Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Inadequate postoperative analgesia and/or an uninhibited perioperative surgical stress response has the potential to initiate pathophysiologic changes in all major organ systems, including the cardiovascular, pulmonary, gastrointestinal, renal, endocrine, immunologic, and/or central nervous systems, all of which may lead to substantial postoperative morbidity. Adequate postoperative analgesia prevents unnecessary patient discomfort and may decrease morbidity, postoperative hospital length of stay, incidence of chronic pain, and thus cost.
Pain after cardiac surgery may be intense and originates from many sources, including the incision (sternotomy or thoracotomy), intraoperative tissue retraction and dissection, vascular cannulation sites, vein harvesting sites, and chest tubes, among others. Achieving optimal pain relief after cardiac surgery is often difficult yet may be attained via a wide variety of techniques, including local anesthetic infiltration, intravenous agents, intrathecal techniques, epidural techniques, paravertebral analgesia, and fascial plane chest wall blocks.
Traditionally, analgesia after cardiac surgery has been obtained with intravenous opioids. However, intravenous opioid use is associated with definite detrimental side effects (nausea/vomiting, pruritus, urinary retention, respiratory depression), and longer-acting opioids such as morphine may delay tracheal extubation during the immediate postoperative period via excessive sedation and/or respiratory depression. Furthermore, opioid-related postoperative adverse events are likely underreported. Thus in the current era of early extubation (fast-tracking), cardiac anesthesiologists are exploring unique options other than traditional intravenous opioids for control of postoperative pain in patients after cardiac surgery.
Although patient-controlled analgesia is a well-established technique (used for more than two decades) and offers potential unique benefits (reliable analgesic effect, improved patient autonomy, flexible adjustment to individual needs, etc.), whether it truly offers significant clinical advantages (compared with traditional nurse-administered analgesic techniques) to patients immediately after cardiac surgery remains to be determined.
Cyclooxygenase-2 (COX-2) inhibitors possess analgesic (opioid-sparing) effects and lack deleterious effects on coagulation. However, current evidence does not suggest that COX-2 inhibitors provide major advantages over traditional nonsteroidal antiinflammatory drugs (NSAIDs). Furthermore, potential links between this class of drugs and cardiovascular complications, sternal wound infections, acute renal failure, and thromboembolic complications need to be fully evaluated.
With goals of reducing perioperative opioid consumption, pain intensity, and persistent postoperative pain, novel drugs are being used in addition to traditional opioids. Several adjunctive analgesic medications have been recently described, including acetaminophen, α 2 -adrenergic agonists, gabapentinoids, ketamine, and magnesium. The numerous clinical investigations regarding this topic use a wide array of heterogeneous techniques (different dosing, timing of administration, length of use) and have prevented clinically useful conclusions.
Administration of intrathecal morphine to patients initiates reliable postoperative analgesia after cardiac surgery, yet its effect on respiratory depression remains unpredictable, potentially delaying tracheal extubation. However, recent studies indicate that low-dose intrathecal morphine may provide reliable analgesia without hindering early tracheal extubation. Intrathecal opioids or local anesthetics cannot reliably attenuate the perioperative stress response associated with cardiac surgery that persists during the immediate postoperative period. Although intrathecal local anesthetics may induce perioperative thoracic cardiac sympathectomy, the hemodynamic changes associated with a “total spinal” make the technique unpalatable in patients with cardiac disease.
Administration of thoracic epidural opioids or local anesthetics to patients initiates reliable postoperative analgesia after cardiac surgery. Administration of thoracic epidural local anesthetics can both reliably attenuate the perioperative stress response associated with cardiac surgery that persists during the immediate postoperative period and induce perioperative thoracic cardiac sympathectomy.
Use of intrathecal and epidural techniques in patients undergoing cardiac surgery remains extremely controversial, particularly with the potential for hematoma. The numerous clinical investigations regarding this topic are suboptimally designed and use a wide array of disparate techniques, preventing clinically useful conclusions.
For a wide variety of reasons (increased use of small incisions, opioid-sparing), the last decade has seen a resurgence and development of regional techniques (including catheter-based approach) in patients undergoing cardiac surgery. Recent clinical studies using intercostal, paravertebral, and fascial plane chest wall blocks indicate that these techniques may have unique clinical advantages, even when compared with traditional intrathecal and epidural techniques.
As a general rule, it is best to avoid intense, single-modality therapy for the treatment of acute postoperative pain. The administration of two analgesic agents that act by different mechanisms (multimodal analgesia) provides superior analgesic efficacy with equivalent or reduced adverse effects. Analgesic therapies should be used only after thoughtful consideration of the risks and benefits for each individual patient. The therapy (or therapies) selected should reflect the individual anesthesiologist’s expertise, as well as the capacity for safe application of the chosen modality in each practice setting. The choice of medication, dose, route, and duration of therapy should always be individualized.
Adequate postoperative analgesia prevents unnecessary patient discomfort and may decrease morbidity, postoperative hospital length of stay, chronic postoperative pain, and thus cost. Because postoperative pain management has been deemed important, the American Society of Anesthesiologists has published practice guidelines regarding this topic. Furthermore, in recognition of the need for improved pain management, the Joint Commission on Accreditation of Healthcare Organizations has developed standards for the assessment and management of pain in accredited hospitals and other health care settings. Patient satisfaction (linked to adequacy of postoperative analgesia) has become an essential element that influences clinical activity of not only anesthesiologists but all health care professionals.
Achieving optimal pain relief after cardiac surgery is often difficult. Pain may be associated with many interventions, including sternotomy, thoracotomy, leg-vein harvesting, pericardiotomy, and/or chest tube insertion, among others ( Fig. 30.1 ). Inadequate analgesia and/or an uninhibited stress response during the postoperative period may increase morbidity by causing adverse hemodynamic, metabolic, immunologic, and hemostatic alterations. Aggressive control of postoperative pain, associated with an attenuated stress response, may decrease morbidity and mortality in patients after cardiac surgery. Adequate postoperative analgesia may be attained via a wide variety of techniques ( Box 30.1 ). Traditionally, analgesia after cardiac surgery has been obtained with intravenous opioids (specifically morphine). However, intravenous opioid use is associated with definite detrimental side effects (nausea/vomiting, pruritus, urinary retention, respiratory depression), and longer-acting opioids such as morphine may delay tracheal extubation during the immediate postoperative period via excessive sedation and/or respiratory depression. Thus in the current era of early extubation (fast-track), cardiac anesthesiologists are exploring unique options other than traditional intravenous opioids for control of postoperative pain in patients after cardiac surgery. The last decade has seen increased use of smaller incisions by cardiac surgeons, prompting clinical investigations into the use of regional anesthesia involving various techniques including intercostal, paravertebral, and fascial plane chest wall blocks. No single technique is clearly superior; each possesses distinct advantages and disadvantages. It is becoming increasingly clear that a multimodal approach/combined analgesic regimen (utilizing a variety of techniques) is the best way to approach postoperative pain (in all patients after surgery) to maximize analgesia and minimize side effects. When addressing postoperative analgesia in cardiac surgical patients, choice of technique (or techniques) should be made only after a thorough analysis of the risk:benefit ratio of each technique in the specific patient in whom analgesia is desired.
Local anesthetic infiltration
Nerve blocks/regional analgesia
Opioids
Nonsteroidal antiinflammatory agents
Acetaminophen
α-Adrenergic agents
Gabapentinoids
Ketamine
Magnesium
Intrathecal techniques
Epidural techniques
Multimodal analgesia
Surgical or traumatic injury initiates changes in the peripheral and central nervous systems that must be addressed therapeutically to promote postoperative analgesia and, it is hoped, positively influence clinical outcome ( Boxes 30.2 and 30.3 ). The physical processes of incision, traction, and cutting of tissues stimulate free nerve endings and a wide variety of specific nociceptors. Receptor activation and activity are further modified by the local release of chemical mediators of inflammation and sympathetic amines released via the perioperative surgical stress response. The perioperative surgical stress response peaks during the immediate postoperative period and exerts major effects on many physiologic processes. However, it is clear that inadequate postoperative analgesia and/or an uninhibited perioperative surgical stress response has the potential to initiate pathophysiologic changes in all major organ systems, including the cardiovascular, pulmonary, gastrointestinal, renal, endocrine, immunologic, and/or central nervous systems, all of which may lead to substantial postoperative morbidity.
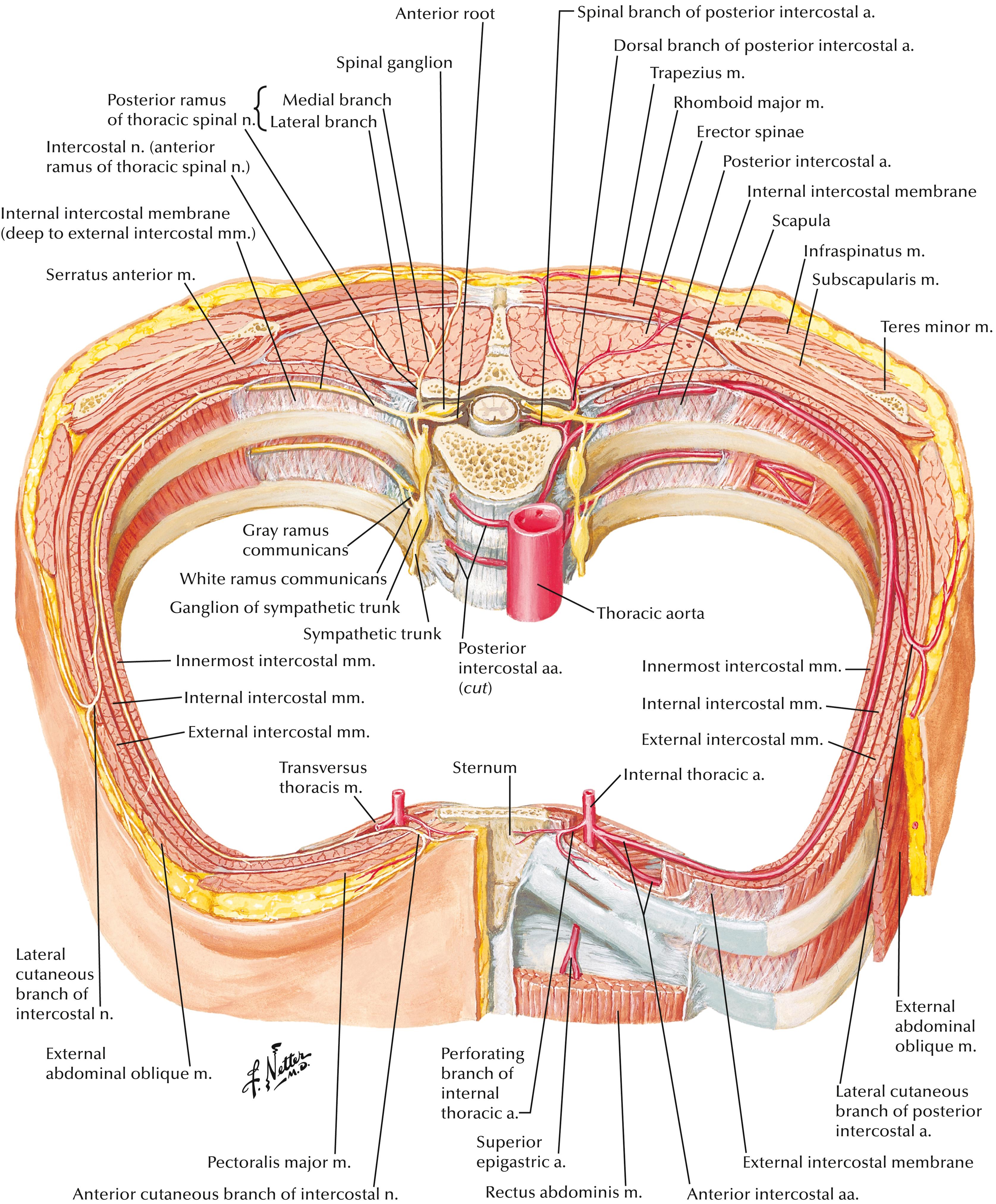
Pain after cardiac surgery originates from many sources, including incision, tissue traction, dissection, chest tube placement, vein-graft harvest, thoracic rib fractures, and positioning.
Patient satisfaction with analgesia is influenced by anticipated level of pain, information on pain treatment modalities, and direct involvement in decision-making.
Quality of postoperative analgesia may influence morbidity.
Quality of postoperative analgesia impacts persistent pain after cardiac surgery.
Persistent pain after cardiac surgery is underestimated.
Persistent pain may result from noxious input from acute injury triggering central nervous system sensitization.
Inadequate analgesia and uninhibited stress response may lead to adverse hemodynamic, metabolic, immunologic, and hemostatic alterations.
Adequate analgesia may decrease perioperative stress response and reduce morbidity including hyperglycemia, lactic acidemia, sepsis, metabolic acidosis, and disseminated intravascular coagulation.
Inadequate postoperative pain control increases the chances of developing persistent pain.
Incidence of persistent pain is high and can occur in up to 30% to 50% of patients.
Risk factors for persistent pain include lower age, female gender, and anxiety.
Persistent pain is defined as developing after surgery and persisting beyond 3 months.
Pain after cardiac surgery may be intense and it originates from many sources, including the incision (sternotomy, thoracotomy, etc.; Fig. 30.1 ), intraoperative tissue retraction and dissection, vascular cannulation sites, dissection of an internal mammary artery, vein harvesting sites, and chest tubes, among others. Maximal pain intensity is typically highest on the first postoperative day and will gradually diminish between postoperative days two and three. Pain distribution also varies from primary incisional pain to other areas of referred pain such as the shoulder, epigastric, or osteoarticular.
Although the most common source of pain in patients after cardiac surgery remains the chest wall, leg pain from vein-graft harvesting can be problematic as well. Such pain may not become apparent until the late postoperative period, which may be related to the progression of patient mobilization as well as the decreasing impact of sternotomy pain (unmasking leg incisional pain). Utilization of minimally invasive vein-graft harvesting techniques (endoscopic vein-graft harvesting) decreases postoperative leg pain intensity and duration compared with conventional open techniques. Although initial harvest times may be prolonged, harvest times become equivalent between the two techniques (endoscopic vs conventional) once a short learning curve is overcome. Furthermore, leg morbidity (infection, dehiscence, etc.) may be less in patients undergoing endoscopic vein harvest compared with patients undergoing conventional open techniques because of different incisional lengths.
Patient satisfaction with quality of postoperative analgesia is as much related to the comparison between anticipated and experienced pain as it is to the actual level of pain experienced. Satisfaction is related to a situation that is better than predicted, and dissatisfaction to one that is worse than expected. Patients undergoing cardiac surgery remain concerned regarding the adequacy of postoperative pain relief and tend to preoperatively expect a greater amount of postoperative pain than that which is actually experienced. Because of these unique preoperative expectations, patients after cardiac surgery who receive only moderate analgesia postoperatively will likely still be satisfied with their pain control. Thus patients may experience pain of moderate intensity after cardiac surgery yet still express very high satisfaction levels. A recent study indicates that obtaining control of pain intensity during the first 48 hours after surgery is a major factor in overall patient satisfaction. In addition, providing patients with information on pain treatment modalities and direct involvement in decision-making leads to increased satisfaction. However, assessment of pain can be challenging in patients who are sedated and mechanically ventilated.
Scientific advances have allowed a better understanding of how and why pain occurs, leading to unique and possibly clinically beneficial pain management strategies. Noxious input from acute injury may trigger a state of central nervous system sensitization, called wind-up . In essence, dorsal horn neurotransmitter release via nociceptive input conditions the central nervous system such that there is enhanced responsiveness (secondary hyperalgesia). Although experimental evidence exists indicating that enhanced responsiveness outlasts the initial provocative insult (induced sensitivity outlasts stimulus), the exact clinical relevance remains to be determined. Advances regarding spinal cord neuropharmacology have led to research aimed at modifying or blocking N -methyl- d -aspartate receptors to influence pain control and to the concept of preemptive analgesia, which is predicated on addressing pain before it initiates peripheral and central sensitization. However, given the redundancy in neurotransmitter receptor systems in the central nervous system, it is unlikely that blocking only one component will result in clear clinical benefits. Although the use of N -methyl- d -aspartate receptor antagonists and the concept of preemptive analgesia are intriguing and certain clinical investigations appear to support their utility, clear and definite clinical benefits in humans remain to be determined. Debate continues over the potential benefits of N -methyl- d -aspartate receptor antagonists and the utility of preemptive analgesic treatment, as well as direction in which research and conceptual development in this exciting field need to proceed.
Inadequate analgesia (coupled with an uninhibited stress response) during the postoperative period may lead to many adverse hemodynamic (tachycardia, hypertension, vasoconstriction), metabolic (increased catabolism), immunologic (impaired immune response), and hemostatic (platelet activation) alterations ( Box 30.3 ). In patients undergoing cardiac surgery, perioperative myocardial ischemia (diagnosed by electrocardiography and/or transesophageal echocardiography) is most commonly observed during the immediate postoperative period and appears to be related to outcome. Intraoperatively, initiation of cardiopulmonary bypass causes substantial increases in stress-response hormones (norepinephrine, epinephrine, etc.) that persist into the immediate postoperative period and may contribute to myocardial ischemia observed during this time. Thus during the pivotal immediate postoperative period after cardiac surgery, adequate analgesia (coupled with stress-response attenuation) may potentially decrease morbidity and enhance health-related quality of life.
Aggressive control of postoperative pain in patients after cardiac surgery may beneficially affect outcome. Administration of intensive analgesia during the immediate postoperative period may suppress sympathetic nervous system activation, thereby having numerous beneficial clinical effects, including alterations in sensitivity of platelets to epinephrine, alterations in fibrinolysis, enhanced regional left ventricular function, and decreased coronary artery vasoconstriction, all potentially leading to a reduced incidence/severity of myocardial ischemia. Postoperative opioid infusions demonstrate a reduced perioperative stress response (assessed via multiple serum mediators), less perioperative morbidity (hyperglycemia, lactic acidemia, sepsis, metabolic acidosis, disseminated intravascular coagulation), and can translate into decreased mortality. Unfortunately, aggressive control of postoperative pain in patients after cardiac surgery with relatively large amounts of intravenous opioids may not allow tracheal extubation in the immediate postoperative period (a goal of current practice).
Chronic or persistent postoperative pain is frequent, impacting quality of life and functional status after surgery ( Box 30.3 ). Chronic postoperative pain, as defined by the International Classification of Diseases 11, is pain that develops after surgery and persists beyond the expected stage of recovery (3 months). The incidence of chronic pain after coronary artery bypass is high and occurs in up to 30% to 50% of patients. The cause of persistent pain after sternotomy is multifactorial, yet tissue destruction, intercostal nerve trauma, scar formation, rib fractures, sternal infection, stainless steel wire sutures, and/or costochondral separation may all play roles. Such chronic pain is often localized to the arms, shoulders, or legs. Postoperative brachial plexus neuropathies also may occur and have been attributed to rib fracture fragments, internal mammary artery dissection, suboptimal positioning of patients during surgery, and/or central venous catheter placement. Postoperative neuralgia of the saphenous nerve has also been reported after harvesting of saphenous veins for coronary artery bypass grafting. Inflammatory mediator release leading to peripheral sensitization at the site of tissue injury has been suggested as a potential mechanism of chronic pain. However, investigations into the utilization of antiinflammatory drugs (dexamethasone) during cardiac surgery do not alter the incidence of persistent pain, suggesting alternative (central) pathways.
Ho et al. conducted a survey with 244 patients after cardiac surgery and median sternotomy and found that persistent pain was reported in almost 30% of patients. The incidence rate of persistent pain at any site was 29% (71 patients) and for sternotomy was 25% (61 patients). However, such persistent pain was usually reported as mild, with only 7% of patients reporting interference with daily living. Thus it was concluded that mild persistent pain after cardiac surgery and median sternotomy is common yet only infrequently substantially interferes with daily life.
Although there are many factors associated with chronic pain, inadequate postoperative pain control increases the chances of developing persistent pain. Investigation into patterns associated with acute postoperative pain (first 7 days after surgery) reveals that moderate to high pain may be defined primarily by patient factors and not surgical factors ( Table 30.1 ). Risk factors include lower age, female gender, higher anxiety, and pain behaviors as predictors of higher levels of immediate postoperative pain. Aggressive analgesic techniques to minimize pain in patients with these risk factors may help decrease the incidence of chronic pain syndromes.
| Risk Factor | Special Considerations |
|---|---|
| Patient demographics | Younger or older age ± female sex |
| Surgical | Thoracic surgery Orthopedic surgery Limb amputation Emergent vs elective |
| Preoperative pain | High preoperative pain scores Chronic pain conditions Preoperative opioid use |
| Psychological | Anxiety/stress Depression Substance abuse Catastrophizing |
| Pain perception | Increased sensitivity to experimental pain: quantitative sensory testing of pressure, thermal, electrical thresholds Increased temporal summation |
| Genetic | Genetic factors affecting pain sensitivity CYP4502D6 enzyme SNP A118G in OPRM1 receptor polymorphisms |
Although the mechanisms of postoperative pain and the pharmacology of analgesic drugs are relatively well understood, the delivery of effective postoperative analgesia remains far from universal. Many techniques are available ( Box 30.1 ). In general, the American Society of Anesthesiologists Task Force on Acute Pain Management in the Perioperative Setting reports that the existing literature supports the efficacy and safety of three techniques used by anesthesiologists for perioperative pain control: regional analgesic techniques, patient-controlled analgesia with systemic opioids, and intrathecal/epidural opioid analgesia. Regarding regional analgesic techniques, the existing literature supports the analgesic efficacy of paravertebral blocks, intercostal nerve blocks, and fascial plane chest wall blocks; however, lack of well-designed, randomized clinical trials limits recommended routine clinical use in cardiac surgery. Regional techniques may have opioid-sparing effects and analgesic benefits when used concurrently with adjunctive modalities (intravenous opioid or other analgesics). Regarding patient-controlled analgesia with systemic opioids, the existing literature supports its efficacy (compared with intramuscular techniques) for postoperative pain management, yet the existing literature is equivocal regarding the efficacy of patient-controlled analgesia techniques compared with nurse- or staff-administered intravenous analgesia. In addition, the existing literature is equivocal regarding the comparative efficacy of epidural patient-controlled analgesia versus intravenous patient-controlled analgesia techniques.
When background opioid infusions are included with patient-controlled analgesia techniques, patients report better analgesia and greater morphine consumption without increased incidence of nausea, vomiting, pruritus, or sedation. Although greater morphine consumption during patient-controlled analgesia with continuous background infusion might predispose patients to respiratory depression, the existing literature is insufficient to reveal this potential adverse effect. Finally, regarding intrathecal and epidural opioid analgesia, the existing literature supports the efficacy of epidural morphine and fentanyl for perioperative analgesia but is insufficient to characterize the spectrum of risks and benefits associated with the use of other specific opioids given by these routes. Pruritus and urinary retention occur more frequently when morphine is given intrathecally or epidurally compared with systemic (intravenous or intramuscular) administration. Furthermore, epidural morphine provides more effective pain relief than intramuscular morphine. Similarly, epidural fentanyl provides more effective postoperative analgesia than intravenous fentanyl. The existing literature is insufficient to evaluate the effects of epidural techniques administered at different times (preincisional, postincisional, postoperative).
Pain after cardiac surgery is often related to median sternotomy (peaking during the first two postoperative days). Because of problems associated with traditional intravenous opioid analgesia and nonsteroidal antiinflammatory drugs (NSAIDs) and cyclooxygenase (COX) inhibitors, alternative methods of achieving postoperative analgesia in cardiac surgical patients have been sought. One such alternative method that may hold promise is continuous infusion of local anesthetic ( Box 30.4 ).
This technique is simple and provides reliable analgesia
Potential disadvantage is tissue necrosis
Can be performed with indwelling infusion catheters or single-shot technique
Must be used with adjunctive analgesic modalities
In a prospective, randomized, placebo-controlled, double-blind clinical trial, White et al. studied local infiltration in 36 patients undergoing cardiac surgery. All patients had two indwelling infusion catheters placed at the median sternotomy incision site at the end of surgery (one in the subfascial plane above the sternum, one above the fascia in the subcutaneous tissue). Patients received 0.25% bupivacaine ( n = 12), 0.5% bupivacaine ( n = 12), or normal saline ( n = 12) via a constant rate infusion through the catheter (4 mL/h) for 48 hours after surgery. Tracheal extubation times were equivocal, and there was a significant reduction in pain scores and morphine use in the 0.5% bupivacaine group. Patient satisfaction was also improved in the 0.5% bupivacaine group; however, there were no significant differences in patient-controlled analgesia morphine use between the 0.25% bupivacaine and control groups. Time to ambulation (1 vs 2 days, respectively) and duration of hospital stay (4 vs 5 days, respectively) were lower in the 0.5% bupivacaine group than in the control group. The authors conclude that continuous infusion of 0.5% bupivacaine at 4 mL/h is effective for decreasing postoperative pain and the need for postoperative supplemental opioid analgesic medication as well as for improving patient satisfaction. These results were replicated by Dowling et al. in 35 patients undergoing cardiac surgery. Patients undergoing elective coronary artery bypass grafting via median sternotomy were randomized to either ropivacaine or placebo groups. At the end of surgery, before wound closure, bilateral intercostal nerve injections from T1 to T12 were performed using 20 mL of either 0.2% ropivacaine or normal saline, followed by infusion at 4 mL/h via catheters. The analgesic technique significantly improved postoperative pain control while decreasing the amount of required opioid analgesia. Additionally, there was a significant decrease in hospital length of stay.
The management of postoperative pain with continuous direct infusion of local anesthetic into the surgical wound has been described after a wide variety of surgeries other than cardiac. Complications encountered with these infusion pump systems include tissue necrosis, surgical wound infection, and cellulitis after orthopedic, gastrointestinal, podiatric, and other surgeries. None of these reported adverse events has involved patients undergoing cardiac surgery. The most commonly reported complication is tissue necrosis. Furthermore, consequences of these reported adverse events were typically severe and required intervention and additional medical and/or surgical treatment. Although these initial reports may be isolated incidents, they may also represent a warning signal, suggesting the need for further investigation into the relation between use of pumps for direct continuous infusion of local anesthetics and other drugs into surgical wounds and subsequent adverse events such as tissue necrosis, serious infections, or cellulitis. Neither of the two clinical investigations involving local anesthetic infusion in patients after cardiac surgery with median sternotomy reported such wound complications. , Regardless, these safety issues merit careful consideration because of the importance of sternal wound complications in cardiac surgery.
With the increasing popularity of minimally invasive cardiac surgery, which uses nonsternotomy incisions (minithoracotomy, endoscopic port sites), the use of regional analgesia for the management of postoperative pain has increased ( Box 30.5 ). Compared to systemic analgesia, regional analgesic techniques, when used in conjunction with other multimodal strategies, reduce pain after cardiac surgery. However, additional benefits of regional anesthesia on morbidity and mortality outcomes after minimally invasive surgery are limited.
When used in conjunction with other analgesic modalities, regional analgesia reliably reduces pain after cardiac surgery.
Additional morbidity/mortality benefits are not consistently demonstrated.
Techniques include intercostal nerve block, paravertebral block, and fascial plane chest wall blocks.
Specific techniques have unique challenges including difficult placement, unreliable analgesia, local anesthetic toxicity, and other complications (pneumothorax, nerve injury).
Because of costal cartilage trauma, tissue damage to ribs, muscles, or peripheral nerves, thoracotomy incisions may induce more intense postoperative pain than that resulting from median sternotomy. Adequate analgesia after thoracotomy incisions is important because pain is a key component in alteration of lung function after this type of incision. Uncontrolled pain causes a reduction in respiratory mechanics, reduced mobility, and increases in hormonal and metabolic activities. Perioperative deterioration in respiratory mechanics may lead to pulmonary complications and hypoxemia, which may in turn lead to myocardial ischemia/infarction, cerebrovascular accidents, thromboembolism, delayed wound healing, increased morbidity, and prolonged hospital stay. Various analgesic techniques have been developed to treat postoperative thoracotomy pain. The most commonly used techniques include intercostal nerve block, thoracic paravertebral block, fascial plane chest wall blocks, and intrathecal and epidural techniques ( Box 30.5 ).
Intercostal nerve blocks have been used extensively for analgesia after thoracic surgery. They can be performed either intraoperatively or postoperatively and usually provide sufficient analgesia lasting approximately 6 to 12 hours (depending on the amount and type of local anesthetic used; Box 30.6 ). Local anesthetics may be administered as a single injection under direct vision before chest closure, as a single preoperative percutaneous injection, as multiple percutaneous serial injections, or via an indwelling intercostal catheter. Blockade of intercostal nerves interrupts C-fiber afferent transmission of impulses to the spinal cord. Various clinical studies have confirmed the analgesic efficacy of this technique, and the technique compares favorably with thoracic epidural analgesic techniques. A major concern associated with intercostal nerve block is the potentially high amount of local anesthetic systemic absorption, yet multiple clinical studies involving patients undergoing thoracic surgery have documented safe blood levels with standard techniques. Clinical investigations involving patients undergoing thoracic surgery indicate that intercostal nerve blockade by intermittent or continuous infusion of bupivacaine (0.25%–0.5%) or ropivacaine (0.5%–0.75%) through indwelling intercostal catheters is an effective method for supplementing systemic intravenous opioid analgesia for postthoracotomy pain. The value of single preclosure injections remains doubtful.
Time-tested, reliable analgesia for 6–12 h, can be single shot or catheter based, and performed preoperatively or postoperatively
May improve pulmonary function in patients after thoracic surgery
Potential risk of local anesthetic toxicity with catheter delivery
Demonstrates equivalent analgesia to thoracic epidural in thoracotomy incisions
Unilateral paravertebral block has decreased incidence of hypotension, nausea, and urinary retention compared to thoracic epidural
Can be single-shot or catheter-based, single-level, or multilevel injection
Role in cardiac surgery is uncertain
Potential risks include symptomatic bradycardia, hypotension, vasovagal episode, hematoma, and local anesthetic toxicity
Described blocks include pectoralis I and II, serratus anterior, pectointercostal, transversus thoracic muscle, and erector spinae blocks
Current literature does not support routine use of fascial plane chest wall blocks in cardiac surgery, due to inconsistent reliability, analgesic effect, and variable technique
These blocks have a good safety profile with a low rate of complications
Potential complications include infection, nerve injury, hematoma, pneumothorax, intravascular injection, and local anesthetic toxicity
Thoracic paravertebral block involves injection of local anesthetic adjacent to the thoracic vertebrae close to where the spinal nerves emerge from the intervertebral foramina ( Fig. 30.2 ). Thoracic paravertebral block, compared with thoracic epidural analgesic techniques, appears to provide equivalent analgesia, is technically easier, and may harbor less risk ( Box 30.6 ). Several different techniques exist for successful thoracic paravertebral block and have been extensively reviewed. It can be performed as a single-level injection, multilevel injection, or continuous catheter technique. The classic technique involves eliciting loss of resistance, however, recent studies advocate a transverse, in-plane ultrasound-guided technique to reduce adverse events. Injection of local anesthetic results in ipsilateral somatic and sympathetic nerve blockade in multiple contiguous thoracic dermatomes above and below the site of injection (together with possible suppression of the neuroendocrine stress response to surgery). Bilateral use of thoracic paravertebral block has also been described and can be used for a sternotomy incision. Continuous thoracic paravertebral infusion of local anesthetic via a catheter placed under direct vision after thoracotomy is also a safe, simple, and an effective method of providing analgesia after thoracotomy. It is usually used in conjunction with adjunct intravenous medications (opioid or other analgesics) to provide optimum relief after thoracotomy. Although supplemental intravenous analgesics are usually required, opioid requirements are substantially reduced.
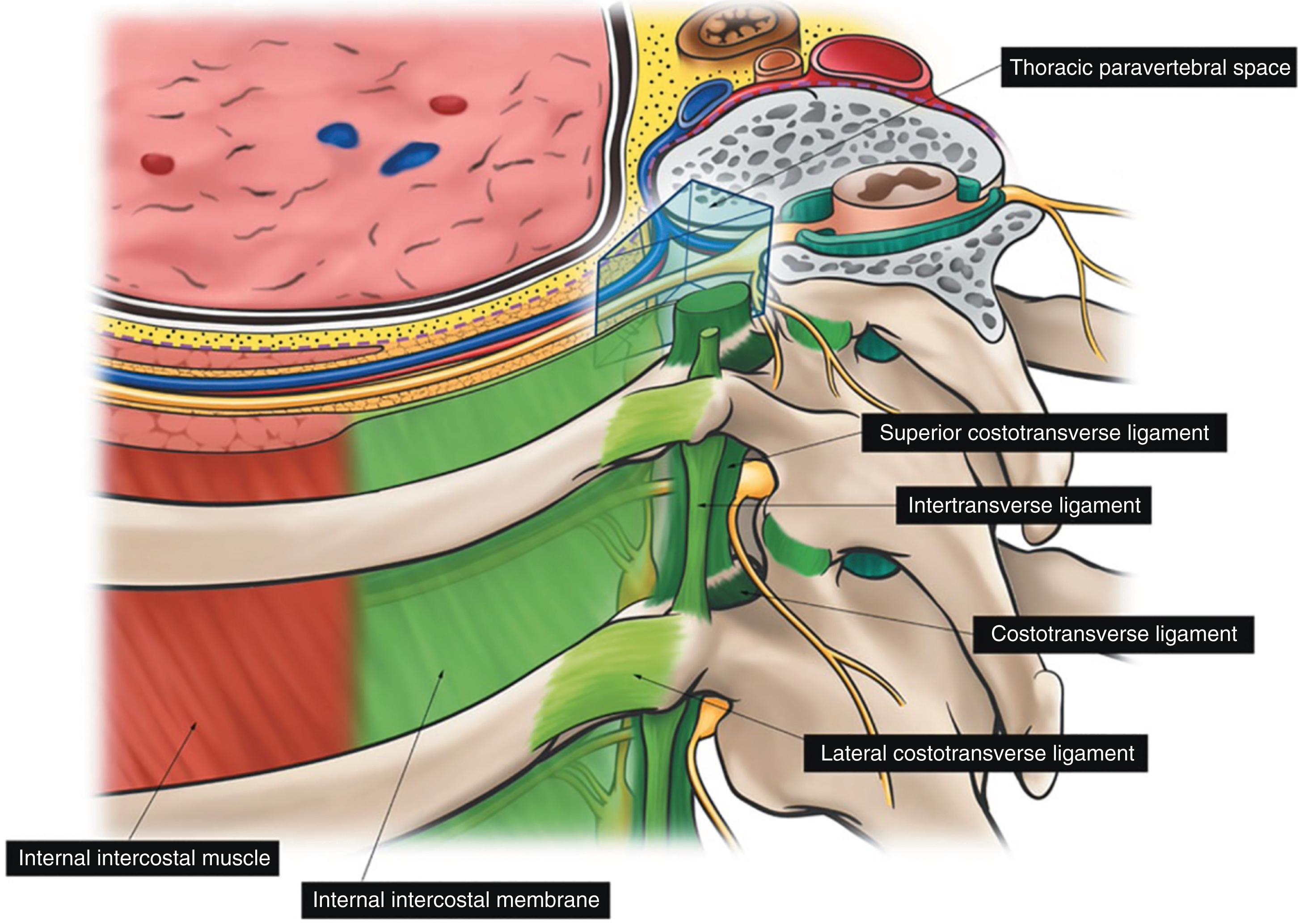
Unilateral paravertebral block is useful for attaining postthoracotomy analgesia. The benefits are a decreased incidence of adverse events (hypotension, nausea, urinary retention) and a decreased risk for systemic local anesthetic toxicity compared to bilateral paravertebral block. Bilateral sympathetic block induced with epidural analgesia results in increased hypotension compared to unilateral paravertebral block. Continuous thoracic paravertebral block as part of a balanced analgesic regimen may provide effective pain relief with few adverse effects after thoracotomy and appears to be comparable with thoracic epidural analgesia. Differences in mortality, major complications, length of hospital stay, chronic pain, and cost benefits are lacking with use of paravertebral block versus placebo or thoracic epidural analgesia. A small study suggests possible decrease in pulmonary complications and incidence of pneumonia when used for postoperative pain during thoracoabdominal aortic aneurysm repair. Serious complications such as pneumothorax, local anesthetic toxicity, and hemodynamic changes are uncommon with ultrasound guidance. In a large cohort of patients ( n = 1427), the adverse event rate was 0.35% for unilateral blockade and 0.88% for bilateral blockade with ultrasound-guided paravertebral block, including symptomatic bradycardia, hypotension, vasovagal episode, and local anesthetic toxicity. Notably, there were no instances of pleural puncture or symptomatic pneumothorax. In contrast, Naja et al. report the overall incidence of pleural puncture at 0.8%, with subsequent pneumothorax in 0.5% of patients, with thoracic paravertebral blocks placed using a landmark technique with a nerve stimulator.
Paravertebral blocks have also been described in minimally invasive cardiac surgery in small clinical studies and case reports. In three patients, undergoing cardiac surgery via right minithoracotomy and cardiopulmonary bypass, paravertebral catheters facilitated postoperative analgesia in two of these patients and early extubation in all patients. In a prospective randomized clinical trial in 60 patients undergoing robotic mitral valve surgery via port incisions, a four-level paravertebral blockade with 0.5% bupivacaine before induction of anesthesia produced reliable analgesia and decreased pain scores. There were no reported instances of hematoma or local anesthetic toxicity in either publication. Patient satisfaction was higher in the paravertebral group.
The role of bilateral thoracic paravertebral block in cardiac surgery with sternotomy remains to be defined. In a pilot study ( n = 8), ropivacaine was administered prior to induction of anesthesia at 3 mg/kg followed by infusion (0.24% at 0.1 mL/kg/h) on each side for a total of 35 mg/h for a 70 kg patient. The bilateral paravertebral block did not eliminate the need for supplemental opioids and the study was stopped early due to concerns of local anesthetic toxicity, with the authors stating “potentially toxic total plasma ropivacaine concentrations were common.” Although small, this study highlights the potential complications with bilateral paravertebral blocks in cardiac surgery. In another larger study in cardiac patients with median sternotomy, bilateral ultrasound-guided thoracic paravertebral blocks facilitated shorter intensive care unit (ICU) length of stay and lower incidence of urinary retention and vomiting compared to thoracic epidural analgesia. However, paravertebral block had less analgesic efficacy than thoracic epidural analgesia.
Although paravertebral blocks and thoracic epidural analgesia are widely used clinically for open thoracotomy, techniques for analgesia after video-assisted thoracoscopic surgery are variable, with no accepted standards for clinical implementation. Clear conclusions are difficult to ascertain from studies due to considerable heterogeneity in methodology and design. A recent randomized, prospective study compared a single-shot paravertebral block (30 mL 0.5% ropivacaine), catheter (0.2% ropivacaine at 10 mL/h), or thoracic epidural (0.125% bupivacaine and 0.05 mg/mL hydromorphone; 40 patients in each group) in patients undergoing video-assisted thoracoscopic surgery. Pain scores and opioid consumption were significantly lower for the epidural group than those of either paravertebral group at 24 and 48 hours, with decreased opioid use for 72 hours. Paravertebral catheter placement took the most time (12 min) versus epidural (5 min) and single injection (6 min). Differences in pain scores were small and not clinically relevant, and the authors concluded that paravertebral catheter placement is not justified due to less efficacy than single-shot technique, increased risk, cost, and time burden. Other studies have demonstrated noninferiority of continuous paravertebral block compared to epidural analgesia with potentially reduced side effects of urinary retention and hypotension. Yet another small study suggests that thoracic paravertebral and epidural may be comparable to patient-controlled analgesia in patients undergoing video-assisted thoracoscopic surgery. In summary, paravertebral blocks are potentially comparable to epidural for thoracotomy incisions, however, further studies are warranted to elucidate analgesic efficacy and safety in cardiac surgery with sternotomy, minimally invasive cardiac surgery, and video-assisted thoracoscopic surgery.
Intercostal nerve block and thoracic paravertebral block offer the advantages of simplicity and efficacy in controlling postoperative pain in patients after thoracic surgery. However, although analgesic efficacy of these techniques may be comparable with intrathecal and epidural techniques, these techniques appear to work best as a part of a multimodal analgesic regimen. Complications associated with infiltrations of large quantities of local anesthetic are a concern when utilizing these analgesic techniques.
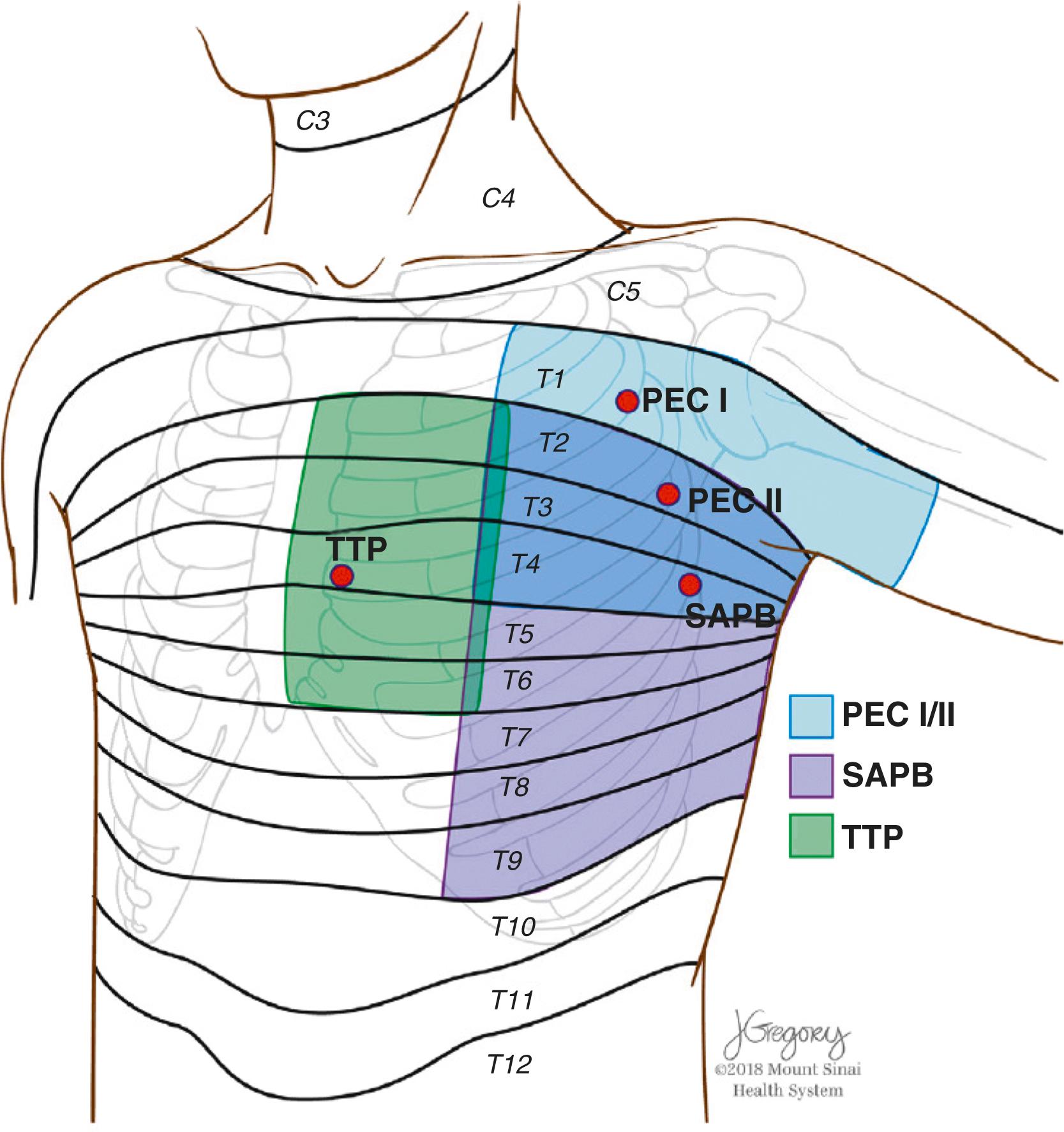
With increased interest in opioid-sparing perioperative analgesic techniques, fascial plane chest wall blocks have emerged as a promising adjunctive strategy for multimodal analgesia in thoracic, minimally invasive cardiac surgery, and conventional (sternotomy) cardiac surgery ( Table 30.2 and Box 30.6 ). Local anesthetic is deposited in fascial planes containing nerve fibers that provide sensory innervation to the targeted chest wall region. After leaving an intervertebral foramen, spinal nerves divide into dorsal and ventral rami. The thoracic chest wall is innervated by the anterior rami of spinal nerves T1–T11, which eventually form the perforating branches of intercostal nerves ( Fig. 30.3 ). Multiple studies on specific fascial plane blocks in cardiac and thoracic surgery demonstrate variable levels of efficacy. Currently described fascial plane blocks include pectoralis I, pectoralis II, serratus anterior, pectointercostal, transversus thoracic muscle, and erector spinae blocks ( Fig. 30.4 ). , Each block is described below with accompanying instructional videos ( , , , ![]() ). Detailed overview of fascial plane chest wall blocks in cardiac surgery is presented by Kelava et al. and key points regarding each block are presented in Table 30.3 .
). Detailed overview of fascial plane chest wall blocks in cardiac surgery is presented by Kelava et al. and key points regarding each block are presented in Table 30.3 .
| Surgical Approach | Block Options | UL/BL |
|---|---|---|
| Sternotomy | PIF or ESP | BL |
| Minimally invasive right anterolateral thoracotomy | PECS I, PECS II, and PIF or ESP | UL right |
| Minimally invasive right thoracotomy (robotic) | PECS II and SAP or ESP | UL right |
| Transapical TAVR | SAP or ESP | UL left |
| Block Key Points |
| PECS I • Blocks medial and lateral pectoral nerves |
|
|
| PECS II • Blocks lateral cutaneous branches of intercostal nerves approximately T2–T6 (long thoracic and thoracodorsal nerves) |
|
|
| SAP • Blocks lateral cutaneous branches of intercostal nerves approximately T3–T9 (long thoracic and thoracodorsal nerves also if superficial SAP) |
|
|
| PIF/TTMP • Blocks anterior cutaneous branches of intercostal nerves |
|
|
|
| ESP • Blocks spinal nerve dorsal and ventral rami |
|
|
Fascial plane blocks have a good safety profile, with a low rate of complications ( Box 30.6 ). Potential complications include infection, nerve injury, hematoma, pneumothorax, intravascular injection, and local anesthetic toxicity. Although there are abundant publications using fascial plane blocks for noncardiac surgery, well-designed, high-quality, randomized controlled trials are lacking in patients undergoing cardiac surgery, and use of these blocks is currently not mainstay clinical practice for postoperative analgesia, nor have they been advocated in guidelines. Current studies do not support any meaningful reduction in opioid-related adverse events, morbidity, or persistent postoperative pain. Black et al. discuss the uncertainty regarding fascial plane blocks, “they represent a paradigm shift … however, despite growing popularity, these novel blocks have their share of controversies, namely inconsistent effect, efficacy, evidence, indication, and technique.” Additionally, fascial plane blocks require additional time, personnel, expertise in performing each technique, and should be used in addition to adjunctive analgesia (opioid and others).
The pectoralis I block was first described by Blanco in 2011 in patients having breast surgery. They injected local anesthetic into the interfascial plane between pectoralis major and minor muscles followed by continuous catheter infusion for up to 7 days. Subsequently the following year, the same author then described a modification to the pectoralis I block, called the pectoralis II block. The block reaches the lateral and medial pectoral nerves, the intercostobrachial, intercostal (3–6), and long thoracic nerve, but spares the anterior branches of the intercostal nerves ( ), ![]() thus providing analgesia to the anterior chest wall. Pectoralis blocks have been described extensively in patients undergoing breast surgery, however, use in adult cardiothoracic literature is limited to case reports and few small prospective studies. In a recent randomized controlled trial, pectoralis block was compared to serratus anterior plane block and intercostal nerve block for postthoracotomy analgesia in cardiac surgery. Patients received 2.5 mg/kg of 0.5% ropivacaine and rescue analgesia with intravenous paracetamol and fentanyl. Early pain scores (<6 h) were comparable between groups, however, pain scores from 6 to 12 hours and rescue fentanyl consumption were lower in patients who received a serratus anterior plane and pectoralis block versus intercostal nerve block. Although there are many studies evaluating the analgesic efficacy of pectoralis blocks in breast cancer surgery patients, these results are difficult to translate to cardiothoracic surgery patients (source of pain, length of surgery, incision/chest tubes) and likely do not apply. There is inadequate evidence for support of use of pectoralis block in cardiothoracic surgery, however, larger randomized controlled trials are underway and may provide answers.
thus providing analgesia to the anterior chest wall. Pectoralis blocks have been described extensively in patients undergoing breast surgery, however, use in adult cardiothoracic literature is limited to case reports and few small prospective studies. In a recent randomized controlled trial, pectoralis block was compared to serratus anterior plane block and intercostal nerve block for postthoracotomy analgesia in cardiac surgery. Patients received 2.5 mg/kg of 0.5% ropivacaine and rescue analgesia with intravenous paracetamol and fentanyl. Early pain scores (<6 h) were comparable between groups, however, pain scores from 6 to 12 hours and rescue fentanyl consumption were lower in patients who received a serratus anterior plane and pectoralis block versus intercostal nerve block. Although there are many studies evaluating the analgesic efficacy of pectoralis blocks in breast cancer surgery patients, these results are difficult to translate to cardiothoracic surgery patients (source of pain, length of surgery, incision/chest tubes) and likely do not apply. There is inadequate evidence for support of use of pectoralis block in cardiothoracic surgery, however, larger randomized controlled trials are underway and may provide answers.
Video 30.1 Pectoralis I and Pectoralis II Blocks (From Kelava M, Alfirevic A, Bustamante S, Hargarve J, et al. Regional anesthesia in cardiac surgery: an overview of fascial plane chest wall blocks. Anesth Analg . 2020;131:127-135.)
The serratus anterior plane block was initially reported as a variation of the pectoral nerve block, designed to block the thoracic intercostal nerves and provide analgesia to the lateral part of the thorax. It covers the lateral cutaneous branches of the intercostal nerves and provides analgesia along the anterolateral chest wall, along a dermatomal distribution from T2 to T9 ( ![]() ). Similar to the pectoralis block, the serratus anterior plane block does not provide adequate analgesia for midline sternotomy and has been extensively used in breast and thoracic surgery. However, case reports of bilateral two-level serratus anterior plane blocks in pediatric patients with median sternotomy demonstrate that the technique may be effective in this population, likely due to size of the patient and volume of distribution. Case reports and clinical trials demonstrate conflicting results in obtaining adequate postoperative analgesia in thoracic and cardiac surgery. Investigational studies are small, lack standardized methodology, and have varied techniques (single-shot and/or continuous catheter). Some studies compare serratus anterior block to other regional anesthesia and intravenous opioids, and some studies compare it to placebo. In general, the literature suggests that continuous serratus anterior plane blocks with catheters are potentially noninferior to paravertebral blocks or thoracic epidural blocks in thoracic surgery and are superior to simple intravenous opioids, however, they may not provide adequate analgesia in cardiac surgery. The role of single-shot serratus anterior plane blocks remains undefined in cardiothoracic surgery.
). Similar to the pectoralis block, the serratus anterior plane block does not provide adequate analgesia for midline sternotomy and has been extensively used in breast and thoracic surgery. However, case reports of bilateral two-level serratus anterior plane blocks in pediatric patients with median sternotomy demonstrate that the technique may be effective in this population, likely due to size of the patient and volume of distribution. Case reports and clinical trials demonstrate conflicting results in obtaining adequate postoperative analgesia in thoracic and cardiac surgery. Investigational studies are small, lack standardized methodology, and have varied techniques (single-shot and/or continuous catheter). Some studies compare serratus anterior block to other regional anesthesia and intravenous opioids, and some studies compare it to placebo. In general, the literature suggests that continuous serratus anterior plane blocks with catheters are potentially noninferior to paravertebral blocks or thoracic epidural blocks in thoracic surgery and are superior to simple intravenous opioids, however, they may not provide adequate analgesia in cardiac surgery. The role of single-shot serratus anterior plane blocks remains undefined in cardiothoracic surgery.
Video 30.2 Serratus Anterior Plane Block (From Kelava M, Alfirevic A, Bustamante S, Hargarve J, et al. Regional anesthesia in cardiac surgery: an overview of fascial plane chest wall blocks. Anesth Analg . 2020;131:127-135.)
A prospective, randomized study in 40 patients scheduled for thoracotomy, comparing thoracic epidural analgesia with serratus anterior plane block, did not find differences in pain or morphine consumption between the two groups. These results indicate noninferiority of serratus anterior plane block with thoracic epidural analgesia in thoracotomy patients. This is corroborated by Hanley et al. in a double-blinded, randomized, noninferiority study with 40 patients, comparing continuous thoracic paravertebral block with continuous serratus anterior block in video-assisted thoracic surgery. They found that both groups were comparable in 48 hour opioid consumption, side effects, length of hospital stay, and patient satisfaction. Another prospective, randomized, triple-blind, placebo-controlled study evaluated 85 patients undergoing video-assisted thoracic surgery with serratus plane block versus placebo, for quality of recovery, pain, opioid consumption, and adverse events. They found that single-injection serratus plane block with 0.4 mL/kg of 0.375% ropivacaine after induction of anesthesia increased quality of recovery for 2 days after surgery and decreased pain scores at 6 hours and opioid consumption at 24 hours. No differences in pain scores were apparent at 24 or 48 hours after surgery. These results are corroborated in a subsequent small retrospective study ( n = 40) in video-assisted thoracoscopic surgery (lobectomy), which found patients had improved postoperative pain scores with cough and less opioid consumption with serratus anterior plane block versus intercostal nerve block. Although these small clinical trials demonstrate mild efficacy of serratus anterior plane blocks, there is an absence of clinically meaningful analgesia and reduction in opioid consumption.
Few studies have assessed serratus anterior plane block in cardiac surgery, and they are small and lack robust methodology. An observational study assessed postoperative pain and opioid consumption in 63 patients undergoing mitral valve surgery with minithoracotomy. Patients received either intravenous analgesia or a serratus anterior plane block. A single-shot injection was performed with 150 mg of ropivacaine 0.375% prior to surgery followed by a peripheral nerve catheter with continuous infusion of ropivacaine 0.3%, 21 mg/h after surgery, with catheter removal 48 hours postoperative. Postoperative pain scores were significantly less at 48 hours and mean morphine consumption was decreased (12 vs 2 mg, P < .01) in the serratus anterior plane group. There were no differences in incidence of postoperative nausea and vomiting, time to extubation, and bowel dysfunction. Alternatively, a retrospective, observational study by Moll et al. found that serratus anterior plane block did not adequately cover incisional and chest tube pain associated with minimally invasive coronary artery bypass and it was inferior to paravertebral block. Further larger, prospective, adequately blinded, randomized studies are needed to evaluate efficacy in cardiac surgery patients.
Thoracic epidural analgesia and paravertebral blocks (both associated with failure rates and complications) are typically used for postoperative thoracotomy pain after lung transplantation. However, recent case reports of bilateral continuous serratus anterior plane catheters in bilateral sequential lung transplantation demonstrate effective pain control and early extubation. , A retrospective study also reported success for postoperative refractory pain management after lung transplant in an enhanced recovery after surgery protocol. These case reports and studies are promising, and larger clinical trials are warranted to understand the safety and efficacy of these blocks for lung transplantation.
The pectointercostal fascial block previously termed the parasternal pectoral block was initially described for breast surgery analgesia. Local anesthetic is deposited 2 cm lateral to the sternum between the pectoralis major and intercostal muscle between the third and fourth rib or fourth and fifth rib, targeting the anterior intercostal nerves in this fascial plane ( ). ![]() The anterior and posterior branches of the intercostal nerves innervate the sternum. Literature consists of case reports in sternal fractures and more recently for anterior, minimally invasive thoracotomy in cardiac surgery. Although the use of parasternal fascial blocks has not been extensively investigated, one small, prospective, randomized, placebo-controlled, double-blind clinical study indicated that parasternal block and local anesthetic infiltration of the sternotomy wound and mediastinal tube sites may be a useful analgesic adjunct for patients who are expected to undergo early tracheal extubation after cardiac surgery. Bilateral pectointercostal fascial block after cardiac surgery with 0.25% ropivacaine significantly reduced pain and fentanyl requirement in a blinded, randomized clinical trial ( n = 40) by Kumar et al. However, another ( n = 80) prospective, randomized trial in adult cardiac surgery patients requiring sternotomy compared ultrasound-guided pectointercostal facial plane block with placebo or bupivacaine and found no difference in cumulative postoperative opioid consumption, although patients who received a pectointercostal fascial block had lower pain scores and no adverse events occurred. In summary, there is insufficient evidence to support routine use of this block in cardiac surgery, with inconclusive results from clinical trials. There may be potential benefits in using this block alongside additional local anesthetic infiltration.
The anterior and posterior branches of the intercostal nerves innervate the sternum. Literature consists of case reports in sternal fractures and more recently for anterior, minimally invasive thoracotomy in cardiac surgery. Although the use of parasternal fascial blocks has not been extensively investigated, one small, prospective, randomized, placebo-controlled, double-blind clinical study indicated that parasternal block and local anesthetic infiltration of the sternotomy wound and mediastinal tube sites may be a useful analgesic adjunct for patients who are expected to undergo early tracheal extubation after cardiac surgery. Bilateral pectointercostal fascial block after cardiac surgery with 0.25% ropivacaine significantly reduced pain and fentanyl requirement in a blinded, randomized clinical trial ( n = 40) by Kumar et al. However, another ( n = 80) prospective, randomized trial in adult cardiac surgery patients requiring sternotomy compared ultrasound-guided pectointercostal facial plane block with placebo or bupivacaine and found no difference in cumulative postoperative opioid consumption, although patients who received a pectointercostal fascial block had lower pain scores and no adverse events occurred. In summary, there is insufficient evidence to support routine use of this block in cardiac surgery, with inconclusive results from clinical trials. There may be potential benefits in using this block alongside additional local anesthetic infiltration.
Video 30.3 Pectointercostal Fascial Plane Block (From Kelava M, Alfirevic A, Bustamante S, Hargarve J, et al. Regional anesthesia in cardiac surgery: an overview of fascial plane chest wall blocks. Anesth Analg. 2020;131:127-135.)
Transversus thoracic muscle plane block is a deeper version of the pectointercostal fascial block. It was first described in case reports of patients undergoing breast cancer resection in 2015, followed by cadaveric study of the spread of injectate in the same issue of the journal. , Subsequently, the same authors described effective analgesia in two patients with bilateral injection of 0.375% levobupivacaine, 20 mL on each side at the fourth and fifth rib prior to median sternotomy. Two years later, the same authors again evaluated complications of this block in 299 consecutive cases. Besides two patients undergoing cardiac surgery who developed an infection at the injection site, there were no instances of hematoma or pneumothorax, suggesting that the block is overall safe. The authors do not provide detail about the two patients, however, they suggest that it was mild infection. The authors do not provide information on analgesic efficacy. Considering that infection in cardiac surgery can have serious consequences, larger clinical trials are necessary to determine safety.
The transversus thoracic muscle plane block affects the anterior branches of intercostal nerves from T2 to T6. Local anesthetic is deposited parasternal in the fascial plane between the transversus thoracic muscle and internal intercostal muscle between the third and fourth ribs or fourth and fifth ribs. The majority of literature accessing efficacy and safety is limited to case reports, however, there are a few recent clinical trials. In a prospective, randomized, double-blind study in patients ( n = 48) having cardiac surgery with median sternotomy, patients were assigned to receive preoperative transversus thoracic muscle plane block with either 20 mL of 0.25% bupivacaine or saline bilaterally. Compared with placebo, the block group had reduced postoperative 24-hour opioid consumption (median, 255 vs 465 μg fentanyl, P < .001) and significantly lower pain scores. The control group had higher incidence of postoperative nausea and pruritus. No complications associated with the block were observed. Although the level of evidence is limited, these promising results warrant larger investigative trials to access safety and long-term outcomes.
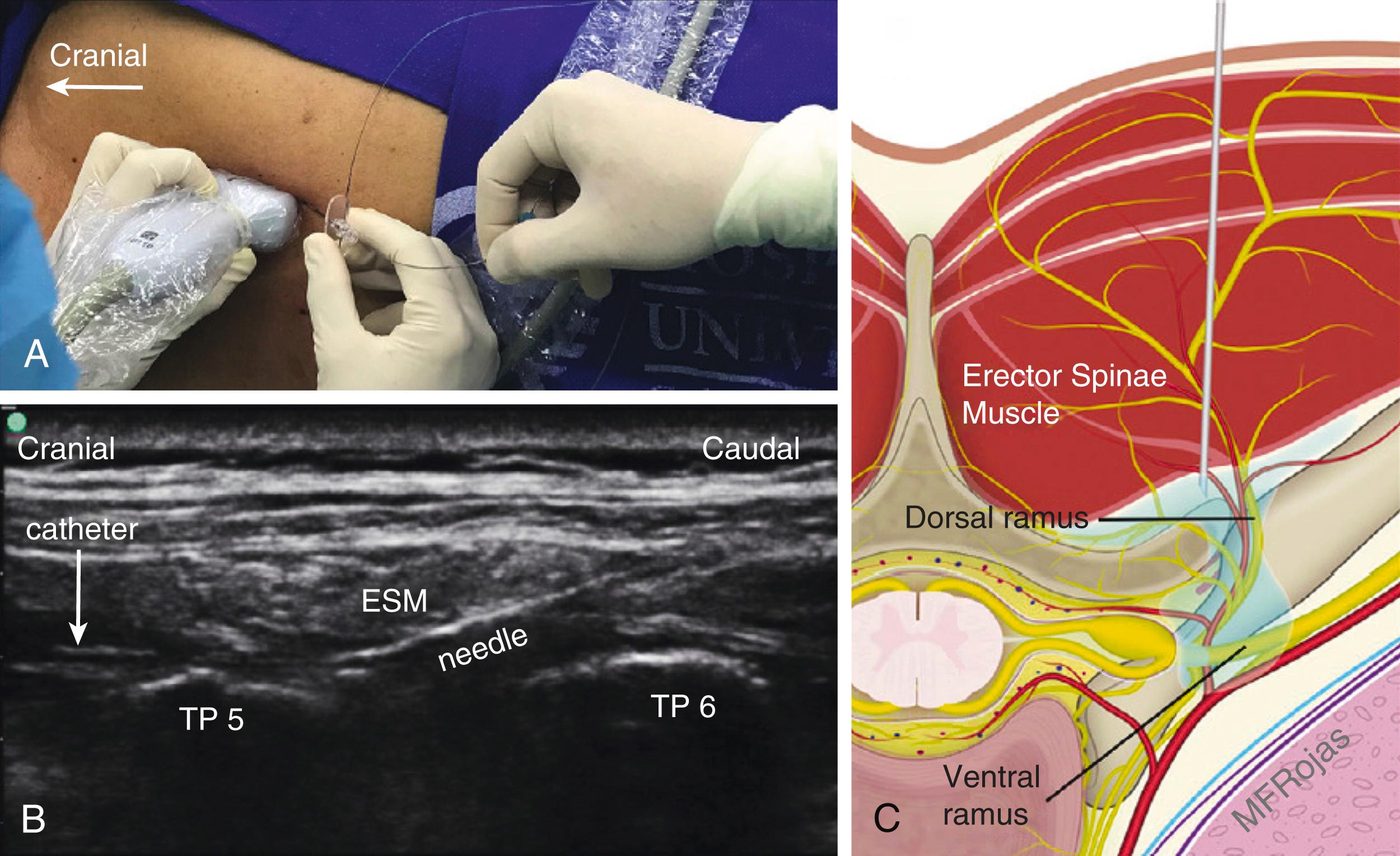
Ultrasound-guided erector spinae plane block, described in 2016 in thoracic surgery patients, is another regional technique extensively studied recently. It is a paraspinal fascial plane block in which local anesthetic is deposited between the erector spinae muscle and thoracic transverse processes ( , ![]() Fig. 30.5 ). A cadaveric study describing the mechanism of action of erector spinae block found that 20 mL of 0.25% methylene blue dye spread cephalocaudal and lateral and toward the dorsal rami posterior to the costotransverse foramen. Purported as being technically easier to perform than thoracic epidural and paravertebral blocks, , some studies suggest comparable pain control as thoracic epidural analgesia and another suggests significant improvement in pulmonary mechanics. In a small ( n = 50), prospective, randomized, clinical trial in cardiac surgery patients with sternotomy, Nagaraja et al. found no differences in pain scores up to 48 hours after surgery, and similar ventilator and intensive care unit duration between thoracic epidural and erector spinae plane block groups, thus providing support of comparable efficacy between thoracic epidural and erector spinae plane blocks.
Fig. 30.5 ). A cadaveric study describing the mechanism of action of erector spinae block found that 20 mL of 0.25% methylene blue dye spread cephalocaudal and lateral and toward the dorsal rami posterior to the costotransverse foramen. Purported as being technically easier to perform than thoracic epidural and paravertebral blocks, , some studies suggest comparable pain control as thoracic epidural analgesia and another suggests significant improvement in pulmonary mechanics. In a small ( n = 50), prospective, randomized, clinical trial in cardiac surgery patients with sternotomy, Nagaraja et al. found no differences in pain scores up to 48 hours after surgery, and similar ventilator and intensive care unit duration between thoracic epidural and erector spinae plane block groups, thus providing support of comparable efficacy between thoracic epidural and erector spinae plane blocks.
Video 30.4 Erector Spinae Plane Block (From Kelava M, Alfirevic A, Bustamante S, Hargarve J, et al. Regional anesthesia in cardiac surgery: an overview of fascial plane chest wall blocks. Anesth Analg . 2020;131:127-135.)
Other literature suggests that erector spinae plane block has greater enhanced opioid-sparing and analgesic effects than either the pectoralis nerve block or serratus anterior plane block , after mitral/tricuspid valve repair via minithoracotomy and minimally invasive thoracic surgery and comparable efficacy to thoracic epidural after thoracotomy. Erector spinae block also performed better than intercostal nerve block after minithoracotomy. In a randomized controlled trial, 60 patients received either of the two blocks (intercostal or erector spinae) and were followed for 48 hours. Erector spinae block patients had statistically significant lower postoperative pain scores, needed lower doses of intraoperative/postoperative opioid/adjunctive analgesia, and had better satisfaction with care.
Continuous bilateral erector spinae blocks in cardiac surgery with sternotomy and cardiopulmonary bypass have also been investigated. After anesthesia induction, bilateral ultrasound-guided blocks were performed, followed by 0.25 mL/kg of 0.5% ropivacaine bolus, with subsequent boluses every 6 hours. The patients used significantly less morphine than the control group in the first 48 hours (mean, 40 vs 0 mg) and exhibited less pain 1 month after surgery. However, comparison was with a historical control group, and substantial limitations with the methodology make the results difficult to interpret. The providers also received 3-day training prior to catheter insertion, making translation of these results to other institutions challenging.
Case reports are abundant, with use of the block in cardiothoracic surgery (median sternotomy, thoracotomy, minimally invasive approach), and some describing promising results. In one particular case series, bilateral erector spinae plane catheters facilitated an opioid-sparing, fast-track technique and intraoperative extubation (four out of five patients) in patients undergoing on-pump cardiac surgery. In a unique case series of five patients, erector spinae block was used for rescue analgesia after left ventricular assist device implantation via left thoracotomy in four patients and preoperatively in one patient. The authors state that it was a safer alternative to thoracic epidural analgesia and thoracic paravertebral blockade in the setting of therapeutic anticoagulation and potential risk of spinal hematoma. In summary, the erector spinae plane block is a promising regional analgesic modality in cardiothoracic surgery with sternotomy and thoracotomy. Several studies have demonstrated opioid-sparing effect and analgesic effect with relative safety. However, the evidence is still preliminary, and much is still unknown regarding the technique (single-shot, catheter), dosing (amount of local anesthetic), timing (preoperative/postoperative), duration of catheter use, and safety (hematoma, local anesthetic toxicity).
The most common local anesthetics used for fascial plane blocks are bupivacaine and ropivacaine in concentrations between 0.0625% and 0.5% and volumes ranging from 10 to 40 mL. Block duration can be prolonged with diluted epinephrine from 1:200,000 to 1:400,000 concentration or use of liposomal bupivacaine (maximum dose of 266 mg, 20 mL). Other adjuvants described in the literature include opioids, α 2 -adrenoceptor agonists, and steroids. Typically, a continuous catheter-based technique facilities prolonged duration of analgesia in majority of case reports and clinical trials.
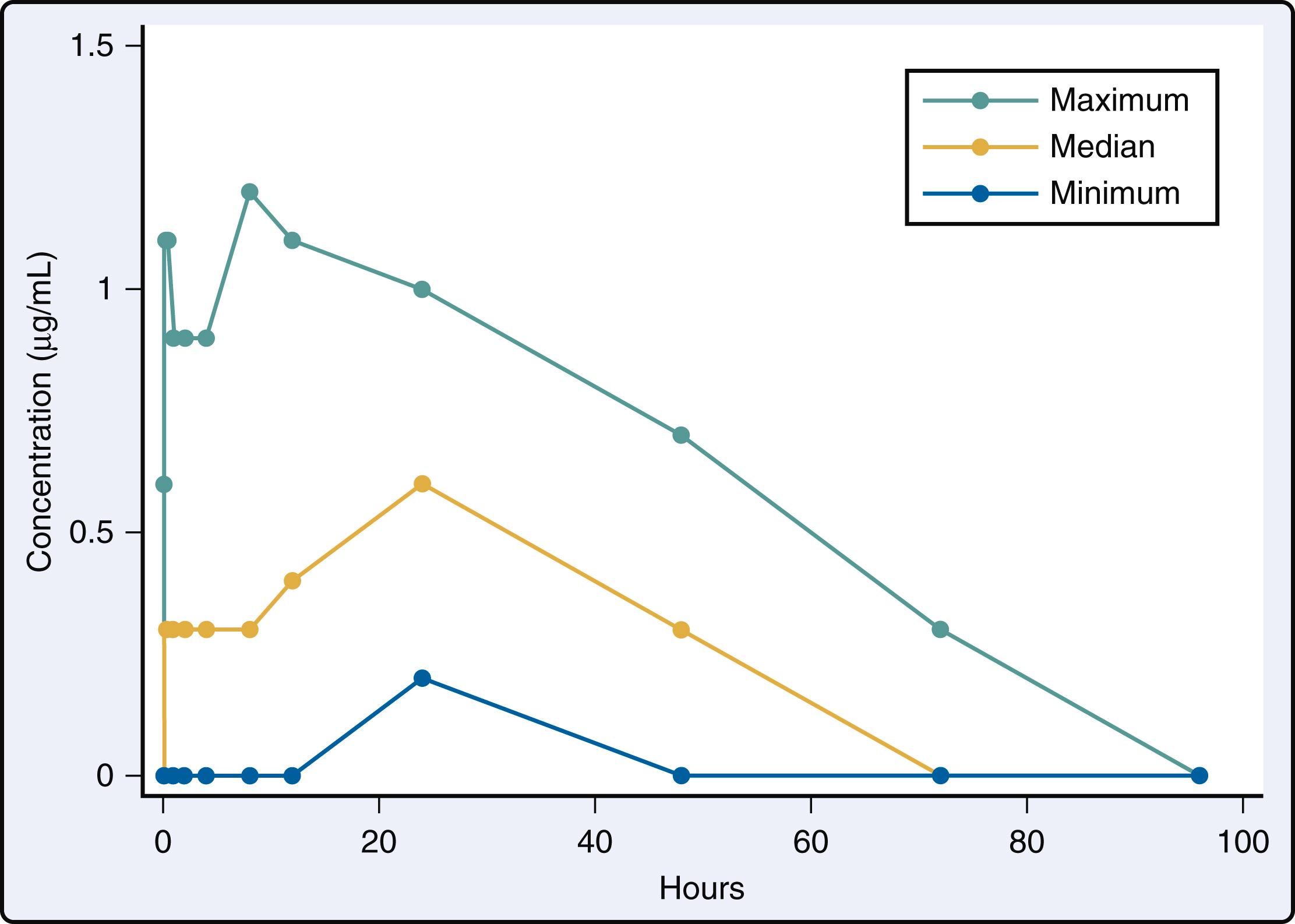
Liposomal bupivacaine, a relatively new medication, with an extended half-life compared to conventional bupivacaine hydrochloride, has been extensively studied in orthopedic and thoracic surgery ( Box 30.7 ). Liposomal bupivacaine is encapsulated in a carrier liposomal molecule, which results in slow release, increasing the duration of action (up to 72 h) and delaying the peak concentration of local anesthetic. A recent prospective observational study demonstrated that the same dose of liposomal bupivacaine (266 mg) intercostal injection for thoracotomy in 15 adults resulted in significant variability and delay in drug absorption and performance ( Fig. 30.6 ). Median time to maximum concentration was 24 hours with a range of 15 minutes to 48 hours, and the median peak serum concentration was 0.6 μg/mL with maximum at 1.2 μg/mL (below toxicity at 2 μg/mL). The peak concentration did not correlate with block duration and could potentially have increased the risk of local anesthetic toxicity with rescue nerve block. Its potential superiority over conventional bupivacaine hydrochloride remains controversial, with some studies demonstrating no difference and others finding benefit with liposomal bupivacaine. Most are small, retrospective, observational studies in thoracic surgery. In a retrospective analysis in thoracic surgery patients, intercostal nerve block with liposomal bupivacaine when compared to 0.5% bupivacaine decreased postoperative in-hospital and post-discharge opioid use. Yet other studies have found no differences in opioid consumption or pain scores between liposomal and conventional bupivacaine when used for intercostal nerve block in minimally invasive thoracic surgery. , The largest retrospective comparative study ( n = 1737) was conducted in patients undergoing lung resection. Patients were stratified by treatment with intraoperative liposomal bupivacaine intercostal block versus epidural analgesia. No clinically relevant pain outcomes were assessed, however, the authors concluded that the use of liposomal bupivacaine was not associated with increased perioperative complications. The average cost of liposomal bupivacaine is substantially higher than bupivacaine hydrochloride, and despite lack of superior performance over conventional bupivacaine, it is clinically available and widely used.
Encapsulated in a carrier liposomal molecule, results in slow release, and has increased duration of action (up to 72 h) and delayed peak concentration
There can be substantial variability in drug absorption and performance
Lack of evidence for superior analgesia compared to conventional bupivacaine
Average cost is substantially higher than conventional bupivacaine
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here