Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Lung cancer is the leading cause of cancer mortality in the United States, accounting for more than 150,000 deaths each year. In addition to lung cancer, the chapter reviews primary malignant and benign pulmonary tumors more commonly encountered in clinical practice based on the latest 2015 World Health Organization (WHO) Classification of Lung Tumors ( Box 21.1 ). Pulmonary lymphoproliferative disorders and benign entities that mimic tumors in the lungs are also discussed. Posttreatment changes related to chemotherapy, radiation therapy, and immunotherapy are briefly described.
Adenocarcinoma
Squamous cell carcinoma
Neuroendocrine tumors
Carcinoid tumors
Small cell carcinoma
Large cell neuroendocrine carcinoma
Sarcomatoid carcinomas
Carcinosarcoma
Adenosquamous carcinoma
Pulmonary blastoma
Pulmonary hamartoma
Chondroma
Synovial sarcoma
Epithelioid hemangioendothelioma
Mucosa-associated lymphoid tissue lymphoma
Diffuse large cell lymphoma
Lymphomatoid granulomatosis
Metastatic Tumors
More than 200,000 patients are diagnosed with lung cancer in the United States each year. Lung cancer is the leading cause of cancer death in men and has surpassed breast cancer as the leading cause of cancer death in women. Lung cancer is one of the most common diseases of the lungs that radiologists encounter in practice. Plain radiography, computed tomography (CT), positron emission tomography (PET), and magnetic resonance imaging (MRI) all play important roles in the imaging evaluation of patients with lung cancer. Please refer to Chapters 3 and 4 for more detailed discussion of PET and MRI findings.
The strongest risk factor for the development of lung cancer is cigarette smoking. An estimated 85% to 90% of lung cancers are directly attributable to smoking. The risk is related to the number of pack-years of smoking, the age at which smoking began, and the depth of inhalation. The risk decreases with cessation of smoking but never completely disappears.
In recent years, genome-wide association studies have helped identify lung cancer susceptibility risk foci mapped to various chromosomal regions, such as 15q and 5p, in different ethnic populations.
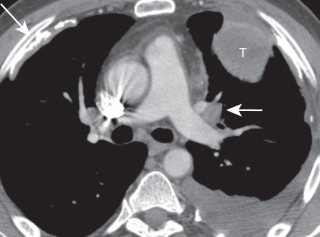
Various occupational and environmental exposures are known to increase the risk of lung cancer, including exposure to asbestos, radon, and silica as well as prior radiation. The combination of asbestos exposure ( Fig. 21.1 ) and cigarette smoking is synergistic and results in a markedly increased risk of lung cancer, particularly if asbestosis is present in the lung parenchyma.
Most of the concomitant lung diseases associated with lung cancer reflect the presence of pulmonary fibrosis, which can be diffuse, as in idiopathic pulmonary fibrosis ( Fig. 21.2 ), or localized, as in tuberculosis. The presence of emphysema on CT has also been linked to increased lung cancer risks among smokers and never-smokers.

Only 10% of patients with lung cancer are asymptomatic. Typically, symptoms are caused by central tumors that result in obstruction of a major bronchus ( Box 21.2 ). This leads to cough, wheezing, hemoptysis, and postobstructive pneumonia. Invasion or compression of adjacent thoracic structures by the tumor may result in pleuritic or chest wall pain, Pancoast syndrome, and symptoms related to obstruction of the superior vena cava. Occasionally, patients may have symptoms that result from distant metastases (e.g., a seizure caused by brain metastases).
10% of patients
Hemoptysis
Cough
Fever caused by postobstructive pneumonia
Pleuritic and chest wall pain
Pancoast syndrome (see Box 21.8 )
Superior vena cava syndrome
Dyspnea
Facial swelling
Arm swelling
Chest pain
Clubbing
Hypertrophic pulmonary osteoarthropathy
Migratory thrombophlebitis
Ectopic hormone production
Adrenocorticotropic hormone: Cushing syndrome
Antidiuretic hormone: hyponatremia
Parathyroid hormone–related protein: hypercalcemia
Neurologic symptoms
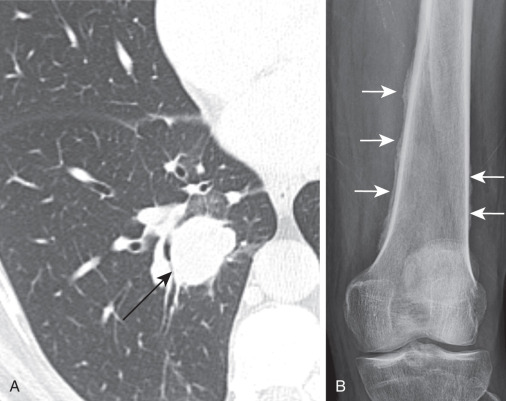
Several paraneoplastic syndromes are associated with lung carcinoma, including clubbing; migratory thrombophlebitis; and ectopic hormone production, including Cushing syndrome from adrenocorticotropic hormone (ACTH) production, hyponatremia associated with syndrome of inappropriate antidiuretic hormone secretion (SIADH), and hypercalcemia caused by excessive parathyroid hormone–related protein (PTHrP) production by the tumor. Hypertrophic pulmonary osteoarthropathy consists of periosteal new bone formation that typically involves the bones of the lower arms and legs and can be painful ( Fig. 21.3 ). There are also a variety of neurologic paraneoplastic syndromes.
Lung cancer is traditionally divided into non–small cell lung cancer (NSCLC) and small cell lung cancer (SCLC) based on histologic features and differences in treatment strategy. NSCLC in the early stages can be treated with surgery alone. The most common NSCLC include adenocarcinoma and squamous cell carcinoma (SCC). SCLC usually presents in the late stage and requires treatment with a combination of chemotherapy and radiation therapy. Based on the latest 2015 WHO classification (see Box 21.1 ), SCLC, large cell neuroendocrine carcinoma (LCNEC), and carcinoid tumors all belong to the category of neuroendocrine tumors.
Adenocarcinoma ( Box 21.3 ) is the most frequent histologic type and accounts for approximately 50% of all lung cancers. It is the most common histologic type in women and in nonsmokers. Typically located in the periphery of the lung, these lesions may not produce symptoms and are found incidentally on routine chest radiography. One of the more unusual symptoms of adenocarcinoma is bronchorrhea, which is the production of more than 100 mL/day of watery sputum.
Most common cell type
Predominant cell type in women and nonsmokers
Occasionally asymptomatic
Bronchorrhea in a small subset of patients
Pulmonary fibrosis is a risk factor
Can be slow growing
Peripheral, subpleural location
±Mucin
Peripheral location
Solitary nodule or mass
Spiculated, lobulated, or ill-defined border
±Air bronchogram
Solid, ground-glass, or mixed attenuation
Consolidation
Multiple nodules
Adenocarcinomas typically show slow growth and tend to metastasize early. They can occur in areas of preexisting pulmonary fibrosis. They are most frequently peripheral and subpleural in location and may arise endobronchially.
The classification of lung adenocarcinoma by the 2011 International Association for the Study of Lung Cancer (IASLC), American Thoracic Society (ATS), and European Respiratory Society (ERS) includes a multidisciplinary diagnostic approach using data from pathology, imaging, and molecular biology. The IASLC/ATS/ERS system introduces the use of clearer terminology with respect to the degree of growth along the alveolar surface (lepidic growth) and invasive components to define pre-invasive and invasive lesions. Preinvasive lesions include atypical adenomatous hyperplasia (AAH) and adenocarcinoma in situ (AIS), both defined as lesions with lepidic growth. AAH is a localized, small (≤5 mm) proliferation of mildly to moderately atypical type II pneumocytes or Clara cells lining alveolar walls. AIS is a localized small (≤3 cm) adenocarcinoma with lepidic growth without evidence of stromal, vascular, or pleural invasion. AAH and AIS are pathologic entities considered to be part of a spectrum and cannot be reliably differentiated on cytology. The term bronchioloalveolar carcinoma (BAC) is no longer used.
Invasive lesions include minimally invasive adenocarcinoma (MIA) and invasive adenocarcinoma. MIA is defined as a predominantly lepidic lesion lacking necrosis and invasion of lymphatics, blood vessels, or pleura and measuring less than 3 cm with an invasive component measuring no more than 5 mm in any one location. Invasive adenocarcinomas are further classified into histologic subtypes according to the predominant histology present: lepidic, acinar, papillary, micropapillary, or solid-predominant patterns. For example, lepidic-predominant adenocarcinoma (LPA) is defined as a lepidic lesion that may have necrosis, invade lymphatics or blood vessels, and the focus of invasion is greater than 5 mm. The nonmucinous adenocarcinomas are more common than mucinous forms. The term invasive mucinous adenocarcinoma has replaced mucinous bronchoalveolar carcinoma .
Various gene mutations have been identified in lung adenocarcinoma. There is a strong association between KRAS mutations in white smokers. Mutations in the epidermal growth factor receptor (EGFR) are more often seen in adenocarcinomas of nonsmokers and Asian women. Rearrangements of the gene encoding anaplastic lymphoma kinase (ALK) are uncommon, mostly seen in young never- or light smokers with adenocarcinoma. Testing of adenocarcinoma for various mutations has become increasingly important because of availability of targeted drug therapies and prognostic significance.
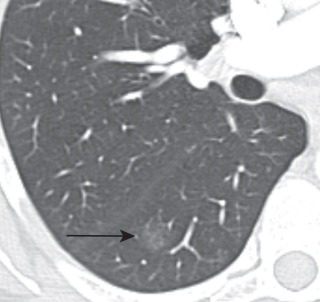
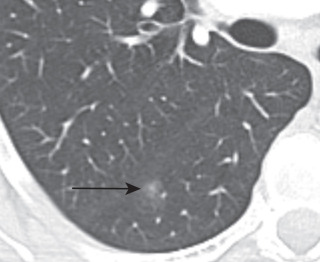
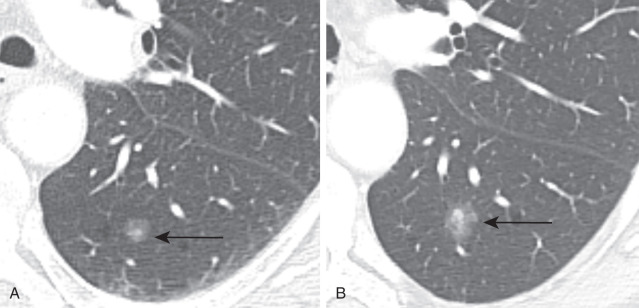
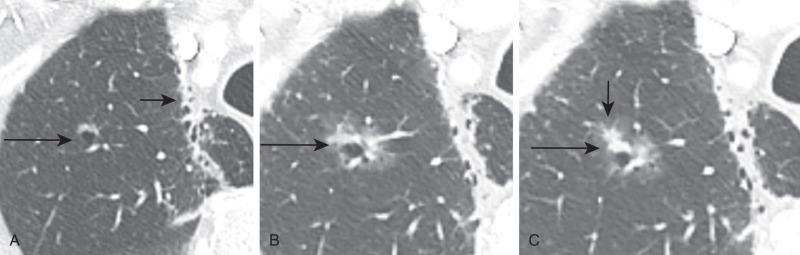
Adenocarcinoma may manifest distinct radiologic patterns. The most common is a solitary peripheral nodule or mass often with lobulated, spiculated, and ill-defined borders. On CT, they may present with pure ground-glass, solid, or mixed (both ground-glass and solid) attenuation. AAH and AIS present as pure ground-glass nodules ( Figs. 21.4 and 21.5 ). MIA is usually of ground-glass or mixed attenuation. Correlation of CT features with histopathologic diagnosis using the IASLC/ ERS/ATS system is still evolving, and studies describing CT manifestations of mixed attenuation nodules relating to the more common nonmucinous forms of adenocarcinoma are summarized in Table 21.1 . In contradistinction, the less common mucinous form of AIS, MIA, and LPA and other forms of invasive adenocarcinomas can manifest as solid nodules. The soft tissue component of mixed attenuation lesions can represent the invasive component or fibrosis and alveolar collapse ( Fig. 21.6 ). The degree of invasion is reported to correlate directly with the size of the soft tissue component on CT. The solitary nodule is associated with an excellent prognosis when it is resected in early-stage disease. Bubbly internal lucencies termed pseudocavitation is caused by alveoli or bronchi spared by tumor infiltration ( Fig. 21.7 ). An air bronchogram may be identified on standard radiography and CT.
| IASLC/ATS/ERS 2011 | Computed Tomography Features |
|---|---|
| Atypical adenomatous hyperplasia | Ground-glass nodule |
| Adenocarcinoma in situ | Ground-glass nodule (possible solid component) |
| Minimally invasive adenocarcinoma | Ground-glass nodule Part solid nodule |
| Lepidic-predominant adenocarcinoma | Part solid nodule Solid nodule |
| Invasive adenocarcinoma classified by predominant subtype | Part solid nodule with ↑↑ solid component Solid nodule |
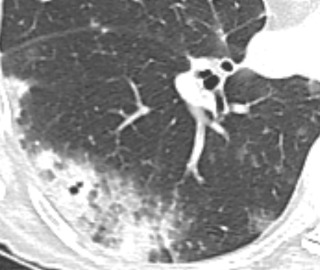
The second appearance is an area of consolidation, which occurs in approximately 20% of cases ( Fig. 21.8 ). This appearance can mimic pneumonia, which is more common than lung cancer. Differential considerations for chronic consolidation also include lymphoma, organizing pneumonia, and chronic infection. The consolidation may be associated with nodules in the same lobe or in other lobes of either lung, reflecting tumor dissemination via the tracheobronchial tree.
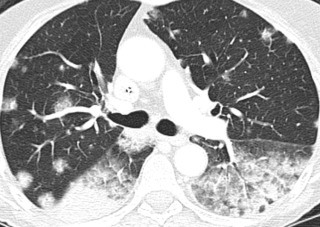
The third appearance is that of multiple nodules and masses scattered throughout the lungs ( Fig. 21.9 ).
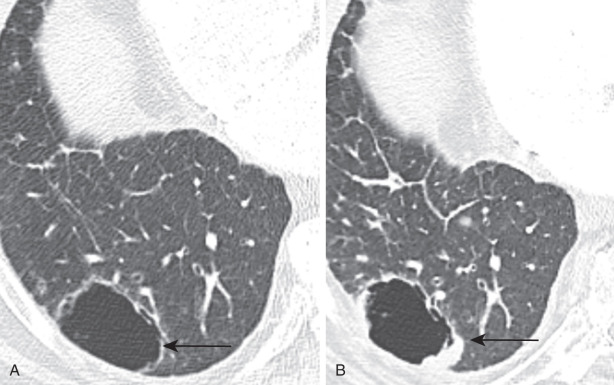
There is also increased awareness in recent years that adenocarcinoma and SCC can initially present as cystic lesions with progressive increase in wall nodularity, thickness, or size ( Fig. 21.10 ).
Adenocarcinoma can have variable degree of fludeoxyglucose (FDG) avidity. Pre- or minimally invasive, ground-glass attenuation, and small-sized lesions are known to have minimal or absent FDG uptake on PET. The degree of FDG avidity pretreatment has been found to have prognostic value for recurrence and survival in those with stage I NSCLC treated with stereotactic body radiotherapy (SBRT). Please see additional detail regarding PET in Chapter 4 .
Squamous cell carcinoma represents approximately 30% of all lung cancers ( Box 21.4 ). There is a strong association with cigarette smoking. SCC is the histologic type most commonly associated with hypercalcemia caused by PTHrP production by the tumor.
One third of all lung cancers
Hypercalcemia related to parathyroid hormone–related protein production
Central, endobronchial location
Local metastases to lymph nodes
Central necrosis
Two thirds in central location
Endobronchial lesion best seen on computed tomography
Postobstructive atelectasis or pneumonia of the lung or lobe
One third in peripheral location
Thick-walled, cavitary mass
Solitary nodule
Squamous cell carcinoma often arises in areas of squamous metaplasia, and there appears to be an orderly progression of alterations in bronchial mucosa in cigarette smokers from squamous metaplasia to invasive carcinoma. Typically, these tumors occur in main, segmental, or subsegmental bronchi, and they grow endobronchially. Bronchial wall invasion occurs with growth proximally along the bronchial mucosa. Spread to regional lymph nodes is common and may occur by direct extension. Central necrosis is a common feature. Histologic features typical for SCCs include the formation of keratin pearls and intercellular bridges.
The radiologic presentation depends on the location of the tumor. The most common presentation is a central mass in the hilar or perihilar region obstructing the bronchus ( Fig. 21.11 ). Involvement of the central bronchus may range from focal thickening to complete occlusion. When the lesion is small, the tumor may not be evident on chest radiography, but the bronchial wall abnormalities are more readily detected on CT. Atelectasis or postobstructive pneumonia is usually identified distal to the obstructed bronchus. Any patient presenting with lobar collapse and signs of infection should be followed radiographically to complete resolution with reexpansion of the involved lobe. Persistent atelectasis strongly suggests a central lung carcinoma.
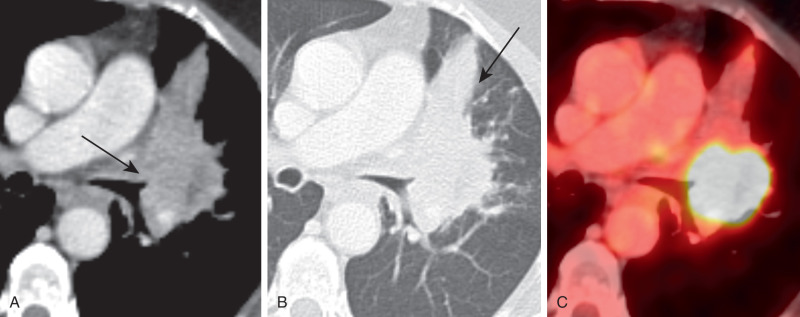
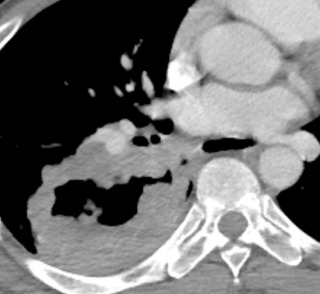
Approximately one third of SCC occurs in the lung periphery. The most characteristic appearance is a thick-walled cavitary nodule or mass that usually does not contain an air–fluid level. The diameter ranges from 2 to 10 cm ( Fig. 21.12 ). The cavitary malignancy may be indistinguishable from a lung abscess based on imaging. A solitary nodule or mass without cavitation can occur in the periphery of the lung parenchyma.
Squamous cell carcinoma is usually FDG avid, although the central necrotic region can be photopenic. PET, MRI, or dual-energy CT is often helpful in distinguishing central tumor from adjacent postobstructive atelectasis.
Superior sulcus tumor ( Box 21.5 ) is not a distinct pathologic entity. It refers to NSCLC arising in the lung apex and is also known as Pancoast tumor. In recent series, adenocarcinomas have accounted for the majority of superior sulcus tumors, although in earlier series, SCC were predominant. Superior sulcus tumors often cause Pancoast syndrome, which is pain in the shoulder girdle and ulnar nerve distribution of the arm and hand. Horner syndrome, characterized by ipsilateral anhidrosis of the face, miosis, and ptosis with narrowing of the palpebral fissure caused by paralysis of the Muller muscle, is seen when the stellate ganglion is involved. These tumors typically invade the chest wall and extend into the neck. Local extension may result in involvement of the brachial plexus, spread to the vertebral bodies and spinal canal, involvement of the sympathetic ganglion, and anterior extension with invasion of the subclavian artery. In patients with potentially resectable tumor, multimodality treatment in which surgery (en bloc resection of the tumor and chest wall) is combined with pre- or postoperative radiation therapy, with or without chemotherapy now forms the standard of care for patients with superior sulcus tumors .
Pain in shoulder and ulnar nerve distribution
Horner syndrome: stellate ganglion involvement
Bone destruction (upper thoracic vertebral bodies, ribs)
Atrophy of hand muscles
Most common: adenocarcinoma
Apical mass or asymmetric thickening
Bone destruction
Magnetic resonance imaging best for brachial plexus involvement
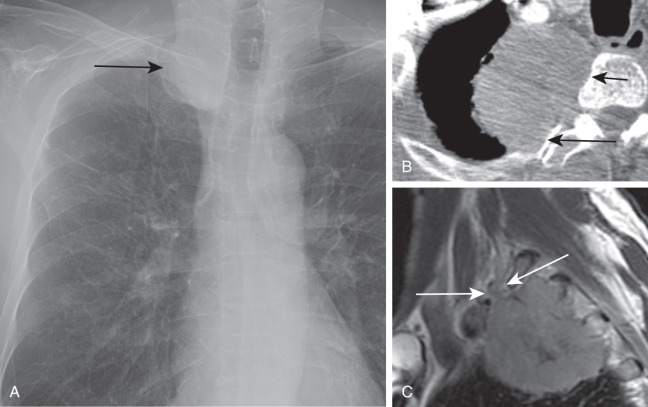
Brachial plexus involvement is suspected when tumor infiltration of the interscalene triangle and around the subclavian artery is observed on CT. MRI is the preferred modality for evaluating superior sulcus tumors because it allows visualization of structures at the apex of the thorax in multiple planes ( Fig. 21.13 ).
Small cell lung carcinoma, the most aggressive form of lung cancer, is characterized by rapid growth and early metastases, which are seen in two thirds of patients at the time of presentation ( Box 21.6 ). It is associated with the poorest prognosis and has the strongest association with cigarette smoking. It accounts for approximately 15% to 20% of all lung cancers.
Most aggressive subtype
Strongest association with smoking
Poorest survival
Accounts for 15% to 20% of cancers
Treated with chemotherapy
Inappropriate ADH production, ectopic ACTH
Central tumor
Tumor necrosis
Hilar or perihilar mass
Massive adenopathy, often bilateral
Lobar collapse
Peripheral nodule
ACTH, Adrenocorticotropic hormone; ADH, antidiuretic hormone.
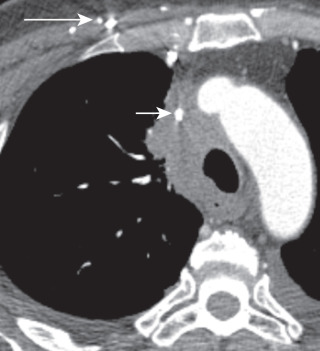
Bulky lymphadenopathy associated with SCLC can cause compression of the superior vena cava (SVC) and lead to SVC syndrome (facial swelling, dyspnea, and headache) ( Fig. 21.14 ). SCLC is also associated with Cushing syndrome (central obesity, hypertension, glucose intolerance, plethora, and hirsutism) and SIADH, resulting in hyponatremia and serum hyposmolarity.
When found in the early stage as an isolated pulmonary nodule, SCLC can be resected and is associated with a better prognosis. However, most patients present in the late stage and are often managed with chemotherapy with or without radiation. The long-term survival is extremely poor because of a high rate of recurrence. Even when treated, the median survival time is 9 to 18 months.
Small cell lung carcinoma is a high-grade neuroendocrine tumor with more than 10 mitoses per 2 mm 2 that manifests histologically as sheets of small oval to slightly spindle-shaped cells with scant cytoplasm and hyperchromatic nuclei with small to absent nucleoli. SCLC is characterized by extensive tumor necrosis and hemorrhage.
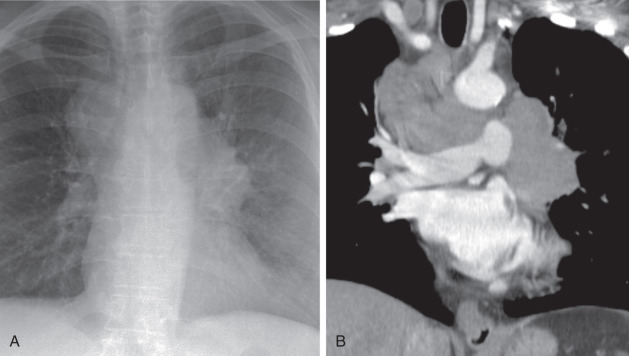
The primary tumor is typically small and often central in location with extensive and bulky hilar and mediastinal adenopathy ( Fig. 21.15 ). Rarely, SCLC manifests as a small, peripheral, solitary nodule. SCLC tends to exhibit avid FDG uptake.
Large cell neuroendocrine carcinomas (LCNEC) account for 3% of lung cancers in surgical series and have a strong association with cigarette smoking. They are characterized by rapid growth, early metastases, and a poor prognosis.
Similar to SCLC, LCNEC is a high-grade neuroendocrine tumor with more than 10 mitoses per 2 mm 2 . It is characterized by large cells with low nuclear to cytoplasmic ratio. Typically, these tumors are peripheral in location. On gross inspection, they are large, with mean diameter of 3 to 4 cm with areas of necrosis.
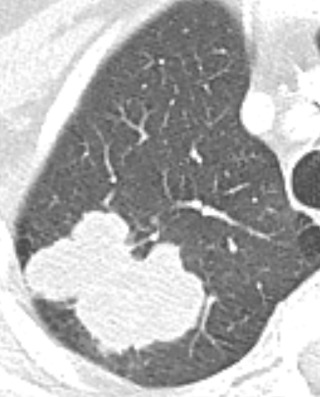
The lesions are usually peripheral and quite large ( Fig. 21.16 ).
Carcinoid tumors are low-grade malignant neoplasms that represent between 1% and 2% of all primary pulmonary malignancies ( Box 21.7 ). Males and females are equally affected over a wide age range. The median age is 50 years. There is no association with cigarette smoking. Patients may present with cough and hemoptysis. Carcinoid tumors may be associated with ectopic hormone production, specifically ACTH. However, these tumors do not produce the clinical carcinoid syndrome unless liver metastases are present. Typical carcinoid (TC) tumors rarely metastasize, but atypical carcinoid (AC) tumors metastasize in 40% to 50% of patients. AC tumors are usually peripheral in location and account for 10% of carcinoid tumors. They may be associated with involvement of hilar and mediastinal lymph nodes and distant metastases, such as the liver or bone.
Median age at diagnosis: 50 years
Men and women equally affected
Cough, hemoptysis
Good prognosis
Typical carcinoid: <2 mitoses per 2 mm 2
Atypical carcinoid: 2–10 mitoses per 2 mm 2
Rare; 10% of carcinoid tumors
Lymph node involvement and distant metastases more likely
Neurosecretory granules
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here