Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Mitral regurgitation (MR) is a significant problem, and the number of patients with MR is growing together with increased numbers of patients with congestive heart failure.
Various percutaneous approaches to mitral valve repair are under preclinical and clinical investigation and show promise. These approaches are predominantly based on established surgical strategies and include edge-to-edge repair, direct and indirect annuloplasty methods, and chamber remodeling strategies.
In recent years, transcatheter mitral valve replacement techniques have gained a foothold with promising results from early trials of new devices.
Replacement of degenerated surgical bioprosthetic valves using the valve-in-valve (ViV) approach of placing a transcatheter aortic valve prosthetic within a degenerated mitral ring or valve is a safe and effective strategy for many patients.
Placement of a transcatheter aortic valve prosthetic in a patient with a degenerated mitral valve ring (valve-in-ring) carries greater risk than the ViV procedure due to valve embolization and left ventricular outflow tract obstruction
Different percutaneous techniques provide specific advantages, depending on the anatomic and functional characteristics of MR. Selection of the appropriate technique or techniques for each patient will ultimately determine the success of these emerging technologies.
Integration of established imaging modalities both in and out of the catheterization laboratory is critical for the safety and efficacy of percutaneous repair technologies. The development of emerging imaging modalities will likely play a role in the future of percutaneous technologies.
Chronic mitral regurgitation (MR) poses a significant public health burden, with more than 3 million people in the United States alone suffering from moderate or severe MR. Left untreated, chronic MR results in heart failure symptoms, left ventricular (LV) cavity dilatation and systolic dysfunction, enlargement of the left atrium (LA), atrial fibrillation, and pulmonary hypertension. Medical therapy may provide some relief from symptoms and is necessary to treat ischemic heart disease and heart failure in patients with MR complicating these underlying disease states. However, there has been no proven benefit of these treatments in MR itself. Therefore, surgical correction with mitral valve repair (MVRe) or replacement remains the mainstay of therapy for chronic MR. However, because of the invasive nature of open-heart surgery and the frequent presence of comorbidities in this group, up to 50% of patients with severe MR may not be offered surgery. This is especially true for older patients and those with impaired LV function. Further, the role of surgery in patients with functional MR (FMR) remains unproven.
As a result, percutaneous technology is poised to significantly alter the treatment paradigm for chronic MR. Percutaneous MVRe offers the potential benefits of decreased morbidity, improved recovery time, and shorter hospital stay compared with open-heart surgery. Current percutaneous options are loosely based on surgical repair techniques, with four primary methods to accomplish a reduction in MR: (1) edge-to-edge (E2E) leaflet repair, (2) indirect annuloplasty via the coronary sinus, (3) direct annuloplasty, and (4) septal-lateral (SL) annular cinching. Percutaneous MVRe has gained significant traction over the past few years, with a number of devices in pre- and clinical stages demonstrating early feasibility. However, despite these early successes, no device has yet achieved commercial approval. Overall, the field is very exciting, with promising initial studies highlighting the need for a better understanding of patient selection for appropriate management of MR.
The mitral valve (MV) complex is composed of the mitral annulus, anterior and posterior leaflets, chordae tendineae, and papillary muscles. The mitral annulus is the elliptical area of attachment of the MV to the base of the LA ( Fig. 52.1 ). The posterior leaflet has three lobes or “scallops”: the lateral (P1), central (P2), and medial scallops (P3). The anterior leaflet scallops are named A1, A2, and A3, respectively, corresponding to the posterior scallops. The anatomic position of the valve is such that the two leaflets meet at the anterolateral and posteromedial commissures. Chordae connect both the leaflets and the anterolateral and posteromedial papillary muscles. The primary chordae connect to the free edge of the leaflet; the secondary chordae (“strut” chords) are thicker and connect to the rough zone of the leaflet; and the tertiary chordae are short and connect the basal zone of the leaflet to the ventricular free wall.
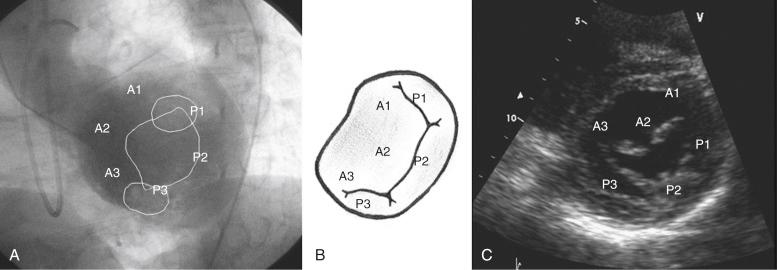
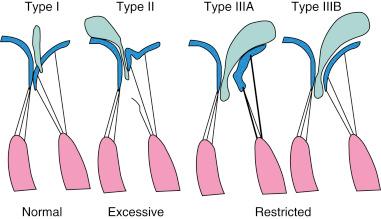
Anatomic or functional abnormalities of any of the structures in the MV apparatus may lead to MR. The disease process leading to MR may be primary MV disease, secondary regurgitation resulting from another cardiac disease, or MV involvement in a systemic inflammatory disease ( Table 52.1 ). Various terminologies are used to characterize the mechanisms of MR. The morphologic description, proposed by Carpentier, classifies the mechanism of regurgitation according to leaflet pathophysiology ( Fig. 52.2 ). Type I regurgitation occurs in the presence of normal leaflet motion and is usually caused by annular dilatation or leaflet perforation. Type II is caused by leaflet prolapse, which is commonly the result of degenerative (myxomatous) disease, chordal elongation or rupture, or papillary muscle elongation or rupture. Type III is caused by restricted leaflet motion, which may arise from posterior wall motion abnormality or papillary muscle dysfunction due to ischemic cardiac disease. The restricted leaflet motion may also be caused by commissural fusion, leaflet or chordal thickening from rheumatic heart disease, or both. This simplification has usefulness in terms of both the surgical approach and the percutaneous approach, because the goal of therapy is to restore normal leaflet function but not necessarily normal valve anatomy.
|
Another common method of categorizing MR is based on the etiology and mechanism of the MR. This classification is commonly used in the literature to study the clinical outcomes of patients ( Table 52.2 ). In this classification scheme, MR is loosely categorized on the basis of an abnormal or normal MV as degenerative or rheumatic disease (primary MR) and functional or ischemic disease (secondary MR) , respectively. However, the terms functional MR , ischemic MR , and secondary MR are often used interchangeably and may represent many different mechanisms and morphologic variants.
| Type I: Normal Leaflet Motion |
| Annular dilatation Dilated cardiomyopathy—“functional MR” a Leaflet perforation Annular calcification |
| Type II: Leaflet Prolapsed |
| Chordal rupture or flail leaflet Chordal elongation—“degenerative MR” Papillary muscle elongation Papillary muscle rupture |
| Type III: Restricted Leaflet Motion |
| IIIA: Fibrosis of the subvalvular apparatus—“rheumatic MR” IIIB: Regional left ventricular remodeling or wall motion abnormality—“ischemic MR” |
a The term “functional MR” is sometimes used to describe both ischemic and nonischemic MR because they share the characteristics of left ventricular geometric remodeling and annular dilatation with normal leaflet morphology.
Degenerative disease includes Barlow disease (myxomatous degeneration) and fibroelastic deficiency, both of which can result in MV leaflet prolapse and degenerative MR (DMR). Fibroelastic deficiency is the most common etiology (approximately 70%) among patients undergoing surgical MVRe in the United States. MR in rheumatic disease is a result of leaflet deformity caused by severe thickening and calcification of the leaflets and subvalvular apparatus and apical leaflet doming resulting in malcoaptation. Although this is a common cause of mitral disease worldwide, it is less frequently encountered in the United States.
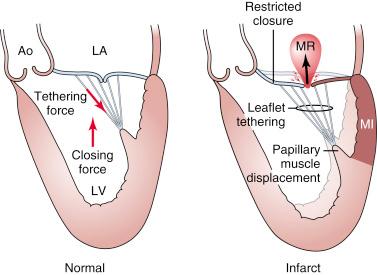
FMR occurs in the setting of LV dysfunction and is seen in patients with coronary artery disease (ischemic MR) or with dilated cardiomyopathy from other causes. Ischemic MR results from decreased closing force and increased tethering force on the leaflets. Various factors can lessen the closing force, including diminished left ventricular ejection fraction (LVEF), LV dyssynchrony, and decreased annular contraction; similarly, many factors can increase the tethering force, including papillary muscle displacement, LV remodeling, and annular dilatation. It is becoming increasingly clear that significant interaction among ventricular, valvular, and annular factors is involved in the generation, perpetuation, and progression of so-called functional MR ( Fig. 52.3 ). More recently, investigators have demonstrated the incidence of typical FMR due to annular dilation but with normal LVEF that results instead from progressive LA enlargement.
The natural history of chronic MR depends on the degree of regurgitation, the cause of the underlying disorder, and the degree of LV dysfunction. Available data on the natural history of the disease are limited by small sample size, selection bias, inconsistent measures of MR severity, and the inclusion of disparate etiologies of regurgitation. However, it appears that many patients with chronic MR remain asymptomatic for many years. Among patients with mild MR, there is an inconsistent rate of progression to severe MR that appears to be independent of medical treatment. When chronic severe MR is present, approximately 5% to 10% of patients per year develop significant symptoms, clinical indication for surgery, death, or all of these.
Whether there is a small risk of sudden cardiac death in patients with severe asymptomatic MR remains controversial, and the data are not compelling enough to subject patients to intervention for this reason alone in the absence of other indications for MVRe. The importance of symptoms in regard to long-term prognosis is demonstrated by the high mortality rate reported for patients with New York Heart Association (NYHA) class III or IV symptoms, even in degenerative MV disease. LVEF is also an important independent predictor of outcome in patients with FMR.
Patients with degenerative MV disease have a favorable long-term prognosis whether they are treated conservatively or, when indicated, surgically. However, patients with degenerative MV disease and coronary disease are fundamentally different from those with degenerative disease alone; the former have a worse prognosis that is dominated by the contribution of coronary disease. Conversely, in the absence of degenerative disease, the presence and degree of MR after myocardial infarction is an independent predictor of mortality, emphasizing the need for accurate quantification; further studies are needed to investigate whether treatment of MR in these patients modifies outcomes.
Echocardiography is the dominant modality for imaging the MV and for the assessment of MR severity. Two-dimensional transthoracic echocardiography (TTE) is useful to evaluate the valvular anatomy, to determine the structure and function of the LV, and to assess the origin and degree of regurgitation. Therefore, TTE provides an understanding of MR severity as well as an insight into the primary or secondary nature of the disease. Furthermore, the anatomic assessment of the MV and the LV also allows determination of whether surgical or percutaneous MVRe may be feasible or whether valve replacement will be necessary. Longitudinal data collected by serial echocardiography may be used to determine the timing of intervention and to follow up the results. If transthoracic images are not adequate, transesophageal echocardiography (TEE) provides an excellent assessment of MV anatomy and severity of regurgitation. This can be particularly useful to determine whether valve repair is feasible.
Evaluation of the severity of valvular regurgitation with echocardiography relies heavily on Doppler methods, including color, pulsed-wave, and continuous-wave Doppler. The American Society of Echocardiography has determined the qualitative and quantitative echocardiographic parameters that are useful in grading MR ( Table 52.3 ). The assessment of severity should rely on the integration of both quantitative and qualitative measures obtained by Doppler techniques. In addition, structural findings such as a flail leaflet or an enlarged LA can add useful information with regard to regurgitation severity.
| Mild | Moderate | Severe | |
|---|---|---|---|
| Structural Parameters | |||
| LA size | Normal | Normal or dilatated | Usually dilatated |
| LV size | Normal | Normal or dilatated | Usually dilatated |
| Mitral leaflets or support apparatus | Normal or abnormal | Normal or abnormal | Abnormal/flail leaflet/ruptured papillary muscle |
| Doppler Parameters | |||
| Color flow jet area | Small, central jet (usually <4 cm2 or <20% of LA area) | Variable | Large central jet (usually >10 cm2 or >40% of LA area) or variable size wall-impinging jet swirling in LA |
| Mitral inflow—PW | A wave dominant | Variable | E wave dominant |
| Jet density—CW | Incomplete or faint | Dense | Dense |
| Jet contour—CW | Parabolic | Usually parabolic | Early peaking—triangular |
| Pulmonary vein flow | Systolic dominance | Systolic blunting | Systolic flow reversal |
| Quantitative Parameters | |||
| Vena contracta width (cm) | <0.3 | 0.3–0.69 | ≥0.7 |
| Regurgitant volume (mL/beat) | <30 | 30–59 | ≥60 |
| Regurgitant fraction (%) | <30 | 30–49 | ≥50 |
| EROA (cm 2 ) | <0.20 | 0.20–0.39 | ≥0.40 |
Color Doppler provides a number of qualitative and quantitative means to determine MR severity. A proximal flow convergence on color Doppler is present in severe regurgitation. The proximal isovelocity surface area (PISA) of this flow convergence can be used to accurately quantitate the effective regurgitant orifice area (EROA). The width of the regurgitant jet at or just downstream from the regurgitant orifice is known as the vena contracta and is slightly smaller than the anatomic regurgitant orifice. This distinction is particularly relevant in some patients with FMR, in which the regurgitant orifice has a slit-like rather than an oval shape. The area of the MR jet occupying the LA can provide a rapid semiquantitative assessment of regurgitation severity. However, this is influenced by instrument factors (e.g., gain) and by jet orientation (e.g., a central jet may appear more severe than an equally large jet that adheres to the atrial wall). In addition, the color jet area is influenced by the driving pressure across the valve and can be enhanced by elevated blood pressure. Color Doppler imaging is also important in the parasternal short-axis view to determine the origin of the MR jet because percutaneous approaches (especially the MitraClip) are more effective in centrally originating jets than in medial or lateral ones.
Regurgitant volume can be assessed with the use of continuous-wave Doppler data. Regurgitant volume is calculated by applying the continuity equation (conservation of mass), in which left-sided regurgitation volume is calculated as the difference between Doppler-derived flows across the aortic and mitral valves. The stroke volume equals the cross-sectional area of the valve annulus, multiplied by the velocity-time integral of flow across the annulus. The regurgitant volume at the MV is calculated as the difference between stroke volumes across the MV and the aortic valve. This can also be expressed as a regurgitant fraction.
Pulsed-wave Doppler is useful to assess the effect of regurgitation on the pulmonary venous flow. A pulmonary venous flow that is blunted or reversed in systole can indicate severe regurgitation. The contour and density of the regurgitant envelope on continuous-wave Doppler is also useful: a dense, early-peaking, or triangular envelope is most consistent with severe regurgitation. In addition, the mitral inflow pattern is typically E-wave dominant (>1.2 m/s) in severe regurgitation, reflecting increased flow across the valve.
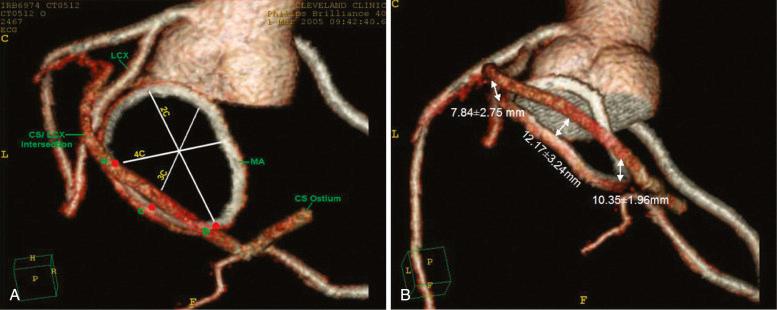
Although historically echocardiography has been the dominant imaging modality in the assessment of MR, cardiac computed tomography (CT), cardiac magnetic resonance imaging (MRI), and three-dimensional (3-D) echocardiography are beginning to play more important roles. Coronary sinus devices that indirectly alter annular geometry are under development, so the relationship of the coronary sinus to the mitral annulus is becoming increasingly important ( Fig. 52.4 ). In addition, the coronary sinus and left circumflex arteries are close to each other and overlap in more than 90% of patients, creating the potential for cinching devices to hinder coronary blood flow. Cardiac CT has the potential to provide significant anatomic details in the screening of patients and procedural planning.
With cardiac MRI, it is possible to obtain significant structural information regarding the geometry of the LV, mitral annulus, and leaflets, as well as quantitative regurgitant volumes. It is likely that regurgitant volumes calculated with the use of cardiac MRI are more accurate and more operator or reader independent compared with those derived from TTE. However, limited experience with this modality currently limits its usefulness for research purposes.
3-D echocardiography provides a more in-depth understanding of the anatomy of the MV apparatus and the changes of MR. Because the position of the MV apparatus in the LV can be disrupted in all directions, 3-D echocardiography is more likely to define the exact mechanism of MR in ischemic disease and to detect multiple jets of MR that may affect the treatment approach. Furthermore, the ability to acquire real-time 3-D images with color flow allows the team to gauge safety and efficacy during both percutaneous and traditional surgical repairs.
The American College of Cardiology and American Heart Association (ACC/AHA) guidelines provide a framework for patient selection and timing of MV surgery. Any patient with acute severe MR should undergo valve surgery. Valve surgery is also recommended for patients with chronic severe MR in the presence of symptoms and for patients without symptoms in the presence of LV systolic dysfunction or LV cavity dilatation (end-systolic dimension >40 mm). Valve surgery results in the preservation of LV function and improved survival.
The onset of atrial fibrillation or the development of significant pulmonary hypertension (pulmonary artery systolic pressure >50 mm Hg at rest or >60 mm Hg with exercise) in an asymptomatic patient with normal LV size and systolic function is a reasonable indication for surgery. Management of asymptomatic patients with severe MR and normal LV size and function remains controversial, but surgery is considered reasonable at centers where repair is likely (>95% chance) and has a low operative risk (<1%); this is provided a class IIa recommendation in the most recent ACC/AHA Guidelines.
MVRe is the preferred method for surgical management of MR to restore normal leaflet function and annular size. When compared with MV replacement, the major advantages of MVRe are improved survival, preservation of LV function, freedom from anticoagulation, and fewer complications. Despite the advantages of MVRe, this technique appears to be underutilized: fewer than 50% of the patients undergoing MV surgery currently receive a repair procedure, even though about 90% are suitable.
MV surgery can be performed via median sternotomy, a minimally invasive approach that uses partial upper sternotomy or small right thoracotomy, or it can be performed robotically through multiple “ports.” Median sternotomy is required if concomitant coronary artery bypass is undertaken. Cardioplegic arrest and cardiopulmonary bypass are necessary regardless of the type of chest wall incision, although typically less than 1 hour is required for a valve repair. Techniques of repair address the annulus (annuloplasty with or without a rigid or flexible ring, decalcification, débridement), the leaflets (triangular or quadrangular resection, sliding annuloplasty, patch enlargement, decalcification, E2E repair), the chordae (resection or elongation of chords), the myocardium itself (e.g., remodeling through the Dor procedure, plication of scar, pericardial cushions, transventricular slings), the papillary muscles (realignment), or some combination of all these.
Isolated mitral annuloplasty is usually reserved for patients with FMR. Available annuloplasty techniques include suture alone, suture with buttressing material, and prosthetic annuloplasty devices. The choice of annuloplasty technique is surgeon and patient specific. A prosthetic annuloplasty band or ring is placed to correct annular dilatation, increase leaflet coaptation by reducing the anteroposterior dimension of the annulus, and prevent future annular dilatation.
Annuloplasty for FMR results in significant improvement in NYHA class, decreased hospital admissions for heart failure, and modest survival rates of 71% to 82% at 2 years and 58% at 5 years. Although favorable changes in LV size, shape, and function have been demonstrated after successful MVRe, a propensity analysis failed to demonstrate any mortality benefit compared with matched patients not undergoing valve surgery. The recurrence rate of FMR after isolated annuloplasty is disappointing (28% at 1 year) and, along with the usually comorbid conditions of this patient population, remains a limitation to widespread use of the procedure.
The combination of annuloplasty and leaflet repair, referred to as Carpentier’s techniques , is most frequently performed for degenerative MV disease. Among the surgical methods of mitral leaflet repair, correction of posterior leaflet or bileaflet prolapse is the most common. Posterior leaflet prolapse occurs in most cases of degenerative MV disease and is the primary cause of regurgitation in approximately 50% of patients. Prolapse results from chordal elongation or rupture and affects the central P2 segment most frequently. This type of problem is most often corrected by posterior leaflet quadrangular resection and plication of the valve annulus, usually accompanied by the placement of a prosthetic annuloplasty ring except in cases of severe calcification of the annulus.
Anterior leaflet prolapse, although less common than posterior leaflet prolapse, is a more challenging problem and has been commonly treated with initial valve replacement due to poor results of repair in this group and greater parity with replacement outcomes. However, when appropriate, several methods have been developed to treat anterior leaflet prolapse with leaflet repair. The most common are chordal transfer, artificial chordae creation, and the Alfieri E2E repair. Chordal transfer is performed by resection of a segment of the posterior leaflet, which is then transferred and sewn to the prolapsing segment of the anterior leaflet. A quadrangular repair of the anterior leaflet completes this procedure. Another method of anterior leaflet repair involves the creation of artificial chordae from Gore-Tex sutures. These artificial chordae are attached to the prolapsing leaflet and the papillary muscle by pledgeted sutures.
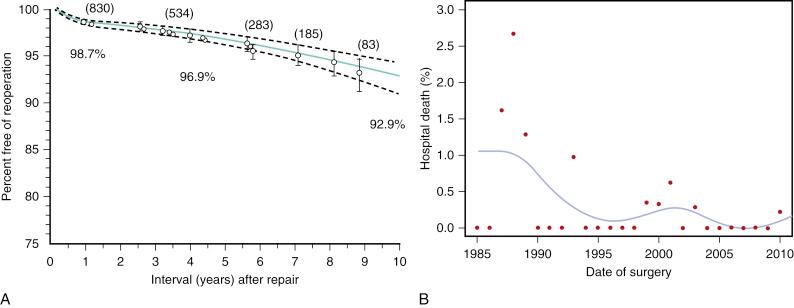
The long-term results for mortality, recurrent MR, and reoperation with MVRe in experienced centers are outstanding, are far superior than those for MV replacement, and the mortality rate is similar to that of the general population (86% to 93% survival at 5 years). At clinical centers of excellence, such as The Cleveland Clinic, MVRe provides durable results with a 10-year freedom-from-reoperation rate of 93% ( Fig. 52.5A ) and contemporary rates of in-hospital mortality of well under 0.1% (see Fig. 52.5B ).
Dr. Ottavio Alfieri (Milan, Italy) pioneered a creative repair known as the double-orifice, or E2E, technique. This procedure was initially developed for anterior leaflet prolapse: the free edges of the anterior and the posterior leaflets are sewn together in an attempt to increase leaflet contact and coaptation and reduce regurgitation. This technique also works for repair of posterior leaflet and bileaflet prolapse. The resulting double-orifice MV usually does not cause stenosis, even when combined with an annuloplasty ring.
The first report of this technique was published in 1998. A larger series, published in 2001, consisted of 260 patients who underwent E2E repair, 81% of whom had DMR, and 80% of whom also had an annuloplasty ring placed. There was a low rate of in-hospital mortality (0.7%), and overall survival was good (94% at 5 years), with 95% freedom from reoperation ( Fig. 52.6 ). Most patients (>80%) were NYHA class I or II. A subsequent report comparing patients who did with patients who did not receive concomitant annuloplasty demonstrated worse freedom from significant MR and a higher rate of reoperation in those with isolated E2E repair.
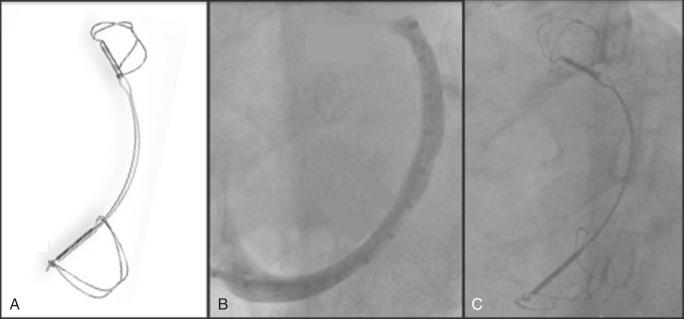
Following Dr. Alfieri’s lead, the technique was adopted into practice at many prominent institutions performing MVRe, although usually as a specialized technique and not as a primary method. The introduction of percutaneous E2E repair has fueled interest in the surgical outcomes of this procedure, leading to the publication of a number of single-center case series. The procedure has been applied to both DMR and ischemic MR with favorable results. Although the surgical double-orifice MVRe has been shown to be effective as a treatment for structural or functional MV disease, the surgical literature is limited by a lack of data on isolated E2E repair.
A series of patients with up to 18 years (mean 9.2 years) of follow-up after surgical E2E repair was recently published by Dr. Alfieri’s group. Of the 61 patients, 36 did not have concomitant annuloplasty due to heavy annular calcification or limited annular dilatation. The average age was 64 years, the average LVEF was 60%, and the indications for surgery were bileaflet prolapse/flail (46%), anterior prolapse/flail (18%), and posterior prolapse/flail (36%). Survival at 12 years was 51%, and freedom from reoperation was only 58%. Of these 36 patients, 55% had 3+ or greater MR, and residual MR greater than 1+ at hospital discharge was a significant predictor of severe MR at follow-up (hazard ratio 3.8; 95% confidence interval 1.7 to 8.2; P = .001). Although this was a small series, these results question the long-term benefit of isolated E2E repair in a group of relatively young patients with DMR and may be relevant to the discussion of percutaneous E2E repair in young and otherwise healthy patients.
The aim of percutaneous strategies for MVRe is to provide relief from severe MR in patients who would otherwise not be candidates for surgical correction or in those who prefer a less invasive approach without the need for cardiopulmonary bypass. The latter is a more difficult proposition, especially in patients with primary (degenerative) MR, given the historical success and safety of surgical MVRe in treating this problem as detailed previously.
There are currently five major approaches to percutaneous repair. The best-studied approach is the E2E repair, based loosely on the surgical repair championed by Dr. Alfieri. The second approach uses the proximity of the coronary sinus to the mitral annulus to accomplish favorable changes in annular geometry, bringing the posterior leaflet toward the anterior leaflet and thereby improving coaptation. In the third approach, LV reshaping, which accomplishes a reduction in septal-to-lateral diameter and improves leaflet coaptation, can reduce MR. In the fourth approach, called the transventricular (direct) approach, mitral annuloplasty is performed by various methods. Another novel approach involves the implantation of artificial chordae. Finally, recent preclinical and clinical work in transcatheter MV replacement has produced interesting results ( Table 52.4 ).
| Device | Development Phase | Ongoing Trial and Stage |
|---|---|---|
| Edge-to-Edge Leaflet Repair | ||
| MitraClip (Abbott Vascular, Minneapolis, MN) | FDA approval for DMR 2013FDA approval for FMR 2019CE Mark approval | No major trials ongoing |
| Indirect Annuloplasty | ||
| CARILLON Mitral Contour System (Cardiac Dimensions, Kirkland, WA) | CE Mark ApprovalPhase II trials published | Phase III Trial:Carillon Pivotal: enrolling (Device vs. Sham + OMT) |
| Direct Annuloplasty | ||
| Mitralign (Mitralign, Salem, NH) | CE Mark Approval | N/A |
| Cardioband (Edwards Lifesciences, Irvine, CA) | CE Mark ApprovalPhase II trial published | Phase III Trial:ACTIVE: enrolling (Device vs. OMT) |
| Cardiac Chamber Remodeling | ||
| Accucinch Percutaneous Ventricular Repair System (Ancora Heart, Santa Clara, CA) | First-in-human implants | Phase II Trial:Planning underway |
| ARTO Septal Sinus Shortening System (MVRx Inc, Belmont, CA) | First-in-human implants | N/A |
| Artificial Chordae Insertion | ||
| NeoChord DS1000 (Neochord, Minneapolis, MN) | CE Mark ApprovalPhase II trial published | N/A |
| Harpoon MVRS (Edwards Lifesciences, Irvine, CA) | Phase II trial published | |
| Transcatheter Mitral Valve Replacement | ||
| Tiara (Neovasc, Inc., Richmond, Ontario) | First-in-human implants | N/A |
| Intrepid (Medtronic Inc., Minneapolis, MN) | Phase II trial published | Phase III Trial:APPOLO: enrolling (TMVR vs. SMVR) |
| Tendyne (Abbott Vascular Santa Clara, CA) | Phase II trial published | Expanded Phase II Trial:Expanded Clinical Study of the Tendyne System: enrolling (TMVR registry alone) |
| CardiAQ (Edwards Lifesciences, Irvine, CA) | First-in-human implants | Phase II Trial:CardiAQ Early Feasibility Trial: enrolling (TMVR registry alone) |
| LivaNova Caisson (LivaNova, Maple Grove, MN) | First-in-human implants | Phase II Trial:PRELUDE: enrolling (TMVR registry alone) |
| SAPIEN M3 (Edwards Lifesciences, Irvine, CA) | First-in-human implants | N/A |
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here