Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The spectrum of pericardial diseases consists of congenital defects, pericarditis (dry, effusive, effusive-constrictive, or constrictive), neoplasms, and cysts. The etiologic classification comprises infectious pericarditis, pericarditis in systemic autoimmune diseases, type 2 (auto) immune process, postmyocardial infarction syndrome, and autoreactive (chronic) pericarditis. ,
Congenital defects of the pericardium are found in 1 in 10,000 autopsies. Pericardial absence can be partial left (70%), partial right (17%), or total bilateral (rare). Additional congenital abnormalities occur in approximately 30% of patients. Most patients with total pericardial absence are asymptomatic. Homolateral cardiac displacement and augmented heart mobility impose an increased risk for traumatic aortic dissection. Partial left-side defects can be complicated by herniation and strangulation of the heart through the defect (causing chest pain, shortness of breath, syncope, or sudden death). Surgical pericardioplasty (Dacron, Gore-Tex, or bovine pericardium) is indicated for imminent strangulation.
Acute pericarditis is dry, fibrinous, or effusive, independent of its etiology. The major symptoms are retrosternal or left precordial chest pain (which radiates to the trapezius ridge, can be pleuritic or simulate ischemia, and varies with posture) and shortness of breath. A prodrome of fever, malaise, and myalgia is common, but elderly patients may not be febrile. A pericardial friction rub can be transient and monophasic, biphasic, or triphasic. A pleural effusion may be present. The heart rate is usually rapid and regular. Echocardiography is essential to detect effusion and concomitant heart or paracardial disease ( Table 75.1 ).
| Diagnostic Measure | Characteristic Findings |
|---|---|
| Obligatory | |
| Auscultation | Pericardial rub (monophasic, biphasic, or triphasic) |
| ECG * | Stage I: anterior and inferior concave ST-segment elevation. PR-segment deviations opposite to P-wave polarity. |
| Early stage II: all ST junctions return to the baseline. PR segments deviated. | |
| Late stage II: T waves progressively flatten and invert. | |
| Stage III: generalized T-wave inversions in most or all leads. | |
| Stage IV: ECG returns to prepericarditis state. | |
| Echocardiography | Effusion types B to D (Horowitz). |
| Signs of tamponade. | |
| Blood analyses | Erythrocyte sedimentation rate, C-reactive protein, lactate dehydrogenase, leukocytes (inflammation markers). |
| Troponin I, † CK-MB (markers of myocardial involvement). | |
| Chest radiograph | Ranging from normal to “water bottle” shape of the heart shadow. |
| Performed primarily to reveal pulmonary or mediastinal pathology. | |
| Mandatory in Tamponade, Optional in Large/Recurrent Effusions or if Previous Tests Inconclusive in Small Effusions | |
| Pericardiocentesis/drainage | Polymerase chain reaction and histochemistry for etiopathogenetic classification of infection or neoplasia. |
| Optional or if Previous Tests Inconclusive | |
| CT | Effusions, pericardium, and epicardium. |
| MRI | Effusions, pericardium, and epicardium. |
| Pericardioscopy, pericardial/epicardial biopsy | Establishing the specific etiology. |
* Typical lead involvement: I, II, aVL, aVF, and V 3 –V 6 . The ST segment is always depressed in aVR, frequently in V 1 , and occasionally in V 2 . Stage IV may not occur, and there are permanent T-wave inversions and flattenings. If an ECG is first recorded in stage III, pericarditis cannot be differentiated by ECG from diffuse myocardial injury, “biventricular strain,” or myocarditis. ECG in early repolarization is very similar to stage I. Unlike stage I, this ECG does not acutely evolve, and J-point elevations are usually accompanied by a slur, oscillation, or notch at the end of the QRS just before and including the J point (best seen with tall R and T waves—large in an early repolarization pattern). Pericarditis is likely if, in lead V 6 , the J point is greater than 25% of the height of the T-wave apex (using the PR segment as a baseline).
† A rise in cardiac muscle troponin I (cTnI) was detected in 38/118 patients (32.2%); more frequently in younger, male patients; and with ST-segment elevation and pericardial effusion at presentation. An increase beyond 1.5 ng/mL was rare (7.6%) and associated with CK-MB elevation. cTnI increase was not a negative prognostic marker for the incidence of recurrences, constrictive pericarditis, cardiac tamponade, or residual left ventricular dysfunction.
Hospitalization and symptomatic treatment are warranted. Nonsteroidal antiinflammatory drugs (NSAIDs) are the mainstay. Indomethacin should be avoided in elderly patients owing to its effect on reducing flow in the coronaries. Ibuprofen (300–800 mg TID) is preferred for its rare side effects, favorable impact on coronary blood flow, and large dose range. Colchicine 0.5 mg at least twice daily for 3 months added to aspirin or another NSAID reduced the recurrence rate impressively in the COPE trial, even at the first episode of pericarditis or even as monotherapy in “idiopathic” effusions. Colchicine is well tolerated, with fewer side effects than NSAIDs. Systemic corticosteroids should be restricted to connective tissue diseases and autoreactive or uremic pericarditis. Intrapericardial steroid application, as long-acting crystalloid triamcinolone, is effective for autoreactive effusions and avoids systemic side effects.
Chronic (>3 months) pericarditis includes effusive (inflammatory or hydropericardium in heart failure), adhesive, and constrictive forms. Symptoms (chest pain, palpitations, and fatigue) are usually mild and related to the degree of cardiac compression and pericardial inflammation. The detection of curable causes (e.g., tuberculosis; toxoplasmosis; myxedema; or viral, autoimmune, and systemic diseases) allows successful specific therapy. Symptomatic treatment and pericardiocentesis should be applied, if indicated. For recurrences, the etiology should be investigated intensely, and if no specific therapy is effective, balloon pericardiotomy or pericardiectomy may be considered.
The term recurrent pericarditis encompasses (1) the intermittent type (symptom-free intervals without therapy) and (2) the incessant type (discontinuation of antiinflammatory therapy precipitates a relapse). Massive pericardial effusion, overt tamponade, or constriction is rare. Symptomatic management relies on exercise restriction and regimens used for acute pericarditis. Colchicine may be effective when NSAIDs and corticosteroids fail to prevent relapses. , It should be considered the first-choice treatment for recurrent pericarditis according to the CORE trial. Corticosteroids should be used only in patients with a poor general condition or in frequent crises. A common mistake could be to use a dose too low to be effective or to taper the dose too rapidly. The recommended regimen is prednisone 1–1.5 mg/kg for at least 1 month. If patients do not respond adequately, azathioprine (75–100 mg/day) or cyclophosphamide can be added.
Corticosteroids should be tapered over a 3-month period. Toward the end of the taper, introduce antiinflammatory treatment with colchicine (0.5 mg BID or TID) or an NSAID. Renewed treatment should continue for 3–6 months. Recently, it has been demonstrated in “idiopathic” pericarditis that previous corticosteroid treatment was a risk factor for recurrence or chronicity. Therefore corticosteroids should be administered after the definite exclusion of viral or bacterial infection of the pericardium. Pericardiectomy is indicated only for frequent and highly symptomatic recurrences resistant to medical treatment.
Pericardial effusion may appear as transudate (hydropericardium), exudate, pyopericardium, or hemopericardium. Large effusions are common with neoplastic, tuberculous, cholesterol, and uremic pericarditis and with myxedema and parasitoses. Loculated effusions are more common when scarring has supervened (e.g., postsurgical, posttraumatic, or purulent pericarditis). Effusions that develop slowly can be remarkably asymptomatic, whereas rapidly accumulating smaller effusions can present as tamponade. Cardiac tamponade is the decompensated phase of cardiac compression caused by effusion accumulation, leading to increased intrapericardial pressure. Heart sounds are distant. Orthopnea, cough, and dysphagia, occasionally with episodes of unconsciousness, can be observed. Insidiously developing tamponade may present as signs of its complications (renal failure, abdominal plethora, shock liver, worsening of glaucoma, and mesenteric ischemia). Tamponade without two or more inflammatory signs (typical pain, pericardial friction rub, fever, or diffuse ST-segment elevation) is usually associated with a malignant effusion (likelihood ratio, 2.9).
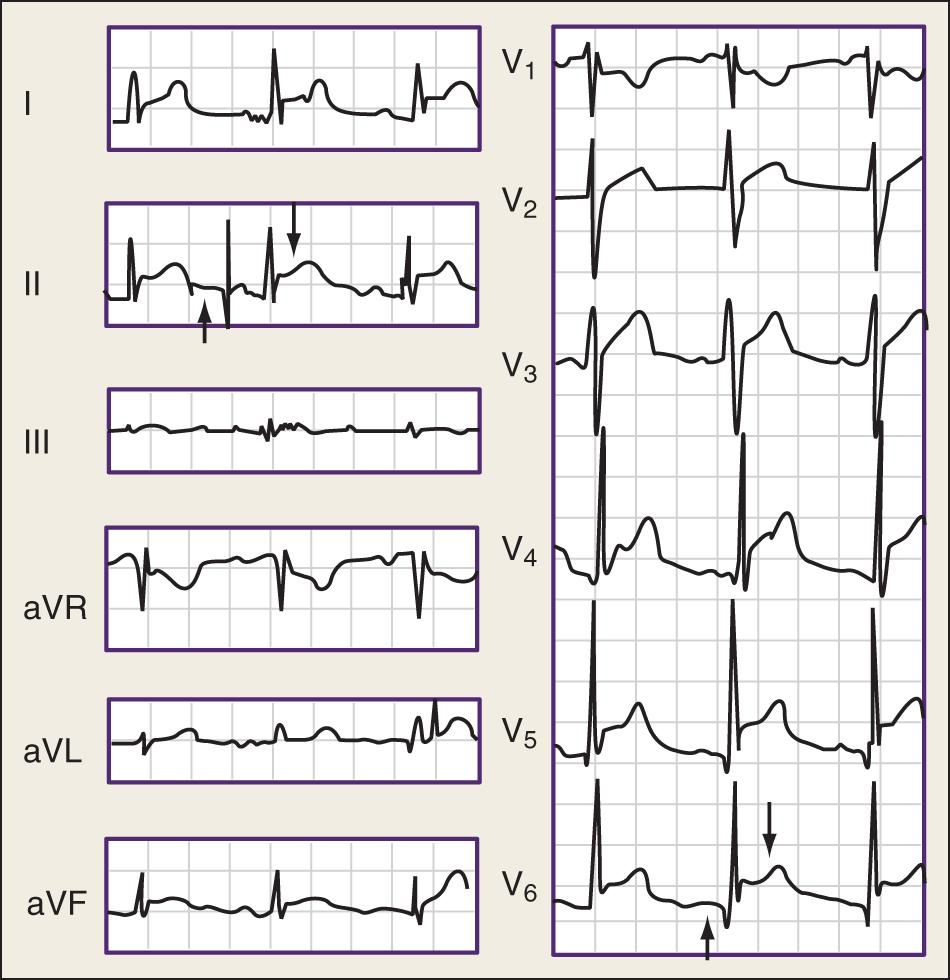
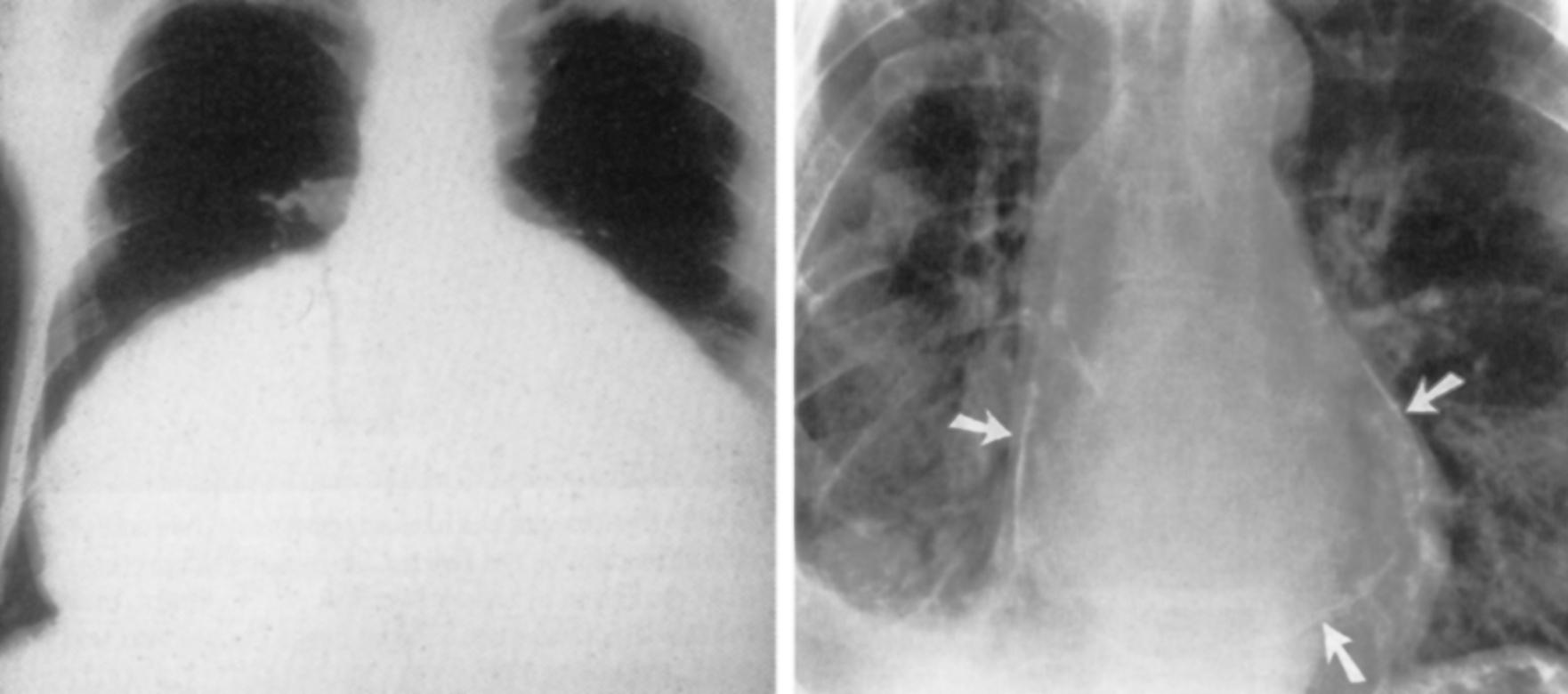
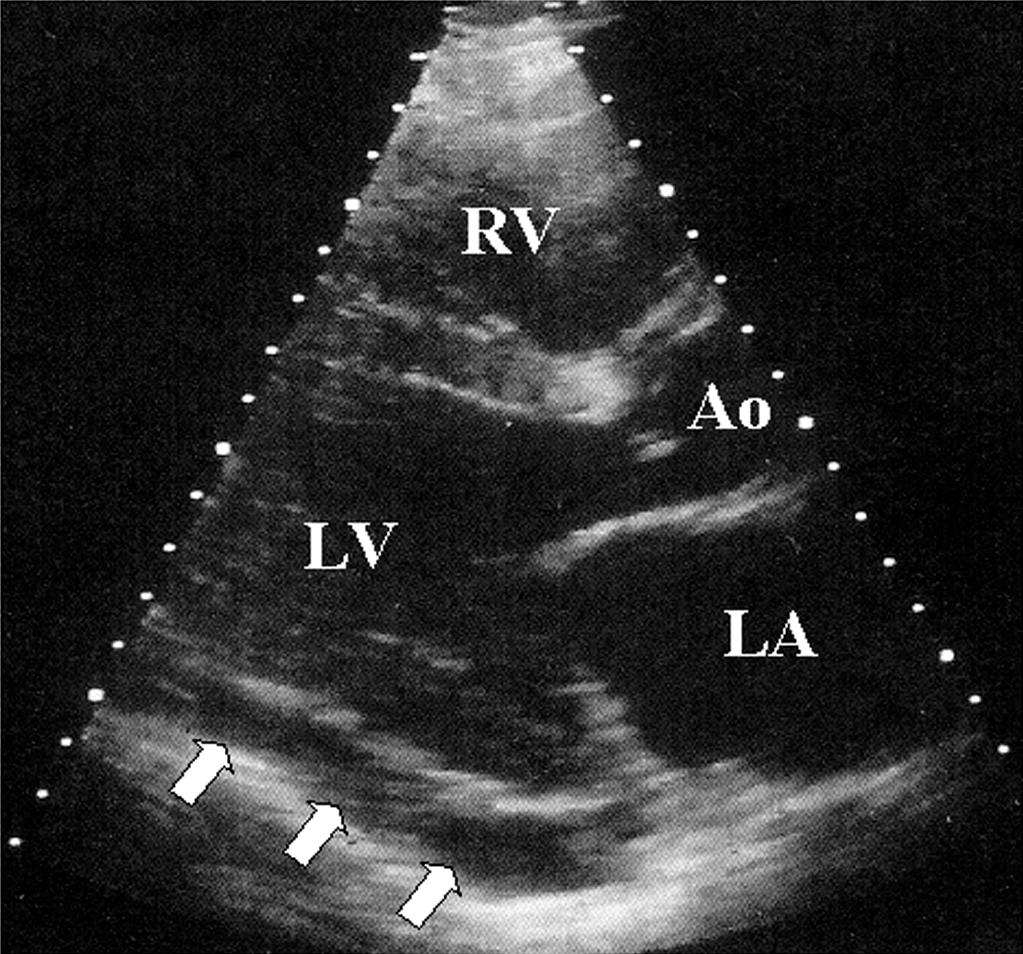
Electrocardiography demonstrates low QRS and T-wave voltages, PR-segment depression ( Fig. 75.1 ), ST-segment/T-wave changes, bundle branch block, and electrical alternans (rarely seen in the absence of tamponade). Microvoltage and electrical alternans are reversible after the drainage of the effusion and resolution of the inflammatory process. In chest radiography, large effusions are depicted as globular cardiomegaly with sharp margins (“water bottle” silhouette) ( Fig. 75.2 ). The size of effusions noted during echocardiography can be graded as (1) small (echo-free space in diastole <10 mm), (2) moderate (10–20 mm; Fig. 75.3 ), (3) large (≥20 mm), or (4) very large (≥20 mm and compression of the heart). In large pericardial effusions, the heart may move freely within the pericardial cavity (“swinging heart”), inducing pseudoprolapse and pseudosystolic anterior motion of the mitral valve, paradoxical motion of the interventricular septum, and midsystolic aortic valve closure ( Table 75.2 ). Up to one-third of patients with an asymptomatic large pericardial chronic effusion develop unexpected cardiac tamponade. Triggers for tamponade include hypovolemia, paroxysmal tachyarrhythmia, and intercurrent acute pericarditis.
| Clinical presentation | Elevated systemic venous pressure, * hypotension, † pulsus paradoxus, ‡ tachycardia, § dyspnea, or tachypnea with clear lungs |
| Precipitating factors | Drugs (cyclosporine, anticoagulants, thrombolytics), recent cardiac surgery, indwelling instrumentation, blunt chest trauma, malignancies, connective tissue disease, renal failure, septicemia || |
| ECG | Can be normal or nonspecifically changed (ST-T wave), electrical alternans (QRS, rarely T), bradycardia (end stage), electromechanical dissociation (agonal phase) |
| Chest radiograph | Enlarged cardiac silhouette with clear lungs |
| M-mode/two-dimensional echocardiogram | Diastolic collapse of the anterior RV free wall, ¶ RA collapse, LA and rarely LV collapse, increased LV diastolic wall thickness “pseudohypertrophy,” IVC dilatation (no collapse in inspiration), “swinging heart” |
| Doppler | Tricuspid flow increases and mitral flow decreases during inspiration (reverse in expiration) |
| Systolic and diastolic flows are reduced in systemic veins in expiration, and reverse flow with atrial contraction is increased | |
| M-mode color Doppler | Large respiratory fluctuations in mitral/tricuspid flows |
| Cardiac catheterization | Confirmation of the diagnosis and quantification of the hemodynamic compromise |
| RA pressure is elevated (preserved systolic × descent and absent or diminished diastolic and descent) | |
| Intrapericardial pressure is also elevated and virtually identical to RA pressure (both pressures fall in inspiration) | |
| RV mid-diastolic pressure is elevated and equal to the RA and pericardial pressures (no dip-and-plateau configuration) | |
| Pulmonary artery diastolic pressure is slightly elevated and may correspond to the RV pressure | |
| Pulmonary capillary wedge pressure is also elevated and nearly equal to intrapericardial and RA pressure | |
| LV systolic and aortic pressures may be normal or reduced | |
| Documenting that pericardial aspiration is followed by hemodynamic improvement ** | |
| Detection of coexisting hemodynamic abnormalities (LV failure, constriction, pulmonary hypertension) | |
| Detection of associated cardiovascular diseases (cardiomyopathy, coronary artery disease) | |
| RV/LV angiography | Atrial collapse and small hyperactive ventricular chambers |
| Coronary angiography | Coronary compression in diastole |
* Jugular venous distention is less notable in hypovolemic patients or in “surgical tamponade.” An inspiratory increase or lack of fall of pressure in the neck veins (Kussmaul sign), when verified by tamponade or after pericardial drainage, indicates effusive-constrictive disease.
† Heart rate is usually greater than 100 beats per minute but may be lower in patients with hypothyroidism or uremia.
‡ Pulsus paradoxus is defined as a drop in systolic blood pressure greater than 10 mm Hg during inspiration, while diastolic blood pressure remains unchanged. It is easily detected by simply feeling the pulse, which diminishes significantly during inspiration. Clinically significant pulsus paradoxus is apparent when the patient is breathing normally. When this sign is present only in deep inspiration, it should be interpreted with caution. The magnitude of pulsus paradoxus is evaluated by sphygmomanometry. If pulsus paradoxus is present, the first Korotkoff sound is not heard equally well throughout the respiratory cycle, but only during expiration at a given blood pressure. The blood pressure cuff is therefore inflated above the patient’s systolic pressure. Then it is slowly deflated, while the clinician observes the phase of respiration. During deflation, the first Korotkoff sound is intermittent. Correlation with the patient’s respiratory cycle identifies a point at which the sound is audible during expiration but disappears when the patient breathes in. As the cuff pressure drops further, another point is reached when the first blood pressure sound is audible throughout the respiratory cycle. The difference in systolic pressure between these two points is the clinical measure of pulsus paradoxus. Pulsus paradoxus is absent in tamponade complicating an atrial septal defect and in patients with significant aortic regurgitation.
§ Occasional patients are hypertensive, especially if they have preexisting hypertension.
|| Febrile tamponade may be misdiagnosed as septic shock.
¶ Right ventricular collapse can be absent in elevated right ventricular pressure and right ventricular hypertrophy or in right ventricular infarction.
** If after drainage of the pericardial effusion, the intrapericardial pressure does not fall below atrial pressure, effusive-constrictive disease should be considered.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here