Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Evaluation of the renal morphology in allograft patients is used to answer two major questions: Is the failure of the graft caused by rejection or some other unrelated lesion? And if rejection is present, what immune mechanism(s) is (are) the cause, and is the lesion potentially reversible using available therapeutic approaches? In the absence of evidence of rejection, it should be ascertained whether the graft failure results from acute tubular injury, acute infectious pyelonephritis, obstruction of the vasculature or urinary outflow tract, presence of recurrent or de novo disease, or toxicity associated with the therapeutic agents used to modulate the immune response. In assessing whether rejection lesions are potentially reversible, it is necessary to evaluate not only the nature of the rejection but also the intensity and chronicity.
The Banff working classification of renal allograft pathology, established in 1997, is an internationally agreed-upon standardized classification of the morphologic changes associated with various types of rejection. It was revised and updated in 2007, 2009, 2013, 2017, and 2019. The XV Banff 2019 conference report detailed clarification of criteria for chronic active T cell–mediated rejection (TCMR), for borderline rejection, and for antibody-mediated rejection (ABMR). Revisions included thresholds for interstitial inflammation in the diagnosis of borderline; peritubular capillaritis grading and C4d scoring in the diagnosis of ABMR; and the interpretation and introduction of a new scoring for interstitial scarring and tubular atrophy ( Tables 8.1–8.5 ). The newer versions of the Banff system were influenced by data from several clinical trials that used the Banff 94 Schema, the results of clinical correlations of the Cooperative Clinical Trials in Transplantation (CCTT), and the findings of the subsequent biannual Banff meetings with the recognition that ABMR plays a significant role in both acute and chronic graft survival. Interstitial infiltration of activated lymphocytes with tubulitis is characteristic of TCMR (type I), with intimal arteritis being indicative of vascular involvement. Although these features are considered the main lesions that are indicative of acute cellular rejection episodes, it is now recognized that antibody-mediated responses can also play a significant role in rejection and can coexist with TCMR. Linear C4d staining of peritubular capillaries (PTCs) by immunofluorescence or immunohistology is characteristic of acute ABMR. The potential contribution of molecular diagnostics to better defining ABMR was discussed at the most recent Banff meeting. These concepts were incorporated into the previous versions of the Banff classification system (see Tables). Ultimately, the goal of the Banff classification system is to be able to give a diagnostic biopsy grade that will provide both a prognostic and a therapeutic tool ( Tables 8.2, 8.2A–C and 8.3–8.5 ). The standardized classification also promotes international uniformity in reporting of renal allograft pathology and is useful to facilitate the performance of multicenter trials of new therapeutic modalities.
| Type of specimen | Wedge biopsy | Core biopsy | ||
|---|---|---|---|---|
| Specimen ID: ______________________________________________ | ||||
| Number of glomeruli: | ||||
| Number of globally sclerosed * glomeruli: | ||||
| Percentage of global glomerulosclerosis: | ||||
| Number of arteries (not-arterioles) ** : | ||||
|
||||
| Circle appropriate findings: | ||||
| Interstitial fibrosis | None | Mild | Moderate | Severe |
| <5%; | 6–25% | 26–50% | >50% of cortex involved | |
| Tubular atrophy | None | Mild | Moderate | Severe |
| 0%; | <25% | 26–50% | >50% of cortical tubules involved | |
| Interstitial inflammation | None | Mild | Moderate | Severe |
| <10%, | 10–25% | 26–50% | >50% of cortex involved | |
| Arterial intimal fibrosis | None | Mild | Moderate | Severe |
| 0%; | <25% | 26–50% | >50% vascular narrowing | |
|
None | Mild * | Moderate * | Severe * |
|
||||
| Glomerular thrombi | None | Mild * | Moderate * | Severe * |
|
||||
| Acute tubular injury/necrosis | None † | Mild † | Moderate † | Severe † |
|
||||
| Other findings: (FSGS, nodular glomerulosclerosis, tumor, etc.) | ||||
|
|
|
|
|
| “Suspicious” for acute T cell–mediated rejection (TCMR; see Table 8.3 for scoring) may coincide with categories 2 and 5 and 6. This category is used when no intimal arteritis is present, but there are foci of tubulitis (t1, t2, or t3) with minor interstitial infiltration (i0 or i1) or moderate to severe interstitial infiltration (i2, i3) with only mild (t1) tubulitis. |
|
| (see Tables 8.3A, 8.3B, 8.3C for scoring); may coincide with categories 2 and 5 and 6. |
|
|
|
| No evidence of any specific etiology (may include nonspecific vascular and glomerular sclerosis, but severity is graded by tubulointerstitial features). |
|
|
| Changes not considered to be because of rejection; may be acute or chronic (e.g., hypertensive changes, calcineurin inhibitor toxicity, obstruction, bacterial pyelonephritis, viral infection) |
|
|
| i0 = <10% of biopsy area with inflammation a i1 = 10%–25% of biopsy area with inflammation i2 = 26%–50% of biopsy area with inflammation i3 = >50% of biopsy area with inflammation |
|
| Grade 1 (IF 1 TA 1) = Interstitial fibrosis and tubular atrophy in <25% of cortical area Grade 2 (IF 2 TA 2) = Interstitial fibrosis and tubular atrophy in 25–50% of cortical area Grade 3 (IF 3 TA 3) = Interstitial fibrosis and tubular atrophy in >50% of cortical area |
|
|
|
There is a significant gap between the need for renal transplants and the availability of donor kidneys. This shortage has led to the expansion of the acceptance criteria for deceased and living kidney organ donors. Extended criteria donors (ECD) include those aged 60 years or older and those over 50 years who also have at least two of the following conditions: a history of hypertension, a serum creatinine level greater than 1.5 mg/dL, or death from a cerebrovascular accident. Frozen section biopsy of the kidney at the time of procurement has become a routine procedure for the assessment of the degree of glomerular sclerosis, the severity of interstitial fibrosis and tubular atrophy, and the degree of arterial and arteriolar sclerosis and hyalinosis. A wedge biopsy is usually performed, but some centers use needle biopsies to obtain a better sampling of larger vessels. These biopsies are also useful in evaluating the degree of acute tubular epithelial injury and the presence of preexisting renal disease. Of note, mild acute tubular injury cannot be reliably ascertained on frozen sections because of inherent frozen section artifact. Each regional organ bank has a standard protocol for reporting the findings. The 2017 Banff working group developed consensus criteria for interpreting preimplantation kidney biopsies. These criteria facilitate the evaluation of biopsies and can be followed by any surgical pathologist (see Table 8.1 ).
The mechanisms involved in allograft rejection are complex and involve both cellular and humoral immunity. Efforts to reduce the immune response to alloantigens (HLA) include cross-matching human leukocyte antigens (HLAs) as closely as possible and blocking the presentation and recognition of these antigens. Additional non-HLA antigens are now also recognized as playing a part in rejection in some patients. In addition, protocols have been developed for preparing patients for ABO-incompatible transplantation. The status of the graft at the time of transplant is also important because outcomes are poorer with prolonged cold ischemia times.
ABMR is divided into two categories: immediate (hyperacute) and delayed. Hyperacute ABMR is allograft failure that occurs within minutes or hours after transplantation. It is thought to be the result of preexisting circulating antibodies in the recipient that are directed against alloantigens, including donor-specific HLA class 1, ABH antigens, and other autoantigens present in the grafted endothelium. Hyperacute ABMR is rarely encountered in today’s practice as the result of prescreening and matching a donor with a recipient. Presensitization of the recipient is often related to previous pregnancies, blood transfusions, or other previous antigenic stimuli; however, hyperacute rejections may also be related to endothelial damage that is not immunologic in nature, such as, for instance, donor-origin disseminated intravascular coagulation (DIC). A separate form of acute graft failure that is not immunologic has been termed acute imminent transplant nephropathy and may be related to injury occurring in the graft during the preservation phase. Acute ABMR refers to situations where graft loss occurs because of the development of antidonor-specific antibodies.
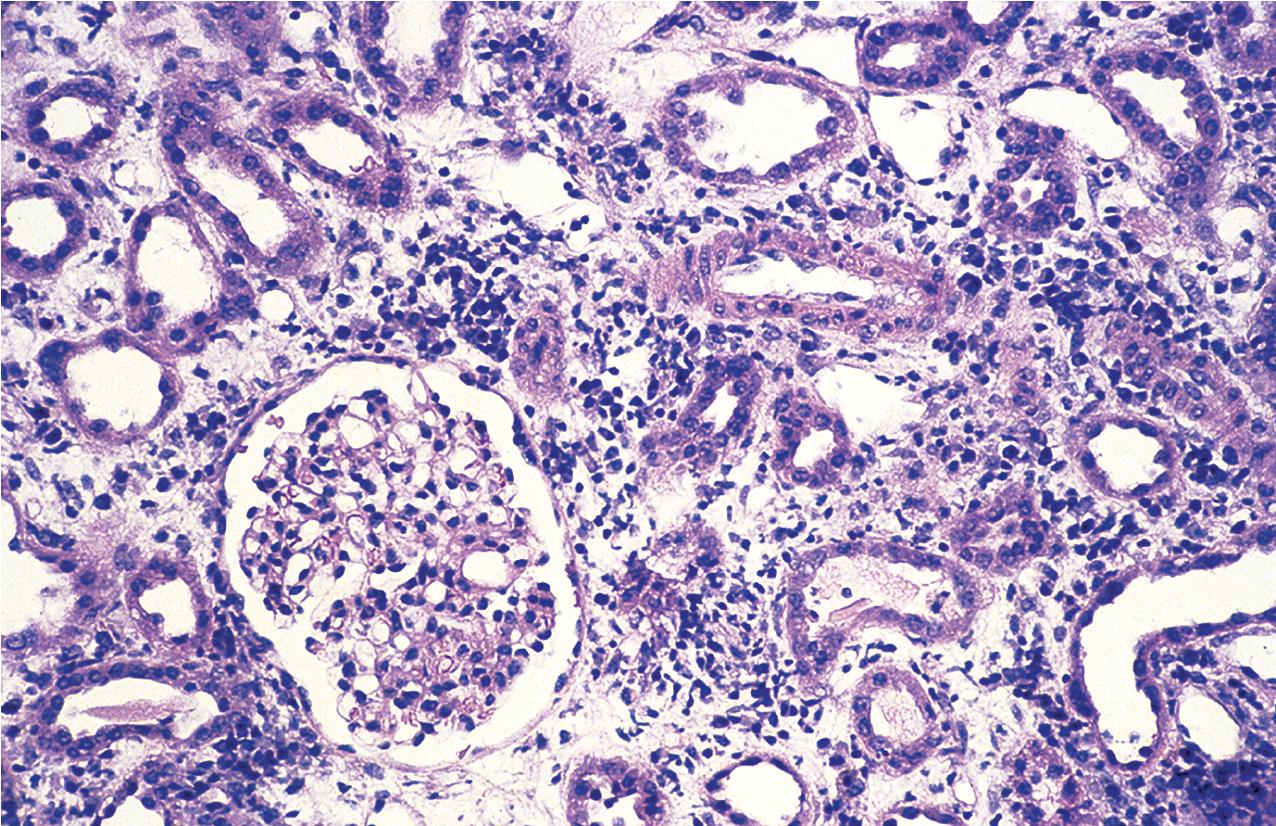
Morphologically, there is evidence of acute tissue injury with at least any one of the following: (1) microvascular inflammation, (2) intimal or transmural arteritis, (3) acute thrombotic microangiopathy (TMA) in the absence of other cause of TMA ( Fig. 8.1 ), or (4) acute tubular injury in the absence of another apparent cause. Active ABMR shows microvascular inflammation involving the glomerular capillaries (glomerulitis) and PTCs (peritubular capillaritis) with prominent leukocyte infiltration, which may include polymorphonuclear leukocytes ( Figs. 8.2 , 8.3 ). Both glomeruli and PTCs must show inflammation, with a combined Banff score of g plus ptc greater than or equal to 2. There may occasionally be fibrin thrombi that mimic a TMA. When vascular thrombosis is present, there is associated infarction and tubular necrosis. The microvascular inflammation is usually accompanied by prominent linear C4d by immunofluorescence on frozen sections (involving at least 10% of PTCs) or immunohistochemistry on paraffin sections (involving any PTC) ( Fig. 8.4 ). A positive C4d test may now substitute for a positive donor-specific antibody test in the diagnosis of active ABMR, so that a C4d-positive test, along with significant microvascular inflammation, is now considered diagnostic of active ABMR. Electron microscopy demonstrates platelets, fibrin-sludged red blood cells, and necrosis of glomerular capillaries and other vascular structures ( Fig. 8.5 ).
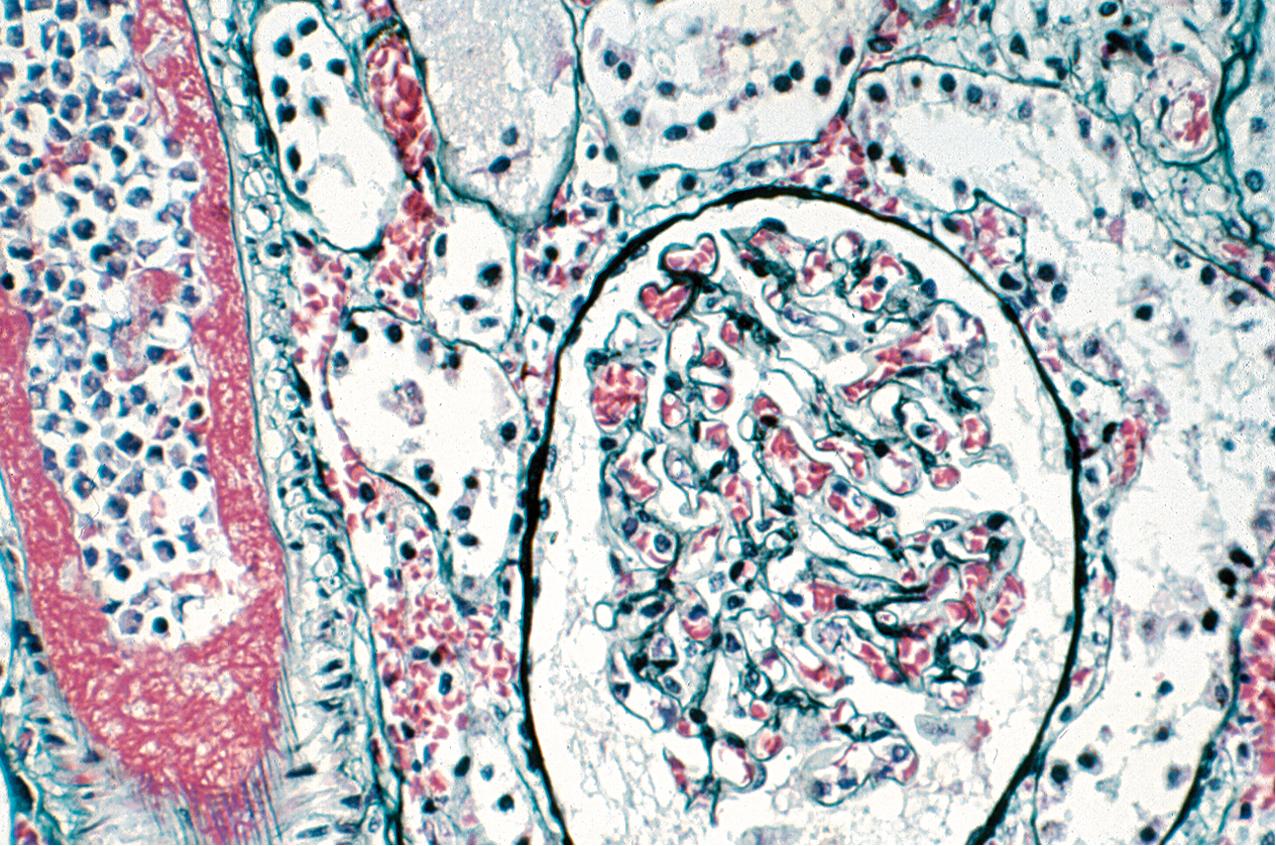
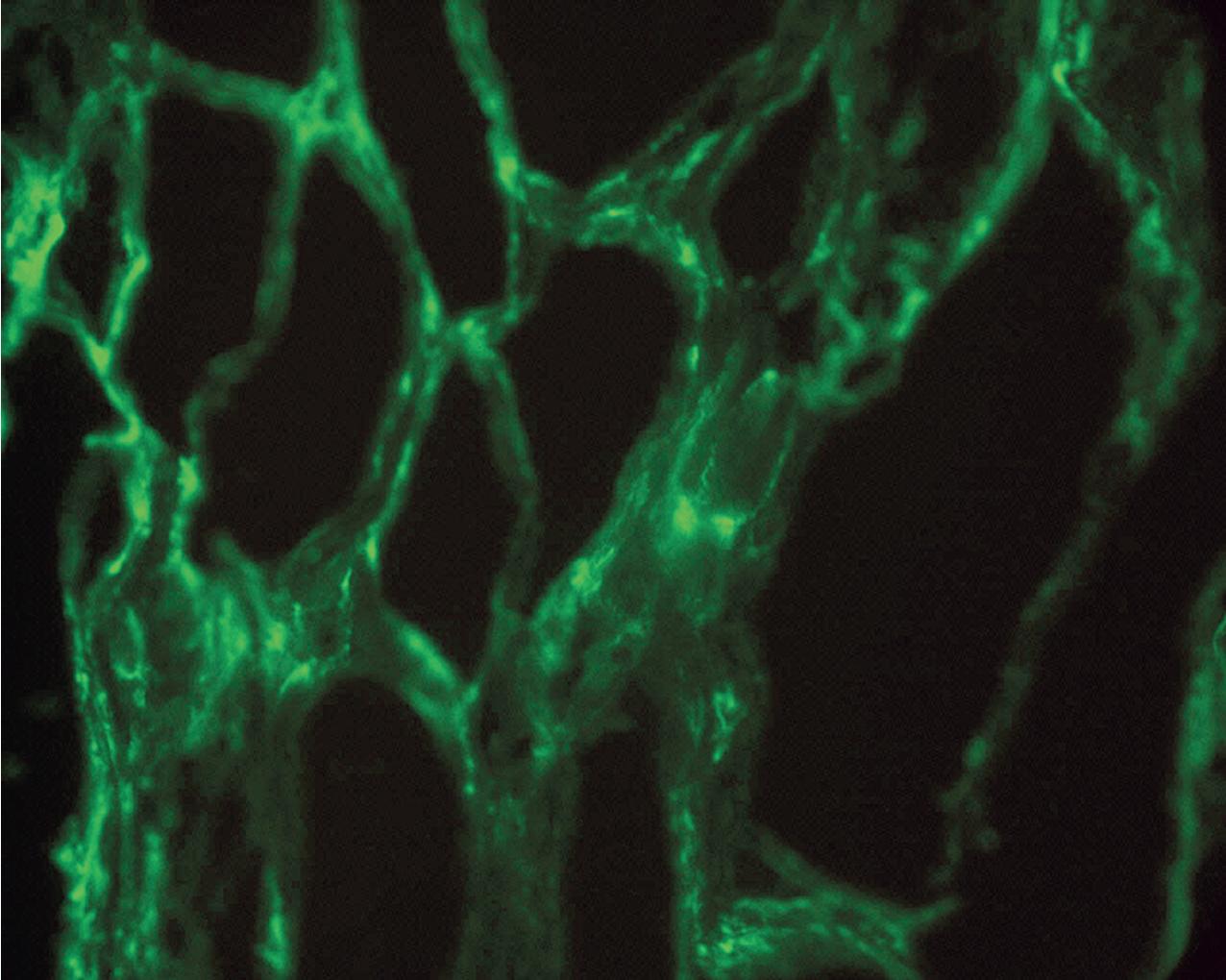
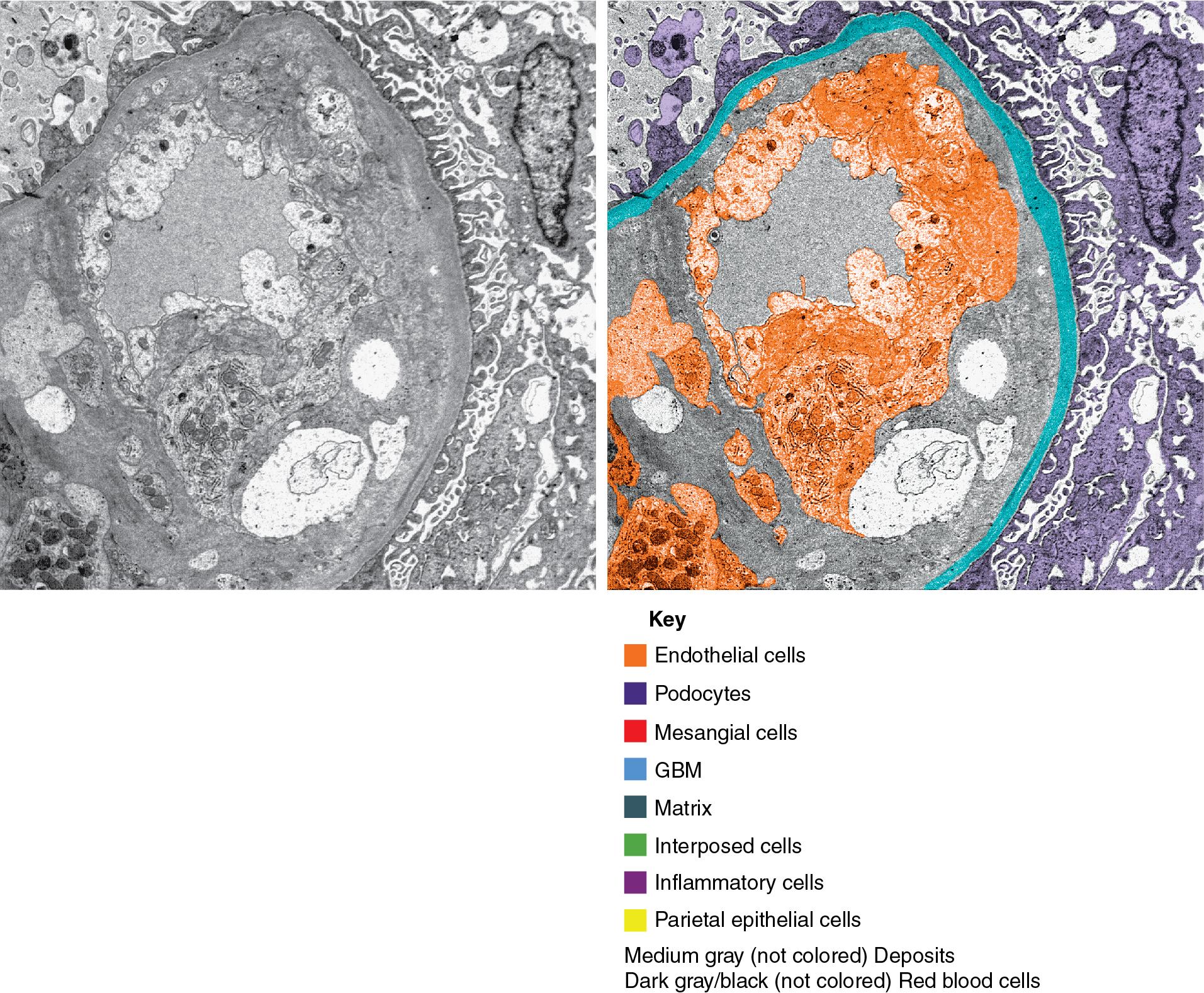
ABMR is now recognized more frequently and is not limited to the early posttransplant period. This, combined with the identification of some relatively specific markers such as PTC staining for C4d, has given rise to a more precise classification ( Tables 8.2, 8.2 A-C ). Molecular studies showing validated increased expression of gene transcripts/classifiers in the biopsy tissue strongly associated with ABMR can be helpful. The current classification also recognizes that ABMR not infrequently accompanies TCMR. Rarely, C4d may be present without morphologic evidence of rejection, and the diagnosis of ABMR should only be made if there is both C4d positivity and microvascular inflammation involving glomeruli and PTCs with g plus ptc scores equal to or greater than 2. In addition, ABMR can exist in the absence of C4d staining secondary to alloantibodies to endothelial or other antigens.
Key diagnostic features of ABMR include PTC leukocytes (peritubular capillaritis) together with glomerulitis (combined score g + ptc ≥ 2), and C4d staining of PTCs (≥10%) on frozen tissue.
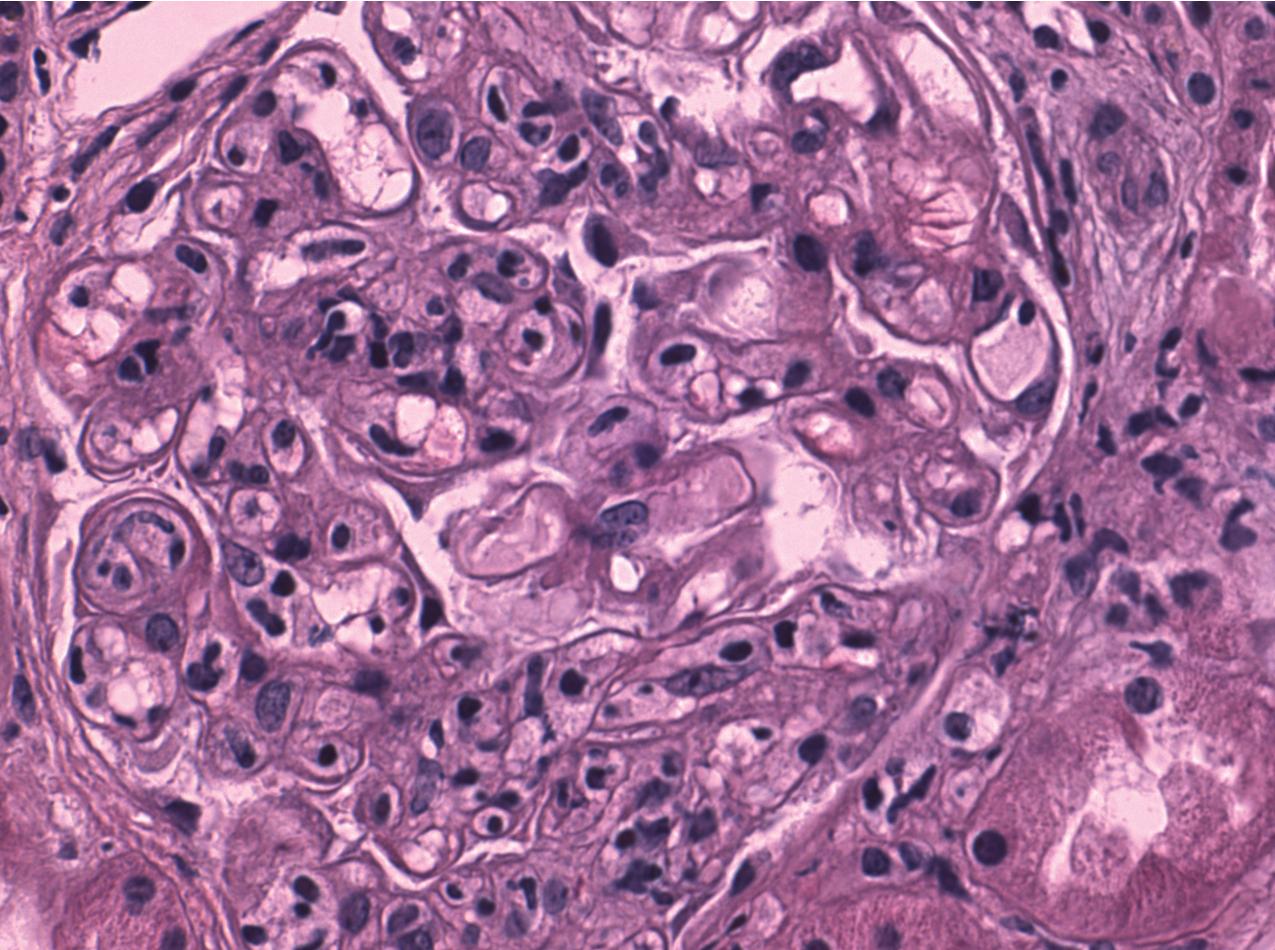
The hallmark of chronic ABMR is the glomerular lesion of transplant glomerulopathy. Glomeruli show varying degrees of lobular accentuation with an increase in mesangial matrix, mesangial interposition, and irregular thickening and double contours of basement membrane without deposits by immunofluorescence ( Figs. 8.2 and 8.6 ). This lesion is associated with significant proteinuria. By electron microscopy, there is separation of the endothelial cells from the basement membrane with the accumulation of a granular material in the subendothelial space. PTC basement membranes also show multilayering, equal to or greater than seven layers, although this is not a specific finding for chronic ABMR. The presence of concentric arterial intimal fibrosis favors ABMR if there is no prior evidence of TCMR. The exact mechanisms involved are still unknown, but humoral immunity directed against donor-specific or vascular endothelial antigens has been suggested as a likely possibility. In addition, many factors involved with progressive fibrosis in the native kidney may play a role in the transplant, such as hypertension, abnormal lipids, and reactive oxygen species, all of which can activate endothelial cells. A method of scoring has been presented at the Banff meeting, including some modified criteria, which take into account the fraction of involved glomeruli ( Table 8.4 ).
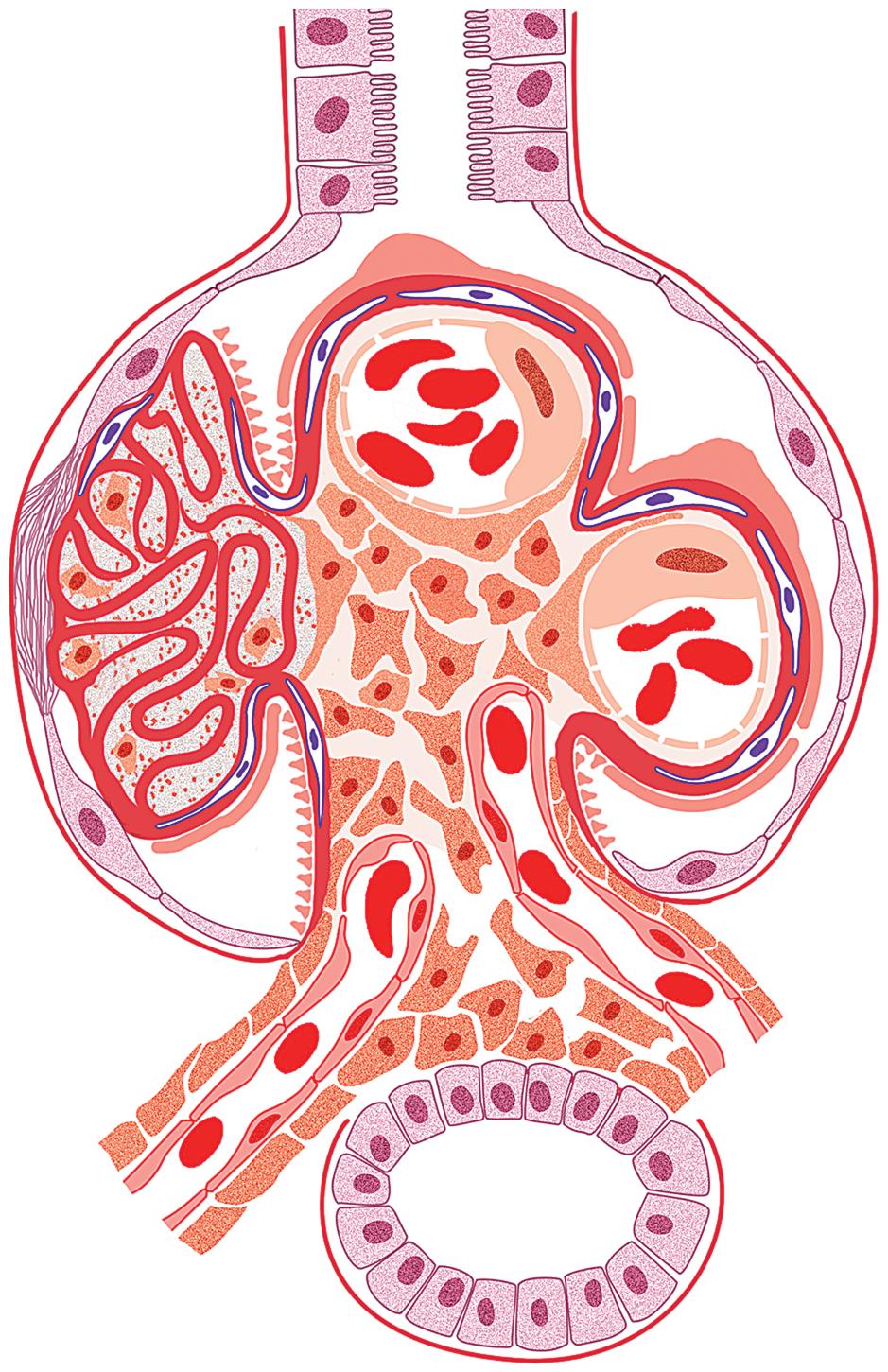
One key diagnostic feature of chronic active antibody-mediated rejection and transplant glomerulopathy is glomerular basement membrane duplication ( Fig 8.6 ).
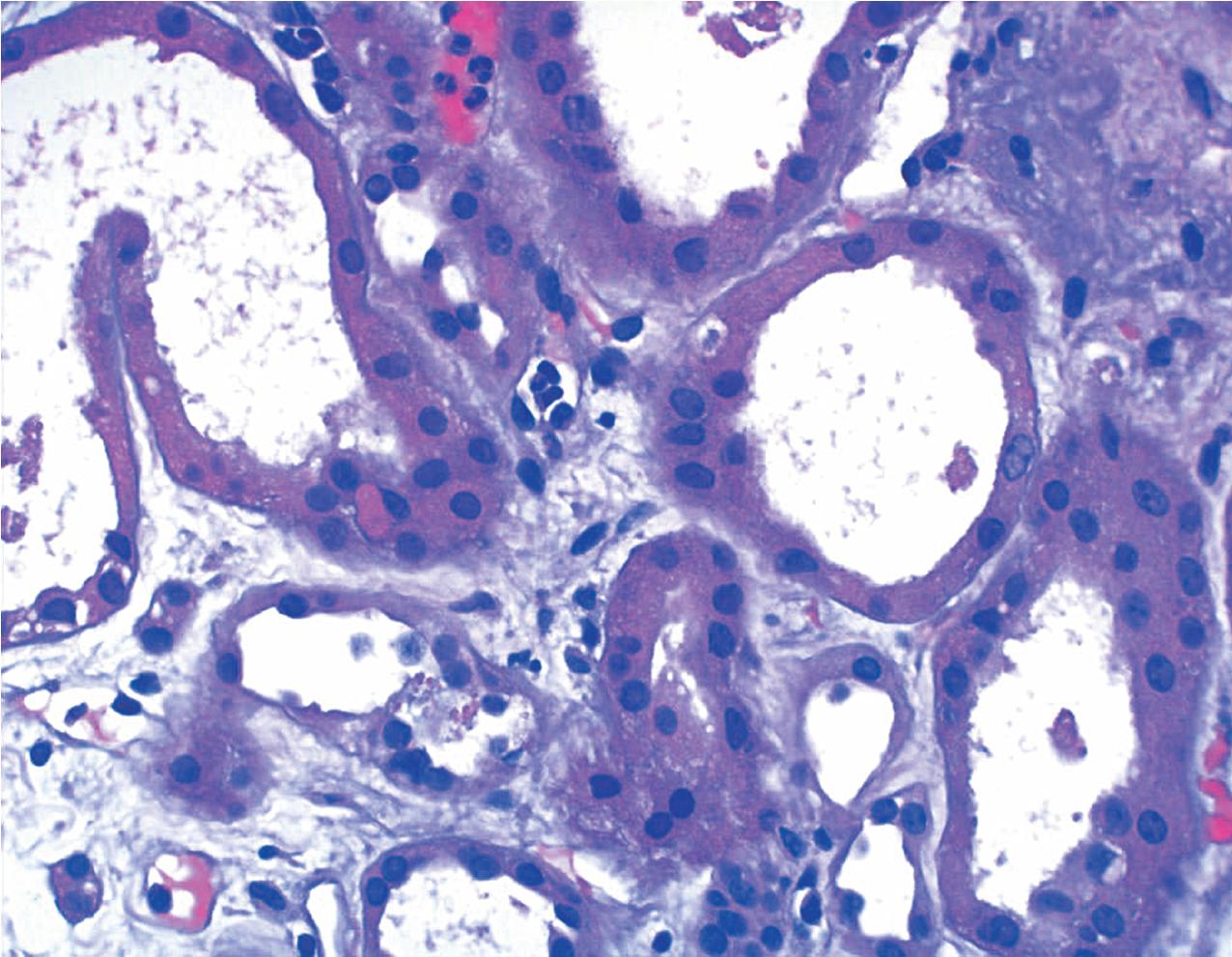
Acute TCMR, despite its name, can occur at any time during the course of the life of the allograft. It is most frequently seen during the initial months after grafting but can also be seen later in graft life, particularly when disturbances of graft therapy are incurred. In the Banff classification, the severity is determined by the degree of tubulitis and the presence or absence of intimal arteritis. This category is used to describe very mild, focal interstitial inflammation involving less than 25% of the cortical parenchyma. No intimal arteritis is present and only mild focal mononuclear cell infiltrates with rare foci of mild tubulitis defined as one to four mononuclear cells per tubular cross section present ( Fig. 8.7 ). Ongoing assessment by the Banff working groups is focused on establishing new cut-offs for borderline versus acute type I rejection. Borderline acute TCMR is now defined as interstitial inflammation involving 10% to 25% of nonscarred cortex (i.e., Banff i1) with at least mild tubulitis (t > 0). Thus cases diagnosed as borderline must have scores for mild interstitial inflammation (i1) and tubulitis of at least t1. Mild tubulitis (t1) along with greater than 25% of inflammation (i2/3) without endothelialitis is also defined as borderline. Biopsies with tubulitis but with minimal interstitial inflammation (<10% of nonscarred cortex, i0) or cases without tubulitis are no longer considered borderline. Some investigators have suggested that such mild persistent infiltrates may contribute to the progression to chronic rejection, and it must be distinguished from other potential nonimmunologic causes of interstitial inflammation, such as bacterial or viral infection or even drug hypersensitivity reactions. Of note, a lower threshold for diagnosing acute TCMR was used by the CCTT classification, requiring only greater than 5% of nonscarred cortex to show inflammation with at least three nonatrophic tubules with any degree of tubulitis within 10 high-power fields. Whether a lower threshold should be used to diagnose acute TCMR by Banff classification is being investigated by one of the Banff working groups.
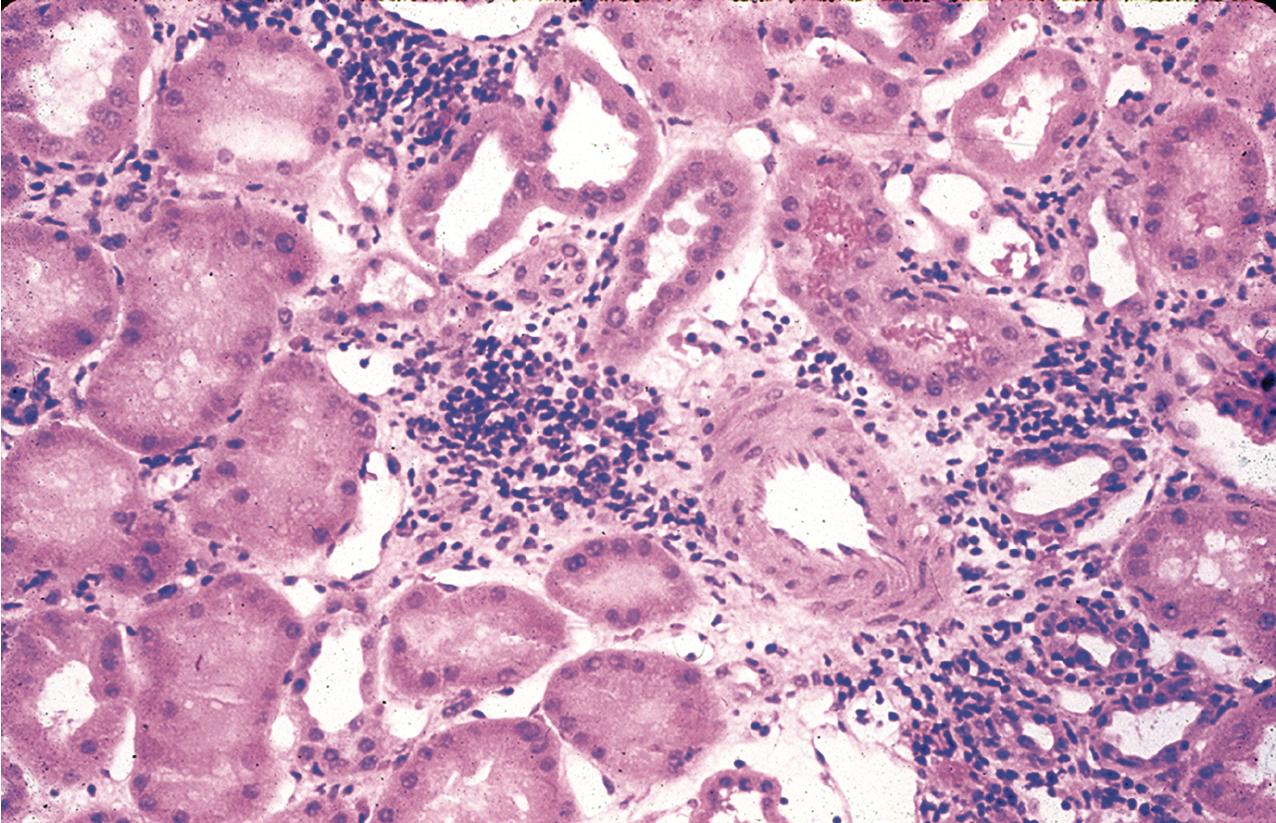
Acute TCMR can involve tubules or arteries and is divided into grades accordingly.
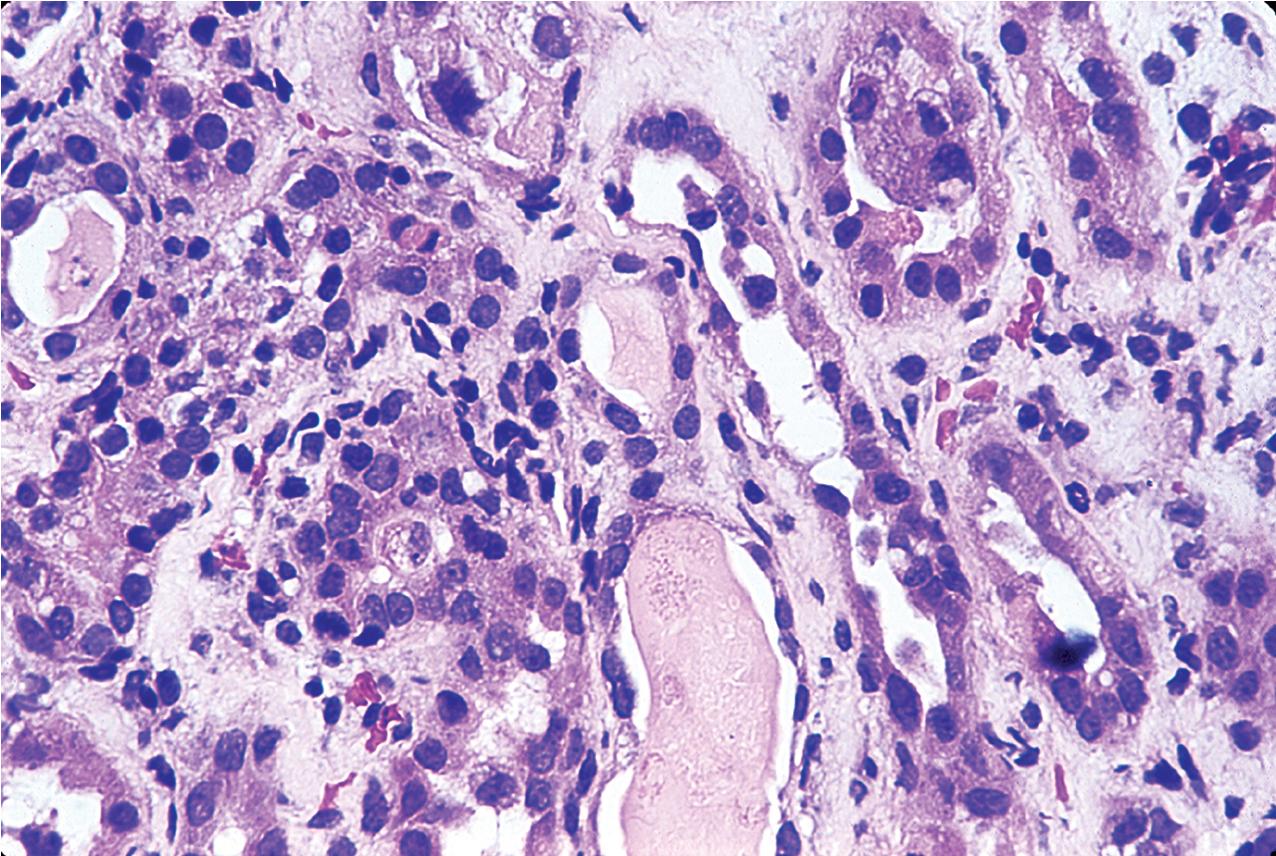
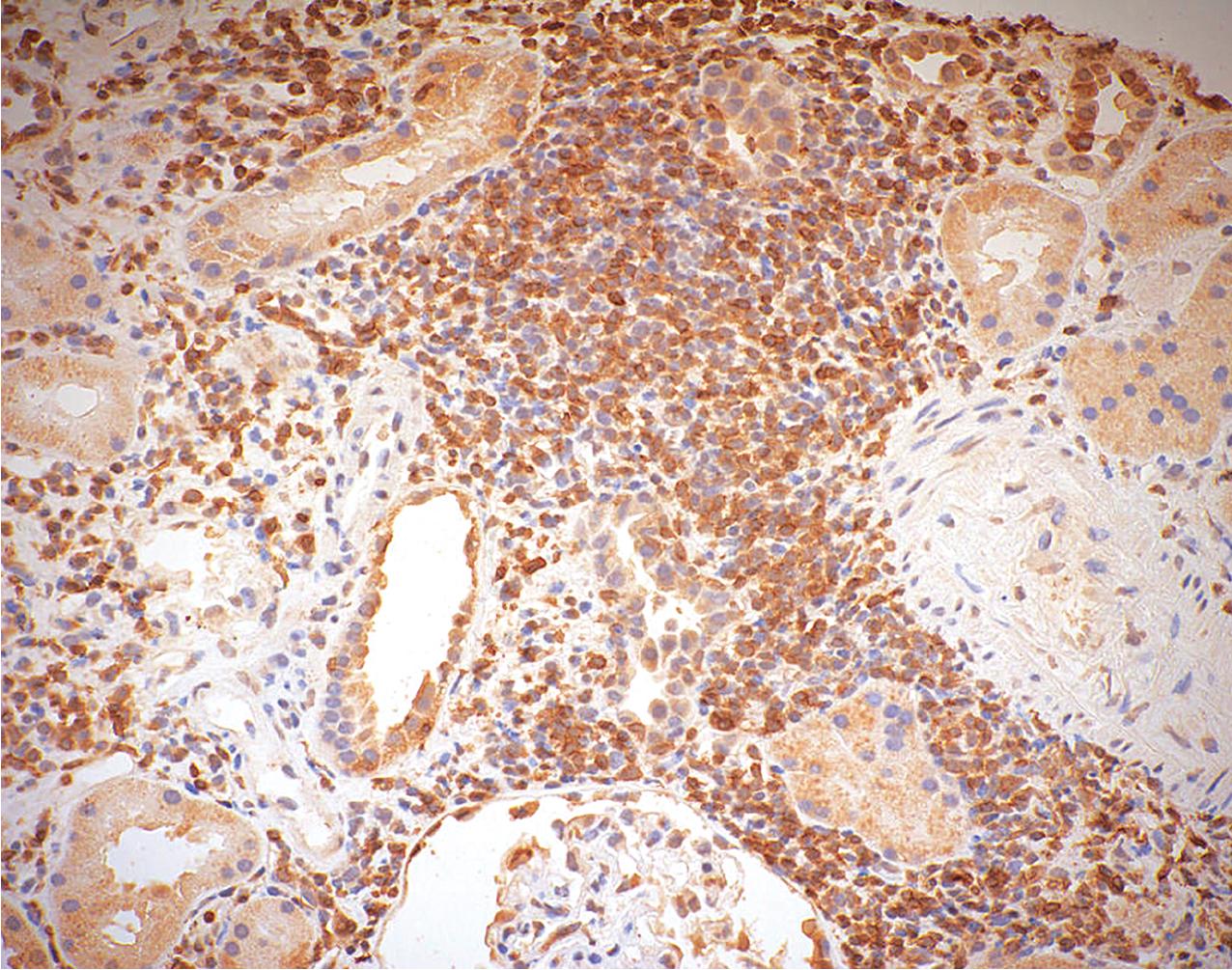
Grade I acute TCMR affects the tubules and is characterized by edema and infiltration of the interstitium by immunoblasts, T lymphocytes, plasma cells, macrophages, and a scattering of polymorphonuclear leukocytes and eosinophils. The infiltrate is generally diffuse but appears somewhat more concentrated around vessels and glomeruli. It is divided into Grade 1A and 1B. In Grade IA, greater than 25% of the nonscarred cortical parenchyma is affected (i2 or i3), and foci of moderate tubulitis with more than four mononuclear cells per tubular cross section or a group of 10 tubular cells is considered characteristic (t2) ( Fig. 8.8 ). Tubules that are severely atrophic are not considered for diagnosis of acute TCMR. In Grade IB, greater than 25% of the parenchyma is also affected (i2 or i3), and numerous foci of severe tubulitis with more than 10 mononuclear cells per tubular cross section or group of 10 tubular cells is considered characteristic (t3) ( Fig. 8.9 ). The lymphocytes include a large population of T cells identifiable by the CD3 antigen ( Fig. 8.10 ) and a greater number of cytotoxic T cells identified by the antigen CD8 than helper inducer CD4+Th cells identified by the presence of the antigen CD4. The ratio of activation antigens CD45RO and CD45RA is also of use in identifying the activity of the rejection. This degree of rejection generally has a good response to antirejection therapy.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here