Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Peritoneal washing cytology (PWC) was introduced in the 1950s as a way to identify microscopic spread of cancer not visible by gross inspection of the peritoneal surface. In some cancer patients, positive PWC may be the only evidence of metastatic disease to the peritoneum. Because positive results correlate with poorer prognosis, cytologic findings are included in the staging system for ovarian and fallopian tube cancers. The yield, however, is low: positive washings by themselves change the surgical stage of only 3% to 5% of women with gynecologic cancers.
PWC is also used to exclude an occult cancer in patients who undergo laparoscopy or laparotomy for presumed benign gynecologic conditions (e.g., endometriosis, leiomyomata) and in women with BRCA1 and BRCA2 mutations undergoing risk-reducing salpingo-oopherectomy. PWC can be used to monitor a patient’s response to treatment for advanced ovarian cancer and other malignancies (the “second-look” procedure), but this is usually limited to patients in research protocols. In some instances, PWC is used to detect peritoneal spread of nongynecologic malignancies such as pancreatic and gastric cancer.
Staging gynecologic malignancies
Ruling out occult cancer
Assessing response to treatment (the “second-look” procedure)
Staging nongynecologic malignancies
On entering the peritoneal cavity, the surgeon evacuates any preexisting peritoneal fluid and submits it separately for cytologic examination. Washings are obtained by instilling 50–200 mL of sterile saline or other physiological solution into several different areas, usually the pelvis, the right and left paracolic gutters, and the undersurface of the diaphragm. The fluid is aspirated and submitted for cytologic evaluation. Segregating washings from different abdominal cavity sites and sending them separately for cytologic evaluation does not have any advantage over combining them into a single specimen.
The specimen should be delivered to the laboratory unfixed and refrigerated at 4°C until slides can be prepared. If a significant delay before cytopreparation is anticipated, an equal volume of 50% ethanol can be added. To prepare slides, the specimen is thoroughly mixed, and an aliquot (often 50 mL) is spun in a centrifuge to a cell sediment/pellet. From this sediment one can prepare smears, cytocentrifuge preparations, or thin-layer preparations (e.g., ThinPrep, SurePath), depending on the resources and preferences of the laboratory. The remaining (or separately centrifuged) cell sediment can also be fixed in 10% formalin, embedded in paraffin, and processed as a histologic specimen (“cell block”). Cell block sections are very useful, especially for morphologic comparison to the patient’s resected neoplasm.
Results of PWC are commonly reported as “negative for malignant cells,” “atypical,” “suspicious for malignancy,” or “positive for malignant cells.” “Atypical” (connoting a low degree of suspicion for malignancy) and “suspicious for malignancy” (connoting a high degree of suspicion) interpretations should be avoided whenever possible because they are not helpful to a physician faced with making a treatment decision. In most cases, only an unequivocally positive diagnosis is used for staging purposes—anything less is treated as a negative result. Side-by-side comparison with the corresponding resection specimen often helps resolve an equivocal case. For involvement of peritoneal washings by a known borderline tumor, “neoplastic cells present” is preferred over “positive for malignant cells” because borderline tumors, strictly speaking, are not malignancies.
Criteria for specimen adequacy have not been established, but it seems reasonable to require some benign mesothelial cells before considering a peritoneal washing specimen adequate and negative for malignant cells. A specimen with malignant cells is always adequate.
Because peritoneal washings are obtained as part of a cancer staging procedure, there is usually a concurrent histologic specimen. For example, when washings are obtained as part of ovarian cancer staging, an oophorectomy is obtained by the laboratory at the same time. Representative slides from the oophorectomy specimen can be helpful in a side-by-side comparison of a diagnostically difficult washing specimen.
Not all patients with metastases to the peritoneum have positive PWC results. In fact, 23% to 52% of patients with biopsy-proven peritoneal involvement have negative results. When peritoneal washings are examined as part of a second-look procedure, the false-negative rate is even higher, ranging from 31% to 86% of patients with biopsy-proven metastases. The higher false-negative rate of second-look procedures may be due to the poor distribution of fluid when the abdominal cavity is altered by adhesions.
False-positive diagnoses are uncommon but well documented, occurring in less than 5% of cases. Causes include reactive mesothelial proliferation with psammoma bodies and endometriosis. In particular, eosinophilic metaplastic atypia in endometriosis can be a source of atypical (but benign) cells that might be misconstrued as malignant. In one instance, ectopic pancreatic tissue was misinterpreted as adenocarcinoma in a patient with a mucinous ovarian cancer.
Peritoneal washings differ morphologically from peritoneal fluid (ascites) in several easily recognizable ways. First, the washing procedure mechanically strips the peritoneal surface of entire sheets of mesothelial cells, whereas sheets of mesothelial cells are not seen in a benign peritoneal effusion. (One exception: peritoneal and pleural effusions suctioned during abdominal or thoracic surgery contain sheets of peritoneal cells that are pulled in by the suctioning process.) Second, skeletal muscle and adipose tissue fragments are common in peritoneal washings, especially in cell block preparations.
Mesothelial cells in sheets
Collagen balls
Histiocytes
Skeletal muscle
Adipose tissue
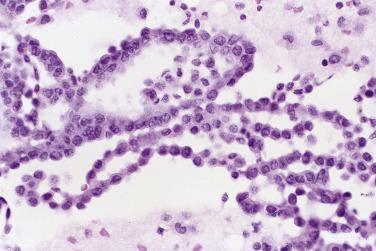
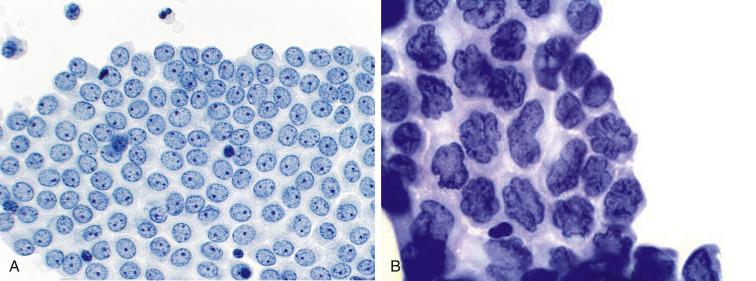
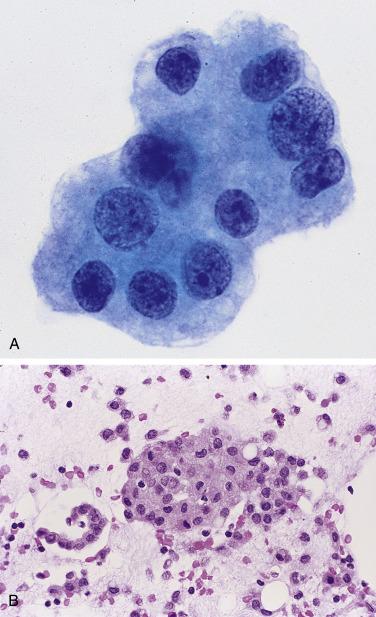
Mesothelial cells in peritoneal washings are arranged predominantly in flat sheets, often large and occasionally folded ( Fig. 5.1 ). In cell block sections the sheets are transected and appear as long, thin ribbons ( Fig. 5.2 ). The cells are evenly spaced, with a moderate amount of cytoplasm. Nuclear membranes are thin, and the chromatin is pale and evenly dispersed; small nucleoli are often present. Although usually round or oval ( Fig. 5.3A ), the nuclei are sometimes scalloped, wrinkled, or grooved, possibly due to fixation artifact. In extreme cases the nuclei have a flower-like appearance (“daisy cells,” Fig. 5.3B ). Isolated mesothelial cells, similar to those seen in ascites, are also encountered.
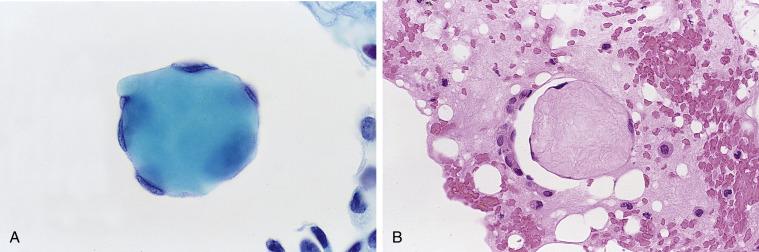
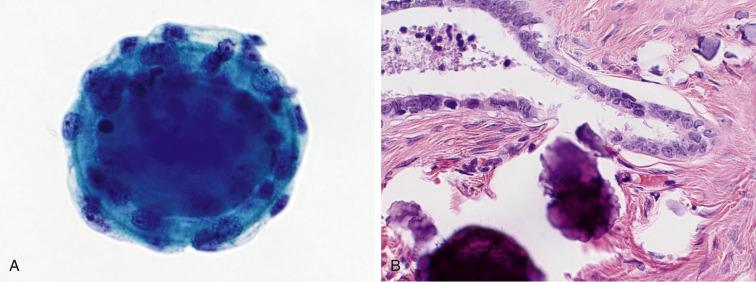
Spherical masses of collagen surrounded by benign, flattened mesothelial cells, known as collagen balls , are seen in up to 50% of peritoneal washings. Aqua in color with the Papanicolaou stain, they have round or bosselated contours ( Fig. 5.4 ). They are usually few in number, but occasionally they can be abundant. They have no known significance (and therefore do not deserve mention on a cytology report). It has been suggested that they result from a pinching off of mesothelial-lined stromal projections known as micropapillomatosis on the surface of the ovaries.
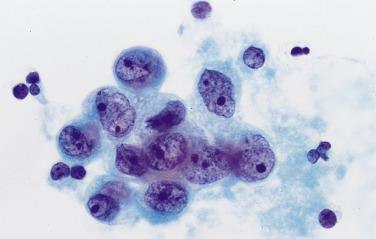
Numerous histiocytes , scattered either as isolated cells or in variably-sized aggregates, are often present ( Fig. 5.5 ). Because they look different from mesothelial cells and are often haphazardly aggregated, they might be misinterpreted as metastatic cancer cells. Attention to the typical nuclear and cytoplasmic features of histiocytes (oval, folded, and kidney-shaped nuclei; pale chromatin; granular and microvacuolated cytoplasm) is helpful in correctly identifying them. Skeletal muscle and adipose tissue fragments are sometimes seen: they fall into the peritoneal cavity when the abdominal incision is made and are suctioned along with the fluid. Detached ciliary tufts , presumably of fallopian tube origin, are a relatively common incidental finding, especially when the washings are obtained during the secretory (luteal) phase of the menstrual cycle. A typical interpretation of a benign peritoneal washing might read as follows: “Negative for malignant cells. Mesothelial cells and histiocytes.”
A group of benign conditions involving the peritoneal and ovarian surfaces results in a proliferation of fallopian tubal-type epithelial cells or mesothelial cells, often with psammoma bodies. These conditions mimic peritoneal involvement by a serous borderline tumor and serous carcinoma; familiarity with them is thus important.
Endosalpingiosis is a proliferation of benign glands and cysts lined by ciliated, fallopian tube–like epithelium. Common locations include the ovarian cortex, uterine serosa, peritoneal surface, and omentum. Psammoma bodies are often seen. Some authors reserve the term endosalpingiosis for proliferations that include tubal-type stroma, as well as glands, and use the term Müllerian inclusion cyst for cases that lack tubal-type stroma. Both endosalpingiosis and Müllerian inclusion cysts, however, are usually incidental microscopic findings in a patient undergoing surgery for something else. Histologically, they raise the possibility of metastatic disease but are identified as benign proliferations because the cells are uniform and bland and lack mitotic activity.
Serous adenofibromas of the ovarian surface are benign ovarian tumors that, like endosalpingiosis, are composed of benign tubal-type glands, cysts, or both, except that they are often a visible mass rather than a microscopic finding, and they have a broad, fibrous, stromal component. The epithelial portion of a serous adenofibroma, such as endosalpingiosis, often contains psammoma bodies and can be confused with peritoneal involvement by a serous borderline tumor or even adenocarcinoma.
Prior surgery, pelvic inflammatory disease, a ruptured cyst, and other conditions cause florid mesothelial hyperplasia , sometimes accompanied by psammoma body formation.
Cuboidal cells (± cilia) with minimal to mild atypia
Psammoma bodies
The conditions described above all have a similar appearance in peritoneal washings. Clusters of cuboidal mesothelial-like cells ( Fig. 5.6 ), some arranged around a psammoma body ( Figs. 5.7 and 5.8 ) or nondescript calcification, are present and can be abundant. Cilia may be present, but often they are absent or impossible to identify. Nuclei are round or oval, with a pale chromatin pattern and small nucleoli. Nuclear atypia is mild, and mitoses are very uncommon.
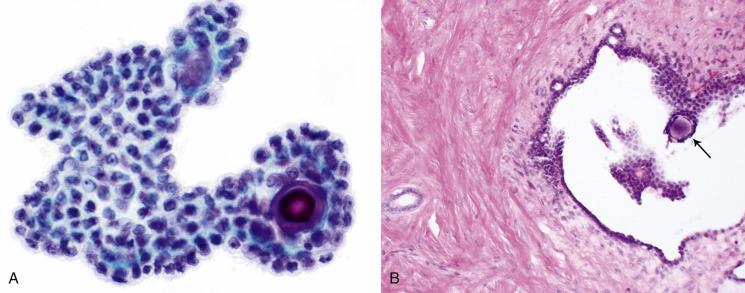
These benign conditions are a potential cause of a false-positive interpretation of involvement by a serous carcinoma or borderline tumor. To avoid a false-positive interpretation, a diagnosis of malignancy should not be based on the presence of psammoma bodies alone. Correlation with the concurrent histologic material is helpful in most cases. Certainly, if the patient does not have a carcinoma or borderline tumor, one should be cautious rendering such a diagnosis on the basis of the peritoneal washings alone. Some patients with endosalpingiosis or benign mesothelial proliferations, however, can also have a serous borderline tumor confined to the ovary. Comparison of the peritoneal washings, particularly cell block preparations, with the histologic sections from the tumor often resolves equivocal cases, but the final interpretation is not always straightforward, and the pathologist is sometimes required to make the best judgment possible. There are few helpful morphologic clues, particularly because many serous borderline tumors have only mild cytologic atypia, equivalent to what can be seen with florid mesothelial hyperplasia and endosalpingiosis. One possible clue: in cell block sections, some papillary fragments of serous borderline tumors have broad stromal cores, a feature not seen in cell blocks containing only mesothelial hyperplasia or endosalpingiosis.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here