Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Periprosthetic elbow fractures are difficult conditions complicating an elbow arthroplasty. They may occur intraoperatively during primary or revision surgery or after high- or low-energy trauma or may represent an end-stage implant loosening and osteolytic periprosthetic disease. This wide spectrum of mechanisms of injury and underlying host conditions requires a specific evaluation sequence, a clear algorithm for treatment decision making, and often special surgical techniques for reconstruction. All these points are discussed in this present chapter.
Fractures around the components of total elbow arthroplasties are uncommon injuries with an estimated prevalence of 5% in primary replacements. However, recent developments in implant design with stronger and more durable implants will probably increase the spectrum of both the indications for elbow replacement and the age of the patients undergoing this operation. The indications for elbow replacement are also shifting from inflammatory conditions, the most frequent indication in the past, to acute trauma in elderly patients and treatment of fracture sequelae in younger individuals, which are much more prevalent in the general population and currently the most increasing indications. All these factors, in combination with an aging and active population, will probably soon lead to an increase in the volume and prevalence of periprosthetic elbow injuries.
The main risk factor for periprosthetic elbow fractures is poor bone stock around components. Potential causes of decreased bone stock can:
be present before the replacement is performed, as in rheumatoid arthritis patients with advanced elbow disease or in old patients with severe osteoporosis with complex distal humeral fractures
be due to surgical factors, such as implant design selection (some stem surface finishes are associated with osteolysis and loosening), inadequate tissue balance, component malalignment (leading to polyethylene wear, metallosis and particle disease, and finally loosening), or poor cementation technique
develop after prosthetic implantation, as mechanical overload or infection, leading to progressive osteolysis and eventually bone fracture
As mentioned already, periprosthetic elbow fractures are commonly the final stage of implant loosening and osteolysis. This is illustrated by many cases (45% and 57% of fractures around the humeral and ulnar stems, respectively) in which the moment at which the fracture occurred was not recalled, or it was described as happening during daily activities, such as during adjustment of the rear-view mirror of a car or getting up from a chair (up to 20%). Falls from standing height are the other main mechanism of production (25% to 45%).
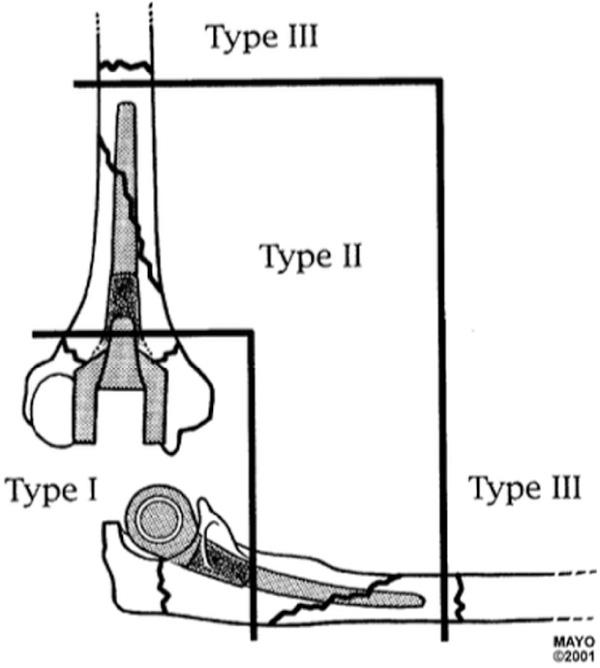
The Mayo classification of periprosthetic elbow fractures is similar to the Vancouver periprosthetic hip fracture classification ( Fig. 103.1 ). The fractures may affect the humerus or the ulna. They may be located at the humeral condyles or olecranon (type I), around the implant stem (type II), or distal to the stem (type III). In type II fractures, when the remaining bone stock is adequate, the component may be well fixed (subtype 1) or loose (subtype 2); the remaining bone stock may be not adequate (subtype 3), with thin, brittle, or even absent cortical areas or medullar insufflation, and may require special reconstructive techniques to restore it. Final classification may require information not only from the initial imaging tests but also information obtained during the surgery, including bone assessment and the final treatment needed to ensure a stable and durable reconstruction.
Evaluation of patients with periprosthetic elbow injuries should always include review of the history and obtaining information about the previous conditions of the elbow and the nature of any prereplacement procedures. The reason for the index arthroplasty or any subsequent revision surgery must be documented, as must the design, year of manufacture/implantation (e.g., precoat ulnar implants used from 1990 to 2000 are associated with bone osteolysis ), and the size of the prosthetic components in place. A thorough investigation is performed, looking for any wound problems in the past or data suspicious of septic antecedents. The history is thus focused on understanding the mechanism of production of the fracture and the underlying conditions affecting bone stock or our treatment decision making (i.e., a periprosthetic fracture in the context of trauma in an otherwise healthy bone with a well-functioning replacement versus a catastrophic bone disruption in an insufflated, osteolytic, and possibly infected bone, in which the fracture is just a step more in the implant failure process).
Physical examination includes a complete neurovascular examination focusing on ulnar and radial nerve symptoms and function, assessing triceps integrity and strength, and determining a functional evaluation by the means of recording the range of motion and elbow stability, if the pain allows this.
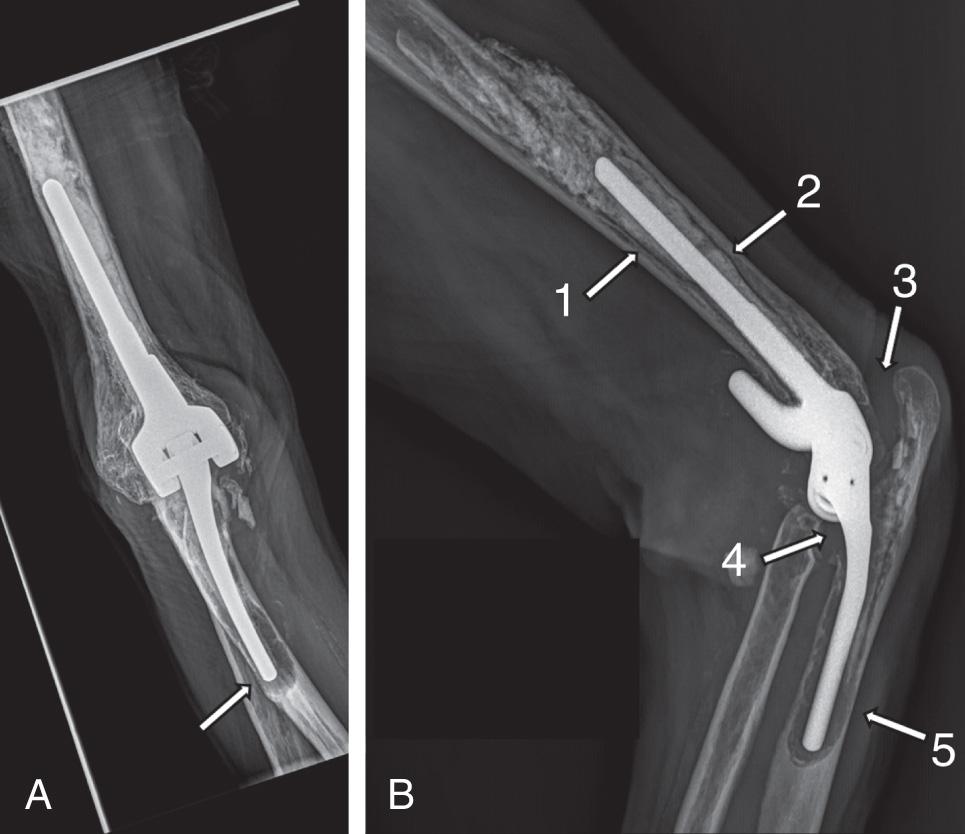
X-rays are usually enough to understand fracture location and comminution, implant fixation status, and bone stock ( Fig. 103.2 ). Long-cassette x-rays including the whole arm and forearm are very useful. To select the best treatment option, the radiologic evaluation should be systematic and aimed at obtaining a clear understanding of the following injury features:
Fracture location (see the earlier classification) and description (orientation of fracture lines, extension, and comminution).
Fixation status of components, looking for radiolucent lines and assessing the quality of the cement mantle. A comparison between the ulnar and the humeral fixation in combination with the time from their implantation can help in understanding the mechanism of failure. Loosening and osteolysis affecting only one of the components is more probably related to a poor-quality cementing technique or problems with a specific component design, whereas a diffuse osteolysis affecting both components could be related to a chronic infection. Migration of a loose component far from the joint deeper in the bone canal should also be documented, as a longer stem than previously thought may be needed for revision if only the native component length was considered (see Fig. 103.2 ).
Bone stock, the assessment of which includes, apart from the fracture itself, documentation of additional cortical thinning areas representing sites of potential fracture in the future, cortical defects and their extension, increases in the medullar diameter (bone insufflation), and the length of the osteolytic bone that needs to be addressed.
Infection should be actively investigated with a thorough clinical history looking for past signs or symptoms of infection, a blood test with leukocyte count, erythrocyte sedimentation rate, and C-reactive protein level; fluid joint aspiration for Gram stain, culture, and an α-defensin test if available; and intraoperative tissue sampling, including membranes around components for intraoperative disease. If the findings of any of the tests mentioned are positive, or any suspicion of infection remains despite negative findings, a formal two-stage procedure should be considered. Additionally, intraoperative soft tissue sampling for culture and implant sonication is performed routinely.
In the first surgical procedure, loose implants are removed and the intramedullary canal is identified with a 3.2-mm drill bit. Then cement is removed with osteotomes, curets, and reaming as needed. Cortical defects are exploited for cement removal. A longitudinal osteotomy of the ulna or a posterior cortical window in the humerus is developed to remove well-fixed components. If a well-fixed implant is found in the context of staging the revision procedure, keeping it in place until results of cultures are available can be considered when the findings for all previously mentioned markers are negative. All synovial membranes, debris, and intramedullary membranes are débrided, and specimens are obtained in every case for histologic analysis and culture. Implants are sent for ultrasonication to increase the sensitivity of periprosthetic tissue culture. Antibiotic spacers are placed, and empiric broad-spectrum intravenous antibiotics are administered. The second stage is performed if cultures remain negative after up to 10 days to rule out infection caused by slow-growing bacteria. If cultures are positive, a second surgical débridement is considered if the first-stage procedure left implants or cement in place, and patients receive intravenous antibiotics for 6 weeks on the basis of the results of the cultures or the sonicated intraoperative specimens. The second stage is performed after 2 weeks of antibiotic cessation and involves removal of spacers as well as repeat débridement, pathologic analysis, and culture. If the pathology results were negative and the clinical picture, including low C-reactive protein concentration, erythrocyte sedimentation rate, and white blood cell count, was reassuring, the reconstructive procedure is then completed with fracture fixation and new prosthetic implantation.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here