Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
There was an estimated 233,000 new cases of prostate cancer in the United States in 2014 and many of these men would have been treated with radical prostatectomy. Incontinence is a well-documented complication of this procedure and, regardless of a robotic or open approach, incontinence rates seem to be similar, ranging from 1% to 40% of all patients at one year. Various definitions of postradical prostatectomy incontinence (PRPI) have been used in the literature, and when the definition is expanded to include occasional dribbling, up to 74% of men will report PRPI. This is often not a temporary issue; urinary incontinence after prostatectomy continues to significantly affect health-related quality-of-life for men many years after surgery.
After patients undergo an appropriate evaluation, they can be offered a variety of therapies that range from conservative management to surgical intervention. Conservative treatments include behavioral modifications and lifestyle changes, penile clamps, condom catheters, biofeedback, pharmacotherapy, electrical stimulation, and extracorporeal magnetic stimulation. If these are unsuccessful, patients can undergo more invasive therapies that include bulking agents, periurethral constrictors, balloon compression of the urethra, bulbar urethral slings, artificial urinary sphincters, and possibly, in the future, stem cell therapy.
Radical prostatectomy can be accomplished by four different approaches: perineal, open retropubic, laparoscopic, or robotic-assisted laparoscopic. The most common procedures used currently are the open retropubic and robotic-assisted laparoscopic prostatectomies. Perineal prostatectomy fell out of favor in the 1980s with the advent of the nerve sparing retropubic prostatectomy and in 2007 less than 2% of the radical prostatectomies performed were by a perineal approach. Pure laparoscopic prostatectomy has been shown to have similar operative and postoperative results as open and robotic prostatectomy, but is considered to be a technically more challenging procedure with a longer learning curve, and is not widely used.
Postradical prostatectomy urinary incontinence can be broadly classified by etiology and contributing factors ( Table 32.1 ). Continence in the male requires
Accommodation of increasing volumes of urine at a low intravesical pressure (normal compliance) and with appropriate sensation.
A bladder outlet that is closed at rest and remains so during increases in intra-abdominal pressure.
Absence of involuntary bladder contractions (detrusor overactivity).
| Functional |
|
| Bladder abnormalities |
|
| Outlet abnormalities |
|
| Combination |
Outlet terminology differs among authors and requires some explanation. We prefer to use the terms smooth and striated sphincter area. The smooth sphincter refers to the smooth muscle of the bladder neck and proximal urethra. This is a physiologic but not anatomic sphincter and is not under voluntary control. Others refer to this area as the internal sphincter, the proximal sphincter, and the bladder neck sphincter. There is virtually no physiologic or pharmacologic response that affects the smooth muscle of the most proximal urethra without affecting the smooth muscle of the bladder neck. Normally, the smooth sphincter is competent and resistance increases in this area during bladder filling and urine storage and decreases during an emptying contraction. In the proximal urethra there is an inner longitudinal smooth muscle layer and a thinner outer circular layer. Many believe the longitudinal layer is continuous with the musculature of the bladder base. Teleologically this is consistent with a tonic role of the circular muscle in helping to maintain closure during filling and storage and a phasic role for the longitudinal layer in contributing to opening of this area during voiding. The function of this entire proximal sphincter mechanism is essentially negated by radical prostatectomy.
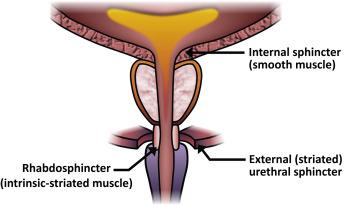
The classic view of the “external urethral sphincter” is that of a sheet of striated muscle within the leaves of a “urogenital diaphragm” that extends horizontally across the pelvis. This is under voluntary control and is responsible for stopping the urinary stream in response to the command “stop voiding.” The striated sphincter concept expands this definition to include intramural and extramural portions ( Figure 32.1 ). The extramural portion, actually periurethral striated muscle, corresponds roughly to the “classic” external urethral sphincter. The intramural portion denotes skeletal muscle that is intimately associated with the urethra from the maximal condensation of the extramural striated muscle to at least the apex of the prostate. This intramural portion is called the rhabdosphincter and includes the intrinsic smooth muscle in that area as well. The prevailing view is that the striated component of the rhabdosphincter contains predominately slow twitch (Type 1) striated muscle, which can maintain resting tone and preserve continence. The fibers of the periurethral (extrinsic to the urethra) striated muscle are fast twitch (Type 2) and capable of rapid on-demand contraction. The striated muscle of the rhabdosphincter is considerably thicker ventrally and invested in a fascial framework that is supported below by a musculofascial plate that fuses with the midline raphe, a point of origin for the rectourethralis muscle. It is this complex that is largely responsible for preservation of the sphincteric function of the outlet after radical prostatectomy. Damage to this area or its innervations, rendering it poorly or nonfunctional, is a major cause of PRPI.
Urinary incontinence after prostatectomy is predominantly sphincter related and the amount can range from a few drops with exercise or changing positions to total or near total leakage. Detrusor dysfunction is often found in such patients but is seldom the sole cause. There are many series that have characterized urodynamic findings in these patients. Two are illustrative. Ficazzola and Nitti found that 27 of 60 men (45%) with PRPI had some element of detrusor overactivity but this significantly contributed to the incontinence in only 16 (27%). Intrinsic sphincter deficiency (ISD) was found in 54 (90%). The PRPI was attributed to ISD alone in 67%, ISD and detrusor overactivity and/or decreased compliance in 23%, and bladder dysfunction alone in 3%. Groutz et al. found that detrusor overactivity was present in 28 of 83 patients (33.7%), but was the only urodynamic finding in 3 patients (3.6%) and ISD was the most common finding and the dominant cause in 73 (88%). They also mention “low urethral compliance” presumably from scarring and fibrosis as a significant cause of ISD in 25 men (30.1%).
Overflow incontinence is one of those entities that is easy to recognize but hard to urodynamically define. It is an uncommon cause of PRPI, usually due to postoperative bladder neck contracture, but one that must be ruled out.
There is a long list of factors that have been associated by some clinicians with the occurrence of PRPI, but unanimous agreement on all is lacking. These are summarized compositely by Koelbl et al. , Herschorn et al. , and Xie and Sandhu and include advanced age, obesity, decreased overall well-being, prior irradiation, prior prostatectomy or ablation for benign disease, anastomotic structure, anastomotic extravasation, and prostate size and configuration. Some factors that may be protective against PRPI include nerve sparing prostatectomy, seminal vesical preservation, bladder neck preservation, puboprostatic ligament preservation, bladder neck intussusception or other alteration, posterior reconstruction of Denonvillier’s musculofascial plate, anterior urethral suspension or support, urethral preservation, and having an experienced surgeon. In the final analysis, meticulous surgical technique of the apex is probably the most important single factor that we as surgeons can influence.
The use of MRI has highlighted the apparent adverse effect of periurethral or perisphincter fibrosis and a “short” urethral length (from verumontanum to proximal bulb). Tuygun et al. have cited a much higher incidence of perisphincter fibrosis in incontinent than continent men at 6 months or more after prostatectomy (22/22 vs. 4/14). The Penn group reported that preoperative increased urethral length was related to decreased time to continence as a dichotomous (<20 mm vs. >20 mm) and continuous variable. Dubbelman et al. reviewed a number of assessment techniques for urethral function as predictors of postradical prostatectomy continence status. Their findings include the following:
There is no role in this respect for sphincter electromyography.
Postoperative abdominal leak point pressures correlated with the presence of ISD but not severity of continence.
Preoperative urethral profilometry “seems to be important” in prediction of PRPI. The maximal urethral closure pressure seems to be the most important parameter.
Preoperative urethral length (apex of prostate to bulb of penis, as defined by Myers et al. ) is associated with PRPI (shorter length, higher chance), although no cut-off values are proposed. Coakley et al. found that of those with a preoperative length of >12 mm 89% were continent at 1 year versus 77% with <12 mm.
MRI evidence of perisphincter fibrosis “might” have a negative effect on sphincter function
Opinions differ regarding the effect on continence of postoperative radiation delivered to the prostatic fossa of postradical prostatectomy patients. Van Cangh et al. and Formenti et al. reported no effect. Suardi et al. found a detrimental effect on continence as did Hofmann et al.
Virtually all men are incontinent immediately following prostatectomy, and it is recommended that they undergo at least 6–12 months of recovery prior to any invasive intervention. The evaluation for PRPI begins with a detailed history and physical examination. It is critical to characterize the type of incontinence (stress or urgency) and the severity. A voiding diary for 2–7 days has been validated as a tool to assess urge and incontinence episodes. Additionally, a 24-h pad test can provide more objective data on degree of incontinence. There are currently efforts in place to validate a questionnaire that accurately assesses quality of life based on pad weight, which may further help delineate degrees of PPI and direct treatments. Physical exam should assess for any previous surgical incision sites and the presence of an inguinal hernia that could affect placement of an artificial urinary sphincter. In addition, all patients should have a urine culture sent as any UTI must be treated prior to intervention. Finally, a postvoid residual, prostate-specific antigen and creatinine level should be sent to evaluate for voiding dysfunction, cancer status, and renal function.
Further evaluation with cystoscopy and multichannel urodynamics is recommended for all men considering surgical intervention. If an anastomotic stricture, bladder neck contracture, or urethral stricture is present it can complicate a surgical intervention and these need to be treated prior to any surgery for incontinence. Furthermore, during cystoscopy the integrity of the external sphincter can be evaluated, which may influence the choice of surgical treatment. Urodynamic testing is used to ascertain bladder capacity, bladder compliance, contractility, and the presence of detrusor overactivity, and can differentiate between stress, urgency, and mixed incontinence. It is important to identify patients with concomitant detrusor overactivity and urgency incontinence, as they may have less successful outcomes following surgery. Relevant Oxford Levels of Evidence (LOE) and Grades of Recommendation (GOR) from the International Consultation on Incontinence with respect to evaluation are :
Basic history, physical exam, urinalysis, and postvoid residual (LOE1-2, GOR A);
Voiding diary (LOE 1-2, GOR B);
Pad tests (LOE 1-2, GOR B);
Cystoscopy, appropriate imaging (LOE 2-3, GOR B);
Multichannel urodynamics may be useful prior to invasive treatment (LOE 3, GOR C).
In the first 6–12 months after prostatectomy, it is generally recommended that patients attempt a variety of conservative, nonsurgical measures that may help improve PRPI. Common sense suggests that all patients should adopt simple lifestyle modifications that include limiting total fluid intake, timed voiding, and avoiding foods and drinks such as alcohol or caffeinated beverages that can cause irritation to the bladder. The ICI Committee on adult conservative management cautions, however, that to date, no trials have addressed the topic of lifestyle interventions alone for men with any type of urinary incontinence. Pelvic floor muscle training may likewise be initiated early in the postoperative period. For those men who are frustrated by pad use but are not interested in pursuing any additional interventions, the use of condom catheters or penile clamps may be considered. These patients need to be carefully monitored for penile erosion. Additional conservative therapies that have been reported to improve symptoms and quality of life include biofeedback, electrical stimulation, external magnetic stimulation, and pharmacotherapy.
Overall the effects of conservative treatments for urinary incontinence in men, both bladder and sphincter related, have received much less attention than in women. As a consequence, ICI recommendations for men are less numerous and less robust. Enthusiastic individual reports are often tempered by lesser ICI Committee recommendations, formulated after consideration of global data.
Pelvic floor muscle training (PFMT), or Kegel exercises, is defined as “any program of repeated voluntary pelvic floor muscle contractions taught by a health care professional.” The goal is to increase the strength of pelvic muscles and improve voluntary control of the pelvic floor via skeletal muscle contraction and support of urethral coaptation. While there is significant variation in treatment regimens, pelvic floor muscle training has been prospectively evaluated in the postprostatectomy population and been found to lessen the time to regain continence, but not change the final percent of those achieving “continence.” Given that there is no harm to pelvic floor muscle training, it is advisable that all men begin an exercise regimen promptly after prostatectomy. Furthermore, pelvic floor muscle training can improve incontinence even in men who have persistent PPI after a year. In a prospective study of over 200 men with incontinence more than a year after prostatectomy, pelvic floor muscle training decreased incontinence episodes by 51%.
Biofeedback (BF) may be used in combination with pelvic floor muscle training to help coach patients how to properly use these muscles. It involves monitoring muscle contractions with EMG so patients can learn how to appropriately contract them. Randomized trials have compared biofeedback enhanced pelvic floor muscle therapy to no training or usual care and have suggested that significantly more men were continent at one to two months after prostatectomy with biofeedback, but this significance was no longer present at 3–4 months. When comparing patients who were treated with biofeedback-enhanced pelvic floor muscle therapy to those who received written or verbal instructions on pelvic floor muscle therapy, three prior randomized studies have shown no significant difference between treatments. However, a recent randomized trial showed good effect. Men undergoing radical prostatectomy who were randomized to biofeedback training and instruction on a structured program of pelvic floor muscle training the day prior to surgery with monthly biofeedback sessions at home had significantly better continence at three and six months after surgery than men who simply received written instructions.
The Committee on Conservative Management of the ICI offered the following conclusions :
The evidence that therapist delivered PFMT with or without BF before or after radical prostatectomy improves continence recovery remains inconsistent.
Some preoperative or immediate postoperative instruction in PFMT may be helpful (GORB B) but whether this is in the form of hands-on therapy or verbal instruction and support remains unclear.
There is some evidence that those who undergo some sort of conservative management, including PFMT, will achieve continence in a shorter time frame but the difference is not significant at 12 months postoperatively (LOE 2).
It is not clear whether PFMT taught by digital rectal exam offers any benefit over verbal or written instruction (GOR B).
The use of BF to assist PFMT is currently a therapist/patient decision based on economics and preference.
For men with PRPI there does not appear to be a benefit to adding electrical stimulation (ES) to a PFMT program (GOR B) although ES may help achieve continence earlier.
In the absence of sufficient data from rigorous and well-reported trials it is not known whether ES, as a stand-alone treatment for male urgency or stress urinary incontinence, is better than no treatment, placebo, or control treatments.
It is not known if pre- or postoperative ES or magnetic stimulation (MS) has a role in reducing PRPI.
The Cochrane Collaboration reviewed conservative management for postprostatectomy incontinence up to November 2009 and concluded that the benefits were uncertain. They further stated that long-term incontinence could be managed by external penile clamp but there are safety concerns.
The European Association of Urology Guidelines are in general agreement and state explicitly that the evidence is inconsistent as to whether ES alone can improve urinary incontinence (LOE 2) and that MS does not cure or improve incontinence (LOE 2).
Pharmacologic therapy is useful management of the overactive bladder symptom syndrome and DO. This subject is exhaustively reviewed in the ICI Consultation and in the ICI Oxford Assessments and the International Consultation on Male LUTS. Since PRPI is mostly an outlet related phenomenon, the reader is referred to these excellent publications for a description of the therapy of OAB-related incontinence and mixed incontinence in the male. It should be noted that the populations studied in the trials considered for the ICI Oxford Assessments consisted primarily of women.
Similarly, the literature relevant to the pharmacotherapy of sphincteric incontinence relates primarily to stress urinary incontinence in women and the treatment of men with stress urinary incontinence after radical prostatectomy has received little attention. Tsakiris et al. searched for articles on drug treatment of male SUI published between 1966 and June 2007 and did a generalized database search in addition. Only nine trials were identified using alpha adrenergic agonists, beta-2 antagonists or SNRSs. Only one of these included a comparison arm, 40 mg twice daily duloxetine plus pelvic floor exercise versus pelvic floor exercise with placebo. Duloxetine is a serotonin–norepinephrine reuptake inhibitor which, in the cat acetic acid model of irritated bladder function, has been shown to significantly increase striated sphincteric activity only during the filling-storage phase of the micturation cycle. The results suggested a positive effect of the drug but were not straightforward. Of those patients completing the 4 month trial, (91/112) 78% of the drug patients versus 52% of the placebo group were “dry.” However, one month after the trial ended, 46% versus 73% were dry, respectively, a shift still observed 2 months later. The authors of the article suggested larger, well-designed studies on duloxetine for this potential usage.
Cornu et al. reported on a series of men with stress or mixed (stress predominant) urinary incontinence postradical prostatectomy who were randomized to duloxetine or placebo after a 2-week placebo run in. Dosage was 20 mg BID for 7 days, 40 mg BID for 67 days, and 20 mg for 14 days. Subjects were at least a year out from surgery. Outcome measures included percent decrease in incontinence episode frequency (IEF), 1 h pad test, and various QOL measures. Statistical significance for IEF percent decrease occurred only at weeks 8 and 12, but there was clearly a trend at 4 weeks as well. There was no statistical difference in 1 h pad test weights but there was in various quality of life scores. A 50–100% decrease in IEF was seen at 12 weeks in over half the patients. Adverse events for drug and placebo included fatigue (50% vs. 13%), insomnia (25% vs. 7%), libido loss (19% vs. 7%), constipation (13% vs. 7%), nausea (13% vs. 7%), diarrhea (13% vs. 7%), dry mouth (6% vs. 0%), anorexia (6% vs. 0%), and sweating (25% vs. 20%). Limitations are the small number of patients (the original proposed sample size was 90) and the lack of any placebo effect on IEF and QOL. One would logically not expect improvement to continue after drug withdrawal unless a permanent change occurred in behavior, anatomy or neuromuscular function. However, in an uncontrolled usage study on men with post-RP SUI, Collado Serra et al. reported that the benefit similarly remained in 85% of the patients after the drug was stopped. In that series, 25% of the patients withdrew because of AEs and 33% because of lack of effect.
Regarding duloxetine the EAU guidelines state that
Duloxetine does not cure urinary incontinence (LOE 1a).
Duloxetine causes significant gastrointestinal and CNS side effects leading to a high rate of treatment discontinuation (LOE 1a).
Duloxetine 80 mg daily can improve SUI in men (LOE 1b).
Duloxetine can be offered to women or men who are seeking temporary improvement in incontinence symptoms (GOR A).
Duloxetine should be initiated using drug titration because of high adverse event rates (GOR A).
Usage of duloxetine for PRPI in the male is off label. Larger controlled and better-designed studies are necessary to provide conclusive positive or negative data on this subject.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here