Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The clinician must be alert for any evidence of underlying neurologic disease, including asymmetrical abdominal reflexes, abnormalities on neurologic examination, or a cavovarus foot deformity. On radiographs, hyperkyphosis over the apex of the curvature, an unusual curve pattern, marked trunk shift, a left thoracic curve, or lack of curve rotation may indicate an underlying neurologic condition. These patients should undergo magnetic resonance imaging (MRI).
Patients presenting with significant pain, those who have rapid progression, and children younger than 10 years of age should also have an MRI screening of the entire spine prior to initiation of treatment.
Shoulder asymmetry, unilateral Sprengel deformity, spasm from acute pain, or a leg-length discrepancy can give the false clinical appearance of scoliosis. Standing full-length scoliosis films are the gold standard for diagnosis.
Bracing and surgery are the mainstays of treatment for scoliosis. Treatment must be tailored to the patient and family. Bracing is typically indicated in growing children with progressive curves measuring 20 to 45 degrees. In attempts to prevent curve progression, bracing is most effective in skeletally immature patients (Risser 0, 1, or 2) and in females who are less than 12 months past menarche. Casting is well tolerated in the young child and may be a preferable technique over bracing because of compliance. It has been shown to be curative in some cases of infantile scoliosis.
When the curve magnitude exceeds 50 degrees in adolescent idiopathic scoliosis, surgical treatment is offered, typically with posterior spinal fusion.
Adult scoliosis is most commonly due to de novo degeneration, progression of adolescent scoliosis, or iatrogenic failure to consider sagittal and coronal balance in spine surgery.
Sagittal plane deformity is significantly disabling, and lack of patient improvement following adult deformity surgery is linked to failure to correct sagittal imbalance.
Goals of adult spinal deformity (ASD) surgery are achieving spinopelvic harmony of lumbar lordosis within 10 degrees of pelvic incidence, sagittal vertical axis <50 mm, and pelvic tilt <25 mm; decompressing symptomatic neurologic elements; and stabilizing sagittal and coronal balance.
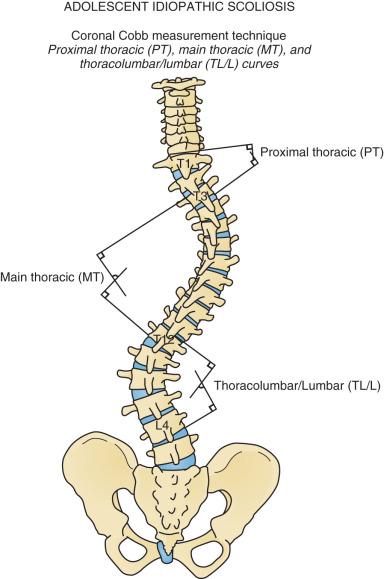
Scoliosis represents a complex three-dimensional deformity of the spine and thorax. Scoliosis is defined as a coronal deformity of the spine with a Cobb angle measuring greater than 10 degrees ( Fig. 37.1 ). Small-magnitude curves may progress during periods of skeletal growth. Larger curves may continue to increase, even in adulthood. Thus a pediatric patient with scoliosis should be followed until skeletal maturity, and young adults with large untreated curves should be monitored periodically for curve progression.
Congenital, neuromuscular, and idiopathic curves are commonly diagnosed in childhood. Congenital scoliosis results from malformation of the vertebral elements, due to either failure of formation or failure of segmentation. A hemivertebra with a contralateral bar results in the greatest risk of curve progression. Particularly in small children, it is difficult to predict which congenital curves will be progressive and will subsequently require surgical intervention. Because of a high associated risk, all children with congenital scoliosis require evaluation for concomitant renal, cardiac, and intrathecal abnormalities. Owing to the structural abnormalities, bracing in congenital scoliosis is typically not indicated. Neuromuscular curves are those associated with an underlying neurologic difference, either structural (Chiari malformation, syrinx, tethered cord, diastematomyelia) or systemic (eg, cerebral palsy, Charcot-Marie-Tooth disease, muscular dystrophy, neurofibromatosis). As idiopathic scoliosis is a diagnosis of exclusion, neurologic causes for the deformity must be ruled out by medical history, clinical examination, radiographs, and, in selected cases, axial imaging.
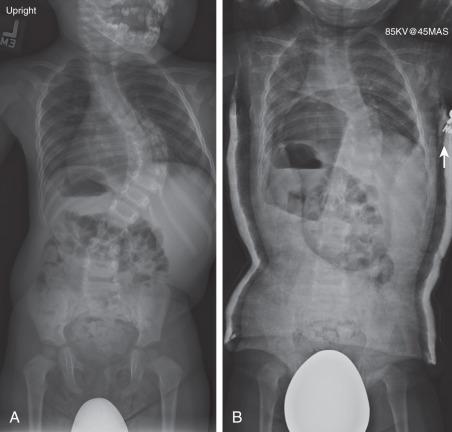
Idiopathic scoliosis is the most common type of scoliosis in children. Based on age at diagnosis, idiopathic scoliosis is traditionally classified into infantile (age less than 3 years), juvenile (age 3 to 10 years), and adolescent (age greater than 10 years). More recently, the term early-onset scoliosis has been used for spinal deformities in children under the age of 5 years—including idiopathic, congenital, and neuromuscular curves—in order to acknowledge the special challenges of treating very young children with scoliosis. Infantile neuromuscular scoliosis is frequently progressive, but if the curve is flexible it may be effectively managed with bracing and casting ( Fig. 37.2 ). Idiopathic infantile scoliosis is more common in males than females, and patients may present with a left-sided curve. Factors associated with progressive infantile idiopathic scoliosis include age at treatment, rib vertebral angle greater than 20 degrees, a phase II rib, or a curve greater than 60 degrees.
Although the cause of idiopathic scoliosis is unknown, family members of affected patients have an increased risk of developing scoliosis, reflecting a genetic component to the disease. Several genes have been associated with familial scoliosis. The incidence of adolescent idiopathic scoliosis (AIS) with a curvature greater than 30 degrees ranges from 1 to 3 in 1000 children. Small curves are common in both males and females, although progression is more common in female children.
Natural history studies of scoliosis reveal the greatest morbidity from early-onset scoliosis, with higher mortality rates and decreased pulmonary function for thoracic curves. One series of patients with untreated juvenile and adolescent scoliosis revealed only one death from cor pulmonale and overall mortality rates similar to those for the average population. Back pain could not be correlated with the severity of the deformity, although patients with lumbar curves had more back pain. Patients with thoracic curves have been noted to have poor pulmonary function because of decreased lung volumes from the coronal plane deformity. Thoracic curves measuring 70 degrees may result in noticeable changes on pulmonary function testing, and curves greater than 100 degrees may result in clinical symptoms.
Curves are at risk for progression during periods of rapid skeletal growth, specifically the first 2 years of life and the adolescent growth spurt. There is increased risk of curve progression for large curves and for skeletally immature patients ( Table 37.1 ). Bone age, peak height velocity, Risser sign, and elbow and hand maturity indices may aid in determining risk of curve progression. Active investigation is under way to identify genetic markers that would assist in predicting curve progression. For adolescent idiopathic scoliosis, thoracic and double curve patterns are at higher risk of progression than thoracolumbar curves. Curves less than 30 degrees at skeletal maturity are thought to be mostly stable and unlikely to progress over time. Curves greater than 50 degrees at skeletal maturity commonly progress at a rate of up to 1 degree per year for thoracic curves. Curves between 35 and 50 degrees at skeletal maturity present a diagnostic dilemma due to insufficient clinical data and should be followed radiographically into adulthood.
| Growing Child | ||
| Open triradiate cartilage, Risser 0, 1, peak height velocity of 8 to 10 cm/year growth. Curves 5–19 degrees have a 22% chance of progression. Curves 20–29 degrees have a 68% chance of progression. |
||
| Curve Size a | Treatment | Follow-up Interval |
| ≤25 degrees | Observation | 4–6 months |
| 25–45 degrees | Bracing (may consider in Risser 0 at 20–25 degrees with documented progression) | 4 months |
| >45 degrees | Surgery—consider anterior release with open triradiate cartilage | |
| Adolescent Nearing Maturity | ||
| Closed triradiate, Risser 3–4, postmenarchal females. Curves 5–19 degrees have a 2% chance of progression. Curves 20–29 degrees have a 23% chance of progression. |
||
| Curve Size a | Treatment | Follow-up Interval |
| ≤30 degrees | Observation | 12 months to document no progression |
| 35–45 degrees | Consider bracing, efficacy not determined | 4 months if braced, 6–12 months otherwise |
| >50 degrees | Surgery | |
| Skeletally Mature Child | ||
| Risser 5, menarche + 2 years in females, cessation of growth. Large thoracic curves >50 degrees may progress up to 0.5–1 degree/year. Lumbar curves >40 degrees may progress. Curves <30 degrees are unlikely to progress. |
||
| Curve Size a | Treatment | Follow-up Interval |
| ≤50 degrees | Observation | 1–2 years |
| >50 degrees | Surgery | |
a Measurement variability for Cobb angle is reported at ±5 to 6 degrees.
Bracing and surgery are the mainstays of treatment for scoliosis. Treatment must be tailored to the patient and family. Bracing is typically indicated in growing children with progressive curves measuring 20 to 45 degrees. In attempts to prevent curve progression, bracing is most effective in skeletally immature patients (Risser 0, 1, or 2) and in females who are less than 12 months past menarche. Numerous types of braces have been described, but a thoracic lumbar spine orthosis (TLSO) is commonly used. Brace wear has been shown to be efficacious. The Bracing in Adolescent Idiopathic Scoliosis Trial (BrAIST), a multicenter randomized controlled trial of bracing AIS patients with 20- to 40-degree curves, showed that bracing significantly decreased curve progression of high-risk curves to the threshold for surgery and benefit increased with longer hours of brace wear. BrAIST further showed that body image and quality of life scores do not have a significant impact on brace wear adherence and are not impacted by brace wear in female adolescents with AIS.
The brace is discontinued at the completion of growth. Casting is well tolerated in the young child and may be a preferable technique over bracing owing to derotational molding of the deformity and improved compliance. It has been shown to be curative in some cases of infantile scoliosis, although in some instances these curves are self-resolving.
When the curve magnitude exceeds 50 degrees in AIS, surgical treatment is offered, typically with posterior spinal fusion. Fusion of smaller curves can be considered in skeletally immature patients who have been documented to have significant progression or in patients who find the appearance and trunk shift unacceptable. Pain is not typically an indication for surgery. Patients with severe deformity or who are skeletally immature (open triradiate cartilages) may also be treated with anterior release or posterior fusion to improve correction and prevent subsequent rotational and coronal or “crankshaft” deformity. In the era of pedicle screw fixation, the necessity of anterior procedures is debated in the literature and may be a matter of surgeon preference. Anterior release, fusion, and instrumentation in the thoracic spine may be performed through a thoracotomy or through a thoracoscopic approach. Anterior procedures, particularly open surgery, may result in decreased pulmonary function. Large curves and complex deformities can also be addressed by osteotomies through a posterior approach to achieve satisfactory correction. These procedures may include, in order of increased correction and neurologic risk, Ponte/Smith-Peterson osteotomies, pedicle subtraction osteotomies, and vertebral column resections.
Neuromuscular scoliosis is guided by similar treatment principles. Progressive imbalance from muscle weakness or spasticity may result in a sweeping thoracolumbar or C-type curvature and subsequent pelvic obliquity, truncal shift, and difficulty with wheelchair seating and caregiving. The role of bracing in the neuromuscular population is not well defined but may be a helpful delay tactic. Custom wheelchair seating systems can also serve as an external brace in many instances. Surgical management is indicated when bracing or wheelchair positioning can no longer accommodate the patient's deformity and the curve progresses to greater than 50 degrees ( Fig. 37.3 ). Smaller curves may continue to progress in adult patients with neuromuscular scoliosis, although curves greater than 50 degrees increase at a faster rate. Patients with underlying cardiac or pulmonary deficits as classically seen in Duchenne muscular dystrophy may benefit from early surgery performed for smaller magnitude curves rather than waiting for the curve to progress while the patient's medical status deteriorates. New corticosteroid treatment protocols for Duchenne muscular dystrophy may redefine these indications. Surgical treatment of nonambulatory patients typically involves fusion from the upper thoracic spine to the pelvis, although the literature has shown improved mobility in paraplegic children fused to L5. Long-term results from this technique are not established. In ambulatory patients with an underlying neuromuscular condition, longer fusions are typically indicated to avoid subsequent decompensation.
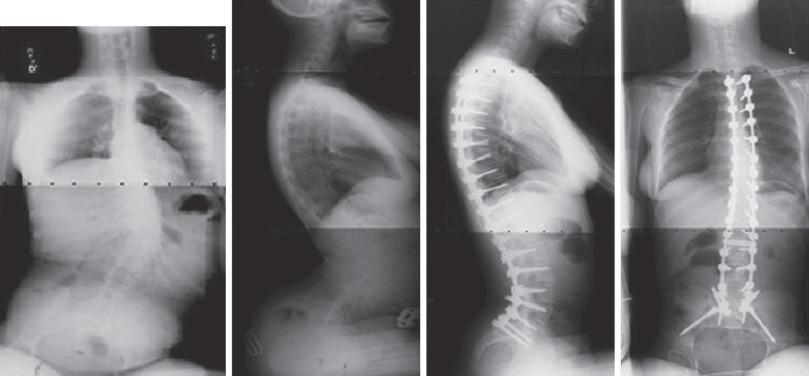
Early-onset scoliosis has unique considerations. Thoracic fusion in a small child is unacceptable and may later lead to thoracic insufficiency syndrome and significant shortening of the trunk. Delay tactics include casting, bracing, and halo gravity traction. When curves do not respond to these measures, surgical intervention may be undertaken to prevent curve progression and allow for spinal growth. Several growing spine systems are in use, including growing spine-based and rib-based devices. There is limited consensus as to the indications and applications of these devices. These are seen as a last resort because the child is then committed to twice yearly lengthening procedures and a high rate of complications, including infection, wound problems, early unintended fusion, implant failure, and, rarely, neurologic complications.
Magnetic controlled growing rod (MCGR) technology has been developed to allow for percutaneous painless distraction of growing rods via handheld magnet. The MCGR procedure can be safely and effectively used in outpatient settings and minimizes surgical scarring and psychological distress while providing similar distraction to traditional growing rods. Because of increased complications seen with single rods, double rods should be utilized. Improvement in pulmonary function has been seen in a small series of early-onset scoliosis patients treated with MCGR. The lengthening procedure can be safely done using ultrasound to measure length of distraction. MCGR achieved cost neutrality to traditional growing rods at 3 years after index surgery.
A planned definitive fusion is performed at the completion of the lengthening procedures. For traditional growing rods, a dual-rod construct has been shown to have fewer complications and improved correction, although a single rod can be used in conjunction with a brace. The implants are frequently quite prominent. Prior to initiating surgical treatment, nutritional and medical status must be optimized. Large curves increase the work of breathing and energy expenditure. Thus children with significant deformity are often malnourished and require dietary supplementation and in many instances gastrostomy tube placement to improve soft tissue coverage and reduce wound complications.
Scoliosis is typically painless in the pediatric population, and children for the most part are asymptomatic. Mild back pain, however, is not uncommon. Screening examinations by pediatricians or school nurses frequently result in referrals. Parents may notice shoulder, scapula, or waist asymmetry, particularly if they are attuned to the diagnosis by a family history of scoliosis.
All patients with presumed idiopathic scoliosis should be evaluated with a medical history, complete neurologic examination, and radiographs. A family history should be taken for scoliosis or a history of underlying neurologic abnormalities. Females should be asked about age at menarche, because this landmark is an excellent predictor of skeletal maturity. On physical examination, the patient's back should be carefully examined for asymmetry, trunk shift, and sagittal plane alignment. AIS is balanced in the coronal plane and typically hypokyphotic, so the presence of kyphosis or excessive truncal shift is an indication for axial imaging ( Fig. 37.4 ). Asymmetrical abdominal reflexes may be associated with a neural axis abnormality. The Adams forward bend test may reveal a thoracic or lumbar rib prominence resulting from rotational deformity. The pelvis should be assessed for an occult leg-length discrepancy, which may give the appearance of scoliosis. The skin should be examined for masses, café au lait patches, and axillary or inguinal freckling indicative of neurofibromatosis. A hairy patch, dimple, or pigmentation over the lumbosacral region suggests spinal dysraphism. The patient should be examined for excess ligamentous laxity or pectus deformity, raising the possibility of an underlying syndrome.
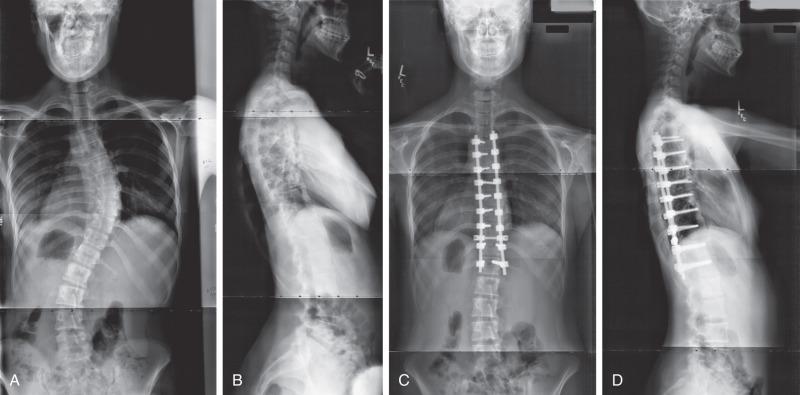
Neural axis abnormalities are found in 13% to 26% of patients with juvenile scoliosis and 2.6% to 14% of patients with adolescent-onset scoliosis. Up to 50% of patients with a left thoracic rather than right thoracic curve may have an underlying neural axis abnormality. As many as 20% to 50% of children with infantile scoliosis may have an underlying neural axis abnormality. No difference in treatment outcome has been reported based on the presence of a neurologic abnormality, although a large syrinx, tethered cord, or Chiari malformation may require surgical intervention ( Fig. 37.5 ). In some instances, scoliosis may improve after treatment of the neural axis abnormality, but this is not predictable.
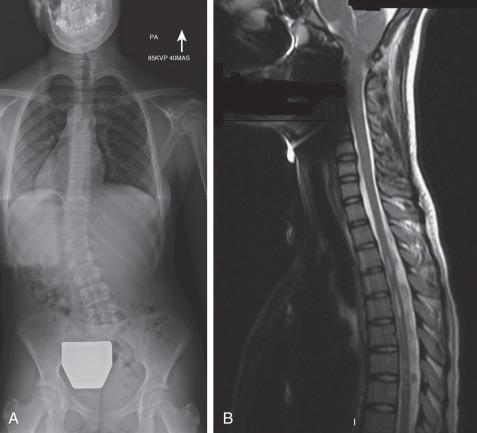
Standing posteroanterior (PA) and lateral spine images should include the neck, clavicles, and pelvis. A lateral image should be taken at presentation in order to document the sagittal plane alignment and rule out congenital deformity. Subsequently, the curve can be followed by standing PA imaging unless there is a concern for kyphoscoliosis.
The idiopathic scoliosis curve types have been classified by King and associates and by Lenke and colleagues ( Fig. 37.6 ). These classifications assist in choosing fusion levels and help identify idiopathic curve types for research purposes. With the Lenke classification, premeasured radiographs have high inter- and intrarater reliability, although there is only poor to good interobserver reliability on unmeasured radiographs. Cobb angle measurement error among different observers is 5 to 6 degrees; thus a more than 5-degree increase in curve size is necessary to document curve progression. Digital imaging systems may improve measurement accuracy. Children who may be candidates for treatment should be followed with radiographs several times annually during periods of rapid growth.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here