Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Paravalvular regurgitation (leak) affects 5% to 17% of all surgically implanted prosthetic heart valves.
Echocardiography is pivotal in the evaluation of both mitral and aortic paravalvular leaks.
Surgical repair is effective but has significantly higher complications rates compared with a percutaneous approach.
Percutaneous paravalvular leak closure is feasible and safe with long-term outcomes comparable to a surgical approach.
Resolution of symptoms and hemolysis as well as long-term survival depend on completeness of defect closure with either approach.
Percutaneous paravalvular closure with an Amplatzer ductal occluder device was first described in 2004 by Kort et al. in a 3–month-old infant who had undergone atrioventricular valve replacement with a 19-mm St. Jude mitral prosthesis in 2004 at 6-weeks of age. Before that the only transcatheter closing techniques described were coil occlusions and the double umbrella device.
Paravalvular regurgitation due to an abnormal communication between cardiac chambers affects 5% to 17% of all surgically implanted prosthetic heart valves (both mechanical- and bioprostheses, with an estimated 500 to 10,200 cases annually). The most common etiologies for paravalvular regurgitation are tissue friability, annular calcification, or infection; patients can present with symptoms of congestive heart failure, hemolytic anemia, or both. In this chapter, we will present appropriate assessment and indications for current percutaneous strategies to close and clinical outcomes of mitral and aortic paravalvular leaks (PVLs).
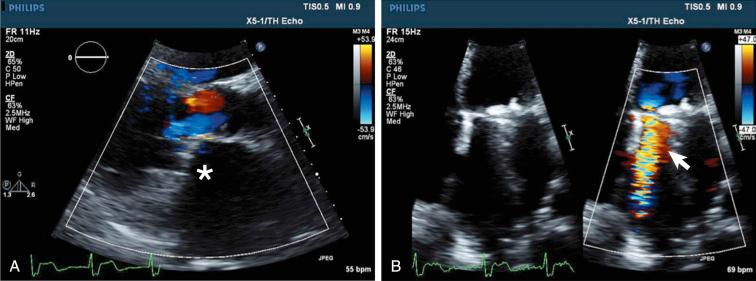
When a patient with a history of aortic- or mitral-valve replacement presents with heart failure symptoms and is found to have a new systolic (mitral PVL) or diastolic (aortic PVL) murmur, a transthoracic echocardiogram is key in the initial workup ( Figs. 62.1 and 62.2A and B ). Assessment of metallic mitral valve prostheses may be difficult due to shadowing from the prosthesis into the left atrium, which can obscure the regurgitation signal (see Fig. 62.1 ). However, a thorough Doppler assessment can help confirm regurgitation as the major culprit.
If the PVL is deemed severe and aggressive medical management fails, there are two management options: (A) surgical reoperation, which usually carries a significant mortality risk, or (B) a percutaneous closure of the PVL.
Due to an increased morbidity and mortality, reoperation is often avoided, especially if the underlying paravalvular tissue is friable (active endocarditis, steroid use) or heavily calcified. This notion is supported by Said et al., who recently reported the Mayo Clinic experience of 206 mitral reoperations for PVL between 1995 and 2015 (10% had an attempted percutaneous closure). Early mortality was 5%; survival at 1, 5, and 15 years was rather low, with 83%, 62%, and 16%, respectively. Recurrence occurred in 43 (21%) patients, and reoperation was required in 19 (9%) patients. Multivariate analysis revealed advanced New York Heart Association (NYHA) class, active endocarditis, chronic steroids, previous coronary artery bypass grafting, baseline creatinine above 1.5, postoperative need for dialysis, and residual PVL ( P < .0001) to be predictors of late mortality. Medical therapy (including standard heart failure management and repeated blood transfusions n cases with hemolysis) can help with symptoms but will not prevent progressive heart failure from volume and/or pressure overload, nor the need for the frequent administration of blood products.
The next step in the workup should include a transesophageal echocardiogram (TEE). The advantage of this approach is less shadowing of mitral valve prostheses and thus a more defined view on the location and approachability of the PVL. The two-dimensional (2D) echocardiogram is routinely combined with three-dimensional (3D) imaging, which further facilitates procedural planning ( Fig. 62.2 ).
![Fig. 62.2, Two-dimensional images (A) and 3D images (B) of the paravalvular leak (arrow) , which reveal an anteromedial location of the paravalvular leak (the interatrial septum (∗) as well as the aortic valve [AV] serve as anatomical markers for orientation purposes). The orifice of the paravalvular leak measured 7 to 8 mm in diameter. Fig. 62.2, Two-dimensional images (A) and 3D images (B) of the paravalvular leak (arrow) , which reveal an anteromedial location of the paravalvular leak (the interatrial septum (∗) as well as the aortic valve [AV] serve as anatomical markers for orientation purposes). The orifice of the paravalvular leak measured 7 to 8 mm in diameter.](https://storage.googleapis.com/dl.dentistrykey.com/clinical/PercutaneousTreatmentofParavalvularLeak/1_3s20B9780323568142000623.jpg)
Despite a circular or oval appearance on 3D TEE, many PVLs follow a serpiginous track, rather than being cylindrical. Thus on the echocardiographic images, the left atrial orifice of the defect may appear large (see Fig. 62.2 ) but the actual size of the track may be much smaller, requiring a smaller closure device than initially estimated. The serpiginous nature of the track can also make crossing of the leak with interventional equipment difficult.
Outcomes for percutaneous PVL closure have been reported by several high-volume centers of excellence. The first reports were published by the groups at Lennox Hill Hospital New York and Mayo Clinic Rochester. Ruiz et al. reported a retrospective review of 57 percutaneous PVL closure procedures in 43 patients between April 2006 and September 2010 (38 paramitral and 11 paraaortic leaks, some with multiple procedures). Successful closure was achieved in 89% of paramitral leaks (68% biological, 32% mechanical) and 73% of paraaortic leaks (55% biological, 45% mechanical). A total of 28 out of 35 patients improved by at least one NYHA functional class (eight by two functional classes, and two by three functional classes). The percentage of patients requiring blood transfusions and/or erythropoietin injections postprocedure decreased from 56% to 5%. The survival rates for patients at 6, 12, and 18 months after PVL closures were 91.9%, 89.2%, and 86.5%, respectively. These data showed that percutaneous repair of aortic and mitral valve PVLs is a viable option, but mortality in this high-risk patient population (mean Society for Thoracic Surgeons (STS) risk score 6.7 ± 5.48) remains high with up to >35% after 3 years. These data demonstrate the potential for durable clinical efficacy, which is dependent on residual regurgitation.
Sorajja et al. published the first prospectively collected acute and 30-day outcome data of 115 patients who underwent PVL closure at Mayo Clinic from February 2004 to September 2010 (15% of patients had multiple defects). Using strict criteria, closure was successful (i.e., ≤1+ residual regurgitation) in 77% of patients. The 30-day complication rate was 8.7% (sudden and unexplained death, 1.7%; stroke, 2.6%; emergency surgery, 0.9%; bleeding, 5.2%). Two devices embolized during the procedure and were retrieved without sequelae. No procedural deaths occurred, but two (1.7%) patients died by 30 days. The 3-year follow-up data (median follow-up was 11 months, range: 1 to 85 months) of the entire cohort demonstrated a 3-year estimate for survival free of all-cause mortality of 64.3%. Survival did not significantly differ for patients with no (69.9%), mild (62.4%), or moderate or severe (58.1%) residual regurgitation, before and after multivariate adjustment. The 3-year estimate of survival free of death or need for cardiac surgery was 63.7%, 59.0%, and 30.9% ( P = .01), respectively. Symptom improvement was confined to those patients who had no or only mild residual regurgitation after PVL closure, indicating near complete closure should be the goal of therapy. Hemolytic anemia was difficult to resolve and persisted in 14 of 29 survivors without an obvious relation between the degree of residual regurgitation and incidence of persistent hemolytic anemia.
More recently, Smolka et al. showed the importance of completeness of PVL closure in the setting of hemolysis. In a limited study group of 116 patients (roughly 43% had signs of hemolysis), percutaneous PVL closure effectively reduced hemolysis if at least 90% reduction of PVL cross-sectional area was achieved.
Persistent hemolytic anemia has been shown to be an independent predictor for poor survival and need for cardiac surgery. Recently the Mayo Clinic authors updated their long-term outcome data, with a median follow-up length of 2.04 (0.7 to 4.9) years. The longer follow-up led to a more differentiated outcome dependent on completeness of PVL closure. Alkhouli et al. showed that of 231 patients with percutaneous PVL repair between 2006 and 2017, survival at 3 years was 61% in patients who had ≤ mild residual PVL and 47% in patients with higher grade of residual PVL ( P = .002). Repeat surgical interventions were also more likely in patients with >mild residual PVL (6% vs. 17%; P = .004).
Similar results have been reported by Calvert et al., a combined UK and Ireland center of excellence experience (20 centers, 2004–15, 259 patients, median follow-up of 110 [7 to 452] days). The one factor independently associated with major adverse cardiovascular events (MACE) was again the degree of persisting leak (>mild) at follow-up (hazard ratio [HR] 3.01; P = .002). In the Spanish HOLE registry (SpanisH real wOrld paravalvular LEaks closure), 469 patients at 19 centers were included. The use of the Amplatzer III device (not available in the United States) and operator experience were independent predictors of procedural success in mitral PVLs; for aortic PVLs the only predictor of procedural success was the leak size (≥10 vs. <10 mm). The overall major adverse events rate (death or emergency surgery or stroke) at 30 days was 5.6%; the only predictor for combined adverse events was NYHA functional class IV (HR 4.2 [1.42 to 12.34]; P = .009).
Redo percutaneous PVL closure is associated with high complication rates. A limited study by Al-Hijji analyzed 16 redo PVL procedures at Mayo Clinic and compared them to successful first attempt procedures. Procedural success was 75% (vs. 85% in the 1st attempt group); 30-day MACE was 12.5% in the redo group, compared with 4.2% in the age- and-sex matched patients who underwent PVL closure for the first time ( P = .35).
While various devices for PVL closure have been used, none of these are U.S. Food and Drug Administration approved for this purpose. Commonly used devices off-label include the Amplatzer Septal Occluder, Duct Occluder, and Muscular Ventricular Septal Defect Occluder (St. Jude Medical, St. Paul, MN). These devices are woven from a larger-caliber nitinol mesh, resulting in a stiffer device with a higher profile, and may be associated with a greater risk of accelerating hemolysis. Our lab still prefers the Vascular Plug II as the primary device for PVL closure.
There are multiple reports comparing long-term results of transcatheter closure versus surgical repair. Zhang et al. analyzed outcomes of 87 consecutive patients with symptomatic PVL who received either transcatheter ( n = 46) or surgical ( n = 41) treatment at Shanghai Chest Hospital between January 2009 and December 2015. Transcatheter closure was safer (MACE 56% vs. 17%; P < .001) and more cost-effective, but 13% of cases showed aggravation of hemolysis. The overall 5-year survival rates after transcatheter and surgical repair, however, were similar at 74% and 72%, respectively ( P = .45).
Wells et al. analyzed 114 patients (56 underwent transcatheter closure, 58 underwent surgical repair) from 2017 to 2016. There were no differences in the primary end point of a composite of death, reintervention for PVL, or readmission for congestive heart failure–related symptoms at 1 year (33.9% vs. 39.7%; P = .53) or 1-year survival (83.9% vs. 75.9%; P = .28). The largest data set come from the Mayo Clinic. Alkhouli et al. analyzed 381 patients who underwent transcatheter ( n = 195) or surgical ( n = 186) treatment of mitral PVL between 1995 and 2015. Technical success was higher (95.5% vs. 70.1%; P < .001), and in-hospital death occurred less often in the surgical group (3.1% vs. 8.6%, P = .027). After risk adjustment, however, there was no significant difference in long-term survival between patients who underwent surgical versus transcatheter treatment of PVLs.
Angulo-Llanos reviewed a one-center experience of percutaneous PVL procedures between years 2008 and 2014 ( n = 51) and compared them to a cohort of surgical patients ( n = 36) with a mean follow-up 785 days. There were no differences in survival free from the composite end point (death from all causes and need for hospital readmission due to cardiovascular causes), according to the treatment received. However, after propensity score analysis in-hospital mortality was higher in the surgical group (30.6% vs. 9.8%, odds ratio 6, P = .01) and clinical improvement was higher in the percutaneous group (71.4% vs. 36.4%, P = .002).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here