Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Keratoprosthesis is a reasonable option in eyes not amenable to conventional keratoplasty.
Advances in the design of the Boston keratoprosthesis type 1, such as the titanium back plate, have improved outcomes.
Changes in the postoperative management of the Boston keratoprosthesis type 1 device have reduced complications.
Postoperative management is critical including the use of antibiotics, steroids, therapeutic bandage contact lens, and intraocular pressure control.
Untreated glaucoma remains the most frequent etiology for irreversible visual loss after keratoprosthesis surgery.
Eyes with severe ocular surface disease have a poorer prognosis
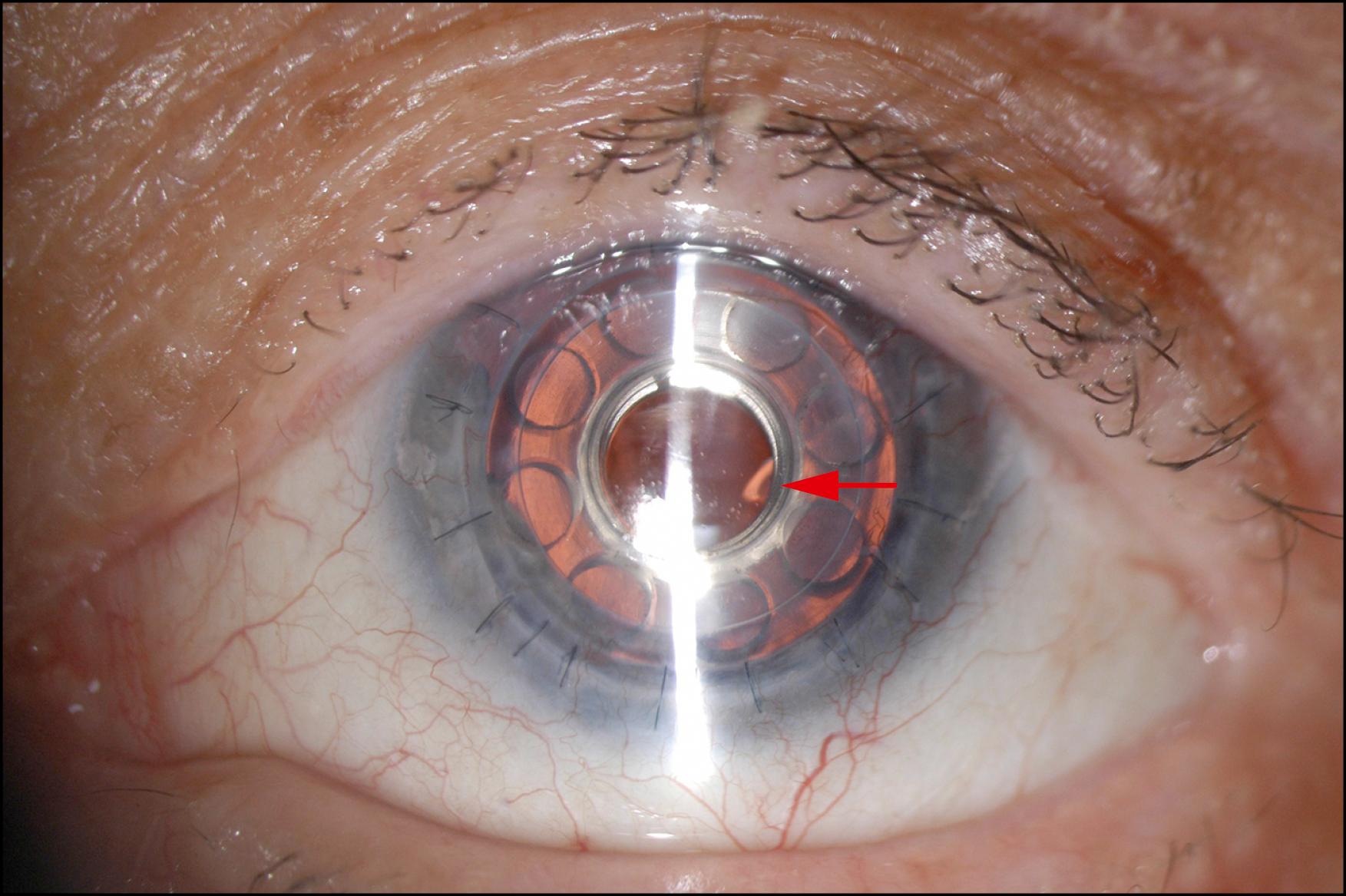
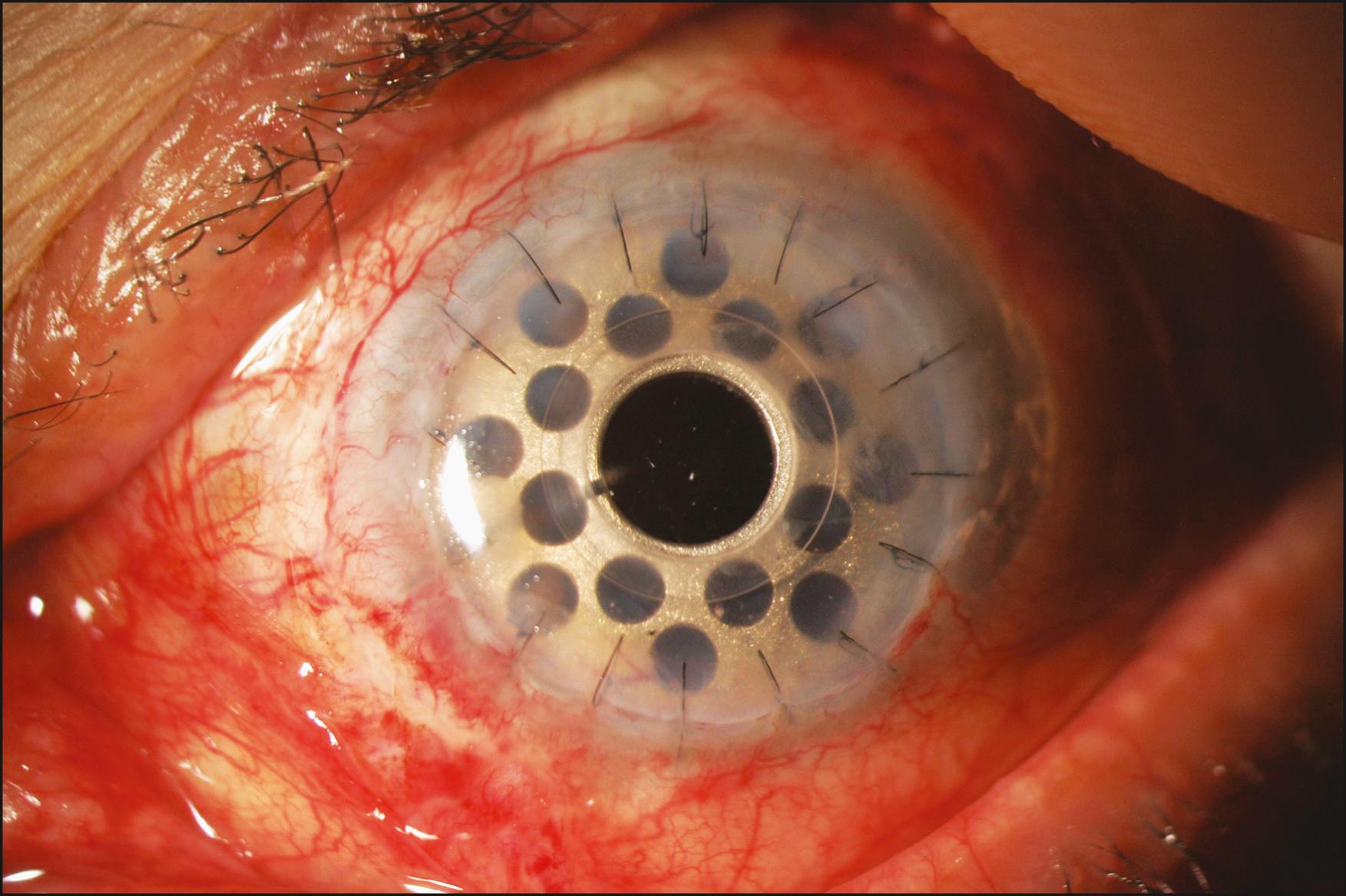
It is not a surprise that keratoprosthesis (KPro) surgery has come of age during the second decade of the 21st century. The advances in the keratoprosthetic devices themselves, surgical techniques, and postoperative management have led to the wide recognition of this procedure as a reasonable surgical alternative to penetrating keratoplasty, if not the procedure of choice, in the setting of corneal opacification not amenable to primary or repeat penetrating keratoplasty. Of the various types of keratoprostheses, including the osteo-odonto-keratoprosthesis (OOKP), the Boston KPro type 1 has emerged as the most commonly used, mainly owing to its ease of insertion, high degree of retention, and frequency of leading to successful visual rehabilitation. With approximately 800 devices implanted worldwide in 2018, and more than 14,000 devices overall since 1992, the Boston KPro type 1 is currently the most commonly implanted corneal prosthetic device in the world. For the indications of repeat graft failure ( Fig. 157.1 ) or noninflammatory limbal stem cell deficiency, the retention rate is approximately 95%, with most of these patients achieving decidedly better vision than preoperatively. Inflammatory categories such as Stevens-Johnson syndrome, mucous membrane pemphigoid ( Fig. 157.2 ), and chemical injuries, as well as conditions resulting in severe ocular surface dryness, are associated with a lower retention rate, not unexpectedly.
The Boston type 1 design modifications of late 2002 include separate front and back polymethylmethacrylate (PMMA) plates that are assembled around the carrier corneal tissue, fenestrations in the back plate to facilitate aqueous nourishment of that tissue, a titanium locking ring to decrease the chance of dislocation of the back plate into the anterior chamber and, most recently, a titanium back plate that may be less prone to inciting anterior segment inflammation ( Fig. 157.3 ). The titanium back plate is now available in either a snap-on version that requires the titanium locking ring or an integrated click-on version that obviates the need for the locking ring. (Note: although the titanium back plate has been considered a positive advance in the design of the Boston KPro, there have been some concerns recently regarding the potentially greater incidence of biofilm development on titanium surfaces compared to PMMA.) With these design modifications came the recommendation of using healthy donor corneal tissue as a carrier in comparison to the historical acceptance of any donor tissue, including tissue that was previously frozen or stored in glycerin. Healthy donor corneal tissue has been associated with better coupling of the donor carrier around the neck of the prosthesis and a decrease in the incidence of corneal melts. Together with the aforementioned design changes in the keratoprosthetic device itself came paradigm shifts in postoperative management, which most KPro surgeons feel have added immensely to its success :
The acceptance of the principle of long-term topical antibiotic coverage: historically, this had been antithetical to the thinking of most external disease specialists because of concern over developing resistance. However, the long-term use of a wide-spectrum antibiotic, such as a newer-generation fluoroquinolone, has led to a decrease in the incidence of intraocular infections. , ,
When intraocular infections did occur in the past they were almost always with gram-positive organisms. As such, the new recommendations include the addition of vancomycin to the fluoroquinolone or the replacement of both with polymyxin B/trimethoprim. In settings where the likelihood of fungal infection is higher, such as those resulting from previous inflammatory conditions and moderate-to-severe dry eye states, consideration should be given to the use of a topical antifungal agent.
Intra- and postoperative use of corticosteroids: consideration may be given to an intravitreal steroid injection at the end of the case followed by a tapering regimen of topical steroids postoperatively.
The maintenance of a bandage contact lens over the KPro indefinitely, whenever possible (see Fig. 157.1 ): this creates a moist environment around the neck of the KPro, and diffuses evaporative forces, leading to a decrease in corneal stromal melts and device extrusion. Moreover, the bandage contact lens decreases mechanical forces between the lid and the anterior plate, which may lead to mobilization of the device.
Aggressive management of glaucoma pre-, intra-, and postoperatively with medication and, if necessary, with the use of a glaucoma tube-shunt drainage device ( Fig. 157.4 ). , , , Measuring the intraocular pressure (IOP) post KPro surgery may be challenging, and periodic optic nerve head and nerve fiber layer imaging and visual fields are necessary.
The above advances and paradigm shift in postoperative treatment, as well as indefinite routine postoperative exams, are felt to be critical to the long-term success of KPro surgery.
KPro surgery patients are evaluated on the first postoperative day, a week later, 2–3 weeks after that, and then on a monthly basis for the first few months before dropping to quarterly follow-up, which may be maintained indefinitely.
Each examination should include measurement of visual acuity as well as an attempt at assessing IOP either with palpation of the globe through the lid or visual inspection of scleral indentation of the globe at the limbus in response to application of digital pressure through the lid or a cotton-tipped applicator at the limbus. Pneumotonometry on the adjacent sclera has a role as well, but applanation and electronic tonometry are not usually useful.
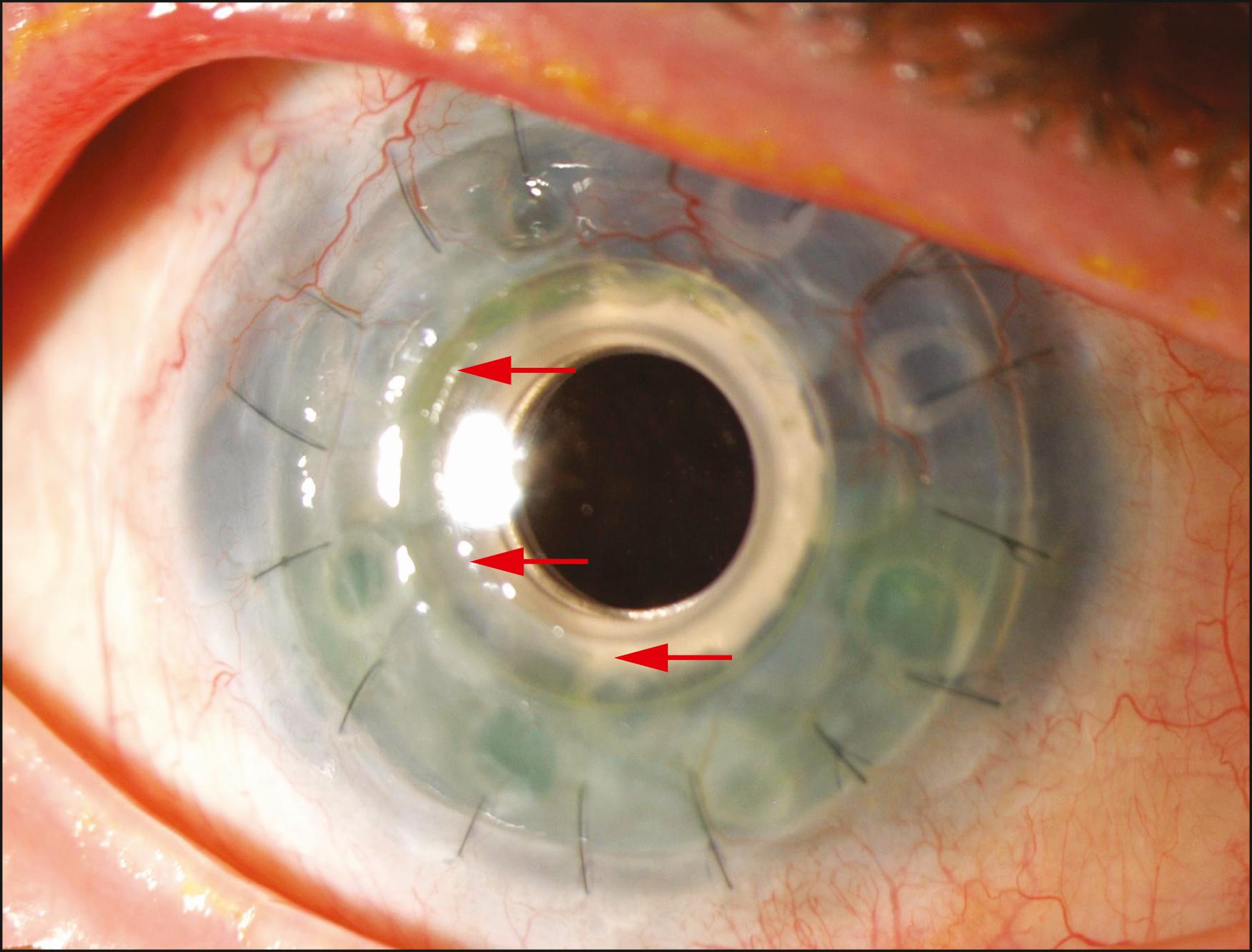
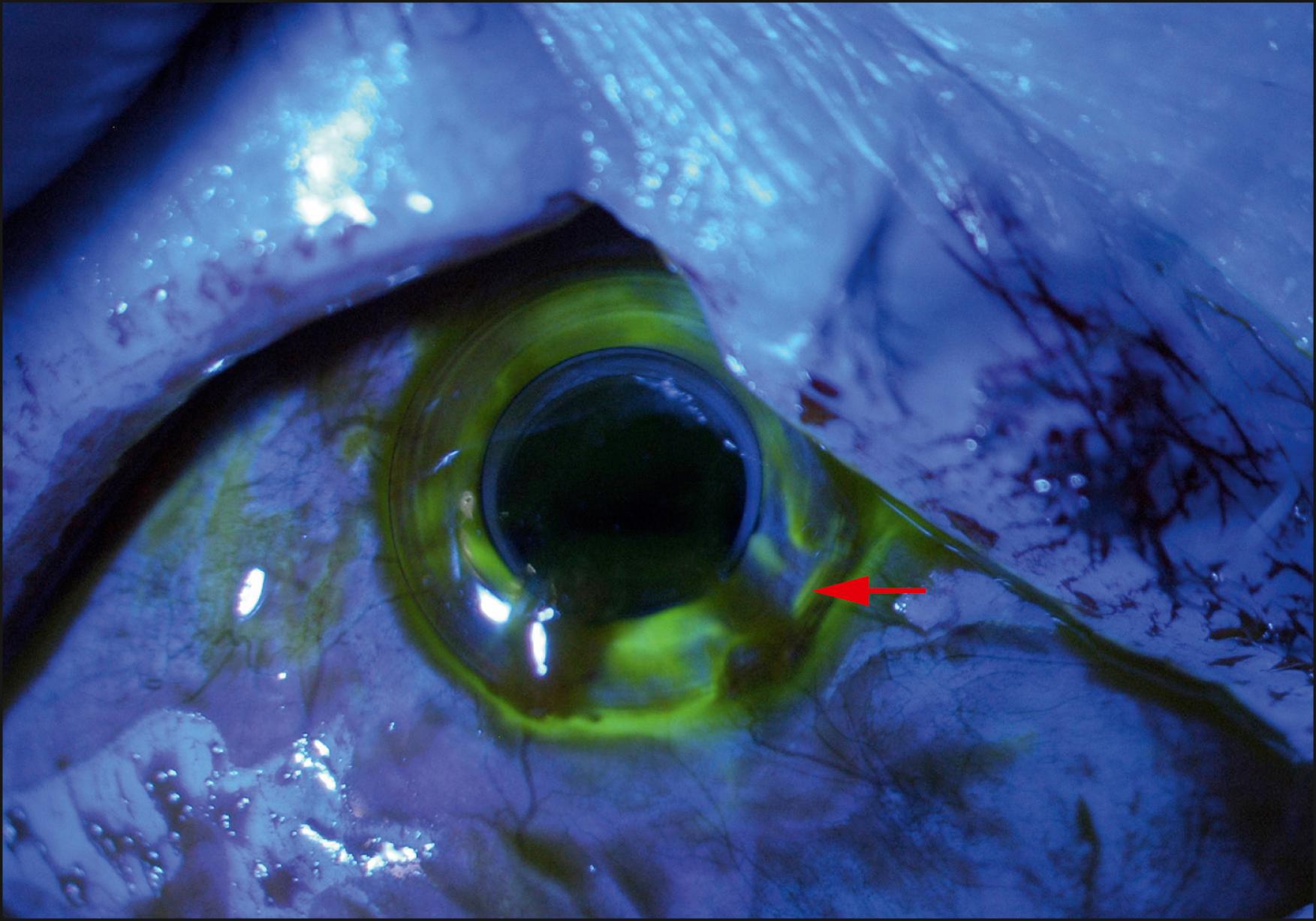
Slit lamp biomicroscopy is employed to evaluate the position of the bandage contact lens, any protein deposits on the contact lens or KPro front plate, the front and back plates of the KPro, and any details of the anterior segment that are visible, including the iris, the intraocular lens implant, and the posterior capsule, when present. Removal of the bandage contact lens may be undertaken to evaluate the corneal surface surrounding the anterior plate. Any epithelial defects should be documented and followed closely. The contact lens should be replaced periodically or when deposits form on the surface, but no less than every 6 months. Some surgeons have observed that if the epithelium grows over the edge of the anterior plate 360 degrees around, therefore sealing off the potential space between the carrier corneal tissue and the neck of the optical element, the bandage contact lens may not be necessary ( Fig. 157.5 ). The examiner may look for leaks using fluorescein ( Fig. 157.6 ). Further along in the postoperative period, early opacification of the fenestrations in the posterior plate may indicate low-to-moderate-grade inflammation that should be approached aggressively with an increase in topical steroids or placement of peribulbar triamcinolone. One can also document the early development of a retroprosthetic membrane (RPM) that is more amenable to the yttrium aluminum garnet (YAG) laser before it becomes thicker, and, on occasion, vascularized ( Fig. 157.7 ).
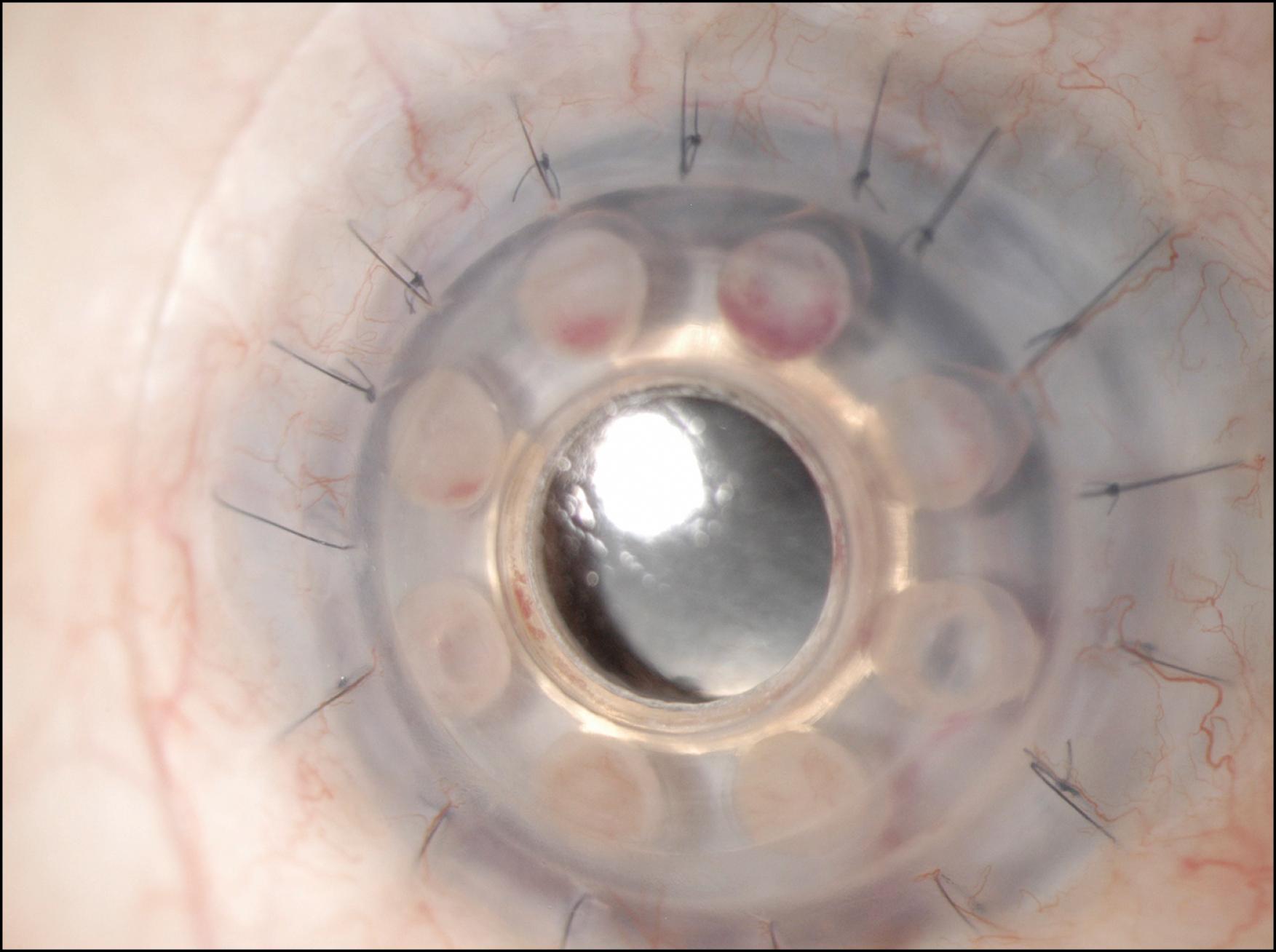
The posterior segment may be evaluated visually with a 90 or 78 diopter lens, which will usually allow a view of the optic nerve and the macula. “Small pupil” diagnostic lenses may allow a wider view of the peripheral retina.
Because follow-up of IOP is not easily achieved after KPro surgery, it behooves the surgeon to continue to monitor the appearance of the optic nerve head, visually or with imaging (e.g., optical coherence tomography [OCT]), as well as to perform periodic threshold perimetry, concentrating on the central field and looking for early evidence of glaucomatous field defects. Interestingly, with the latest Boston KPro type 1 design, most patients are capable of a 60-degree angle of visual field. As undetected glaucoma is one of the major causes of long-term visual loss in these patients, it is reasonable to have all KPro patients followed by a glaucoma specialist in addition to the cornea specialist. ,
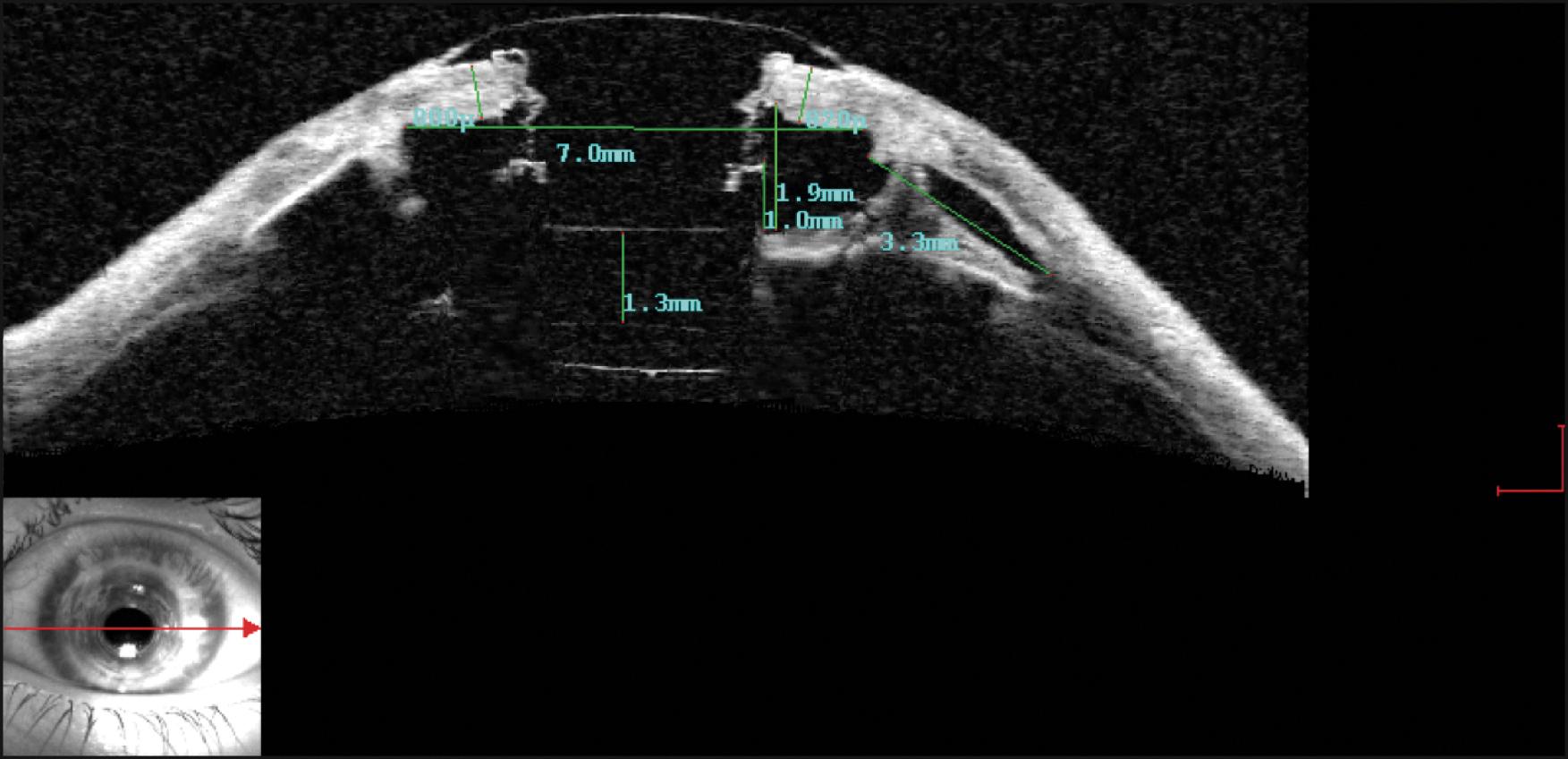
B-scan ultrasonography may be performed whenever peripheral retinal pathology or vitreous hemorrhage is suspected, or there is any evidence of intraocular inflammation that suggests endophthalmitis or sterile vitritis. Finally, subspecialty services may employ ultrasound biomicroscopy (UBM) as well as anterior segment optical coherence tomography (AS-OCT) to look for subtle clues that may explain any difficulties encountered in the postoperative period, including the relationship between the iris and the posterior plate, the positions of the tube shunt and intraocular lens implant, the development of RPMs, and the status of the anterior vitreous ( Fig. 157.8 ). OCT may also be used to identify any gaps between the neck of the optical element and the surrounding carrier corneal tissue, as well as detecting fibrous ingrowth, its source, and pattern of progression.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here