Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Acute renovascular ischemia is defined as a sudden interruption of arterial and/or venous renal blood flow; it may involve either partial or complete ischemia of one or both kidneys. In the setting of acute renal ischemia, salvage of a functioning kidney requires a high index of suspicion, rapid diagnosis, and prompt implementation of a definitive treatment plan. Acute renal ischemia can progress rapidly to irretrievable loss of renal function. Appropriate clinical management is therefore quite time-sensitive, and the duration and severity of renal ischemia are key considerations for decision-making.
Causes of acute renal ischemia include renal artery thrombosis, embolism, trauma, aortic or renal artery dissection, iatrogenic injury, and renal vein thrombosis ( Box 130.1 ). Evidence supporting specific management strategies for acute renal ischemia is limited and primarily comes from case reports and small cohort studies.
Renal artery thrombosis
Atherosclerotic disease
Aneurysmal disease
Dissection (aorta or renal artery)
Fibromuscular dysplasia
Arteritis (e.g., Takayasu)
Stent thrombosis
Hypercoagulable state
Renal artery embolism
Chronic atrial fibrillation
Valvular heart disease
Aortic plaque
Paradoxical embolism in setting of patent foramen ovale
Cardiac tumor
Trauma
Iatrogenic
Direct injury or embolization
Ostial occlusion by aortic endograft
Renal vein thrombosis
Hypercoagulable state
Mechanical obstruction
Malignancy
Ischemic renal injury is characterized by glomerular collapse and tubular necrosis, resulting in reduced glomerular filtration and loss of tubular function. Post-ischemic reperfusion further exacerbates endothelial dysfunction through excess cytokine secretion and the expression of adhesion molecules that promote leukocyte influx. Reperfusion injury also leads to production of nitric oxide and reactive oxygen species, creating additional parenchymal damage and impaired autoregulation of renal blood flow.
Baseline kidney function and the duration of warm renal ischemia affect the potential for recovery of function and are therefore important considerations when revascularization is being considered. Age and diabetes have been identified as risk factors associated with increased susceptibility to injury from acute renal ischemia in experimental models. ,
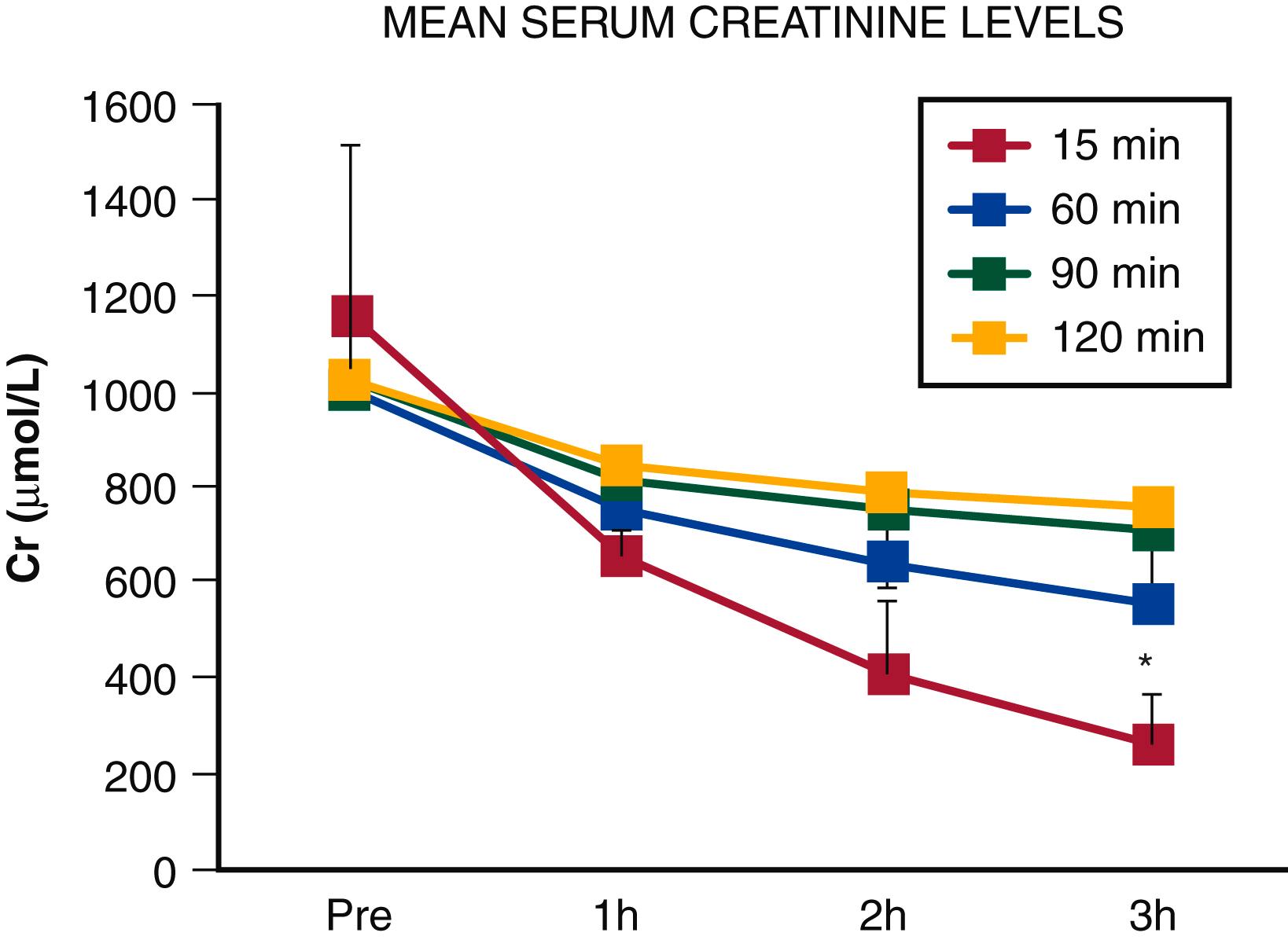
In a normal kidney, 1 hour of warm renal ischemia is associated with loss of 70% to 80% of function, but complete recovery is possible within weeks. Increasing the ischemia duration to 2 hours limits long-term recovery to 30% to 50% of baseline function. In vivo models of acute, total renal artery occlusion have demonstrated irreversible ischemia after 3 to 4 hours of warm ischemia, but this varies by species. , Figure 130.1 shows serum creatinine levels in a porcine model after warm renal ischemia lasting 15, 60, 90, and 120 minutes. Prolonged ischemia time (>45 minutes) has been identified as a predictor of poor early graft function following human living donor kidney transplantation, and more than 90 minutes of acute warm ischemia has been suggested as a cut point for the retrieval of renal function. Technically successful revascularization performed after longer durations of acute ischemia may not achieve functional recovery, especially when overt renal failure or anuria is already present. ,
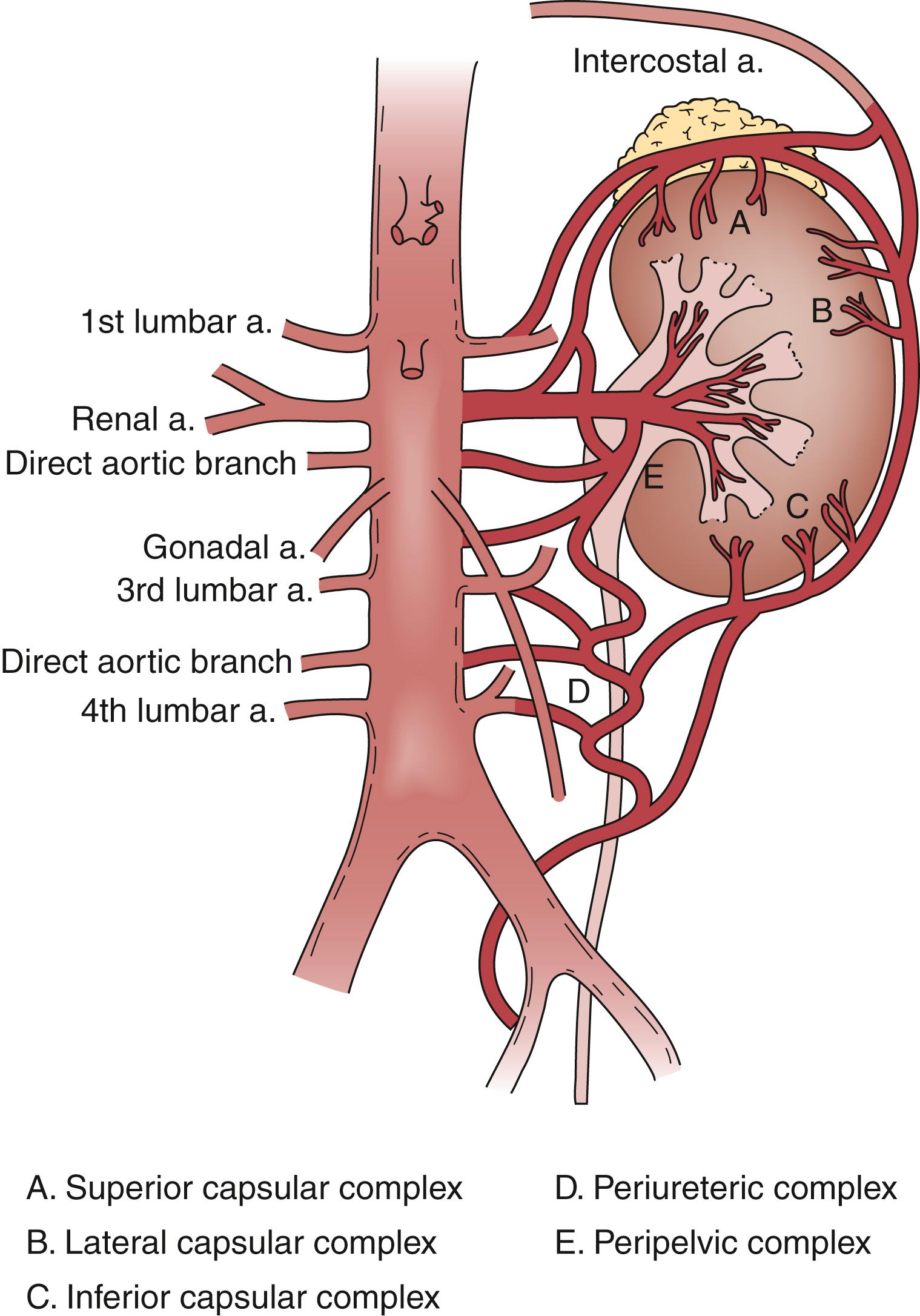
In contrast to acute renal artery occlusion, gradual main renal artery occlusion (e.g., main renal artery stenosis that progresses in severity over months or years) may induce development of collaterals and lessen the severity of the ischemic insult. Renal artery collaterals originate from the inferior adrenal, gonadal, ureteral, internal iliac, lumbar, intercostal, and capsular arteries ( Fig. 130.2 ). Less commonly, collaterals may also originate from the inferior mesenteric artery. Pre-renal communication may exist at the hilum, while intrarenal communication occurs through capsular perforating arterioles. As much as 80% of renal arterial collateral communication may be independent of the main renal artery, potentially allowing maintained functional viability despite main renal artery occlusion (as in acute thrombosis of a chronic critical stenosis). , Accessory renal arteries (alternatively referred to as aberrant, duplicated, or extra renal arteries) have a prevalence of 24% to 42% based on imaging and cadaver studies and may also preserve direct flow from the aorta.
With gradual reduction of renal perfusion, maintained tubular function (reflected by low fractional excretion of sodium and normal N -acetyl-glucosaminidase excretion) has been observed despite reductions in both glomerular filtration rate and filtration fraction. In a canine model of gradual renal artery occlusion induced incrementally over 7 weeks, collateral circulation developed and was sufficient to maintain both renal viability and life. Clinical reports of functional recovery following periods of main renal artery occlusion exceeding 24 hours underscore the importance of collateral perfusion and demonstrate that the potential for kidney salvage is not solely a function of warm ischemia time. Main renal artery occlusion accompanied by the visualization of good renal vein flow on duplex ultrasound suggests the presence of collateral arterial flow.
Renal vein thrombosis can also produce acute renal ischemia, and may occur primarily or in association with inferior vena cava thrombosis. As in acute arterial ischemia, the degree of renal functional impairment and severity of clinical presentation depend on the acuity of venous occlusion and collateral flow. When renal vein thrombosis occurs rapidly in the absence of developed collaterals, congestion, and edema of the affected kidney can produce pain from capsular distension as well as hemorrhagic infarction. , Experimental models have demonstrated that renal injury and blood flow reduction are both greater from venous than arterial occlusion.
Symptoms and signs of acute renal ischemia include abdominal or back pain, dyspnea, nausea, vomiting, hematuria, anuria, and acute hypertension. , , , , Since many of these symptoms are nonspecific, the differential diagnosis for acute renal ischemia is broad and includes a variety of nonrenal pathologies. Renal colic with nephrolithiasis is a common initial presumptive diagnosis with a similar presentation, and several reports have recommended that absence of renal calculi on an unenhanced computed tomography (CT) scan performed for suspected renal colic should prompt a contrast-infused study to rule out acute ischemia. Other diagnoses with symptoms similar to renal ischemia include pyelonephritis, renal carcinoma, mesenteric ischemia, cholecystitis or biliary colic, gastritis, splenic infarction, myocardial infarction, and pulmonary embolism. , , , ,
Clinical presentation of acute renal ischemia is often delayed for hours or days after symptom onset, adversely affecting the possibility of functional salvage through revascularization. In a series of patients with embolic renal artery occlusion secondary to atrial fibrillation described by Hazanov et al., the majority of patients presented over 24 hours after symptom onset ; Huang et al. reported a median time of 31 hours between symptom onset and emergency department presentation for acute renal infarction.
Renal vein occlusion may also present with unilateral congestion and tenderness, although the enlarged kidney is seldom palpable in adults. In a retrospective cohort study of neonates with renal vein thrombosis, only 13% of patients had the “classic triad” of flank mass, gross hematuria, and thrombocytopenia.
No single laboratory test is diagnostic of acute renal ischemia, so tests must be selected and interpreted in combination with the clinical presentation and physical exam findings. Laboratory abnormalities reported with acute renal ischemia include leukocytosis, elevated lactate dehydrogenase (LDH), gross or microscopic hematuria, proteinuria, elevated D-dimer, and eosinophilia. , , , , , , , Eosinophilia may be indicative of atheroembolism. , In a series of 20 patients with acute renal ischemia, 19 patients had an elevated serum LDH, while 16 had the triad of flank/abdominal pain or tenderness, elevated serum LDH, and proteinuria. Only half of patients presenting with acute renal ischemia have an elevated serum creatinine, so a normal serum creatinine does not exclude this diagnosis. In the setting of a contralateral kidney with normal function, asymptomatic unilateral renal ischemia may be identified as an incidental imaging finding of unknown acuity.
Imaging plays a critical role in the diagnosis of acute renal ischemia because the history, physical exam, and laboratory findings are often nonspecific. The ideal diagnostic imaging test for renal ischemia would be rapid, widely available, noninvasive, inexpensive, and with no need for either radiation exposure or contrast administration. Although no imaging technique shares all of these characteristics, consideration of these attributes can be helpful in choosing the most appropriate study.
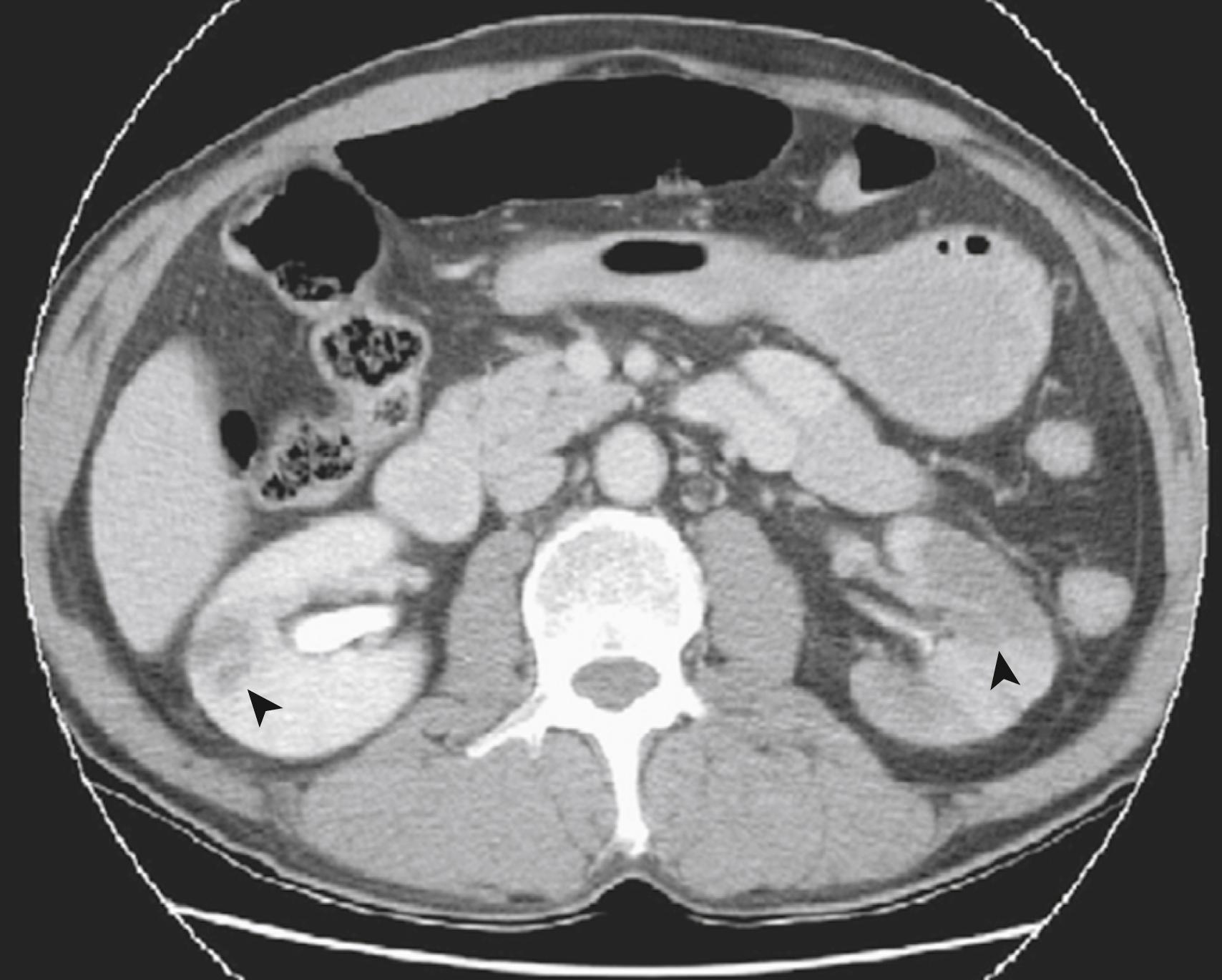
Computed tomography angiography (CTA) is widely available, rapid, and useful for ruling out other soft tissue abnormalities when acute renal ischemia is suspected. CTA is less time-consuming than MRA and has 80% sensitivity for acute renal ischemia (which is superior to ultrasound). CTA also evaluates aortic and iliac anatomy, which is useful for planning revascularization if indicated. CTA should include both noncontrast and venous phase images, and is also the study of choice for renal vein thrombosis, with a sensitivity and specificity that both approach 100%. Features of a renal infarct on CTA imaging include areas of hypoattenuation with associated mass effect ( Fig. 130.3 ) or a “cortical rim sign,” described as a rim of functioning nephrons supplied by capsular collaterals surrounding an otherwise nonfunctioning kidney. , , ,
Renal MRA is capable of the anatomic evaluation plus physiologic assessment through flow-dependent imaging. Unenhanced MRA evaluation for renal artery disease has similar reliability to CTA; its capability for evaluating renal segmental arteries and parenchymal disease make it potentially useful when contrast imaging is contraindicated. These advantages have resulted in increasing utilization of MRA for the evaluation of chronic renal artery disease, but MRA is used less frequently as first-line imaging for acute renal ischemia. MRA has practical drawbacks when rapid imaging is required because it is more time-consuming and may require extensive image post-processing by a technologist. Compared with CTA and/or ultrasound, access to rapid MRI is more limited. MRI/MRA may have a benefit when assessing for renal cell carcinoma concomitant with renal vein thrombosis.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here