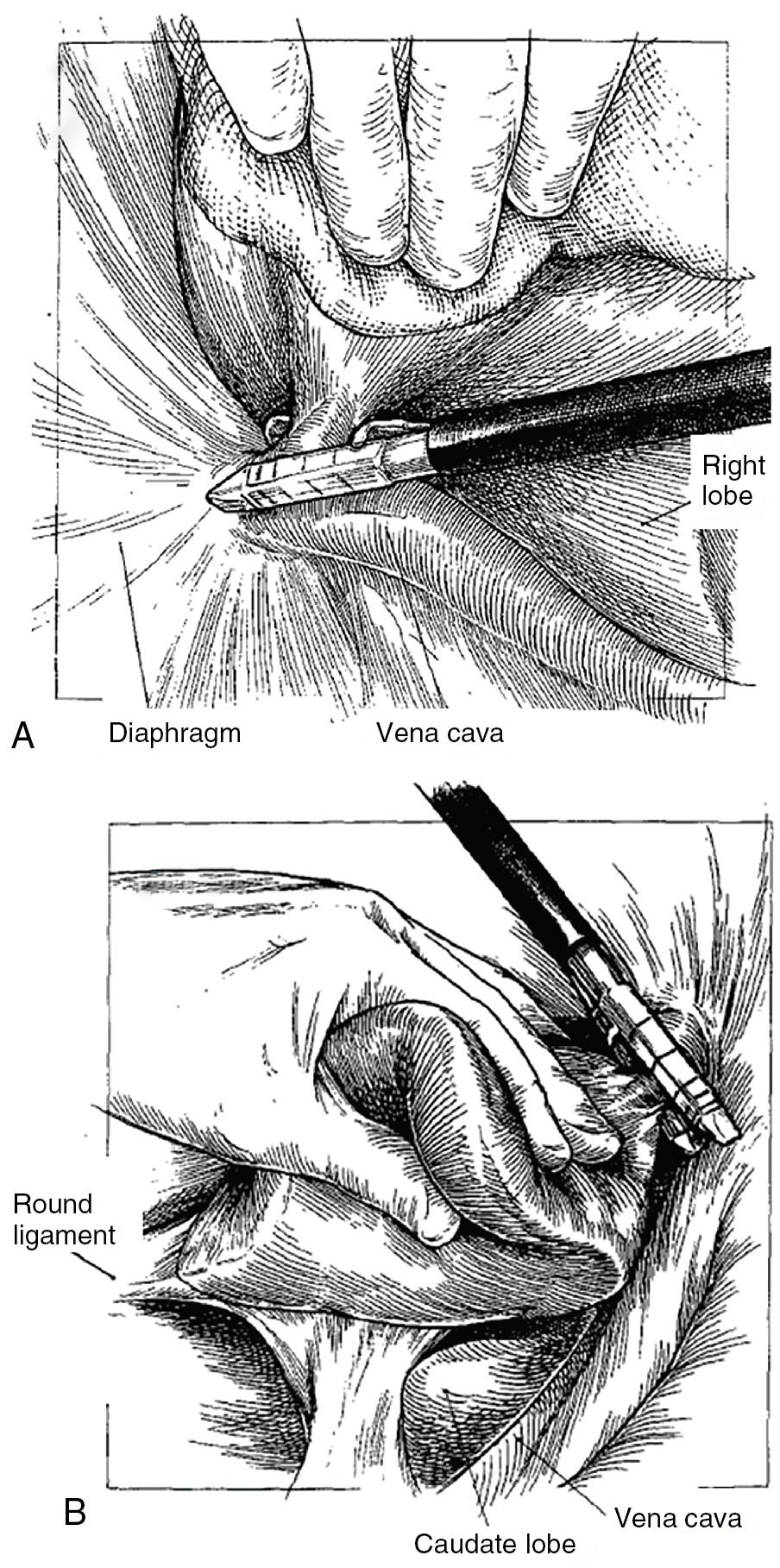
Special mention must be made of the perforator veins behind the caudate process (see Fig. 118B.7 ). This is the bridge of liver tissue belonging to segment VI that bridges this segment and the caudate lobe. If a large inferior right hepatic vein is present (as it is in 21% of patients, often draining segment VI), it usually lies behind the caudate process (see Chapter 2 ). This can be ligated during mobilization, or one may elect to leave it in place as the primary drainage for the remaining right-sided parenchyma. There may also be a tongue of liver tissue that passes from the back of the right lobe of the liver around the vena cava to the caudate on the left. In others, this structure may only be ligamentous and has been called the dorsal ligament, Makuuchi ligament, hepatocaval ligament, or the caudate-caval ligament. This tissue may contain vasculature and can be divided using a stapler. To do this, the liver is retracted medially, and a stapler is passed from below, parallel to the inferior vena cava.
The left liver mobilization begins by dividing the lesser omentum along the inferior edge of segment III. This usually is avascular, but if present, a replaced or accessory left hepatic artery will run in this plane. A laparotomy pad is placed under segment II, with a portion extending beyond the edge of the liver. This is to help protect the esophagus and stomach, which are just to the left of the IVC. The left hand is used to retract the left liver downward, and electrocautery is used to divide the loose attachments of the left triangular ligament in a lateral to medial fashion until the white of the laparotomy pad is visualized. As dissection proceeds medially, attention should be paid to the takeoff of the left hepatic vein, which again can be identified by tracing the drainage of the left phrenic vein on the diaphragm.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here