Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Venous thromboembolism (VTE) has protean clinical manifestations and is a continuum from deep venous thrombosis (DVT) to the main life-threatening complication of pulmonary embolism (PE).
Left untreated, patients with a diagnosis of PE have a significant recurrence rate and mortality; this can be considerably reduced with anticoagulation.
Diagnostic and treatment decisions rely on good risk stratification, preferably using validated scores such as the Wells scores and the pulmonary embolism rule-out criteria (PERC rule) to avoid excessive investigation and unnecessary therapy as well as to guide the management and disposition of those with PE.
The investigative algorithm should include electrocardiography (ECG), chest x-ray (CXR) and measures of oxygenation (plus other investigations for alternative diagnoses) to stratify risk, guide radiological testing and search for alternative causes. Computed tomographic pulmonary angiography (CTPA) is the most commonly used diagnostic test. Several further modalities may be utilized—including validated combinations of D-dimer (as well as age-adjusted D-dimer), lower limb ultrasound, ventilation/perfusion (V/Q) scan, single-photon emission computed tomography (SPECT or V/Q SPECT) and CTPA—to refine probability. Which are chosen will depend on local resources.
The decision to treat is based on reaching a diagnostic threshold (>80% chance of PE), where PE morbidity/mortality outweighs the risks of anticoagulation.
In massive (clinically unstable) PE, transthoracic or, if available, trans-oesophageal echocardiography is the recommended initial investigation.
Novel oral anticoagulants (NOACs), now being referred to as direct oral anticoagulants (DOACs), are the recommended first-line treatment for most cases of PE, although in some circumstances sub-segmental PE (SSPE) may be appropriately left untreated.
Thrombolysis (or embolectomy if thrombolysis is contraindicated or has failed) is indicated only for haemodynamically unstable/shocked PE. Stable patients with evidence of right ventricular strain should be monitored vigilantly, with thrombolysis initiated urgently if they deteriorate clinically.
Prognosis can be usefully assessed and used to guide disposition and management, with combined clinical and investigation parameters using validated scores, such as the Simplified Pulmonary Embolism Severity Index (SPESI).
Pulmonary embolus (PE) is the third most common cardiovascular disease, more often seen in the elderly with co-morbidity (cancer, trauma, immobility, surgery or severe medical disease). Historically, when diagnosed clinically and untreated, it has a high mortality. Treating PE with anticoagulation reduces in-hospital mortality to between 4% and 12%.
The diagnosis and management of PE is often difficult. It requires careful clinical assessment, documented risk stratification (preferably a validated score such a Wells, PERC, etc.) and appropriate selection of diagnostics, none of which are truly definitive. Possible alternative serious conditions should always be considered. All PE diagnostic tests (including D-dimer) need risk assessment to ensure safe and appropriate usage (see risk assessment, further on). Generally, with PE risk below 5% (most low-risk emergency patients), an alternative diagnosis should be sought, with PE investigation pursued if no definitive alternative is found. Overly aggressive radiological investigation in 5% risk populations potentially finds more false-positive venous thromboembolism (VTE) than real PE/deep venous thrombosis (DVT). The diagnostic threshold is 70% to 80% PE probability, where treatment benefits clearly outweigh treatment risks. Most diagnostic guidelines use Wells, and/or PERC (if low-risk Wells) and/or D-dimer (if low-risk Wells and positive PERC) for most emergency department (ED) patients, followed by computed tomographic pulmonary angiography (CTPA) or occasionally nuclear ventilation/perfusion scan (V/Q) for definitive diagnosis. Venous ultrasound (US or CT venography) may assist after indeterminate imaging. Echocardiography, if available, is useful for PE risk stratification and particularly for managing unstable cases ( Fig. 5.5.1 ).
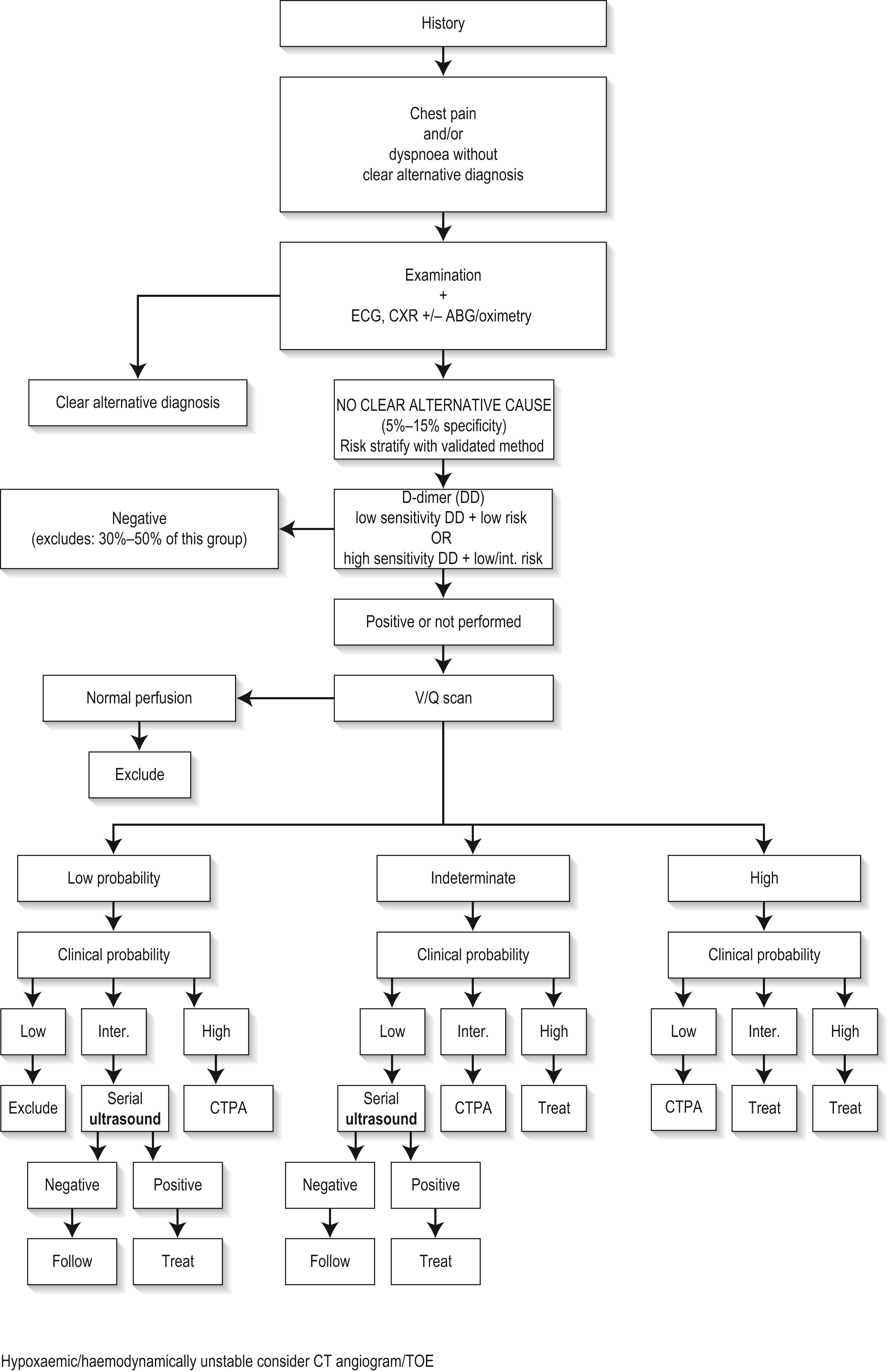
Almost all cases of diagnosed PE will be anticoagulated, the majority on direct oral anticoagulants (DOACs) or novel oral anticoagulants (NOACs), some with heparin/warfarin. Thrombolysis or embolectomy (when thrombolysis is contraindicated or fails) is indicated in shocked patients (massive PE, blood pressure [BP] <90 mm Hg for >15 minutes or if patient is requiring inotropic support due to PE); thrombolysis is not routinely administered for submassive PE. Disposition decisions (e.g. home therapy, intensive monitoring) are assisted by prognostic decision making based on validated scores (e.g. the Simplified Pulmonary Embolism Severity Index [SPESI]) or PE severity markers (e.g. biomarkers such as brain natriuretic peptide [BNP], troponin, etc.) or echocardiography.
Up to 30% of PEs, particularly ED presentations, are idiopathic. Most risk factors act via more than one item in Virchow’s triad (e.g. vessel wall injury, venous stasis or hypercoagulable states). The major risk factors associated with secondary (provoked) PE are as follows:
Surgery or trauma; particularly pelvis/ lower abdomen, lower limb or central nervous system (CNS) (provokes 15% to 30% of PE)
Cancers (15% to 25%)
Systemic disease with immobilization (>24 hours bed rest), particularly heart disease/strokes (15% to 25%)
A history of DVT/PE (particularly unprovoked, 5% to 15%).
PE incidence rises exponentially with age, with those above 85 years of age having 60 times the incidence of 18- to 25-year-olds, mainly driven by co-morbidity. Hypercoagulable states (e.g. antithrombin III deficiency, factor V Leiden, antiphospholipid syndrome, protein C/S deficiencies etc.), indwelling venous devices, hormone contraception or therapy, obesity, later pregnancy/post-partum, many vasculitic/autoimmune diseases, smoking, use of non-steroidal anti-inflammatory drugs and long-haul air travel increase the risk of PE.
The greatest preventable cause of DVT/PE is surgery. Stasis from illness-related bed rest—particularly cancer, stroke and cardiopulmonary disease—are also common. Low-dose preventive low-molecular-weight heparin (LMWH) or NOACs/DOACs make surgery safer and more effective and improve outcomes for high-risk hospitalized medical patients. Thrombo-prophylaxis risk assessment should be performed and started as early as possible.
The history provides important clues for diagnosing PE. Virtually all PE patients present with either recent-onset dyspnoea (particularly if rapid/recurrently episodic), chest pain (any type), or both (sensitivity 97%, specificity 10%). Syncope either with respiratory symptoms or signs (even if transient), where VTE risk is high, often suggests severe disease. DVT symptoms should be sought in all patients. Haemoptysis has some predictive value but is uncommon. Associated risk factors, divided into major and minor, increase PE likelihood and should be documented and incorporated into risk assessments.
Other symptoms (e.g. clear musculoskeletal symptomatology) are less important for diagnosis but may suggest or exclude other causes, particularly in low-risk patients. No single symptom (or sign) has the sensitivity or specificity to either establish or exclude the diagnosis.
Physical signs confirming PE (e.g. persistent unexplained tachycardia [>100] at rest) are rare but can significantly increase PE diagnostic probability; this is incorporated into most prediction rules. Leg (or arm) DVT signs (e.g. a swollen, oedematous leg with pain in the venous distribution or significant thrombophlebitis) increase PE risk and mandate imaging. Other features include tachypnoea (50% to 80%), cough (10% to 20%), mild fever (<38.5°C), wheeze and pleural effusion/rub; but these are not discriminatory for PE. Occasionally elevated jugular venous pressure (JVP), a loud cardiac S(P)2 heart sound or a pulmonary systolic murmur may suggest right ventricular strain.
The pre-test probability (PTP) calculation used to decide on investigation strategies is based on history, examination and investigations, including CXR, arterial blood gas analysis/oximetry, ECG and investigations for alternative diagnoses. For non-experts, this estimate is best made using validated scoring systems. The best validated and most widely disseminated is the Wells rule ( Table 5.5.1 ). If the Wells score is low (<2) the patient should have a PERC score ( Box 5.5.1 ) applied to decide whether D-dimer is indicated; if PERC is negative PE is excluded (<2% PE incidence) without need for D-dimer testing. If PERC is positive in this low-risk Wells group an age adjusted D-dimer is performed followed by CTPA/VQ/SPECT if positive. If Wells is higher (>4), then the D-dimer is not performed and the patient progresses directly to CTPA/VQ/SPECT.
| Clinical signs of DVT | 3.0 |
|---|---|
| Pulse rate >100 (at rest) | 1.5 |
| Immobilized ≥3 days | 1.5 |
| Surgery <4 weeks | 1.5 |
| Past history PE/DVT | 1.5 |
| Haemoptysis | 1.0 |
| Current/recent neoplasm | 1.0 |
| No alternative diagnosis more likely than PE | 3.0 a |
| Score b | |
| Low | <2 |
| Moderate | 2–6 |
| High | >6 |
a Including information from ECG, ABG, CXR and other tests for alternative diagnoses.
b A dichotomized (modified Wells) scale of ≤4 (low) or >4 (high) is also validated and allows lower sensitivity D-dimers (and age-adjusted D-dimer using sensitive latex agglutination tests) to exclude more patients. On line version at http://www.mdcalc.com/wells-criteria-for-pulmonary-embolism-pe/
| Parameter | Score |
|---|---|
| Age <80 years | 1 |
| History of cancer | 1 |
| Chronic cardiopulmonary disease | 1 |
| Pulse ≥110 bpm | 1 |
| Systolic blood pressure <100 mm Hg | 1 |
| Arterial oxyhaemoglobin saturation level <90% | 1 |
| 0 points = 30-day mortality risk 1.0% (95% CI, 0.0%–2.1% a ≥1 point(s) = 30-day mortality risk 10.9% (95% CI, 8.5%–13.2%) b |
|
a Jimenez D, Aujesky D, Moores L, et al. Simplification of the pulmonary embolism severity index for prognostication in patients with acute symptomatic pulmonary embolism. Arch Intern Med . 2010;170:1383–1389.
b Konstantinides SV, Torbicki A, Agnelli G, et al. 2014 ESC guidelines on the diagnosis and management of acute pulmonary embolism. Eur Heart J . 2014;35:3033–3069, 3069a–3069k.
The initial screening tests for PE are discussed below. For patients considered high risk for PE (Well’s >6) without contraindications to anticoagulation or individual agents, initiate either a DOAC, heparin (LMWH or unfractionated heparin (UFH) or fondoparinux. For intermediate risk ED patients (PE in 15% to 25%) consider bleeding risks versus signs of severity for PE or underlying cardio-respiratory instability when deciding to start anticoagulants before confirmed PE. All unstable patients (including hypoxia, tachycardia/hypotension/lactate excess) should be anticoagulated immediately unless absolute contra-indications exist with heparin in addition to receiving thrombolysis or embolectomy.
A normal chest CXR with significant hypoxia is somewhat suggestive of PE. However, the CXR is abnormal in 80% to 90% of PE. If definitely present, an enlarged/plump descending pulmonary artery, pulmonary oligaemia or cut-off (Westermark sign), and particularly a ‘Hampton hump’ (a semi-circular opacity with the base abutting the pleural surface) are quite specific (70% to 90%). However, these may be subtle (e.g. often identified in retrospect) and have poor sensitivity. Pleural effusion, plate atelectasis, enlarged heart and non-specific consolidation are common but seen no more often than in other cardio-pulmonary conditions. The main role of CXR is to identify alternative diagnoses and to assist in deciding if a V/Q scan will provide adequate discrimination.
At least 21 potential ECG features have been postulated for PE, but they are insensitive and most lack specificity. The most significant are tachycardias (particularly atrial), right bundle branch block (often incomplete/transient), right axis deviation (particularly S1-S3), T-wave inversion (especially deep V1-V3) and the S1-Q3-T3 pattern. The more features present, the more suggestive for right ventricular (RV) strain. These changes have been associated with poorer PE outcomes, particularly the rare S1-Q3-T3 pattern.
In recent years, the role of ABG in PE has been challenged. A PaO 2 below 80 mm Hg without other cause makes PE more likely, but 12% of PE patients have PaO 2 greater than 80 mm Hg. An abnormal A–a gradient marginally increases PE risk, but 20% of PE patients have normal A–a gradients. In most patients, oximetry is sufficient to exclude significant hypoxaemia (<92%) and little is gained from ordering a routine ABG/venous analysis.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here