Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The interrelationship between disease and nutrition has been recognised since at least the time of Hippocrates, who is credited with the phrase ‘let food be thy medicine’. Attention to and correction of nutritional status remain fundamental to holistic medical care. Nevertheless, approximately one-third of all patients admitted to an acute hospital will have evidence of protein-calorie malnutrition. In many cases, these abnormalities are not adequately recognised or addressed and, remarkably, two-thirds of patients leave the hospital with either no improvement or even deterioration in their nutritional status. Malnutrition has damaging effects on psychological status (depression), activity levels and appearance. In the surgical patient, low body fat content may be viewed as an advantage, making technical aspects of surgery easier. There is, however, clear evidence that patients with significant protein-energy malnutrition have a significantly greater incidence of postoperative mortality and morbidity, including pneumonia, pressure sores, surgical site infection and prolonged hospital stay.
It is important to recognise that the definition of malnutrition does not simply include nutritional depletion. In Western society there is equally a steadily growing epidemic of obesity. Whilst obese individuals generally have a matching increase in lean body mass, some have underlying muscle wasting (sarcopenic obesity), and these patients are at high risk of metabolic syndrome and postoperative complications. Patients with sarcopenic obesity may be difficult to recognise clinically because their muscle wasting is hidden by overlying fat.
Nutritional disorders in surgical practice have two principal components. First, starvation can be initiated by the effects of the disease, by restriction of oral intake, or both. Simple starvation results in progressive loss of the body’s energy and protein reserves (i.e., subcutaneous fat and skeletal muscle). Second, there are the metabolic effects of stress/inflammation; namely, increased catabolism and reduced anabolism. These result in low serum albumin concentration, accelerated muscle wasting and water retention. Although malnutrition may be the result of simple starvation, in the majority of surgical patients it results from a more complex and interrelated combination of increased demand related to the catabolic effects of illness, reduced food intake and metabolic changes that may result not only in nutrient depletion but also in impaired nutrient utilisation ( Fig. 5.1 ).
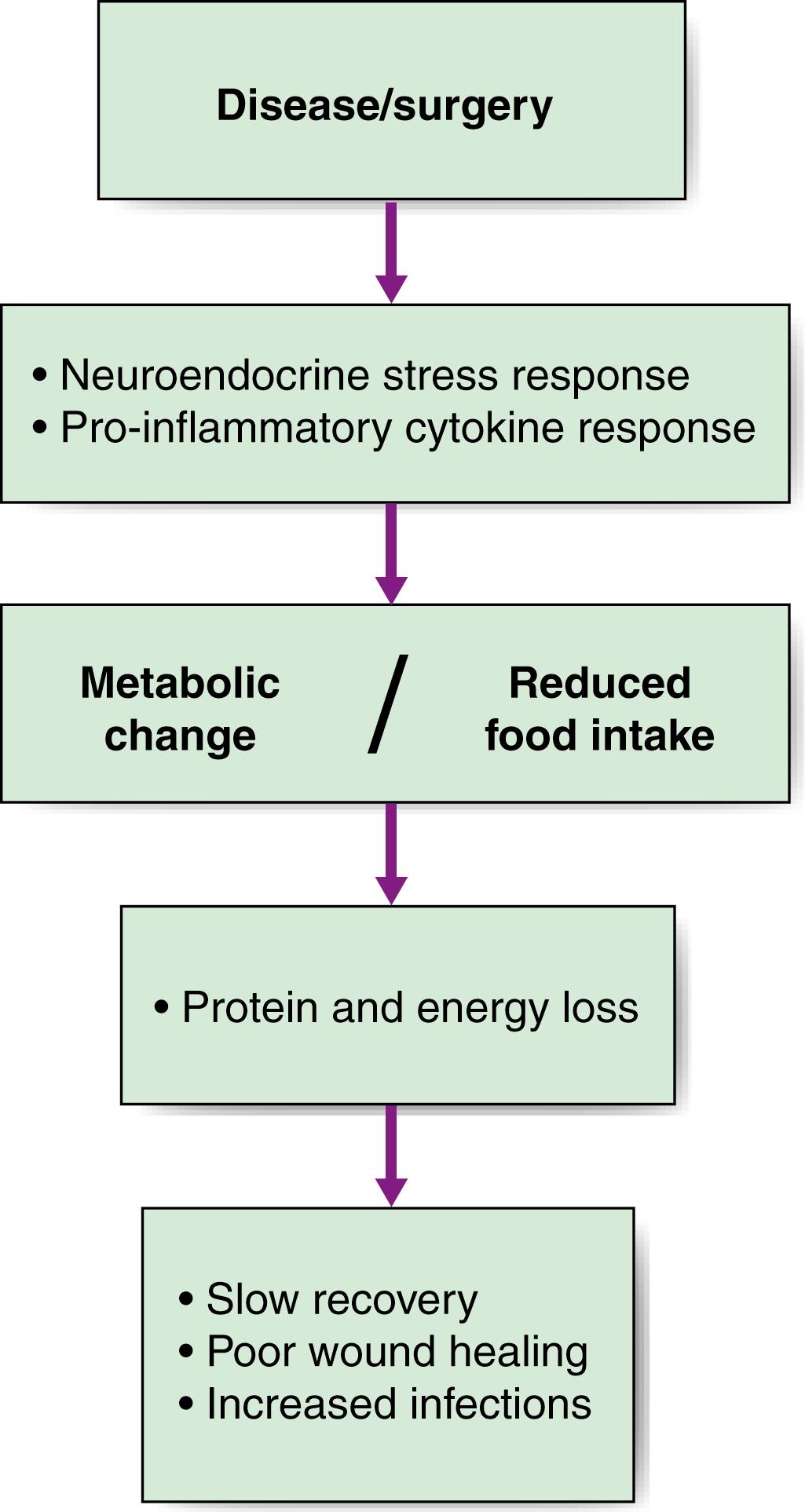
The body’s main energy reserves are found in subcutaneous and intraabdominal fat depots, but loss of fat does not usually impair function. In contrast, there are no true protein reserves in the body. In response to starvation or stress, structural tissues such as skeletal muscle and even the gut are broken down (catabolism) to liberate amino acids for the generation of glucose precursors, and this may ultimately result in functional impairment that can impede recovery.
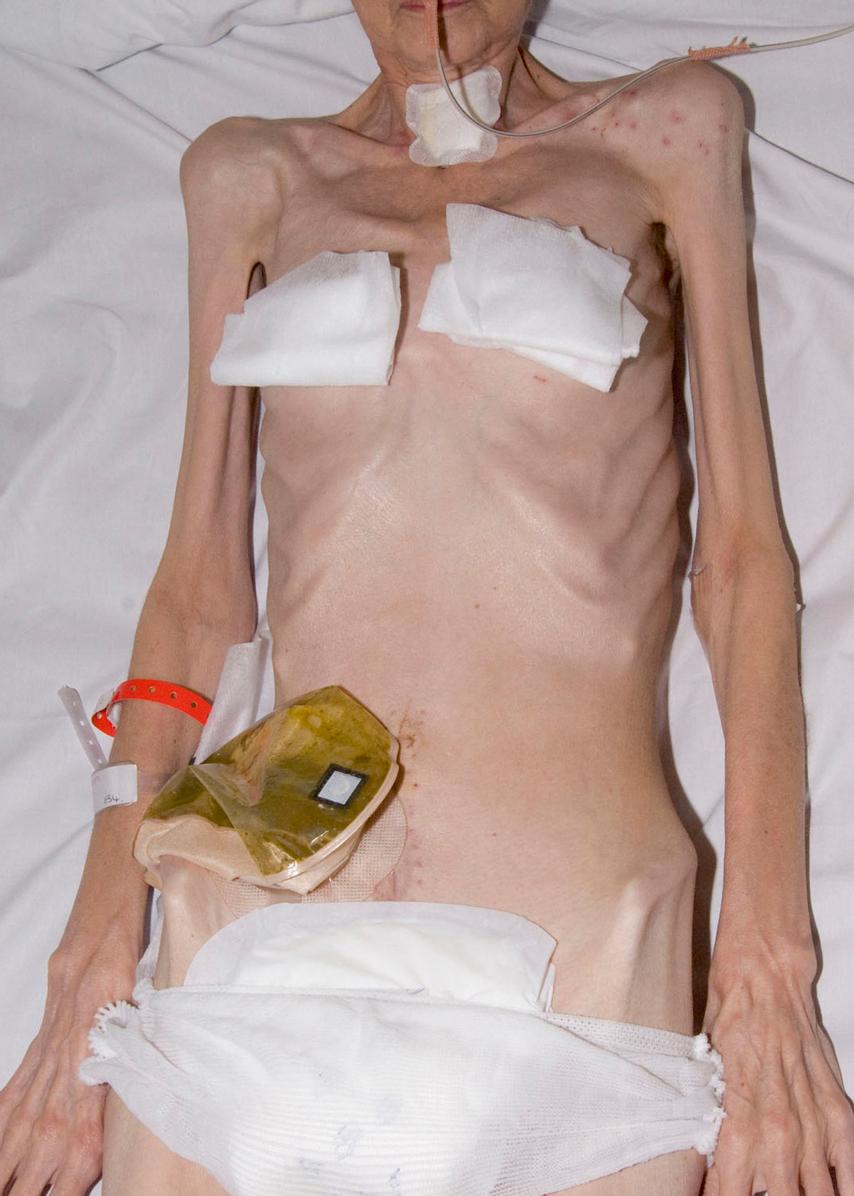
The key elements of nutritional assessment include current food intake, levels of energy and protein reserves, and the patients’ likely clinical course ( Fig. 5.2 ). Patients who have not had an adequate dietary intake for 5 days or more require nutritional support, and those with symptoms such as anorexia, nausea, vomiting or early satiety are at risk of a reduced food intake and hence undernutrition. Energy reserves are most easily assessed by examining for loss of subcutaneous fat (skinfolds), whereas protein depletion most commonly manifests as skeletal muscle wasting ( Fig. 5.3 ). A history of weight loss of more than 10–15% is highly significant, because this level of nutritional depletion has been repeatedly shown to be associated with impaired outcome of surgical treatment. Patients can also be assessed according to their body mass index (BMI), which is calculated as weight (in kg) divided by height (in m 2 ). Normal BMI is between 18.5 and 24.9 kg/m 2 . A BMI of less than 18 suggests significant protein-calorie undernutrition. Finally, it is important to recognise that in assessing the nutritional status of patients, knowledge of their likely clinical course is vital ( Fig. 5.4 ). For example, if patients are well nourished, they should be able to withstand a brief period of reduced food intake associated with major surgery. However, if patients are already severely malnourished (e.g., weight loss of 15%, BMI 17), then even a short period of starvation or catabolism may result in further life-threatening nutritional depletion. Taken together, a patient’s food intake, level of reserve and likely clinical course should indicate the need for nutritional support, and these assessments should form part of the routine daily appraisal of every patient during a surgical ward round.
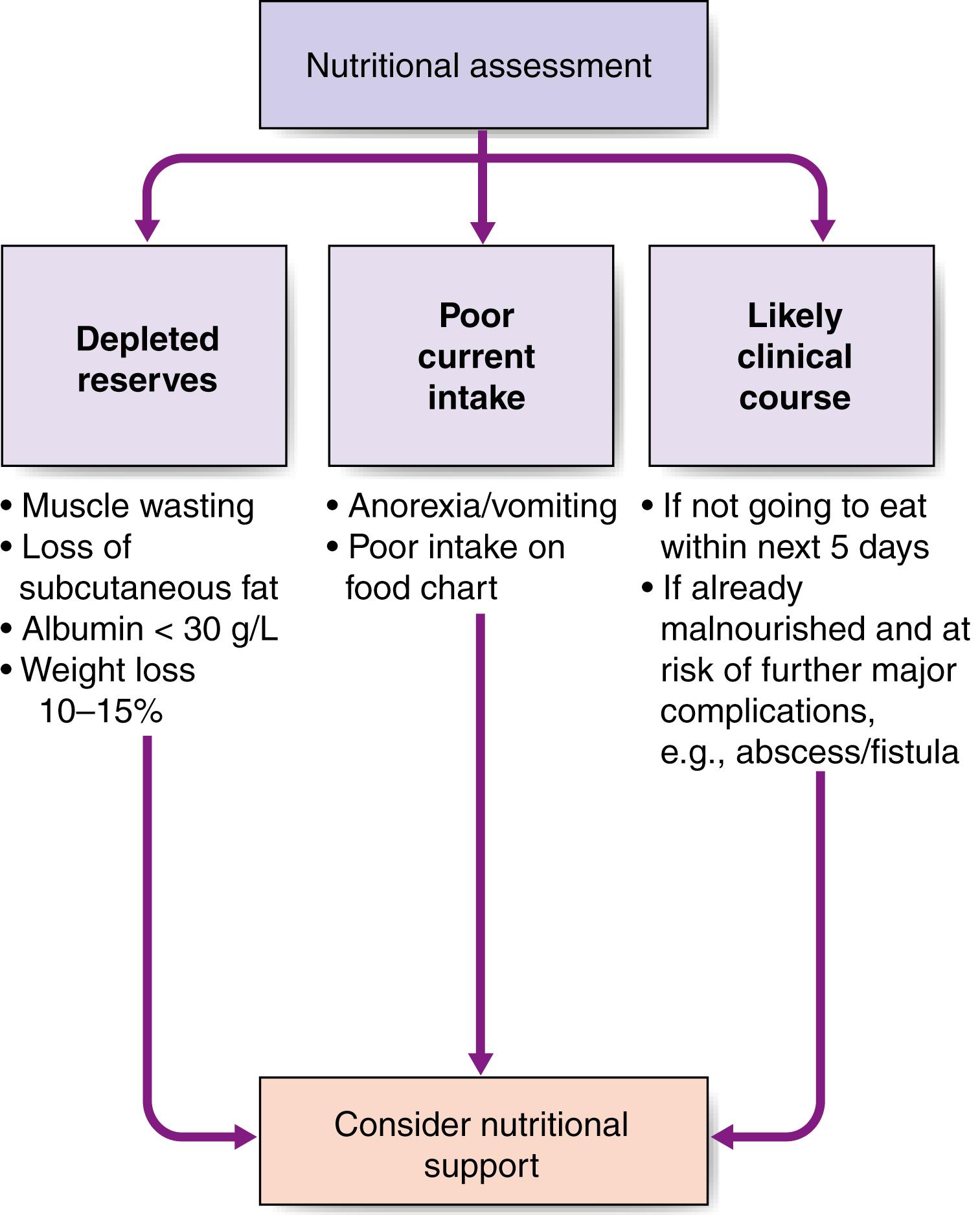

Simple screening tools used on admission can also identify patients at risk. The ‘Malnutrition Universal Screening Tool’ (‘MUST’) was developed by the British Association of Parenteral and Enteral Nutrition (BAPEN) to aid in the identification of malnutrition and should be routinely employed when any patient is admitted to hospital. The MUST tool uses a five-step process to derive a score based upon BMI, percentage unplanned weight loss and the ‘acute stress’ effect of disease, following which patients at ‘high risk’ of nutritional depletion require referral for dietetic support.
BMI = weight (kg) / height (m 2 )
| < 18.4 | Underweight for height |
| 18.5–24.9 | Ideal weight for height a |
| 25–29.9 | Over ideal weight for height |
| 30–39.9 | Obese (severe if BMI>35) |
| > 40 | Morbidly obese |
a There is a view that this should be 18.5–22.9 in South Asia.
Energy and protein/nitrogen requirements vary, depending on weight, body composition, clinical status, level of physical activity and dietary intake. For most patients, an approximation based on weight and clinical status is sufficient. Few adult patients require more than 25 to 30 kcal/kg/day (approximately 1800–2200 kcal in an adult of average body mass; Table 5.1 ). Additional calories are unlikely to be used effectively and may even constitute a metabolic stress, leading to excessive activation of the sympathetic nervous system (diet-induced thermogenesis), that may result in fever and difficulty weaning from mechanical ventilation as a consequence of the excessive carbon dioxide load. Particular caution should be exercised when ‘refeeding’ the chronically starved patient because of the dangers of potentially fatal cardiac dysrhythmias due to hypokalaemia and hypophosphataemia (refeeding syndrome – see below).
Nutritional status in surgical patients may be adversely affected by starvation (effects of disease such as oesophageal cancer, restricted intake), the effects of inflammation (increased catabolism) and the effects of the operation itself (stress/inflammatory response).
Nutritional status is assessed by current food intake, levels of reserves and likely clinical course.
| Uncomplicated | Complicated/stressed | |
|---|---|---|
| Energy (kcal/kg/day) | 25 | 30–35 |
| Protein (g/kg/day) a | 1.0 | 1.3–1.5 |
a Grams of protein can be converted to the equivalent amount of nitrogen by dividing by 6.25.
The most common method for assessing protein/nitrogen requirement is based on body weight ( Table 5.1 ). Although more accurate assessment for patients receiving nutritional support can be derived from measurement of 24-hour urinary urea excretion, which can be converted to an estimate of 24-hour urinary nitrogen loss, this is seldom necessary in routine clinical practice.
Enteral diets normally provide protein, whereas parenteral nutrition delivers nitrogen (N) in the form of amino acids. The nitrogen equivalent of protein is calculated by multiplying protein content (in grams) by a conversion factor of 6.25. In practice, nitrogen requirements are usually estimated based upon predicted calorie intake and the level of metabolic stress. Most patients require 1 g N per 200 kcal of energy daily (typically 10 g N) in the absence of catabolism, but nitrogen requirements increase significantly with the catabolic response to stress and may rise to as much as 18 to 20 g N/day in critically ill, burned or septic patients. Even if losses exceed this, more than 18 g N/day (equivalent to 112 g protein) is seldom given, because it is unlikely to be used effectively as protein is diverted to generate energy precursors associated with the stress response. It is thus currently impossible to prevent substantial loss of protein reserves and lean body mass in critically ill patients, and the aim of meeting requirements under these circumstances is primarily to limit the losses resulting from catabolism.
The ideal way for surgical patients to obtain sufficient nutrition is for them to eat or drink an adequate quantity of palatable and nutritious food. Unfortunately, socioeconomic factors may make this difficult. Hospital catering is not traditionally noted for its palatability. In addition, illness is usually associated with a change in eating behaviour, from taking three large daily meals to frequent consumption of smaller amounts of food, and this is extremely difficult to manage in a hospital environment. Eating is a social activity for most people, and lying in solitude in a hospital bed is not conducive to adequate food intake. Other reasons for poor food intake include the patient being too weak and anorexic, poor dentition, having a mechanical problem such as obstruction of the gastrointestinal tract and even the cumulative effects of repeated periods of fasting to undergo investigations. Patients may have difficulty ingesting sufficient food to meet increased metabolic demands. Some patients suffer from ‘intestinal failure’, that is, a state in which the amount of functioning gut is reduced below a level where enough food can be digested and absorbed for nourishment. Intestinal failure can be acute (when it is usually reversible) or chronic (when it is usually permanent). Acute intestinal failure is relatively common, especially after abdominal surgery, when it usually results from the development of surgical complications, whereas chronic intestinal failure is comparatively rare.
The principal causes of acute intestinal failure are mechanical intestinal obstruction and paralytic ileus, frequently associated with abdominal sepsis, as well as intestinal fistula formation, in which bowel content is lost externally or short-circuited (internal fistula) before it can be adequately digested and absorbed. Chronic intestinal failure may result from short bowel syndrome, following extensive small bowel resection, extensive small bowel disease, such as Crohn’s disease, and motility disorders, such as chronic intestinal pseudoobstruction. In some patients with short bowel syndrome, the remaining intestine may adapt over a period of months or years by a process of progressive dilatation and mucosal hyperplasia, allowing the patient to regain nutritional independence. Reconstructive surgery may also improve the function or even be employed to increase the functional length of remaining intestine in selected cases. Intestinal transplantation may be undertaken, especially in patients with loss of venous access preventing intravenous feeding, or in those who develop complications of chronic intestinal failure such as liver disease, for which transplantation may also be required.
Specialised nutritional treatment is required for intestinal failure if the patient is to remain adequately nourished. The provision of nutrition in such patients is further complicated by the metabolic consequences of ongoing inflammation or sepsis. As a general rule, this results in increased energy requirements and impaired ability to use administered nutrients, rendering nutritional support less effective. The overwhelming priority in providing effective nutritional support for such patients is therefore to eliminate sepsis.
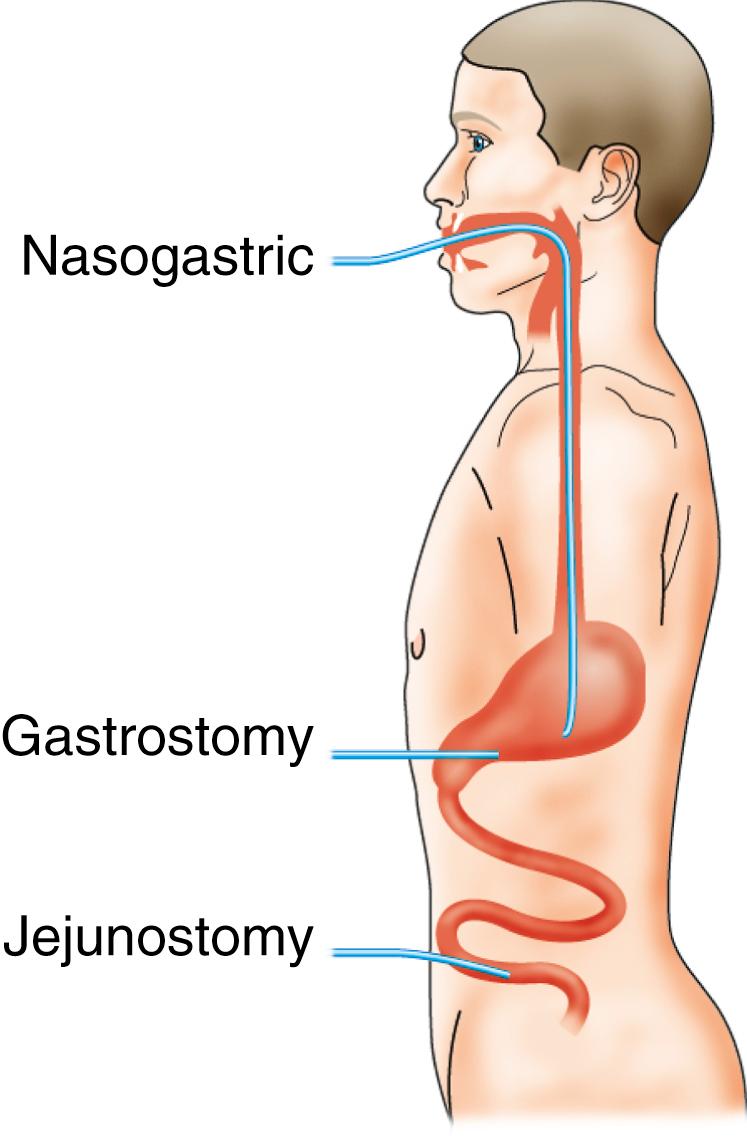
Nutrients can be given via the gastrointestinal tract (enteral nutrition) or intravenously (parenteral nutrition) ( Fig. 5.5 ). Parenteral nutrition is indicated only for patients in whom enteral feeding is not feasible. Enteral feeding is both safer and cheaper than parenteral nutrition ( EBM 5.1 ), which is particularly relevant in developing countries where the cost of parenteral nutrition is prohibitive and availability is limited. All patients with a normal length of functioning gastrointestinal tract, and even the majority with a reduced length, can be fed adequately by the enteral route. Furthermore, the ingestion of even suboptimal amounts of food may help maintain the gut function and may have beneficial metabolic, immunologic and psychological consequences. A flexible and pragmatic approach employing a combination of enteral and parenteral nutrition is therefore optimal, tailoring the route of nutrient provision to the patient’s ability to tolerate and benefit from it.
‘Enteral nutrition should be first choice for nutritional support in the critically ill surgical patient.’
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here