Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
A nodule or diffuse enlargement of the salivary glands may be caused by a cystic lesion, an inflammation, a degenerative process, a benign or malignant neoplasm. For adequate management, the exact nature of the process must be revealed, which can be done only by microscopic evaluation. Incisional or cutting needle biopsies have not been accepted as the procedure of choice for obtaining tissue for diagnostic purposes because of various complications. Fine-needle aspiration (FNA), however, poses a negligible risk of complications, and in medical centers where the technique is routinely used, approximately a third of all patients with salivary gland lesions are spared surgery. Of equal importance is that a FNA diagnosis allows an individualized treatment plan when surgery is required. The clinical impact of FNA has been demonstrated in several studies.
The technique of aspiration does not differ from that used for other superficial sites. The FNA procedure using a 23- or 25-gauge needle is rarely painful to the degree that it prevents successful sampling. Rarely, a small hematoma may occur or some blood may be seen in the saliva after aspiration.
Slides of aspirates are preferably made for both air-drying and alcohol fixation. The slides are stained with May–Grünwald–Giemsa and Papanicolaou stains, respectively. Both stains are helpful because each method places emphasis on different features.
Immunophenotyping can be of value in subtyping salivary gland tumors and is essential in differentiating reactive from neoplastic lymphoid infiltrates. Cytospin material performs excellently in such studies and the technique for procuring such material has been described.
Air-dried and formalin-fixed smears can be used for analysis of nuclear antigens such as cell proliferation after staining with a Ki-67 antibody.
A representative set of slides produced from FNA biopsy of a salivary gland tumor is shown in Fig. 22-1 .
The parotid, the submandibular, and the sublingual glands compose the major salivary glands. The parotid glands are of ectodermal origin, and their secretion is serous and drains through Stensen's duct into the oral cavity. The submandibular glands are of endodermal derivation and secrete a mixed but predominantly serous substance. They drain via Wharton's duct at the sublingual caruncle close to the frenulum of the tongue. The sublingual glands lie in the floor of the mouth adjacent to the midline and produce a mixed, primarily mucous secretion.
Minor salivary glands are non-encapsulated aggregates of mucous or mixed mucous–serous glands located superficially over the lips, throughout the oral cavity, nasopharynx, sinuses, trachea, and bronchi.
The glands are organized into lobules. Each lobule consists of a cluster of acini around a terminal duct system. The acini are made up of pyramid-shaped epithelial cells that have eccentric round nuclei ( Fig. 22-2 ). The cytoplasm is abundant, finely granular, and acidophilic in serous glands and clear in mucus-secreting glands. Flat myoepithelial cells with elongated nuclei form an outer layer around each acinus. From the lobules, the secretion is conveyed via interlobular ducts to the main secretory ducts. The smallest excretory ducts are lined by cuboidal cells, whereas those lining larger ducts are tall and columnar. Near the orifice in the oral cavity, the ducts are lined by stratified squamous epithelium. A layer of myoepithelial cells is present between the lining epithelial cells and the basement membrane.
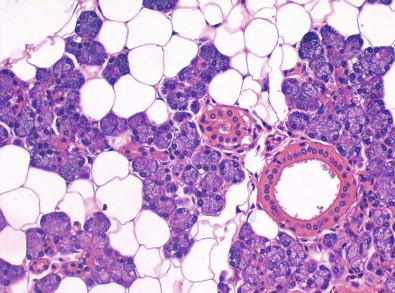
With increasing age, the cells of acini and ducts occasionally transform into oncocytes. An oncocytic cell is a large epithelial cell with distinct cell borders, eosinophilic finely granular cytoplasm, and a round nucleus, often with a central prominent nucleolus.
The parotid glands normally contain small lymphoid aggregates and one or more nodes. Salivary ducts and acini have also been found in lymph nodes outside the capsule of parotid glands.
Aspiration of normal or near-normal salivary glands usually yields only a small amount of epithelial cells. Normal structures are often seen as “contamination” in aspirates from pathologic lesions. The normal structures are viewed as basically acinar cells in well-preserved cohesive ball-like formations ( Fig. 22-3 ) and as ductal cells in monolayered sheets or small tubuli.
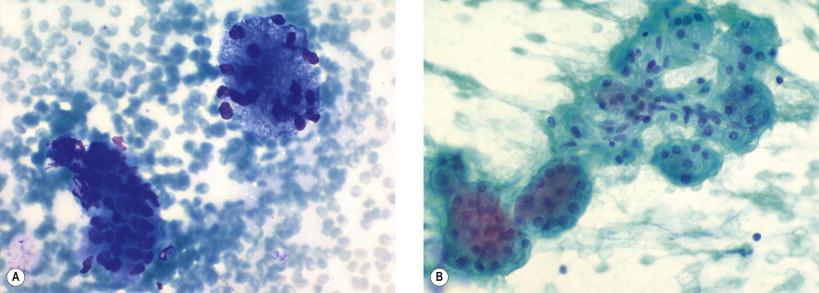
The acinar cells have a central or eccentric, small rounded nucleus with a small nucleolus. The cytoplasm is abundant, granular, and rather foamy ( Fig. 22-3 ). Bare cell nuclei are usually observed in the background. Terminal secretory ducts appear as short tubular structures, and larger ducts appear as flat clusters or sheets of cuboidal or columnar cells with evenly spaced monomorphous nuclei ( Fig. 22-3 ). Myoepithelial cells are only occasionally noted as small dark nuclei superimposed in the clusters.
Benign and malignant salivary gland lesions may have a cystic component. Cystic aspirates therefore cause a special problem because the cyst fluid alone does not allow a morphologic evaluation. The most common cyst is a ductal retention cyst due to obstruction (sialolithiasis), and it contains watery fluid or, in cases of inflamed cysts, a cloudy fluid.
Retention cysts of the major salivary glands that are deep seated are, at palpation, felt to be rather firm. They are therefore likely to be diagnosed clinically as solid neoplasms; however, FNA readily discloses the nature of the lesion. In such cases, a clinical follow-up after a few weeks should suffice. If the cyst recurs, it should be reaspirated to attempt a cure and to obtain a further sample for cytologic evaluation.
If a residual mass persists, however, the aspiration should be repeated because some benign and malignant salivary gland tumors may have both a solid and a cystic component. An initial sampling from a cystic portion of the tumor may therefore be misleading, and a repeat biopsy should be performed from the walls of the cyst to help establish a diagnosis.
Branchial cleft cysts can occasionally appear in the anterior portion of a parotid gland and on clinical examination may be mistaken for a salivary gland lesion.
Small superficial cysts are often encountered in minor salivary glands of the lips and the oral mucosa. They are in general accurately diagnosed clinically. At aspiration, such cysts yield a clear mucous fluid and leave no residual mass.
An aspirate from a benign retention cyst is usually clear and has a watery to viscous consistency. It can, however, be cloudy and yellowish when secondary inflammation has taken place. The smear usually contains only a few histiocytes and degenerative epithelial cells. Epithelial cells of cuboidal, columnar, or squamous type can be found together with inflammatory cells and histiocytes.
If the palpatory findings normalize after aspiration and the smear contains only a few phagocytes or occasional epithelial cells, the lesion is probably a retention cyst.
Water-like or viscous mucoid aspirate
Histiocytes and other inflammatory cells
Epithelial cells – cuboidal and squamoid.
Sialoadenosis is a benign, non-inflammatory, non-neoplastic enlargement of one or both parotid glands. The cause of sialoadenosis is unknown but may be associated with diabetes, liver cirrhosis, or nutritional and hormonal disturbances.
FNA demonstrates cellular smears of normal or slightly enlarged acinar cells.
Nodular oncocytic hyperplasia refers to a condition with more than one nodule, whereas oncocytoma refers to a single neoplasm. The distinction between the two may at times be arbitrary. Nodular oncocytic hyperplasia is often combined with significant clear cell change, which may create a similarity to acinic cell carcinoma.
Aspirates contain cohesive clusters of cells showing a transition between oncocytes and acinic cells, with abundant finely vacuolated or finely granular cytoplasm. Cells from an acinic cell carcinoma tend to have more prominent nucleoli and occasionally also a red granulation (May–Grünwald–Giemsa) of cytoplasm. The cell patterns, however, can in extreme cases overlap to a certain extent but should in a majority of cases cause no diagnostic problems.
In young individuals, viral infections have a predilection for involving the parotid glands, which become diffusely enlarged. The diagnosis is usually obvious on clinical grounds and rarely requires morphologic confirmation.
Acute bacterial sialoadenitis is characterized by diffuse painful enlargement of the salivary gland. It occurs with equal frequency in the parotid and submandibular salivary glands, mostly in elderly patients. Poor oral hygiene with dehydration, sialolithiasis, and impaired immune response are generally thought to be immediate or contributory causes of this condition. Acute sialoadenitis with or without abscess formation can clinically simulate a malignant tumor.
Aspirates from acute inflammatory salivary gland lesions contain fibrin, cellular debris, neutrophils, lymphocytes, and histiocytes ( Fig. 22-4 ). In cases with sialolithiasis, calculi may also be encountered.
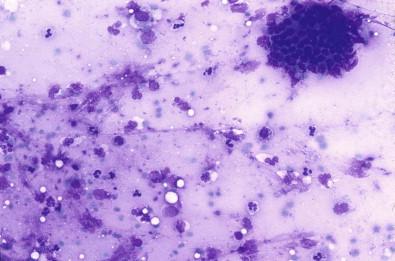
Debris, fibrin
Neutrophils, macrophages, histiocytes
Epithelial cells of both acinar and ductal type.
Nonspecific chronic sialoadenitis is often secondary to stricture and obstruction of the ducts. In elderly patients, decreased secretion of saliva and retrograde infections may result in a gradual diffuse enlargement of the salivary glands. It is frequently seen after radiotherapy of the oral cavity. Symptoms include recurrent mild pain and diffuse swelling related to eating. In more chronic cases of sialoadenitis, the gland becomes fibrotic and hard, with secondary atrophy of acini and proliferation of ductal epithelium. In obstructive sialoadenitis caused by calculi, the epithelium may undergo squamous and mucoid metaplasia and the ducts become dilated by mucous material.
Aspirates from non-obstructive sialoadenitis contain sparse cellular elements. The acinic cells are atrophic and may barely be seen in the smears. The duct cells that predominate are often small and tightly packed in clusters. Scattered fibroblasts, lymphocytes, and granulocytes may also be observed.
Aspirates from the obstructive type of chronic sialoadenitis may contain mucous, ductal cells with evidence of squamous metaplasia, and cylindric cells, some of which may even have cilia. In addition, macrophages with vacuolated cytoplasm imitating mucus-producing cells, lymphocytes, granulocytes, and crystals of various shapes may be observed ( Fig. 22-5 ).
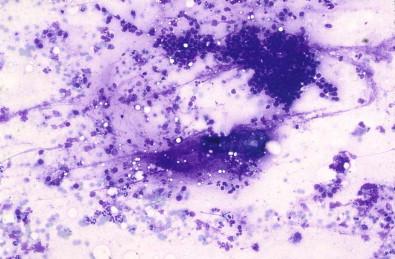
Mucoid background (sometimes)
Ductal epithelium in clusters and sheets
Scanty acinar groups with atrophy of epithelium
Occasional fibroblasts, lymphocytes, neutrophils, and macrophages.
Small lymph nodes are often found within the parotid gland. When such lymph nodes enlarge, they can clinically be difficult to differentiate from neoplasms. FNA can identify the lymphoid nature of the lesion and often differentiates among reactive lymph node hyperplasia, nonspecific lymphadenitis, granulomatous lymphadenitis, and lymphoma. A conclusive categorization of the lymphoid population will, however, require an immunologic analysis. The cytology pertaining to intraparotid lymph nodes does not differ from that of lymph nodes from other sites and is described elsewhere.
The benign lymphoepithelial lesion is an autoimmune disease also known as Sjögren's syndrome. It is characterized by insidious swelling of one or several salivary and lacrimal glands ( Fig. 22-6 ). The condition may result in decreased secretion from the glands, causing xerostomia and keratoconjunctivitis.

The aspirates contain lymphoid cells with the same morphologic variability noted in reactive hyperplasia of lymph nodes. Small mature lymphocytes are admixed with follicular center cells, plasma cells, and histiocytes ( Fig. 22-7 ). This appearance corresponds to the histology of such affected glands, which have abundant lymphoid tissue with large germinal centers. The tissue contains scattered solid epithelial rests, which represent myoepithelial proliferations, but cytologically few, if any, epithelial or epithelioid cells are encountered.
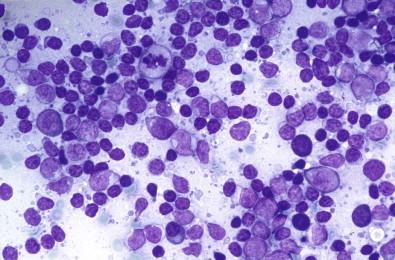
A careful cytologic evaluation of the lymphoid infiltrate is needed to preclude a low-grade lymphoma. In fact, an immunocytochemical evaluation of the lymphoid cells is the only possible way to conclusively differentiate a reactive from a neoplastic infiltrate.
Several investigators have reported the possibility of transition from benign lymphoepithelial lesions to malignant lymphomas. Low-grade malignant lymphomas can have either a monotonous or a mixed cell pattern, whereas smears from high-grade lymphomas are dominated by blasts. A cytologic diagnosis of malignant lymphoma should always be confirmed using immunocytochemistry to show that the lymphoid cells are monoclonal.
Mixed population of lymphoid cells
Epithelial cells, mostly ductal, sparse
Cystic fluid may dominate.
Aspirates from a Warthin's tumor can in rare cases be dominated by lymphoid elements. An abundance of lymphocytes should, however, not cause diagnostic problems, because the aspirates in general contain some sheets and clusters of oncocytic cells. A detailed description of Warthin's will follow.
The incidence of salivary gland neoplasms is approximately 2 per 100 000 persons annually. Tumors are more frequent in the parotid glands than in the other salivary glands, whether major or minor. A 4 : 1 ratio is found between benign and malignant tumors. In addition, enlargement of the salivary glands is more often due to a non-neoplastic disorder than to the presence of neoplasms.
Pleomorphic adenomas constitute about 75% of major salivary gland tumors. Mixed tumors can occur in minor salivary glands as well. The tumor usually is a well-circumscribed, solitary, painless, and slow-growing nodule. Most of the tumors arise in the superficial portion of the parotid gland ( Fig. 22-8A ).
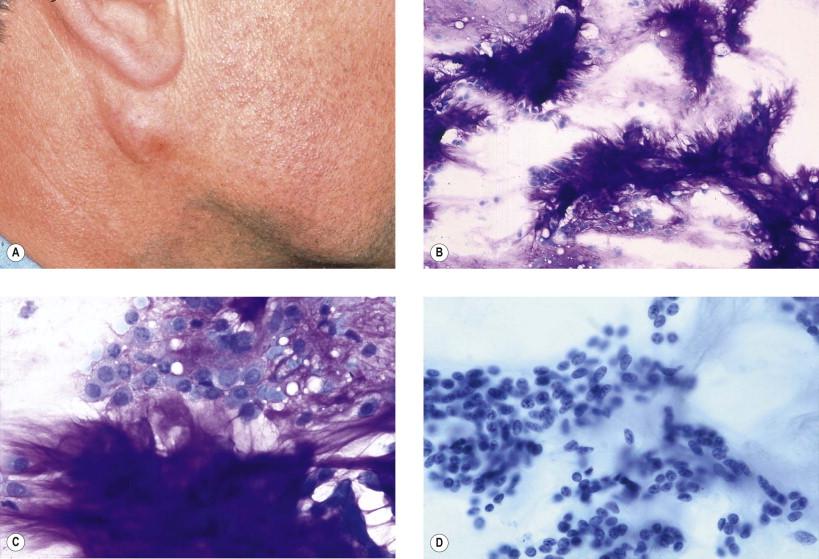
Histologically, pleomorphic adenomas have a capsule that often is incomplete, a fact that explains recurrences after apparent complete excision. The tumors are composed of epithelial and myoepithelial elements and a sparse to abundant stroma. The epithelial component may have a trabecular, glandular, solid, or adenoid cystic (cylindromatous) pattern. Foci of squamous metaplasia, oncocytic changes, mucus production, and cells with sebaceous differentiation may be encountered within the same tumor. The stromal component can be myxoid, chondroid, or myxochondroid.
The ratio of epithelial-to-mesenchymal components varies widely. Some pleomorphic adenomas may be quite cellular, composed almost entirely of epithelial or spindly myoepithelial cells, whereas others have predominantly mesenchymal components with very few epithelial cells. The variability in histologic pattern is typically reflected in FNA.
Aspirates from pleomorphic adenomas have a thick, gelatinous consistency. In May–Grünwald–Giemsa-stained smears, the mesenchymal fragments appear as a red to dark purple, fibrillar, and chondromyxoid substance ( Fig. 22-8B,C ). In Papanicolaou-stained smears, the mesenchymal substance is gray to pale pink and usually has a distinct fibrillar structure ( Fig. 22-8D ). Spindled or plasmacytoid myoepithelial cells dominate and have a moderate to rich often homogeneous cytoplasm ( Fig. 22-9 ).
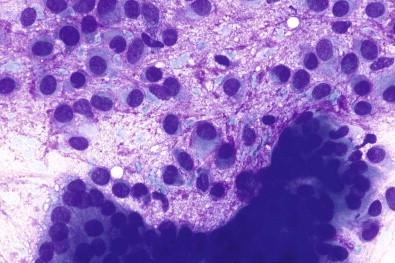
The second cell type is epithelial and forms small tubules or sheets ( Fig. 22-9 ). This cell type is not seen in smears from all pleomorphic adenomas. They are seen dispersed or in groups within or separated from the stromal substance. The epithelial and myoepithelial cells can be difficult to evaluate because the May–Grünwald–Giemsa stain penetrates the chondromyxoid tissue poorly, giving only a pale blue color to the nuclei.
Fibrillary, and/chondromyxoid stroma
Myoepithelial cells of ovoid to plasmacytoid type
Uniform, medium-sized epithelial cells, forming ducts or small sheets
Clusters and single cells gradually merging with the mucoid mesenchymal elements
It is not rare to find some large myoepithelial cells in an otherwise typical aspirate of pleomorphic adenoma ( Fig. 22-10 ). Such cells can cause concern about malignancy. In addition, cystic changes can also lead to diagnostic difficulties since such lesions may show atypical squamous cells and even mucus-producing cells ( Fig. 22-11 ). However, in such cases any features suggestive of pleomorphic adenoma should overrule occasional findings that may be suggestive of malignancy. This is the “dogma of pleomorphic adenoma,” which is practiced in our clinic.
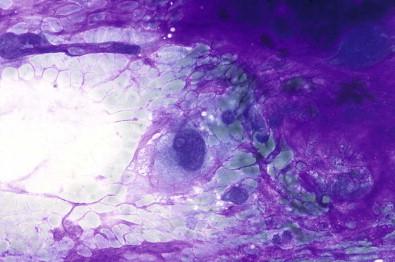
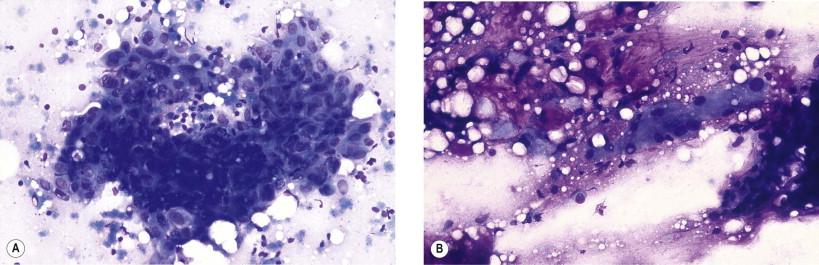
Distinguishing between pleomorphic adenoma and adenoid cystic carcinoma can in rare cases be difficult. Aspirates from pleomorphic adenomas may also contain structures which mimic adenoid cystic carcinoma, such as homogeneous globules surrounded by monomorphous epithelial cells ( Fig. 22-12A ). Sampling performed from different parts of the tumor should reduce this risk of diagnostic error. Immunophenotyping will, in difficult cases, allow a correct diagnosis. Some pleomorphic adenomas are to such an extent dominated by a mature chondroid substance that a true chondroid neoplasm cannot be excluded ( Fig. 22-12B ).
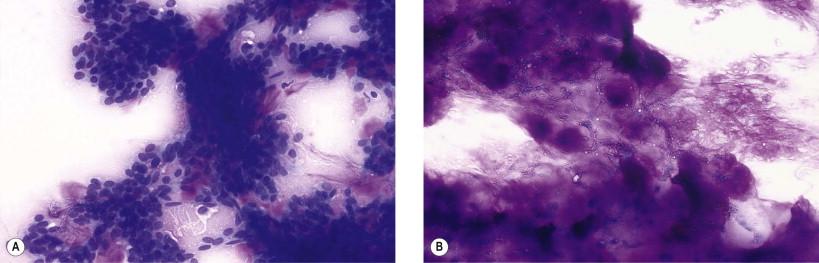
Malignant transformation within a pleomorphic adenoma (carcinoma ex pleomorphic adenoma) can occur. It is then most often an invasive, undifferentiated carcinoma with markedly abnormal malignant cells and will thus not cause diagnostic problems. More differentiated malignant tumors such as adenoid cystic carcinoma and mucoepidermoid carcinoma have also been reported.
This rare benign tumor is found in both major and minor salivary glands. All ages seem to be affected.
Histologically, the tumor is composed of a single cell type of myoepithelial origin often of plasmacytoid or spindle cell type. Epithelial cells as found in many pleomorphic adenomas are rare. There is no chondromyxoid stroma but a mucoid or collagenous substance may be found.
Monomorphic myoepithelial cells of either plasmacytoid or spindle cell type are abundant ( Fig. 22-13 ). Binucleated cells and cells with pseudonucleoli can be found. Mitoses are rare. There is a sparse collagenous stroma and/or mucoid ground substance.
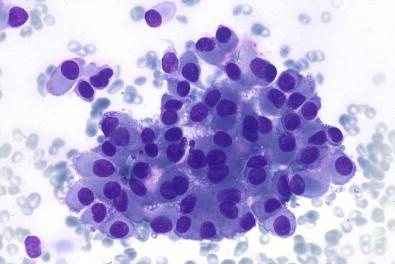
Cohesive fragments of tumor cells as well as dissociated cells
Small collagenous fragments, sometimes mucoid substance
Monomorphous plasmacytoid or spindle cells.
This tumor can be difficult to distinguish from a pleomorphic adenoma but the absence of chondromyxoid stroma favors myoepithelioma. Their separation does, however, not seem very important since their treatment is the same.
Adenolymphomas usually occur within the lower portion of the parotid gland and are thought to develop from salivary gland inclusions in lymph nodes adjacent to the lower pole of the parotid gland ( Fig. 22-14 ). They constitute approximately 5% of all parotid tumors and are rarely found outside the parotid area. Adenolymphomas are bilateral in 5–10% of cases. Previously, these tumors were said to occur mainly in men; however, later reports of tumor incidence cite a male-to-female ratio of about 1.4 : 1. The majority of patients are middle-aged to elderly.
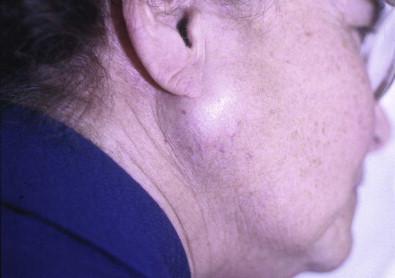
Physical examination usually reveals a soft, doughy, painless swelling with poorly defined borders. The tumors vary in size. Without treatment, they tend to reach a certain size of about 3–5 cm and then stop growing. Adenolymphomas are most often partly cystic but solid variants occur.
Histologically, the cystic spaces are lined by a double-layered oncocytic epithelium often forming papillary projections. The epithelium is usually double layered, and, in the case of inflammation, the epithelium may undergo squamous metaplasia. Mucus-producing goblet cells are occasionally observed. The contents of the cystic portion of the tumor vary in consistency from thin and watery to thick and viscous. It may have an admixture of cholesterol crystals, inflammatory cells, histiocytes, and desquamated epithelial cells with pyknotic nuclei. The supporting stroma contains lymphoid elements, which often form large germinal centers.
Cystic material with a thin watery-to-mucoid appearance predominates in aspirates from adenolymphoma. The smears contain a granular and amorphous substance, sheets of cells with oncocytic changes, cellular debris, and macrophages. A lymphoid component is almost always present, but the amount varies.
The characteristic and diagnostic feature in adenolymphoma is the presence of flat, irregular sheets of polyhedral, finely granular oncocytic cells with well-defined cellular borders ( Fig. 22-15A ). With Papanicolaou stain, these sheets have a honeycomb arrangement ( Fig. 22-15B ). Papillary fragments with the lymphoid stroma and overlying oncocytic cells can occasionally be observed. The nuclei of the oncocytic cells are round, with a granular chromatin pattern and a central small nucleolus. Mast cells are a frequent finding in May–Grünwald–Giemsa-stained material but are not easily seen in Papanicolaou-stained smears. They are found in association with the sheets of oncocytic cells. Mast cells may also occur, but less frequently, in mucoepidermoid carcinoma and pleomorphic adenomas.
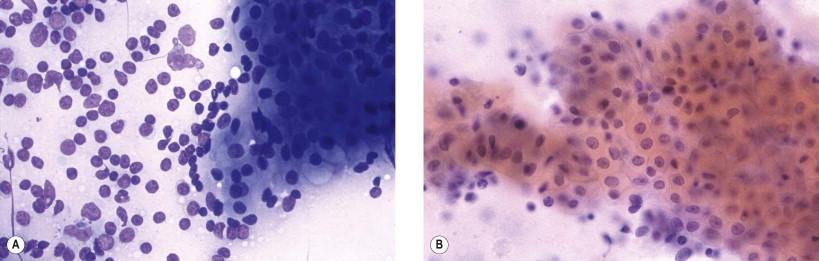
Monolayered sheets of oncocytic cells with distinct cell borders
Thin to thick mucoid cystic material
Inflammatory cells and cellular debris
Mixed population of lymphocytes often in tissue tangles.
An aspirate containing only fluid or mucoid material is obviously not diagnostic but raises the possibility of adenolymphoma if the aspiration is made in the parotid gland region.
Cystic, well-differentiated mucoepidermoid carcinomas may also yield aspirates of abundant mucoid material containing small clusters of epithelial cells with minimal nuclear atypia and numerous lymphocytes. The epithelial cells in this case usually are superimposed and have a finely vacuolated cytoplasm, a sign of mucus production. These cells do not usually appear in monolayered sheets, and the cellular borders are indistinct.
Metaplastic epidermoid cells in adenolymphomas can be atypical and even show true keratinization. They can, thus, be confused with squamous carcinoma cells ( Fig. 22-16 ). We therefore advocate that a conclusive diagnosis of a well-differentiated squamous cell carcinoma in the parotid area should be made only if a primary carcinoma can be found in the oral cavity or upper respiratory tract.
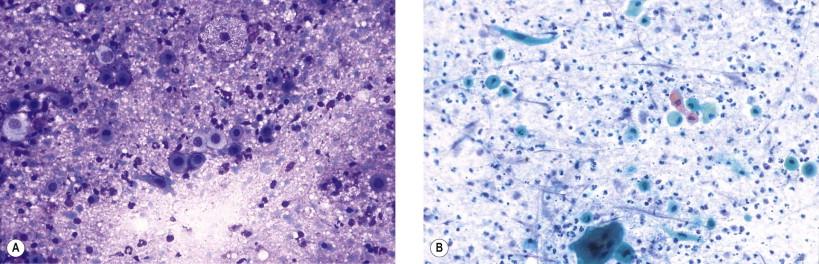
Aspirates from a predominantly solid adenolymphoma with only a few lymphocytes may be indistinguishable from oncocytoma. Distinction between these two neoplasms is not critical because their management should be identical. Some workers believe that these two tumors are variants of the same entity.
Oncocytomas are rare benign tumors that typically appear in elderly patients as a hard nodule. They are most often found in the parotid glands but can be found in all major salivary glands. The tumor is composed of cells with abundant granular eosinophilic cytoplasm and round, central, or eccentric nuclei with distinct nucleoli. The cells are uniform and arranged in solid acinar groups or parallel columns with sparse, delicate supporting stroma. Unlike most Warthin's tumors, oncocytomas lack lymphoid infiltrates. A malignant variant has been described.
In aspirates from oncocytomas, the principal feature is solid plugs and sheets of polygonal, monomorphous oncocytic cells in a clean or slightly bloody background ( Fig. 22-17 ). These cells are identical to the oncocytes of Warthin's tumor. Lymphoid cells and a proteinaceous precipitate are not observed in oncocytomas.
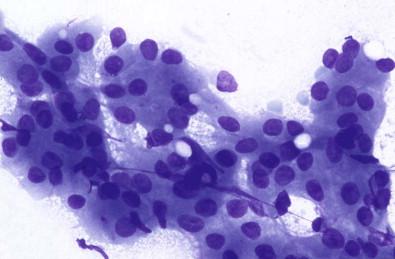
Clusters of uniform large cells with abundant finely granular cytoplasm (oncocytes)
Round nuclei usually centrally located within the cell and containing distinct nucleoli
Absence of fluid, debris, and lymphoid cells.
Oncocytomas may be confused with non-neoplastic oncocytic nodular hyperplasia, acinic cell carcinoma, and Warthin's tumor. A precise cytologic distinction between various benign lesions containing oncocytic cells does not seem important because all are cured by simple surgical excision. It is more important to differentiate between a benign oncocytic lesion and a malignant tumor, which may require a different type of treatment. In the malignant variant of oncocytic origin mitotic figures are often seen as well as necrosis. In aspirates from acinic cell carcinoma, the cells tend to a more poorly outlined cytoplasm which is more granular and finely vacuolated than typical cells from oncocytoma. In May–Grünwald–Giemsa stain, the cytoplasm of cells from acinic cell carcinoma usually appears slate-gray and may contain a fine red granulation not seen in benign oncocytic cells.
Pleomorphic adenoma and mucoepidermoid carcinoma may have focal oncocytic changes. This is, however, seldom a diagnostic problem if adequate sampling of material from different parts of these tumors has been carried out.
Basal cell adenomas account for about 2% of all primary tumors of the major salivary glands and are mostly seen in elderly patients. The term basal cell adenoma is used to identify a neoplasm with a single, well-defined, basaloid histologic pattern, which can have a diverse architecture such as trabecular, tubular, papillary, and solid patterns. Cystic changes may be superimposed on these patterns. The mesenchymal chondromyxoid elements of the type encountered in pleomorphic adenomas are generally lacking.
The FNA features of basal cell adenoma have been described by several investigators. Smears are cellular and show cohesive solid groups and irregularly branching cords ( Fig. 22-18 ). The individual cells are uniform, with scant poorly defined cytoplasm, oval nuclei with finely granular chromatin ( Fig. 22-18 ). Nucleoli may be seen in occasional cells. Single cells usually appear as naked nuclei, and small amounts of fibrous to amorphous material may be seen at the edges of cell clusters. This matrix is homogeneous, stains bright red with May–Grünwald–Giemsa stain, and is almost translucent in Papanicolaou-stained slides.
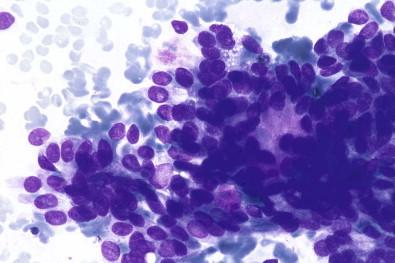
Small clusters of branching cords composed of small uniform cells with round or oval nuclei
Sparse homogeneous background material
Individual cells appear as naked nuclei or have a scant amount of cytoplasm.
FNAs from basal cell adenomas and from the solid form of adenoid cystic carcinomas have similar cytologic features. The size and configuration of cell groups, as well as the individual cell morphology and the presence of single cells, are virtually identical. The solid adenoid cystic carcinoma is lacking the metachromatic globules or cylinders that characterize cribriform adenoid cystic carcinoma. The distinction between these two prognostically quite different tumor entities is difficult. Clinically, pain or nerve damage indicates malignancy.
The cytologic differentiation from pleomorphic adenoma is sometimes difficult but myoepithelial cells are never seen. The distinction between these two entities seems to be less important because the management is similar.
Clear cell adenomas are composed of prominent clear cells that form tubular structures. The differential diagnosis in tissue sections includes nodular clear cell/oncocytic hyperplasia, acinic cell tumor, and mucoepidermoid tumor. Although we have not encountered this uncommon tumor, we believe that the same differential diagnostic problem would occur in FNA.
Sebaceous cell adenoma is a very rare solid or sometimes cystic neoplasm made up of flat polygonal cells similar to the sebaceous cells usually seen in smears from epidermoid cysts ( Fig. 22-19 ).
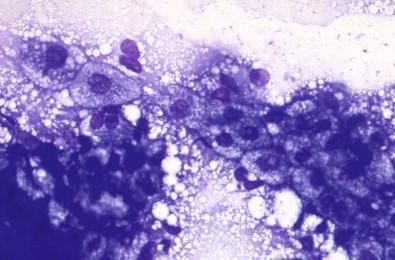
Sebaceous cells are occasionally seen in tissue specimens from major salivary glands and within neoplasms such as pleomorphic adenoma and Warthin's tumor.
Tumor cells often in dense clusters
Few dispersed cells
Monomorphic cells with round nuclei and a rich vacuolated/granular cytoplasm.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here