Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Total hip arthroplasty (THA) and total knee arthroplasty (TKA) are surgical procedures that have been shown to improve pain, function, and quality of life. However, these procedures are notoriously painful, and postoperative recovery can be challenging. Aside from patient discomfort, poorly controlled pain can prolong length of stay, limit participation in rehabilitation, and increase the cost of care. Severe acute postoperative pain is also a risk factor for developing chronic persistent postsurgical pain (PPSP), which is estimated to affect 7% to 20% of TKA and 2% to 8% of THA cases. In contrast, well-controlled pain allows for early mobilization, lowers complication rates, and may enhance overall recovery. For these reasons, a standardized, multimodal analgesic (MMA) regimen is a recommended component of clinical pathways. , In this chapter, we will review and summarize the clinical evidence for the various analgesic modalities available for THA and TKA.
The development of a perioperative analgesic pathway requires an understanding of the mechanisms involved in the generation, transmission, and perception of pain. Surgical incision causes a release of inflammatory mediators, such as substance P and prostaglandins (PGs), that bind to and activate nociceptors. Prostaglandin E2 (PGE2) is an important mediator of acute pain and has been implicated in peripheral and central sensitization. Nociceptors are specialized receptors found on the free nerve terminals of primary afferent neurons. These receptors detect noxious stimuli and convert them into an electrical signal that travels from the periphery to the spinal cord. Aδ and unmyelinated C-fibers are the sensory afferent neurons that convey pain throughout the body. Cutaneous and articular structures, such as the hip and knee, are densely innervated by these fibers. , Aδ and unmyelinated C-fibers enter the spinal cord via Lissauer’s tract and synapse on spinothalamic tract neurons, which are located on the dorsal horn of the spinal cord. Axons from STT neurons cross the midline and ascend the spinal cord until they synapse in the thalamus. Thalamic axons project to cortical structures that are involved in the sensory-discriminative, affective, and cognitive aspects of pain. The principal sites of pain modulation are the periphery, spinal cord, and supraspinal structures. Existing therapeutics target these areas of the pain pathway to produce analgesia. The sites of action for various analgesics are illustrated in Fig. 3.1 .
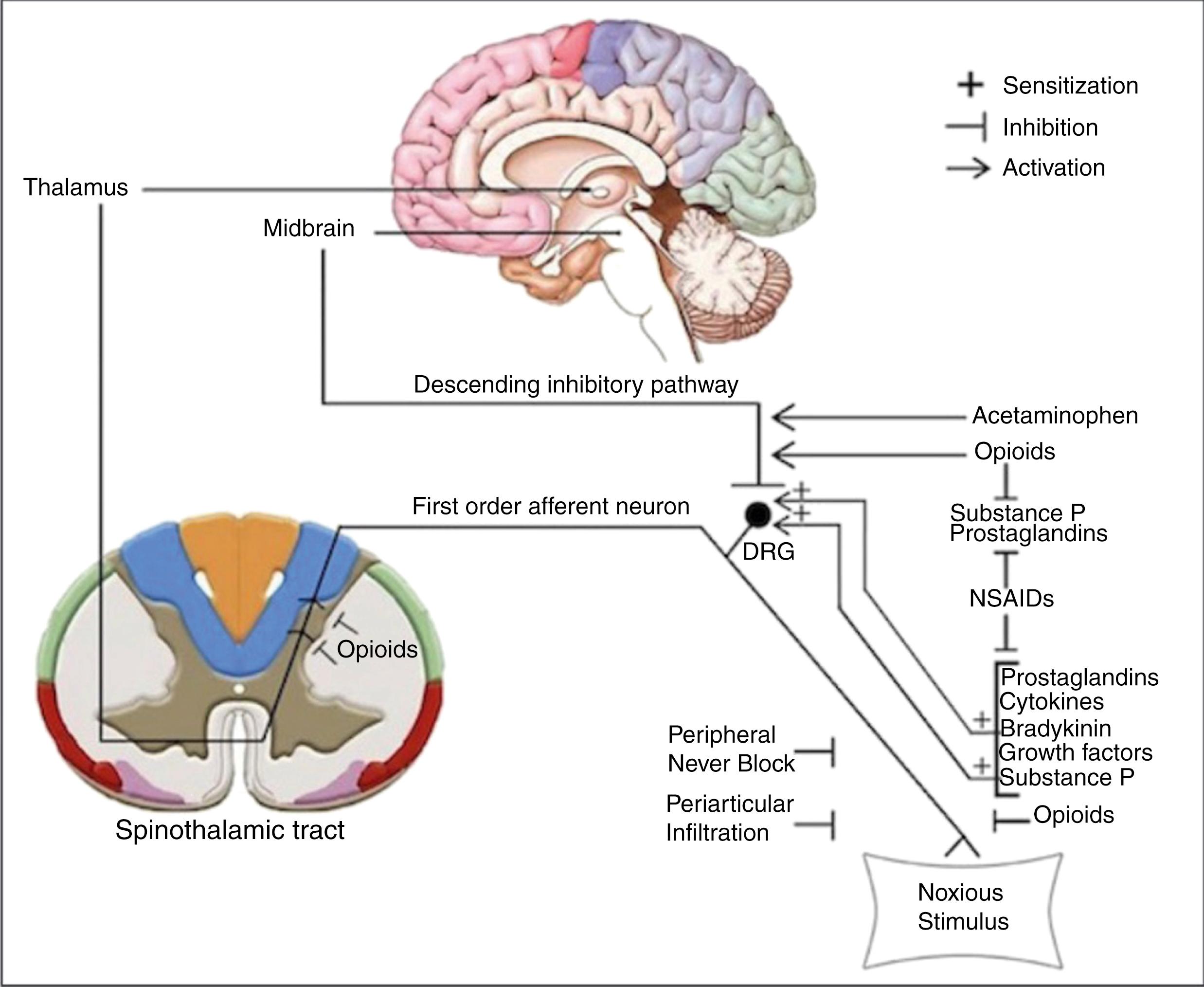
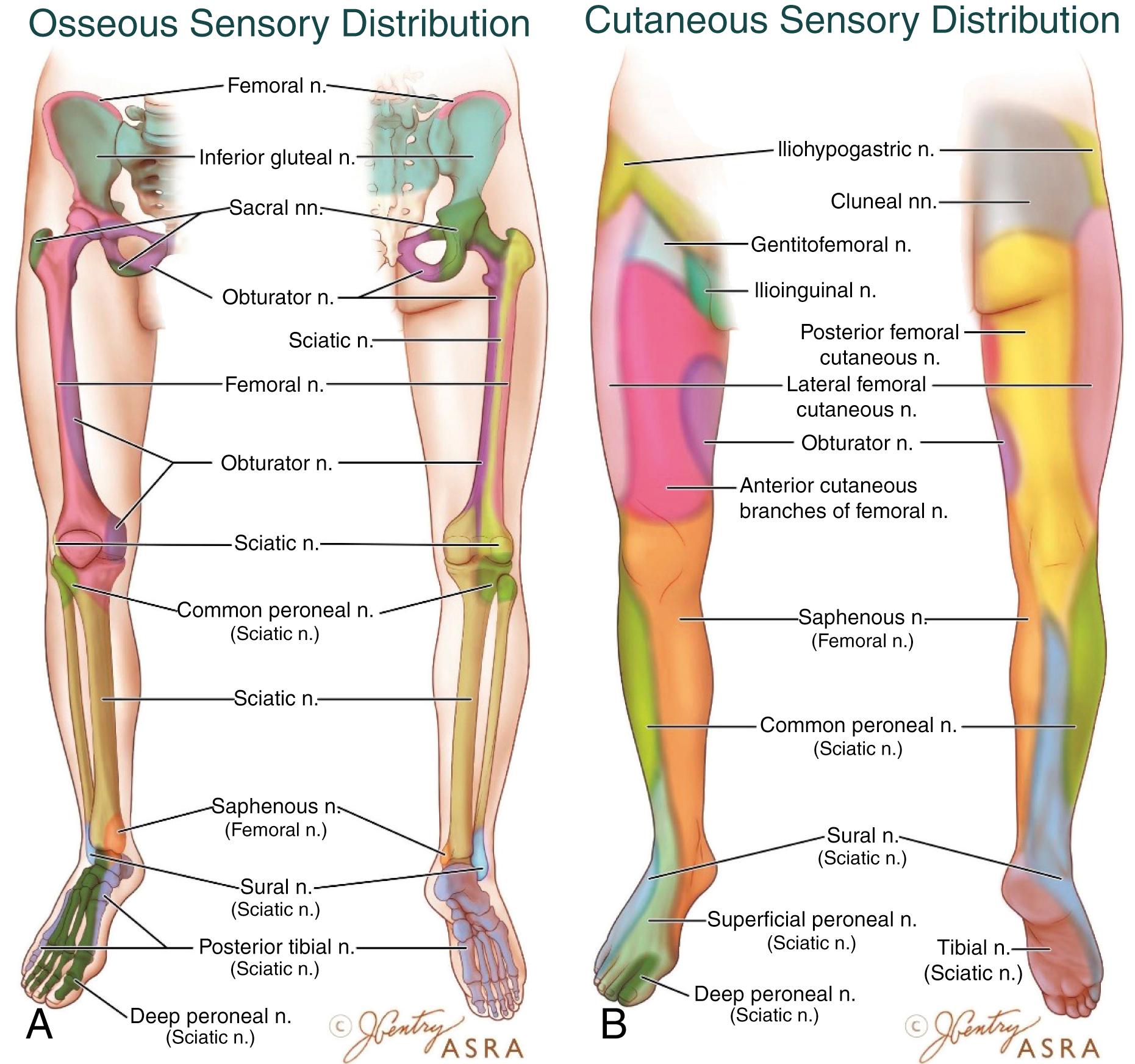
Both the hip and knee joints are densely innervated by branches of the lumbosacral plexus ( Fig. 3.2 ). Branches of the femoral nerve (FN), obturator nerve (ON), and sciatic nerve (SN) innervate the anterior, medial, and posterior knee, respectively. Innervation to the hip joint is more complex, but the nociceptor-rich region of the anterior capsule is believed to transmit the majority of painful stimuli via the FN and ON.
Historically, postoperative analgesia for total joint arthroplasty (TJA) was achieved with intravenous (IV) opioids delivered via patient-controlled analgesia (PCA) or continuous epidural infusions of local anesthetic with or without opioids. Although these techniques produced high-quality analgesia, their side effects conflicted with the goals of enhanced recovery. For example, opioids can cause respiratory depression, gastrointestinal dysfunction, and sedation. Compared with systemic opioids, epidural analgesia produces a denser level of analgesia. However, unintended hypotension, motor blockade, and impaired ambulation may also occur as the result of the nonselective blockade of sympathetic, sensory, and motor fibers.
Limitations of conventional analgesic techniques led to the development of MMA, which is an analgesic strategy that incorporates two or more nonopioid analgesics of different pharmacologic classes to target different receptors along the pain pathway. This strategy has been shown to improve the quality of analgesia and reduce opioid-related adverse effects. Multimodal analgesia has replaced opioid-based analgesia as the recommended analgesic strategy for many enhanced recovery after surgery (ERAS) pathways.
The preoperative phase is a critical period for perioperative optimization. Prior to surgery, patients are risk stratified and evaluated for the presence of modifiable risk factors. For example, obesity, diabetes mellitus, tobacco use, and malnutrition are modifiable risk factors. Preoperative optimization has been shown to reduce complications and improve postoperative outcomes.
Board-certified arthroplasty surgeons of the American Association of Hip and Knee Surgeons (AAHKS) were surveyed, and greater than 90% of respondents said that they routinely incorporate preemptive analgesia into their clinical practice. Preemptive analgesia is defined as the administration of analgesics prior to surgical incision. Studies suggest that early antinociceptive interventions aimed at attenuating the inflammatory response may reduce peripheral and central sensitization and the severity of postoperative pain. Preemptive analgesic strategies vary across orthopaedic practices but commonly include a combination of acetaminophen, nonsteroidal antiinflammatory drugs (NSAIDs) and/or gabapentinoids. Current literature is conflicting regarding effects of preemptive administration of ketamine on postoperative pain and opioid consumption in major orthopaedic surgeries, but it appears that the analgesic effect of ketamine is independent of time of administration. Preemptive opioid administration is not recommended because it may increase the risk of opioid-induced respiratory depression and sedation.
Opioid-tolerant patients include those with a history of chronic opioid therapy, former and active opioid abusers, and former abusers enrolled in methadone or buprenorphine treatment programs. The perioperative management of these patients can be challenging because they are tolerant to the analgesic effects of opioids and, thus, require increasingly higher doses to achieve analgesia. Additionally, these patients exhibit a phenomenon called opioid-induced hyperalgesia, which is an increased sensitivity to pain as a result of opioid exposure.
Recent evidence suggests that chronic opioid use is associated with increased rates of postoperative complications. In a study by Zywiel et al., patients on chronic opioid therapy prior to TKA had higher rates of complications and were more likely to require reoperation for persistent pain and/or stiffness compared with opioid-naïve patients. Pivec et al. reported similar adverse outcomes in patients on chronic opioid therapy prior to THA. A 2016 study by Nguyen et al. sought to evaluate the impact of preoperative opioid weaning on clinical outcomes following TJA. In this retrospective matched-cohort study, patients who reduced intake of chronic opioids by more than 50% experienced greater improvements in functional outcomes than chronic opioid users who did not wean. Furthermore, the improvement in clinical outcomes was comparable to patients not on preoperative opioids, suggesting that preoperative opioid use may be a modifiable risk factor in TJA.
Despite evidence suggesting that preoperative opioid use can adversely affect outcomes, there is insufficient evidence to recommend preoperative opioid weaning, and the decision to wean should be made after close coordination with the patient’s prescribing physician. For chronic pain patients unable or unwilling to reduce their preoperative opioids, early referral to a pain specialist to optimize preoperative pain management and formulate a perioperative pain management regimen is recommended.
Methadone and buprenorphine are opioid medications used for treating opioid use disorder (OUD) and/or chronic pain. Methadone is a full mu opioid receptor agonist with a long half-life. It should be taken on the day of surgery and resumed as soon as possible after surgery.
Buprenorphine is a partial mu receptor agonist and kappa receptor antagonist. It binds strongly to and slowly dissociates from the mu opioid receptors. As the daily dose of buprenorphine increases, the number of unoccupied receptors decreases. For example, patients taking a daily dose of 2 mg of buprenorphine have 27% to 47% of their opioid receptors occupied, whereas those taking 16 mg have 80% to 92% of their opioid receptors occupied. As a result, patients taking buprenorphine doses of 16 mg or more per day are unlikely to receive significant benefit from additional opioid medications.
Managing the patient receiving buprenorphine medication-assisted treatment (MAT) for OUD is challenging. These patients are typically profoundly opioid tolerant, and opioids alone are unlikely to provide adequate analgesia. A multimodal approach with regional anesthesia is advisable. Discontinuation of buprenorphine exposes these patients to standard opioids, which may lead to relapse. Further, converting from buprenorphine to standard opioids does not improve analgesia. Recent recommendations support continuing buprenorphine MAT uninterrupted throughout the perioperative period. Clinical guidelines for the perioperative management of buprenorphine from the American Society of Regional Anesthesia and Pain Medicine (ASRA) and other societies is expected in the near future.
Many of these techniques are amenable to continuous blockade via a perineural catheter and should also be considered in this patient population as a means to extend the duration of analgesia.
Acetaminophen is a weak antiinflammatory but potent analgesic and antipyretic medication whose clinical efficacy is well established. It possesses minimal adverse effects, but fulminant hepatic failure can occur if daily consumption exceeds the recommended 4 g per day. Unlike NSAIDs, acetaminophen does not impair platelet or kidney function or cause gastric ulceration. Although its exact analgesic mechanism is unknown, central inhibition of PG synthesis via the cyclooxygenase (COX) pathway is believed to be involved.
When incorporated into a multimodal analgesic regimen, acetaminophen has been shown to reduce pain and opioid consumption after TJA. Prophylactic acetaminophen has also been shown to reduce the incidence of postoperative nausea and vomiting (PONV).
Oral and IV acetaminophen are the most common routes of perioperative administration, and both have been shown to reduce pain and opioid consumption when incorporated into a multimodal analgesic regimen. When IV acetaminophen (Ofirmev; Cadence Pharmaceuticals, San Diego, CA) was first approved by the US Food and Drug Administration (FDA) in 2010, it was hoped that it would augment the benefits of oral acetaminophen. However, studies comparing the two formulations did not demonstrate that IV acetaminophen was associated with superior outcomes. , , Therefore, without a clear clinical advantage, the routine use of the more expensive IV acetaminophen is not recommended. Instead, oral acetaminophen, which is comparable in safety and efficacy but also more cost-effective, is the recommended formulation. ,
Perioperative oral NSAIDs have consistently demonstrated a reduction in opioid consumption and pain intensity. NSAIDs reduce pain and inflammation by inhibiting COX-1 and COX-2 enzymes, which catalyze the conversion of arachidonic acid into PGs and thromboxane A2 ( Fig. 3.3 ). The PGs produced by the constitutively active COX-1 enzyme are responsible for protecting the gastrointestinal mucosal lining, platelet aggregation, and other homeostatic functions. In contrast, COX-2 is an inducible isoform associated with the production of PGs that produce pain, inflammation, and fever.
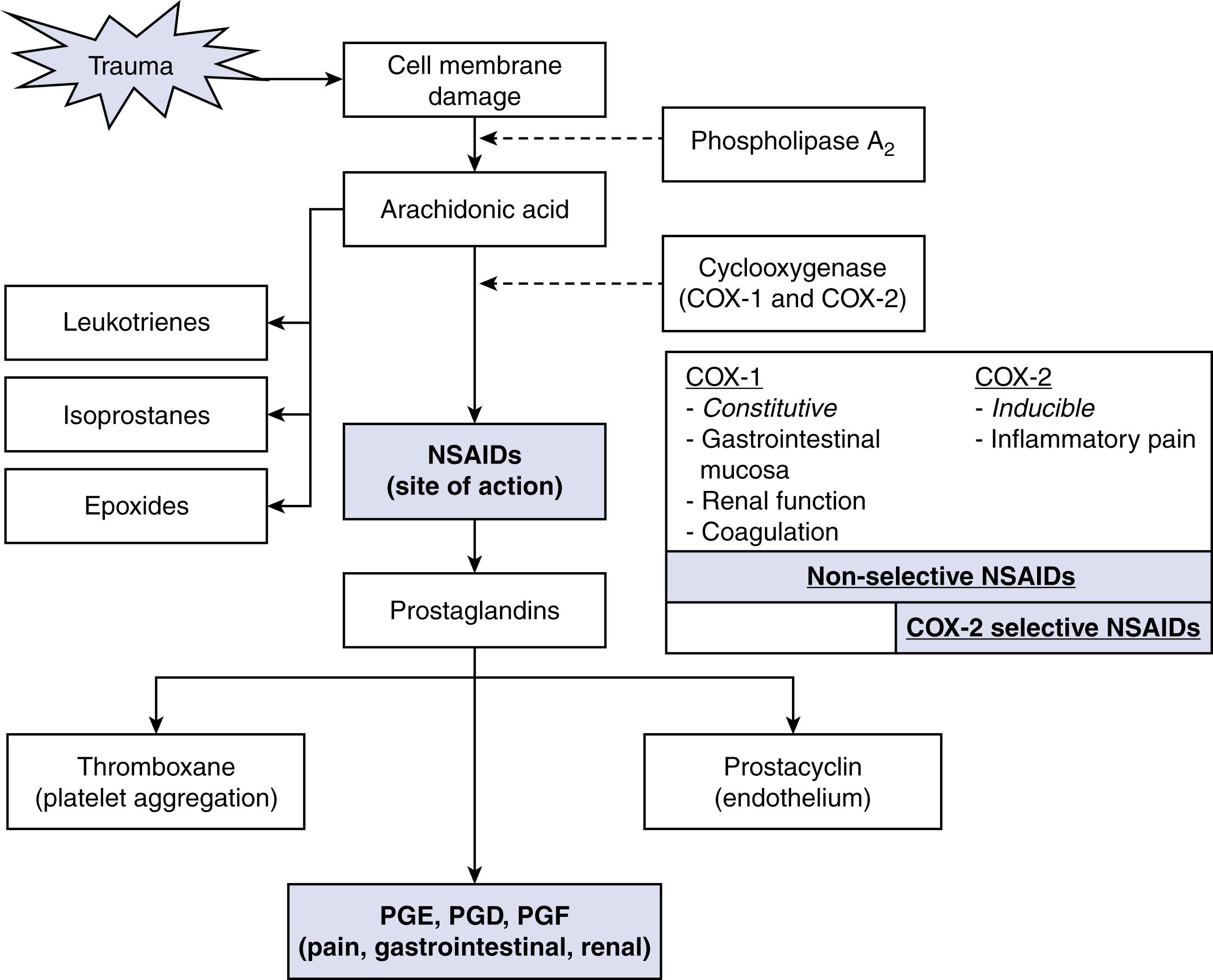
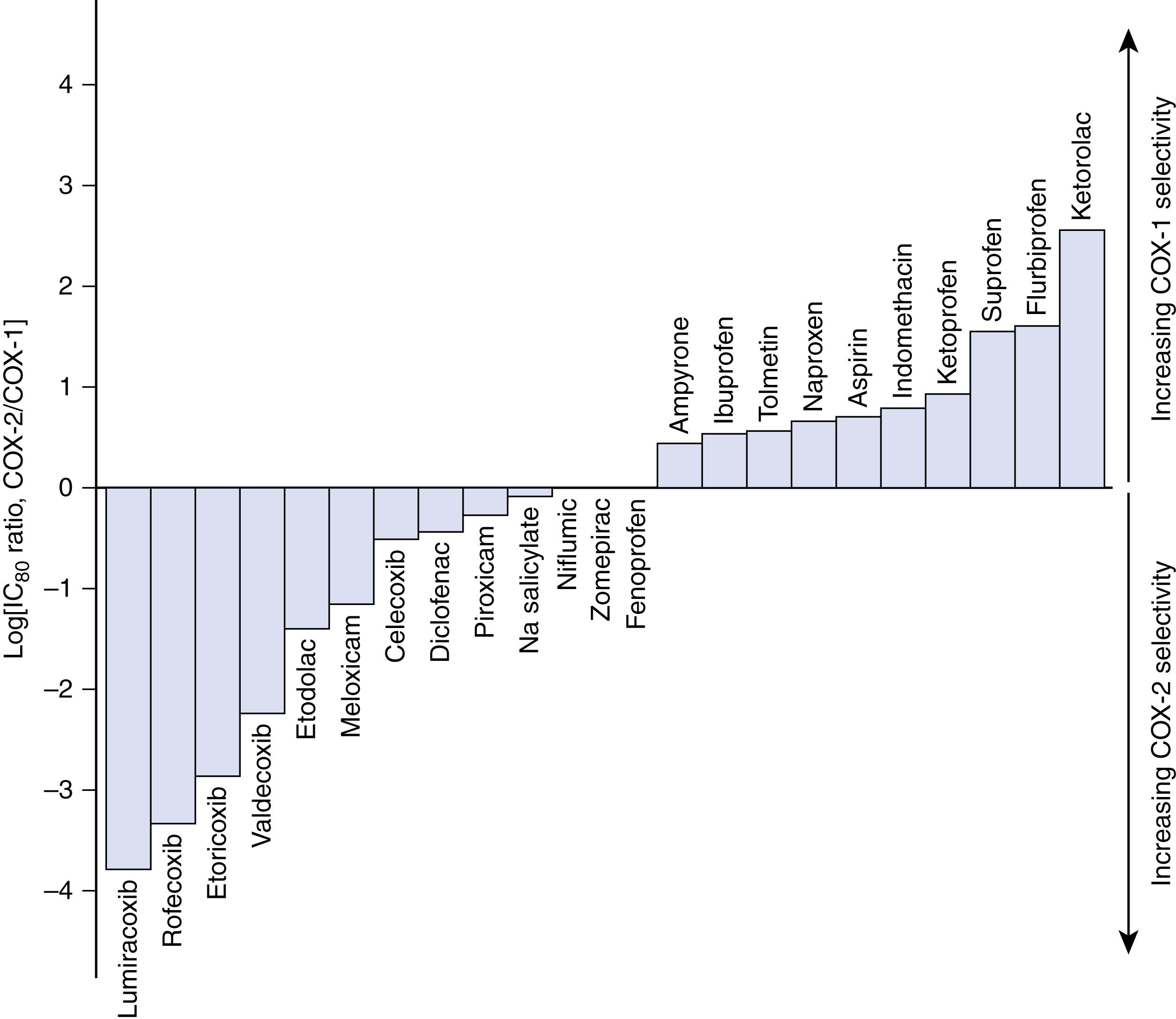
NSAIDs are classified as either selective COX-2 inhibitors or nonselective (COX-1 and COX-2) inhibitors. Both classes of NSAIDs have been shown to consistently reduce pain and opioid consumption, but COX-2 inhibitors are believed to possess a superior adverse effect profile. This claim is controversial; all NSAIDs, regardless of class, carry a warning from the FDA due to their risk of gastrointestinal bleeding, stroke, myocardial infarction, and acute kidney injury.
Celecoxib is the only highly selective COX-2 NSAID available in the United States ( Fig. 3.4 ). Perioperative celecoxib has been shown to significantly improve resting pain at 48 and 72 hours, decrease opioid consumption, and improve active range of motion following TKA. In a study by Buvanendran et al., the perioperative administration of a COX-2 inhibitor reduced opioid consumption, pain, PONV, sleep disturbance, and improved knee range of motion after TKA.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here