Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
There are a variable number of glands (2 to 10), but usually four are present, symmetrically arranged within the upper and lower poles of the bilateral thyroid gland. Approximately 5% of people have more than four glands. The glands are usually soft, pliable, and measure < 0.5 cm (5 mm), with a combined weight of approximately 120 to 140 mg. It is agreed that no single gland should be more than 60 mg, although the lower glands are often slightly larger than the upper glands. Embryologically, the upper pair arise from the fourth branchial pouch, whereas the lower pair arise from the third branchial pouch. Notably, glands can be found anywhere along the normal route of migration, resulting in glands embedded within the thymus, pericardium, esophagus, and mediastinum.
The parathyroid gland is composed of four cell types:
The chief cell (6 to 8 µm), a small polygonal cell with central round nucleus and abundant cytoplasmic secretory granules, is the basic functional cell involved in parathyroid hormone (parathormone, PTH) secretion.
The oxyphilic or oncocytic cell (12 µm) is slightly larger than chief cells, with abundant granular cytoplasm, ultrastructurally shown to be packed with mitochondria. These cells increase in number as the patient ages and tend to occur in nodules.
The water-clear cell (quite rare) has well-defined cell borders and abundant clear cytoplasm due to excessive glycogen.
Adipocytes, which also increase with age, compose up to 60% of the cellular mass. A general rule of thumb: 50% parenchymal fat at 50 years of age. There also tends to be more stromal fat at the poles of the gland(s).
The cellular components are arranged in cords, sheets, and pseudoglandular or pseudoacinar patterns. Age, sex, body fat, and other factors affect normal parathyroid gland cellularity.
In short, the principal function of the parathyroid glands is calcium homeostasis, briefly discussed here (spatial constraints limit this discussion). Calcium homeostasis is maintained within narrow limits (approximately 8 to 10 mg/dL) by the balance between variables increasing serum calcium (dietary intake, intestinal absorption, and bone resorption) and those decreasing serum calcium (bone accretion and urinary/fluid excretion). Nearly 99% of calcium is stored in the skeletal system, and osteoclastic resorption is most important. The three most important hormones are PTH, calcitonin (secreted by thyroid C cells), and vitamin D.
PTH is a polypeptide hormone (84 amino acid residues; the n-terminal is active, whereas the c-terminal is assayed) that increases blood calcium levels by stimulating osteoclastic resorption of bone and decreasing urinary excretion of calcium, while increasing renal phosphate excretion. This results in hypophosphatemia and can cause metabolic acidosis due to inhibition of HCO 3 . The half-life of PTH is 20 to 30 minutes. Abnormal or decreased function of the parathyroid glands results in disorders of calcium metabolism, characterized by too little (e.g., osteomalacia, pseudo hypoparathyroidism) or too much (e.g., milk-alkali syndrome, calcinosis) calcium. PTH also stimulates production of 1,25 dihydroxy-cholecalciferol (calcitriol), the hormonally active form of vitamin D, which increases the uptake of calcium from the gut, decreases urinary excretion of calcium, and increases the release of calcium from bone.
Parathyroid non-neoplastic disorders include aplasia, cysts, parathyroiditis, and hyperplasia. The discussion will be limited to hyperplasia.
Hyperparathyroidism is a state of elevated serum PTH as a result of excessive secretion. It is separated into primary, secondary, and tertiary, with primary hyperparathyroidism caused most commonly by adenoma (80%), hyperplasia (15%), and carcinoma (5%). The inappropriately increased PTH is due to an intrinsic abnormality in the gland(s) rather than a known stimulus for PTH secretion. Most hyperplasia results from secondary hyperparathyroidism, in which there is an increase in parathyroid parenchymal cell mass within multiple glands in response to a known clinical stimulus for increased secretion of PTH, such as chronic renal failure, malabsorption, and abnormalities of vitamin D metabolism. It is usually characterized by hypocalcemia and hyperphosphatemia. Similarly, tertiary hyperparathyroidism is generally characterized by four-gland hyperplasia in a state of autonomous hypersecretion.
Primary chief cell hyperplasia is a non-neoplastic absolute increase in the parenchymal cell mass within multiple parathyroid glands without a known clinical stimulus for increased PTH secretion. There is an annual incidence of approximately 4 per 100,000 persons per year in the United States.
Non-neoplastic absolute increase in parathyroid parenchymal cell mass within all parathyroid tissue without a known stimulus
Approximately 40/million population
Calcium metabolism abnormality
Cardiovascular disease may cause death
Females > males (2-3 : 1)
Adults, 40-60 years (mean, 5th decade)
Younger (25 years) in syndrome associated patients
20% may be familial
Asymptomatic, discovered during multichannel analyzer studies
If symptomatic: fatigue, lethargy, anorexia, weakness, vomiting, depression, polyuria, polydipsia, hypertension
“Bones, stones, abdominal moans” not seen often
Nephrolithiasis, nephrocalcinosis
Biochemical findings include elevated calcium, decreased inorganic phosphorus, and increased parathyroid hormone levels
Excellent, although recurrences occur (~15%)
Surgery with autotransplantation
Approximately 15% of all primary hyperparathyroidism is caused by primary chief cell hyperplasia. Women are affected much more commonly than men (2 to 3 : 1), with the overall incidence increasing with age (especially in postmenopausal women). The mean age at presentation is the 5th decade. Most patients present with sporadic disease (80%), although approximately 20% of patients have familial disease (most commonly multiple endocrine neoplasia [MEN] syndromes). Patients tend to present much younger (mid-twenties) when syndrome associated, without a sex predilection. MEN1 (Wermer syndrome) is the most common syndrome, with nearly 90% of patients having parathyroid hyperplasia. The MEN1 gene is localized on chromosome 11q13. Up to approximately 30% of patients with MEN2A (Sipple syndrome) have parathyroid proliferative disease but it is very rare in MEN2B. Patients may also present as part of autosomal dominant familial isolated hyperparathyroidism, calcium-sensing receptor (CASR) mutation, familial hypocalciuric hypercalcemia, neonatal severe primary hyperparathyroidism, and hyperparathyroidism–jaw tumor (HPT-JT) syndrome.
A fair number of patients are asymptomatic, the disorder discovered incidentally during routine multiphasic screening for other reasons. The presentation is often varied but vague, including fatigue, lethargy, anorexia, weakness, nausea, vomiting, constipation, polyuria, polydipsia, hypertension, arthralgias, and psychiatric symptoms. The classic triad of “bones, stones, and abdominal moans” is rarely seen in modern care but refers to osteitis fibrosa cystica (brown tumor of bone), kidney stones, and peptic ulcer disease, respectively. Work-up often reveals nephrocalcinosis, nephrolithiasis, and metastatic calcifications. Psychiatric or mental changes are frequent and include emotional instability, depression, psychosis, and confusion. Symptoms relate specifically to the degree (serum level) and duration of calcium elevation. Rarely, patients present with a neck mass.
Biochemical findings include an elevation of serum ionized calcium levels, with corresponding decrease in serum inorganic phosphorus concentrations and associated high serum alkaline phosphatase. Serum PTH levels, using the intact hormone assay, will usually be increased above the normal range of 10-65 pg/mL. In addition, there will be a high urine calcium, cAMP, hydroxproline, and phosphate.
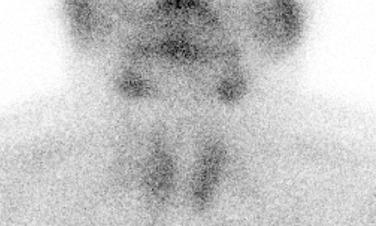
99m Tc sestamibi imaging is the preferred technique for detecting the topographic location of parathyroid tissue ( Fig. 26.1 ), although limited in detecting hyperplasia, with greater clinical utility for detecting adenoma or carcinoma. In general, radiographic studies are primarily used in the setting of recurrent disease after failed surgery. Single-photon emission computed tomography (SPECT) with the use of 99m Tc sestamibi as the radiotracer, especially when combined with computed tomography, can help with preoperative localization. Unusual locations of (ectopic) parathyroid tissue may be identified with magnetic resonance imaging using gadolinium and fat-suppression techniques. However, hyperplasia does not enhance as much with gadolinium as does an adenoma. Ultrasound (US), when used by an experienced investigator, can be a good alternative if scintigraphic studies are inconclusive. Furthermore, US may be used to guide fine needle aspiration (FNA) or guide retrograde venous sampling to determine PTH levels.
All glands affected, although not equally (two glands sampled for accurate diagnosis)
Diffuse or nodular enlargement
Soft, tan-brown glands
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here