Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
A 35-year-old man sought medical attention for leg weakness. He noticed difficulty getting up from low chairs and ascending stairs. Looking back, he reported that 10 years previously he had noticed some difficulty when trying to stand after squatting to paint the baseboard in his dining room; he had to push on his thighs to straighten his knees. He denied weakness in his arms and had no problems with chewing or swallowing. He had no sensory complaints. He reported occasional muscle cramps and twitches in his legs. His examination demonstrated mild weakness in bilateral shoulder abductors and moderate weakness in the proximal legs, mainly hip flexors, hip extensors, and knee extensors. He had normal facial and tongue strength as well reflexes and sensation. Nerve conduction studies were normal, including sensory potentials. Electromyography showed diffuse, predominantly chronic denervation changes and survival motor neuron (SMN) genetic testing revealed a homozygous deletion in the SMN1 gene, with five copies of SMN2, consistent with the diagnosis of spinal muscular atrophy type 4. Ten years later he remained active in his job and ambulated independently.
In this chapter we address the motor neuron diseases (MNDs) other than amyotrophic lateral sclerosis (ALS). MNDs are disorders that produce painless weakness, atrophy, cramps, and fasciculations; they are a consequence of the degeneration of anterior horn cells and selective cranial motor nuclei. We also address multifocal motor neuropathy (MMN) and hereditary spastic paraparesis (HSP). MMN and HSP are considered separate categories of disorders from MNDs, as MMN and HSP preferentially target motor nerves and corticospinal tracts, respectively, rather than motor nuclei. However, these conditions may have overlapping phenotypes, with MMN sometimes resembling lower motor neuron–dominant ALS and HSP mimicking primary lateral sclerosis (PLS); thus they deserve to be discussed in this chapter.
Many of the disorders discussed in this chapter have known or suspected genetic mechanisms. The spinal muscular atrophies (SMAs) are conceptualized as largely inherited disorders in which there is predominant degeneration of anterior horn cells and selective motor cranial nerve nuclei. In childhood SMAs, mutations of a single gene and derangement of a single gene product are responsible for the majority of cases, and the resultant phenotype is fairly homogeneous. In other disorders, for example, HSP, there are a variety of recognized genotypes associated with a heterogeneous array of phenotypic variations.
SMA types I–IV are allelic disorders of the survival motor neuron (SMN) gene 1 located on chromosome 5q12.2–q13.3. When there is more than one affected individual in a given family, the phenotype is typically homogeneous but may be variable in some cases. In normal individuals, there are two copies each of the SMN1 and SMN2 genes. Although both genes produce similar proteins, the SMN2 gene appears to produce an unstable and rapidly degrading protein that compensates to a variable degree for the lack of the SMN1 protein. There are no known clinical consequences from mutations of the SMN2 gene alone.
It is estimated that 95% of SMA I–III patients are homozygous for deletion of exons 7 and 8 of the SMN1 gene. The remainder are thought to be compound heterozygotes with absence of exons 7 and 8 on one allele and a point mutation of the other SMN1 allele. The severity of the SMA phenotype appears to be related to the number of SMN2 copies available to compensate for the deleted SMN1 gene. Homozygotes devoid of SMN1 who harbor two copies of SMN2 tend to manifest as an SMA I phenotype. An increasing number of SMN2 copies correlates with proportionately milder (SMA II–IV) forms of the disease. Individuals homozygous for the SMN1 mutation with five copies of the SMN2 gene have been reported to be asymptomatic. Why motor neurons remain selectively vulnerable to SMN deficiency remains unknown.
SMA is considered to be the most common cause of mortality in infancy among autosomal recessive disorders. The natural history of SMA is variable. For this reason clinical subgroups have historically been defined based on the best motor function attained during development. Type I SMA infants do not sit independently, type II SMA individuals sit at some point but are not able to walk, and type III SMA children and adults are able to walk independently during their childhood.
Of the multiple SMA phenotypes, the infantile and childhood forms are the most prevalent. SMA type I or Werdnig-Hoffman disease is the most severe form ( Fig. 63.1 ). Its incidence is between 4 and 10 in 100,000 lives births depending on the population studied. Clinical manifestations become evident within the first 6 months of life. In some cases recognition of reduced movement occurs in utero or within the first few days of life. Affected infants are hypotonic with symmetric weakness that is either generalized or proximally predominant. As in ALS, facial weakness is typically mild and extraocular muscles are spared. Fasciculations are seen in the tongue but rarely in limb muscles, presumably because of the ample subcutaneous tissue of neonates. Manual tremor, characteristic of SMA types II and III, is rarely present. Deep tendon reflexes are typically absent. Abdominal breathing, a weak cry, and a poor suck are common. Ventilatory difficulties stem primarily from intercostal rather than diaphragmatic weakness. Consequently pectus excavatum and a diminished anteroposterior diameter of the chest are seen. Mild contractures may occur, but arthrogryposis is not part of the classic phenotype. Intellectual development is normal. Without mechanical ventilation, death is inevitable, almost always within a year or two. An earlier age of onset correlates with a shorter life expectancy.
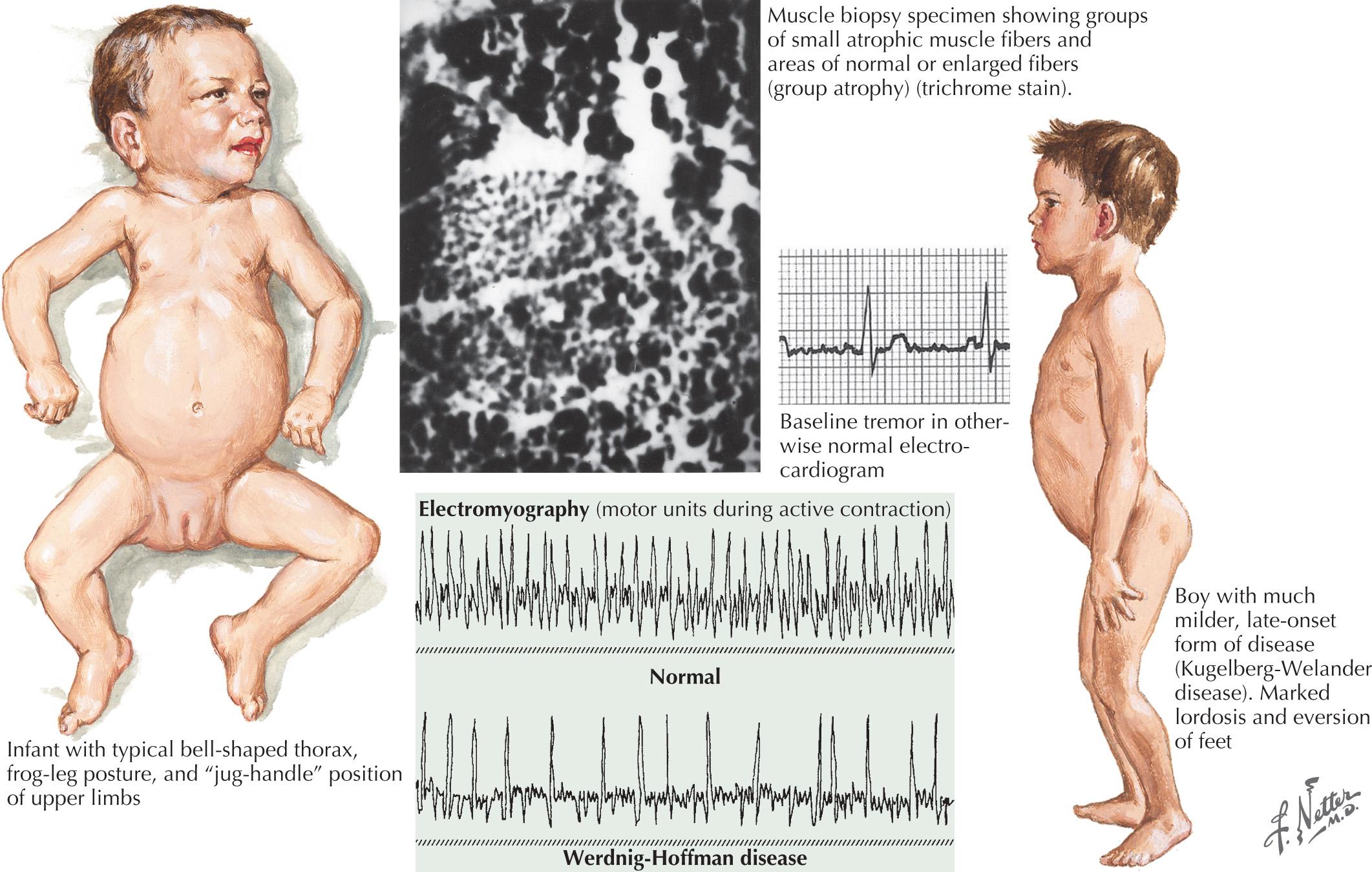
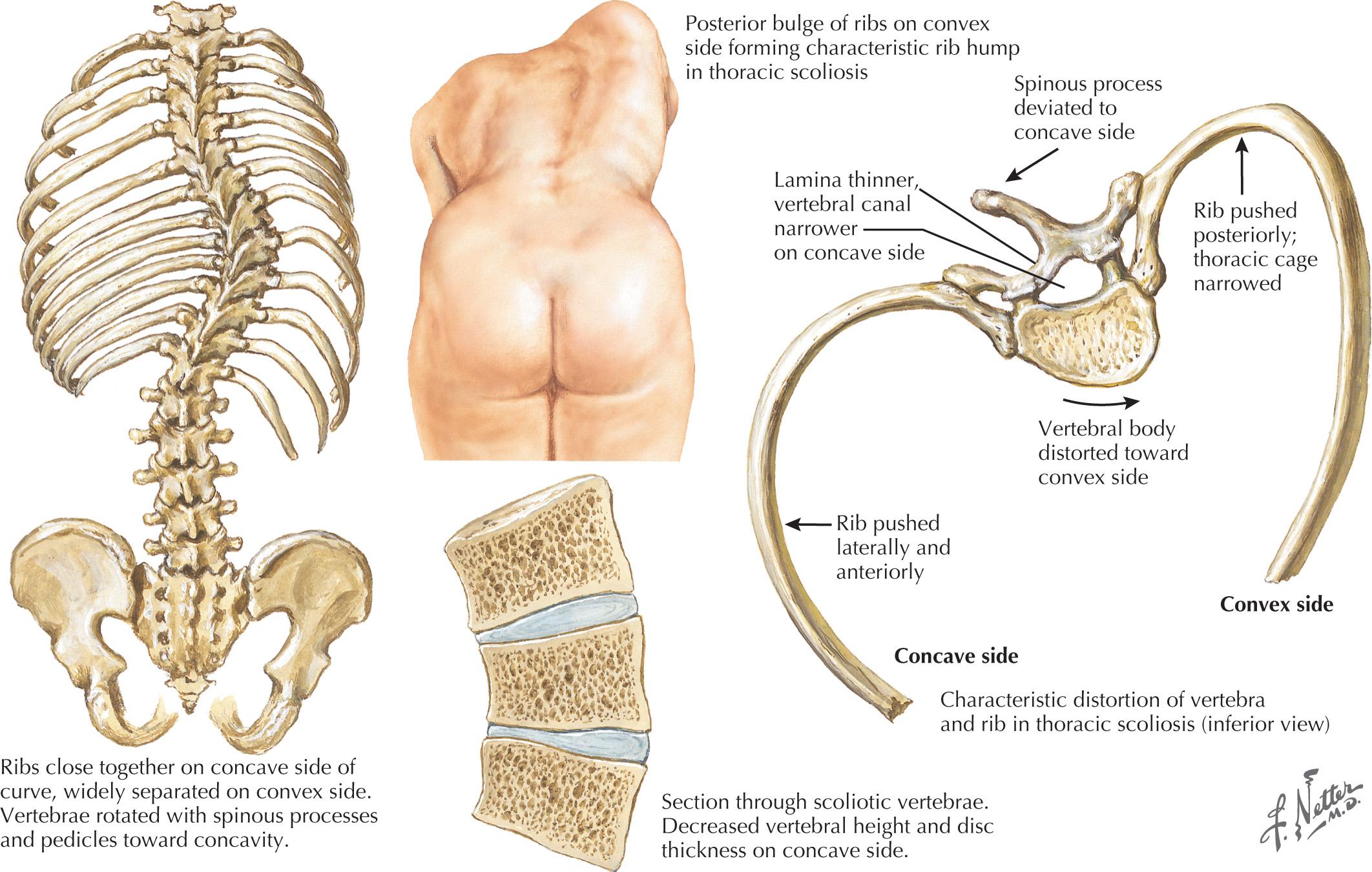
The intermediate form of SMA II typically begins between 6 and 18 months of age. The disorder is clinically defined by a child who sits independently but never walks. Postural hand tremor is the only significant phenotypic variance from Werdnig-Hoffman disease. Tongue fasciculations, areflexia, and a generalized to proximally predominant and symmetric pattern of weakness mimic the SMA I phenotype. Approximately 98% of these individuals survive to age 5 and two-thirds to age 25. In view of the more protracted course and of wheelchair dependency, SMA II and SMA III patients commonly acquire kyphoscoliosis and joint contractures ( Fig. 63.2 ).
SMA III or the Kugelberg-Welander syndrome differs from the intermediate form only in the age of onset, milestones achieved, and life expectancy. Affected individuals develop the ability to stand and walk. Onset age is typically 18 months or more. Some authors have attempted to divide SMA III into type a and type b, based on age at onset of symptoms, with the intention of better defining the natural history in individual patients. In SMA type IIIa, defined as symptom onset before 3 years, it is estimated that 70% will remain ambulatory 10 years after symptom onset and 20% will still ambulate 30 years after symptom onset. In SMA type IIIb, defined as symptom onset after 3 years, virtually all patients will remain ambulatory at 10 years and 60% at 40 years after symptom onset. Life expectancy extends into the sixth decade and may be normal in many individuals. Initial symptoms are typically related to proximal weakness. Hand tremor, areflexia, and tongue fasciculations are commonplace. Fasciculations in limb muscles are more evident than in SMA types I and II.
Adult-onset SMA IV is a rare genetically heterogeneous disorder. SMA IV children achieve motor milestones at normal ages. Onset of weakness is typically in the third or fourth decade in the recessively inherited cases. Initial symptoms are typically proximal weakness of the lower extremities, particularly the hip flexors, hip extensors, and knee extensors. Shoulder abductors and elbow extensors are the most frequently affected muscles of the arms. Tongue fasciculations, hand tremor, and in some cases calf hypertrophy may occur. Life expectancy in SMA IV is normal. Unlike SMA I-III, SMA IV may be autosomal dominant or recessive.
As in ALS, a multidisciplinary model of care is used in SMA to address socioemotional, nutritional, orthopedic, physical/occupational therapy, and ventilatory issues. Genetic counseling is recommended for families. Comanagement of patients with orthopedic colleagues is important in these patients, particularly in regard to surveillance for spinal curvature and bone health once a patient becomes nonambulatory.
In 2016, the US Food and Drug Administration (FDA) approved the first agent, nusinersen, for SMN-related SMA following multiple studies including a sham-controlled phase 3 trial. Nusinersen is an antisense oligonucleotide that is delivered intrathecally and allows SMN2 to produce full-length protein. It is hoped that ongoing studies will provide insight into its long-term safety and efficacy in children. To date, the evidence of efficacy appears to be highest for treatment of infantile and childhood-onset SMA, and the benefit for older SMA patients remains unclear.
In 1968, Kennedy and colleagues first described the syndrome of X-linked bulbospinal muscular atrophy, and in 2001, LaSpada and colleagues identified the associated trinucleotide repeat mutation on exon 1 of the androgen receptor gene. This relatively rare disorder typically affects men in their 40s. Initial symptoms are often nonspecific and consist of muscle cramps, tremor, and subtle weakness of bulbar or proximal muscles; as the symptoms can be slow and insidious, there can be a delay in diagnosis.
As the name implies, the clinical manifestations are largely referable to degeneration of the lower cranial nerve motor nuclei and anterior horn cells of the spinal cord. The weakness progresses insidiously and is proximally predominant and symmetric in pattern. Typically, symptoms referable to the lower extremities have the greatest initial impact. Approximately 10% of the time the initial symptoms pertain to difficulty with swallowing, speaking, or chewing and may include jaw drop. Facial weakness is common. Perioral and tongue fasciculations are common and represent helpful clinical clues. As in the case of virtually all MNDs, ptosis and ophthalmoparesis are absent. As with other SMAs, postural tremor is common. There is an associated but frequently asymptomatic sensory neuropathy that may be recognized only by nerve conduction studies. Clinical heterogeneity exists. Asymmetry of muscle weakness at onset has been emphasized by some authors. Occasionally rapidly progressive weakness occurs. The median age of wheelchair dependency is 61 years, or approximately 15 years after onset of weakness. Women who are heterozygous for the Kennedy disease mutation may rarely be symptomatic.
The effects of X-linked bulbospinal muscular atrophy (BSMA) are not restricted to the neuromuscular system. Affected males suffer the consequences of androgen insensitivity, including gynecomastia, impotence, testicular atrophy, and potential infertility. There is also an increased incidence of diabetes mellitus.
In 1963, Hirayama described a slowly progressive focal MND affecting one upper extremity and sometimes both. Males represent 60% of the cases. Hirayama disease is perhaps best considered a segmental or regional form of MND. Onset is typically between ages 15 and 25. Although most commonly reported in persons of Asian origin, it may occur in individuals of any ethnic background.
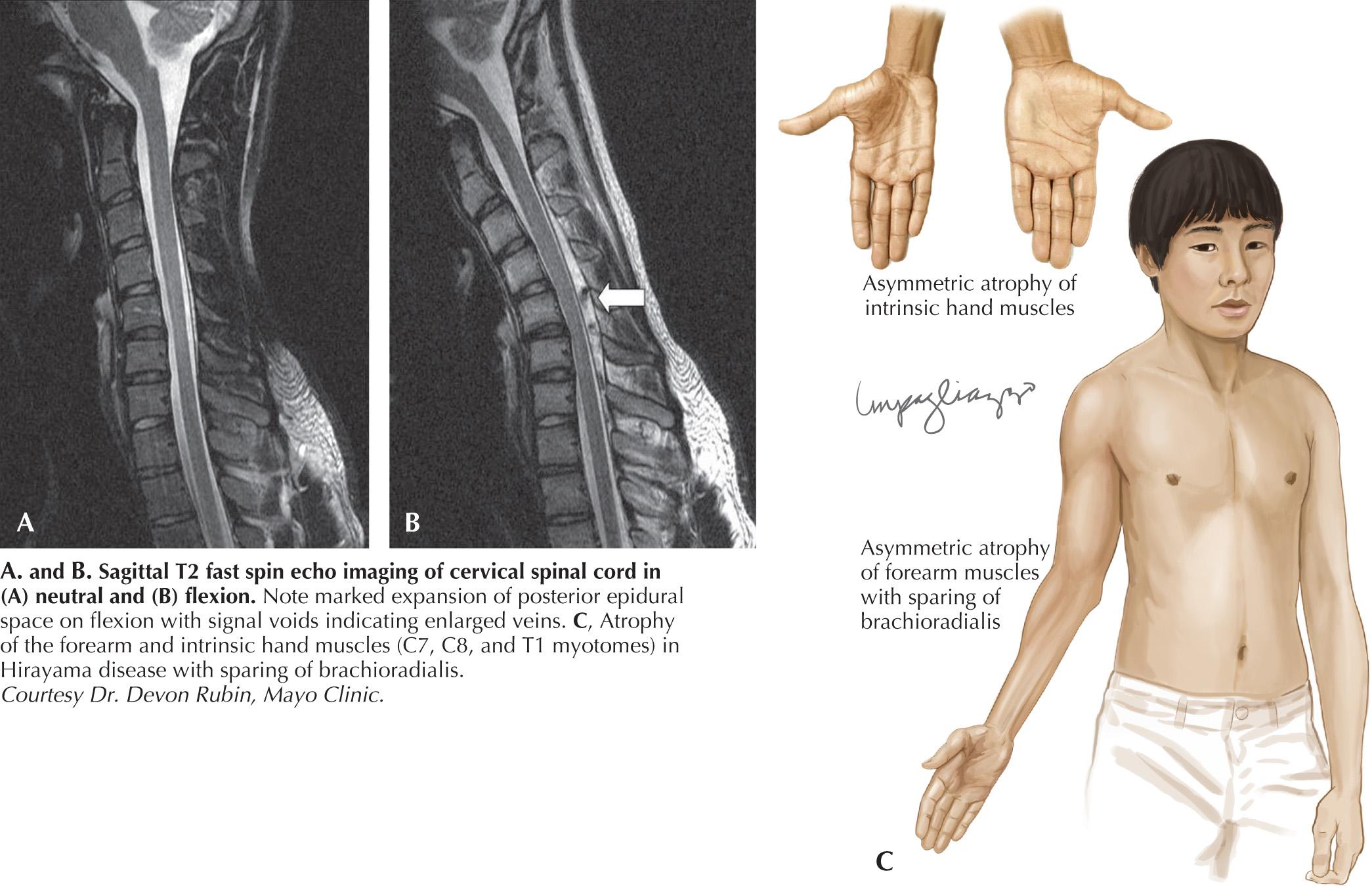
Characteristically the disorder causes the insidious development of weakness and atrophy of the C7–T1 muscles of the hand and forearm. It begins unilaterally, typically in the dominant extremity. Over the course of months to years, the weakness may gradually spread to involve more proximal muscles. In one-third of the cases, there is clinical weakness of the opposite limb. An even higher percentage will have bilateral upper extremity involvement on electrodiagnostic studies. Tendon reflexes in the involved limb may be spared, although neither overt pyramidal nor bulbar involvement occurs. Reflex preservation may reflect the restricted nature of the disease and the lack of a reliable C8–T1 muscle stretch reflex. As in the heritable SMAs, tremor may occur. In most cases, there is an arrest of further progression after 6 years or less. Although a significant decline in affected limb function in the cold is common with all MNDs, “cold paresis” is emphasized in this population. Hyperhidrosis of the involved limb has also been described.
Ischemic changes in the cervical spinal cord of a single autopsied case of Hirayama disease led to the hypothesis of a compressive mechanism. In 2000, Hirayama reported the results of dynamic imaging in 73 patients and 20 controls. Ninety-four percent of the patients had significant forward displacement and flattening of the posterior surface of the cervical cord during neck flexion ( Fig. 63.3 ). Anterior shifting of the cervical and thoracic dura was seen, at times with associated engorgement of the venous plexus on dynamic magnetic resonance imaging (MRI) of the cervical spine. The presumptive mechanism is that the blood supply to the spinal cord is mechanically compromised, the anterior horn being a watershed zone particularly vulnerable to ischemic injury. Other observations supporting this hypothesis include the recognition that the spinal cord is often asymmetrically flattened in this disorder with the weaker arm correlating with the side of the greatest spinal cord compression; it is believed that a similar phenotype may occur in individuals with different mechanisms of cervical spinal cord compression. Specifically, a slowly progressive, asymmetric pattern of bibrachial amyotrophy has been reported in individuals with epidural cerebrospinal fluid (CSF) collections associated with chronic spinal CSF leaks.
A scapuloperoneal pattern of weakness may result from either neurogenic or myopathic disorders. The neurogenic form of the scapuloperoneal syndrome has been referred to by the eponym Davidenkow disease. It has been considered to represent a SMA variant even though distal sensory loss was common in Davidenkow's original series. Symptomatic onset typically occurs in late childhood related to asymmetric weakness of scapular fixators or foot dorsiflexors. Weakness typically progresses into a more generalized pattern. Some patients with a neurogenic scapuloperoneal syndrome have been found to have mutations within the PMP-22 gene. This suggests that the disorder might be more correctly characterized as a hereditary neuropathy.
The syndrome of benign calf amyotrophy (BCA) is a disorder that presents as slowly progressive weakness and atrophy in the distal lower extremity, typically the calf. In a small series of 8 patients with BCA, 50% had unilateral symptoms. Symptoms plateaued after 1 to 3 years. Electromyography (EMG) showed mainly chronic reinnervation changes in distal greater than proximal S1 myotomes, particularly in the calves. The mechanism for BCA is not known.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here