Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Paroxysmal nocturnal hemoglobinuria (PNH) is an opportunity to expound upon three major topics: hemolytic anemia, autoimmune-mediated bone marrow failure, and thrombosis. For the physician explaining a new diagnosis to the patient, PNH is now a cause for cautious optimism, as we have seen a tremendous improvement in the outcome over the past decades. Indeed, we can now alleviate symptoms and prevent complications in almost all patients, and life expectancy of a newly diagnosed patient is now close to that of age-matched controls. The complement-mediated hemolysis in PNH has inspired the development of a new class of drugs that target complement proteins (see Chapter 23 ), and C5 blockade has been shown to not only ameliorate hemolysis but also reduce the risk of thrombosis in PNH. A novel C3 inhibitor was approved recently for PNH and may be particularly useful for those with persistent hemolytic anemia despite C5 inhibition. Anti-complement therapy has also revolutionized the treatment of another hematologic disorder (atypical hemolytic anemic syndrome, aHUS) (see Chapter 132 ), as well as two neurologic disorders, with more drugs and additional indications likely to come.
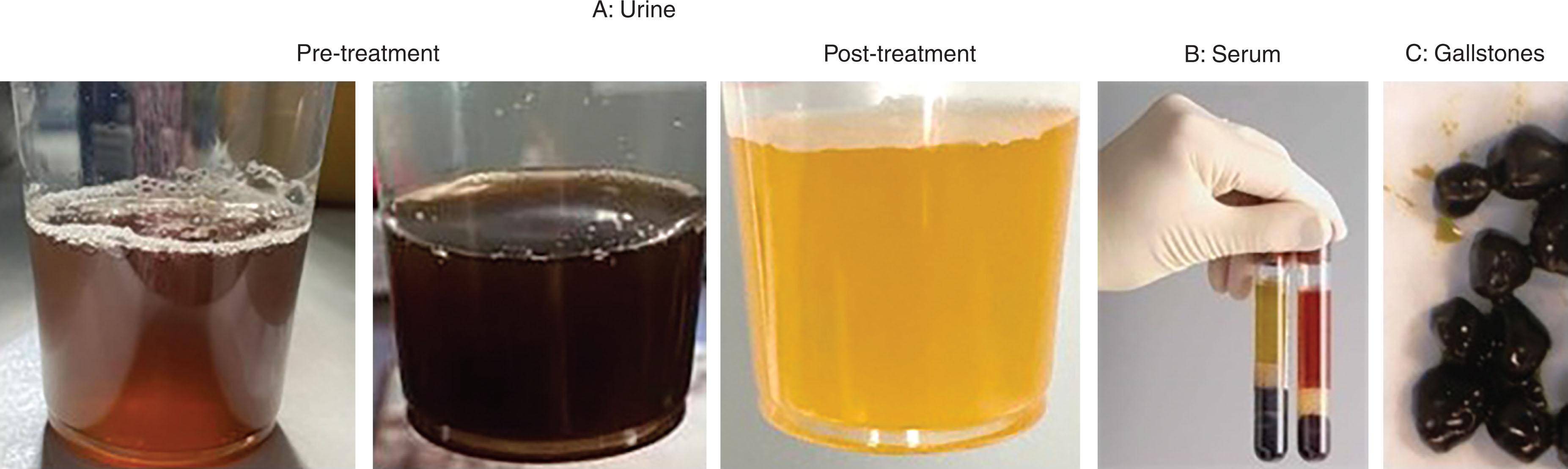
In considering the earliest descriptions of patients who might have had PNH, we must keep in mind that most patients throughout history with dark urine, for sure, have not had this rare disorder. However, William Gull reported on a patient in 1866 with an intermittent bloody-appearing urine that did not contain red cells, with the color of the morning samples more pronounced than later in the day —possibly the first reported case. Paul Strübing in 1882 described a young man with urine that appeared black or dark red ( Fig. 32.1A ), with associated fatigue and abdominal pain. He made the critical observation that the attacks were not precipitated by exposure to cold, differentiating this from paroxysmal cold hemoglobinuria (PCH), which, due to its association with untreated syphilis, was then much more common. Strübing deduced that the hemolysis was intravascular, as the patient’s plasma turned red after severe attacks (see Fig. 32.1B ). Van den Bergh later characterized the disorder in a patient, having ruled out PCH by the Donath Landsteiner test, and demonstrated that red cells under conditions of acidification with CO 2 were lysed by autologous and ABO blood type-matched donor serum. Unlike antibody-mediated hemolysis, hemolysis in vitro could be abrogated by heat treatment of the serum, which was known to inactivate complement. Soon after, it was determined that urine hemosiderin staining could be seen between attacks, and we now know that in spite of the paroxysms that give the disease its name, hemolysis is, in fact, an ongoing process. The disorder was briefly named for the authors Marchiafava and Micheli who recognized this point and was eventually termed paroxysmal nocturnal hemoglobinuria by Enneking in 1928.
A major breakthrough occurred in the late 1930s with the development of the Ham test, which provides an estimate of the percentage of red cells that are susceptible to lysis by activation of the alternative pathway, after the acidification of serum in conditions of optimized magnesium concentration. Of note, there are always some red cells that will not lyse in the Ham test even upon repeat exposure to acidified serum, and the severity of the condition roughly correlated with the percentage of cells that were susceptible. Rosse and Dacie in the 1960s delineated these two populations and showed that the abnormal cells were exquisitely sensitive to low levels of human complement in an assay that activated the classical pathway. In 1970, Oni, Osunkoya, and Luzzatto made the critical observation, using G6PD allozymes to infer X-inactivation patterns, that the abnormal red cells in PNH belong to a clone derived from a single cell.
By that time, it had already been recognized that there was a cellular defect in acetylcholinesterase in PNH erythrocytes and alkaline phosphatase in neutrophils, but the lack of these particular enzymes on a clonal population did not yet explain complement sensitivity. That came with the discovery of a regulator of early complement proteins, CD55 (decay accelerating factor)—and the observation by Anne Nicholson-Weller and her colleagues that this protein is missing on PNH red cells and leucocytes. It was soon appreciated that there is a second regulator, membrane inhibitor of reactive lysis (MIRL, CD59), which inhibits the late complement proteins that can form a membrane attack complex, and it was found that like CD55, CD59 is also missing on blood cells in PNH ( Fig. 32.2 ).
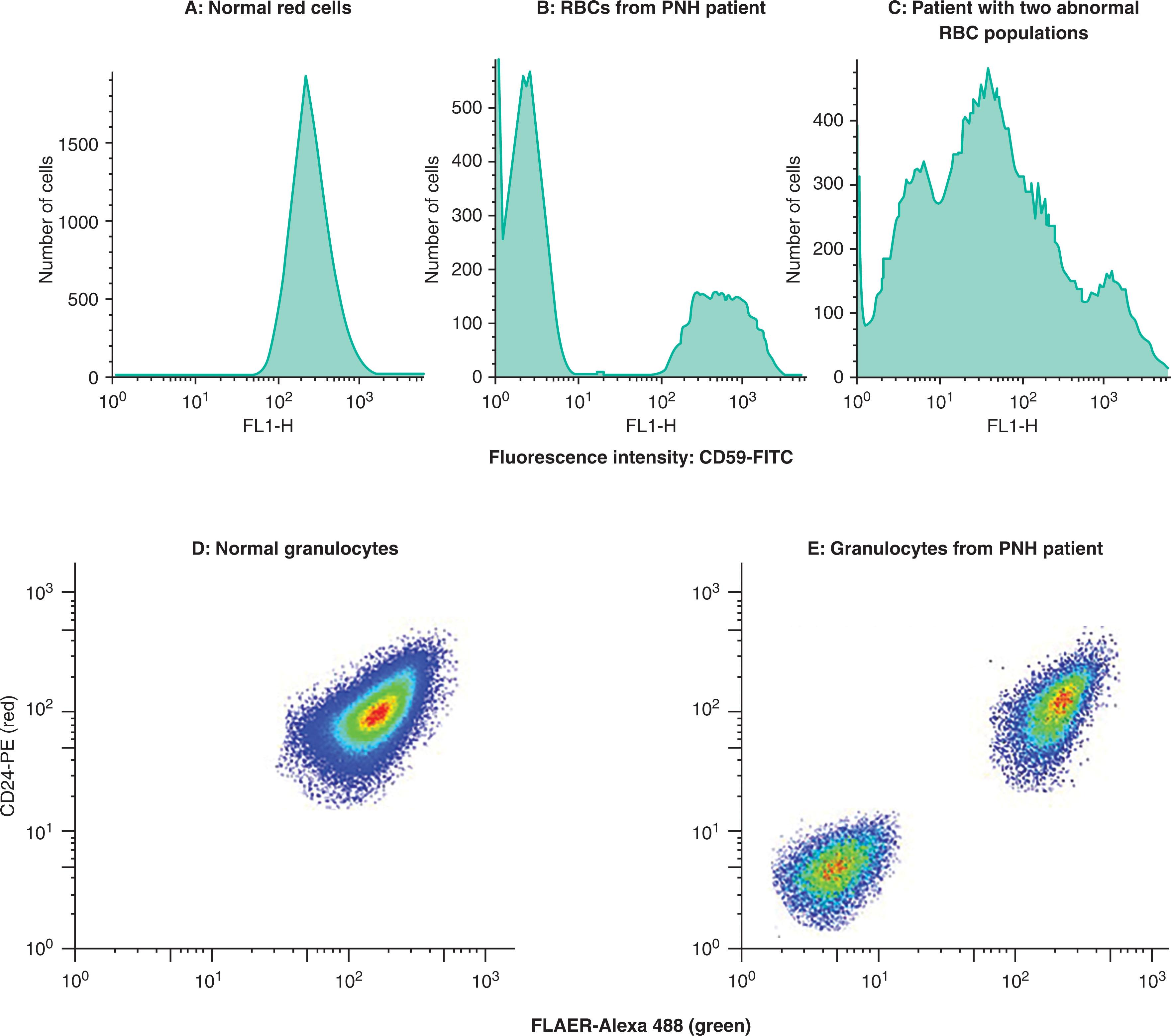
The missing membrane proteins ( Table 32.1 ) have in common the glycosylphosphatidylinositol (GPI) anchor, which is covalently bound to the carboxy terminus of certain proteins and tethers them to the membrane. Taroh Kinoshita’s group and colleagues in Osaka first showed in the early 1990s that PNH cells are defective in the first step in the biosynthesis of GPI and then went on to isolate the gene responsible for this step by complementation cloning in cell lines. They named this gene PIGA (phosphatidylinositol glycan A), and demonstrated that there is an acquired somatic mutation in this gene in all patients with PNH they studied. While there are over 20 genes necessary for the production and trafficking of GPI ( Table 32.2 ), it was understood that PIGA was the only gene found to be mutated in PNH because it maps to the X-chromosome, whereas all the other genes in the pathway are autosomal. Thus, the clonal GPI-negative population would be much more likely to be produced by a single event on the X-chromosome, rather than two separate events involving any of the other genes in the pathway. Identifying the membrane defect in PNH led to a simple flow cytometry test performed on peripheral blood (see Fig. 32.2 ), which by the 1990s was replacing the Ham test. The identification of abnormal leucocyte populations with the same surface defect as red cells and harboring the PIGA mutation confirmed that PNH is indeed not just a red cell disease, but actually is a stem cell disorder.
| Antigen | Hematopoietic Lineage | Classification |
|---|---|---|
| CD55: decay accelerating factor | All blood cells | Complement regulator |
| CD59: membrane inhibitor of reactive lysis | All blood cells | Complement regulator |
| CD58: lymphocyte function associated antigen-3 | All blood cells | Adhesion molecule |
| Acetylcholinesterase | Red blood cells | Enzyme |
| CD14: monocyte differentiation antigen | Granulocytes, monocytes, macrophages | Endotoxin-binding receptor |
| CD16: Fcγ receptor III | Granulocytes, NK cells | Receptor |
| CD66b | Granulocytes | Adhesion |
| Neutrophil alkaline phosphatase | Granulocytes | Enzyme |
| CD87: urokinase (plasminogen activator) receptor | Monocytes, granulocytes | Receptor |
| Leukocyte alkaline phosphatase | Granulocytes | Enzyme |
| CD52: Campath-1 antigen | Lymphocytes, monocytes | Small peptide of unknown function, target of Campath |
| CD24 | B lymphocytes, granulocytes | B-cell differentiation |
| CD48 | All leukocytes | Adhesion molecule |
| CD73: ecto-5′-nucleotidase | Some B and T lymphocytes | Enzyme |
| Dombrock-Holley/Gregory-bearing protein | Red blood cells | Blood group antigen |
| Folate receptor | Myeloid and erythroid cells | Receptor |
| CD109 | Activated platelets and T cells | Unknown |
| CD157 | Mature monocytes | Adhesion and transmigration of monocytes |
| Step | Genes | Enzymatic Function | Human Neurologic Phenotype | Human Hematologic Phenotype |
|---|---|---|---|---|
| 1 | PIGA | UDP-GlcNAc:PI α1–6 N-acetylglucosaminyltransferase (GlcNAc-T) | Yes (MCAHS2) | Acquired mutation→PNH Inherited hypomorphic mutations→no hemolysis reported |
| 1 | PIGH | GlcNAc-T | Yes | No |
| 1 | PIGC | GlcNAc-T | Yes | No |
| 1 | PIGQ | GlcNAc-T | Yes | No |
| 1 | PIGP | GlcNAc-T | Yes | No |
| 1 | DPM2 | GlcNAc-T | Yes | No |
| 1 | PIGY | GlcNAc-T | Yes (HPMRSG) | No |
| 2 | PIGL | GlcNAc-PI deacetylase | Yes (CHIME syndrome) | No |
| 3 | PIGW | Inositol acyltransferase | Yes, hyperphosphatasia | No |
| 4 | PIGM | Mannose transferase | Yes | Inherited promoter mutation→Budd Chiari syndrome. |
| 4 | PIGX | Mannose transferase | No human cases reported | No |
| 5 | PIGV | Mannose transferase | Yes | No |
| 6 | PIGN | Ethanolamine phosphate transferase | Yes | No |
| 7 | PIGB | Mannose transferase | Germline bi-allelic mutations resulting in severe neurologic phenotype | Inherited mutation and CN-LOS→PNH |
| 8 | PIGO | Ethanolamine phosphate transferase | Yes, hyperphosphatasia | No |
| 8 | PIGF | Ethanolamine phosphate transferase | No human cases reported | No |
| 9 | PIGG | Ethanolamine phosphate transferase | Yes | No |
| 9 | PIGF | Ethanolamine phosphate transferase | Yes | No |
| 10 | GPAA1 | GPI transamidase (transfers peptide to the anchor) | Yes | No |
| 10 | PIGK | GPI transamidase (transfers peptide to the anchor) | Yes | No |
| 10 | PIGT | GPI transamidase (transfers peptide to the anchor) | Germline bi-allelic inactivation: multiple congenital abnormalities with hypotonic seizures (MCAHS) | Inherited mutation combined with an acquired mutation: hemolysis responding to eculizumab, aseptic meningitis, urticaria, bowel inflammatory condition (PIGT-PNH) |
| 10 | PIGS | GPI transamidase (transfers peptide to the anchor) | Yes | No |
| 10 | PIGU | GPI transamidase (transfers peptide to the anchor) | Yes | No |
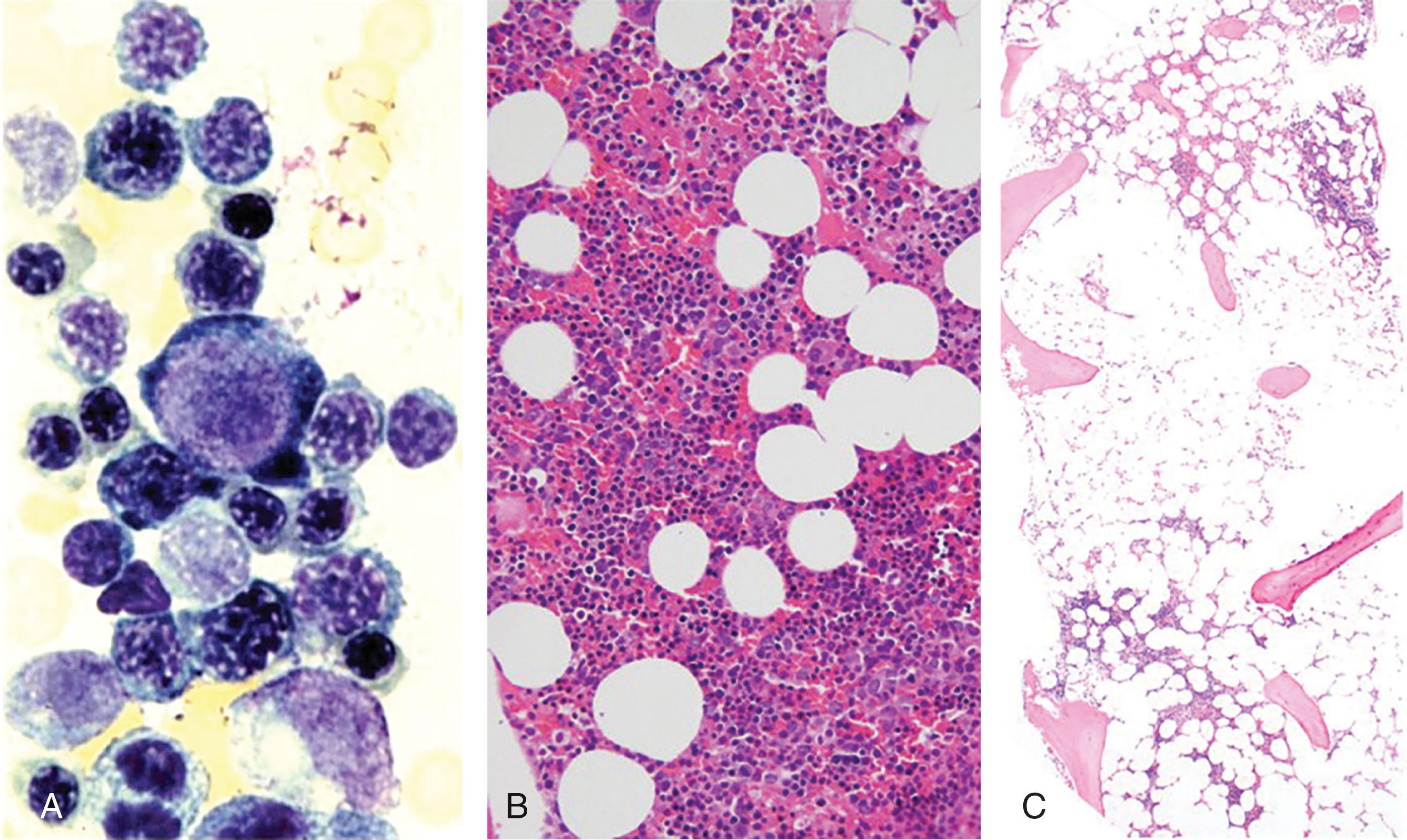
The link between the mutation, GPI-anchor synthesis defect, and loss of complement inhibition now closed the circle on the most prominent clinical phenotype—hemolytic anemia. But this failed to explain the paradox of why this stem cell clone—whose mature red cell progeny, at least, seem to be weakened—would have a growth advantage. To explain this, in 1989 Rotoli and Luzzatto considered the critical earlier observation of Dacie and Lewis that PNH, a rare disease, is highly associated with another rare and seemingly very different hematologic disorder—immune-mediated aplastic anemia (see Chapter 31 ) ( Fig. 32.3 ). This had been commented upon by Dameshek in 1967 and then revisited by Young in 1992. The working hypothesis for the past 32 years has thus been the “immune escape model,” whereby the PNH stem cell has a selective growth advantage in an aplastic marrow environment, because it is less sensitive to immune attack than normal stem cells. This model has not been proven, but it explains many key features of PNH and there is experimental evidence supporting it.
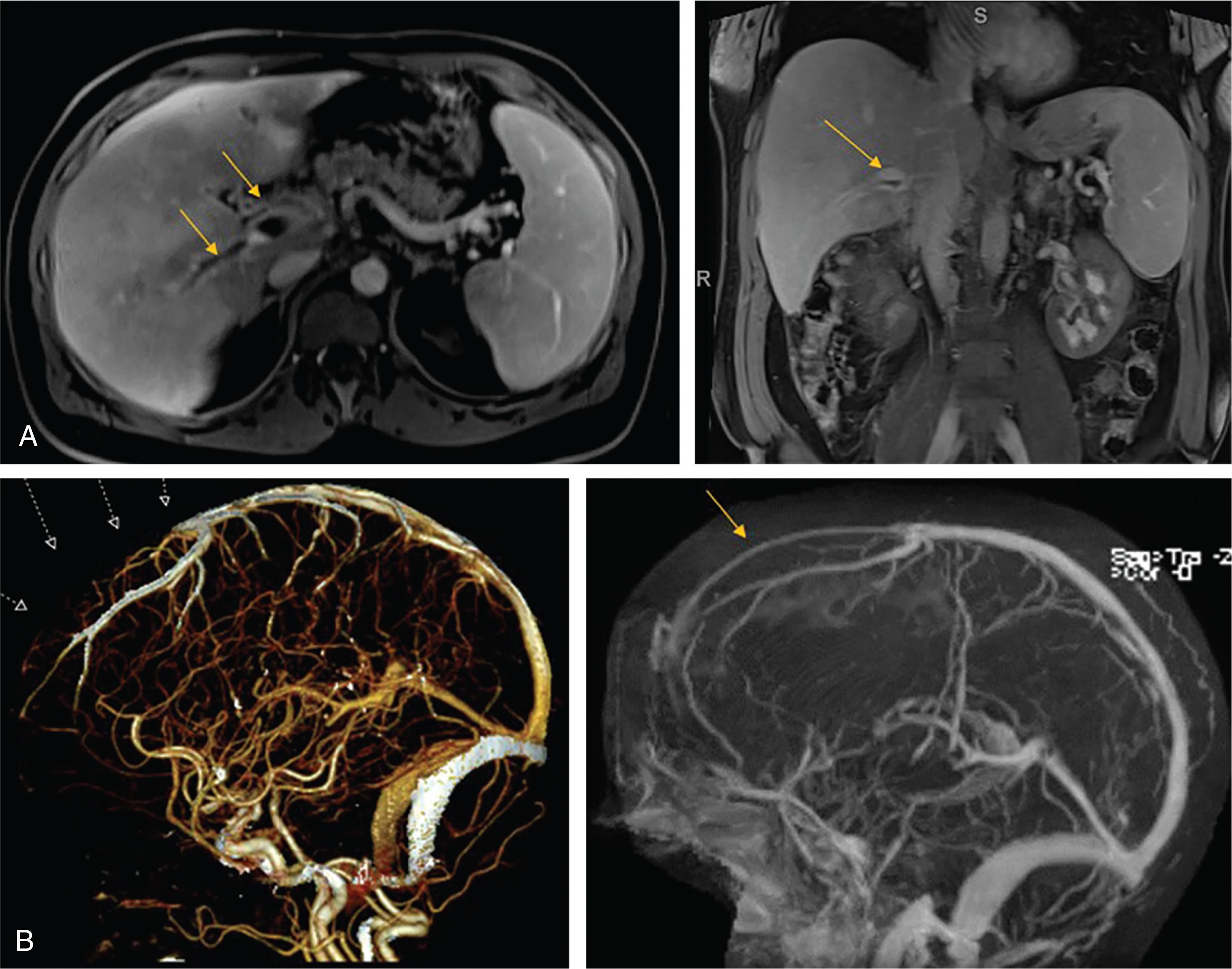
The other major piece of the puzzle has been to understand the pro-thrombotic tendency in PNH, noted 70 years ago by Crosby to be a frequent cause of death, often involving unusual sites such as the cerebral dural venous sinuses and intra-abdominal veins ( Fig. 32.4 ). While there are a few proposed mechanisms for this, the group in the United Kingdom made two major breakthroughs. First, using historical controls, they demonstrated that thrombotic complications in PNH could be largely prevented by primary prophylaxis with warfarin. Then, as reported by Hillmen et al., they pioneered in 11 patients the use of eculizumab, a monoclonal antibody targeting the C5 complement protein (see review by Rother et al. and Chapter 23 ). This led to the first randomized trial in PNH (the TRIUMPH Study), and the drug’s approval in 2007 as a treatment for hemolysis in PNH, for which it can be highly effective. Most importantly, it was subsequently shown that eculizumab reduces thrombotic risk by more than an order of magnitude —from 14 per 100 observation-years in untreated patients to approximately 1 per 100 observation-years in treated patients. Probably as a result of this effect, the UK group has now demonstrated a near normal life-expectancy in treated patients.
Eculizumab has now been engineered in a clever manner with a set of substitutions that increase its half-life about fourfold. However, unfortunately, some patients respond only partially to C5 inhibition. In 2009, Risitano and colleagues made the critical observation that despite C5 blockade, C3 degradation products can still opsonize PNH red cells (due to their lack of CD55), resulting in their uptake in the reticuloendothelial system—a milder form of extravascular hemolysis that had previously been masked by the severe intravascular hemolysis seen in untreated patients (see Fig. 32.1 ). These partial responders represented an unmet clinical need, which has led to the development of the first-ever inhibitor of the C3 complement protein—pegcetacoplan. This drug was designed to prevent both forms of hemolysis and has recently been approved by the Food and Drug Administration (FDA) for the treatment of PNH, after it compared favorably with eculizumab in a randomized trial with an endpoint of hemolytic anemia.
The incidence of PNH in the United Kingdom is reported to be 1.3 per million per year, with a prevalence of approximately 16 per million. In some Asian countries such as Thailand, where the incidence of aplastic anemia is higher, PNH is probably also more common, though still only a few cases per million. Approximately 30% of patients with PNH will have a prior history of acquired aplastic anemia, and this is by far the greatest risk factor for PNH. In contrast, there is a single case report of PNH arising out of an inherited bone marrow failure syndrome (see Chapter 30 ), and the emergence of PNH is essentially unheard of after chemotherapy or radiation-induced marrow hypoplasia. Approximately 7.5% of patients with Budd Chiari syndrome will have a concurrent diagnosis of PNH as an underlying cause, and PNH flow cytometry should be considered part of the hypercoagulability workup for unusual site thromboses, or thrombosis with an abnormal CBC or LDH. The median age of patients at diagnosis is about 40, children can be affected, and males and females are at similar risk.
Patients with a flow cytometry test indicating a PNH clone can be classified as “Classic PNH,” “PNH with another hematologic disorder,” and “laboratory/subclinical PNH.” Patients with classic PNH typically present with intravascular hemolysis or thrombosis. Patients with “PNH and another hematologic disorder” typically have aplastic anemia (AA/PNH) and their PNH clone is often detected based on routine screening or an elevated LDH. PNH/MDS and PNH/MPN overlap syndromes are rare. Patients with “laboratory/subclinical PNH” will have a PNH clone detected as part of a workup for mild cytopenias that defy explanation, or a mildly elevated LDH, MCV, or reticulocyte count, and they by definition do not require treatment. Nevertheless, the identification of a small subclinical clone helps to classify the patient as having an immune-mediated marrow injury. Some patients with PNH will have a population of cells that have only a partial deficiency in expression of CD59 (termed PNH II cells) often in co-existence with cells with a total lack of CD59 (PNH III cells) as well as normal (PNH I) cells (see Fig. 32.2C ). Those patients whose abnormal red cell populations are mainly of the PNH II type may have less severe hemolysis but can have serious thrombotic complications.
Patients with classic PNH will present with dark urine or symptoms of anemia and features of intravascular hemolysis: an elevated LDH and reticulocyte count, anemia, a positive urine hemosiderin stain, with low or absent haptoglobin. The chronic loss of hemoglobin in the urine can result in iron deficiency. The patient will often report that the urine is darkest in the morning; it is unclear if this is due to increased hemolysis at night, as once thought, or increased concentration of the urine due to nocturnal fasting. To clarify what patients mean by “dark urine” it is helpful to ask them to compare the color to some familiar liquid. Patients will often describe their darkest urine as appearing as cola, black ink, soy sauce—or appearing red. This may depend upon the oxidation status of the iron in the free hemoglobin. Bouts of darkened urine are interspersed with periods of time with apparently normal appearing urine, as per the term “paroxysmal,” sometimes with upward swings of a chronically elevated LDH and an exacerbation of anemia.
Patients will sometimes have had extensive urologic evaluations prior to referral to a hematologist: while a dipstick that is positive for “blood” in the absence of red cells on a microscopic examination will be a clue that the problem is not hematuria, some patients with PNH will actually have microscopic hematuria as well. In contrast, jaundice, hyperbilirubinemia, dark gallstones, and iron overload are features of extravascular hemolysis not typically seen on presentation—but are seen commonly in patients on C5 inhibitors. In untreated patients, free hemoglobin in the plasma can scavenge nitrous oxide and result in symptoms of smooth muscle dysfunction: abdominal pains, esophageal spasms, and male erectile dysfunction. Pulmonary arterial hypertension and renal dysfunction can occur, but this is usually subclinical. Sometimes a darkening of the kidneys is picked up on magnetic resonance images (MRI) as an incidental finding, a result of hemosiderin deposition. Attacks of hemolysis can be triggered by infection and fever, and due to abdominal discomfort, it may be difficult for patients to follow the typical instructions to keep well-hydrated. This can require a visit to the emergency room (ER) or infusion center for intravenous fluids, even if transfusions are not required at the time.
Patients with classic PNH can also present with thrombosis (with or without symptomatic hemolysis), typically involving unusual sites, such as the cerebral dural venous sinuses, the portal or hepatic veins, or other intra-abdominal veins, such as the inferior vena cava, the renal veins, splenic veins, or inferior or superior mesenteric veins. Thromboses in most of these sites can be detected by a sonogram with Doppler or a contrast computed tomography (CT), but mesenteric vein thrombosis can be detected only on an MR Venogram, which is also sensitive enough to identify thromboses in smaller branches of the hepatic veins as well. Splenic vein and mesenteric vein thromboses are particularly dangerous because they can lead to splenic rupture and bowel infarction respectively: both are surgical emergencies. Patients with thrombosis and secondary gastrointestinal hemorrhage (or central nervous system hemorrhage due to extensive cerebral vein thrombosis) also represent a particular challenge. Hypersplenism or marrow failure can result in thrombocytopenia, and PNH is therefore one of the conditions where the difficult combination of thrombocytopenia and thrombosis can occur. While the aspartate aminotransferase (AST) can be elevated simply due to hemolysis, an elevated alanine transaminase (ALT) in patients with PNH must be taken as a sign of Budd-Chiari syndrome until proven otherwise. Dermal venous thrombosis has been reported and can be complicated by necrosis. More familiar sites of thrombosis such as extremity deep venous thrombosis, pulmonary emboli, arterial cerebral vascular accidents (CVAs), and myocardial infarction can also occur in patients with PNH.
Patients presenting with AA/PNH fall along a spectrum, where they can resemble patients with aplastic anemia, or classic PNH, or both. As mentioned above, patients with AA/PNH can have thrombosis and thrombocytopenia at the same time due to marrow failure, resulting in a situation where anticoagulation would be dangerous. In AA/PNH, the reticulocyte count and neutrophil count can be decreased as well. Aplastic anemia can precede the diagnosis of PNH, or can occur later. A fall in blood counts in a patient with PNH who has been previously stable should prompt a bone marrow biopsy and aspirate to rule out progression to MDS or AML.
The peripheral smear findings will often show various red cell abnormalities, but none is specific to PNH. However, the smear will be useful to rule out findings suggestive of competing causes of intravascular hemolysis and can indicate concurrent iron deficiency. In those with an adequate erythropoietic response and an elevated reticulocyte count, the most prominent feature on the smear can be polychromasia. The mean corpuscular volume (MCV) is usually elevated, even in the absence of reticulocytosis, as occurs in aplastic anemia.
The bone marrow in patients with classic PNH is typically hypercellular with an erythroid hyperplasia, as can be seen in any patient with reticulocytosis (see Fig. 32.3 ). However, the marrow can range from hypocellular to hypercellular, and there is often a patchy distribution of the marrow. A normocellular or even hypercellular marrow does not rule out AA/PNH, and cytopenias may still respond to immune suppression. An increase in mast cells is sometimes noted. Almost a quarter of patients with PNH will have a cytogenetic abnormality, which is sometimes transient and by itself does not make a diagnosis of myelodysplasia (MDS) (see Chapter 61 ). Likewise, morphological abnormalities can often be seen in PNH that overlap with those seen in MDS. However, excess blasts and complex cytogenetic abnormalities are diagnostic of MDS and are an indication for referral for stem cell transplantation—as may be the case for abnormalities of chromosome 7, which is associated with a poor prognosis when seen in aplastic anemia. Elevated platelets, white blood cell counts, or fibrosis are rare findings but can be seen in the PNH/MPN overlap syndrome (see Chapters 69 and 72 ) and should prompt testing for mutations in JAK2 as well as BCR-ABL fusions.
Understanding the pathophysiology of PNH requires a consideration of the GPI anchor. Although there are variations between species as well as modifications, the core GPI structure consists of protein-ethanoloamine- P -6Manα1–2Manα1–6Manα1 (P-ethanolamine)-4Glucoseamine- phosphatidylinositol ( Fig. 32.5A ). Unlike in yeast, GPI is not essential for the survival of mammalian cells but is essential for embryogenesis. The anchor is synthesized in the endoplasmic reticulum (ER) membrane and involves 10 reactions and almost 30 different genes ( Table 32.2 ). The first two steps take place on the cytoplasmic side and the remaining steps in the lumen of the ER. The first step is the transfer of N-acetylglucosamine (GlcNAc) from uridine diphosphate (UDP)-GlcNAc to phosphatidylinositol (PI) to yield GlcNAc-PI: this is the step that is deficient in PNH. This reaction is catalyzed by GlcNAc-PI α1–6 GlcNAc transferase, an enzyme whose subunits are encoded by seven different genes: PIGA, PIGC, PIGH, PIGQ, PIGY, PIGP, and DPM2 (see Fig. 32.5B ). The PIGA gene encodes a membrane protein in the endoplasmic reticulum that has the active site of this enzyme complex; it has a nonfunctional intron-less pseudogene on chromosome 12.
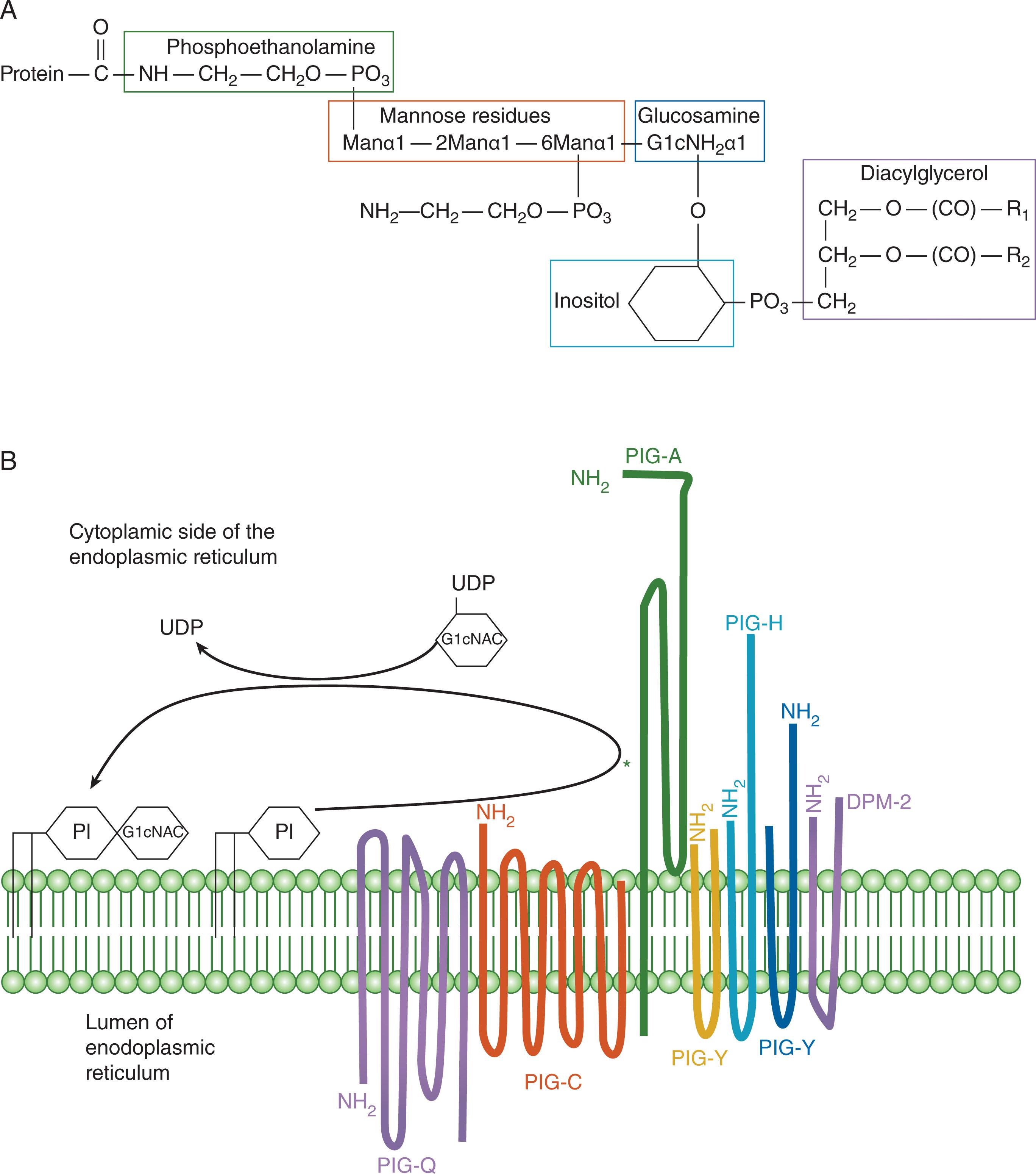
Because of embryonic lethality, the mutations seen in PNH are almost always acquired somatic mutations occurring in a hematopoietic stem cell. Because PIGA is X-linked ( Fig. 32.6 ), a mutational “single hit” will generate a GPI-negative (PNH) phenotype. Of note, although PIGA is near the pseudo-autosomal region, it is completely silenced on the inactive X-chromosome in female cells. Therefore, on the cellular level, any female with a mutation on the active X chromosome that disrupts PIGA will have an equally severe phenotype as the same mutation in a hemizygous male. Thus females are as severely affected as males.
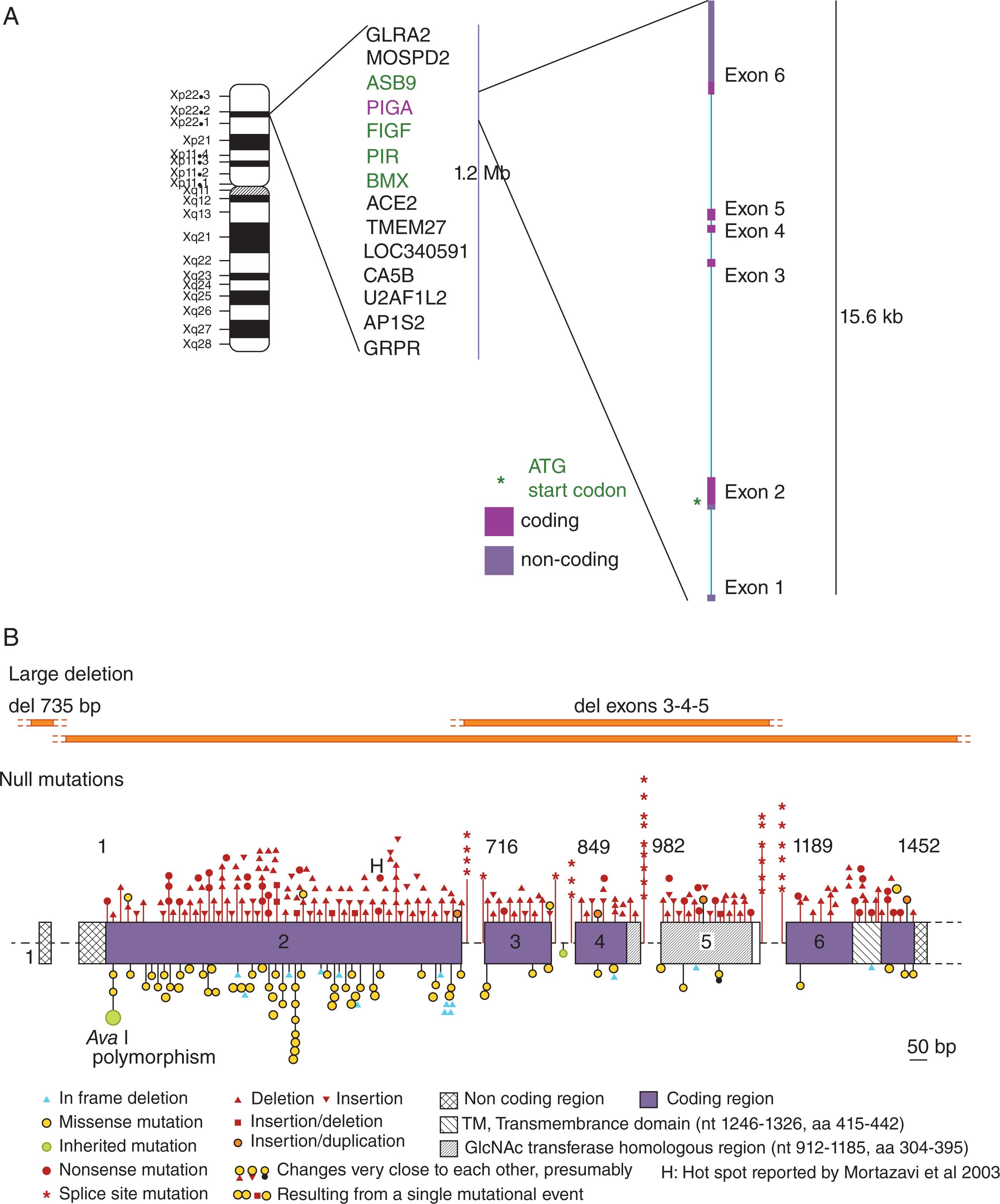
The PIGA gene was identified in the early 1990s by expression cloning and was found to correct the defect in GPI-anchor synthesis when expressed in cell lines from patients with PNH. Since then, PIGA has been found to be mutated in almost all patients. The PIGA gene encodes a protein of 484 amino acids, with a transmembrane domain inserted in the endoplasmic reticulum. Most of the protein is on the cytoplasmic side (see Fig. 32.5B ), with the GlcNAc transferase homologous region containing the active site near the transmembrane domain. There are 6 exons, the longest is exon 2, which contains the translation initiation codon (see Fig. 32.6 ); 600 bp of upstream sequence is required for full gene expression, which has features of a housekeeping gene promoter, and PIGA is listed as a housekeeping gene—as is the case for almost all of the genes required for GPI-anchor assembly.
The somatic inactivating PIGA mutations in PNH are quite diverse and include missense mutations, in frame deletions, nonsense and frameshift mutations, splice site mutations, deletions of hundreds of base pairs —and much larger deletions that also involve contiguous genes on the X-chromosome (see Fig. 32.6 ). Missense mutations are often seen in patients where there is a partial loss of CD59 (PNH II cells), suggesting that there is some residual enzymatic activity, whereas truncating mutations are generally associated with complete loss of CD59. It is not uncommon to observe 3 distinct populations of cells demonstrated by flow: normal cells, PNH II cells with a partial deficiency, and PNH III cells with a complete deficiency of CD59, corresponding to differing degrees of complement senstitivity (see Fig. 32.2 ). In keeping with this, multiple PIGA mutations have been identified in some patients. The oligo clonality sometimes seen in PNH must be taken into account in any model of how PIGA mutant clones expand.
The PIGA gene has also proven useful to other fields. Particularly, because flow cytometry can identify rare mutants among a much larger population of normal cells, PIGA represents an excellent reporter gene for measuring the frequency and rate of spontaneous and induced mutations in human cells and in animals. This provides an important tool in the preclinical evaluation of the genotoxic effects of medications. Furthermore, because of the ease of cell sorting for GPI-positive and GPI-negative cells, and because mutants can be selected by aerolysin (which selectively kills normal cells in a GPI-dependent manner) PIGA represents a selectable and counter-selectable marker for genome editing and genome writing projects.
There is a recently discovered alternative pathway to produce the PNH phenotype: germline loss of one of the autosomal genes involved in GPI-anchor synthesis and a second acquired event that eliminates the remaining allele in hematopoietic stem cells. This was first reported for the PIG T gene on chromosome 20q, and the resulting syndrome has been termed “PIGT-PNH.” Here the first event is an inherited chain-terminating mutation on the maternal allele and the second mutation is a deletion of chromosome 20q ranging from 8 to 18 Mb on the paternal allele. In addition to features of PNH, these patients also develop recurrent aseptic meningitis, urticaria, arthralgia, and bowel inflammation. Of note, the PIG T mutation affects the 10th step in the pathway, where the protein is transferred to the anchor, and accumulation of the completed but un-conjugated anchor may result in the inflammatory phenotype. Recently, a family has been reported where something similar occurs in the PIG B gene on chromosome 15q. Here there is a germline missense mutation in PIG B as well as a 70 kb deletion that is telomeric to PIG B on the same chromosome that may greatly increase the rate of copy neutral loss of heterozygosity events (CN-LOH). A woman with a history of aplastic anemia was found to have the inherited PIG B mutation, the 70 kb deletion, and CN-LOH resulting in the PNH phenotype in almost 100% of blood cells. Her asymptomatic mother, who inherited the same mutations, was found to have approximately 4% PNH leukocytes. Another individual in the pedigree with the missense mutation but without the deletion did not have PNH blood cells.
A partial loss of GPI is not embryonic lethal, and inherited germline hypomorphic alleles have been reported for many of the genes involved, including for PIG A. This produces a severe neurologic phenotype (see Table 32.2 ), but there is generally sufficient residual GPI anchor production so that these patients do not have thrombosis or hemolysis. An exception to this has been the patient reported by Almeida et al. with a PIGM promoter mutation resulting in Budd Chiari syndrome as well as seizures—which responded to epigenetic modification therapy.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here