Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The algorithms used for left ventricular assessment cannot always be applied to the right ventricle (RV), which differs in morphology, shape, and function. The RV has a complex geometrical shape with a thinner and more trabeculated myocardium than the left ventricle (LV). The chest x-ray is a preliminary imaging tool, with the lateral view best suited to detect RV enlargement. Echocardiography is the mainstay for assessment of the RV, with published adult and pediatric guidelines. Cardiac magnetic resonance imaging (CMR) is the current gold standard for RV assessment and tissue characterization. Gated cardiac computed tomography angiography (CTA) is an alternative when CMR is contraindicated.
The retrosternal location of the right ventricular outflow tract (RVOT) and pulmonary valve (PV) makes echocardiography difficult, especially in adolescents and adults. CMR and CTA allow detailed assessment of anatomy, valve mobility, presence of poststenotic dilation, and associated condition of the branch pulmonary arteries (PAs). With current CT scanners, image acquisition can be completed within seconds or less and with a short breath hold. CTA is excellent for assessment of proximal and distal PAs. CMR is excellent for sequential assessment of the PV. Anatomy and function of the RVOT and PV can be assessed by balanced steady state free precession (SSFP) CMR imaging. Turbo spin-echo with blood suppression technique is very useful for imaging RVOT and branch PAs especially in patients with metallic implants causing artifacts. Through-plane and in-plane phase contrast quantification of flow across the PV and branch PAs provides comprehensive functional assessment including valve stenosis or regurgitation, and differential pulmonary blood flow. Magnetic resonance angiography (MRA) is an alternative to CTA for assessment of extracardiac structures.
Ebstein anomaly accounts for fewer than 1% of all cases of congenital heart disease. Associated malformations include atrial septal defect (ASD), ventricular septal defect (VSD), pulmonary stenosis and atresia, tetralogy of Fallot, congenitally corrected transposition of the great arteries, and patent ductus arteriosus (PDA). Conduction system abnormalities and arrhythmias, including Wolff–Parkinson–White syndrome, have been seen in 22% to 42% of patients.
Wilhelm Ebstein provided the first description of this abnormality in 1866. It is characterized by abnormal development and positioning of the tricuspid valve (TV) leaflets, with apical displacement of the septal and posterior leaflets. This phenomenon leads to partitioning of the RV into an atrialized basal inlet segment and an apical outflow chamber. The displaced valve leaflets may be adherent to the RV wall. The anterior valve leaflet tends to be normally positioned, however, it is frequently large and redundant, and sometimes adherent to the RV free wall with a “sail-like” appearance.
The pathophysiology of Ebstein anomaly varies, depending on the degree of dysplasia and displacement of the TV leaflets and the presence and severity of associated defects. With greater degrees of dysplasia and displacement, the severity of tricuspid insufficiency increases, resulting in elevated right atrial pressures and thus right-to-left shunting at the atrial level. Patients with mild forms of Ebstein anomaly may be asymptomatic in childhood and present with symptoms of cyanosis, right heart failure, or arrhythmia in adulthood. Patients with severe forms present in early neonatal life with severe combined cyanosis and acidosis, and a ductal-dependent circulation.
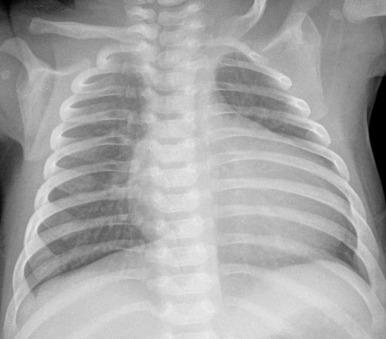
Classically, the chest radiographic appearance includes a globular or box-shaped heart as a result of right atrial enlargement associated with normal to diminished pulmonary vascularity ( Fig. 74.1 ). Findings can vary by clinical presentation. Patients with significant valvar dysplasia and severe tricuspid insufficiency have marked right heart enlargement and decreased antegrade pulmonary blood flow. With less marked valve displacement and tricuspid insufficiency, only mild right heart enlargement may be present ( e-Fig. 74.2 ).
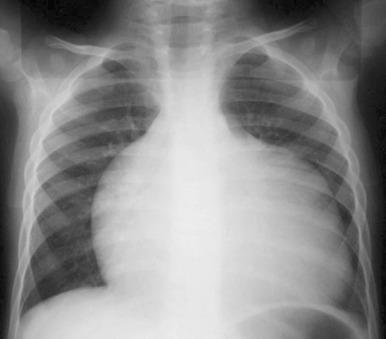
Ebstein anomaly can be readily diagnosed by both prenatal and postnatal echocardiography. Echocardiography and CMR allow for detailed anatomic and functional evaluation. The characteristic features include displacement and tethering of the septal and posterior leaflets of the TV, dilation of the right atrium, and atrialization of the RV ( Fig. 74.3 ). Imaging goals include evaluation of the TV and severity of tricuspid regurgitation; evaluation of right-to-left shunting through the ASD or patent foramen ovale, evaluating volume and function of the RV including the atrialized portion; delayed enhancement in the thin atrialized ventricular wall and septum; PV and branch PA stenoses; left ventricular size and systolic function; and aortic arch anomalies. Axial imaging provides reliable information about the atrialized RV. A total right/left ventricular volume index obtained by CMR has been found to correlate with heart failure markers and disease severity.
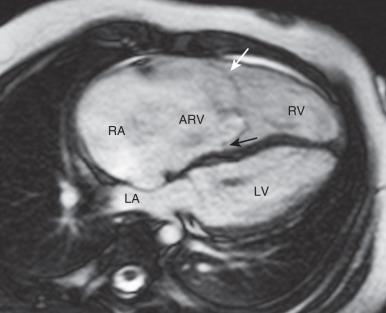
Patients with mild forms of Ebstein anomaly may not require surgical treatment, or the need for surgery may not arise until later in life until right heart dysfunction develops. Indications for surgical intervention/reintervention and/or medical therapy include limited exercise capacity (greater than New York Heart Association class II), increasing heart size (cardiothoracic ratio >65%), cyanosis (resting oxygen saturations <90%), severe tricuspid regurgitation with symptoms, progressive right ventricular dilation or dysfunction, transient ischemic attack, or stroke.
Surgery includes valvuloplasty with either the Carpentier or Da Silva's cone procedures. The Carpentier procedure involves reimplantation of the anterior and posterior TV leaflets at the level of the neotricuspid annulus, resulting in a monocuspid or bicuspid valve configuration. More recently described “cone reconstruction” includes mobilization and re-anastomosis of the TV leaflets and subvalvar apparatus to form a cone-shaped valve with improved competence. Other possible treatments include TV replacement or, rarely, oversewing of the TV and creation of a central shunt in infancy with an ultimate single ventricle type palliation. Transcatheter closure of the ASD may be considered if severe cyanosis is present without tricuspid insufficiency necessitating valve repair. Rarely, cardiac transplantation may be indicated. Clinically significant arrhythmias develop in some patients and require transcatheter or surgical ablation procedures.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here