Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Staging has significant impact on prognostic assessement, treatment decision-making, and clinical trial planning and analysis.
Primary tumor (Breslow) thickness and ulceration continue to represent important prognostic factors for survival and define T-category strata in cutaneous melanoma.
Mitotic rate, defined as mitoses per square millimeter, is an important independent prognostic factor, particularly for patients with thin (T1 ≤1 mm) melanoma, and replaces Clark level of invasion as a primary criterion for defining T1b melanoma.
Lymphatic mapping and sentinel node biopsy constitute important components of melanoma staging to identify occult regional lymph node (stage III) disease among patients who present with clinical stage IB or II cutaneous melanoma.
Nodal status is a dominant independent predictor of survival in patients with clinically node-negative primary cutaneous melanoma.
The AJCC melanoma staging system currently represents the most widely accepted algorithm for assessing melanoma prognosis at initial diagnosis.
A uniformly accepted, contemporary, and standardized cancer staging sytem is an essential requirement for meaningful comparisons to be made across patient populations. Elements of a staging system include incorporation of relevant prognostic factors using consistent definitions based on a clinical understanding that reflects current practice and knowledge of disease-specific cancer biology. Whenever possible, an evidence-based approach should be used.
Cutaneous melanoma is one of the few potentially lethal cutaneous conditions to confront medical professionals. Although advances in screening and early detection, as well as increases in overall 5-year survival, are encouraging, the incidence and number of deaths attributable to melanoma continue to increase. Knowledge of prognostic factors and staging are critically important for prognostic assessment, surgical, adjuvant, and systemic treatment planning, and clinical trial design and analysis.
In addition to its obvious clinical relevance, advancing understanding of the factors that affect melanoma prognosis may also improve the understanding of the complex pathophysiology of this disease. As the molecular biology of melanoma progression is unmasked, it is anticipated that, in addition to enhanced prognostic capabilities, therapies will be developed that specifically target dysregulated pathways (see Chapter 55 ). In this chapter, the significance of a myriad of prognostic factors will be presented using as a framework the American Joint Committee on Cancer (AJCC) melanoma staging system and associated recent collaborative database analysis.
As the understanding of the biology of this often capricious disease has improved, the melanoma staging system has been revised numerous times to reflect the identification of increasingly powerful prognostic factors. The AJCC Melanoma Staging Committee significantly revised the melanoma staging system in 2001 (6th edition of the AJCC Cancer Staging Manual ), and this was based on an improved understanding of melanoma-associated prognostic factors, including data from the largest international melanoma prognostic factor survival analysis ever conducted at the time. Features of the revised system, formally introduced into clinical practice in 2002, included new strata for primary tumor thickness, incorporation of primary tumor ulceration as an important staging criterion in both the T and N classifications, revision of the N classification to reflect the importance of regional nodal tumor burden, and new categories for stage IV. A comprehensive and expanded AJCC collaborative melanoma database analysis was performed in 2008; prior revisions were also validated in the 7th edition (2009) AJCC melanoma staging system. In addition to discussion of this analysis, relatively minor modifications to the T, N, and M classifications included in this recently published edition and formally implemented January 2010, are discussed in detail below ( Tables 26.1 and 26.2 ).
Highlights of the recommended changes for the 7th edition of the AJCC Cancer Staging Manual include the following:
Melanoma thickness and primary tumor ulceration continue to define T category strata. Primary tumor mitotic activity (defined as mitoses/mm 2 ) is an important adverse predictor of survival. For T1 melanoma, a mitotic rate of at least 1 mitosis/mm 2 replaces level of invasion as a primary criterion for defining T1b.
The presence of nodal micrometastases can be defined by either hematoxylin and eosin (H&E) or immunohistochemical staining (previously, only H&E could be used).
There is no lower threshold of tumor burden defining the presence of regional nodal metastasis. Based on a consensus that volume of regional metastatic tumor deposits <0.2 mm in diameter (an empiric lower threshold previously used by the AJCC for defining nodal metastasis in breast cancer patients) is clinically significant in melanoma patients, nodal tumor deposits of any size are included in staging nodal disease. An evidence-based lower threshold of clinically insignificant nodal metastases has not yet been defined.
Survival estimates for patients with intralymphatic regional metastases (i.e. satellites and in-transit metastases) are somewhat better than for the remaining cohort of stage IIIB patients; nonetheless, the current stage definition for intralymphatic regional metastasis has been retained, since this stage grouping represents the closest statistical fit.
The prognostic significance of microsatellites remains controversial. This uncommon feature has been retained in the N2c category, largely because previous published studies were insufficient to substantiate revision of the definitions used in the 6th edition of the staging manual.
The site of distant metastases remains the primary component of the M category (non-visceral, M1a [cutaneous, subcutaneous, distant nodal]; lung, M1b; and visceral metastasis, M1c). An elevated serum lactic dehydrogenase (LDH) level remains a powerful adverse predictor of survival; patients with an increased LDH are all categorized as M1c, regardless of the sites of distant disease.
No subgroups of stage IV melanoma are recommended.
The definition of metastatic melanoma from an unknown primary site was clarified; isolated metastases arising in lymph nodes, skin, and subcutaneous tissues are categorized as stage III rather than stage IV.
Lymphatic mapping and sentinel lymph node (SLN) biopsy are important components of melanoma staging and should be used (or at least discussed with the patient) in defining occult stage III disease among patients who present with clinical stages IB and II melanoma.
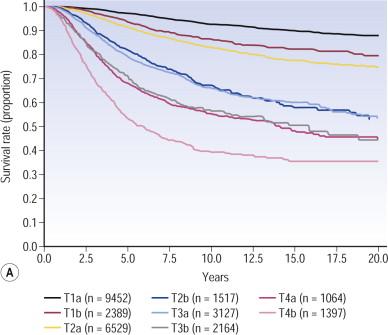
The 7th edition melanoma staging system TNM categories are defined in Table 26.1 and the stage groupings in Table 26.2 . These revisions are based on the updated AJCC collaborative melanoma database that contained prospective data on over 50,000 patients with stages I–III melanoma, and nearly 10,000 patients with stage IV melanoma. Interestingly, 5-year survival rates over the 5-year interval between this and the prior staging system demonstrate some increase in survival, likely due to the improvements in melanoma staging (i.e. more widespread use of sentinel node biopsy for patients with T1b–T4 lesions) and treatment ( Table 26.3 ). Serving as an introduction to melanoma staging and prognosis, the overall heterogeneity of survival among patients with varying stages of disease is illustrated by the survival estimates for patients with stages I to IV melanoma ( Fig. 26.1 ).
| T Classification | Thickness | Ulceration Status |
|---|---|---|
| Tis | NA | NA |
| T1 | ≤1.00 mm | a: w/o ulceration and mitosis <1/mm 2 |
| b: with ulceration or mitoses ≥1/mm 2 | ||
| T2 | 1.01–2.0 mm | a: w/o ulceration |
| b: with ulceration | ||
| T3 | 2.01–4.0 mm | a: w/o ulceration |
| b: with ulceration | ||
| T4 | >4.0 mm | a: w/o ulceration |
| b: with ulceration |
| N Classification | # of Metastatic Nodes | Nodal Metastatic Burden |
|---|---|---|
| N0 | 0 | NA |
| N1 | 1 | a: micrometastasis * |
| b: macrometastasis † | ||
| N2 | 2–3 | a: micrometastasis * |
| b: macrometastasis † | ||
| c: in-transit met(s)/satellite(s) without metastatic nodes | ||
| N3 | 4+ metastatic nodes, or matted nodes, or in-transit metastases/satellites with metastatic nodes |
| M Classification | Site | Serum LDH |
|---|---|---|
| M0 | No distant metastases | NA |
| M1a | Distant skin, subcutaneous, or nodal metastases | Normal |
| M1b | Lung metastases | Normal |
| M1c | All other visceral metastases | Normal |
| Any distant metastasis | Elevated |
* Micrometastases are diagnosed after sentinel lymph node biopsy.
† Macrometastases are defined as clinically detectable nodal metastases confirmed pathologically.
| Clinical Staging * | Pathologic Staging † | ||||||
|---|---|---|---|---|---|---|---|
| T | N | M | T | N | M | ||
| 0 | Tis | N0 | M0 | 0 | Tis | N0 | M0 |
| IA | T1a | N0 | M0 | IA | T1a | N0 | M0 |
| IB | T1b | N0 | M0 | IB | T1b | N0 | M0 |
| T2a | N0 | M0 | T2a | N0 | M0 | ||
| IIA | T2b | N0 | M0 | IIA | T2b | N0 | M0 |
| T3a | N0 | M0 | T3a | N0 | M0 | ||
| IIB | T3b | N0 | M0 | IIB | T3b | N0 | M0 |
| T4a | N0 | M0 | T4a | N0 | M0 | ||
| IIC | T4b | N0 | M0 | IIC | T4b | N0 | M0 |
| III | Any T | N > N0 | M0 | IIIA | T1–4a | N1a | M0 |
| T1–4a | N2a | M0 | |||||
| IIIB | T1–4b | N1a | M0 | ||||
| T1–4b | N2a | M0 | |||||
| T1–4a | N1b | M0 | |||||
| T1–4a | N2b | M0 | |||||
| T1–4a | N2c | M0 | |||||
| IIIC | T1–4b | N1b | M0 | ||||
| T1–4b | N2b | M0 | |||||
| T1–4b | N2c | M0 | |||||
| Any T | N3 | M0 | |||||
| IV | Any T | Any N | M1 | IV | Any T | Any N | M1 |
* Clinical staging includes microstaging of the primary melanoma and clinical/radiologic evaluation for metastases. By convention, it should be used after complete excision of the primary melanoma with clinical assessment for regional and distant metastases.
† Pathologic staging includes microstaging of the primary melanoma and pathologic information about the regional lymph nodes after partial (i.e. sentinel lymph node biopsy) or complete lymphadenectomy. Pathologic stage 0 or stage IA patients are the exception; they do not require pathologic evaluation of their lymph nodes.
| T Classification | 5-Year Survival Rate (SE) | |||
|---|---|---|---|---|
| 6th Edition (2001) | 7th Edition SLN Subset * | |||
| % ± SE | (n) | % ± SE | (n) | |
| T1a | 95 ± 0.4 | 4150 | 97 ± 0.2 | 9452 |
| T1b | 91 ± 1.0 | 1380 | 95 ± 1.5 | 597 |
| T2a | 89 ± 0.7 | 285 | 95 ± 0.6 | 2842 |
| T2b | 77 ± 1.7 | 958 | 86 ± 2.0 | 614 |
| T3a | 79 ± 1.2 | 1717 | 85 ± 1.5 | 1158 |
| T3b | 63 ± 1.5 | 1523 | 76 ± 2.5 | 677 |
| T4a | 67 ± 2.4 | 63 | 76 ± 3.6 | 338 |
| T4b | 45 ± 1.9 | 978 | 67 ± 3.6 | 386 |
* Patients staged based on sentinel node staging for T1b–T4.
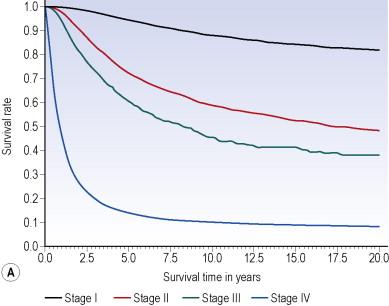
The overall prognosis for patients who present with localized melanoma without regional lymph node or distant metastases is generally favorable ( Fig. 26.1A,B ). In the AJCC analysis, among 25,734 patients with localized melanoma (documented clinically or pathologically), tumor thickness and ulceration were the dominant independent predictors of survival ( Table 26.4 ). Similar results were obtained in the cohort of 9883 patients without clinical evidence of nodal metastases at initial presentation, whose regional nodes were pathologically staged primarily by sentinel node biopsy ( Table 26.5 ). In this latter group, nodal status was the most significant independent predictor, and level of invasion was no longer a predictor of outcome.
| Variable | Without Mitotic Rate in Model (N=25,754) | With Mitotic Rate in Model (N=10,233) | ||||
|---|---|---|---|---|---|---|
| Chi-Square | P | HR (5% CI) | Chi-Square | P | HR (95% CI) | |
| Tumor thickness | 420.1 | <0.0001 | 1.35 (1.31–1.39) | 84.6 | <0.0001 | 1.25 (1.91–1.31) |
| Ulceration | 257.3 | <0.0001 | 1.93 (1.78–2.10) | 47.2 | <0.0001 | 1.56 (1.38–1.78) |
| Clark level | 46.3 | <0.0001 | 1.22 (1.16–1.30) | 8.2 | 0.0041 | 1.15 (1.04–1.26) |
| Mitotic rate | – | – | – | 79.1 | <0.0001 | 1.26 (1.20–1.32) |
| Site | 75.6 | <0.0001 | 1.41 (1.30–1.52) | 29.1 | <0.0001 | 1.38 (1.23–1.54) |
| Gender | 46.5 | <0.0001 | 0.76 (0.71–0.83) | 32.4 | <0.0001 | 0.70 (0.62–0.79) |
| Age | 90.0 | <0.0001 | 1.16 (1.13–1.20) | 40.8 | <0.0001 | 1.16 (1.11– 1.22) |
| Variable | Chi-Square | Hazard Ratio (95% CI) | P |
|---|---|---|---|
| Nodal status | 222.5 | 2.52 (2.23–2.84) | <0.0001 |
| Thickness | 162.1 | 1.33 (1.27–1.38) | <0.0001 |
| Ulceration | 113.4 | 1.93 (1.71– 2.18) | <0.0001 |
| Site | 36.1 | 1.45 (1.28–1.63) | <0.0001 |
| Age | 47.6 | 1.20 (1.14–1.26) | <0.0001 |
| Gender | 11.3 | 0.80 (0.71–0.91) | 0.0008 |
| Clark level | 2.1 | 1.11 (0.96–1.29) | 0.1512 |
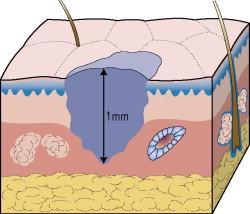
One of the earliest descriptions of tumor thickness was published by Breslow in 1970, when he theorized that primary melanoma tumor volume correlates with prognosis. Realizing that accurate and reproducible tumor volume measurements are essential yet difficult to perform, he reasoned that the maximal thickness of the primary melanoma (measured from the granular layer to the deepest component of the tumor using an ocular micrometer) ( Fig. 26.2 ) would be an accurate, reproducible surrogate for tumor volume and a significant predictor of prognosis. This concept has since been validated in multiple studies, including the AJCC analysis. In the 7th edition AJCC staging system, the cutoff points for tumor thickness strata remain at 1 mm, 2 mm, and 4 mm on the basis of prior statistical modeling, the lack of naturally occurring break points in the analysis, and utility as thresholds for clinical decision-making ( Tables 26.1 and 26.2 ). To date, tumor thickness remains the most powerful primary tumor-derived prognostic factor in melanoma.
When a patient presents with a lesion suggestive of melanoma, biopsy followed by histologic examination is essential. While specific biopsy techniques are addressed elsewhere, full-thickness biopsy into the subcutaneous tissue must be performed to permit proper microstaging of the lesion. In this setting, shave biopsies are generally discouraged, since failure to obtain a full-thickness specimen may result in inaccurate microstaging if melanoma is diagnosed.
Primary tumor ulceration has also been recognized as an important predictor of survival in clinically node-negative patients. Ulceration is defined as the absence of an intact epidermis (i.e. loss of continuity of the overlying epithelium) overlying the primary tumor on histologic examination and is a reproducibly identifiable feature of melanoma ( Fig. 26.3 ).
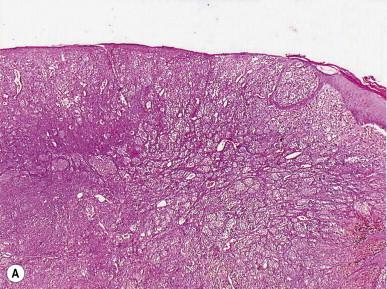
Numerous studies have shown that patients with ulcerated primaries have a higher likelihood of metastases and a worse prognosis compared to patients with non-ulcerated tumors when controlled for tumor thickness. Although the biological basis of ulceration remains speculative, it may be related to the inherent aggressiveness of the tumor itself or possibly other stromal factors, such as angiogenic support. As noted in both the 2001 and the 2008 AJCC prognostic factor analyses, patients with ulcerated primaries had outcomes approximately the same as those of patients in the next-higher tumor thickness stratum whose lesions were not ulcerated ( Fig. 26.4 ).
In the AJCC T classification, ulceration at each tumor thickness level is categorized as either absent or present. Spatz et al. developed a clear definition of histologic criteria for traumatic ulceration, an important contribution since this form of ulceration has no prognostic value, but non-traumatic ulceration has independent prognostic significance. The incidence of ulceration among primary tumors is quite heterogenous, ranging from approximately 3% in patients with T1 lesions to 57% among those with T4 melanoma.
Importantly, ulceration is a primary tumor feature that also carries independent prognostic significance in patients with stage III (regional node) disease (see below). Ulceration has been included in the AJCC staging system for melanoma since 2002.
Accumulating evidence supports the notion that primary tumor mitotic rate is an important independent prognostic factor in patients with cutaneous melanoma. In 1983, Schmoeckel et al. found a correlation between mitotic rate and prognosis. In the largest single institution study to date, the Sydney Melanoma Unit determined by Cox multivariate analysis that tumor mitotic rate (expressed as the number of mitoses/mm 2 ) was the second most powerful independent prognostic factor after tumor thickness, an observation also noted by others investigators.
As a result of numerous studies showing an inverse association between primary tumor mitotic rate and survival, this factor was included for the first time in the 2008 AJCC collaborative melanoma database analysis. The number of mitoses/mm 2 correlated strongly with tumor thickness. By univariate analysis, there was a significant inverse correlation between the number of mitoses/mm 2 (using the so-called ‘hot spot’ approach) and survival. Remarkably, among the 11,664 patients with stage I or II melanoma in whom mitotic rate was known, it was the second most powerful independent adverse predictor of survival ( Table 26.4 ). Tumor ulceration remained highly significant, whereas level of invasion maintained only borderline significance. Among patients with T1 (i.e. ‘thin’) melanomas, the presence of at least 1 mitosis/mm 2 was an independent adverse predictor of survival, and contributed to the decision to include mitotic rate as a primary tumor (T-category) criterion for patients with thin melanoma ( Tables 26.1 , 26.2 , and 26.6 ).
| Thickness (mm) | Mitosis per mm 2 | N | Survival Rate ± SE | |
|---|---|---|---|---|
| 5-Year | 10-Year | |||
| 0.01–0.50 | <1.0 | 1194 | 99.1 ± 0.4 | 97.4 ± 8.6 |
| 0.01–0.50 | ≥1.0 | 327 | 97.0 ± 1.2 | 95.2 ± 1.7 |
| 0.51–1.00 | <1.0 | 1472 | 97.7 ± 0.5 | 93.0 ± 1.0 |
| 0.51–1.00 | ≥1.0 | 1868 | 93.5 ± 0.6 | 87.1 ± 1.2 |
Standardized assessment of tumor mitotic rate using the ‘hot spot’ method (expressed as mitoses/mm 2 ) is now recommended by the AJCC as a required element of the primary tumor pathology report. The recommended approach to mitotic rate measurement is detailed in the 7th edition AJCC Cancer Staging Manual . This involves finding the so-called ‘hot spot’ by identifying the area in the dermis containing the greatest concentration of mitotic figures. After counting the mitoses in the hot spot, the mitoses in adjacent fields are counted until an area corresponding to 1 mm 2 has been assessed. Usually this will mean assessment of four high-power fields at 400× magnification, but calibration of individual microscopes is required since field sizes may vary considerably. If no hot spot can be identified and mitoses are sparse and/or randomly distributed throughout the tumor, then a single mitosis is selected, and beginning with the count in that field, the mitoses in adjacent fields are counted until an area of 1 mm 2 has been assessed. All mitotic rate results should be reported as mitoses/mm 2 . When the invasive component of a melanoma is <1 mm 2 in area, the mitotic rate can be reported as ≥1/mm 2 (if at least 1 mitosis is identified) or 0/mm 2 (if no mitoses are identified).
In 1969, Clark and colleagues described the histologic classification of melanoma by its level of invasion into the dermis or subcutaneous tissue. Five categories of invasion analogous to the current five Clark levels were described ( Fig. 26.5 ). However, the prognostic significance and reproducibility of Clark level have been debated. In most, but not all, comparative studies, including analyses of large prospective databases, tumor thickness was shown to be a more powerful prognostic factor and more reproducible measurement than the Clark level.
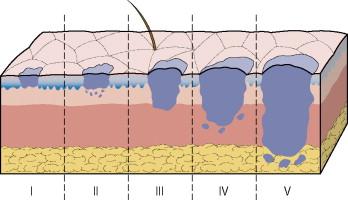
More recent analyses have confirmed the importance of tumor thickness as a prognostic factor and found that level of invasion was significant only for melanoma lesions ≤1 mm in thickness. Similarly, in the 2002 AJCC database analysis, the level of invasion had greater prognostic significance than ulceration only for tumors ≤1 mm. While Clark level was included in the 2002 staging system for T1 lesions only, it has been supplanted by mitotic rate as a primary criterion for T1b melanoma (with ulceration) in the 7th edition melanoma staging system (except in those rare instances when mitotic rate cannot be determined) since it was not found to be prognostic when both mitotic rate and ulceration were in the model.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here