Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The population is aging as people are living longer.
The geriatric population comprises a significant proportion of otolaryngologic practice.
Presentation of diseases in older adults may be different than in the younger population, and the spectrum of disease may be different.
Diagnosis, prognosis, and goals of therapy need to be discussed with patients and their families.
Sensory disorders that affect hearing, speech, taste, smell, and balance are common and may significantly affect the quality of life of the geriatric patient.
Awareness of how otolaryngologic processes affect the geriatric population will allow for better satisfaction for patients, their families, and the otolaryngologists involved in caring for this population.
The social and economic significance of the so-called graying of America has received a great deal of attention. The US population of adults over age 65 is expected to double by 2050, growing from 43.1 million in 2012 to a projected 83.7 million and representing 20% of the total population. Disability and disease are more prevalent in older populations, and nearly half of lifetime-per-capita health expenditures occur after the age of 64. The expected growth of the geriatric population therefore portends a significant impact on society's social, economic, medical, and ethical needs and obligations.
Together, geriatricians and other specialists in geriatric medicine and surgery are developing a system of care that keeps older adults healthier, more functional, and more independent at older ages than was previously possible. Otolaryngologists play a role as communication specialists and are a key resource for helping older adults avoid isolation. The American Society of Geriatric Otolaryngology was formed in 2007 to help the specialty present a unified position in treating geriatric patients with otolaryngologic conditions.
The normal process of aging affects all parts of the ear, but the greatest clinical impact is on cochlear and vestibular function. Age-related hearing loss, or presbycusis, is the most common type of auditory dysfunction and is thought to be due to a series of insults over time that include age-related degeneration, noise exposure, and diseases of the ear. Presbycusis is greatly affected by genetic background, diet, and systemic disease. Vestibular symptoms are present in more than half of older adults. Because balance depends on input from the ears, eyes, and peripheral sensory systems, all of which degenerate over time, impaired function in any of these systems contributes to vestibular complaints.
The pinna is commonly involved in actinic disorders, especially basal and squamous cell carcinoma, which makes sun protection and frequent inspection essential. The external auditory canal suffers a decrease in cerumen production because of degeneration of cerumen glands and a reduction in the total number of glands. This may lead to drier cerumen that is less protective of the underlying skin and may result in higher incidence of impaction and infection. Ear canal skin also undergoes atrophy, which results in itching, fragility, and subsequent self-induced lacerations. The use of topical emollients has been recommended for difficult cases.
Hearing loss is highly prevalent in older adults and has been associated with multiple adverse outcomes that include cognitive decline, incident dementia, risk of hospitalization, loneliness, driving impairment, falls, and difficulty walking. Based on data from the National Health and Nutritional Examination Survey (NHANES), 28.1 million US adults aged 60 years and older have clinically significant hearing loss, defined by World Health Organization criteria as a speech-frequency pure tone average (0.5, 1, 2, and 4 kHz) of greater than 25 dB of hearing loss in both ears. Hearing loss prevalence doubles in each decade of life from the second to seventh decade and is present in nearly two-thirds of US adults 70 years and older. Presbycusis may have a devastating effect on older individuals by reducing their ability to communicate, thereby jeopardizing autonomy and limiting opportunities to be active members of society. This decrease in social engagement can have profound consequences, and loneliness is a known determinant of morbidity and mortality in the elderly.
Gacek and Schuknecht initially defined four histopathologic types of presbycusis ( Table 13.1 ). Subsequently, two more categories were added: mixed and indeterminate. The indeterminate category alone may account for 25% of cases, and a mixture of pathologic changes may be present most of the time.
| Histopathologic Type | Audiometric Characteristics | Histologic Findings |
|---|---|---|
| Sensory |
|
|
| Neural |
|
|
| Strial (metabolic) |
|
|
| Inner ear conductive |
|
|
Presbycusis is a multifactorial condition that represents the lifetime accumulation of both intrinsic and extrinsic insults on the inner ear, including the inner and outer hair cells, stria vascularis, and afferent spiral ganglion neurons.
Recent epidemiologic studies on age-related hearing loss have suggested four primary categories of risk factors for presbycusis: cochlear aging, noise exposure, genetic predisposition, and health comorbidities. The mechanistic pathways for these risk factors tend to be overlapping, but specific changes are also relevant to each category.
Increasing age has shown a strong, consistent association with risk of hearing loss across multiple studies. Increased mutations and deletions in mitochondrial DNA are thought to be a contributing factor in this association. Temporal bone studies have observed higher frequency of common aging deletions within the cochlea in subjects with age-related hearing loss compared with normal hearing controls. Markaryan and colleagues found a significant difference in the level of common aging deletions in mitochondrial DNA in the cochlea based on severity of hearing loss.
Noise exposure is a well-established cause of hearing loss and risk factor for presbycusis. Recent studies have shown substantial overlap in the pathways involved in noise-induced cochlear damage and the cumulative effects of aging. Reactive oxygen species are believed to play a major role in cochlear aging, and they are also generated in response to noise exposure. While the direct correlation between noise-induced inner ear damage and the frequency, intensity, and duration of noise exposure has been well established, some argue that noise exposure causes hearing loss at any age and is not true presbycusis. Indeed, recent studies on the interaction of noise-induced hearing loss and age-related hearing loss are contradictory and variable, likely secondary to the underlying influence of other intrinsic and environmental variables on both mechanisms. More research is needed to determine the differential effects of noise exposure on the elderly.
Male sex and race have both been associated with hearing loss in multiple studies. When evaluating the relationship between hearing loss and race, blacks consistently demonstrate 60% to 70% lower odds of hearing loss compared with whites. One hypothesis for this association is that the higher melanin production that results in darker skin color also indicates higher levels of melanin produced by strial melanocytes within the cochlea, and this intracochlear melanin protects the stria vascularis by acting as a free-radical scavenger.
Aside from the genetic contributions of sex and race, presbycusis has been found to cluster strongly in families. Approximately half of the variability in presbycusis may be attributed to genes. The effect of genes is more pronounced for the strial atrophy pattern of hearing loss (flat audiogram) than the sensory phenotype (high-frequency loss). Interestingly, many of the candidate genes associated with hearing loss are also associated with oxidative stress and atherosclerosis. Proposed genes in recent studies include those that code for glutathione peroxidase and superoxide dismutase, two antioxidant enzymes active in the cochlea. Endothelin 1, a potent vasoactive peptide involved in the development of atherosclerosis, can also produce long-term constriction of the spiral modiolar artery, leading to ischemia in the inner ear. An association between a single-nucleotide polymorphism within the END1 gene and hearing loss has been observed in middle-aged and elderly Japanese.
Other factors associated with age-related hearing loss include smoking and circulatory disorders such as hypertension, cardiovascular and cerebrovascular disease, and diabetes. In the Framingham cohort, coronary artery disease, stroke, intermittent claudication, and hypertension were all linked to hearing loss. Many of the gene candidates associated with hearing loss are also associated with atherosclerosis. Overall the data on cardiovascular disease and hearing loss are inconclusive, however, and more work is needed to further characterize the relationship between hearing loss and atherosclerosis.
Hearing loss correlates with smoking and diabetes. In a population of over 3700 adults aged 48 to 92 years, Cruickshanks and colleagues found that smokers were 1.69 times more likely to have hearing loss than nonsmokers. A dose-dependent effect of smoking on high-frequency hearing loss was also noted in a recent multicenter study. An interaction between noise exposure and smoking was also apparent in the NHANES cohort: heavy smokers who reported firearm use demonstrated a significantly greater hearing loss than nonsmokers with similar noise exposure. Diabetes has also been associated with hearing loss in multiple studies and may involve physiologic mechanisms similar to cardiovascular disease and smoking.
Despite the complexity of factors that contribute to age-related hearing loss, substantial overlap exists in the mechanistic pathways involved. Regardless of the etiology or mechanistic pathway involved, presbycusis tends to produce the same functional outcome—namely, a disproportionate effect on speech understanding and perception in noise. Elderly who suffer from age-related hearing loss tend to struggle with an inability to understand words, which leads to complaints such as, “I can hear you, but I can't understand you,” or “You're just mumbling.” As presbycusis worsens, the ability to extract meaning from day-to-day conversations diminishes, such as hearing “I’ll see you someday ” instead of “I’ll see you Sunday. ”
Recent studies have observed an independent association between hearing loss in older individuals and cognitive decline. Hearing loss has been associated with increased risk for incident dementia and incident cognitive impairment in multiple longitudinal observational cohorts. This relationship has substantial implications for the health and well-being of elderly patients with presbycusis. A pilot randomized trial to evaluate whether hearing loss treatment in older adults can reduce cognitive decline has been completed. The full trial, called Aging and Cognitive Health Evaluation in Elders (ACHIEVE), began enrollment in late 2017. The results of this study may have substantial implications for reducing risk of cognitive decline and dementia in aging populations.
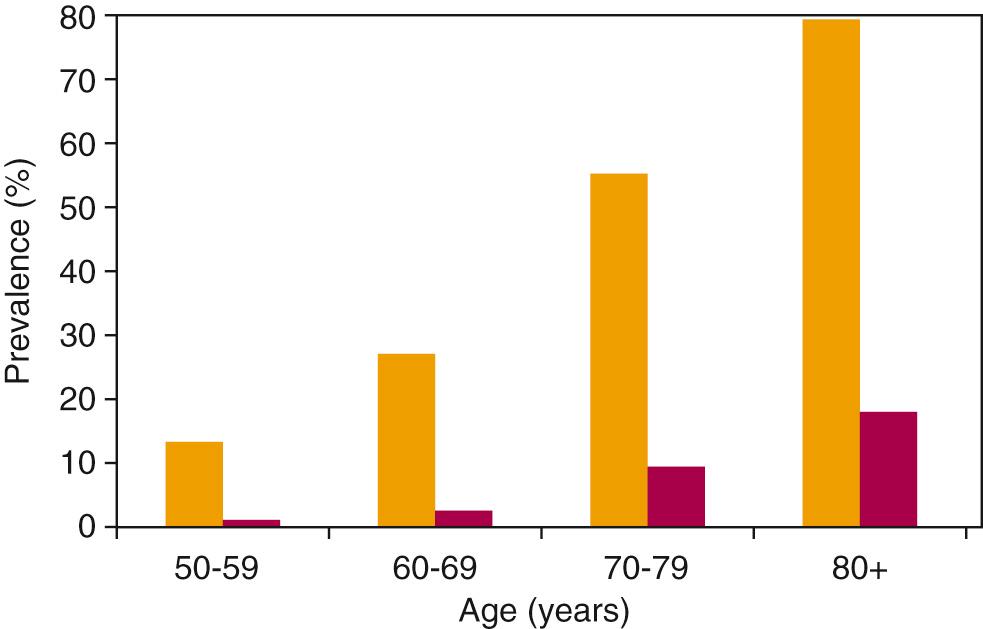
Amplification represents the mainstay of treatment for age-related hearing loss; however, the current use of hearing aids in the United States remains low. Only 3.8 million people, or 14.2% of individuals over age 50 with hearing loss in the NHANES cohort, wear hearing aids. While hearing aid use increases with every decade of life, the prevalence of untreated hearing loss grows even faster in each age category ( Fig. 13.1 ). There are an estimated 20 million US adults over age 60 living with untreated, clinically significant hearing loss. The cost of conventional amplification likely contributes to disparities in access to hearing care, particularly for the 6 million low-income older adults living with untreated hearing loss in the United States. Both racial and socioeconomic differences in hearing aid utilization have been reported. Other barriers to hearing aid use include stigma, perceived ineffectiveness, discomfort, ongoing maintenance costs, and cosmesis.
With the high cost of traditional amplification and continued disparities in access to hearing care, recent emphasis has been placed on personal sound amplification products (PSAPs) and low-cost, community-based hearing interventions for older adults. A recent study by Reed and colleagues compared the efficacy of PSAPs to conventional hearing aids. Three of the five PSAPs evaluated in this study demonstrated improvement in speech understanding that was comparable to improvements with a conventional aid, while one PSAP showed no improvement and one model actually worsened patients’ speech understanding. A pilot randomized trial of a community-delivered hearing intervention using over-the-counter amplification devices demonstrated feasibility and preliminary efficacy of the intervention. Another pilot study of an over-the-counter, community-based hearing intervention for dementia patients demonstrated feasibility and was viewed favorably by caregivers in their qualitative responses ( Box 13.1 ).
There is a growing national debate over how to provide accessible and affordable hearing health care. The National Academies of Sciences, Engineering, and Medicine published a report in 2016 on improving hearing health care for adults that has served as a harbinger for change. Similar to the President's Council of Advisors on Science and Technology recommendation in late 2015, the Academies’ committee advised that the Food and Drug Administration (FDA) create a new category of “over the counter” wearable hearing devices for mild to moderate hearing loss to provide an alternative, consumer-driven approach to the current medical model for hearing loss treatment. Legislation was signed into law in 2017 that requires the FDA to create a new category of over-the-counter hearing devices. By opening this market, the 2017 legislation is expected to increase access, decrease costs, and expand amplification options available to patients.
Aside from amplification, correction of health factors that may impact age-related hearing loss—such as smoking, hypertension, and cholesterol levels—should also be considered. A randomized trial is concluding at the time of this writing to assess whether a daily-administered 100 mg aspirin tablet decreases the progression of presbycusis by addressing inflammation and microvessel disease that may be contributing to age-related hearing loss.
Cochlear implantation may play a role in treating older adults with severe to profound sensorineural deafness. Such a degree of hearing loss is most often due to an underlying pathologic process such as Meniere disease or otosclerosis in combination with presbycusis; the latter does not produce this degree of hearing impairment on its own. A study of 749 adolescent and adult cochlear implant recipients found that age was a clinically insignificant predictor of audiologic outcome from cochlear implantation, compared with duration of profound deafness and residual speech recognition. Multiple studies have found improvements in quality-of-life scores in elderly cochlear implant recipients, and one study noted a significant reduction in symptoms of loneliness after cochlear implantation that was not achieved with hearing aids. As discussed previously, comorbid chronic health conditions seen more commonly in older populations will play a role in surgical planning and perioperative management.
Overall, the goal of treatment for presbycusis is to enable effective communication in all settings. Although addressing peripheral functioning deficits through hearing aids, over-the-counter amplification devices, and cochlear implants is essential, the growing body of evidence demonstrating a strong link between hearing loss and cognitive decline suggests that a more comprehensive management strategy is necessary. Expansion of diagnostic testing to include speech in noise and cognitive evaluation, as well as incorporation of rehabilitation and counseling, will contribute to this more comprehensive approach.
Presbystasis, the dysequilibrium of aging, is a group of disorders that affect the mobility of a large number of older persons. Because of the degeneration of the vestibular, proprioceptive, and visual senses, the ability to walk and drive can be reduced to the point of incapacitation; lessening spatial-orientation abilities contribute to this as well. Falls are one of the most common concerns relating to imbalance in older adults, and falls often lead to functional decline, anxiety, depression, social withdrawal, and increased medical costs.
Input from the vestibular, visual, proprioceptive, and other systems can be thought of as providing input into a common central processor that in turn controls posture and eye movement. Disorders of these sensory organ systems have traditionally been treated by otolaryngologists, neurologists, and ophthalmologists, depending on the organ system causing the most obvious dysfunction. However, development of the unifying discipline of neurotology has led to an integrated approach to, evaluation of, and care for older adults who experience dysequilibrium. Otolaryngologists must be aware of other causes of dysequilibrium or dizziness, because a variety of organ systems may contribute to these difficulties. For example, side effects of psychotropic medications, abnormalities in blood pressure, leg muscle weakness, neuromotor disorders such as Parkinson disease, and generalized loss of coordination can contribute to feelings of dysequilibrium and dizziness. The failure of one organ system can be overcome with compensation, but with multisystem failure, increasingly severe deficits occur.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here