Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Examination of the placenta is a critical component in evaluating the etiology of intrauterine demise. This is particularly true as pregnancy progresses, because the cause of demise beyond the first trimester is less likely to be due to intrinsic fetal disease and more likely due to “errors of the intrauterine environment”; that is, as a result of causes extrinsic to the fetus. As a general principle, the longer intrauterine stressors that ultimately result in demise are present, the more likely that placental “footprints” of those stressors will (1) accrue and (2) be detectable by gross or microscopic examination. This chapter lists segregated specific mechanisms of demise based on the timing of evolution from onset to demise. These categories are somewhat fluid, in that some forms of acute demise may spill into an early subacute time frame and vice versa, just as some typically subacute mechanisms of demise may evolve for weeks preceding death.
Many conditions are covered in additional detail in Chapter 31 with respect to placental evaluation and in Chapter 33 with respect to clinical gestation-specific disorders.
Of great clinical relevance are the following concepts, the literature for which remains in evolution. These concepts pertain largely to subacute and chronic intrauterine pathologies, although the following superimposed acute manifestations of preexisting disorders can add to the overall burden of fetal stress and concomitant risk for untoward fetal/neonatal outcome.
As a general rule, the more advanced or extensive a pathologic process, the greater the potential risk for untoward fetal/neonatal outcome.
Threshold criteria are accruing for identifying the severity/extent of specific placental lesions that herald an increased risk of untoward outcome.
The coexistence of separate pathologic processes, each of which can have fetal/neonatal implications, compounds their cumulative effects in at least an additive, if not multiplicative manner.
The same processes that lead to unexpected fetal demise can also result in neonatal morbidity and mortality.
The hallmark disorders that fall into this conceptual framework include the following diagnostic categories. Diagnostic criteria for several histologic findings within the categorical diagnoses have recently been published in a consensus manuscript ( Box 32.1 ):
Fetal vascular malperfusion
Amniotic fluid infection
Chronic villitis without evidence of infection
Maternal vascular malperfusion, usually in the context of reduced fetal reserve for tolerating additional intrauterine stressors
Definition: Poorly branched (sparse) and pathologically small distal villous branches with increased syncytial knots, resulting in diminished terminal branches relative to number of stem villi
Constraints: Applicable area bounded by lower two-thirds of a full thickness non-marginal disc slide
Extent:
Focal: Involves at least 30% of bounded area on one slide
Diffuse: Involves at least 30% of bounded area on two or more slides
Definition: Enlarged distal villi with reduced syncytiotrophoblasts and centrally placed capillaries; present in more than 10 villi/focus, involving more than 30% of villi in one full thickness disc slide
Constraints: Applicable to gestational ages more than 36 weeks; rare if less than 34 weeks
Extent:
Focal: Involves one slide
Diffuse: Involves two or more slides
Definition: Lesions described under the umbrella of fetal vascular malperfusion are likely due to obstruction in fetal blood flow
Constraints: Reflects antemortem changes, even if in postmortem placentas
Extent:
Distribution:
Segmental: Chorionic or stem villus thrombosis (± avascular villi)
Presumptive cause: Thrombotic occlusion of chorionic or stem villous vessels, or stem vessel obliteration
Global: Venous ectasia, intramural fibrin in chorionic or stem villi (± small foci [<5] avascular villi)
Presumptive cause: Partially obstructed umbilical blood flow
Intensity:
High grade:
More than 45 avascular villi in three disc sections (15 avascular villi/section); or
Two or more occlusive/non-occlusive thrombi in chorionic ± stem villi; or
Multiple (3+) non-occlusive thrombi
Specific considerations:
Intramural fibrin (formerly intimal cushion), likely reflects global fetal vascular malperfusion:
Focal: Involves one slide
Recent: Subendothelial/intramural
Remote: Calcified
Avascular villi:
Small foci: Three or more foci of two to four avascular villi/focus
Intermediate foci: Three or more foci of five to 10 avascular villi/focus
Large foci: Three or more foci of 10 or more avascular villi/focus
Villous stromal-vascular karyorrhexis (formerly hemorrhagic endovasculitis ):
Three or more of two to four terminal villi showing karyorrhexis of fetal cells with preservation of surrounding trophoblast
Definition: Maternally derived lymphohistiocytic infiltrate within chorionic villi
Constraints: Three disc blocks will capture 62% of cases; six to seven blocks will capture 85% of cases
Extent:
Low grade: Less than 10 affected villi; more than one focus required
Focal: One slide
Multifocal: More than one slide
High grade: Multiple foci/slide, on more than one slide; at least one focus with more than ten villi
Patchy unless more than 30% of all villi involved
“Ungradable, possibly low grade”: Less than 10 villi, one focus
“Ungradable, possibly high grade”: More than 10 villi, one focus
Modifiers:
Location: Parabasal/basal, random, subchorionic, mixed
± stem villous obliteration (formerly obliterative fetal vasculopathy )
± associated avascular villi
In the context of fetal death, “primary placental disorders” as a concept refers to antemortem pathologic changes within the placenta of sufficient severity or degree to result in intrauterine demise. Thus, much of this chapter is devoted to the explanation and illustration of these findings.
Acute catastrophic demise is a consequence of profound, rapidly evolving intrauterine hypoxia, from sudden disruption of umbilical or uteroplacental circulation. The common etiologies, and those covered herein, include acute cord “accidents,” such as prolapse (see Chapter 31 for discussion of spontaneous umbilical cord hematoma), placental abruption, transected vasa previa, uterine rupture, and amniotic fluid embolism. These conditions are almost always unforeseen and may result in fetal death within minutes to hours. Acute modes of demise are almost always sporadic and are thus without risk of recurrence in future pregnancies.
Umbilical cord prolapse is an uncommon obstetric emergency; the incidence is around 0.1% to 0.6% of all births. It may occur at any time during the antenatal period or the intrapartum. Prolapse of the umbilical cord is defined by the descent of umbilical cord below the presenting part of the fetus.
Any condition in which the presenting fetal part does not completely occupy the maternal pelvis can lead to cord prolapse. The risk factors include abnormal fetal presentation, multifetal gestation, multiparity, low birth weight, prematurity, polyhydramnios, spontaneous rupture of membranes, and obstetric interventions, such as amniotomy or placement of internal monitors. Any form of cord prolapse may result in sudden compression of umbilical vessels, leading to compromise of the umbilical circulation and therefore insufficient fetal oxygenation.
Cord prolapse may be subdivided into three types: (1) overt cord prolapse, (2) funic presentation, and (3) occult prolapse. With overt umbilical cord prolapse, the cord descends through the cervix into the vaginal canal after the membranes are ruptured. Funic presentation means that a loop of umbilical cord lies between the fetal presenting part and the still-intact fetal membranes covering the cervical os. Occult cord prolapse occurs when the cord is located alongside the presenting part, but it is not detectable by the examiner.
Cord prolapse should be suspected when ruptured membranes are accompanied by prolonged fetal bradycardia or repetitive moderate to severe variable decelerations, the latter a clinical sign of umbilical cord compression. The diagnosis can be confirmed by palpating or visualizing the cord within the vagina, preceding the presenting part.
The overall perinatal mortality associated with cord prolapse has declined significantly in the past few decades, likely because of improvements in fetal monitoring and, more generally, to increased clinical attention in circumstances that predispose to and complicate preterm birth. Perinatal mortality is less than 3% when cord prolapse occurs in a hospital setting with continuous fetal monitoring, whereas the rate rises to 38% to 44% in non-hospitalized deliveries. Outcome is also modified by the diagnosis-to-delivery interval, cord decompression (vaginal manipulation of the presenting part away from the cord), and mode of delivery.
Pathologic confirmation of cord prolapse is usually not possible, because hypoxia progresses more rapidly than pathologic changes can evolve. In most cases, the absence of overt pathology supports an acute, catastrophic event. Morphologic evidence, if present, may be manifested solely by congestion of the fetal vascular tree.
Q: Can I diagnose acute cord prolapse?
A: No, cord prolapse has no pathologic correlates; it is a clinical diagnosis.
Placental abruption is a serious obstetric complication and an important cause of both maternal and perinatal morbidity and mortality. Placental abruption has been reported in approximately 1% of all pregnancies, with an incidence of 5.9 to 6.5 per 1000 singleton births and 12.2 per 1000 twin births. Bleeding with placental abruption is usually maternal, although disruption of the placental disc with secondary fetal bleeding from villous vessels can occur.
The primary causes of atraumatic placental abruption include maternal systemic disorders with a component of vascular disease or intrauterine conditions that predispose to maternal bleeding. Traumatic abruption can of course occur in the absence of a predisposing maternal systemic or intrauterine milieu. Associated conditions thus include trauma, cigarette smoking, cocaine use, multiple gestation, hypertension, preeclampsia, thrombophilia, advanced maternal age, prior abruption, preterm premature rupture of membranes (PPROM), intrauterine infection, and polyhydramnios. Some pregnancies will be afflicted with multiple conditions; these usually have etiologic overlap (e.g., PPROM and intrauterine infection).
The physiologic pathogenesis of placental abruption may be unknown in an individual case, although in general the process is usually explained by vascular or placental abnormalities, including increased vessel fragility, vascular malformations, or abnormalities in placentation. The mechanisms of abruption in term and extremely preterm pregnancies are in most cases distinct. At term or near term, abruption may be caused by one of two main pathways: (1) an acute direct shearing force or (2) an acute or chronic process associated with abnormal uteroplacental perfusion. A direct shearing force can occur when there is a strong direct impact to the uterus (trauma) or with sudden uterine decompression, such as after delivery of the first fetus in twin pregnancy. In the second and more common pathway, deranged uteroplacental blood flow occurs in the setting of physically abnormal spiral arterioles. Vessel rupture may occur acutely during a hypertensive episode, perhaps mediated by vasospasm, or with chronic conditions where abnormally modeled arterioles are prone to rupture, even in the absence of elevated blood pressure (e.g., with some forms of autoimmunity).
In normal pregnancies, spiral arterioles undergo physiologic remodeling, transforming from muscular arteries to vessels to non-muscular vessels with dilated lumens. This physiologic change leads to lowered vascular resistance and increased blood flow that is relatively immune to changes in maternal systemic blood pressure. The absence of this physiologic process, which is commonly found in pregnancies complicated by hypertensive disorders, results in hypoxically mediated changes to vessel walls that predispose to vessel rupture. The histologic counterpart of failed conversion is the spectrum of decidual arteriopathy (see Chapter 31 and Fig. 32.14 , presented later in the chapter).
An example of vasospasm that can lead to abruption occurs with maternal cocaine use, which increases the risk of abruption by nearly fourfold when compared with the general population. Placental abruption occurs because of the vasoconstrictive effect of the cocaine, leading to acutely decreased uteroplacental circulation.
In contrast to abruption at term, midgestational abruption cases are significantly associated with inflammation (presumptively acute deciduitis) in the setting of amniotic fluid infection. This mechanism, which we have coined “inflammatory abruption,” in the previable period raises concern for an increased risk of recurrent previable/preterm delivery, including clinically assessed cervical incompetence (a more detailed discussion of this topic is presented in Chapter 33 ).
Placental abruption, when clinically manifest, can present with any of the following maternal and/or fetal signs and symptoms: onset of vaginal bleeding accompanied by painful contractions and uterine rigidity, ultrasound demonstration of retroplacental hemorrhage, and/or signs of fetal distress as assessed by fetal monitoring. However, not all placental abruption is clinically evident. Maternal vaginal bleeding may be absent, for example, in cases of so-called concealed abruption.
The maternal mortality rate is approximately 1%, primarily the result of severe hemorrhage and its complications, such as disseminated intravascular coagulation (DIC) and renal failure. The perinatal mortality rate is higher, ranging from 4.4% to 6.7%, depending on accessibility to neonatal care facilities and gestational age. Acute catastrophic demise is significantly higher with extensive placental abruption, particularly if it involves more than 75% of the placenta. Even less dramatic abruption, as low as 25% placental separation, is associated with an increased risk of preterm birth. Although neonatal outcomes have improved because of advances in neonatal care, surviving children still have an increased risk of long-term complications, most significantly neurologic impairment.
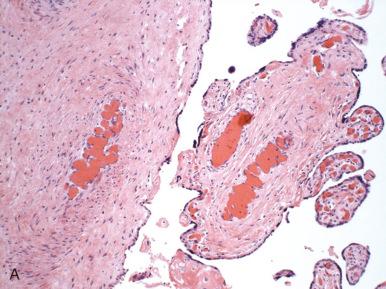
Placental abruption is defined as premature separation of a normally sited placenta from the underlying maternal surface. Hemorrhage occurs at the decidual/placental interface and may be of arterial or venous origin. Grossly, there may be a retroplacental organized blood clot with a depressed overlying maternal surface ( Fig. 32.1A ). However, if the abruption occurs shortly before delivery, there may be insufficient time for grossly visible pathologic features to evolve, and the placenta may appear normal. In general, acute abruption unassociated with mechanical trauma is arterial, whereas chronic abruption is venous. The retroplacental hematoma can extend behind the membranes as a retromembranous hemorrhage (see Fig. 32.1B ).
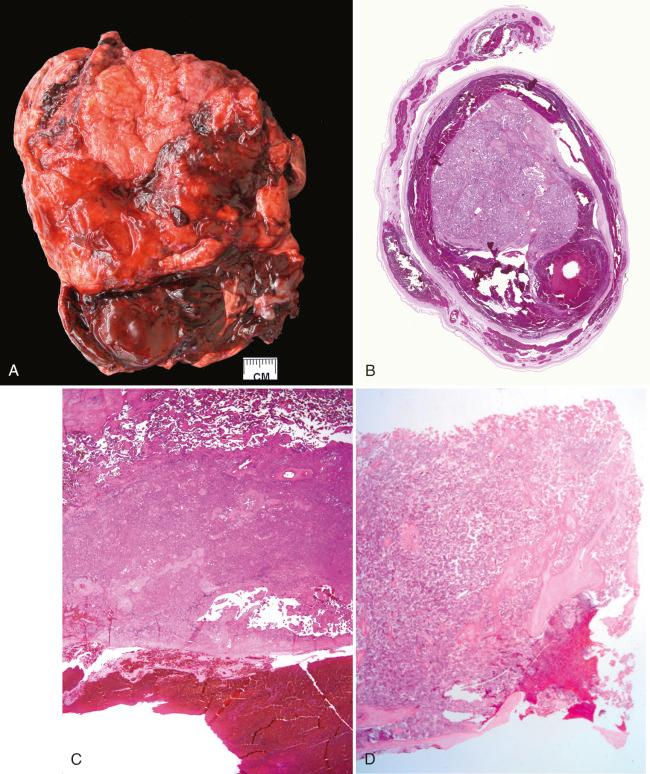
With arterial abruption, the retroplacental blood acts as a barrier, preventing still-flowing arterial blood from entering the intervillous space. The timing of abruption cannot be determined precisely by gross or microscopic examination, although some guidelines can be followed, which are broadly outlined herein. If abruption begins within a relatively few hours of delivery, the only gross finding may be a fresh loosely adherent retroplacental blood clot. After several hours, the adherent blood clot will become affixed to the maternal surface, and compression of the overlying placental tissue will ensue. Recent placental infarction, of up to a day or so in duration, appears as condensed red parenchyma grossly. As the interval between abruption, infarction, and delivery widens to days and weeks, the blood clot evolves from red to brown, as the infarct changes from red to pink to off-white.
Microscopically, sudden acute abruption may be seen as recent hemorrhage dissecting into the decidua or basal intervillous space (see Fig. 32.1D ). With time, the intervillous space overlying the abrupted area collapses, because the retroplacental blood bars fresh maternal blood from entering the placenta. This leads to parenchymal compression, and the affected villi undergo ischemic infarction (see Fig. 32.1C ). With prolonged abruption, red blood cells within the clot will fragment or degenerate through dissolution of cell integrity, and laminated fibrin will produce a layered appearance to the hematoma. Hemosiderin-laden macrophages may be seen as early as a few days following pathologic bleeding, but they are readily identified once several (4 to 5) days have elapsed. One very early microscopic parenchymal change that can be seen with abruption is fresh intravillous hemorrhage, caused by the disruption of villous capillary integrity, probably the result of mechanical forces ( Fig. 32.2 ). Otherwise, the microscopic evolution of placental infarction in the setting of abruption follows that of infarction in general, as detailed in Chapter 31 .
Placental abruption occurring shortly prior to delivery may not manifest pathologic changes.
If the third stage of labor (placental separation) is delayed, additional blood may accumulate behind the placenta and/or membranes, thus mimicking abruption.
Q: Is it the responsibility of the pathologist to diagnose abruption?
A: No, placental abruption is foremost a clinical diagnosis; pathologic correlation is intended to corroborate and therefore strengthen the clinical diagnosis.
Q: When can I confidently diagnose placental abruption?
A: The pathologic diagnosis requires retroplacental bleeding with overlying infarction; intravillous hemorrhage can add additional pathologic evidence of abruption if delivery was not by cesarean section.
Vasa previa, although uncommon, is highly likely to result in fetal mortality if undetected before membrane rupture. It occurs in approximately 1 of 2500 pregnancies and is associated with velamentous or marginal cord insertions, in vitro fertilization, placenta previa, multifetal gestations, and bilobed or succenturiate placentas. The incidence is highest with velamentous cord insertions, estimated to be 1 in every 50 cases. Velamentous vessels in vasa previa course through the membranes below the presenting part and are thus vulnerable to traumatic injury by the presenting fetal part, or transection at membrane rupture ( Fig. 32.3A ). Vasa previa can be detected in asymptomatic women as early as the second trimester by ultrasonography and color flow Doppler study.
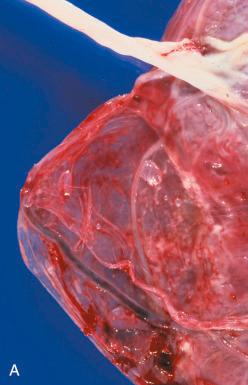
Because of the unpredictability of vasa previa and the high fetal mortality rate, screening transvaginal ultrasonography and Doppler study have been recommended in all cases at risk for vasa previa (e.g., low-lying placentas). However, management later in gestation remains controversial; the primary issue involves when in gestation to intervene, weighing potential complications of prematurity against the likelihood of spontaneous membrane rupture leading to mortality.
The diagnosis is not often made antenatally, such that patients present with painless vaginal bleeding at the time of spontaneous or artificial rupture of membranes. Rapid fetal exsanguination proceeds within minutes, manifested as sudden profound fetal bradycardia followed shortly by demise. In cases with velamentous cord insertions, the clinical presentation may be persistent fetal bradycardia from compression of velamentous vessels by the fetus' presenting part.
The fetal mortality rate ranges from 22.5% to 100% in cases where vasa previa is not recognized before the onset of labor. Fetal death occurs from asphyxia and hypovolemic shock.
One study demonstrated a significant decrease in fetal mortality in cases with prenatally diagnosed vasa previa undergoing elective cesarean delivery at 35 weeks' gestation or earlier, as compared with cases not diagnosed prenatally. In this study, the perinatal mortality rate was elevated to 56% in cases in which delivery was delayed until 38 weeks' gestation. In prenatally undiagnosed cases resulting in live birth, neonatal survival depends on prompt recognition of the condition, urgent cesarean delivery, aggressive resuscitation, and early postnatal blood transfusion.
There may be recent red (oxygen-rich) hemorrhage in the membranes adjacent to ruptured velamentous vessels, usually secondary to fetal placental venous bleeding (see Fig. 32.3B ).
Although there are no absolute microscopic criteria with which to diagnose antenatally ruptured velamentous vessels, in the appropriate clinical context features of vascular transection and accompanying damage, such as early myocyte ischemic necrosis, murally adherent platelet/fibrin aggregation, and early noninfectious neutrophil migration to damaged tissues provide compelling ancillary evidence in the appropriate clinicopathologic context.
Post-delivery mechanical trauma can disrupt placental fetal vascular integrity. Therefore mural transection alone is insufficient; additional histologic and clinical corroboration is needed.
Amniotic fluid embolism is a physiologically complex, poorly understood, and uncommon disorder with high maternal and fetal mortality rates.
When there is disruption of the physical barrier separating the maternal circulation and the amniotic fluid, amniotic fluid and its contents (fetal squames, vellus hairs) may enter the maternal bloodstream. This process may accompany labor, delivery, or cesarean section; following amniocentesis or trauma; or during termination of pregnancy. Rarely, amniotic fluid embolism occurs unrelated to an external event. The entry of amniotic fluid–laden fetal material into the maternal circulation initiates a complex physiologic reaction involving chemokines and cytokines that stimulate maternal anaphylaxis. The initial clinical phase consists of pulmonary and systemic hypertension. Circulation from the right to the left heart may fail because of pulmonary vasoconstriction. Transient but profound oxygen desaturation occurs, resulting in neurologic injury in most survivors. In one study, only 8% of women who experienced cardiac arrest in the setting of amniotic fluid embolism survived neurologically intact. Decreased systemic vascular resistance and the need for unsustainable increased left ventricular stroke effort follow this initial phase, leading to cardiovascular collapse. Oxygenation failure and coagulopathy, due in part to the presence of amniotic fluid contents entrapped in alveolar capillaries and small arterioles, stimulating the formation of platelet thrombi, occur in the secondary phase.
The three classic clinical hallmarks of maternal presentation include sudden hypotension, hypoxia, and consumptive coagulopathy. However, there is variation in clinical presentation, such that reported clinical findings include maternal hypotension, pulmonary edema, acute respiratory distress syndrome, cardiopulmonary arrest, cyanosis, coagulopathy, dyspnea, and seizure, in addition to fetal distress. The clinical diagnosis of amniotic fluid embolism is based on these characteristic signs and symptoms with exclusion of other causes.
Amniotic fluid embolism occurs in approximately 1 case per 20,000 deliveries yet accounts for 9% of pregnancy-related maternal deaths. Fetal outcome is also poor and critically related to the cardiac arrest-to-delivery interval. Overall neonatal survival is 70%, but residual neurologic impairment occurs in nearly half the survivors.
There are no gross examination correlates of amniotic fluid embolism.
Historically, squamous cells or other debris of fetal origin in the central pulmonary circulation was believed to be specific evidence of amniotic fluid embolism. Several studies have suggested that this finding is neither specific nor sensitive, pointing out that squamous cells, syncytiotrophoblast, and other debris of fetal origin may be found in the central circulation of women with conditions other than amniotic fluid embolism. However, these observations fail to take into account the presence of platelet thrombi and leukocyte margination in the pulmonary vessels of fatal amniotic fluid embolism, features that reflect failed oxygenation and anaphylaxis, respectively.
Hysterectomy, if performed antemortem, may demonstrate amniotic fluid elements in myometrial and parauterine vessels, lending supportive evidence to the diagnosis.
Placental examination is uninformative, with perhaps meconium or normoblastemia as reflective of hypoxic fetal stress.
DIC due to disorders other than amniotic fluid embolism can exhibit platelet-fibrin thrombi in microvasculature. However, in DIC, organs in addition to the lung, such as renal glomeruli, should also demonstrate microthrombi. Too, with DIC there is often perivascular, petechial, and stromal bleeding in multiple visceral sites, which does not occur with amniotic fluid embolism.
Uterine rupture may follow a preexisting injury or anomaly, may be associated with trauma, or may in rare cases afflict a previously unscarred uterus. The causes of uterine rupture may be classified into two main categories: (1) uterine intervention or predisposing anomaly present before the current pregnancy, and (2) uterine injury or abnormality during a current gestation. Uterine defects that antedate a current pregnancy include previous surgery involving the myometrium, such as cesarean section or myomectomy, uterine scarring following curettage or trauma, and congenital uterine anomaly. Uterine abnormalities that can result from a current pregnancy can occur before delivery from uterine overdistention (e.g., hydramnios, multifetal gestation), blunt or sharp external trauma, or inappropriate labor stimulation with oxytocin or prostaglandin; during delivery from internal version, a difficult obstetrical maneuver, or vigorous externally applied uterine pressure; and acquired from other causes, such as placenta percreta or gestational trophoblast neoplasia. Among these various etiologies, the most common cause of uterine rupture is separation of a previous cesarean hysterotomy scar.
The location of a uterine rupture site depends on its precipitating cause. Rupture of a previously intact uterus during labor primarily involves the lower uterine segment, which is physiologically thinner than the fundus. If there is extensive cervical dilatation, the tear is usually longitudinal. By contrast, dehiscence of a previous low transverse cesarean section scar may result in horizontal tearing that may extend to involve the broad ligament(s).
The clinical presentation may vary from a subtle non-reassuring fetal heart rate pattern to sudden maternal hypovolemic shock. Complete rupture leads to massive hemorrhage and extrusion of the fetus or placenta in the abdominal cavity.
Uterine rupture is one of the most life-threatening, although rare, obstetrical emergencies, estimated to occur in 0.1% of pregnancies, with a high risk of maternal and perinatal morbidity and mortality.
Pathologic confirmation of uterine rupture is straightforward, in that the uterus displays loss of tissue integrity and acute hemorrhage.
What may be problematic are issues of timing: namely, when the rupture began, when it resulted in intrauterine stress, and when rupture became complete. Gross and microscopic evaluation of the uterus, placenta, or fetus, depending on which specimens are examined, may shed light on some elements of timing. However, the timing of irreversible fetal injury, as with many mechanisms of intrauterine stress, is usually beyond the purview of pathologic determination alone.
Q: Is it my responsibility to time the onset of uterine rupture?
A: Although the evolution of placental findings in the setting of placental abruption is fairly stereotypical, the gross and microscopic features occur across time intervals. Specific timing must be correlated with the clinical history.
Subacute modes of demise comprise a heterogeneous etiologic group but all have their origins extrinsic to the fetus. Even infectious agents that may ultimately cause demise after entry into the fetal body are not intrinsically of fetal origin. Subacute modes of demise are usually sporadic, without risk of recurrence in future pregnancies. The evolution of disease before the demise in this group is typically of day(s) to a week or so, not minutes to hours as in acute catastrophic demise, and not weeks to months as with chronic modes of death.
Thrombosis of the fetal vasculature is found in approximately 5% of “routine” placentas. In addition to its strong association with intrauterine growth restriction (IUGR), fetal vascular thrombosis is seen most commonly with maternal diabetes, fetal death (particularly that resulting from umbilical cord “accidents,” discussed later), and gross abnormalities of the umbilical cord; less frequently it is associated with fetal chromosomal disorders, infection, or chorangiomas. Fetal thrombosis may also occasionally result from fetal hypercoagulation (e.g., secondary to protein S or protein C deficiency).
The pathogenesis of fetal vascular malperfusion, regardless of location within the fetal vascular tree, reflects non-acute reduction of fetal blood flow, resulting, for example, from stasis caused by subacute-to-chronic umbilical cord compression or the result of structural cardiac abnormalities. However, many other conditions predispose to non-acute fetal blood flow restriction, in addition to umbilical cord abnormalities (nuchal or body-wrapped cords, marginal and velamentous cord insertions, umbilical hypercoiling, tethered cords) and structural fetal heart defects. These categories include intrauterine conditions that limit cord buoyancy within amniotic fluid (such as, oligohydramnios and uterine structural anomalies), and intrinsic fetal blood flow hypercoagulability (e.g., fetal hyperglycemia due to poorly controlled maternal diabetes, leukocytosis due to congenital transient myeloproliferative disorder).
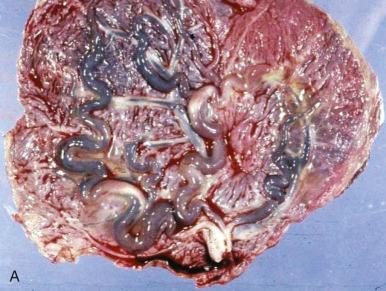
Thrombi in umbilical or fetal surface/chorionic plate vessels can occasionally be grossly visualized as dilated dark red vessels (because of recent thrombosis) or as shrunken, tan-white, sometimes calcified vessels (because of remote thrombosis) ( Fig. 32.4B and C ). Occasionally, when large regions of avascular villi are present, placental cross sections may show macroscopic pallor (so-called white infarct); this change is poorly appreciated in unfixed specimens but readily evident in formalin-fixed specimens.
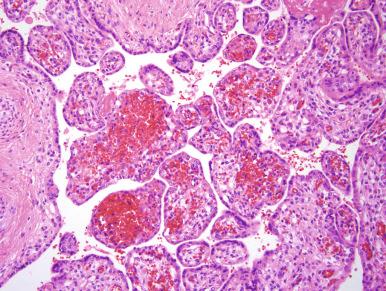
Several distinctive patterns of fetal thrombosis–related histologic lesions can be seen in the placenta, such as (1) thrombi in large muscular fetal vessels, including vessels of the umbilical cord, chorionic plate (see Fig. 32.4A ) and large stem villus vessels, and (2) fetal thrombotic vasculopathy (FTV) involving stem villi and non-muscular villous capillaries. The latter category includes both villous stromal karyorrhexis ( Fig. 32.5B ) and avascular villi (see Fig. 32.5A and C ). An additional distal villous pattern that can be seen with non-acute fetal flow restriction is that of delayed villous maturation, with centrally placed capillaries due to failure of villous modeling into a mature phenotype characterized by formation of vasculosyncytial membranes (see Fig. 32.5D and E ). Several of these patterns frequently coexist in one placenta.
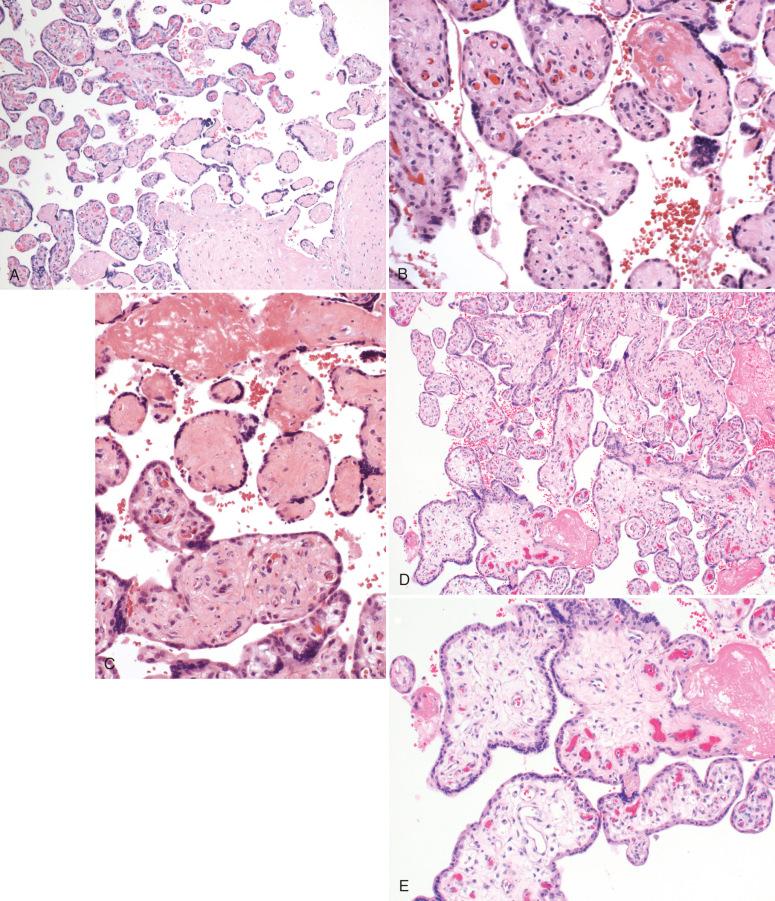
Histologically, non-occlusive to totally occlusive thrombi may be seen in umbilical vessels, chorionic surface vessels, or stem villous vessels (see Fig. 32.4D–H ). Thrombi in umbilical and chorionic vessels can often be identified as involving arteries or veins; however, in more distal fetal vessels, this is often not ascertainable from histologic assessment. Intermediate lesions in muscular vessels are eosinophilic fibrin cushions (see Fig. 32.4D ) attached to or organized within a superficial vascular neo-intima; when thrombi are most recent, the endothelium in these areas is discontinuous, and fragmented red cells are seen along the edge of the vessel (see Fig. 32.4E ). The oldest lesions are organized thrombi with dystrophic calcification (see Fig. 32.4H ).
Occlusive thrombi are often seen upstream of, or presumed to be responsible for, a cluster of avascular terminal villi. The latter lesion, one component of FTV, is defined by clusters of at least 15 avascular terminal villi on at least two distinct histologic sections. FTV is often not recognized grossly, because lesions are focal and unevenly distributed in the placental parenchyma. Histologically, FTV is characterized by either (1) fully avascular villi, which appear completely collagenized without vascular remnants (see Fig. 32.5A and C ), or (2) an intermediate-aged lesion, referred to as villous stromal karyorrhexis, characterized by nuclear debris and fragmented red cells scattered over villous stroma (see Fig. 32.5B ).
It is important to note that the changes of thrombosis and FTV develop over time, and as such they are not associated with acute cord compression (i.e., as in cord prolapse); in addition, because of the temporally heterogeneous nature of FTV, it can be easily missed, particularly in poorly sampled placentas. For this reason, we recommend a generous sampling of the placental parenchyma (at least four sections) to include fetal surface vessels in evaluation of stillbirths, as well as in placentas with gross umbilical cord abnormalities.
The most difficult aspect of fetal vascular thrombosis to interpret, and indeed to comprehend, is the simultaneous presence of both “active” and “passive” changes within a placenta in cases of non-acute antenatal or antemortem restriction of umbilical blood flow. To illustrate, consider a region of the fetal vascular tree that undergoes active venous thrombosis (as described earlier) secondary to umbilical cord compression, resulting in restriction of umbilical venous outflow and umbilical arterial inflow. In an affected placental parenchymal region, the cessation of venous outflow results in a downstream cessation of villous capillary flow, manifest by villous stromal karyorrhexis or, eventually, avascular villi. Concomitantly, because arterial inflow is impaired, the arterial stem villus vessels will exhibit either hypovolemia, in more acute cases, or, in longer-standing FVT, the nondistended septated appearance of muscular vessels that have fully ceased to experience inflow. Thus, the arterial counterpart to active, upstream venous flow obstruction is a simultaneous passive involution, which may be confused with global passive postmortem changes unless the observer can interpret all fetal vascular changes in their entirety.
Active or antenatal thrombosis, occurring while the fetus is still alive, should be distinguished from fetal vascular involution (“passive” thrombosis), which occurs after cessation of fetal circulation because of intrauterine demise. Although true (“active”) thrombosis usually occurs in dilated vessels (see Fig. 32.4F ), vascular involution ( Fig. 32.6A ) is often characterized by vessels with small septated lumens interrupted by bands of fibroblast ingrowth. Such lesions have been referred to historically as hemorrhagic endovasculitis, a highly misleading term, because it involves neither true hemorrhage nor inflammation. Active thrombosis can be difficult to distinguish from postmortem passive involutional change, particularly after a demise-to-delivery time greater than several days (see Fig. 32.6B ). One helpful distinction is temporal heterogeneity associated with active thrombosis. Whereas prolonged retention of a fetus after demise leads to vascular involution evenly distributed throughout the chorionic villi, active thrombosis occurring before demise is more likely to be regionally confined, reflected by clusters of avascular villi or villous stromal karyorrhexis present in a multifocal distribution.
The combination of thrombosis in placental fetal vessels along with FTV may help point to compromised umbilical cord blood flow (non-acute “cord accident”) as the mechanism of fetal demise. When evaluating placentas of third-trimester stillbirths, we found that the combination of (1) dilated large caliber fetal vessels, (2) thrombosis in such vessels, and (3) FTV, present as either avascular villi or villous stromal karyorrhexis, had a specificity of 95% for cord accident.
In live-birth placentas with gross umbilical cord abnormalities predisposing to compression (i.e., nuchal cords, strictured cords, long cords, hypercoiled cords, cords with true knots, abnormally inserted cords), the presence of thrombosis or FTV on histologic examination pointed to a subset of cases with adverse perinatal outcome, including fetal growth restriction. In addition, FTV alone, particularly when multifocal and extensive, has also been associated with adverse fetal sequelae, including antenatal predisposition to postnatally determined neurologic injury, severe growth restriction, and fetal demise.
Q: Can I indicate an increased risk poor outcome or cause of stillbirth in placentas with features of fetal vascular thrombosis?
A: Yes. There are criteria, overlapping but not identical, that indicate both an increased risk of untoward outcome in live births and the cause of stillbirth due to fetal vascular thrombosis.
Whereas small, even multiple, intervillous thrombi are common clinically insignificant findings in the placenta, likely representing small fetal-maternal hemorrhages in which maternal coagulation is activated to curb fetal bleeding, acute or chronic significant fetal blood loss into the maternal circulation via the intervillous space is a rare but often catastrophic event.
Fetal-maternal hemorrhage is usually silent; when this is the case, mothers present with decreased fetal movement, non-reassuring fetal tracings, or intrauterine demise. When a cause for fetal-maternal hemorrhage is known, it is invariably associated with mechanical trauma to the maternal abdomen. Causes include motor vehicle accidents, unintentional forward falls, and intentional assault.
Grossly, a fresh intervillous thrombus may be identified in a minority of placentas with documented fetal/maternal hemorrhage, which is often only 1 to 2 cm in greatest dimension (see Fig. 31.23 ). This is likely due to failed maternal coagulation to be activated in the face of fetal-to-maternal blood space bleeding. In cases associated with stillbirth, fetal surface vessels that have been drained appear hypovolemic to euvolemic ( Fig. 32.7A and B ). The fetus and the placenta are often grossly pale secondary to exsanguination of fetal blood (see Fig. 32.7C ).
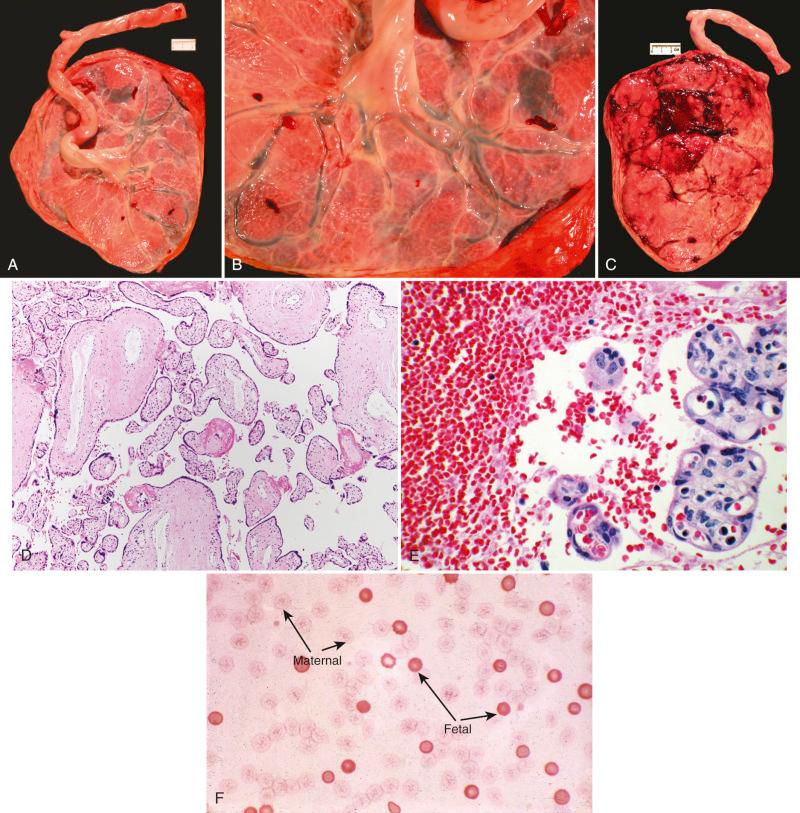
On microscopic examination, fetal vessels are often profoundly hypovolemic (see Fig. 32.7D ); nucleated red blood cells (nRBCs) are also seen (see Fig. 32.7E ). In placentas containing a fresh intervillous thrombus, the presence of nRBCs points to the fetal origin of the hemorrhage (see Fig. 32.7E ). The diagnostic test of choice for fetal/maternal hemorrhage is either flow cytometry or the Kleihauer-Betke test (see Fig. 32.7F ), which can determine the presence and quantitate the volume of fetal red blood cells in the maternal blood circulation.
The maternal Kleihauer-Betke test, if obtained days following delivery, may be falsely low due to maternal clearing of fetal blood. In these circumstances, the preponderance of evidence must rely on clinical history and gross and microscopic placental (and fetal, if performed) evaluation.
Severe acute or chronic fetal-maternal hemorrhage is highly associated with untoward outcome, including profound fetal anemia, hydrops, and demise.
Q: What clinical and pathologic features can I use to assist in rendering a diagnosis of fetal/maternal hemorrhage?
A: Clinically, Kleihauer-Betke testing on maternal blood, if done shortly after recognition of fetal demise, provides an estimation of antemortem fetal blood loss. The clinic history may elicit an instance of maternal abdominal trauma. Pathologically, gross and microscopic placental evaluation may demonstrate fetal hypovolemia, normoblastemia, and, in a minority of cases, a fresh intervillous hemorrhage containing nRBCs.
Amniotic fluid infection is also discussed in Chapter 31, Chapter 33 . It is mentioned here because it can result in fetal loss at any stage of pregnancy, although typically after the first trimester. As described elsewhere, the most common mechanism of fetal demise with amniotic fluid infection is PPROM or preterm labor (PTL), leading to previable delivery and stillbirth, or preterm birth with associated complications of infection in the setting of prematurity.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here