Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Papulosquamous eruptions comprise a group of disorders characterized by the presence of superficial papules and scale. These conditions account for a large number of patients in both pediatric dermatology and pediatric primary-care practice. In disorders of keratinization (psoriasis, pityriasis rubra pilaris, keratosis follicularis, ichthyosis, hyperkeratosis of the palms and soles, and porokeratosis), cutaneous lesions develop as a result of either genetically programmed retention or increased production of scale in the epidermis. In the inflammatory dermatoses (dermatitides, pityriasis rosea, pityriasis lichenoides, lichenoid dermatoses, and fungal infections), clinical findings result from epidermal response to dermal inflammation. An algorithmic approach to diagnosis for the papulosquamous disorders is summarized at the end of the chapter (see Fig. 3.55 ).
Psoriasis is a common disorder characterized by red, well-demarcated plaques with a dry, thick, silvery scale ( Fig. 3.1 ). The condition affects 1–3% of Americans, of whom an estimated 40% develop the eruption before the age of 20 years. Psychosocial impairment in addition to physical symptoms make early diagnosis and intervention critical in children and adolescents.
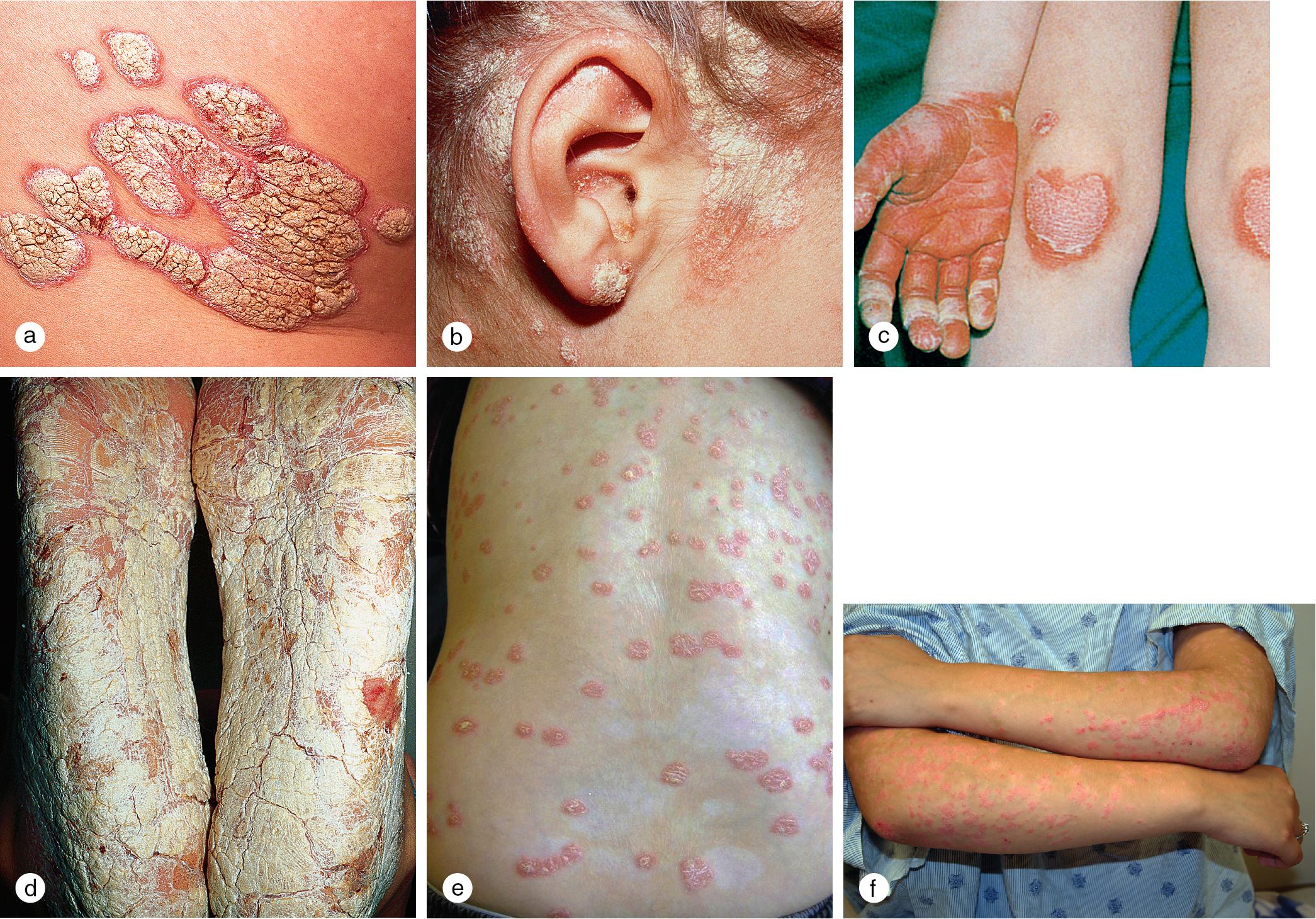
Psoriasis is a multifactorial disorder with both hereditary and environmental components. In more than one-third of patients, other family members are affected. A number of human leukocyte antigen (HLA) (e.g. HLA Cw6) types have been associated with psoriasis in different populations. Several genetic loci have been under investigation, and studies suggest a major susceptibility region for early-onset psoriasis on chromosome 6p21.3. Although the factors that initiate the rapid turnover in epidermal cells, which contributes to psoriatic plaques, are unknown, upper respiratory tract viral and streptococcal infections, urinary tract infections, and other infections are known to precipitate outbreaks in genetically predisposed individuals. Triggers of psoriasis also include drugs (interferon, lithium, carbamazepine, indomethacin, beta-blockers, terfenadine, terbinafine, isotretinoin, antimalarials), climate, cigarette smoking, alcohol, psychologic stress, and malignancy. Once the process is initiated, keratinocytes within the epidermis, under the influence of Th1 cytokines and cells of the immune system, amplify the psoriatic cascade, resulting in angiogenesis, hyperproliferation, and altered cell differentiation.
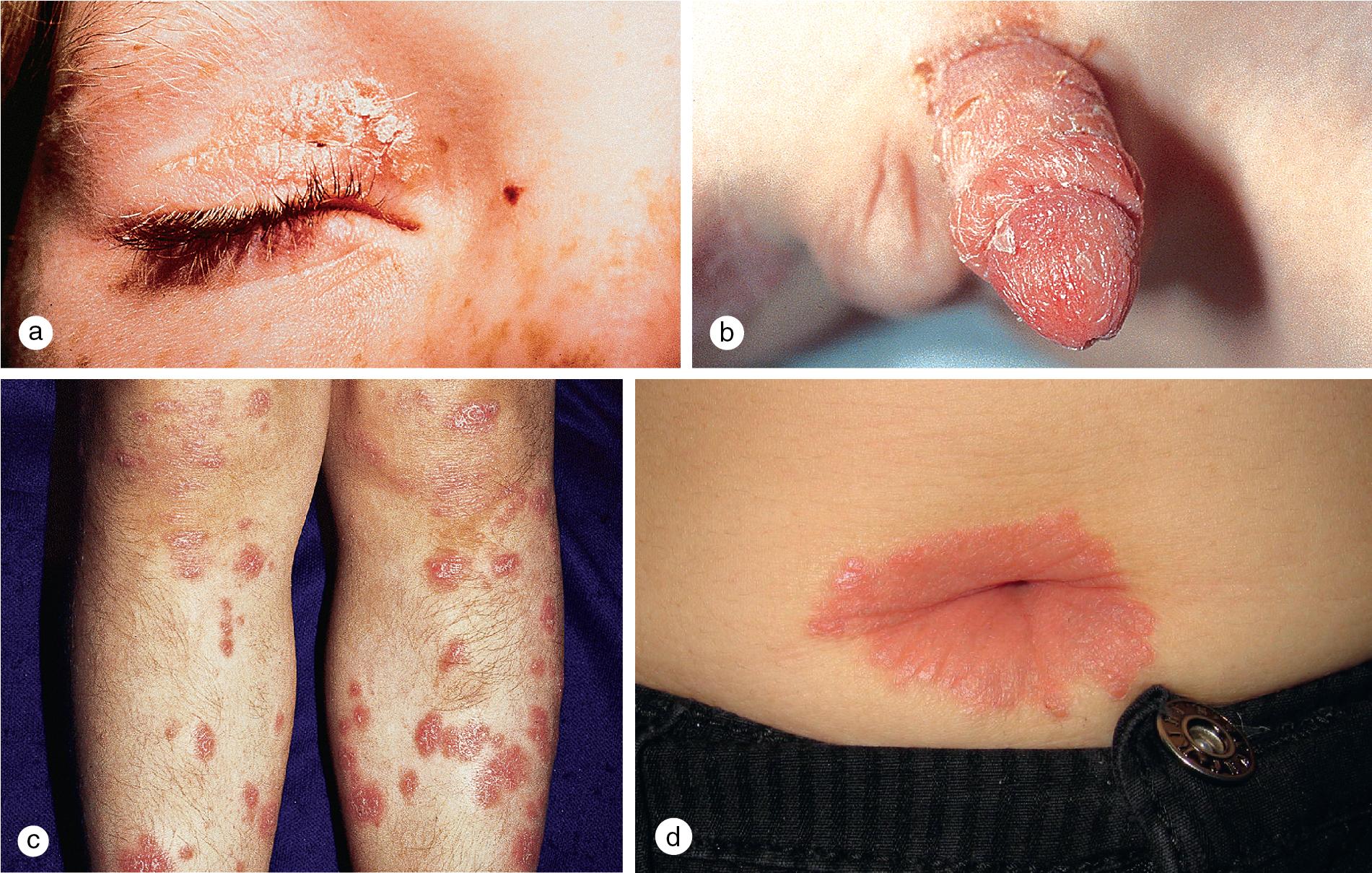
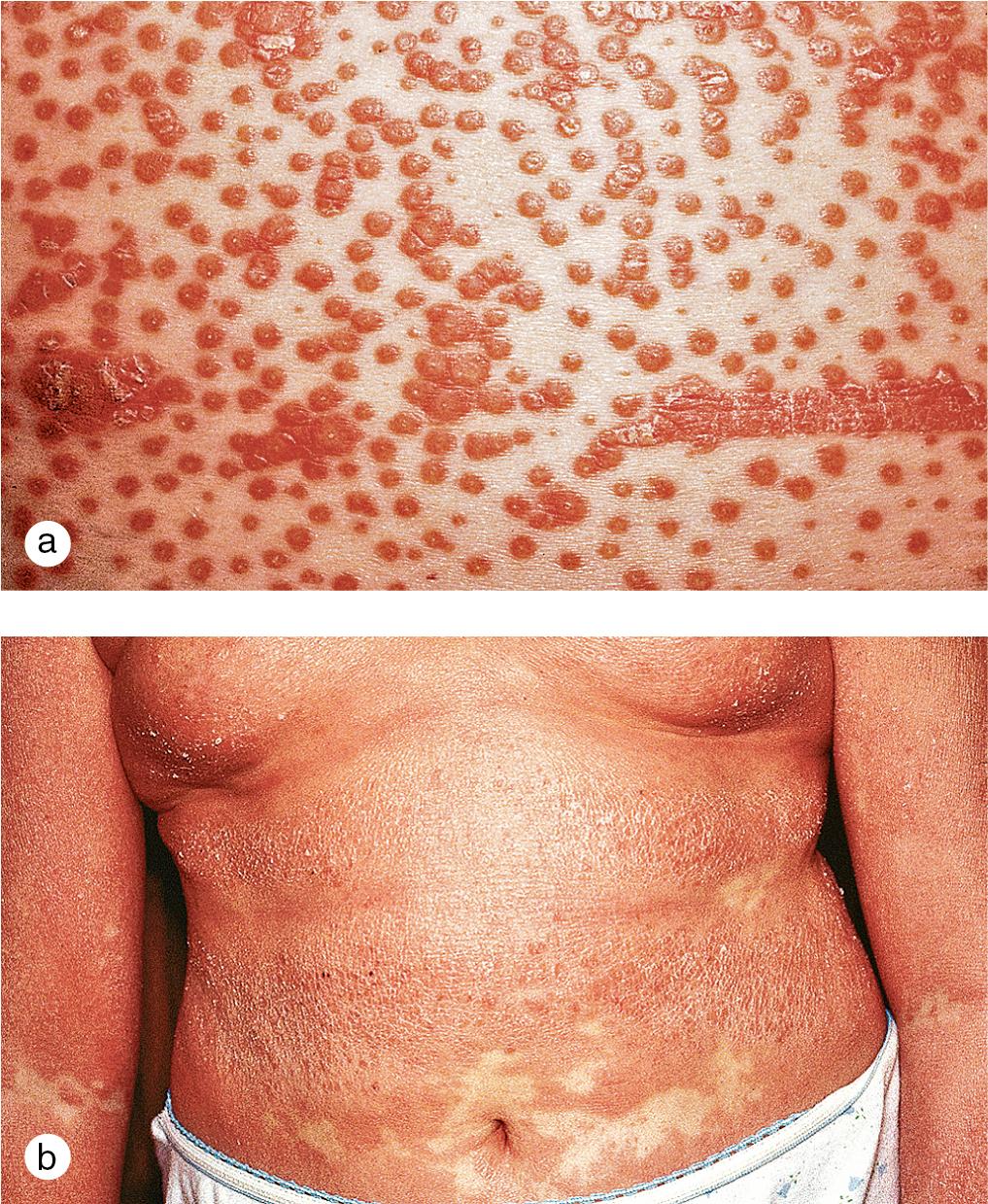
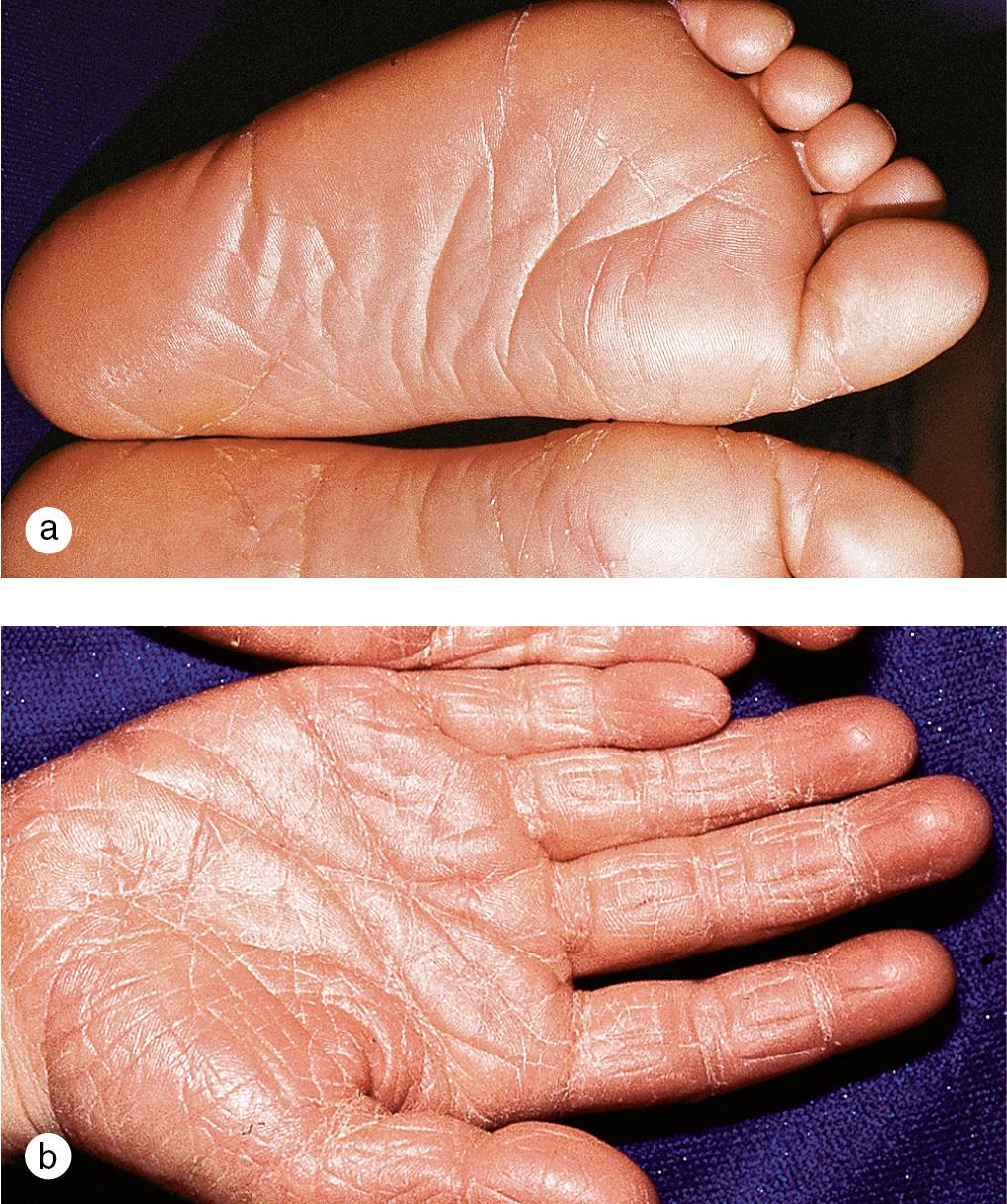
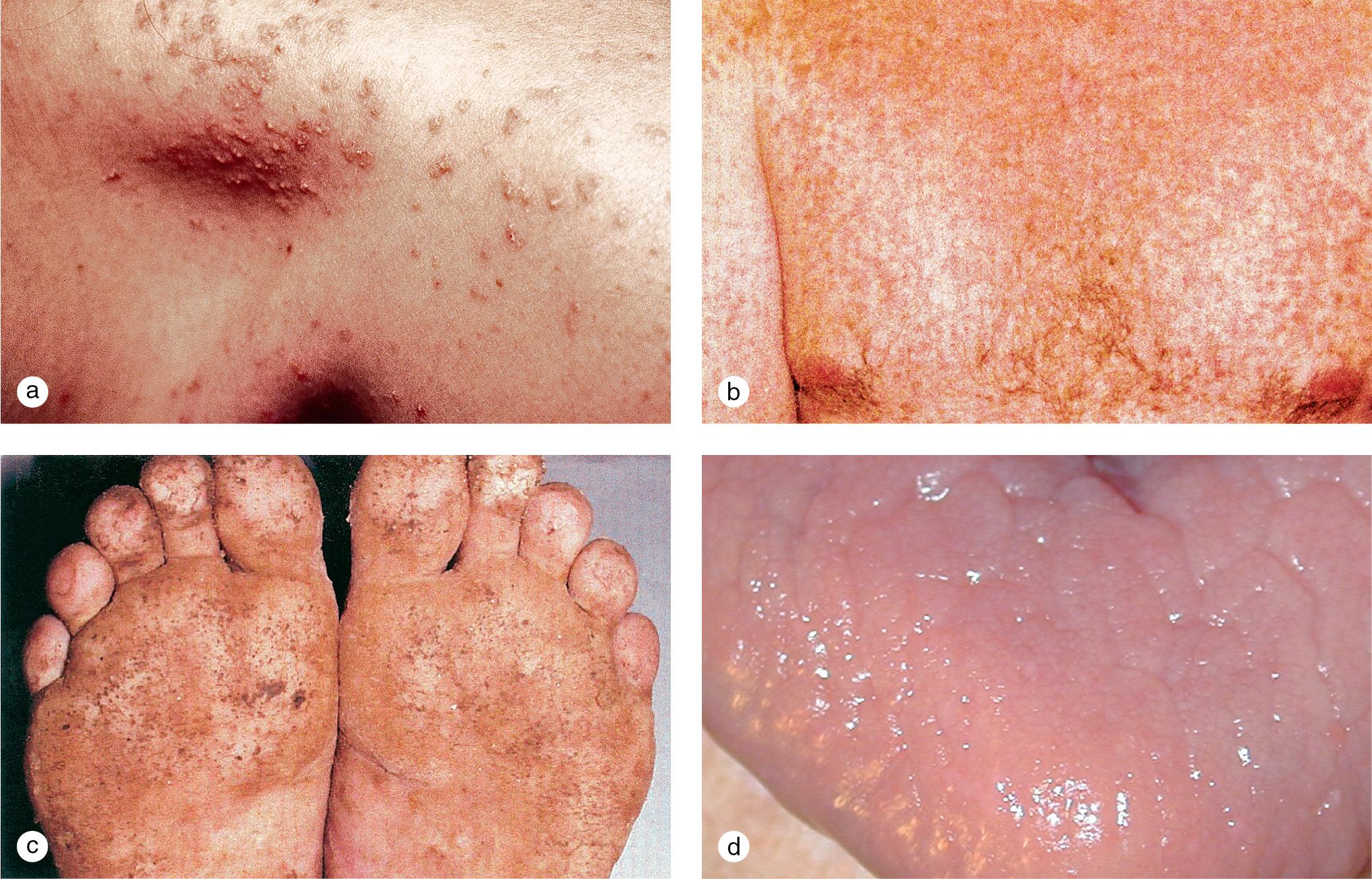
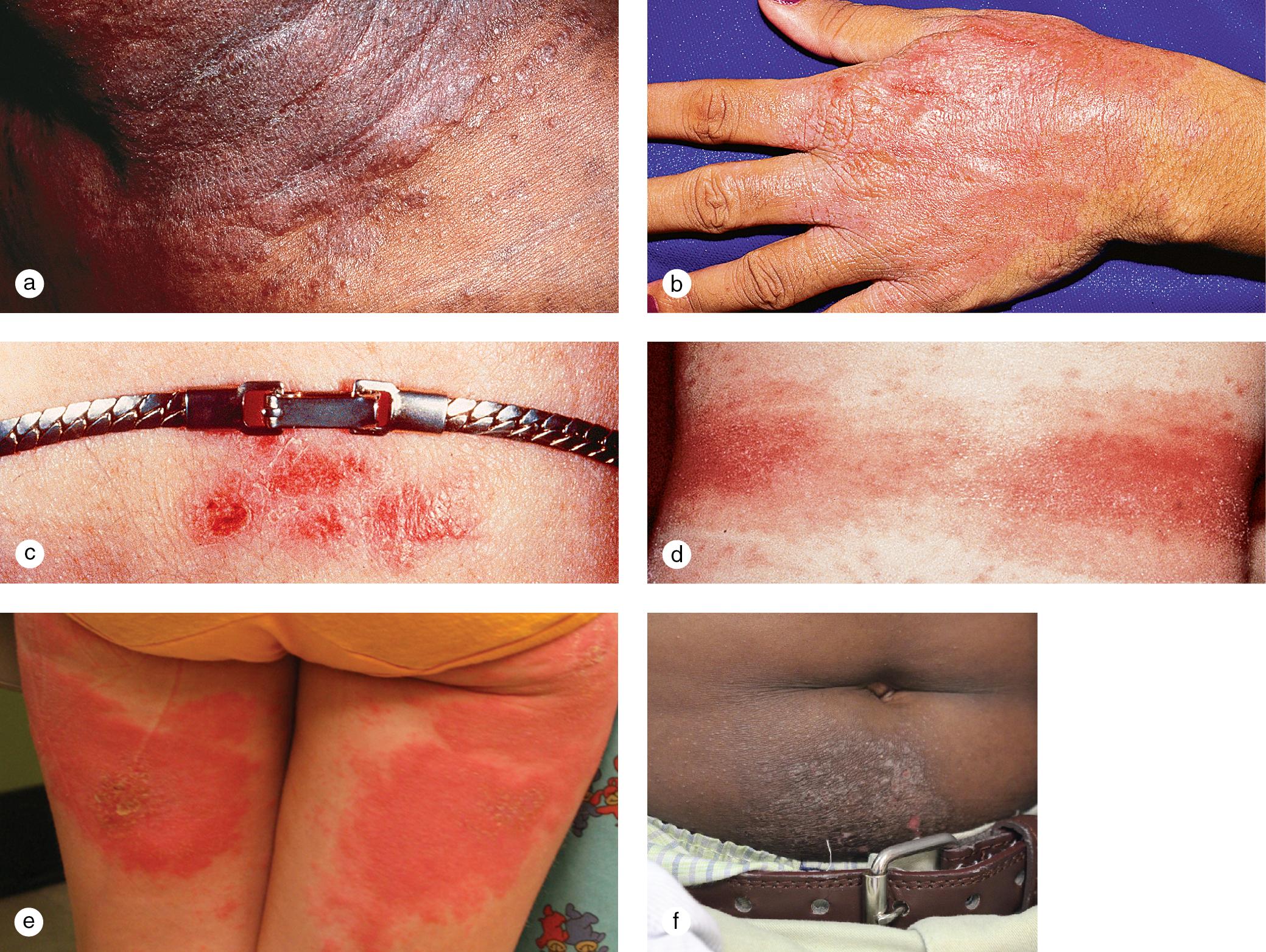
Cutaneous lesions tend to locate on the scalp, the sacrum, and the extensor surfaces of the extremities. About 50% of children present with large plaques over the knees and elbows. Thickening and fissuring of the skin of the palms and soles (keratoderma) may also be present ( Fig. 3.1 c,d). In one-third of children, many drop-like lesions (guttate psoriasis) are scattered over the body, including the face, trunk, and extremities ( Fig. 3.1 e). In infancy, psoriasis may present as a persistent diaper dermatitis. In older children, the eyelids, genitals, and periumbilical areas are commonly involved ( Fig. 3.2 ). Scalp disease may develop as an isolated finding but is often seen with other variants ( Fig. 3.1 b). Itchy, red plaques with thick, tenacious scale are often evident at the frontal hairline and around the ears. Nail changes, which may also occur as the first manifestation of psoriasis, include onycholysis (separation of the nail plate from the nail bed to produce “oil drop changes”), nail pitting, yellowing, increased friability, and subungual hyperkeratosis (see Fig. 8.58 a,b).
Of psoriasis patients, close to 10% suffer from psoriatic arthritis, one of the seronegative spondyloarthropathies. In half of these individuals, arthritis develops before the skin rash. Examination of the joints characteristically demonstrates heat, pain, and swelling of multiple joints of the hands and feet, particularly the distal interphalangeal joints. The arthritis tends to be progressive with the development of flexure deformities and contractures. In addition to the typical lesions of plaque psoriasis, patients often demonstrate severe psoriatic involvement of the hands, nails, and feet ( Fig. 3.3 ). The HLA B-27 antigen is usually positive.
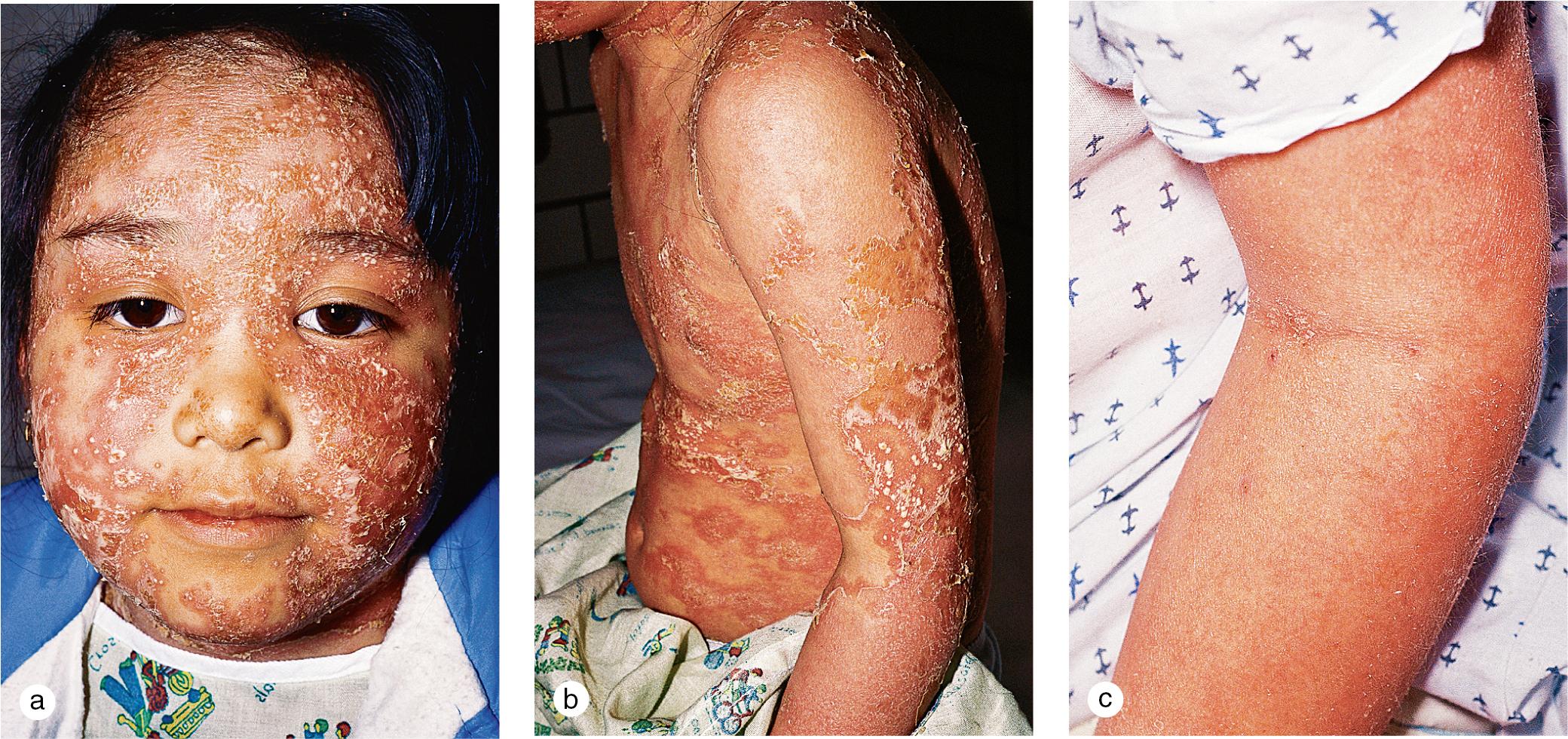
Rarely, children develop erythrodermic psoriasis with acute widespread erythema and scaling or pustular psoriasis with generalized erythema and pustule formation ( Fig. 3.4 ). These variants are associated with high fevers, chills, arthralgias, myalgias, and severe, cutaneous tenderness. Fluid and electrolyte losses and leukocytosis may be marked. Secondary bacterial infection and sepsis can occur.
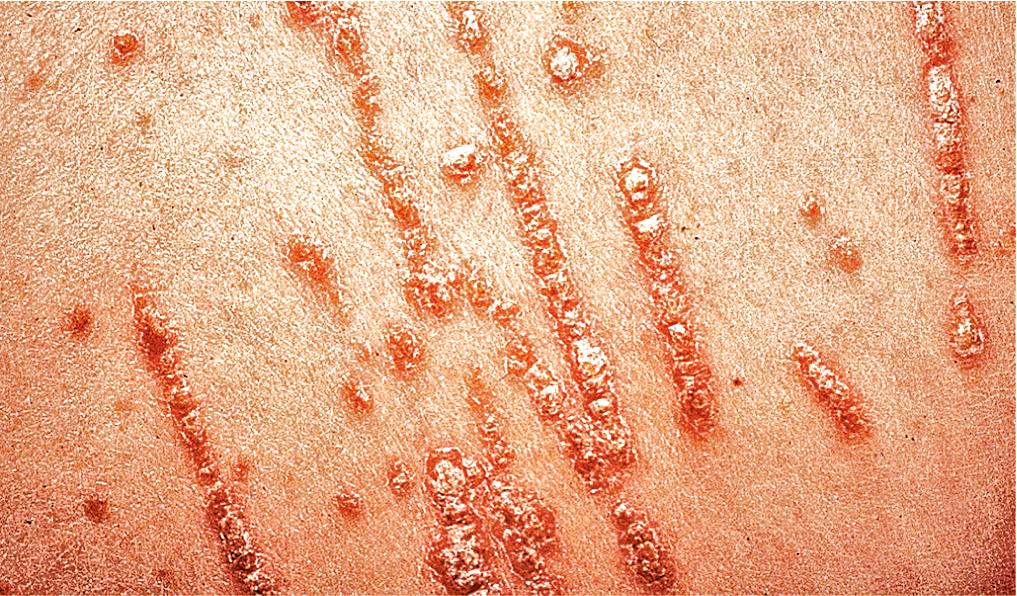
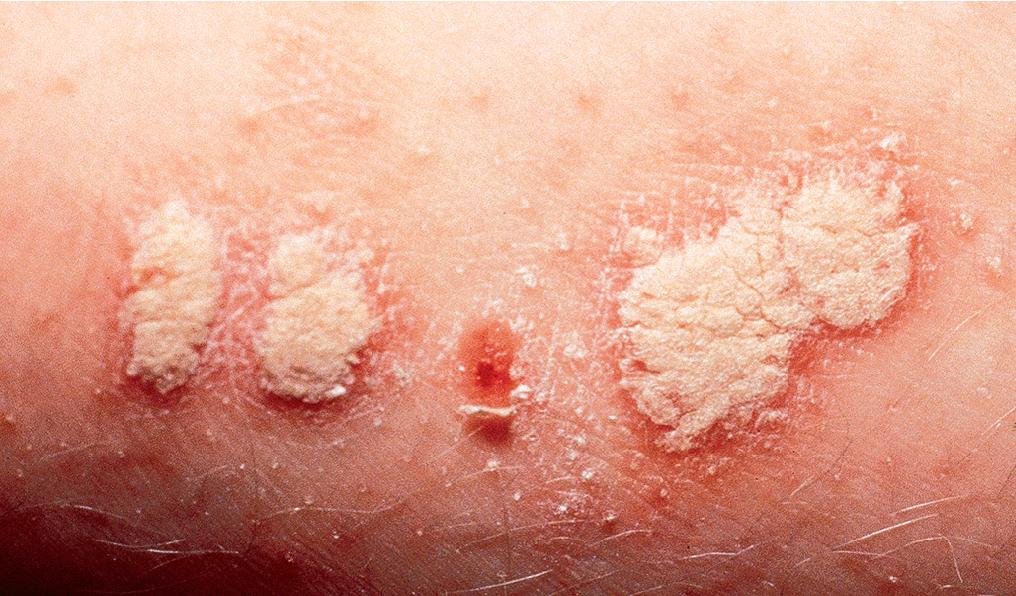
Psoriatic lesions are often induced in areas of local injury, such as scratches, surgical scars, or sunburn, a response termed the Koebner or isomorphic phenomenon ( Fig. 3.5 ). In areas of thick scale, tortuous capillary loops proliferate close to the surface. Gentle removal of the scale results in multiple, small bleeding points, referred to as the Auspitz sign ( Fig. 3.6 ).
The diagnosis is usually made by identifying the typical morphology and by the distribution of skin lesions. Confirmatory skin biopsy findings include regular thickening of the epidermal rete ridges, elongation and edema of the dermal papillae, thinning of the epidermis overlying tortuous dermal capillaries, absence of the granular layer, parakeratosis, spongiform pustules of Kogoj, and Munro microabscesses. In children, skin biopsies may not always be diagnostic, particularly in early disease and certain anatomic sites such as the keratoderma of the palms and soles.
The course of psoriasis is chronic and unpredictable, marked by remissions and exacerbations. A number of different topical agents, which include lubricants, corticosteroids, tar, dithranol (anthralin), and keratolytics, are useful in managing cutaneous lesions. Topical calcipotriol (and other vitamin D3 analogs), tazarotene (a member of the acetylenic class of retinoids), and topical corticosteroids with ultra-high potency may also play a role in the treatment of thick, localized plaques in some children. Occlusive dressings and pulsed-dye laser, which targets tortuous vessels at the base of psoriatic plaques, may be useful in localized disease. Disseminated, chronic or recalcitrant disease may require ultraviolet (UV) light therapy (broadband UVB 290–320 nm, narrowband UVB 311–313 nm, psoralen photosensitizer plus UVA [PUVA] 320–400 nm). Where available, narrowband UVB light therapy has replaced broadband UVB and PUVA in children because of improved safety and efficacy comparable with PUVA. Life-threatening erythrodermic and pustular psoriasis and psoriatic arthritis usually respond to oral retinoids (acitretin) and antimetabolites (methotrexate). Cyclosporine, an immunosuppressant that inhibits T-cell activation has been approved for use in adults with severe psoriasis and may be useful in children with severe, recalcitrant, disfiguring, incapacitating, or life-threatening disease. Recent studies with systemic tacrolimus and pimecrolimus have also shown these agents to be highly effective, but further data are needed on long-term safety. Both calcineurin inhibitors are also available in a topical formulation that may be effective for treating selective patients with facial and intertriginous (inverse) psoriasis. The use of systemic agents requires close laboratory and clinical monitoring.
A relatively new group of pathogenesis-based treatments capable of selective disruption of the psoriatic cascade show promise for management of severe disease in children. Apremilast is a small molecule inhibitor of phosphodiesterase 4, and the anti-tumor necrosis factor agents (adalimumab, etanercept, and infliximab), interleukin (IL)-17 signaling blockers (brodalumab, ixekizumab, and secukinumab), anti-IL-23 (guselkumab, risankizumab, and tildrakizumab), and the monoclonal antibodies directed against IL 12–23 pathway (ustekinumab) have been shown to be effective in adults. Because safety and efficacy data of these various classes are accumulating for the treatment of psoriasis in pediatric patients, the American Academy of Dermatology and the National Psoriasis Foundation issued joint recommendation guidelines for the management and treatment of psoriasis in this special cohort in late 2019. Etanercept and ustekinumab are U.S. Food and Drug Administration (FDA) approved for patients with psoriasis 4 years and older and 12 years and older, respectively. Recently, the manufacturers of ixekizumab received FDA approval for the treatment of the pediatric cohort from ages 6 to 18 with moderate to severe plaque psoriasis. In Europe, etanercept and ustekinumab along with adalimumab are also approved. Several other biologics are used off-label both here and abroad.
Pityriasis rubra pilaris (PRP) is an uncommon disorder of keratinization characterized by small follicular papules, widespread orange-red, scaly plaques surrounded by islands of spared skin, and marked thickening of the skin on the palms and soles ( Fig. 3.7 and Fig. 3.8 ). Onset of disease occurs most commonly in prepubertal children and in adults over 50 years old, and it has been associated with trauma, acute, self-limited illness, and immunocompromised states such as HIV. Most cases are sporadic and acquired, but a familial variant has been reported. Overall, 75% of cases resolve spontaneously within 3–4 years, but in familial disease, persistence is the rule.
In childhood, the circumscribed variant, which accounts for a majority of the cases, begins with the development of coalescing hyperkeratotic papules on the elbows and knees and a palmoplantar keratoderma ( Fig. 3.8 ). Superficial, red, scaly plaques occasionally appear on the face and trunk. Less commonly, children develop a pattern that mimics the classic adult variety. The eruption begins with follicular, hyperkeratotic patches on the back, chest, and abdomen, which expand to involve interfollicular skin. Lesions on the scalp and other sebaceous areas develop simultaneously or soon thereafter and may disseminate. Even in widespread disease, islands of normal appearing spared skin are characteristic. Discrete, hyperkeratotic papules may also remain present over the knuckles, wrists, elbows, and knees. In erythrodermic PRP, facial edema and scale may result in ectropion formation. Nail dystrophy with subungual hyperkeratoses may also be present.
The clinical presentation, particularly with keratoderma of the hands and feet and follicular papules, helps differentiate PRP from psoriasis, seborrheic dermatitis, atopic dermatitis, and pityriasis rosea. A skin biopsy that demonstrates interfollicular orthohyperkeratosis and perifollicular parakeratosis is typical, but not diagnostic.
Although PRP is usually self-limited in childhood, severe disabling disease may require systemic therapy with retinoids or methotrexate. Variable response to therapy has been reported with topical vitamin D analogs, phototherapy, and systemic immunosuppressive agents including steroids, cyclosporine, and azathioprine. Recently, there has been increasing clinical experience with the systemic biologic agents in adults and children with this disorder (see section on psoriasis).
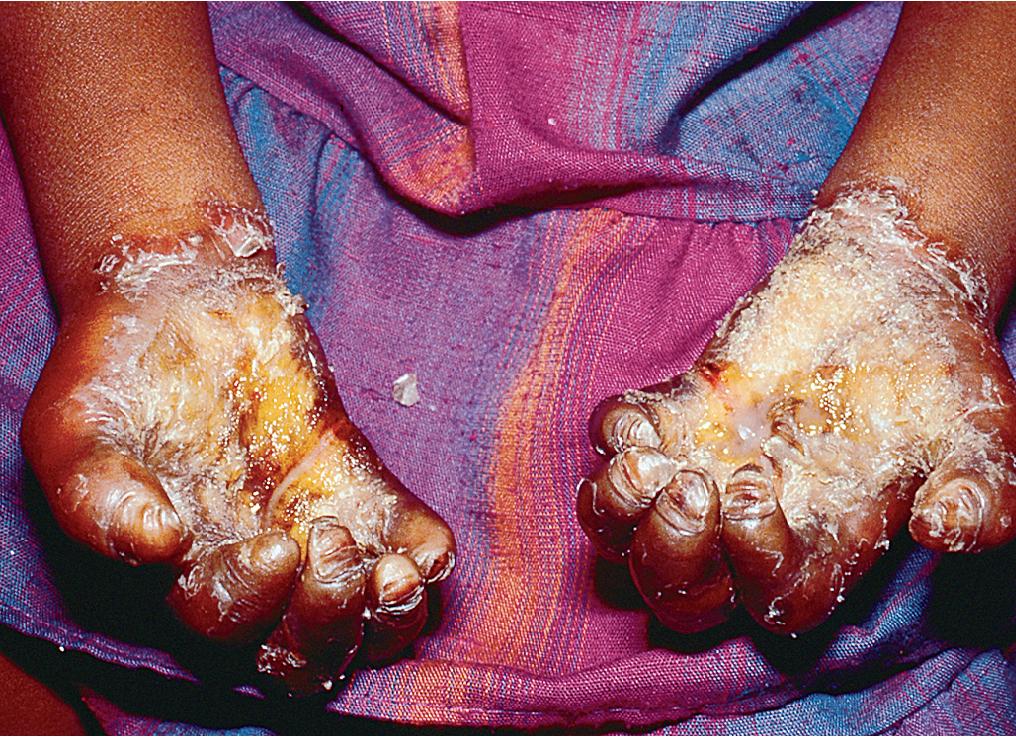
Keratosis follicularis is an autosomal-dominant disorder that typically presents in children from 8 to 15 years old and is characterized by hyperkeratotic follicular papules on the face, scalp, neck, and seborrheic areas of the trunk ( Fig. 3.9 ). Although the onset is usually insidious, a rapidly progressive course may follow an inciting event, such as intense sun exposure or a viral infection. Red, scaly papules coalesce to form widespread, thick, odoriferous, greasy plaques, particularly on the scalp and forehead and around the ears, shoulders, mid-chest, and mid-back. Flexures may also be involved, with moist vegetative plaques.
Other characteristic lesions include flat-topped, warty papules on the dorsum of the hands and tiny hyperkeratotic papules and pits on the palms and soles. Subtle pebbly papules on the oral mucosa may simulate leukoplakia. Nail dystrophy, with thickening or thinning of the nail plate, fracture of the distal nail plate, longitudinal white and red streaks, and subungual hyperkeratosis, may also be present.
The genetic defect in Darier disease has been mapped to chromosome 12q23–q24.1, which results in dysfunction of an endoplasmic reticulum Ca 2+ ATPase (SERCA2) and subsequent disruption of intracellular Ca 2+ signaling. Although the cause is unclear, there is also a predisposition to certain bacterial, viral, and fungal infections. The tendency of affected individuals to develop disseminated herpes simplex (Kaposi varicelliform eruption) and recurrent staphylococcal infections has been recognized for years.
The characteristic clinical picture permits easy differentiation of Darier disease from other papulosquamous disorders, such as seborrheic dermatitis, PRP, and psoriasis. Classic histologic changes from skin biopsy, which include dyskeratosis (with the formation of corps ronds and corps grains), suprabasal acantholysis (leading to the formation of suprabasal clefts), and the formation of villi by upward proliferating dermal papillae, confirm the diagnosis.
Although Darier disease tends to persist throughout life, many patients experience episodic flares and remissions in disease activity. Topical vitamin A acid may be helpful in managing early lesions. However, its use is limited by a high risk of irritation. Experience with oral retinoids has been promising, but prolonged use may be associated with unacceptable complications, which include hyperostosis, epiphyseal plate changes, increased skin fragility, and teratogenicity. Aggressive protection from the sun and treatment of secondary bacterial and viral infections also control exacerbations.
The ichthyoses are a heterogeneous group of scaling disorders characterized by retention hyperkeratosis (ichthyosis vulgaris, X-linked ichthyosis) or increased proliferation of epidermal cells (lamellar ichthyosis, epidermolytic hyperkeratosis). These diseases may be differentiated by clinical findings, histopathology, biochemical markers, and specific genetic mutations, as outlined in Chapter 2 .
Keratoderma is a heterogeneous group of disorders of keratinization defined by the presence of focal or generalized thickening of the skin of the palms and/or soles. Although difficult to characterize, keratodermas can be classified based on whether they are acquired or inherited, isolated or associated with other cutaneous or systemic findings, and specific morphologic features and extent of involvement of lesions on the palms and soles ( Table 3.1 ). Inherited keratodermas can be subdivided based on the mode of inheritance, presence of other ectodermal and non-ectodermal findings, and specific genetic and molecular markers. The astute clinician can often make a specific diagnosis by searching for key findings in the Online Mendelian Inheritance in Man database supported by the National Library of Medicine and the Johns Hopkins University School of Medicine.
| Type | Disorder | Genetics | Onset | Involvement | Hyperhidrosis | Associated Findings |
|---|---|---|---|---|---|---|
| Diffuse palmoplantar keratoderma | Unna–Thost syndrome |
|
Infancy | Palms, soles | Severe |
|
| Keratoma hereditarium mutilans (Vohwinkel syndrome) |
|
Infancy | Honeycomb keratoderma; star-shaped plaques on hands and feet, elbows, knees | ± |
|
|
| Mal de Meleda keratoderma |
|
Infancy | Palms, soles, elbows, knees, including dorsal surfaces (transgridiens) | ± |
|
|
| Tylosis (Howel–Evans syndrome) |
|
Adolescence | Soles, sometimes palms | ± |
|
|
| Papillon–Lefèvre syndrome | Autosomal recessive Mutation on 11q14.2 |
Birth to 2–3 years | Soles more severe than palms, intense erythema, wrists, ankles, elbows, knees; follicular hyperkeratoses | ± | Nail dystrophy; periodontitis; calcification of falx cerebri; mental retardation; arachnodactyly | |
| Olmsted syndrome |
|
Infancy | Progressive, diffuse involvement of hands, feet, leads to flexion contractures, digital amputation | (Anhidrosis) | Periorificial hyperkeratosis Oral leukokeratosis |
|
| Focal palmoplantar keratoderma | Punctate palmoplantar keratoderma | Autosomal dominant (2–5% African Caribbean individuals) | Childhood, adolescence | Palms, soles | – | Variable |
| Tyrosimia type II (oculo-cutaneous tyrisonosis, Richner–Hanhart syndrome) | Autosomal recessive 16q22.2 | Infancy through adulthood | Fingertips, palms | Corneal ulcerations; mental retardation | ||
| Porokeratosis palmar, plantar, and disseminated 1 | Autosomal dominant (rare) | Childhood through adulthood | Extremities, trunk | – | Squamous cell carcinoma, increased with immuno-suppression |
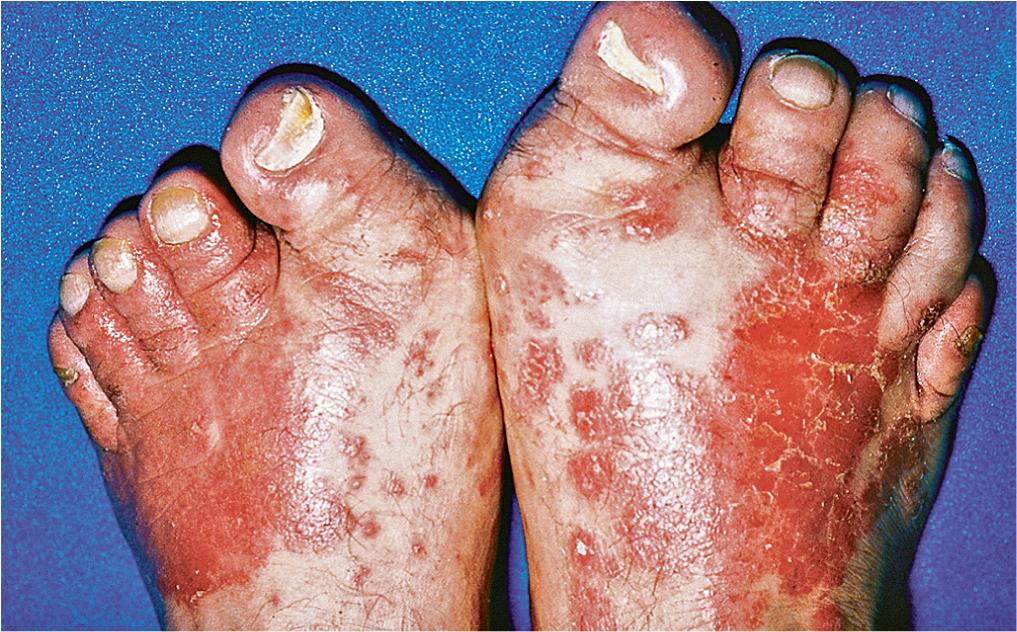
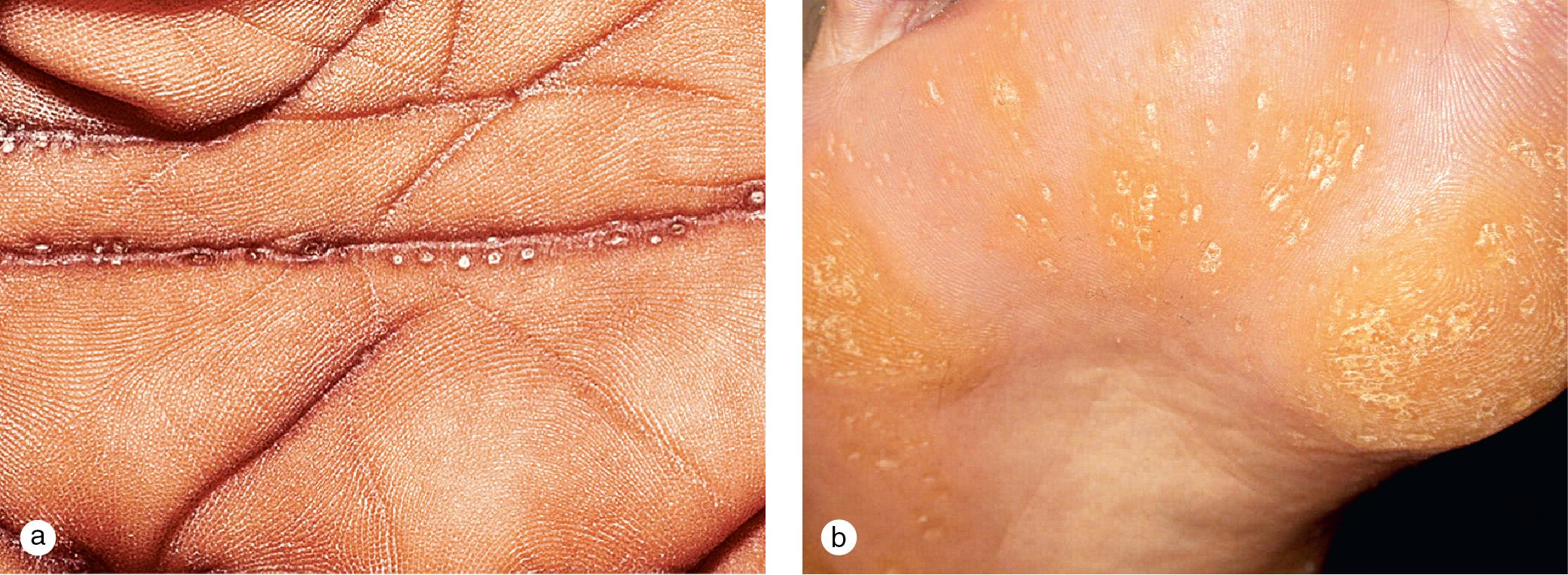
Unna-Thost (non-epidermolytic type), the most common genetic variant, is inherited in an autosomal dominant pattern and is associated with a defect in type 1 and type 16 keratins. This keratoderma presents in the first year of life with diffuse hyperkeratosis restricted to the palmar and plantar surfaces ( Fig. 3.10 ). Although lesions may be asymptomatic, hyperhidrosis may lead to maceration and the formation of painful fissuring, blisters, and bacterial superinfection. Unna-Thost must be differentiated from a number of unusual keratodermas associated with hyperkeratoses that extend to the dorsal surfaces of the hands and feet, elbows, and knees and develop at distant sites ( Fig. 3.11 ).

Focal keratoderma with discrete papules and plaques on the palms and soles also appears as an isolated phenomenon or in association with other cutaneous findings and systemic disease. A mild autosomal-dominant variant with pits and hyperkeratotic papules in the hand and foot creases occurs in 2–5% of black individuals ( Fig. 3.12 ). Occasionally, this keratoderma is painful and requires surgical treatment.
Neurosensory deafness, carcinoma of the esophagus (Howel–Evans syndrome), and peripheral neuropathy (Charcot–Marie–Tooth disease) are rarely associated with palmoplantar keratoderma and may be excluded by auditory screening and a careful family history.
Hyperkeratosis of the palms and soles that presents as part of a diffuse disorder of keratinization, such as psoriasis or PRP, is differentiated by a thorough examination of the integument for other diagnostic clinical findings. Unfortunately, it may be difficult to make a specific diagnosis until other features of the underlying disorder manifest.
Porokeratosis is a disorder of keratinization characterized by annular, sharply demarcated plaques with raised hyperkeratotic borders ( Fig. 3.13 ). Although many rare subtypes have been described, four distinctive variants are recognized, based on morphology and distribution of the lesions, time of onset, triggering factors, mode of inheritance, and, in some instances, specific genetic mutations.
Subtle, reddish-brown, scaly papules that slowly enlarge to form irregularly shaped plaques with hypopigmented atrophic centers and raised, grooved (double-edged) borders are typical of this condition, which invariably develops in childhood ( Fig. 3.13 a–c). Plaques are usually unilateral and vary in size from a few millimeters to several centimeters in diameter. One to three lesions are usually present. This rare, autosomal-dominant variant appears most commonly on the extremities, thighs, and perigenital skin, although any area that contains mucous membranes can be involved. Biopsies of the longitudinal furrow in the border demonstrate the diagnostic coronoid lamella, which consists of a parakeratotic column that fills a deep, epidermal invagination.
This variant has been associated with clonal chromosomal abnormalities in chromosome 3 in the region 3p14–p12. Chromosomal instability may rarely result in the development of squamous cell carcinoma within these slowly progressive lesions. Although excision or carbon dioxide laser surgery may be useful, recurrences have been reported.
This variant presents much like the Mibelli variant. However, plaques are linearly arranged on the distal extremities and trunk, where they demonstrate a zosteriform pattern. Onset is also in early childhood, but the mode of inheritance has not been established.
Disseminated superficial actinic porokeratosis (DSAP) is a common autosomal-dominant variant with delayed expression, found primarily in lightly pigmented individuals of Celtic extraction. Many small, 2–4 mm diameter, hyperkeratotic papules appear symmetrically on sun-exposed surfaces of the extremities during the second and third decades of life ( Fig. 3.13 d). These brown-, red-, or skin-colored lesions may coalesce to form irregular, circinate patterns. Progression of papules, which occurs particularly during the summer months, can be slowed by aggressive use of sun protection. A mutation in some families with DSAP has been identified at 12q23–24.1.
Punctate porokeratosis affects the palms and soles and has been described as a distinct entity. However, it usually presents in association with the Mibelli or linear variant. Punctate porokeratosis may also occur as a widely disseminated form reminiscent of disseminated superficial actinic porokeratosis, but with involvement of both sun-exposed and sun-protected areas. It is included in the differential diagnosis of punctate keratoderma.
Many of these disorders demonstrate changes of both acute (erythema, edema, vesiculation, crusts) and chronic (scaling, lichenification, hypopigmentation, hyperpigmentation) inflammation. Microscopically, dermatitis is recognized by the presence of intercellular edema (spongiosis), variable epidermal thickening (acanthosis), and the presence of dermal inflammatory cells, usually lymphocytes. Research and development of a number of new medications designed to selectively inhibit the dermatitic inflammatory cascade have led to more specific understanding of the molecular basis of these disorders. Various dermatitides may be differentiated by clinical features and specific histologic patterns.
A reasonable way to think of these conditions is as exogenous (“outside job”) versus endogenous (“inside job”) phenomena. Exogenous disorders include irritant and allergic contact dermatitis and photodermatitis. Endogenous dermatitides include atopic dermatitis, dyshidrotic eczema, nummular dermatitis, and seborrheic dermatitis. Juvenile palmar and plantar dermatosis and perioral dermatitis are triggered by a combination of inside and outside factors. Pityriasis rosea and parapsoriasis present with distinctive dermatitic patterns, but their causes are unclear.
Contact dermatitis refers to a group of conditions in which an inflammatory reaction in the skin is triggered by direct contact with environmental agents. In the most common form, irritant contact dermatitis, changes in the skin are induced by caustic agents such as acids, alkalis, hydrocarbons, and other primary irritants. Anyone exposed to these agents in a high enough concentration for a long enough period of time develops a reaction. The rash is usually acute (occurring within minutes), with itching or burning, well-demarcated erythema, edema, blister formation, and/or crust formation.
By contrast, allergic contact dermatitis is a T-cell–mediated immune reaction to an antigen that comes into contact with the skin. In the initial phase of the reaction, antigens are processed by Langerhans cells in the epidermis under the influence of keratinocyte-derived cytokines. T-cells subsequently migrate to regional lymph nodes where there is clonal expansion of the specifically sensitized lymphocytes. Elicitation of an allergic response occurs on subsequent exposure to the allergen with proliferation of T lymphocytes in the skin and regional lymph nodes.
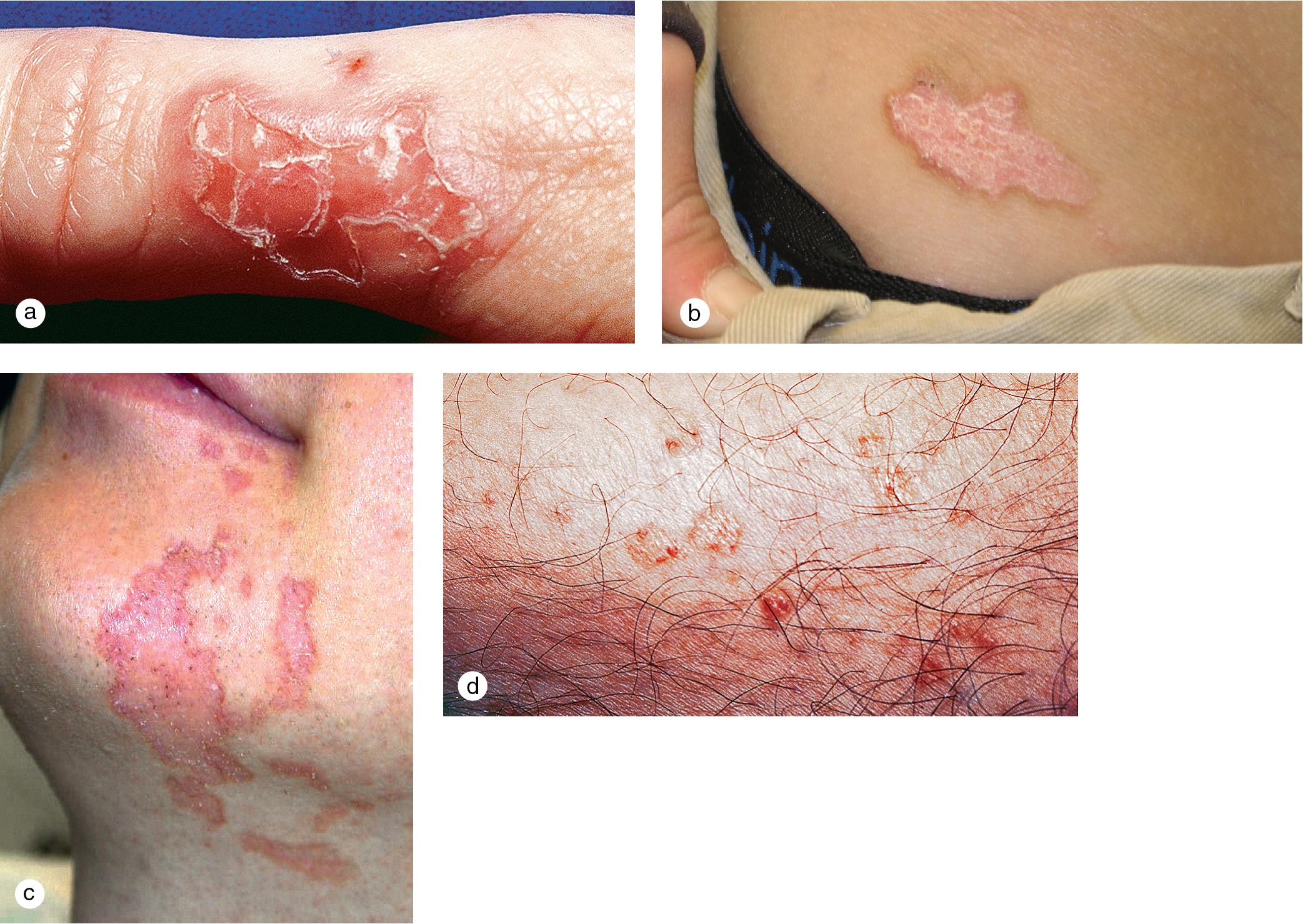
Although contact dermatitis frequently presents with dramatic onset of erythema, vesiculation, and pruritus, the rash may become chronic with scaling, lichenification, and pigmentary changes ( Fig. 3.14 ). At times the allergen is obvious, such as poison ivy or nickel-containing jewelry. Often, however, some detective work or formal patch testing is required to elicit the inciting agent ( Fig. 3.14 f). The initial reaction occurs after a period of sensitization of 7–14 days in susceptible individuals. Once sensitization has occurred, re-exposure to the allergen provides a more rapid reaction, sometimes within hours. This is a classic example of a type IV delayed-hypersensitivity response.
The most common allergic contact dermatitis in the United States is poison ivy or rhus dermatitis, which typically appears as linear streaks of erythematous papules and vesicles ( Fig. 3.15 ). However, with heavy exposure or in exquisitely sensitive individuals, the rash may appear in large patches. When lesions involve the skin of the face or genitals, impressive swelling can occur and obscure the primary eruption. Direct contact with the sap of the plant (poison ivy, poison oak, or poison sumac), whether from leaves, stems, or roots, produces the dermatitis ( Fig. 3.16 ). Indirect contact, such as with clothing or pets that have brushed against the plant, with logs or railroad ties on which the vine has been growing, or with smoke from a fire in which the plant is being burned, is another means of exposure. Areas of skin exposed to the highest concentration of rhus antigen develop the changes first. Other sites that have received lower doses react in succession, which gives the illusion of spreading. However, once on the skin, the allergen becomes fixed to epithelial cells within about 20 min and cannot be spread further. Thorough washing within minutes of exposure may prevent or reduce the eruption. Barrier creams applied before exposure may afford some protection.
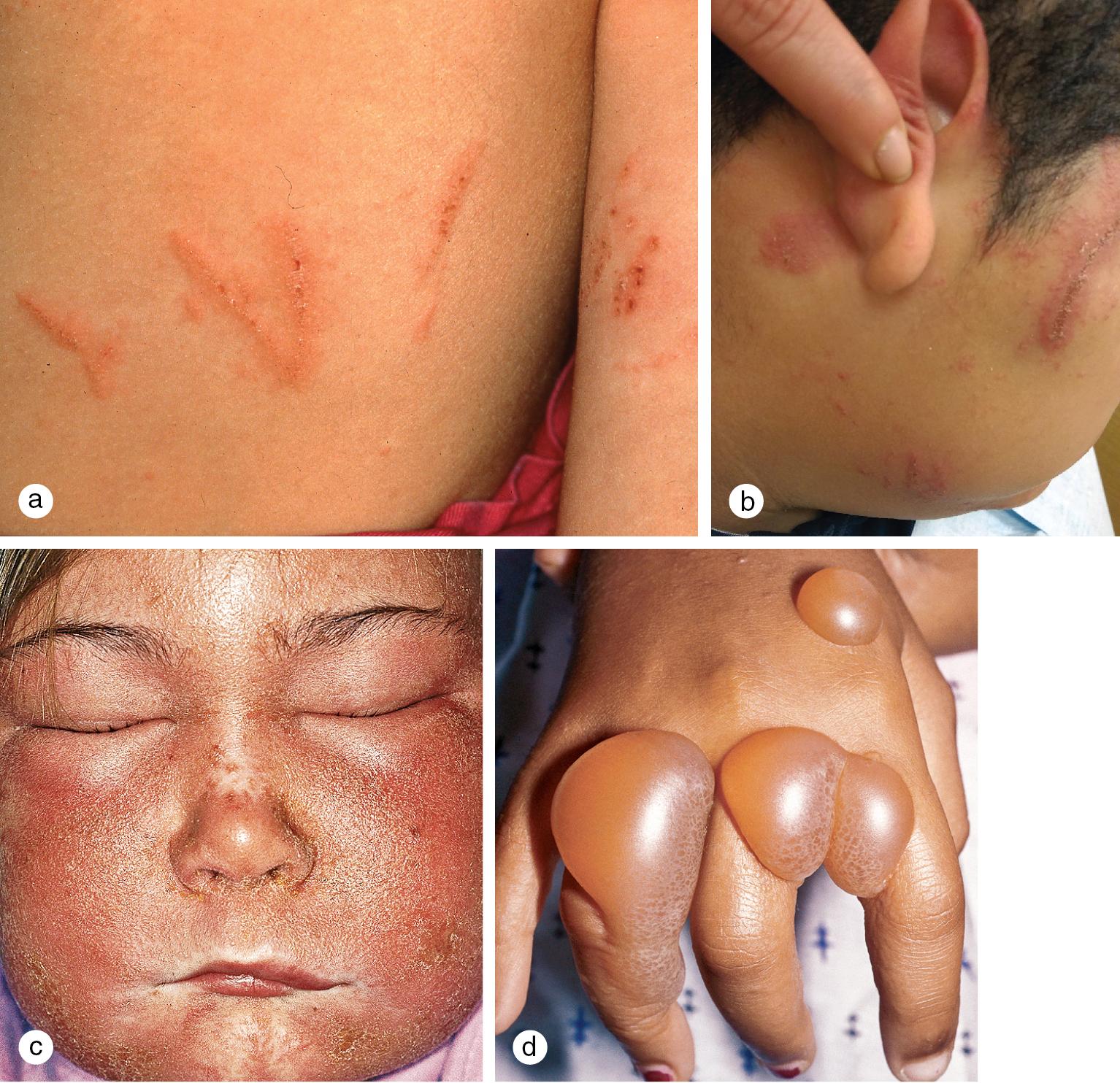

Other common contact allergens include nickel, formaldehyde resin, wool alcohols, fragrance mix, and cobalt chloride. Several studies using standard patch-test techniques show that the prevalence of contact sensitivity to these allergens in children is similar to that of adults. Moreover, patch testing in children is safe and effective, and clinically significant findings may exceed 70% in carefully selected patients.
Some allergens, known as photosensitizers, require sunlight to become activated. Photocontact dermatitis caused by drugs (e.g. tetracyclines, sulfonylureas, and thiazides) characteristically erupts in a symmetric distribution on the face, the “V” of the neck, and the arms below the shirt sleeves. Topical photosensitizers (e.g. dyes, furocoumarins, halogenated salicylanilides, p-aminobenzoic acid) produce localized patches of dermatitis when applied to sun-exposed sites. These agents are found in cosmetics, sunscreens, dermatologic products, germicidal soaps, and woodland and house plants (see Chapter 7 ).
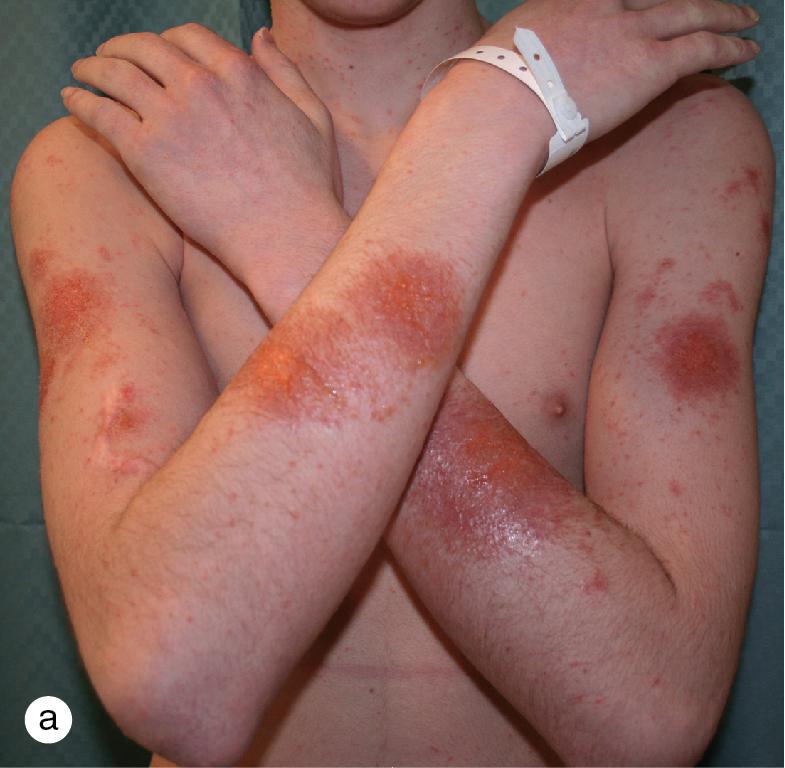
Occasionally, the local reaction in a contact dermatitis is so severe that the patient develops a widespread, immunologically mediated, secondary eczematous dermatitis. When the dermatitis appears at sites that have not been in contact with the offending agent, the reaction is referred to as “autoeczematization” or an “id” reaction ( Fig. 3.17 ).
A careful history usually reveals the source of contact dermatitis. In some patients, patch testing with a standardized tray of common antigens or the suspected antigen and several controls is necessary to confirm the diagnosis.
Although small areas of contact dermatitis are best treated topically, widespread reactions require and respond within 48 h to a 2- to 3-week tapering course of systemic corticosteroid; these begin at 0.5–1.0 mg/kg per day. Patients may experience rebound of the rash when treated with a shorter course. Oral corticosteroids may also be indicated in severe local reactions that involve the eyelids, extensive parts of the face, the genitals, and/or the hands, where swelling and pruritus may become incapacitating.
Also known as eczema, atopic dermatitis is a chronically recurrent, genetically influenced skin disorder of early infancy, childhood, and occasionally adult life. Although it was initially described in the nineteenth century, it was not until 1935 that Hill and Sultzberger first characterized the clinical entity. The term atopy , derived from the Greek word that means “not confined to a single place,” was introduced in 1923 by Coca and Cooke to describe a cohort of patients with asthma and allergic rhinitis who demonstrated immediate wheal and flare reactions upon skin testing with a variety of environmental allergens. The sera of these patients contained skin-sensitizing antibodies that were subsequently characterized as IgE immunoglobulins. It was later recognized that these atopic individuals also frequently manifested the itchy, eczematous dermatitis that was labeled atopic dermatitis.
The term eczema , which means “boil over,” is used by many physicians when referring to atopic dermatitis. However, most dermatologists use the word eczematous as a morphologic term to describe the clinical findings in various sorts of acute (erythema, scaling, vesicles, and crusts) and chronic (scaling, lichenification, and pigmentary changes) dermatitis. Both acute and chronic dermatitis may be present in atopic patients at different sites at the same time and the same site at different times during the course of the disease.
Although data are not precise, recent surveys reveal that atopic dermatitis is rather common with an incidence of 10% among Americans. The prevalence is highest among children, affecting 15–20% of all children between 6 months and 10 years of age. Subtle findings may be present during the first few months of life, and nearly 60% of patients have an initial outbreak by their first birthday. Another third develop disease between 1 and 5 years. The onset of eczema in adolescence and adulthood is unusual and should alert the clinician to the possibility of other diagnoses.
In families with a history of allergic rhinitis or asthma, nearly one-third of the children are expected to develop skin lesions of atopic dermatitis. Inversely, in patients with atopic dermatitis, one-third are expected to have a personal history of allergic rhinitis or asthma, with two-thirds showing a family history of these disorders. Half of those who manifest the dermatologic condition in infancy or childhood ultimately develop allergic respiratory symptoms. Atopic dermatitis appears to be linked to the histocompatibility locus antigen (HLA-DRB1), as has been described in allergic rhinitis. Still, it seems to be inherited as an autosomal trait with multifactorial influences.
Many external factors, which include dry skin, soaps, wool fabrics, foods, infectious agents, and environmental antigens, act in concert to produce pruritus in susceptible individuals. The resultant scratching leads to acute and chronic changes typical of atopic dermatitis.
The pathogenesis of inflammation is unclear, and no single factor has been implicated in the initiation of the inflammatory cascade. However, there is evidence that expansion of type 2 cytokine-secreting lymphocytes plays a major role in the acute phase of atopic dermatitis ( Fig. 3.18 ). This results in increased levels of proinflammatory cytokines such as IL-4, -10, and -13, and of IgE and a corresponding decrease in interferon-gamma. Inflammatory dendritic cells have also been shown to increase in atopic skin, which further fuels inflammation and the itch–scratch cycle.
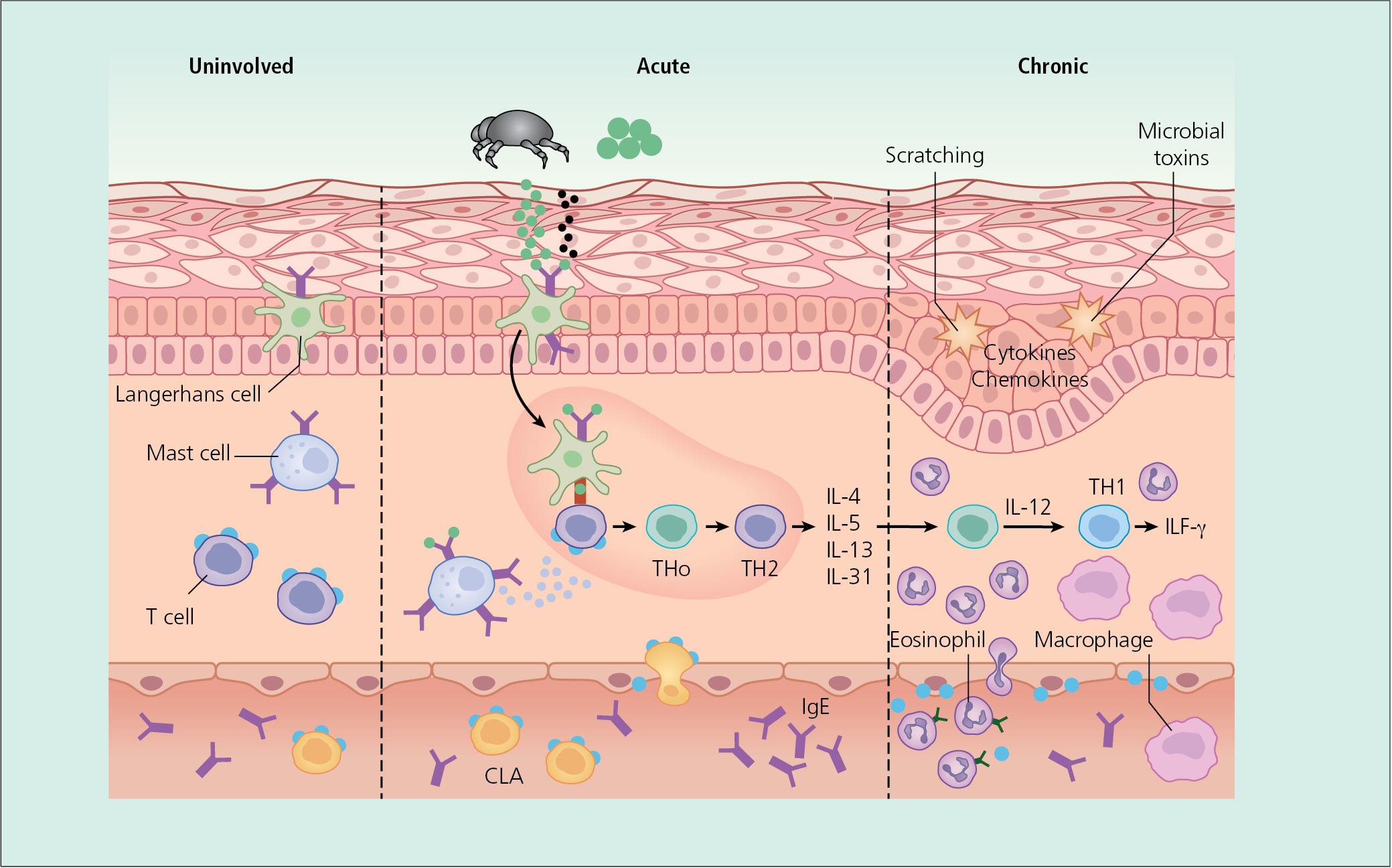
In a subset of atopics, defects in barrier function may play an initiating role. Mutations in the filaggrin gene are associated with the development of ichthyosis vulgaris and barrier dysfunction and an increased risk of developing atopic dermatitis. Predisposition to atopic dermatitis has also been associated with alteration in ceramide content of stratum corneum and disturbed maturation of lamellar bodies, which serve as water-retaining molecules in the barrier.
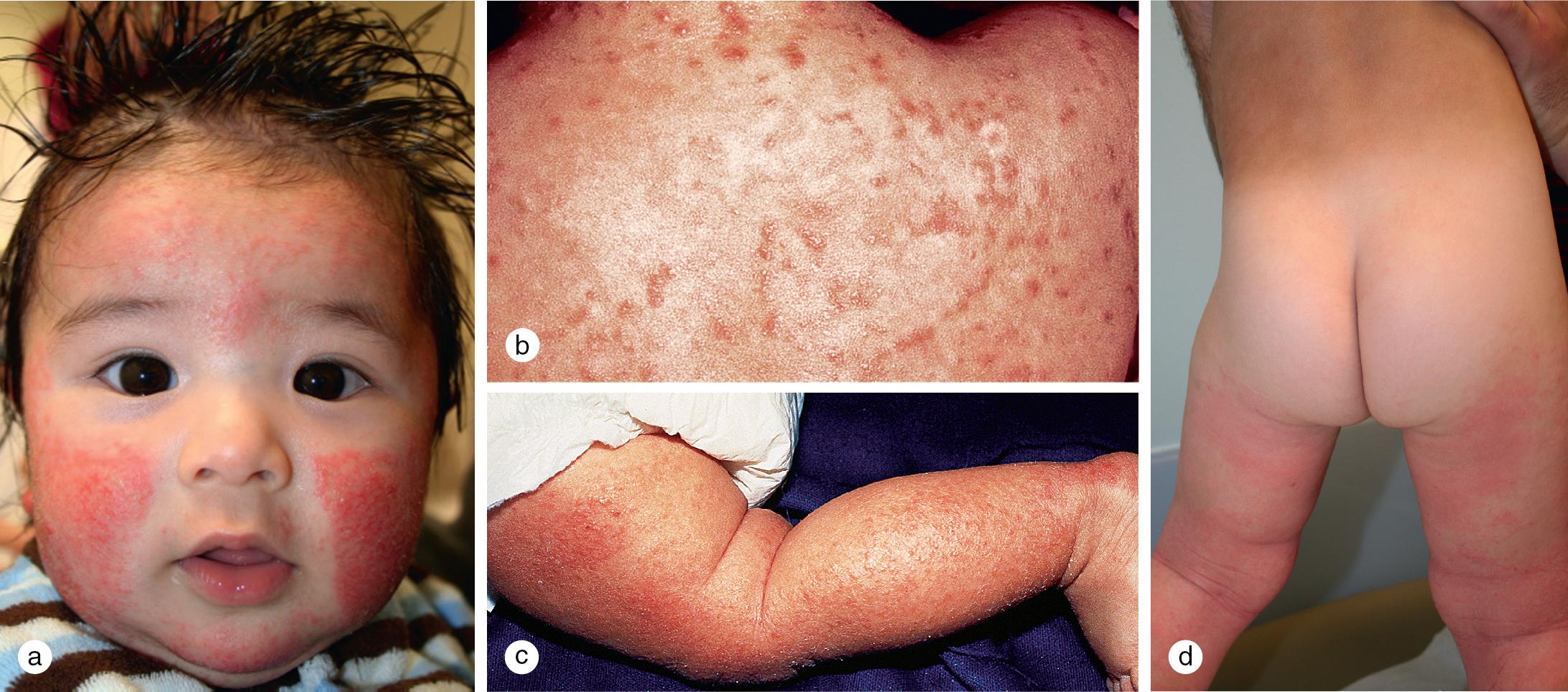
Although there is no one specific laboratory study or clinical finding in atopic dermatitis, over the years, clinicians and investigators have developed a series of major and minor diagnostic criteria ( Table 3.2 ). In addition to a history of pruritus, the distribution, morphology, and evolution of skin lesions in atopic dermatitis follow a characteristic pattern ( Fig. 3.19 ). The infantile phase begins between shortly after birth and 6 months of age and lasts about 2–3 years. Typically, the rash is composed of red, itchy papules and plaques, many of which ooze and crust. Lesions are symmetrically distributed over the cheeks, forehead, scalp, trunk, and the extensor surfaces of the extremities ( Fig. 3.20 ). The diaper area is usually spared.
| Major Criteria (All Required for Diagnosis) | Common Findings (At Least Two) | Associated Findings (At Least Four) |
|---|---|---|
|
|
|
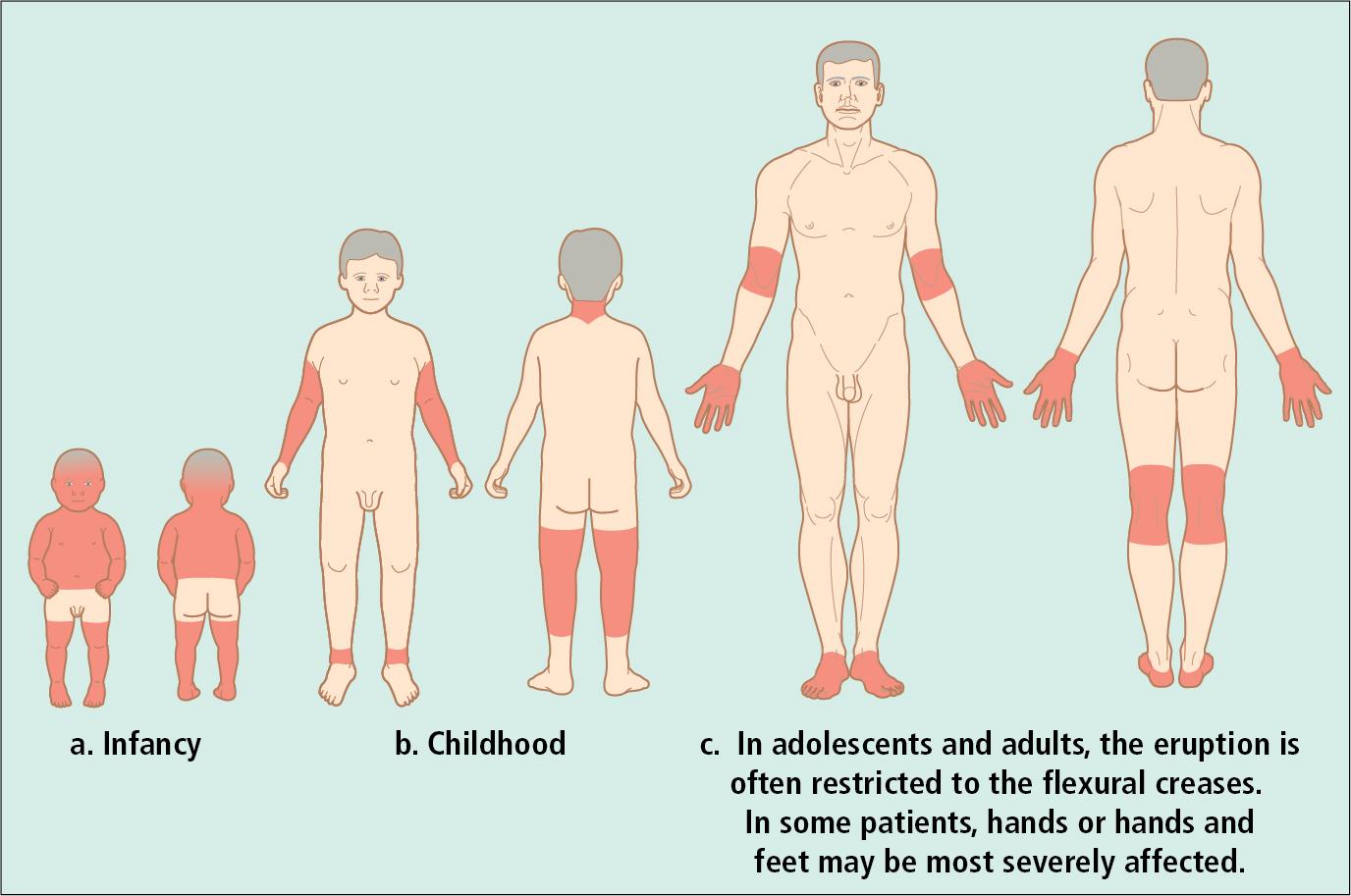
The childhood phase of atopic dermatitis occurs between 4 and 10 years of age. Circumscribed, erythematous, scaly, lichenified plaques are symmetrically distributed on the wrists, ankles, and flexural surfaces of the arms and legs ( Fig. 3.21 ). The ear creases and back of the neck are also commonly involved. These areas develop frequent secondary infections, probably as a result of the introduction of organisms by intense scratching. Although the eruption may become chronic and rarely generalized, remissions may occur at any time. Most children experience improvement during the warm, humid summer months and exacerbations during the fall and winter.
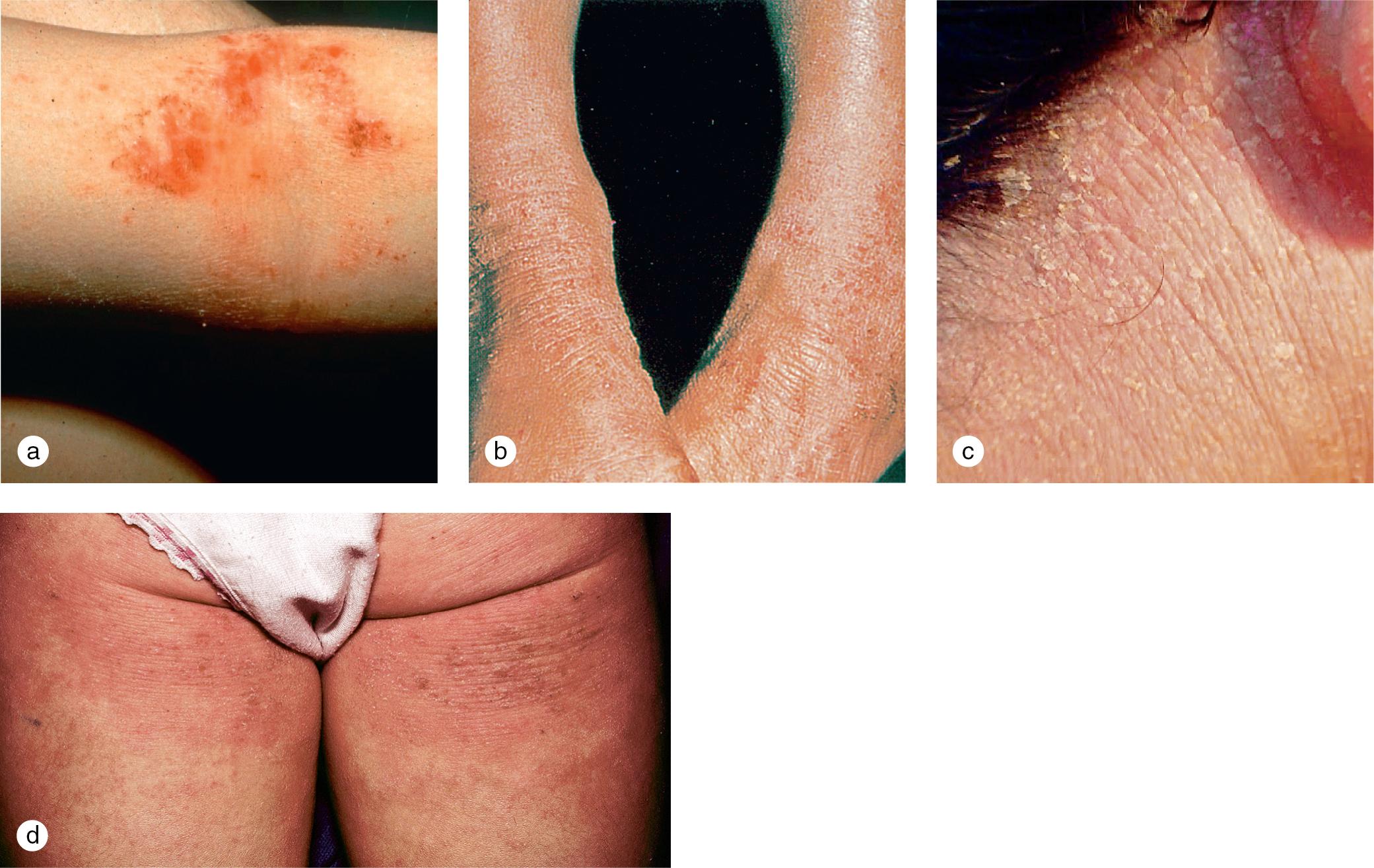
Of children with atopic dermatitis, 75% improve by 10–14 years of age; the remaining individuals may go on to develop chronic adult disease. Major areas of involvement include the flexural creases of the arms, neck, and legs. Chronic dermatitis may be restricted to the hands or feet, but some patients develop recurrent, widespread lesions.
Although nummular eczematous dermatitis was initially described as distinct from atopic dermatitis, clinicians frequently use this term to describe the discrete, coin-shaped, red patches seen on many patients with atopic dermatitis ( Fig. 3.22 ). Lesions typically appear as tiny papules and vesicles, which form confluent patches on the arms and legs. Nummular lesions may be extremely pruritic, and they are difficult to treat, particularly during the winter months, when the incidence seems to peak.
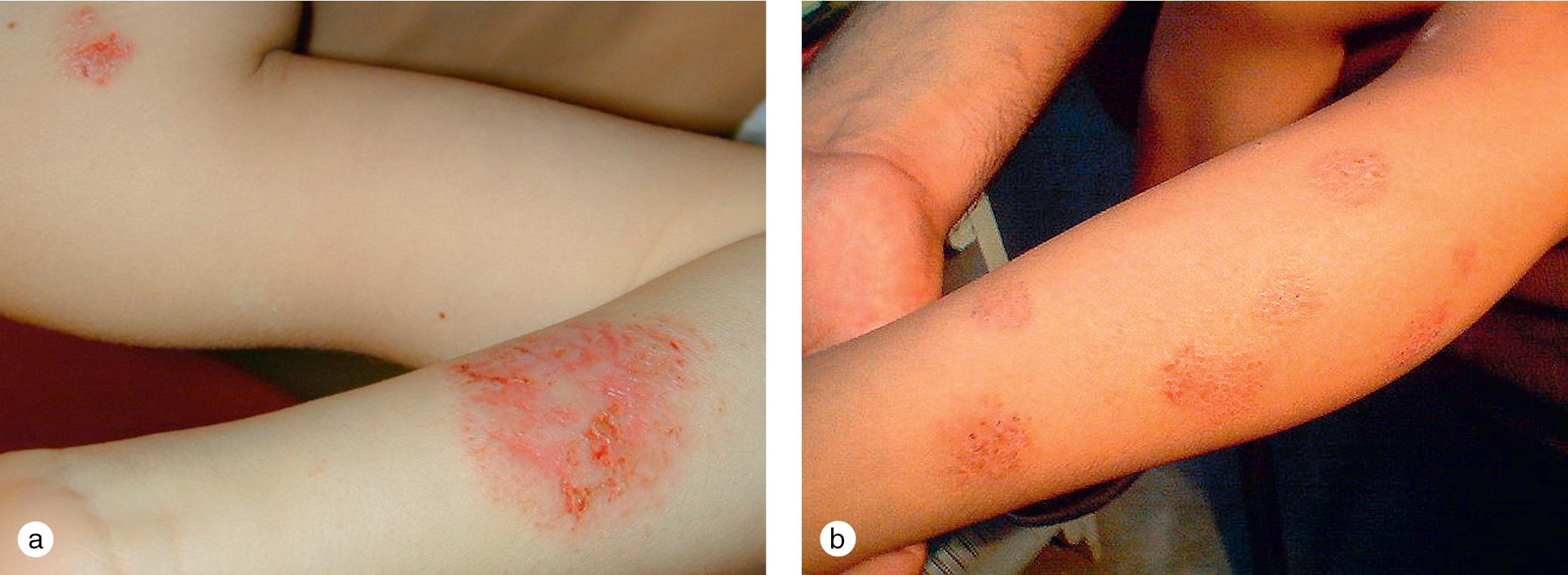
Although chronic rubbing results in lichenification, and scratching in linear excoriations, individuals who pick and gouge at their itchy, irritated skin tend to produce markedly thickened papules known as prurigo nodules ( Fig. 3.23 ). Although prurigo nodularis is not specific to atopic dermatitis, many patients with these nodules also have an atopic diathesis, which manifests as allergic rhinitis, asthma, or food allergy. Frequently, other stigmata of atopic dermatitis are present as well. Prurigo lesions tend to localize to the extremities, although widespread cutaneous involvement is observed in some cases.
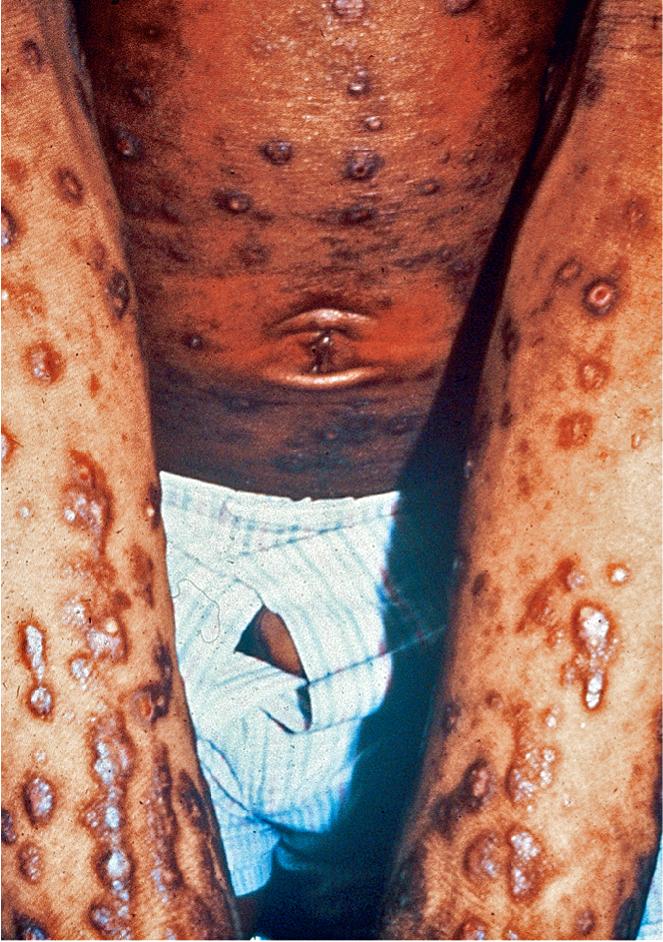
Although a few atopic patients present initially with a predominance of follicular papules, virtually all patients develop these 2–4 mm diameter follicular lesions sometime during their clinical course ( Fig. 3.24 ). Lesions are usually widespread on the trunk, but careful observation also reveals their presence on the extremities, particularly early in flares of disease activity. In children with chronic disease, discrete papules may be obscured by excoriations and lichenification, particularly in the flexural creases.
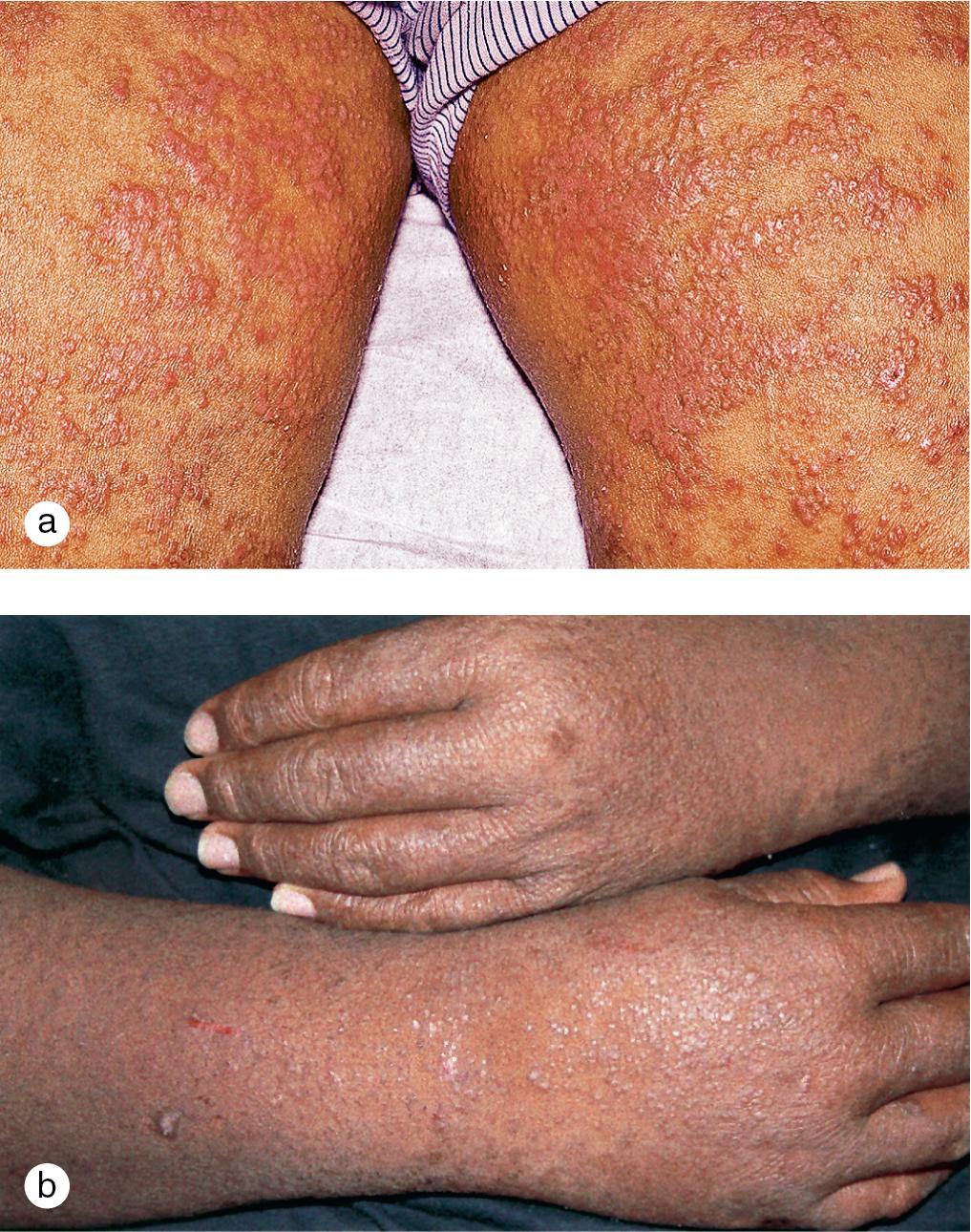
Although ichthyosis vulgaris (IV) is inherited independently of atopic dermatitis, hyperlinearity of the palms and soles typical of IV is a frequent finding in patients with atopic dermatitis. Moreover, discoveries of the filaggrin loss-of-function mutations causing IV and the prevalence of these defects in atopics have highlighted the potentiation of atopic dermatitis in IV-affected individuals ( Fig. 3.25 ).
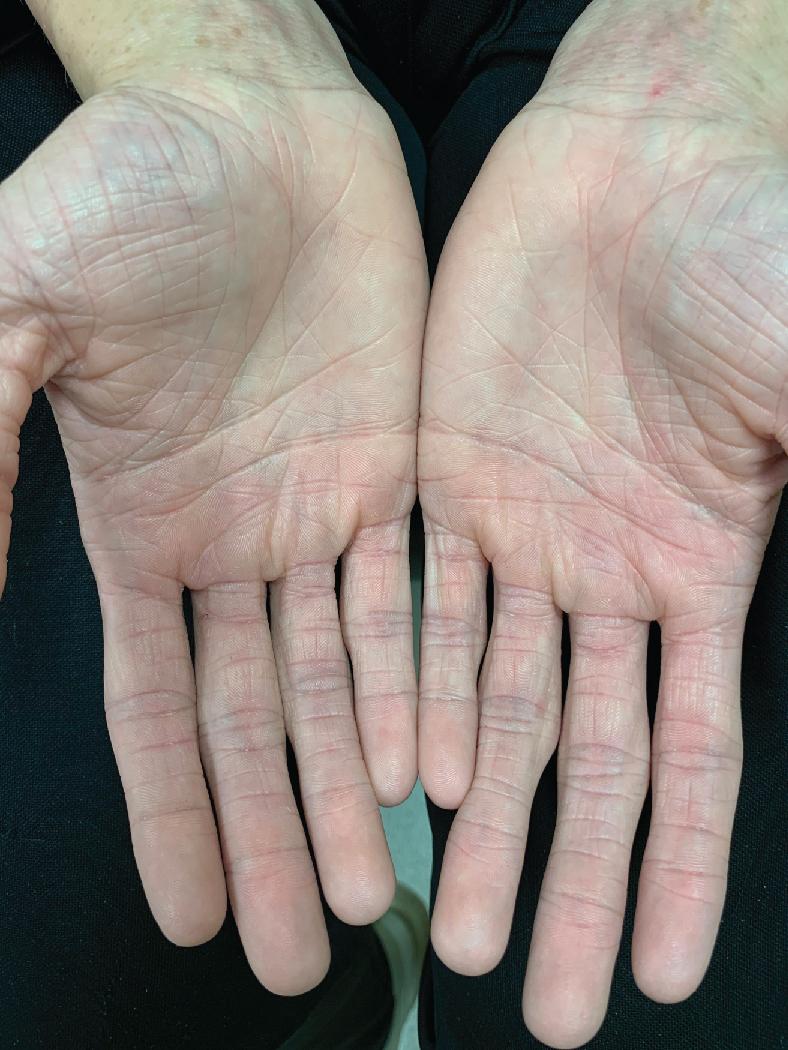
IV, which is inherited in an autosomal-dominant pattern with an incidence of 1:250, is associated with a defect in profilaggrin that is converted into filaggrin, the major protein comprising keratohyalin. The severity of IV increases with decreasing levels of functioning filaggrin. Retained polygonal scales are usually evident on the distal lower extremities (see Fig. 2.34 ), but they may also show a generalized distribution. Xerosis associated with ichthyosis may contribute to the pruritus present in atopic dermatitis.
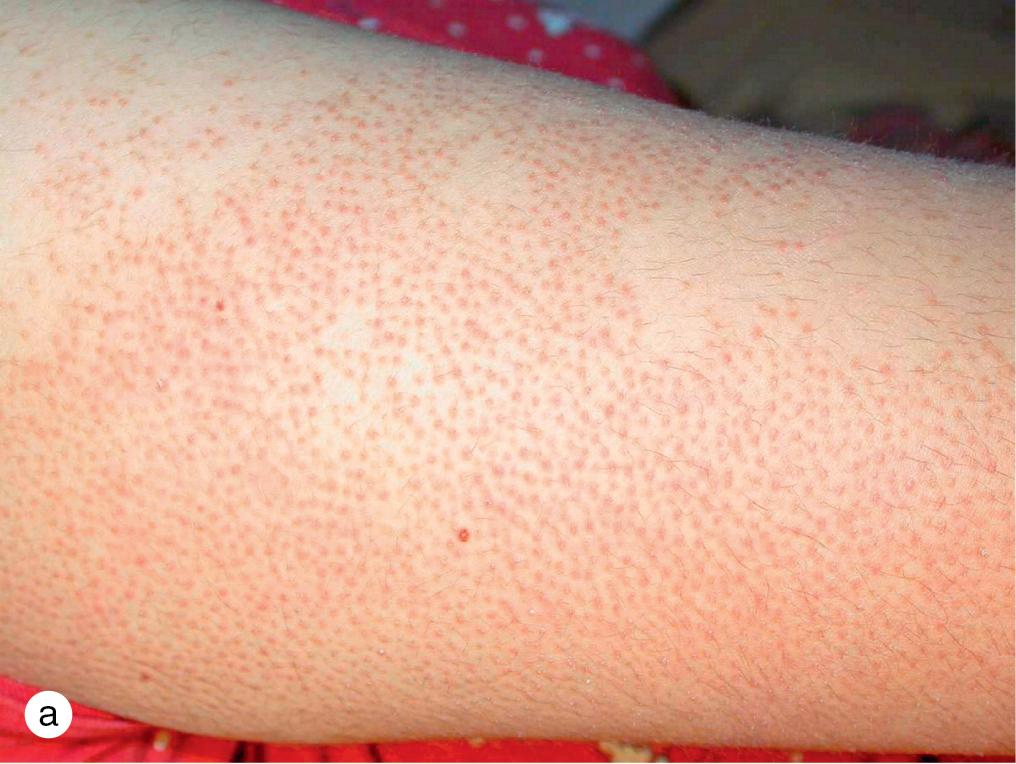
Although keratosis pilaris is often an isolated finding, it is commonly associated with atopic dermatitis and/or IV. Keratosis pilaris results from retention of scales in the follicular infundibulum, which clinically is characterized by horny follicular papules and erythema on the upper arms, medial thighs, and cheeks ( Fig. 3.26 ). Occasionally, more widespread papules appear on the back, buttocks, and legs.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here