Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Proteinuria can be detected by various means, and the most common is the dipstick test, a calorimetric assay that spots only albumin and not low-molecular-weight proteins. In addition, false-positive dipstick assessment can be seen with highly concentrated urine, alkaline urine, the presence of contrast media, vaginal secretions, or semen. False negatives are less common but can be seen with very dilute urine. Though 24-hour urine collection is the gold standard to quantify the proteinuria, spot urine protein-to-creatinine ratio can be used for initial confirmation after a positive screen with dipstick or to trend proteinuria ( Table 22.1 ). A ratio <0.2 protein mg/creatinine mg is considered normal in children older than 2 years of age and a ratio <0.5 mg/mg is considered normal in younger children between 6 months and 2 years of age. In timed collection, protein excretion >240 mg/m 2 in 24 hours in children younger than 6 months of age and >150 mg/m 2 in older children is considered abnormal, and over 40 mg/m 2 /hr (>3 g/1.73 m 2 /day) is considered nephrotic range. Qualitative analysis of protein in urine by immunonephelometry or electrophoresis helps distinguish glomerular from tubular proteinuria.
| Method | Indications | Normal Range | Comments |
|---|---|---|---|
| Dipstick testing | Routine screening for proteinuria performed in the office | Negative or trace in a concentrated urine specimen (specific gravity: ≥1.020) Test interpretation:
|
False-positive test can occur if urine is very alkaline (pH >8.0) or very concentrated (specific gravity: >1.025), when there is pus, vaginal secretions, or semen present |
| 24-hr urine for protein and creatinine ∗ excretion | Quantitation of proteinuria (as well as creatinine clearances) | <150 mg/m 2 /24 hr | More accurate than spot urine analysis; inconvenient for patient; the creatinine content should be measured to determine whether the specimen is truly a 24-hr collection. The amount of creatinine in a 24-hr specimen can be estimated as follows: females, 15–20 mg/kg; males, 20–25 mg/kg |
| Spot urine for protein/creatinine ratio—preferably on first morning urine specimen | Semiquantitative assessment of proteinuria | <0.2 mg protein/mg creatinine in children older than 2 yr old <0.5 mg protein/mg creatinine in those 6–24 mo old |
Simplest method to quantitate proteinuria; less accurate than measuring 24-hr proteinuria |
| Microalbuminuria | Assess risk of progressive glomerulopathy in patients with diabetes mellitus | <30 mg urine albumin per gram of creatinine on first morning urine | Therapy should be intensified in diabetics with microalbuminuria |
Proteinuria in children can be transient, orthostatic, or persistent. Transient and orthostatic proteinuria are benign conditions and require no treatment. Several factors including fever, stress, hypovolemia, exercise, and seizures can lead to transient proteinuria ( Table 22.2 ). Orthostatic proteinuria is defined as increased protein in urine only when upright. In this condition, absence of proteinuria when horizontal and resting can be confirmed by documenting absence of protein in a first morning void. Split day/night urine collection is the gold standard to diagnose orthostatic proteinuria, which is a common benign cause of proteinuria, especially in adolescents. Persistent proteinuria requires meticulous evaluation to rule out renal pathology.
| Transient Proteinuria |
|
| Orthostatic (Postural) Proteinuria |
| Glomerular Diseases Characterized by Isolated Proteinuria |
|
| Glomerular Diseases with Proteinuria as a Prominent Feature |
|
| Tubular Diseases |
|
Evaluation of proteinuria begins with a detailed history and physical examination. Pertinent histories that help distinguish pathologic from benign proteinuria include history of respiratory symptoms concurrent with or preceding the proteinuria, presence of red urine, edema, positive family history of kidney disease, or hearing loss. Findings of edema and hypertension suggest pathologic proteinuria. Repeating urine dipstick in asymptomatic children with a negative history can eliminate unnecessary further testing for transient proteinuria. If still positive, spot urine protein-to-creatinine ratio can help confirm the presence of proteinuria. If confirmed, a first morning void protein-to-creatinine ratio can then identify orthostatic proteinuria. Once the benign conditions are ruled out in asymptomatic children, further testing is similar to that of symptomatic children and these children should be referred to nephrologists. This more detailed evaluation begins with 24-hour urine collection where possible, complete urinalysis, and sediment evaluation looking for glomerular or other parenchymal pathology that could be causing the proteinuria ( Fig. 22.1 ). Positive leukocyte esterase, nitrite, and presence of pyuria or bacteriuria suggest a urinary tract infection. If not resolved with treatment of infection, proteinuria will need further evaluation. Low molecular proteins, such as β2-microglobulin, α1-microglobulin, lysozyme, and retinol-binding protein, are found in tubular proteinuria as is seen in Fanconi syndrome or Dent disease. Red blood cell (RBC) casts are pathognomonic of glomerulonephritis . Serum chemistry including creatinine, BUN, electrolytes, albumin, and cholesterol will also help separate proteinuria secondary to glomerulonephritis or nephrotic syndrome. Lupus antibody studies, streptococcal infection, and complement C3 and C4 levels along with viral studies can help delineate the various causes of glomerulonephritis and nephrotic syndrome. Renal ultrasound should be considered to rule out any gross parenchymal etiology for the proteinuria, such as dysplastic kidney and cystic kidney disease. Renal biopsy may be indicated if there is evidence for worsening of proteinuria, hypoalbuminemia, deteriorating renal function, or a poor response to the initial therapy.
Differential diagnoses for proteinuria are extensive, as described in Table 22.2 . The initial evaluation of a patient with proteinuria is presented in Table 22.3 . Indications for a referral to a pediatric nephrologist are described in Table 22.4 . If there is obvious edema with proteinuria, the diagnostic evaluation noted in Table 22.3 advances directly to the second phase and, if necessary, to the third phase.
| Pediatrician’s Work-up: Phase I |
|
| Pediatrician’s Work-up: Phase II |
|
| Pediatric Nephrologist’s Work-up: Phase III |
|
|
The combination of proteinuria, hypoalbuminemia, edema, and hyperlipidemia are the defining features of nephrotic syndrome. Nephrotic syndrome may be a result of many primary etiologic factors, with varying renal pathologic processes and long-term consequences. Proteinuria that causes edema is always clinically significant, although not all edema is secondary to proteinuria ( Table 22.5 ). All children with nephrotic syndrome invariably have “nephrotic range” proteinuria, necessitating detailed evaluation, and most require treatment. In rare cases, a child with asymptomatic proteinuria has nephrotic-range proteinuria. If there is concomitant hypoalbuminemia and hyperlipidemia, the work-up proceeds as if the child presented with nephrotic syndrome, despite the absence of edema. Even without hypoalbuminemia and hyperlipidemia, nephrotic-range proteinuria is less likely to be benign than is less marked asymptomatic proteinuria.
| Kidney Diseases |
|
| Heart Failure |
| Liver Failure |
| Nutritional and Gastrointestinal Disorders |
|
| Endocrine Disorders |
|
| Miscellaneous |
|
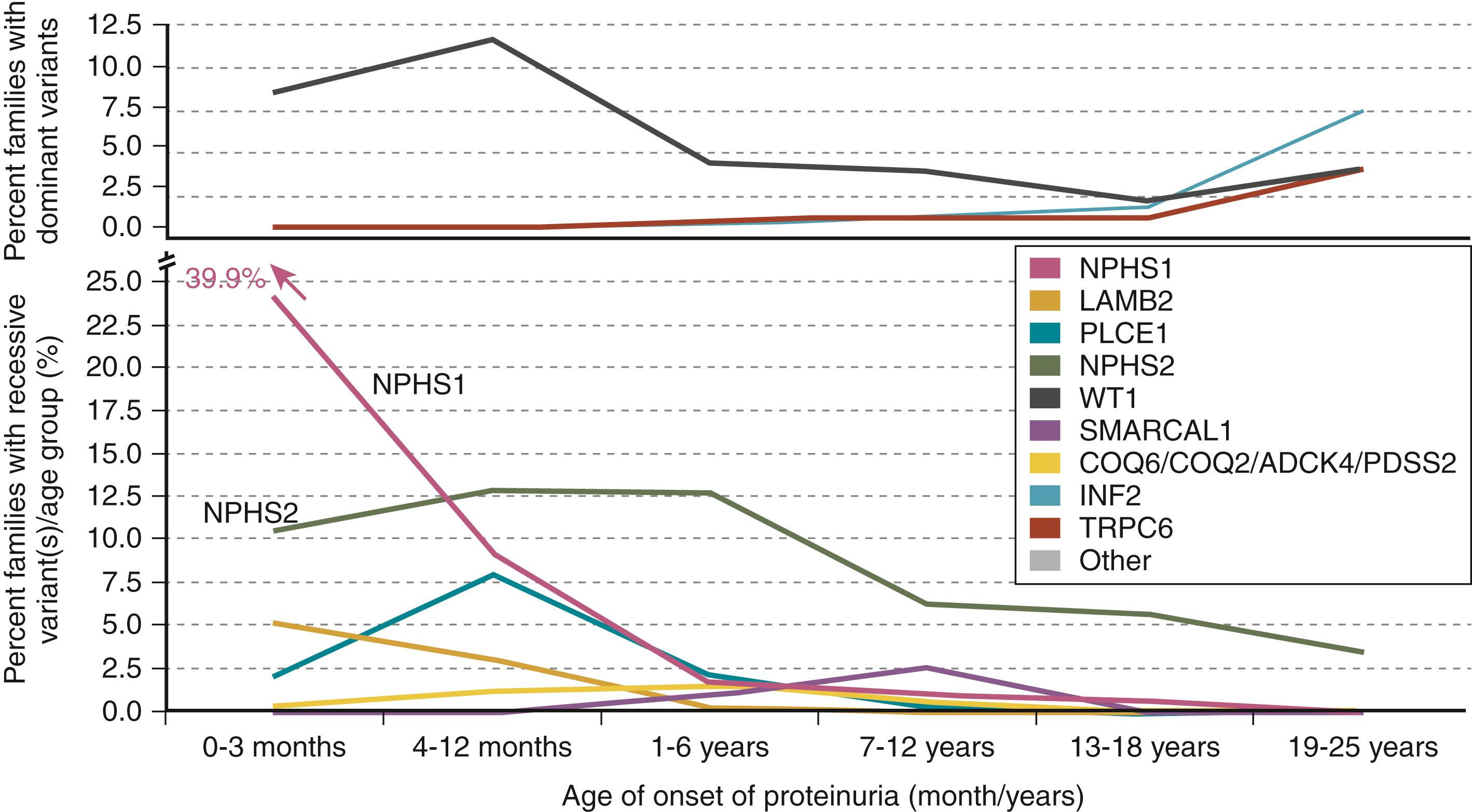
Three diseases constitute all cases of isolated nephrotic syndrome: minimal change disease (the most common), focal segmental sclerosis (also called focal glomerular sclerosis), and membranous glomerulopathy. These classifications are based on pathologic findings. Thus, these presentations could be primary or secondary due to other causes. In addition, nephrotic syndrome can be present along with glomerulonephritis (GN), such as postinfectious GN, immunoglobulin A (IgA) GN, or membranoproliferative GN. Systemic diseases also cause childhood nephrotic syndrome, accounting for 10% of cases. The three foremost considerations include systemic lupus erythematosus (SLE), IgA vasculitis (Henoch-Schönlein purpura), and hemolytic uremic syndrome. These diseases have extrarenal manifestations in addition to the proteinuria and must be considered in any child who presents with systemic illness and significant proteinuria. Hereditary forms of nephrotic syndrome are a genetically heterogeneous group of disorders representing a spectrum of hereditary renal diseases ( Table 22.6 and Fig. 22.2 ). Over 45 recessive or dominant genes have been associated with steroid-resistant nephrotic syndrome (SRNS)/hereditary nephrotic syndrome in humans ( Table 22.7 ). Several of the more common disorders along with other causes of nephrotic syndrome are noted in Tables 22.8 and 22.9 . Causes of congenital nephrotic syndrome (in infants 3 months of age or younger) are noted in Table 22.10 .
| Location | Phenotype | Inheritance | OMIM | Gene/Locus |
|---|---|---|---|---|
| Primarily Renal | ||||
| 1q23.3 | Nephrotic syndrome, type 22 | AR | 619155 | NOS1AP |
| 1q25.2 | Nephrotic syndrome, type 2 | AR | 600995 | PDCN |
| 1q42.13 | Nephrotic syndrome, type 18 | AR | 618177 | NUP133 |
| 3p21.31 | Nephrotic syndrome, type 5, with or without ocular abnormalities | AR | 614199 | LAMB2 |
| 7q21.11 | Nephrotic syndrome, type 15 | AR | 617609 | MAGI2 |
| 7q33 | Nephrotic syndrome, type 13 | AR | 616893 | NUP205 |
| 10q22.1 | Nephrotic syndrome, type 14 | AR | 617575 | SGPL1 |
| 10q23.33 | Nephrotic syndrome, type 3 | AR | 610725 | PLCE1 |
| 11p13 | Nephrotic syndrome, type 4 | AD | 256370 | WT1 |
| 11p11.2 | Nephrotic syndrome, type 19 | AR | 618178 | NUP160 |
| 12p12.3 | Nephrotic syndrome, type 6 | AR | 614196 | PTPRO |
| 12q14.1 | Nephrotic syndrome, type 21 | AR | 618594 | AVIL |
| 12q15 | Nephrotic syndrome, type 11 | AR | 616730 | NUP107 |
| 16p13.13 | Nephrotic syndrome, type 10 | AR | 615861 | EMP2 |
| 16q13 | Nephrotic syndrome, type 12 | AR | 616892 | NUP93 |
| 17q22 | Nephrotic syndrome, type 7 | AR | 615008 | DGKE |
| 17q22 | Hemolytic uremic syndrome, atypical, susceptibility to, 7 | AR | 615008 | DGKE |
| 17q25.1 | Nephrotic syndrome, type 17 | AR | 618176 | NUP85 |
| 17q25.3 | Nephrotic syndrome, type 8 | AR | 615244 | ARHGDIA |
| 19p13.2 | Nephrotic syndrome, type 16 | AR | 617783 | KANK2 |
| 19q13.12 | Nephrotic syndrome, type 1 | AR | 256300 | NPHS1 |
| 19q13.2 | Nephrotic syndrome, type 9 | AR | 615573 | COQ8B |
| Xq22.3 | Nephrotic syndrome, type 20 | XL | 301028 | TBC1D8B |
| 19q13.2 | Glomerulosclerosis, focal segmental, 1 | AD | 603278 | ACTN4 |
| 11q22.1 | Glomerulosclerosis, focal segmental, 2 | AD | 603965 | TRPC6 |
| 6p12.3 | Glomerulosclerosis, focal segmental, 3 | CD2AP-AD | 607832 | CD2AP |
| 14q32.33 | Glomerulosclerosis, focal segmental, 5 | INF2-AD | 613237 | INF2 |
| 15q22.2 | Glomerulosclerosis, focal segmental, 6 | AR | 614131 | MYO1E |
| 10q24.31 | Glomerulosclerosis, focal segmental, 7 | AD | 616002 | PAX2 |
| 2q35 | Glomerulopathy with fibronectin deposits 2 | AD | 601894 | FN1 |
| 7p14.2 | Focal segmental glomerulosclerosis 8 | AD | 616032 | ANLN |
| 9q33.3 | Focal segmental glomerulosclerosis 10 | AR | 256020 | LMX1B |
| 1q32 | Glomerulopathy with fibronectin deposits 1 | AD | 137950 | GFND1 |
| 2q35 | Glomerulopathy with fibronectin deposits 2 | AD | 601894 | FN1 |
| Systemic Disorders | ||||
| 1p33-p31.1 | Forsythe-Wakeling syndrome | AR | 613606 | |
| 17q21.33 | Interstitial lung disease, nephrotic syndrome, and epidermolysis bullosa, congenital | AR | 614748 | ITGA3 |
| 3p21.31 | Pierson syndrome | AR | 609049 | LAMB2 |
| 15q25.2 | Galloway-Mowat syndrome 1 | AR | 251300 | WDR73 |
| Xq28 | Galloway-Mowat syndrome 2, X-linked | XLR | 301006 | LAGE3 |
| 14q11.2 | Galloway-Mowat syndrome 3 | AR | 617729 | OSGEP |
| 20q13.12 | Galloway-Mowat syndrome 4 | AR | 617730 | TP53RK |
| 2p13.1 | Galloway-Mowat syndrome 5 | AR | 617731 | TPRKB |
| 21q22.3 | Galloway-Mowat syndrome 6 | AR | 618347 | WDR4 |
| 12q15 | Galloway-Mowat syndrome 7 | AR | 618348 | NUP107 |
| 1q42.13 | Galloway-Mowat syndrome 8 | AR | 618349 | NUP133 |
| 16p13.3 | Congenital disorder of glycosylation, type Ik | AR | 608540 | ALG1 |
| 11p13 | Denys-Drash syndrome | AD, SMu | 194080 | WT1 |
| 2q11.2 | Autoimmune disease, multisystem, infantile onset, 2 | AR | 617006 | ZAP70 |
| 4q21.1 | Epilepsy, progressive myoclonic 4, with or without renal failure | AR | 254900 | SCARB2 |
| 2q35 | Schimke immunoosseous dysplasia | AR | 242900 | SMARCAL1 |
| 10q24.32 | Immunodeficiency, common variable, 10 | AD | 615577 | NFKB2 |
| 9q33.2 | Amyloidosis, Finnish type | AD | 105120 | GSN |
| 16p13.2 | Congenital disorder of glycosylation, type Ia | AR | 212065 | PMM2 |
| 11p13 | Frasier syndrome | AD, SMu | 136680 | WT1 |
| 2q33.1 | Autoimmune lymphoproliferative syndrome, type II | AD | 603909 | CASP10 |
| 1p31.3 | Autoinflammation, immune disregulation, and eosinophilia | AD | 618999 | JAK1 |
| Xp11.23 | Congenital disorder of glycosylation, type IIm | SMo, XLD | 300896 | SLC35A2 |
| 4q31.3 | Amyloidosis, familial visceral | AD | 105200 | FGA |
| 11q23.3 | Amyloidosis, 3 or more types | AD | 105200 | APOA1 |
| 12q15 | Amyloidosis, renal | AD | 105200 | LYZ |
| 15q21.1 | Amyloidosis, familial visceral | AD | 105200 | B2M |
| 2q24.2 | Aicardi-Goutieres syndrome 7 | AD | 615846 | IFIH1 |
| 3p21.1 | Autoimmune lymphoproliferative syndrome, type III | AR | 615559 | PRKCD |
| 6q13 | Sialic acid storage disorder, infantile | AR | 269920 | SLC17A5 |
| 2q36.3 | Alport syndrome 2, autosomal recessive | AR | 203780 | COL4A4 |
| 2q36.3 | Alport syndrome 2, autosomal recessive | AR | 203780 | COL4A3 |
| 9q33.3 | Nail-patella syndrome | AD | 161200 | LMX1B |
| Xq22.3 | Alport syndrome 1, X-linked | XLD | 301050 | COL4A5 |
| 16p13.3 | Familial Mediterranean fever, AR | AR | 249100 | MEFV |
| 19p13.3 | C3 deficiency | AR | 613779 | C3 |
| 12q24.31 | Mucopolysaccharidosis-plus syndrome | AR | 617303 | VPS33A |
| 10p13 | Omenn syndrome | AR | 603554 | DCLRE1C |
| 11p12 | Omenn syndrome | AR | 603554 | RAG1 |
| 11p12 | Omenn syndrome | AR | 603554 | RAG2 |
| 11p15.5 | Nephropathy with pretibial epidermolysis bullosa and deafness | AR | 609057 | CD151 |
| 22q12.3 | End-stage renal disease, nondiabetic, susceptibility to | 612551 | APOL1 | |
| 22q12.3 | Glomerulosclerosis, focal segmental, 4, susceptibility to | 612551 | APOL1 | |
| 9q33.3 | Ventriculomegaly with cystic kidney disease | AR | 219730 | CRB2 |
| 1q44 | Muckle-Wells syndrome | AD | 191900 | NLRP3 |
| 12q13.3 | Pseudo-TORCH syndrome 3 | AR | 618886 | STAT2 |
| Xq13.3 | Intellectual disability, X-linked 98 | XLD | 300912 | NEXMIF |
| 15q26.1 | Arthrogryposis, renal dysfunction, and cholestasis 1 | AR | 208085 | VPS33B |
| 11p15.4 | Immunodeficiency 10 | AR | 612783 | STIM1 |
| 14q32.32 | Imerslund-Grasbeck syndrome 2 | AR | 618882 | AMN |
| 10p14 | Hypoparathyroidism, sensorineural deafness, and renal dysplasia | AD | 146255 | GATA3 |
| 12q24.11 | Hyper-IgD syndrome | AR | 260920 | MVK |
| Xp11.23 | Immunodisregulation, polyendocrinopathy, and enteropathy, X-linked | XLR | 304790 | FOXP3 |
| Xq26.2-q26.3 | Lesch-Nyhan syndrome | XLR | 300322 | HPRT1 |
| 12q23.2 | Microcephaly 24, primary, autosomal recessive | AR | 618179 | NUP37 |
| 1q21 | Nephropathy-hypertension | AD | 161900 | |
| 15q22.31 | Melorheostosis, isolated, somatic mosaic | 155950 | MAP2K1 | |
| 7q11.23 | Cutis laxa, autosomal dominant | AD | 123700 | ELN |
| 2p21 | Cystinuria | AD, AR | 220100 | SLC3A1 |
| 19q13.11 | Cystinuria | AD, AR | 220100 | SLC7A9 |
| Gene | Protein | Inheritance | Locus | Phenotypes |
|---|---|---|---|---|
| Slit Diaphragm and Adaptor Proteins | ||||
| NPHS1 | Nephrin | AR | 19q13.1 | CNS, SRNS (NPHS1) |
| NPHS2 | Podocin | AR | 1q25–q31 | CNS, SRNS (NPHS2) |
| PLCE1 | Phospholipase C, ε1 | AR | 10q23 | DMS, SRNS (NPHS3) |
| CD2AP | CD2-associated protein | AD/AR | 6p12.3 | SRNS (FSGS3) |
| FAT1 | FAT1 | AR | 4q35.2 | NS, ciliopathy |
| Cytoskeleton Components | ||||
| ACTN4 | α-Actinin-4 | AD | 19q13 | Late-onset SRNS (FSGS1) |
| INF2 | Inverted formin-2 | AD | SRNS (FSGS5), Charcot-Marie-Tooth disease with glomerulopathy | |
| MYH9 | Myosin, heavy chain 9 | AD | 22q12.3–13.1 | Macrothrombocytopenia with sensorineural deafness, Epstein syndrome, Sebastian syndrome, Fechtner syndrome |
| MYO1E | Myosin IE | AR | 15q22.2 | Childhood-onset SRNS (FSGS6) |
| ARHGDIA | Rho GDP-dissociation inhibitor (GDI) a1 | AR | 17q25.3 | Childhood-onset SRNS (NPHS8), seizures, cortical blindness |
| ARHGAP24 | Arhgap24 (RhoGAP) | AD | 4q22.1 | Adolescent-onset FSGS |
| ANLN | Anillin | AD | 7p14.2 | FSGS8 |
| GBM and Basal Membrane Proteins and Related Components | ||||
| LAMB2 | Laminin subunit β2 | AR | 3p21 | Pierson syndrome DMS, FSGS (NPHS5) |
| ITGB4 | Integrin-β4 | AR | 17q25.1 | Epidermolysis bullosa, anecdotic cases presenting with NS and FSGS |
| ITGA3 | Integrin-β3 | AR | Epidermolysis bullosa, interstitial lung disease, SRNS/FSGS | |
| CD151 | Tetraspanin | AR | 11p15.5 | Epidermolysis bullosa, sensorineural deafness, ESRD |
| EXT1 | Glycosyltransferase | AR | 8q24.11 | SRNS |
| COL4A3,4 | Collagen (IV) α3/α4 | AD/AR | 2q36–q37 | Alport syndrome, FSGS |
| COL4A5 | Collagen (IV) α5 | XD | Xq22.3 | Alport syndrome, FSGS |
| Apical Membrane Proteins | ||||
| TRPC6 | Transient receptor potential channel 6 | AD | 11q21–q22 | SRNS (FSGS2) |
| EMP2 | Epithelial membrane protein 2 | AD | 16p13.2 | Childhood SRNS/SSNS (MCD) (NPHS10) |
| Nuclear Proteins | ||||
| WT1 | Wilms tumor protein | AD/AR | 11p13 | SRNS (NPHS4), Denys-Drash syndrome, Frasier syndrome, WAGR syndrome |
| LMX1B | LIM homeobox transcription factor 1-β | AD | 9q34.1 | Nail-patella syndrome, NS |
| SMARCAL1 | HepA-related protein | AR | 2q35 | Schimke immunoosseous dysplasia |
| PAX2 | Paired box gene 2 | AD | 10q24.3–q25.1 | Adult-onset FSGS (FSGS7), renal coloboma syndrome |
| MAFB | A transcription factor | AD | 20q11.2–q13.1 | Carpotarsal osteolysis progressive ESRD |
| LMNA | Lamins A and C | XD | 1q22 | Familial partial lipodystrophy, FSGS |
| NXF5 | Nuclear RNA export factor 5 | XR | Xq21 | SRNS/FSGS cardiac conduction disorder |
| GATA3 | GATA binding protein 3 | AD | 10p14 | HDR syndrome (hypoparathyroidism, sensorineural deafness, renal abnormalities) |
| NUP93 | Nucleoporin 93kD | N/A | 16q13 | SRNS |
| NUP107 | Nucleoporin 107kD | N/A | 12q15 | Early-childhood-onset SRNS/FSGS |
| Mitochondrial Proteins | ||||
| COQ2 | 4-Hydroxybenzoate polyprenyltransferase | AR | 4q21–q22 | Early-onset SRNS, CoQ10 deficiency |
| COQ6 | Ubiquinone biosynthesis monooxygenase COQ6 | AR | 14q24.3 | NS with sensorineural deafness, CoQ10 deficiency |
| PDSS2 | Decaprenyl-diphosphate synthase subunit 2 | AR | 6q21 | Leigh syndrome, CoQ10 deficiency, FSGS |
| MTTL1 | Mitochondrially encoded tRNA leucine 1 (UUA/G) | Maternal | mtDNA | Mitochondrial diabetes, deafness with FSGS, MELAS syndrome |
| ADCK4 | aarF domain containing kinase 4 | AR | 19q13.1 | Childhood-onset SRNS (NPHS9), CoQ10 deficiency |
| Lysosomal Proteins | ||||
| SCARB2 | Scavenger receptor class B, member 2 (LIMP II) | AR | 4q13–q21 | Action myoclonus-renal failure syndrome, lysosomal storage disease |
| NEU1 | Sialidase 1 | |||
| N-Acetyl-α-neuraminidase | AR | 6p21.33 | Nephrosialidosis, SRNS | |
| Other Intracellular Proteins | ||||
| APOL1 | Apolipoprotein L1 | AR | 22q12.3 | FSGS in African-Americans (FSGS4) |
| PTPRO | Tyrosine phosphatase receptor-type O (GLEPP1) | AR | 12p12.3 | SRNS (NPHS6) |
| CRB2 | Crumbs homolog 2 | AR | 9q33.3 | Early-onset familial SRNS (FSGS9) |
| DGKE | Diacylglycerol kinase-ε | AR | 17q22 | Atypical hemolytic uremic syndrome, membranoproliferative lesions (NPHS7) |
| ZMPSTE24 | Zinc metalloproteinase | AR | 1q34 | Mandibuloacral dysplasia, FSGS |
| PMM2 | Phosphomannomutase 2 | AR | 16p13.2 | CDG syndrome, FSGS |
| ALG1 | β1,4 Mannosyltransferase | AR | 16p13.3 | CDG syndrome, congenital NS |
| CUBN | Cubilin | AR | 10p13 | Childhood-onset SRNS megaloblastic anemia |
| TTC21B | IFT139 (a component of intraflagellar transport-A) | AR | 2q24.3 | Nephronophthisis (NPHP12), FSGS |
| WDR73 | WD repeat domain 73 | AR | 15q25.2 | Galloway-Mowat syndrome, SRNS/FSGS |
| Idiopathic Nephrotic Syndrome |
|
| Genetic Disorders Associated with Proteinuria or Nephrotic Syndrome |
|
| Nephrotic Syndrome (Typical) |
|
| Proteinuria with or Without Nephrotic Syndrome |
|
| Multisystem Syndromes with or Without Nephrotic Syndrome |
|
| Metabolic Disorders with or Without Nephrotic Syndrome |
|
| Secondary Causes of Nephrotic Syndrome |
| Infections |
|
| Drugs |
|
|
| Immunologic or Allergic Disorders |
|
| Associated with Malignant Disease |
|
| Glomerular Hyperfiltration |
|
| Secondary Causes |
|
| Primary Causes |
|
| Primary CNS |
|
|
|
|
|
|
|
|
|
| Secondary CNS |
|
|
|
|
|
|
Most cases of nephrotic syndrome in children are caused by minimal change nephrotic syndrome, defined as normal histologic features of the kidney by light microscopy and immune stains. Preschool-aged children constitute the age group in which minimal change nephrotic syndrome is most common. Patients often present with asymptomatic edema, which may manifest as swollen or puffy eyes upon awakening in the morning; increasing abdominal girth (increased waist or belt size) from ascites; pedal or leg edema, which causes difficulty in putting on their regular-sized shoes, especially after being upright during the daytime; or swelling in other sites, such as the scrotum, penis, vulva, and scalp. Tense edema or ascites is occasionally painful.
Minimal change nephrotic syndrome is slightly more common in males than in females. The hallmark of this disease is total clearing of the proteinuria with oral prednisone therapy. A common misconception is that neither hematuria nor hypertension is present in children with minimal change disease. Microscopic hematuria and hypertension are present in up to 20% of children who have minimal change disease. The BUN or serum creatinine level may also be elevated in up to 30% of the cases, usually from prerenal causes. Serum complement studies are normal. Older age, hematuria, hypertension, and azotemia may occur with minimal change nephrotic syndrome, but the combination suggests another disease.
Studies that would help confirm that a patient with nephrotic syndrome has minimal change disease include urinalysis; serum chemistry including BUN, creatinine, albumin, and cholesterol levels; and complements and lupus antibody titers.
The urinalysis would be expected to show 3+ to 4+ protein, which is correlated with a urine concentration of 300–2,000 mg/dL. The urine may also occasionally yield positive results for blood. Microscopic examination of the urine sediment often shows oval fat bodies and/or refractile granular casts, which are seen when there is significant lipiduria. Red blood cells might also be present, but it is unusual to see red blood cell casts. Their presence would suggest a diagnosis of glomerulonephritis (see Chapter 23 ).
The complement C3 and C4 levels are normal in minimal change disease and are depressed in some other causes of nephritis (see Chapter 23 ). The serum cholesterol values are elevated in minimal change nephrotic syndrome and are usually >250 mg/dL; levels in the range of 500–600 mg/dL may occur. The serum albumin concentration is invariably <2.5 and often <2.0 g/dL. A renal biopsy is not immediately indicated because most patients (>90%) with minimal change disease respond to prednisone, a response that is considered diagnostic.
With a presumptive diagnosis of minimal change nephrotic syndrome, it is recommended that patients be placed on a therapeutic course of prednisone, 60 mg/m 2 /day or 2 mg/kg/day, up to a maximum of 60 mg for 4–6 weeks, followed by a dose of 40 mg/m 2 or 1.5 mg/kg (maximum 40 mg) given every other day for another 6 weeks. In most patients, there is total resolution of proteinuria within 10–21 days of initiating therapy. Patients who do not respond to prednisone therapy should be considered candidates for a renal biopsy to guide further therapy.
Total clearing of proteinuria in response to prednisone is an excellent prognostic sign. Very few patients progress to renal failure, although many patients (∼80%) who initially respond to prednisone therapy with total clearing of proteinuria may have relapses and require intermittent prednisone therapy for many years. Approximately 18% of patients treated with prednisone for minimal change nephrotic syndrome respond to therapy and never experience a relapse.
Patients with recurrent nephrotic syndrome are subgrouped into those who experience frequent and infrequent relapses. A patient with infrequent relapse has fewer than two relapses in any 6-month period; a person with frequent relapse has two or more relapses within 6 months. Prednisone should be reinitiated at a dose of 60 mg/m 2 /day or 2 mg/kg/day until a maximum of 60 mg/day and continued until the urine test results are negative for protein for 3 consecutive days. After that, alternate-day prednisone is given at a dose of 40 mg/m 2 or 1.5 mg/kg (maximum 40 mg) in the morning for another 4 weeks and then discontinued altogether. Relapses are frequent during the influenza virus seasons; any minor upper respiratory infection may trigger a relapse of nephrotic syndrome. Patients who suffer infrequent relapses may be treated with prednisone alone.
Patients with frequently relapsing nephrotic syndrome may be steroid dependent and require constant daily prednisone therapy to maintain a remission. Because constant daily prednisone has significant untoward side effects (growth failure, cushingoid facies, osteoporosis, cataracts, opportunistic infections, hypertension, and glucose intolerance), other therapies need to be considered. A renal biopsy is recommended prior to initiating alternative agents to confirm the diagnosis of minimal change nephrotic syndrome. Treatment strategies with corticosteroid-sparing agents for patients with frequent relapse who develop steroid-related adverse effects include alkylating agents, cyclophosphamide or chlorambucil, and more recently rituximab.
Even in patients with the frequent relapse variant of minimal change disease, the incidence of renal failure is only 1%. The reported mortality rate remains higher, at approximately 5%.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here