Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Pleomorphic sarcomas are defined as malignant mesenchymal neoplasms characterized at a histologic level by prominent cellular pleomorphism and often brisk mitotic activity, with or without a readily identifiable line of differentiation.
Cellular pleomorphism in mesenchymal neoplasms is probably best defined as a significant variation in the size and shape of individual cells within a lesion, primarily as a result of striking alterations in nuclear and cytoplasmic morphology. It may either be a consistent and defining feature of a particular neoplasm (e.g., pleomorphic fibroma of the skin, atypical fibrous histiocytoma, symplastic [“bizarre”] leiomyoma of the uterus, pleomorphic hyalinizing angiectatic tumor [PHAT], pleomorphic myogenic sarcomas) or fall within the accepted histologic spectrum of certain well-defined soft tissue tumors (e.g., schwannoma with degenerative atypia, pleomorphic, and spindle cell lipoma). It may also involve a lesion diffusely (e.g., pleomorphic fibroma of the skin) or be present only focally (e.g., schwannoma, pleomorphic lipoma). From the previously mentioned examples, and Boxes 7.1 and 7.2 , it becomes apparent that cellular pleomorphism may be present in lesions encompassing a broad spectrum of biologic behavior, ranging from those that are completely harmless to those with a malignant and extremely aggressive clinical course. Pleomorphism is therefore not in itself an indicator of aggressive behavior.
Pleomorphic fibroma of skin
Atypical (pseudosarcomatous) fibrous histiocytoma
Atypical fibroxanthoma
Pleomorphic lipoma
Symplastic (bizarre) leiomyoma of the uterus
Schwannoma with degenerative atypia (“ancient” schwannoma)
Pleomorphic hyalinizing angiectatic tumor (probably benign and characterized by repeated local recurrences but long-term biologic behavior uncertain)
Myxoinflammatory fibroblastic sarcoma (probably of low-grade malignancy with rare distant metastases but long-term biologic behavior is uncertain)
Myxofibrosarcoma, high-grade
Pleomorphic leiomyosarcoma
Pleomorphic rhabdomyosarcoma
Pleomorphic liposarcoma
Dedifferentiated liposarcoma, morphologically high-grade
Extraskeletal osteosarcoma
Malignant mesenchymoma
Undifferentiated pleomorphic sarcoma
Mitotic activity is a separate histologic feature, independent of the presence or absence of cellular pleomorphism. Mitotic activity (as for cellular pleomorphism) is not per se an indicator of aggressive behavior (e.g., brisk mitotic activity in benign lesions such as nodular fasciitis and giant cell tumor of tendon sheath).
However, the combination of cellular pleomorphism and mitotic activity is generally a more ominous finding and is often associated with malignancy in soft tissue neoplasms. Rare exceptions to this rule exist: atypical fibroxanthoma (AFX), a neoplasm with significant cellular pleomorphism and brisk mitotic activity, when strictly defined, is generally accepted as being a benign non-metastasizing lesion.
Benign and malignant mesenchymal neoplasms may exhibit cellular pleomorphism, either diffusely or focally.
Benign and malignant neoplasms may exhibit mitotic activity.
Cellular pleomorphism together with mitotic activity in a mesenchymal neoplasm is highly suggestive of malignancy.
Consequently, when confronted with a pleomorphic “sarcomatoid” neoplasm, the principal issues are the following:
To exclude nonsarcomatous lesions (e.g., pleomorphic/anaplastic carcinoma as typically seen in the thyroid gland, lung, and pancreas; melanoma; large cell lymphoma), which may mimic high-grade sarcomas. This is readily achieved with an appropriate panel of immunohistochemical markers ( Table 7.1 ).
| Pleomorphic Carcinoma | Melanoma | ALCL | DLBCL | |
|---|---|---|---|---|
| Keratins | + | − | − | − |
| S-100 protein | − | + | − | − |
| HMB45 | − | ± | − | − |
| Melan A | − | ± | − | − |
| CD45 | − | − | ± | + |
| CD20/CD79a/PAX5 | − | − | − | + |
| CD2/CD3 | − | − | ± | − |
| CD30 | − | − | + | − |
| ALK | − | − | ± | − |
To avoid classifying a benign lesion as malignant. This requires a familiarity with benign mimics of pleomorphic sarcomas (see Box 7.1 ).
To maximize efforts in subclassifying the lesion, including the use of ancillary techniques such as immunohistochemistry, various (cyto)genetic modalities (including fluorescence in situ hybridization), and (rarely) electron microscopy. This is important for the selection of appropriate treatment strategies and inclusion in relevant and meaningful clinical trials.
To minimize classification as an undifferentiated pleomorphic sarcoma. This is a “wastebasket” label for a heterogeneous group of tumors that remain unclassifiable despite the use of currently available ancillary diagnostic techniques.
Accurate classification of pleomorphic soft tissue tumors is therefore best regarded as a “gestalt” based on a summation of the clinical setting, histologic features (including results of ancillary techniques), and clinicopathologic experience.
Pleomorphic/anaplastic carcinoma, melanoma, and anaplastic large cell lymphoma are notorious mimics of pleomorphic sarcoma.
Benign mesenchymal lesions may mimic pleomorphic sarcoma.
A host of benign mesenchymal lesions may exhibit pleomorphic features (see Box 7.1 and discussion in the relevant chapters elsewhere in this book). This chapter primarily addresses malignant mesenchymal lesions with a pleomorphic phenotype, ranging from pleomorphic sarcomas readily classifiable on the basis of recognizable histologic features (with or without the aid of ancillary techniques) to those lesions that remain unclassifiable with currently available techniques, previously categorized under the nosologically meaningless rubric malignant fibrous histiocytoma (MFH). Now, in keeping with the most recent World Health Organization (WHO) consensus classification, these latter lesions are designated as undifferentiated (high-grade) pleomorphic sarcoma (see Box 7.2 ).
When strictly defined, AFX, also discussed in Chapter 15 , is no longer regarded as a malignant neoplasm, but it is nevertheless briefly covered in this section because it is histologically and immunohistochemically indistinguishable from undifferentiated pleomorphic sarcoma. AFX is an intradermal tumor, probably of fibroblastic origin, arising almost exclusively in actinically damaged skin of the head and neck area of older adults, with pleomorphic and/or spindle-cell features, usually with an expansile growth pattern, and without invasion into subcutaneous tissue, vascular invasion, or necrosis. Cases of so-called AFX described at sites without actinic damage and in younger patients are possibly examples of atypical (pseudosarcomatous) fibrous histiocytoma, although convincing clinicopathologic studies supporting this hypothesis are as yet not available.
AFX presents as a skin-colored nodular lesion in the head and neck area of older patients. The nodule is usually less than 2 cm in diameter, and frequently there is a history of rapid growth. Ulceration may be present, but is, in our experience, relatively uncommon.
Histologically, all cases of AFX are centered in the dermis, generally abutting the epidermis, although occasionally a grenz zone may be present. The lesion is usually well circumscribed, often polypoid, and the overlying epidermis may form a collarette around the tumor ( Fig. 7.1A and B ). AFX is a cellular neoplasm composed of a haphazard and/or fascicular arrangement of highly pleomorphic and spindle-shaped cells. Multinucleated giant cells with monomorphic or pleomorphic nuclei are frequently present. The cytoplasm is as a rule easily discernible, eosinophilic, or amphophilic, and may occasionally contain lipid droplets; variable amounts of clear cell change and granular cell change may rarely be present. Mitotic activity, including both typical and atypical mitotic figures, is generally marked (see Fig. 7.1C ). A spindle-cell (nonpleomorphic) variant of AFX characterized by a fascicular proliferation of spindle-shaped cells with pale eosinophilic cytoplasm and without an overtly pleomorphic component is recognized (see Fig. 7.1D ). AFX has an expansile growth pattern, frequently with a “pushing” border, and may be surrounded by a mononuclear inflammatory infiltrate. The overlying epidermis is normal, but when ulceration is present, the lesion often cannot be evaluated for the presence of epidermal dysplasia or junctional activity, important in excluding the differential diagnostic options of sarcomatoid (spindle-cell) carcinoma and melanoma. Invasion into subcutaneous fat (even minimal, or deeper) or the presence of necrosis, lymphovascular, or perineural invasion are not features of AFX and exclude AFX as a diagnostic consideration.
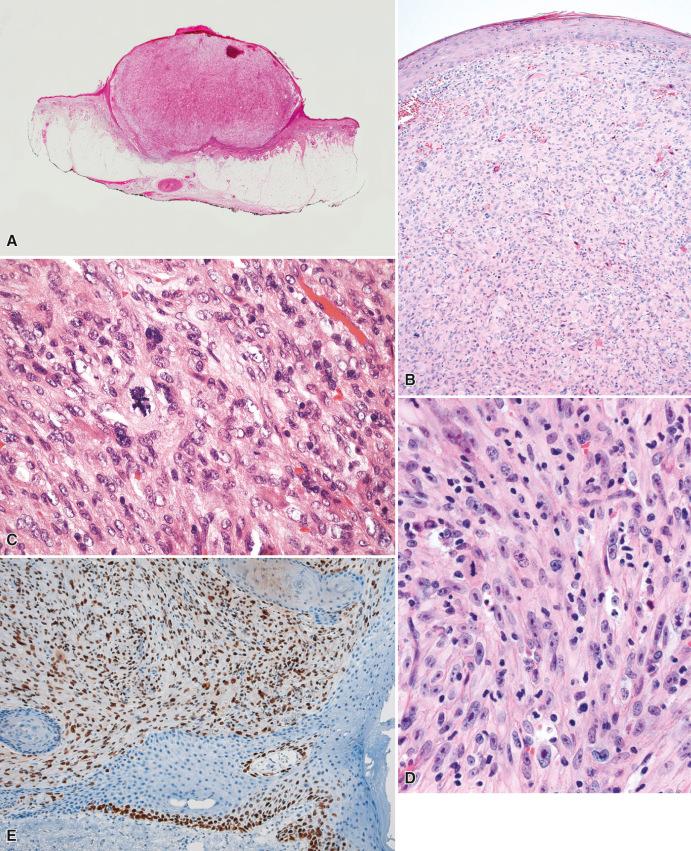
In addition to nonspecific staining for vimentin, the lesions are frequently positive for CD10 and characteristically show strong, diffuse staining for p53 (see Fig. 7.1E ), although, in our experience, a very small number of otherwise typical examples of AFX (<5%) do not stain for p53. Weak positivity for smooth muscle actin may focally be present. Most importantly, S-100 protein and keratins are negative.
Ultraviolet light–induced TP53 mutations, and, more recently, ultraviolet light–induced TERT promoter mutations, all supporting a sun exposure–induced etiology, have been identified in AFX.
The differential diagnosis includes primarily spindle-cell (sarcomatoid) carcinoma, melanoma, atypical intradermal smooth muscle tumor, and cutaneous involvement by (metastatic) leiomyosarcoma, which may all exhibit a pleomorphic and spindle-cell appearance. Diligent clinical correlation (to exclude metastatic disease from a known or unknown primary lesion elsewhere), together with an appropriate immunohistochemical panel, including several keratins, melanoma markers, smooth muscle actin, and desmin, is generally sufficient to reach the correct diagnosis. Lesions otherwise typical of AFX but characterized by infiltration into subcutaneous fat, necrosis, lymphovascular, or perineural invasion, all predictive of aggressive behavior including (distant) metastases, are best classified as pleomorphic dermal sarcoma, not otherwise specified. Therefore the diagnosis of AFX cannot be rendered based on superficial (shave) biopsies, where the base of the lesion cannot be visualized. In such samples, the interim designation atypical intradermal spindle-cell/pleomorphic neoplasm , with a comment recommending complete excision prior to definite diagnosis, is appropriate.
Surgical excision with free margins is curative. Although there is some debate as to whether AFX is benign, if the previously mentioned criteria are strictly applied, AFX behaves in a benign manner with a risk of local recurrence if incompletely excised. There are rare reports of AFX with metastases, but such cases are often incompletely illustrated for optimal evaluation of the minimal diagnostic criteria for AFX (discussed earlier), or stem from the pre- and early immunohistochemistry era (without or with inadequate immunohistochemical workup). Consequently, these lesions may not represent typical AFX and possibly include malignant nonmesenchymal tumors such as poorly differentiated and sarcomatoid carcinoma and amelanotic melanoma. A very recent and more convincing study of putative AFX with (distant) metastases has been published; nevertheless, the conclusions need to be interpreted with caution, and further studies are required to support the validity of these findings.
Atypical fibroxanthoma (AFX) is a tumor of older adults, arising in actinically damaged skin, almost exclusively in the head and neck area.
AFX is usually a pleomorphic tumor, although a spindle cell variant is also recognized.
AFX does not infiltrate the subcutaneous fat and does not exhibit necrosis, vascular invasion, or perineural invasion.
AFX is a diagnosis of exclusion; sarcomatoid carcinoma, melanoma, and leiomyosarcoma must be excluded.
AFX is regarded as benign and does not metastasize.
Undifferentiated pleomorphic sarcoma, previously termed MFH , is defined as a high-grade pleomorphic neoplasm with no identifiable line(s) of differentiation using currently available diagnostic techniques. In the 1960s, Stout and colleagues first introduced MFH as a diagnostic label for a group of malignant mesenchymal tumors composed primarily of pleomorphic and spindle-shaped cells with a predominantly storiform growth pattern, which did not appear to fit into any of the recognized sarcoma categories. The genesis of this term was based partly on (1) the fibroblast-like appearance of the neoplastic cells at a light microscopic level, and (2) the observation that tumor cells in tissue culture appeared to acquire ameboid and phagocytic properties—hence they presumed the cells to be of “fibrohistiocytic” origin.
Following these original studies, MFH soon gained widespread acceptance as a clinicopathologic entity, and by the end of the 1980s, storiform/pleomorphic MFH was the single largest category of sarcomas. During this period, four additional variants were added to the MFH family: giant cell MFH, inflammatory MFH, myxoid MFH, and angiomatoid MFH. Nevertheless, with the passage of time, skepticism concerning the validity of this diagnostic concept grew, and, facilitated by the advent of electron microscopy and the development and refinement of immunohistochemical techniques, it became increasingly clear that there was no real scientific evidence for a “fibrohistiocytic” line of differentiation in this group of tumors. In fact, in a seminal paper in 1992, Fletcher convincingly demonstrated that so-called storiform/pleomorphic MFH (the “flagship” MFH category) represented a heterogeneous group of tumors including not only a variety of different sarcoma types but also melanomas, carcinomas, and lymphomas, unified by a common histologic appearance ( Fig. 7.2 ). In this retrospective re-analysis of 159 tumors diagnosed as MFH, 97 cases (61%) turned out to be specific sarcomas with an identifiable line of differentiation, 20 (13%) were nonmesenchymal neoplasms (pleomorphic carcinoma, melanoma, and lymphoma), and only 42 (26%) showed no identifiable line(s) of differentiation. Of these remaining cases, 21 (13%) were of a sufficient quality to be evaluable and potentially eligible for a diagnosis of storiform/pleomorphic MFH , although importantly, these lesions did not share any consistent features, negating the possibility that they might form part of a homogeneous tumor category. Clearly the terms fibrohistiocytic and fibrous histiocytoma are misnomers for this group of lesions, uniting a variety of different tumor types that are probably unrelated. The category storiform/pleomorphic MFH therefore seems to have served primarily as a “wastebasket” for a heterogeneous group of unclassifiable neoplasms with pleomorphic morphology. A proportion of these tumors will in all likelihood prove to be of (myo)fibroblastic lineage, but, as of yet, due to the lack of reliable and reproducible diagnostic criteria by which such a line of differentiation can be consistently recognized, accurate diagnosis of high-grade myofibroblastic sarcomas remains problematic. Diagnostic criteria for malignant “true” histiocytic neoplasms have gradually been defined in the past decade, facilitating accurate recognition of this group of tumors.
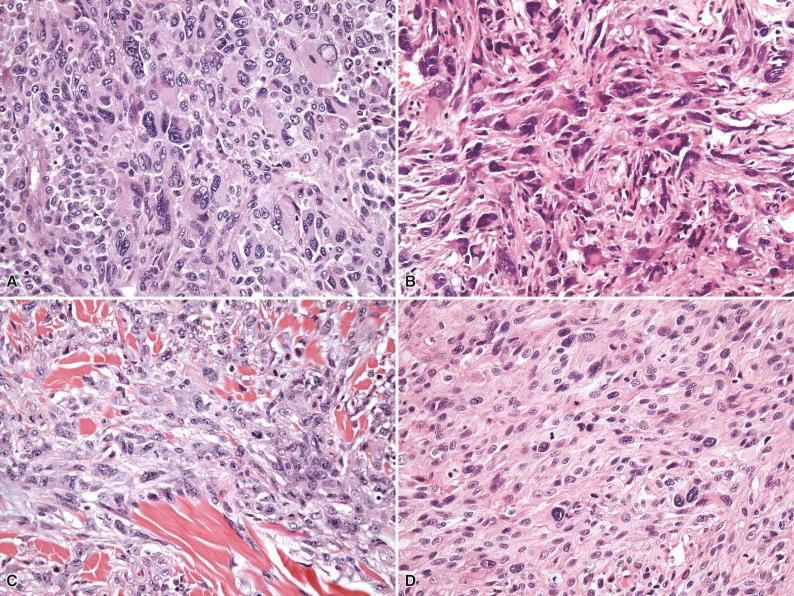
The remaining four MFH variants have similarly been critically re-evaluated. Giant cell MFH, once nonmesenchymal tumors such as osteoclast-rich (metastatic) carcinoma have been excluded, appears to represent a morphologic pattern shared primarily by (malignant) giant cell tumor of soft tissue, extraskeletal osteosarcoma, and leiomyosarcoma with osteoclastic giant cells (see Chapter 11 ). Inflammatory MFH remains a poorly defined category and, once pleomorphic lymphoreticular neoplasms and inflammatory sarcomatoid carcinomas have been excluded, probably consists predominantly of dedifferentiated liposarcoma (DDLPS) with inflammatory features and inflammatory myofibroblastic tumor (see Chapter 10 ). Myxoid MFH, a distinct entity discussed separately later, is synonymous with myxofibrosarcoma, a term that is a more accurate representation of its fibroblastic lineage and myxoid properties. Angiomatoid MFH shares no features with the other members of the MFH family and has been renamed angiomatoid fibrous histiocytoma as a result of its intermediate biologic potential (rarely metastasizing), falling within the category of tumors of “uncertain differentiation” (see Chapters 3 and 10 ). Consequently the diagnostic label “undifferentiated pleomorphic sarcoma” has been gradually and informally introduced to replace “MFH” and should be regarded as a diagnosis of exclusion to be used only when all efforts to identify a specific line of differentiation have failed.
This shift in thinking is reflected in the recent WHO classifications (2002, 2013), and general consensus at last appears to have accepted that the rubric “MFH” is scientifically inaccurate, no longer of diagnostic value, and represents a collection of heterogeneous tumors with variable morphology (pleomorphic, spindle cell, epithelioid, round cell) that are not readily classifiable by current methods. As such, the WHO has chosen to replace this term (“MFH”) with the neutral and more accurate label of undifferentiated (high-grade) sarcoma , and in the most recent WHO classification (2013), this group has been further categorized to include the subcategories undifferentiated pleomorphic sarcoma, undifferentiated spindle cell sarcoma, undifferentiated round cell sarcoma, undifferentiated epithelioid sarcoma, and undifferentiated sarcoma not otherwise specified ( Table 7.2 ).
| Old Terminology | New Terminology |
|---|---|
| Storiform/pleomorphic MFH Giant cell MFH Inflammatory MFH |
Undifferentiated sarcoma with pleomorphic, spindle cell, round cell and epithelioid subtypes Undifferentiated pleomorphic sarcoma with giant cells Undifferentiated pleomorphic sarcoma with prominent inflammation |
| Myxoid MFH | Myxofibrosarcoma |
| Angiomatoid MFH | Angiomatoid fibrous histiocytoma |
Undifferentiated sarcomas as a whole probably account for 5% to 10% of sarcomas in adults older than 40 years of age. These tumors generally arise in the deep (subfascial) soft tissues of older patients (in the sixth decade and older). They have a predilection for the extremities (lower limbs), followed by the trunk, and a small proportion (no more than 10%) arise in the subcutaneous tissues and retroperitoneum.
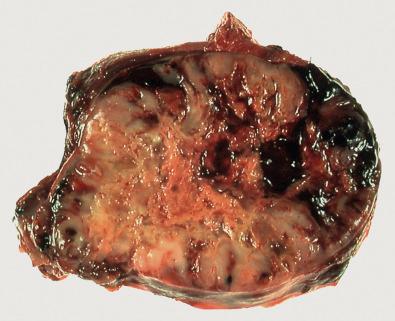
Grossly, the tumors vary in size from 5 to 15 cm, are usually firm with a grayish color, are usually well circumscribed (with or without a pseudocapsule derived from adjacent compressed tissue) but may show infiltrative margins, and frequently contain visible areas of necrosis and/or hemorrhage ( Fig. 7.3 ). Histologically, tumors are composed of a haphazard, storiform, fascicular, or nested arrangement of predominantly or variably highly pleomorphic, spindle-shaped, epithelioid or round cells, with a variable amount of eosinophilic or amphophilic cytoplasm and numerous typical and atypical mitoses. Stroma is usually collagenous with or without an inflammatory infiltrate and occasionally (osteoclastic) giant cells may be prominent ( Fig. 7.4 ). Ancillary ultrastructural studies are, by definition, not helpful in identifying a particular line of differentiation.
Usually only the nonspecific marker vimentin is convincingly positive. The degree of immunohistochemical positivity required to define a particular line of differentiation remains a controversial issue in soft tissue pathology, although generally one requires more than just rare positive cells for an immunohistochemical marker to support specific differentiation ( Table 7.3 ).
| KRT | EMA | S-100 | HMB45 | CD30 | ALK | SMA | DES | h-CD | MYOG | |
|---|---|---|---|---|---|---|---|---|---|---|
| Carcinoma | + | ± | − | − | − | − | − | − | − | − |
| Melanoma | − | − | + | ± | − | − | − | − | − | − |
| ALCL | − | − | − | − | + | ± | − | − | − | − |
| PLMS | ± | ± | − | − | − | − | + | + | + | − |
| PRMS | − | − | − | − | − | − | ± | + | − | + |
| PLPS | − | − | ± | − | − | − | ± | − | − | − |
| DDLPS | − | − | − | − | − | − | ± | ± | − | − |
| MPNST-R | − | ± | ± | − | − | − | ± | + | − | + |
| MFS | − | − | − | − | − | − | ± | − | − | − |
| IMT | − | − | − | − | − | ± | ± | ± | − | − |
Cytogenetically, karyotypes are usually highly complex and nonspecific ( Table 7.4 ).
| Type of Sarcoma | Cytogenetic Abnormality |
|---|---|
| MFS | Nonspecific complex karyotypes |
| PLMS | Nonspecific complex karyotypes |
| PRMS | Nonspecific complex karyotypes |
| PLPS | Nonspecific complex karyotypes |
| DDLPS | Ring and giant marker chromosomes derived from amplification of 12q13–15 (variable amplification of MDM2 , SAS , CDK4 , HMGA2 ) |
| Additional nonspecific complex abnormalities | |
| EO | Nonspecific complex karyotypes |
| PHAT/MIFS/HFLT | t(1;10)(p22;q24) (maps to TGFBR3 and MGEA5 ) and loss of chromosomes 3 and 13 |
| Supernumerary ring chromosomes derived from segments of chromosome 3 and a derivative chromosome 13 | |
| der(10)t(1;10) in combination with aberrations of chromosome 3 | |
| t(2;6)(q31;p21.3) | |
| UPS | Nonspecific complex karyotypes |
| Extensive intratumoral heterogeneity | |
| Aneuploidy (haploid, triploid, tetraploid) | |
| Genomic imbalances, mutations in TP53 , RB1 , CDKN2A |
The differential diagnosis includes (metastatic) sarcomatoid carcinoma, (metastatic) melanoma, anaplastic large-cell lymphoma, and other high-grade pleomorphic sarcomas (high-grade myxofibrosarcoma, pleomorphic leiomyosarcoma, pleomorphic rhabdomyosarcoma, pleomorphic liposarcoma, DDLPS, and malignant peripheral nerve sheath tumor [MPNST] without or with heterologous rhabdomyoblastic differentiation [malignant triton tumor]). The clinical context and an appropriate immunohistochemical panel usually resolve any diagnostic dilemmas in the distinction from carcinoma, melanoma, and lymphoma (see Table 7.1 ). Generous sampling and the judicious use of immunohistochemistry (see Table 7.3 ) should facilitate the identification of foci with myogenic or neurogenic features (leiomyosarcoma/rhabdomyosarcoma or MPNST), lipoblasts (pleomorphic liposarcoma), contiguous foci of atypical lipomatous tumor (ALT)/well-differentiated liposarcoma (WDLPS), or areas with myxoid stroma and curvilinear vessels (myxofibrosarcoma). Because undifferentiated (high-grade) sarcoma remains a diagnosis of exclusion (as detailed previously), extensive sampling is mandatory in order to maximize the chance of identifying a (histologically, immunohistochemically, or ultrastructurally) recognizable line of differentiation, facilitating accurate and clinically useful classification.
Wide surgical excision with free margins and adjuvant radiotherapy is the primary therapeutic modality of choice. Prognostically, tumors are usually high grade, have distant metastases at presentation in 5% to 10% of cases, and have a 5-year survival of 50% to 60%. Tumor size, tumor depth, grade, the presence of necrosis, and local recurrence appear to correlate with metastatic rate and survival, although these do not per se appear to be independent variables for the prediction of outcomes.
The importance of maximizing efforts in accurate classification becomes apparent when one realizes that DDLPS has a 5-year metastatic rate of only 15% to 20%, compared with a 5-year metastatic rate of 30% to 35% for high-grade myxofibrosarcoma, and a far higher 5-year metastatic rate (>60%), together with a significantly inferior relapse-free survival rate, for pleomorphic leiomyosarcoma and rhabdomyosarcoma.
MFH is a meaningless rubric, does not exhibit convincing “fibrohistiocytic” differentiation, represents a “wastebasket” for a heterogeneous collection of unclassifiable neoplasms with pleomorphic, spindle cell, epithelioid or round cell morphology, and has been designated undifferentiated sarcoma in the current World Health Organization classification.
When confronted with a morphologically unclassifiable neoplasm, all efforts need to be made to exclude poorly differentiated carcinoma, melanoma, and lymphoma.
The realization that sarcomas exhibit a wide range of biologic behavior, varying from indolent to highly aggressive, underscores the need for maximal effort in the accurate classification of sarcomas.
Undifferentiated sarcoma is a diagnosis of exclusion and should be used only when all efforts to identify a specific line of differentiation have failed.
Accurate classification of sarcomas allows for a better definition of their clinical behavior and facilitates participation in meaningful clinical trials.
PHAT (of soft parts) is a recently described and enigmatic pleomorphic mesenchymal neoplasm. Although (distant) metastases and tumor-related mortality have not as yet been described, PHAT is reviewed in this section because it may represent an indolent low-grade sarcoma with disease-related morbidity and mortality after longer-term follow-up.
PHAT is currently defined as an (as yet) nonmetastasizing locally aggressive tumor of uncertain lineage characterized by mitotically inert spindle-shaped and pleomorphic cells; ectatic thin-walled vessels exhibiting fibrinoid change; and a variable inflammatory infiltrate composed predominantly of eosinophils, lymphocytes, and plasma cells.
Recognition of putative morphologic and (cyto)genetic overlap with hemosiderotic fibrolipomatous tumor (HFLT) and myxoinflammatory fibroblastic sarcoma (MIFS) would appear to support a morphologic continuum within a single entity (with associated implications for management and follow-up), although the exact nature of this relationship remains an active area of debate.
PHAT is a rare tumor occurring in adults (age range 10 to 80 years; median age 50 years) with an equal gender distribution. It arises primarily in the subcutaneous tissues of the lower limbs and less frequently at other sites. Patients usually present with a slowly enlarging painless swelling, which not infrequently has been present for a long period of time before medical attention is sought.
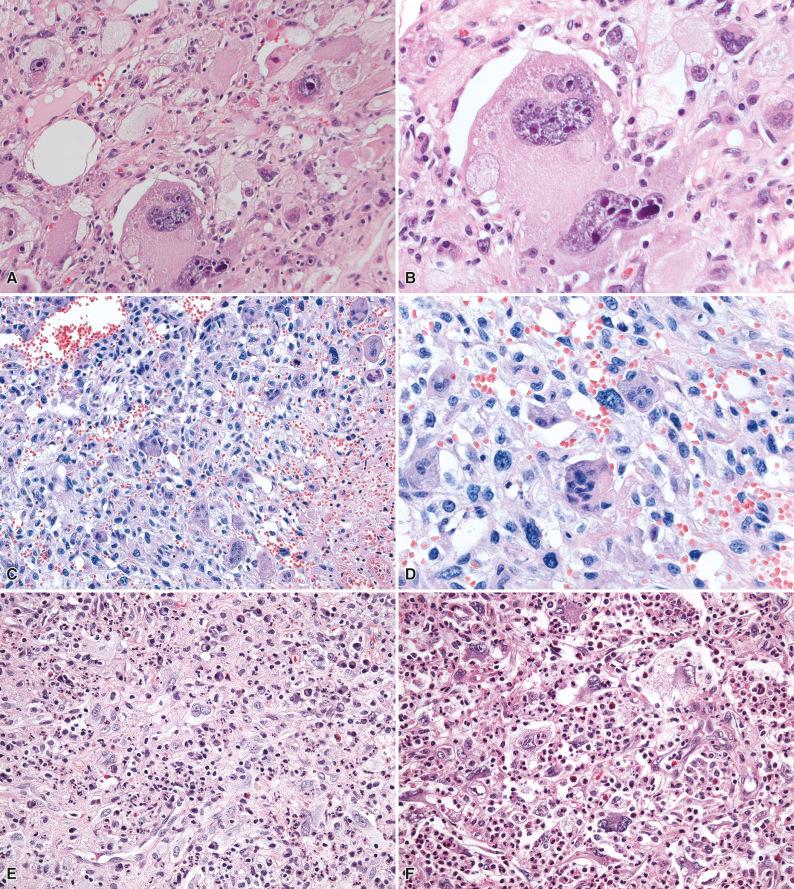
Grossly, tumors are usually poorly circumscribed with infiltrative margins and vary in size from less than 1 cm to more than 10 cm in diameter, with an average size of 3 to 4 cm. On sectioning, tumors are grayish-brown to tan in color, depending on the extent of intralesional vascularity and hemosiderin deposition. Histologically, clusters of thin-walled ectatic vessels, which may be either hyalinized or show extensive fibrinoid change involving the full thickness of the vessel wall, frequently with intraluminal thrombosis, are prominent ( Fig. 7.5A and B ). The stromal cells vary from being spindle-shaped to highly pleomorphic with bizarre, hyperchromatic nuclei often containing intranuclear pseudoinclusions (derived from the cytoplasm; see Fig. 7.5C and D ). Mitoses are scarce, usually less than 1 per 50 high-power fields, and any increase in mitotic activity is generally regarded as not being compatible with a diagnosis of PHAT. A variable inflammatory infiltrate composed of eosinophils, lymphocytes, and plasma cells is usually present, and deposits of hemosiderin may occasionally be prominent. Importantly, in a number of cases of putative PHAT, primarily in peripheral regions of the tumor, variably cellular areas composed of hemosiderin-laden spindle-shaped cells with wavy nuclei and a fascicular orientation morphologically similar to HFLT are present, infiltrating adjacent fat, while cases of putative PHAT with morphologic overlap with MIFS have also been reported, supporting grounds for a morphologic continuum within these three entities.
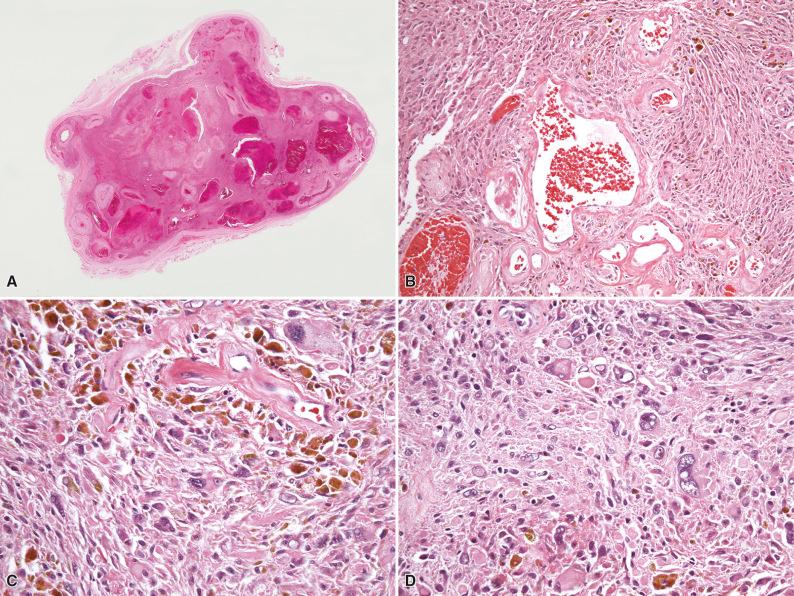
Ancillary ultrastructural studies to date have not been of help in identifying a line of differentiation.
The neoplastic cells do not demonstrate a specific immunohistochemical profile. Lesional cells are positive for CD34 in roughly half of cases, but they are negative for S-100 protein, important in the distinction from “ancient” schwannoma.
At a cytogenetic level, a variety of molecular alterations have been identified in PHAT, some of which (primarily a t[1;10]) appear to be shared (to a limited extent) with MIFS and HFLT, and consequently additional data are required to define the exact nature of the relationship between these three entities.
The differential diagnosis primarily includes ancient schwannoma and a hemangioma with degenerative changes (symplastic hemangioma). The immunohistochemical negativity for S-100 protein excludes schwannoma, whereas the presence of a vascular component intimately associated with obviously atypical stromal cells argues against a vascular lesion.
As a result of the increasing recognition and awareness of overlapping morphologic and cytogenetic features between PHAT, HFLT, and MIFS, although still preliminary, experts are beginning to believe that PHAT, HFLT, and MIFS may fall within one morphologic spectrum.
Wide surgical excision with free margins is the primary therapeutic modality of choice. Typical cases of PHAT are characterized primarily by nondestructive local recurrences, although longer-term follow-up data are needed to determine more accurately the exact biologic behavior of this entity; if indeed there is a biologic continuum with MIFS (which may show high-grade progression, as discussed later) and HFLT, this will have implications for treatment and follow-up guidelines.
PHAT may be mistaken for ancient schwannoma or hemangioma with degenerative changes (symplastic hemangioma).
Many experts now believe that, based on unifying morphology and cytogenetics, PHAT, HFLT, and MIFS may fall within one morphologic spectrum.
MIFS, also discussed in some detail in Chapters 5 and 10 , has previously also been referred to as acral MIFS, inflammatory myxohyaline tumor of the distal extremities with virocyte or Reed-Sternberg–like cells, and inflammatory myxoid tumor of the soft parts with bizarre giant cells. The tumor is defined as a low-grade sarcoma occurring primarily in the distal extremities. It is characterized by myxoid stroma, an inflammatory infiltrate, and distinctive virocyte- or Reed-Sternberg–like cells. Recognition of putative histologic overlap with HFLT and PHAT, the identification of hybrid lesions, and the discovery of unifying cytogenetic data support a morphologic continuum within a single entity.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here