Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The prostate is a pear-shaped glandular organ that weighs up to 20 g in the normal adult male and that depends for its differentiation and subsequent growth on androgenic hormones synthesized in the testis, acting through a poorly understood mesenchymal–epithelial interaction. It is most commonly divided into the anterior fibromuscular stroma and three distinct glandular zones as described by McNeal: peripheral zone, transition zone, central zone. The transition zone envelops the urethra in the mid portion of the prostate and is the anatomic region enlarged by benign prostatic hyperplasia. The central zone (which together with the seminal vesicles is thought to be derived from the wolffian duct) is shaped like an inverted pyramid at the base of the prostate and contains the ejaculatory ducts as they course to the prostatic urethra at the verumontanum, a posterior prominence in the prostatic urethra. The prostatic glands of the central zone may have a unique morphology with more deeply eosinophilic cytoplasm and/or more complex intraluminal architecture consisting of papillary infolding or epithelial bridges ( Fig. 26.1 ). The peripheral zone envelops the transition zone and extends caudally to comprise most of the apex. Finally, the anterior tissues consist of smooth muscle (extending from the base), skeletal muscle from the anterior fibromuscular sling (extending from the apex), and adipose tissue in the extraprostatic compartment. The prostate is enveloped by a fibromuscular layer usually referred to as a “capsule,” but it has been pointed out that this is most definitely not a well-defined anatomic structure with constant features.
The glandular component of the organ is traditionally divided into acini and ducts, the latter subdivided into large (primary, major, excretory) and peripheral (secondary, minor); however, unlike other organs, there is little morphologic difference between the two. Both acini and ducts contain luminal secretory cells, a surrounding outer layer of basal cells, and scattered neuroendocrine cells (which are often unrecognizable). The secretory cells, which are located in the luminal side of the gland, contribute a wide variety of products to the seminal fluid. Histologically, these cells form an undulating luminal surface and are characterized by relatively pale cytoplasm that contains minute closely packed vacuoles that may be difficult to appreciate. They produce prostate-specific acid phosphatase (PSAP) and prostate-specific antigen (PSA), both of which can be readily identified immunohistochemically and have been proved of great diagnostic utility because of their organ-related specificity. Secretory cells coexpress various keratins, but they do not include high-molecular-weight types such as 34βE12, a fact of diagnostic significance. More recently, immunoreactivity for NKX3.1 (nuclear) and prostate-specific membrane antigen (PSMA; cytoplasmic) has been demonstrated in prostatic secretory cells, which may very useful diagnostically. The normal prostatic secretion is a neutral mucosubstance. On occasion, one encounters scattered individual columnar mucin-secreting cells in non-neoplastic prostatic epithelium, particularly in areas of atrophy.
The basal cells form a thin layer that separates the luminal secretory cells from the basement membrane. Morphologically, basal cells often appear as small inconspicuous, almost pyknotic nuclei at the base of the secretory cells. When prominent (such as in hyperplasia), they are more basophilic than secretory cells and commonly have large nuclei with open chromatin and small pin-point nucleoli. They characteristically express high-molecular-weight cytokeratin (e.g. 34βE12 and CK5/6) and p63, a fact that has been exploited in the differential diagnosis between well-differentiated carcinomas (in which basal cells are absent) and benign conditions that simulate it (in which they are generally present, although sometimes in a patchy discontinuous fashion). Under normal conditions these basal cells do not have the phenotype of myoepithelial cells, in the sense that they lack immunoreactivity for S-100 protein or smooth muscle actin. However, they can be viewed as being equivalent to the myoepithelial cells of the breast and other glandular organs. Support for this interpretation derives from the fact that they can undergo clear-cut myoepithelial metaplasia in conditions such as sclerosing adenosis. They do not express PSA or PSAP, but they have been shown to exhibit strong immunoreactivity for androgen receptors focally. They are thought to represent a multipotential population that gives rise to all epithelial lineages present in the normal, hyperplastic, and neoplastic prostate.
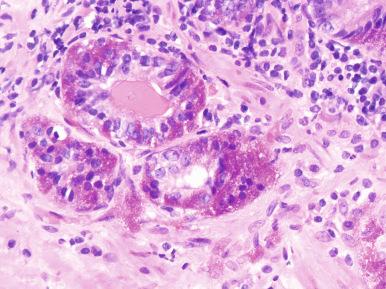
The neuroendocrine cells express chromogranin A and B, secretogranin II, and various peptide hormones such as somatostatin, calcitonin, and bombesin ; they coexpress PSA, suggesting a common origin with the secretory cells. However, they are negative for androgen receptors. While often inconspicuous, occasional cells with red neuroendocrine granules may be seen ( Fig. 26.2 ).
Near the urethra, the large prostatic ducts may be lined by urothelium that is continuous and indistinguishable from that lining the prostatic urethra. In contrast to bladder epithelium, its surface does not display umbrella cells, but rather a single layer of columnar cells that are immunoreactive for PSA and PSAP. On occasion, this epithelium undergoes squamous metaplastic changes; these were very common at the time estrogen therapy was widely employed for prostatic carcinoma.
The prostatic stroma is notable because of its large content of smooth muscle fibers. It has been pointed out that the presence of this muscular stroma duplicates the function of myoepithelial cells in other organs, such as breast, and makes the presence of myoepithelial cells in the prostate superfluous. Prostatic stromal cells have been found to contain androgen receptors.
Peripheral nerves are evenly distributed in the apex, mid gland, and base ; they are of importance to pathologists because of the high frequency with which the loose connective space that surrounds them is involved by adenocarcinomas of this organ.
The prostatic lymph vessels drain into the pelvic lymph nodes and from there into the retroperitoneal chain.
Ectopia of prostatic tissue is a very rare event, save for the benign prostatic-type polyps discussed in Chapter 25 (Bladder). It has been described in the bladder (especially in the trigone and in association with urachal remnants), root of the penis, epididymis, testis, seminal vesicle, retrovesical space, submucosa of the anal canal, pericolic fat/pararectal space, uterine cervix and vagina, and spleen.
Benign prostatic hypertrophy is the usual name applied to a common benign disorder of the prostate that, when extensive, results in varying degrees of urinary obstruction, sometimes requiring surgical intervention. The term nodular hyperplasia , as proposed by Moore in his classic study, is a more exact designation. The disease represents a nodular enlargement of the gland caused by hyperplasia of both glandular and stromal components. This results in an increase in the weight of the organ well beyond the 20 g regarded as normal for adult individuals. The clinical incidence of this disease is only 8% during the fourth decade, but it reaches 50% in the fifth decade and 75% in the eighth decade.
No predisposing or protecting factors (other than castration) have been identified. It has been established that prostatic nodular hyperplasia occurs only in individuals with intact testes and that it is an androgen-dependent disorder. Specifically, it has been suggested that nodular hyperplasia may result from an initial activation of mesenchymal clones with embryonal functions that stimulate development of the glandular component. In turn, this may result from dihydrotestosterone accumulation within the gland, resulting from decreased catabolism of the molecule and enhanced intracellular binding. A peculiar immunohistochemical finding that may throw some light on this issue is the demonstration that p27 protein (a negative regulator of the cell cycle) is prominently expressed in the epithelial and stromal cells of the normal prostate, but is essentially negative in nodular hyperplasia.
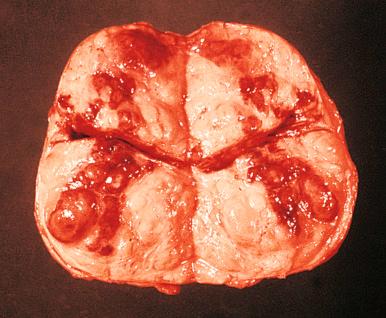
At autopsy the average weight of a prostate gland affected by nodular hyperplasia is 33 g ± 16 g. Specimens obtained surgically weigh 100 g on average, but on rare occasions weights of over 800 g have been recorded. Grossly, variously sized nodules with a gray to yellow color and a granular appearance are seen projecting above the cut surface ( Fig. 26.3 ).
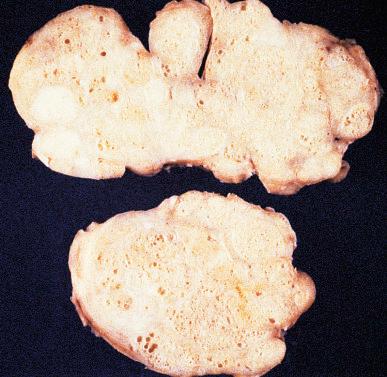
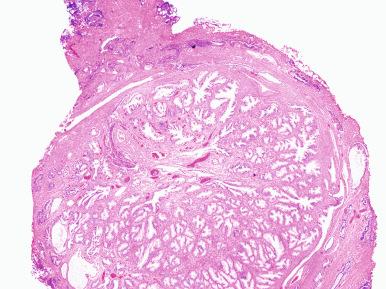
A cross section of an entire gland with early involvement clearly shows that nodular hyperplasia begins in the transition zone. This fact supports the interpretation that this zone of the gland reacts differently to hormonal stimuli than the outer zones. In most instances the nodules congregate on both sides of the urethra, causing compression and urinary obstruction. In others, the disease may result in a midline dorsal nodule at the bladder neck protruding into the bladder lumen, so-called “median-lobe hypertrophy.” With increased growth, the peripheral zone of the organ is pushed aside and compressed; in florid examples, the transition zone may extend into and comprise much of the apex.
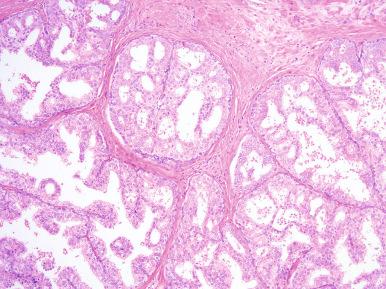
Microscopically, the earliest change is a stromal proliferation about small sinusoidal spaces in the periurethral regions and, to less degree, in the periductal and intralobular areas ( Fig. 26.4 ). This stromal proliferation (which in the periductal areas may have a concentric or an eccentric quality) contains more smooth muscle and less elastic tissue than the normal stroma. This is followed by hyperplasia of the glandular component, so that in the well-developed disease the nodules are composed of varying proportions of both elements. These proportions are somewhat different in patients with symptomatic and those with asymptomatic nodular hyperplasia. The glands are dilated or even cystic and often contain an inspissated secretion of glycoprotein nature (corpora amylacea), which is sometimes calcified. The epithelium ranges from flat to columnar, sometimes facing each other in the same gland (“functional polarization”); the cytoplasm is pale, and the nuclei are regular and centrally located ( Fig. 26.5 ). The nucleoli are inconspicuous. Papillary infoldings are common. A basal cell layer is seen immediately above a well-developed basement membrane.
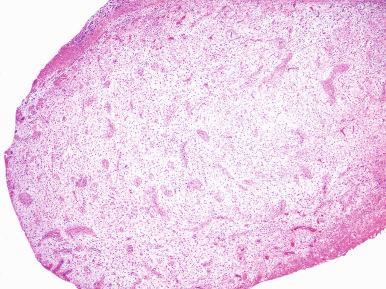
Small clusters of lymphocytes are common in the interstitium and around the ducts. They are probably the result rather than the cause of the hyperplasia ; a diagnosis of chronic prostatitis is not warranted because of their mere presence.
Many morphologic variations of this basic theme exist, some of them resulting from the overgrowth of one component over the other and others form the emergence of distinctive patterns. It should be pointed out that there is little relation between a diagnosis of nodular hyperplasia made on a biopsy specimen and either the weight of the gland or the score system used to measure urinary obstructive symptoms.
The treatment for nodular hyperplasia includes medical or surgical management. Medical alternatives to surgery include various medications aimed at blocking the actions of androgens by preventing their secretion or their conversion to their tissue active form or at relaxing the stromal muscle cells. Among these, the drug most widely used is finasteride, which works by inhibiting 5-α reductase, the enzyme that converts testosterone to the potent androgen dihydrotestosterone. The morphologic changes induced by this compound in the prostate are thought to be relatively minor and nonspecific. The involved area may be excised by various techniques, such as transurethral resection (TUR), suprapubic prostatectomy, and laser enucleation. It should be realized that these procedures remove only the newly formed nodules. The compressed peripheral portions of the gland remain; these expand by stromal growth to surround the prostatic urethra and may be the source of recurrent hyperplasia. Not surprisingly, the chance of a patient undergoing a second operation for this disorder is substantially higher after a TUR than an open prostatectomy.
Infarct of the prostate occurs predominantly in large prostates affected by nodular hyperplasia. Its reported incidence is probably related to the thoroughness of the microscopic examination. In the glands carefully studied by Moore, it was found to be present in 18%–25% of cases. It has been traditionally reported in TUR specimens, but it can also be recognized in prostatic needle biopsies. The size and number of the infarcts are directly related to the degree of prostatic hyperplasia. True infarcts occurring on a vascular basis should be distinguished from necrotic changes involving a gland or group of glands but sparing the stroma, a change sometimes seen in nodular hyperplasia.
The mechanism of infarct is unknown but may be related to the presence of prostatic infection or trauma resulting from an indwelling catheter, cystitis, or prostatitis, all of which may result in thrombosis of the intraprostatic portion of the urethral arteries.
Grossly, prostatic infarcts vary in size from a few millimeters up to 5 cm. They are speckled, gray-yellow, and often contain streaks of blood. The peripheral margins are usually sharp and hemorrhagic and may impinge on the urethra ( Fig. 26.6 ). Microscopically, the infarcts are of ischemic type, with sharply outlined areas of coagulative necrosis involving glands and stroma . Prominent squamous metaplasia may develop in the ducts at the periphery of the infarct, a change that should not be confused with squamous cell carcinoma ( Fig. 26.7 ). This metaplastic change is confined to the expanded ducts, keratinizes only rarely, and does not extend to the surrounding prostatic tissue. It should be remembered that true squamous cell carcinomas of the prostate are exceptionally rare and are most often seen after radiation therapy.
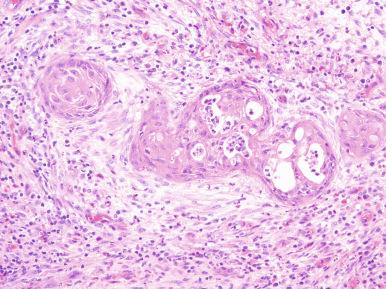
Most prostatic infarcts are clinically silent. Occasionally, they cause acute urinary retention because of the accompanying edema. Since they are often adjacent to the urethra, gross hematuria can also occur. Diffuse oozing of blood from the overlying mucosa may be seen cystoscopically. They may cause serum elevation of PSAP and PSA. Removal of the infarcted area promptly returns these levels to normal; if it does not, further work-up is indicated.
Acute prostatitis is rarely seen in surgical specimens. In general, prostatitis is a clinical, not a pathologic diagnosis. So-called chronic prostatitis is more common, but it is important to distinguish the true infectious processes of this organ from the inconsequential mononuclear infiltrates often seen in biopsies or accompanying nodular hyperplasia. While the latter phenomenon has been dignified by terms such as “chronic nonbacterial prostatitis” or “lymphocytic prostatitis,” it is generally best to reserve the term “prostatitis” for clinical infections. Therefore, we do not report the presence of chronic inflammation on biopsy unless it is quite marked and only with the descriptive term “marked chronic inflammation.” In bacterial prostatitis, the route of infection remains uncertain in most cases. Some cases follow gonococcal or nongonococcal urethritis, and others result from periurethral infection associated with indwelling urethral catheterization. Gram-positive bacteria are the most common pathogens found in cultures from prostatic fluid. Adenovirus prostatitis , as seen in immunosuppressed patients, can exhibit prominent necrotizing features. While biopsy would be unusual for a clinically apparent infection, microscopically it usually appears as a localized process involving a small number of ducts or acini. The lumina are distended and filled with secretion mixed with inflammatory cells, among which neutrophils predominate. The stromal component, on the other hand, is mainly mononuclear and is composed of a mixture of lymphocytes, plasma cells, and histiocytes. In the presence of a monotonous infiltration of mature lymphocytes throughout the organ, the alternative diagnosis of involvement by chronic lymphocytic leukemia should be considered. We have seen rare prostate biopsies with CMV inclusions within epithelial and endothelial cells.
Prostatitis is often accompanied by elevation of serum PSA, particularly following a digital rectal examination, which should return quickly to normal following successful antibiotic treatment.
Autoimmune prostatitis (IgG4-related sclerosing disease) has recently been proposed as a disease entity, characterized by elevated serum IgG4 levels and sometimes associated with systemic or multifocal lesions involving other organs. In the histologically typical case, a mixed inflammatory infiltrate rich in plasma cells is seen, accompanied by fibrosis and obliterative phlebitis.
In the past, the majority of prostatic abscesses resulted from gonorrhea. At present, most have an obstructive etiology and are the manifestation of secondary infection of the prostate from an infected pool of residual urine. Escherichia coli is the organism usually responsible. In one series, 36% presented with acute urinary retention and 31% with perineal or suprapubic pain. Prostatic fluctuation on digital rectal examination is the most characteristic sign, transrectal ultrasound is the most reliable diagnostic method, and transurethral drainage under antibiotic coverage is the treatment of choice.
A relatively common form of prostatic abscess in east Asia and northern Australia is melioidosis , an infectious disease produced by the gram-negative bacterium Burkholderia pseudomallei .
The prostate, epididymis, and testis are the organs most commonly involved in tuberculosis of the male genital system. Of 105 autopsy cases included in the classic study by Auerbach, the prostate was involved in 100, and in 35 it was the sole site in the system. In most cases, the infection is the result of hematogenous spread from the lungs (or, less often, from the skeletal system), but it may also result from direct invasion from the urethra.
Early tuberculous lesions in the prostate are seldom detected on palpation. It is only when the disease is advanced that enlargement occurs and fluctuant, tender zones may be felt. Grossly, the lesions are usually bilateral. Confluent caseous zones occur with liquefaction and cavitation, until finally the prostate becomes an enlarged mass with multiple cavities. It may perforate into the urethra and extend into the urinary bladder. With still further spread, sinus tracts may form into the rectum, perineum, and peritoneal cavity. Healing with calcification may supervene, a change detectable by radiographic examination. In the late stages, the prostate becomes shrunken, fibrotic, and hard, to the point that it may simulate carcinoma on palpation.
Microscopically, the initial lesion is in the stroma but quickly spreads to the acini. Well-developed lesions show confluent foci of caseation with incomplete fibrous encapsulation. There is little tendency for the formation of typical tubercles.
Patients treated with intravesical bacillus Calmette–Guérin (BCG) for bladder carcinoma may develop granulomas in the prostate similar to those seen more often in the bladder itself (see Chapter 25 ). These granulomas may be of noncaseating or caseating type. They may be located along the periurethral or transitional zone or involve the gland diffusely. Stains for acid-fast organisms are usually negative, although occasionally a few organisms are visualized.
The prostate can be involved by blastomycosis , coccidioidomycosis , actinomycosis , cryptococcosis , histoplasmosis , aspergillosis , Cytomegalovirus (CMV), and candidiasis . Many of these infections occur in the setting of immunosuppression resulting from AIDS or other disorders.
Chlamydia trachomatis and Trichomonas vaginalis have been identified in the prostate gland, but their possible role as etiologic agents of prostatitis remains to be determined. The Chlamydia organism can be detected in tissue by immunohistochemical and in situ hybridization techniques.
Nonspecific granulomatous prostatitis, sometimes described as “idiopathic,” is applied to a rare prostatic disorder thought to represent an initially immune-mediated process accompanied by a reaction to the prostatic secretions released from obstructed ducts. Most cases occur in glands affected by nodular hyperplasia in patients over 50 years of age. The clinical triad of high fever, symptoms of prostatitis, and a hard prostate on palpation is present in one-fifth of cases and may suggest the diagnosis. A preoperative diagnosis of carcinoma is suspected in about 30% of cases because of the firmness of the lesion, caused by the dense fibrosis.
Grossly, the gland is firm to stony hard. The cut surface shows obliteration of the architecture, with formation of yellow granular nodules. Microscopically, large nodular aggregates of histiocytes, epithelioid cells, lymphocytes, and plasma cells are seen. Characteristically, these granuloma-like formations are centered in the lobules ( Fig. 26.8 ). A tubercle-like reaction with multinucleated giant cells, as well as collections of neutrophils, eosinophils, and detritus within the ducts, also may be seen. Microorganisms and caseation necrosis are absent. The microscopic changes can simulate carcinoma in needle biopsy specimens. Immunohistochemical studies have shown a concentration of T cells in and around damaged ducts and glands.
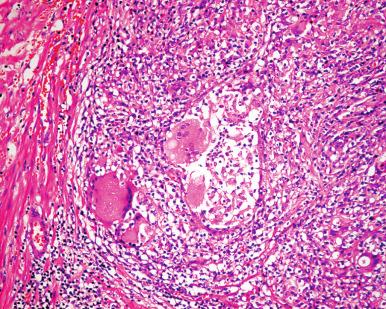
Most cases of prostatitis associated with a predominant eosinophilic infiltrate, sometimes intense, belong to one of the following categories :
Eosinophilic prostatitis (allergic prostatitis, allergic granuloma of prostate), characterized by small stellate necrobiotic nodules surrounded by palisading epithelioid histiocytes and eosinophils, resembling rheumatoid nodules. Vasculitis may be found. Patients often have a history of allergy and asthma and usually exhibit peripheral eosinophilia; in some cases, systemic vasculitis is present. Some of these cases have been thought to be a component of the Churg–Strauss syndrome. Elevation of serum PSA levels may occur in this disorder.
Iatrogenic inflammation , a morphologically similar condition in patients lacking these systemic symptoms that develops after a surgical procedure to the area, usually occurs in a TUR but sometimes a prostatic needle biopsy. The interval between the surgical procedure and the appearance of the prostatitis ranges from less than 1 month to several years; eosinophils are more numerous when this interval is shorter. An associated granulomatous response may represent a reaction to collagen altered as a result of the surgery or to metal deposition from the instruments themselves. Some of these granulomas are elongated and tortuous, whereas others are wedge shaped, with their base facing the cauterized tissue.
Parasitic infestation resulting from metazoa are rarely reported and, as in other sites, may be associated with an intense eosinophilic infiltrate.
Malakoplakia can involve the prostate, usually in association with bladder disease (see Chapter 25 ). As in the latter site, it should be viewed as a peculiar form of tissue reaction to bacterial infection. The infiltrate is usually located around prostatic ducts and has a mixed composition. The diagnosis is possible on needle biopsy specimens. A similar condition but lacking Michaelis–Gutmann bodies has been described as nodular histiocytic prostatitis . Reactive histiocytic infiltrates may closely mimic the morphologic appearance of prostatic adenocarcinoma after hormonal ablation. Malakoplakia may sonographically simulate prostatic carcinoma. It can also be seen in prostates having carcinoma in other portions of the gland.
Vasculitis of either necrotizing, fibrinoid, or granulomatous type can involve the prostate as an isolated event.
Hair granuloma , described exceptionally in TUR specimens, is thought to result from hair from the perineal area being introduced into the prostate by an earlier perineal prostatic needle biopsy.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here