Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The goal of this chapter is to promote practical techniques that enhance safety during ultrasound-guided peripheral nerve block (PNB). Ultrasound-guided PNB can be performed with a level of precision and purpose that is not feasible with other techniques. Ultrasound imaging and guidance for PNB is unique among all the nerve localizing tools and monitors because the needle trajectory can be tracked and controlled, reducing the risk of trauma to nerves and other critical structures. However, operators find it challenging to maintain the needle in the plane of the ultrasound beam in all patients and anatomical locations. Therefore, anesthesiologists require appropriate training and education to develop this skill. As with all regional anesthesia techniques, the safety and efficacy of ultrasound-guided techniques require meticulous attention to indication, anatomy, equipment, and technique.
Inadvertent vascular puncture and injection of local anesthetic may result in local anesthetic systemic toxicity (LAST). Inadvertent vascular puncture may result in persisting bleeding or discomfort from bruising in the subcutaneous tissues or muscles. More serious cases of trauma have resulted in trauma to critical organ sites including the liver, kidney, intestinal wall, pelvis, retroperitoneum, and mediastinum. Sequelae of bleeding and hematoma have included pain, neurologic complications, renal failure, postponement of surgery, surgical intervention to correct the injury, sepsis, delayed recovery, prolonged hospitalization, dyspnea, tracheostomy, and even death. Fortunately, serious hematoma and injury are rare. However, these have occurred in patients with both normal and impaired hemostasis and following both superficial and deep PNB. Therefore, knowledge of the frequency and origins of the vasculature in close proximity to block locations is important so as to avoid these vessels and reduce the incidence of vascular complications. A Cochrane meta-analysis of randomized, controlled trials concluded that ultrasound guidance was associated with a reduced incidence of vascular puncture compared to nerve stimulator guidance. This result is supported by observational studies.
Identify vessels on the sonogram along the intended needle trajectory .
Many PNB locations have a high level of vascularity, and vessels can be imaged using two-dimensional (2D) or color Doppler modes on the ultrasound machine. The authors recommend that practitioners routinely perform a pre-block survey sonogram to identify major and minor vessels so that a safe needle trajectory can be planned. An example presented in Fig. 74.1 is a pre-block survey sonogram obtained prior to a supraclavicular brachial plexus block. This pre-block sonogram identified a large arterial structure (the dorsal scapular artery) between the neural elements of the brachial plexus. It is important to note that when arterial flow is at 90 degrees to the angle of insonation, there will be no color Doppler signal. Some vessels may be difficult to identify on the sonogram related to size, circumflex course, and depth.
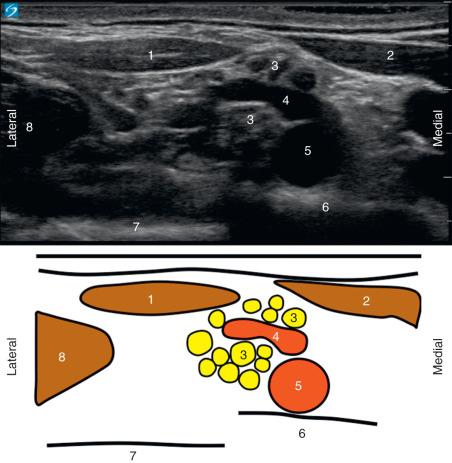
Knowledge of common vascular branches .
Because the operator may not always identify vessels using ultrasound, knowledge of the location of key vasculature is of paramount importance. In this section, we provide two examples in the neck and inguinal region. The dorsal scapular, suprascapular, and superficial cervical arteries arise directly from the subclavian artery or from the thyrocervical trunk. There is substantial variation in the anatomy and nomenclature with at least four different arterial trunks described emerging directly from the subclavian artery or from the thyrocervical trunk. In a combined cadaver and clinical study, Muhly documented that arterial branches were often in close proximity to or passed between branches of the brachial plexus ( Fig. 74.1 ). In the supraclavicular region, arterial branches, usually the dorsal scapular artery, were adjacent to the plexus in 43/50 (86%) patients. In the interscalene region and arterial vessel (likely the superficial cervical artery), they were identified in 45/50 (90%) of patients. In a similar study performed by the same authors, at the level of the femoral crease, there was a 12% incidence of arterial vessels (profunda femoral, lateral circumflex femoral) located lateral to the femoral artery.
Plan an atraumatic needle trajectory taking into account both identified and expected vessels .
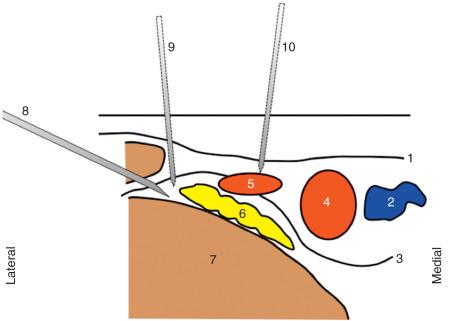
The anesthesiologist can implement strategies to reduce the risk of inadvertent vascular trauma (if a vessel is identified on a pre-block sonogram) including a complete change of needle trajectory, a minor change to the transducer angle so that the vessel is no longer in the planned needle trajectory, or another approach to the nerve or plexus. Using the example in Fig. 74.1 , if the vessel is large, it may be sensible to choose a different approach to the brachial plexus. Using an example at the femoral block level, circumflex vessels have a medial to lateral course, vessels may be anterior to the femoral nerve, and injury to this vessel may be less likely if an anteromedial approach is avoided and a lateral to medial approach is used. In Fig. 74.2 , examples of trajectories and relative risk of vascular injury during femoral block are illustrated.
LAST is a feared complication of regional anesthesia due to its potential to result in a fatal outcome. LAST is variable in timing of onset and presentation; therefore, diagnosis can be delayed or even missed. Management of this complication has improved with the use of crisis resource algorithms, cognitive aids, checklists, and the use of intravenous lipid emulsion (ILE) therapy. ILE represents a major milestone in improving safety. Despite these advances, the most important strategy for improving patient outcomes is preventing LAST.
Recent epidemiological and registry studies have captured LAST events including presenting features and incidence. Table 74.1 summarizes the incidence of LAST, block types involved, practice setting, and severity/type of LAST.
| Author, Year | n | N | Incidence | Block Type | Practice Setting | Severity and Nature of Presenting Features | |
|---|---|---|---|---|---|---|---|
| n per 1000 | % | ||||||
| Ecoffey, 2010 | 16 | 29,290 | 0.54 | 0.05 | Neuraxial: 10,554 Peripheral: 18,735 |
Pediatric | All major LAST, Seizure 1; cardiac 15 (15 occurred under GA) |
| Orebaugh, 2012 | 6 | 14,498 | 0.41 | 0.04 | Peripheral | Single-center academic | Major LAST (seizures) all with non-ultrasound technique |
| Sites, 2012 | 0 | 12,668 | – | Peripheral | Single-center academic | Events sought were seizures | |
| Polaner, 2012 | 0 | 14,917 | 0–2 a | Neuraxial: 9,156 Peripheral: 5,761 |
Pediatric, 6 academic centers | ||
| Barrington, 2013 | 22 | 25,336 | 0.87 | 0.09 | Peripheral (all) | Multi-center, mixed academic, community | Minor LAST – 13 (prodromes, agitation); major LAST – 9 (seizures 6, loss of consciousness 3, cardiac arrest 1,) |
| Rohrbaugh, 2013 | 8 | 15,014 | 0.53 | 0.05 | Interscalene | Details not provided | |
| Gurnaney, 2014 | 3 | 1954 | 1.53 | 0.2 | Peripheral | Pediatric, Single-center academic | Outpatient catheters, all minor (prodromes) |
| Heinonen, 2015 | 15 | 217,700 | 0.07 | 0.007 | Nationwide center | Major LAST 15 (CV 1, seizures 14) | |
| Liu, 2016 | 3 | 80,661 | 0.04 | 0.004 | Peripheral | Single-center, high-volume practice | Major LAST (seizures) – 3 |
| Allegri, 2016 | 10 | 29,545 | 0.3 | 0.03 | Peripheral | Multi-center | Minor LAST – 3 (prodromes); major LAST –7 (seizures 4, CV 3) |
| Morwald 2017 | 434 | 238,473 | 1.8 | 0.18 | Peripheral | Administrative database | Surrogate outcomes |
a 95% confidence interval per 10,000 presented for zero events, calculated by author; one event presented as unconsciousness followed by seizure.
In 2013, a dataset from the Australian and New Zealand Registry of Regional Anaesthesia (AURORA) comprising 25,336 PNBs from 20,021 patients was analyzed. There were 22 cases of LAST (13 mild severity, 8 major, and 1 cardiac arrest) with an overall incidence of LAST of 0.87 per 1000 PNB. Analysis of this dataset using logistic regression and propensity scoring demonstrated that ultrasound guidance was associated with a 60% to 65% reduced risk of LAST. Factors associated with an increased risk of LAST included site of injection (upper extremity and paravertebral block having an increased risk of LAST compared to lower extremity block), increased local anesthetic dosage, increased local anesthetic dosage per weight, and reduced patient body weight. Lidocaine was associated with an increased risk of LAST compared with ropivacaine. This result was surprising because conventional wisdom would predict that lidocaine would present a lower risk of LAST. Interestingly, lidocaine dosage was increased in ultrasound-guided techniques (4.55 [1.54 to 7.5] mg/kg) compared to nonultrasound techniques (3.38 [1.10 to 6.67] mg/kg); presented as median (10th to 90th centiles). This was in contrast to ropivacaine dosage 1.48 (0.73 to 2.71) mg/kg being reduced for ultrasound-guided techniques compared to non-ultrasound techniques 1.63 (0.74 to 2.88) mg/kg. Bupivacaine was only used in 4% of 25,336 PNB; therefore, it was not possible to determine if this was a potential risk factor. During the time period of the AURORA registry (2007 to 2012), epinephrine was not uniformly captured to the database; therefore, this could not be entered as a covariate in the logistic regression model. It is generally accepted that patient comorbidities and individual susceptibility to local anesthetic may increase the risk of LAST.
In 2016, the incidence of LAST reported by Liu was 0.04 per 1000 PNBs from a cohort of 80,661 PNB from the Hospital for Special Surgery. This incidence is substantially lower than other reports. However, it was derived from a high-volume PNB practice that may have expertise not representative of the wide spectrum of clinical practice. A nationwide study in Finland used an electronic survey to record outcomes over a 3-year period, 2011 to 2013, where it was estimated that 211,700 PNB were performed. Fifteen episodes of LAST were reported giving an overall incidence of LAST of 0.7 (95% CI, 0.4 to 1.2) per 10,000. Ultrasound guidance was used in two of three cases from the Hospital for Special Surgery and 5 of the 15 cases of LAST in the study from Finland. Ultrasound guidance does not completely prevent LAST from occurring.
Despite advances in management of LAST poor outcomes still occur. In a recent summary of 53 published case reports of LAST following patient PNB from 59 patients between 2010 and 2014, mortality was the outcome in 6 of 59 patients with LAST events. From a recent cohort study that utilized a large administrative database, the authors' concluded that although the incidence of LAST is low, it remains clinically significant.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here