Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Pericarditis and myocarditis have chest pain, laboratory results, and ECG findings that can mimic acute myocardial infarction (AMI). When the diagnosis is not clear, early coronary angiography definitively identifies or excludes AMI.
Acute pericarditis is treated with aspirin or nonsteroidal antiinflammatory drugs. Adding colchicine decreases the rate of recurrence.
Cardiac tamponade presents many ways. Patients with dyspnea, distended neck veins, hypotension, and muffled heart sounds should have the diagnosis made by bedside ultrasound. Pericardiocentesis is therapeutic and may help establish the etiology.
Myocarditis should be considered in any patient with viral illness symptoms and chest pain.
Patients with newly diagnosed hypertrophic cardiomyopathy should avoid strenuous exertion until evaluated by a cardiologist. Beta-blockers are the mainstay of therapy; nitrates should be avoided.
Dilated cardiomyopathy presents with symptoms of heart failure or rhythm disturbance.
Many cardiomyopathies have genetic origins. Send patients for both cardiology and genetics follow-up.
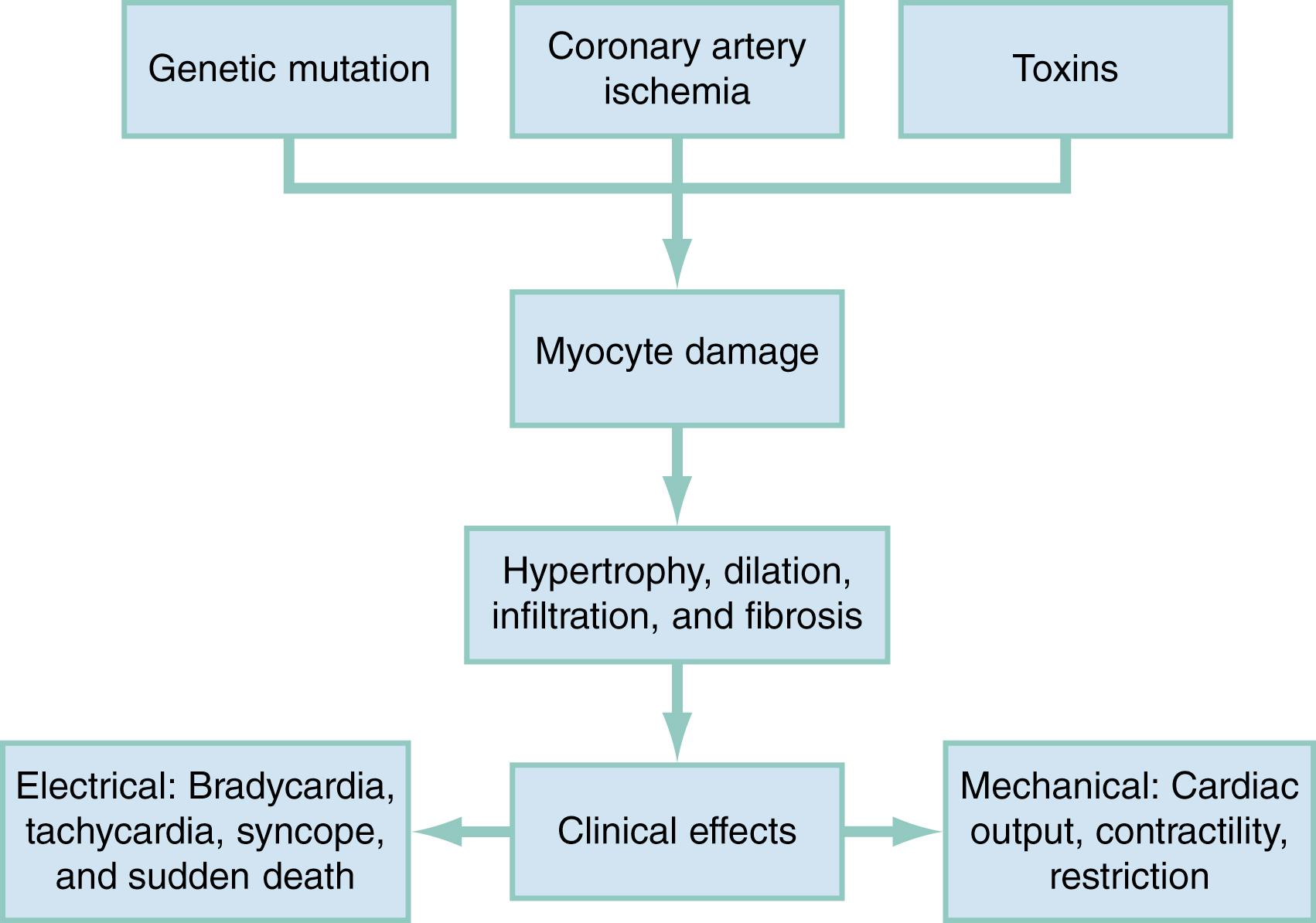
Cardiomyopathies are a heterogeneous group of diseases associated with myocyte injury, which leads to ventricular hypertrophy, fibrosis, or dilation. Clinical symptoms result from the underlying pathology.
Myocytes can be injured by genetic and nongenetic factors ( Fig. 68.1 ). More than one thousand mutations in numerous genes have been identified as etiologies. When injury occurs, pathophysiologic pathways are activated that include neurohumoral factors, immune factors, and cytokines. These cause myocyte dysfunction followed by remodeling, involving either hypertrophy or dilation. There is an increase in interstitial fibrosis, which impairs ventricular filling and leads to increased metabolic demand. At a cellular level, these may involve the troponin complex, intracellular concentration of calcium, myocardial subproteins, or the sarcomere. There is an alteration in the myocyte’s ability to contract, and the electrophysiology is altered. The correlation between genotype, phenotype, nongenetic factors, and clinical presentation is not known.
Dilated cardiomyopathy (DCM) is characterized by ventricular dilatation and decreased contractility. Clinical DCM is the final common response to many genetic and non-genetic processes. The incidence of DCM is estimated to be 35 cases per 100,000 persons. The true incidence is probably underestimated because many asymptomatic cases are undiagnosed. DCM occurs in adults and children. Up to one-third of cases are caused by mutation of genes encoding the cytoskeleton, sarcomere, and nuclear envelope proteins. Secondary neurohumoral changes contribute to remodeling and ongoing myocyte damage. Many of the genetic causes are mutations coding for the protein titan, which provides passive force and regulates sarcomere contraction and signaling. The histology is irregular myocyte hypertrophy with fibrosis or necrosis, which leads to muscle changes. The result is impaired myocardial force generation, which initiates a vicious cycle that increases the burden on the remaining cells and leads to increased stress, more work, and more cell death. This results in the clinical symptoms of heart failure or dysrhythmias.
Symptoms have an insidious onset. Heart failure occurs as the initial manifestation in 75% of adults, with dyspnea (usually with exertion or while supine) being the major symptom. Chest pain, peripheral edema, dysrhythmias, syncope, and sudden death also occur. Rhythm disturbances implicate conduction system involvement. Shortness of breath and fluid overload are the most common presentations in children.
Abnormal ventricular contractility defines DCM, and an ejection fraction less than 45% is required for diagnosis. Echocardiography shows left ventricular (LV) dilation, reduced systolic function, and variable wall motion abnormalities. End-diastolic and systolic volumes are increased by more than two standard deviations from normal. Pulmonary arterial line placement is often performed, revealing elevated pulmonary capillary wedge pressure and central venous pressures.
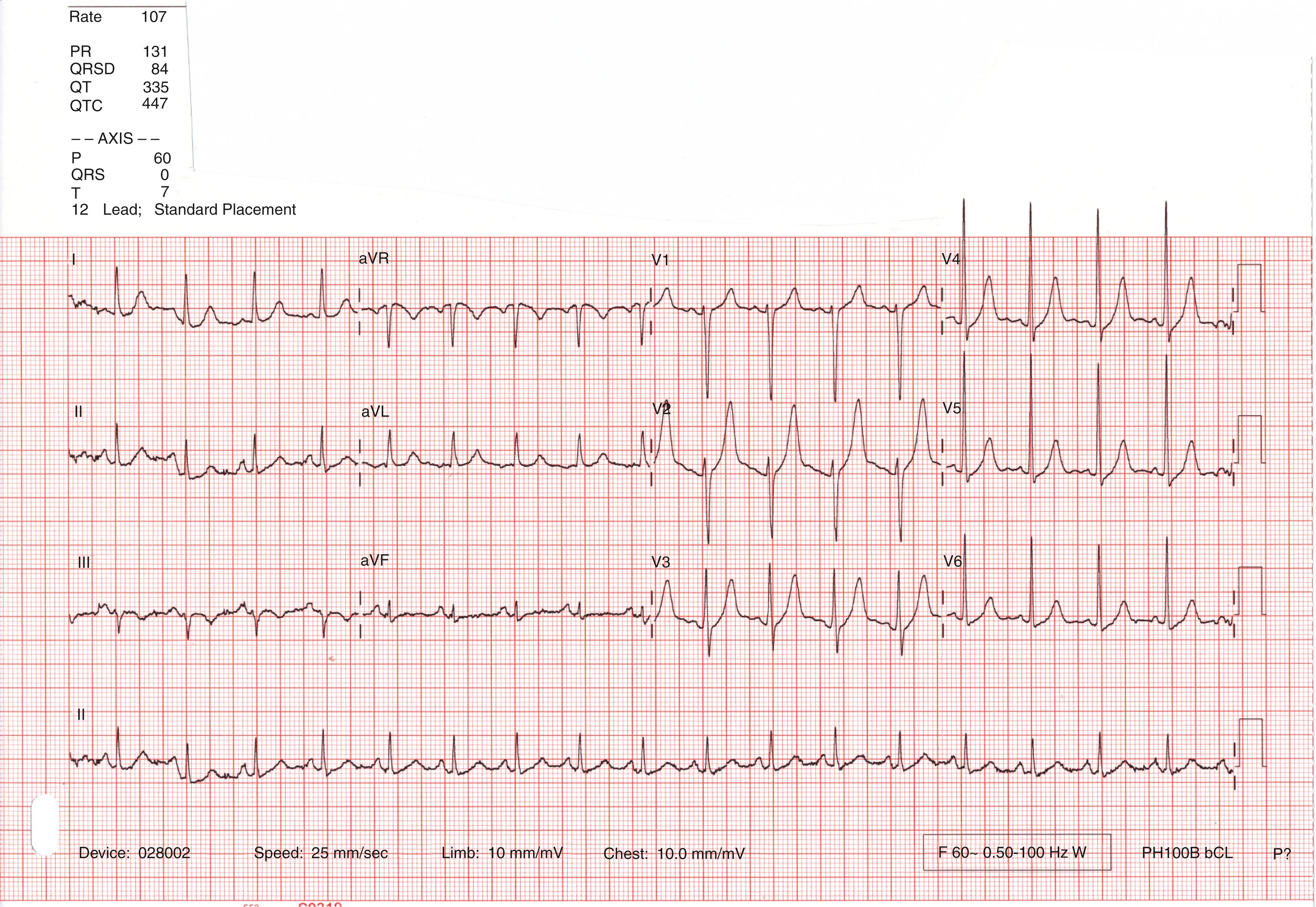
Electrocardiogram (ECG) findings are nonspecific and may include poor R wave progression, intraventricular conduction delay, left bundle branch block or left ventricular hypertrophy ( Fig. 68.2 ) with or without repolarization changes. Ectopy is common. Chest x-ray shows cardiomegaly and possible pulmonary congestion. Troponin and BNP may be elevated.
MRI showing delayed gadolinium enhancement can indicate necrosis or scarring. Right-sided failure can develop from primary right-sided disease, persistent volume overload from left-sided disease, or may represent occult atrial septal defect. The echocardiogram or ventricular imaging during coronary angiography evaluate for right-sided pathology as well. For definitive diagnosis, endomyocardial biopsy may be necessary, although histologic abnormalities are nonspecific.
The most common nongenetic cause of DCM is ischemic cardiomyopathy caused by coronary artery disease. Other etiologies include toxins such as alcohol, cocaine, methamphetamine, chemotherapy with anthracycline related agents, hemochromatosis, pseudoephedrine, ephedra, phenothiazines, lithium, anabolic steroids, clozapine, and hydroxychloroquine.
Treatment is supportive to reduce heart failure, rhythm disturbances, and conduction system issues. Supportive measures include adequate rest, weight control, abstinence from tobacco, moderate salt and ethanol consumption, and structured physical activity.
Medical treatment includes diuretics and vasodilators for patients who present with dyspnea or hypoxemia. We recommend starting with intravenous furosemide at one to two times the patient’s baseline dose, or 10 mg if diuretic naïve. For vasodilators, nitroglycerine can be initiated at 5 mcg/minute and titrated upward, closely monitoring blood pressure and symptoms.
To improve long-term outcomes and reduce heart failure exacerbations, patients should be on a closely monitored heart failure regiment. For patients with mild exacerbations who may be discharged, the emergency clinician might initiate or titrate a medication in coordination with the patient’s cardiologist. Angiotensin-converting enzyme inhibitors (ACEIs), beta-blockers, and angiotensin receptor blockers (ARBs) have mortality benefit. Among beta-blockers, carvedilol (starting dose 3.125 mg BID, titrated up to target dose 25 mg BID) is well studied and has proven effective. , ARB treatment produces beneficial LV remodeling in nonischemic dilated cardiomyopathy. Maximal beneficial remodeling is achieved within 12 to 16 months. Newer treatments include angiotensin receptor-neprilysin inhibitor valsartan/sacubitril, which has shown early promise in the management of dilated cardiomyopathy. Experimental treatments include sinoatrial modulator ivabradine and autologous somatic tissue-derived stem cells transplanted into a DCM heart to stimulate native regeneration. Both have shown limited clinical promise to date.
Biventricular pacing to coordinate both ventricles can help clinical function. ICD therapy reduces all-cause and cardiovascular mortality in patients with nonischemic cardiomyopathy. Current guidelines call for implantable cardioverter-defibrillator (ICD) placement when ejection fraction (EF) is less than 35%. Patients with EF less than 35% who have not had electrophysiologic evaluation should be referred for consideration of ventricular synchronization pacing. A search for a more precise way to predict the need for an ICD is ongoing.
Patients with DCM show progressive deterioration. There is an increased risk of sudden death, especially with some genetic types. Because medical therapy usually fails, with a mortality of 20% by 3 years, DCM is the leading indication for cardiac transplantation.
Emergency department (ED) patients should be admitted if they have new rhythm disturbances that cannot be adequately controlled with oral medications, deterioration of cardiac function as determined by symptoms or diagnostic data, shock, or significant pulmonary fluid overload that cannot be reversed. Patients whose DCM is first diagnosed in the ED should be hospitalized unless the symptoms are mild and a cardiology consult is available in the ED. If the emergency clinician can control or convert atrial rhythm disturbances, obtain sequential testing for mildly elevated troponin levels, and control symptoms in patients with known DCM, discharge is possible. Medication adjustments, such as increased diuretic dose or adding a beta-blocker or ACEI, should be considered, but follow-up within 48 hours is indicated. Heart failure clinics have been shown to reduce morbidity and mortality, making them an excellent option if available locally. Otherwise, the patient should be referred to a cardiologist.
Hypertrophic cardiomyopathy (HCM) is one of the first diseases understood at the genetic, anatomic, pathophysiologic, and clinical levels. It is the most commonly inherited cardiac disease. HCM is caused by sarcomere gene mutations that result in left ventricular hypertrophy and scar formation, which then lead to dysrhythmias and heart failure. It is inherited as autosomal dominant, but genotype-phenotype correlations have been inconsistent. More than 200 mutations have been identified. Mutations for beta-myosin heavy chain and myosin binding protein C are the two most common genetic defects, but 40% of cases have no genetic abnormality identified to date. The mutations lead to abnormal cardiac proteins that affect actin-myosin bridging, stroke power, sensitivity to adenosine triphosphate (ATP), and sensitivity of the muscle to calcium. To compensate for abnormal protein, the muscle hypertrophies and fibroses. The combination of myocyte disarray, hypertrophy, and fibrosis leads to the clinical manifestation of dysrhythmias and heart failure. Nongenetic factors such as protein translation and the environment also affect phenotype and clinical outcomes. Specific mutations correlate with sudden cardiac death. Only half of patients with Arg403Gln mutation survive past age 45 years.
The defining anatomic feature of HCM is a hypertrophied left ventricle in the absence of another cause. The ventricular cavities are small or normal, and atrial dilation is common. Hypertrophy is asymmetrical and generally most severe at the base of the intraventricular septum near the aortic valve. Left ventricle (LV) outflow is obstructed in one-third of cases and can be provoked, or dynamic, in one-third more. Histology is myocyte hypertrophy and disarray with interstitial fibrosis. The myocytes are enlarged with bizarre shapes and pleiotropic nuclei. The abnormal cells are widely distributed with a preference for the intraventricular septum.
Although HCM occurs at all ages, most patients are diagnosed between ages 30 and 40 years. Approximately 2% of cases are diagnosed in children younger than 5 years old, and 7% are diagnosed before 10 years old.
Presentation varies widely. Common symptoms are a result of diastolic dysfunction (exertional dyspnea, orthopnea, peripheral edema), LV outflow obstruction (exertional dyspnea or syncope), imbalance between myocardial oxygen supply and demand (chest pain caused by decreased blood flow into the coronary arteries), and dysrhythmias (palpitations, atrial fibrillation in 20%, ventricular tachycardias, and sudden death). Physical examination shows a displaced left precordial impulse, strong peripheral pulses, a harsh midsystolic grade 3 to 4 over 6 murmur loudest between the apex and the left lower sternal border. The Valsalva maneuver or changing from standing to squatting changes the murmur. Often, the first presentation is sudden death, which most commonly occurs during periods of exertion.
In the ED, the diagnosis should be considered in anyone with a family history, characteristic murmur, and cardiopulmonary symptoms not explained by another condition.
In individuals with murmurs, HCM may be confused with valvular diseases or a ventricular septal defect. In the absence of a murmur, symptoms may suggest mitral valve prolapse, primary pulmonary hypertension or coronary artery disease.
The ECG is abnormal in approximately 90% of patients. The most common abnormalities are atrial fibrillation, left ventricular hypertrophy, ST segment alterations, T wave inversion, left atrial enlargement, abnormal Q waves, and diminished or absent R waves in the lateral leads. The chest radiograph may be normal or may show left ventricular or atrial enlargement.
Echocardiography findings include asymmetrical left ventricular hypertrophy, left ventricular outflow tract narrowing, a small left ventricular cavity, and reduced septal motion. Unlike fixed ventricular flow obstructions, the amount of obstruction imaged changes from beat to beat. This dynamic characteristic of HCM distinguishes it from the discrete forms of obstruction. Left ventricle obstruction at rest is an independent predictor of heart failure.
MRI can help with diagnosis and prognosis. It provides an accurate measure of wall thickness and identifies the location and extent of fibrosis. Extensive late gadolinium enhancement, which represents scarring, is associated with sudden death. MRI for HCM is rarely available to the EP and should be used by the cardiologist to manage long-term care.
Beta-blockers and calcium channel blockers are used to control exercise-related symptoms. A beta-blocker such as metoprolol 12.5 mg BID and titrate up to 100 mg BID is the first-line treatment. For those intolerant of beta-blockers, diltiazem 30 mg QID is indicated. Disopyramide (100 to 150 mg Q6H) is often added to reduce resting gradient across the aortic valve. These treatments do not alter the natural history of disease. Spironolactone is not efficacious. ARB agents are being studied. A small model inhibitor of myosin MYK-461 has been effective in preventing HCM in mouse models. Nitroglycerin should be avoided in HCM-associated chest pain because it decreases ventricular volume, exacerbating outflow tract obstruction.
Atrial fibrillation can cause marked hemodynamic compromise and severe CHF. Cardioversion and rate control should be attempted. If the patient is stable, start with diltiazem 0.25 mg/kg (based on weight) or esmolol 50 mcg/kg/min. These doses may be repeated or an infusion started. If not successful at rate control (HR < 100) or the patient is hypotensive (BP < 90 systolic), then electrical cardioversion is indicated. For HCM patients with atrial fibrillation, the risk of stroke is high, and anticoagulation (rivaroxaban 20 mg QD, apixaban 5 mg BID, or dabigatran etexilate 150 mg BID) should be started from the ED. Amiodarone (600 mg QD divided into 2 or 3 doses) is the drug of choice long term to treat atrial fibrillation as well as ventricular dysrhythmias. Phenylephrine is the vasopressor of choice for persistent hypotension when intravenous fluids have not improved blood pressure. Beta-agonists, such as dobutamine, increase the gradient across the outflow tract obstruction and should be avoided.
Invasive procedures, such as myomectomy or septal alcohol ablation, may be needed to treat severe cases. Dual-chamber pacing decreases outflow gradient and improves symptoms but does not improve mortality.
Implantable cardioverter-defibrillators are indicated for patients who survive sudden cardiac death (SCD) or have certain risk profiles. Risk calculation is based on patient age, family history of one or more first-degree relatives with SCD at age less than 40 years or with confirmed HCM, size of left ventricle and atrium, left ventricle outflow gradient, presence of ventricular tachycardia, and unexplained syncope. These factors all reflect the extent of interstitial fibrosis.
A patient with angina, syncope, near-syncope, dysrhythmias, and abrupt hemodynamic changes should be hospitalized. A patient suspected to have HCM should be seen by cardiology in the ED or hospitalized to expedite making the diagnosis.
The natural history of HCM is variable and probably reflects the many different genetic causes. The clinical course is either heart failure, atrial fibrillation, or sudden death. With modern therapy, most patients have low premature mortality.
The evidence for the role of exercise is unclear. Newer evidence shows that the risk of SCD may be lower than previously reported, and moderate or recreational exercise may be beneficial. HCM remains the most frequent cause of nontraumatic sudden death in the young, however, and some still recommend that patients not participate in competitive sports. Current evidence is unclear about which athletes benefit from SCD risk screening. Given the current evidence, emergency physicians should advise their discharged patients to eliminate strenuous exercise until cardiology evaluation.
Restrictive cardiomyopathy (RCM) is a gradual and progressive limitation of ventricular filling secondary to myocardial infiltration. There is stiffness of the ventricles with normal diastolic volume and normal ventricular wall thickness. RCM is the least common type of cardiomyopathy, accounting for less than 5% of all cases. The most common etiology in the United States is amyloidosis. Other causes include sarcoidosis, hemochromatosis, scleroderma, neoplastic cardiac infiltration, glycogen storage disorders, Fabry disease, Gaucher disease, and mutations related to myocardial muscle proteins.
Restriction of ventricular filling and compliance results in low ventricular volumes, high end-diastolic ventricular pressures, and decreased cardiac output. Systolic function is maintained. Endomyocardial biopsy shows interstitial fibrosis and myocyte hypertrophy, neither of which is pathognomonic. There are no specific genetic mutations. Grossly, there is atrial enlargement and small ventricles. As the disease progresses, the ventricular cavities may become obliterated by fibrous tissue, scarring, or thrombus.
Most patients are diagnosed in their 50s. The most common symptom is dyspnea. Signs and symptoms may include exercise intolerance (cardiac output cannot be increased because ventricular filling is compromised), pulmonary congestion, elevated central venous pressure, peripheral edema, pulmonary edema, and S 3 and S 4 gallops. Clues to making the diagnosis in children include trouble eating, lack of weight gain, and difficulty exercising.
Serum troponin and chemistry panel should be obtained to exclude myocardial infarction or renal and hepatic mimics. Because some causes of RCM can be treated or managed, it is important to search for etiologic causes, which requires extensive serologic testing and cardiac MRI. These can be performed as an inpatient or outpatient. Comprehensive echocardiography should be obtained to confirm the diagnosis.
ECG findings are not specific. Chest x-ray shows cardiomegaly and often pulmonary congestion. BNP may be elevated. Point of care ultrasound (POCUS) shows thickened ventricles and no change in isovolemic relaxation time with respiration. Central venous pressure tracings show rapid filling causing a sudden dip in pressure and then a sudden plateau, yielding a curve similar to a square root sign. CT can also differentiate RCM from constrictive pericarditis. Pericardial calcification or history of radiation therapy favors constrictive pericarditis over RCM.
Most of the underlying causes of RCM are untreatable, with the exception of hemochromatosis. Patients rely on high filling pressures, making volume management challenging. A trial of beta-blockers (metoprolol 25 mg BID or atenolol 25 mg QD) or calcium channel blockers (diltiazem 30 mg QID) may help increase filling time and thus cardiac output. ACEIs (lisinopril 10 mg QD) and ARBs (losartan 50 mg QD) may also help. Patients with RCM should be maintained in sinus rhythm because they are dependent upon the atrial contribution to cardiac output. RCM is relentless, with 64% of patients dying within 5 years of diagnosis.
Patients should be hospitalized for shortness of breath or hypotension that does not respond to ED treatment. Patients with either tricuspid regurgitation or small LV end-diastolic volume have a poor prognosis.
Peripartum cardiomyopathy (PPCM) was first described in 1849. It is a rare, often dilated, cardiomyopathy with systolic dysfunction that presents in late pregnancy or the early postpartum period. Although the condition is prevalent worldwide, women with African ancestry seem to be at greatest risk even after confounders are eliminated. Other risk factors include preeclampsia, advanced maternal age, tocolytic use, twins, obesity, and cocaine use. The incidence is thought to be between 1 in 1500 and 1 in 4350 live births and increasing, possibly because of improved detection or rising maternal age. The exact pathogenesis is unknown. Some women have an underlying sarcomere gene mutation.
PPCM is similar to DCM, with heart failure symptoms predominating on initial presentation. Patients have dyspnea and edema and may also have chest pain or palpitations. Unfortunately, these symptoms can be expected in normal pregnancy, making distinguishing pathology from expected physiologic changes challenging at times. Acute alterations in symptoms, sudden or worsening chest pain or dyspnea, or difficulty performing routine activities are diagnostic clues. Physical examination often reveals tachycardia, tachypnea, pulmonary rales, and an S3 heart sound.
The ECG may show LV hypertrophy or nonspecific ST-T wave changes. On echocardiography, all four chambers are enlarged, with a reduction in left ventricular systolic function.
Other causes of symptoms should be evaluated. The differential diagnosis includes preeclampsia, myocardial ischemia including coronary dissection, pulmonary embolism, pneumonia, and primary rhythm disturbance.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here