Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Cardiac anesthesia has fundamentally shifted from a high-dose narcotic technique to a more balanced approach using moderate-dose narcotics, shorter-acting muscle relaxants, and volatile anesthetic agents.
This new paradigm has also led to renewed interest in perioperative pain management involving multimodal techniques that facilitate rapid tracheal extubation such as regional blocks, intrathecal morphine, and supplementary nonsteroidal antiinflammatory drugs.
This approach has prompted a change from the classical model of recovering patients in the traditional intensive care unit manner, with weaning protocols and intensive observation, to management more in keeping with the recovery room practice of early extubation and rapid discharge.
Fast-track cardiac anesthesia appears to be safe in comparison with conventional high-dose narcotic anesthesia, but if complications occur that would prevent early tracheal extubation, the management strategy should be modified accordingly.
The initial management in the postoperative care of fast-track cardiac surgical patients consists of ensuring an efficient transfer of care from operating room staff to cardiac recovery area staff, while at the same time maintaining stable patients' vital signs.
Pulmonary complications following cardiopulmonary bypass are relatively common, with up to 12% of patients experiencing some degree of acute lung injury and approximately 1% requiring tracheostomy for long-term ventilation.
Risk factors for respiratory insufficiency include advanced age, presence of diabetes or renal failure, smoking, chronic obstructive lung disease, peripheral vascular disease, previous cardiac operations, and emergency or unstable status.
Patients with preexisting chronic obstructive lung disease have higher rates of pulmonary complications, atrial fibrillation, and death.
Operating room events that increase risk include reoperation, blood transfusion, prolonged cardiopulmonary bypass time, and low–cardiac output states, particularly if a mechanical support device is required.
Hospital-acquired infections are important causes of postoperative morbidity. Strategies to reduce the incidence of ventilator-associated pneumonia include early removal of gastric and tracheal tubes, formal infection control programs, hand washing, semirecumbent positioning of the patient, use of disposable heat and moisture exchangers, and scheduled drainage of condensate from ventilator circuits.
Patients at risk for acute lung injury and those developing acute respiratory distress syndrome should be switched to a lung-protective ventilation strategy, which involves maintaining peak inspiratory pulmonary pressure less than 35 cm H 2 O and restricting tidal volumes to 6 mL/kg of ideal body weight.
Permissive hypercapnia may be necessary to implement a lung-protective ventilation strategy. It should be used judiciously in patients with pulmonary hypertension because acidosis can exacerbate pulmonary vasoconstriction and further impair right ventricular function and cardiac output.
Impediments to weaning from mechanical ventilation and extubation include delirium, unstable hemodynamic status, respiratory muscle dysfunction, renal failure with fluid overload, and sepsis.
Short-term weaning success can be achieved with any variety of ventilation modes. The patient receiving long-term ventilatory support requires an individualized approach that may encompass pressure-support ventilation, synchronized intermittent mandatory ventilation weaning, or T-piece trials. Noninvasive ventilation may assist in the transition from full support to liberation from mechanical ventilation.
A few patients are not able to be weaned from ventilation support. Characteristics of these patients include a persistent low-output state with multisystem organ failure. Long-term weaning may be best accomplished in a specialized unit rather than an acute cardiovascular recovery area.
Cardiac anesthesia itself has fundamentally shifted from a high-dose narcotic technique to a more balanced approach using moderate-dose narcotics, shorter-acting muscle relaxants, and volatile anesthetic agents. This change primarily has been driven by a realization that high-dose narcotics delay extubation and recovery after surgical procedures. This new paradigm also has led to renewed interest in perioperative pain management. In addition to changes in anesthetic practice, the type of patients presenting for cardiac operations is changing. Patients are now older and have more associated comorbidities (stroke, myocardial infarction [MI], renal failure). Change also has taken place in the recovery of patients undergoing cardiac procedures. Although cardiac surgical procedures often were associated with a high mortality rate and long intensive care unit (ICU) stays, the use of moderate doses of narcotics has allowed for rapid ventilator weaning and discharge from the ICU within 24 hours. This change has prompted a shift from the classical model of recovering patients in the traditional ICU manner, with weaning protocols and intensive observation, to management more in keeping with the recovery room practice of early extubation and rapid discharge.
Few trials have compared inhalation agents for fast-track cardiac anesthesia (FTCA). Several studies examined the effectiveness of propofol versus an inhalation agent; these studies demonstrated reductions in myocardial enzyme release (creatine kinase myocardium band [CK-MB], troponin I) and preservation of myocardial function in patients receiving inhalation agents.
The choice of muscle relaxant in FTCA is important to reduce the incidence of muscle weakness in the cardiac recovery area (CRA) that may delay tracheal extubation. Several randomized trials compared rocuronium (0.5–1 mg/kg) with pancuronium (0.1 mg/kg) and found significant differences in residual paralysis in the ICU, with delays in the time to extubation in the pancuronium-treated group.
Several trials examined the use of different short-acting narcotic agents during FTCA. In these trials, fentanyl, remifentanil, and sufentanil all were found to be efficacious for early tracheal extubation. The anesthetic drugs and their suggested dosages are listed in Box 29.1 .
Narcotic
Fentanyl 5–10 µg/kg
Sufentanil 1–3 µg/kg
Remifentanil– infusions of 0.5–1.0 µg/kg per min
Muscle Relaxant
Rocuronium 0.5–1 mg/kg
Vecuronium 1–1.5 mg/kg
Hypnotic
Midazolam 0.05–0.1 mg/kg
Propofol 0.5–1.5 mg/kg
Narcotic
Fentanyl 1–5 µg/kg
Sufentanil 1–1.5 µg/kg
Remifentanil infusions of 0.5–1.0 µg/kg per min
Hypnotic
Inhalational 0.5–1 MAC
Propofol 50–100 µg/kg per min
Narcotic
Morphine 0.1–0.2 mg/kg
Hypnotic
Propofol 25–75 µg/kg per min
MAC, Minimum alveolar concentration.
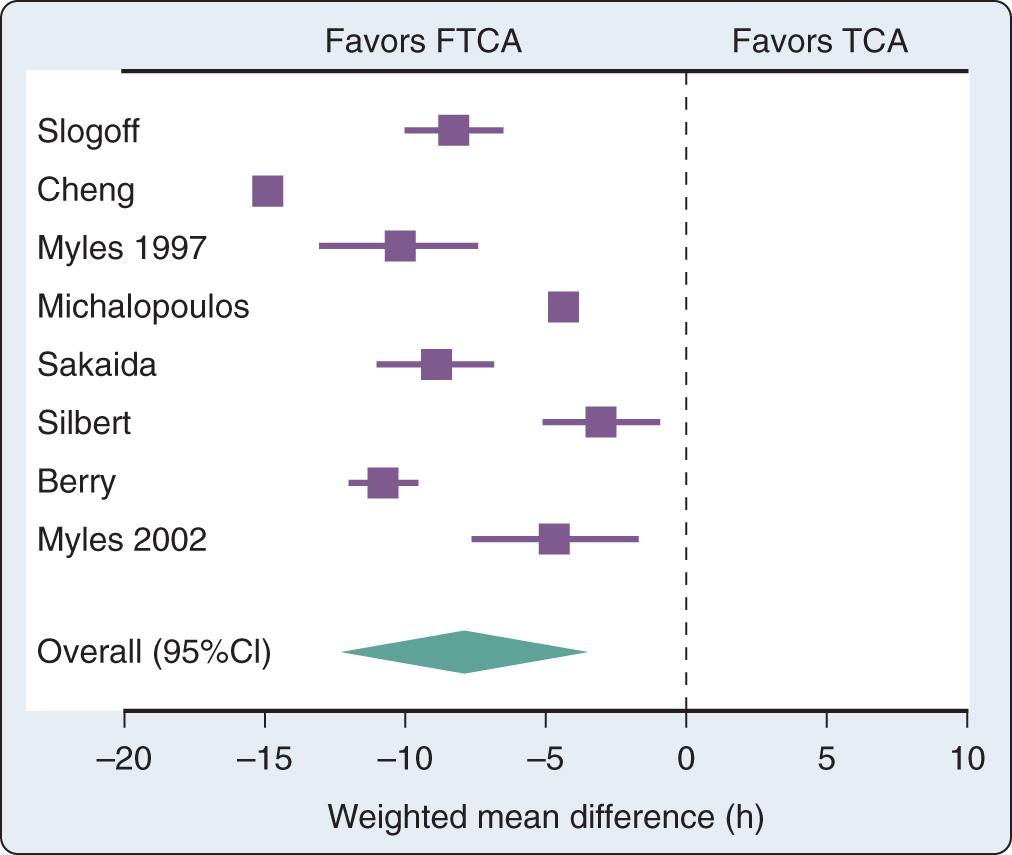
Several randomized trials and one metaanalysis of randomized trials addressed the question of safety of FTCA. None of the trials was able to demonstrate differences in outcomes between the fast-track anesthesia group and the conventional anesthesia group. The metaanalysis of randomized trials demonstrated a reduction in the duration of intubation by 8 hours ( Fig. 29.1 ) and in the ICU length of stay (LOS) by 5 hours in favor of the fast-track group. However, the hospital LOS was not statistically different.
FTCA appears safe in comparison with conventional high-dose narcotic anesthesia. It reduces the duration of ventilation and ICU LOS considerably without increasing the incidence of awareness or other adverse events. FTCA appears effective at reducing costs and resource use. As such, it is becoming the standard of care in many cardiac centers. The usual practice at many institutions is to treat all patients as candidates for FTCA with the goal of allowing early tracheal extubation for every patient. However, if complications occur that would prevent early tracheal extubation, the management strategy is modified accordingly. Investigators have demonstrated that the risk factors for delayed tracheal extubation (>10 hours) are increased age, female sex, postoperative use of intraaortic balloon pump (IABP), inotropes, bleeding, and atrial arrhythmia. The risk factors for prolonged ICU LOS (>48 hours) are those of delayed tracheal extubation in addition to preoperative MI and postoperative renal insufficiency. Care should be taken to avoid excessive bleeding (antifibrinolytic agents) and to treat arrhythmias either prophylactically or when they occur (β-blockers, amiodarone).
On arrival in the CRA, initial management of cardiac patients consists of ensuring an efficient transfer of care from operating room (OR) staff to CRA staff while at the same time maintaining stable patient vital signs. The anesthesiologist should relay important clinical parameters to the CRA team. To accomplish this, many centers have devised handoff sheets to aid in the transfer of care. The patient's temperature should be recorded, and, if low, active rewarming measures should be initiated with the goal of rewarming the patient to 36.5°C. Shivering may be treated with low doses of meperidine (12.5–25 mg, intravenously). Hyperthermia, however, is common later within the first 24 hours after cardiac operations and may be associated with an increase in neurocognitive dysfunction, possibly a result of hyperthermia exacerbating cardiopulmonary bypass (CPB)–induced neurologic injury ( Box 29.2 ).
Normothermia
Hemoglobin >7 g/dL
Pa co 2 35–45 mm Hg
Sa o 2 >95%
Mean blood pressure >50–70 mm Hg
Potassium: 3.5–5.0 mEq/L
Blood glucose <10.0 mmol/L (<200 mg/dL)
Pa co 2 , Partial pressure of arterial carbon dioxide; Sa o 2 , arterial oxygen saturation.
Ventilatory requirements should be managed with the goal of early tracheal extubation in patients ( Box 29.3 ). Arterial blood gases (ABGs) are initially drawn within one-half hour after admission and then repeated as needed. Patients should be awake and cooperative, hemodynamically stable, and have no active bleeding with coagulopathy. Respiratory strength should be assessed by hand grip or head lift to ensure complete reversal of neuromuscular blockade. The patient's temperature should be more than 36°C, preferably normothermic. When these conditions are met and ABG results are within the reference range, tracheal extubation may take place. ABGs should be drawn approximately 30 minutes after tracheal extubation to ensure adequate ventilation with maintenance of partial pressure of arterial oxygen (Pa o 2 ) and partial pressure of arterial carbon dioxide (Pa co 2 ). Inability to extubate patients as a result of respiratory failure, hemodynamic instability, or large amounts of mediastinal drainage necessitates more complex weaning strategies.
A/C at 10–12 beats/min
Tidal volume 8–10 mL/kg
PEEP 5 cm H 2 O
pH 7.35–7.45
Pa co 2 35–45 mm Hg
Pa o 2 >90 mm Hg
Saturations >95%
Arterial blood gases as above
Awake and alert
Hemodynamically stable
No active bleeding (<400 mL/2 h)
Temperature >36°C
Return of muscle strength (>5 s, head lift/strong hand grip)
A/C, Assist-controlled ventilation; Pa co 2 , partial pressure of arterial carbon dioxide; Pa o 2 , partial pressure of arterial oxygen; PEEP, positive end-expiratory pressure.
Some patients may arrive after extubation in the OR. Careful attention should be paid to these patients because they may subsequently develop respiratory failure. The patient's respiratory rate should be monitored every 5 minutes during the first several hours. ABGs should be drawn on admission and 30 minutes later to ensure that the patient is not retaining carbon dioxide. If the patient's respirations become compromised, ventilatory support should be provided. Simple measures such as reminders to breathe may be effective in the narcotized or anesthetized patient. Low doses of naloxone (0.04 mg, intravenously) also may be beneficial. Trials of continuous positive airway pressure (CPAP), bilevel positive airway pressure (BiPAP), or noninvasive ventilation (NIV) may provide enough support to allow adequate ventilation. Reintubation should be avoided because it may delay recovery; however, it may become necessary if the earlier mentioned measures fail, with resulting hypoxemia, hypercarbia, and a declining level of consciousness.
Chest tube drainage should be checked every 15 minutes after ICU admission to assess a patient's coagulation status. Although blood loss is commonly divided into two types, surgical or medical, determining the cause of bleeding is often difficult. When bleeding exceeds 400 mL/hour during the first hour, 200 mL/hour for each of the first 2 hours, or 100 mL/hour over the first 4 hours, returning the patient to the OR for chest reexploration should be considered. The clinical situation must be individualized for each patient, however, and in patients with known coagulopathy, more liberal blood loss before chest reexploration may be acceptable ( Box 29.4 ).
Review activated coagulation time, prothrombin time, international normalized ratio, and platelet count
Administer protamine if bleeding is caused by excess heparin (reinfusion of pump blood)
Treat the medical cause with platelets, fresh-frozen plasma, and cryoprecipitate if bleeding is secondary to decreased fibrinogen
Factor VIIa should be considered if bleeding continues despite a normal coagulation profile
Treat the surgical cause with reexploration
Hypokalemia is common after cardiac surgical procedures, especially if diuretic agents were given intraoperatively. Hypokalemia contributes to increased automaticity and may lead to ventricular arrhythmias, ventricular tachycardia, or ventricular fibrillation. Treatment consists of potassium infusions (20 mEq potassium in 50 mL of D 5 W infused over 1 hour) until the potassium level exceeds 3.5 mEq/mL. In patients with frequent premature ventricular contractions caused by increased automaticity, a serum potassium level of 5.0 mEq/mL may be desirable. Hypomagnesemia contributes to ventricular preexcitation and may contribute to atrial fibrillation (AF). This disorder is common in malnourished and chronically ill patients, a frequent occurrence in the cardiac surgical setting. Management consists of intermittent boluses of magnesium: 1 to 2 g over 15 minutes. Hypocalcemia also is frequent during cardiac operations and may reduce cardiac contractility. Intermittent boluses of calcium chloride or calcium gluconate (1 g) may be required ( Box 29.5 ).
SSx: muscle weakness, ST-segment depression, “u” wave, T-wave flat, ventricular preexcitation
Rx: IV KCl at 10–20 mEq/h by central catheter
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here