Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The conditions presented in this chapter represent a wide spectrum of physiologic and pathologic endometrial changes. Their proper identification is important to exclude malignancy and provide correlation with the clinical context. A list of simplified evidence and consensus-based terminology for the most important benign endometrial pathology findings is shown in Table 9.1 .
|
|
a As described by Parra-Herran et al; see complete reference including explanatory notes in Can J Pathol . 2018;10(1):13–24.
The endometrium responds to the sex hormonal environment displaying consistent architectural and cytologic patterns according to the phase of the menstrual cycle. Examination of the normal endometrium and its progression in the cycle is important for two reasons: (1) it establishes a background for the evaluation of abnormal changes, either metaplastic or neoplastic; and (2) in reproductive-age women, it can help document ovulation and normal progression of the luteal phase. Nonetheless, precise determination of the date suffers from significant interobserver variation, and nowadays is variably requested by the gynecologist depending on practice site. The following paragraphs describe the morphology of “classic” normal endometrium as a framework for the evaluation of women in active (reproductive-age), gestational, and inactive states.
Cycling endometrium is further subdivided into six phases reflecting the progression over a prototypical (although not fully realistic) 28-day cycle that starts with the first day of menstruation. Variations from the standard 28-day cycle, which are common, are mostly due to variations in the length of the preovulatory (proliferative) phase. In contrast, the postovulatory (secretory) phase is remarkably constant at 14 days of duration. It is important to note that in endometrial biopsy and curetting materials, variable amounts of lower uterine segment and endometrium basalis will be present. They frequently retain an inactive appearance or do not respond to the hormonal environment as consistently as the endometrium functionalis; therefore, they should be excluded from date estimation.
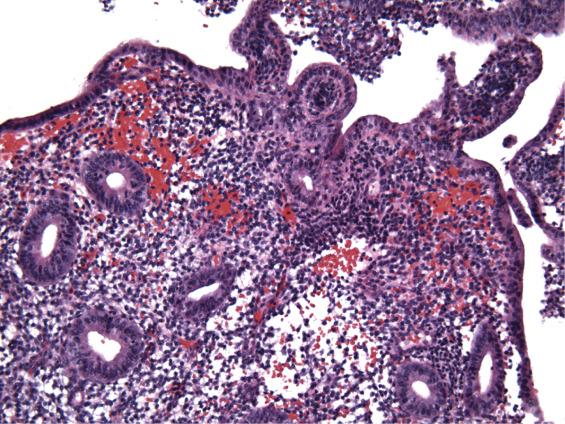
During the first few days after menstruation, rising levels of estrogen cause vasoconstriction, cessation of bleeding, and reepithelization of the endometrium basalis. Basal glands are uniformly round or tubular and show minimal mitotic activity and nuclear stratification. Detachment of the surface epithelium also occurs. Patchy residual stromal breakdown is seen in the form of dense, discrete aggregates of stroma admixed with blood and inflammatory cells ( Fig. 9.1 ).
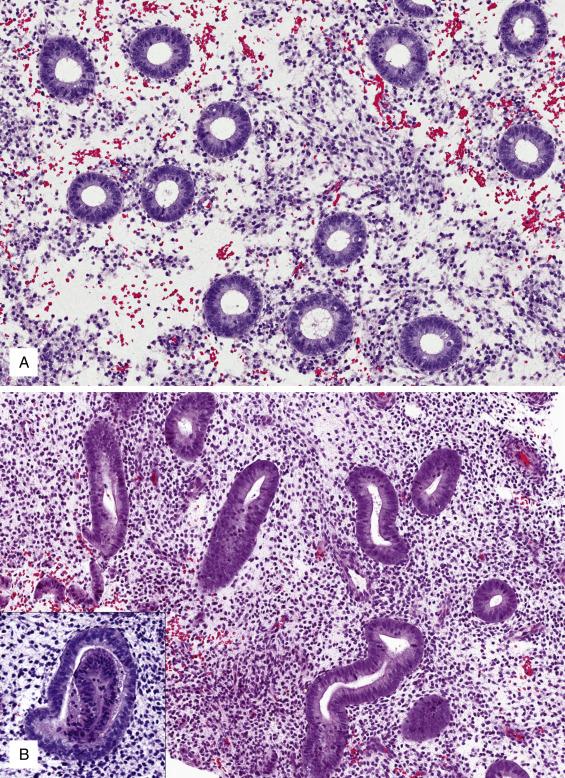
As levels of circulating estrogen increase, endometrial glands and stroma undergo active proliferation ( Fig. 9.2A ). The glandular compartment is evenly distributed and is composed of uniformly sized round to tubular glands. They are lined by columnar epithelium with smooth apical borders and pseudostratified, round to elongated hyperchromatic nuclei. Mitoses are readily evident. A common artifact encountered in this phase is gland telescoping (one glandular segment incorporated into another gland’s lumen, Fig. 9.2B , insert). The stroma is also mitotically active; stromal cells are uniform and round or spindled, and its appearance ranges from densely cellular to mildly edematous. In the latter scenario, the amount of intercellular space may mimic predecidual change; however, the cells have much less cytoplasm than predecidual cells, and the presence of glandular and stromal mitoses and the lack of prominent spiral arterioles are useful features arguing against this possibility. For practical purposes, subdividing the proliferative phase has not been shown to have diagnostic or management utility as it is poorly reproducible.
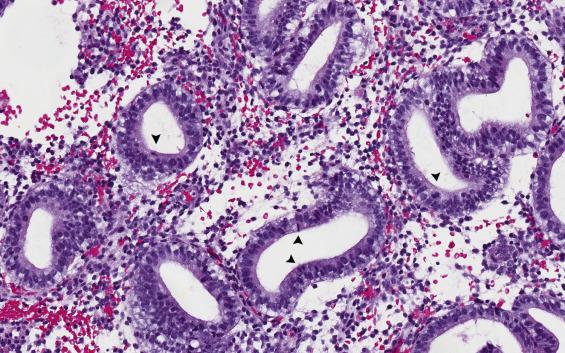
In the prototypical 28-day cycle, day 16 marks the point when ovulation occurs. Periovulatory endometrium is characterized by (1) gland tortuosity suggesting secretory endometrium at low power magnification, (2) focal and discontinuous subnuclear vacuolization involving less than 50% of the cells in each gland, and (3) readily identifiable mitotic figures ( Fig. 9.3 ).
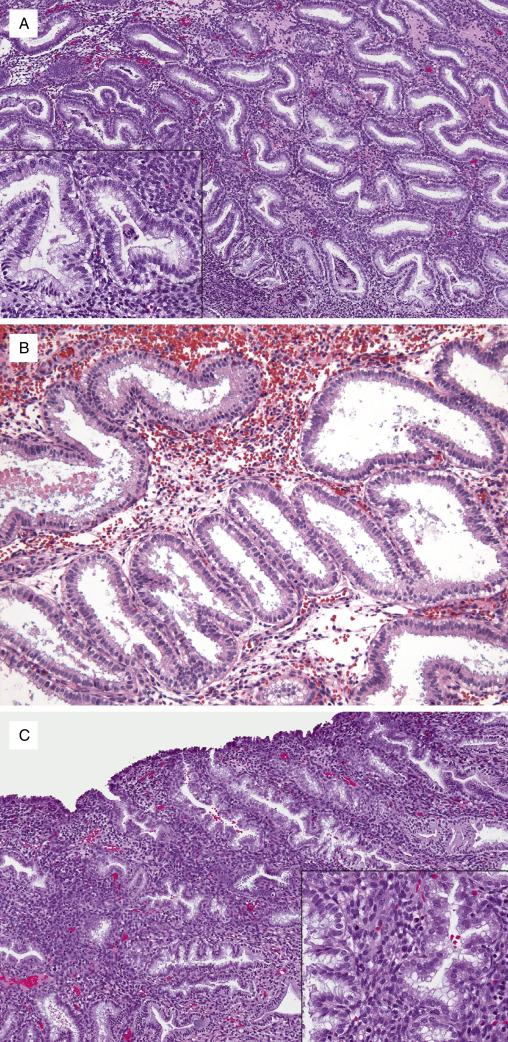
The first morphologic correlates of the secretory phase are the presence of uniform and diffuse subnuclear vacuolization (seen in more than 50% of the cells) and a significant decrease in proliferative activity. These changes are diagnostic of day 17 in the cycle and confirm that ovulation has occurred ( Fig. 9.4A ). Vacuoles progressively migrate from the subnuclear to supranuclear portions of the cytoplasm: a mixture of cells with subnuclear and supranuclear vacuoles is seen in day 18 with the nuclei mostly occupying the midpoint of the cytoplasm ( Fig. 9.4B ), and a predominance of supranuclear vacuoles is characteristic of day 19 ( Fig. 9.4C ). By day 19, nuclei are once again located at the base of the cell and intraluminal secretions start to appear.
During the early secretory phase, endometrial glands progressively increase in size and tortuosity; as the glandular compartment grows, its relative proportion of surface area increases (exceeding the gland to stroma ratio of 1:1 seen in the proliferative phase). The vertical parallel arrangement of the glands, perpendicular to the endometrial surface, also becomes more evident. As the amount of cytoplasm in glandular cells increases, nuclei become more uniform and evenly spaced, displaying round to ovoid shape and a fine chromatin pattern. Surrounding stroma remains dense to mildly edematous.
Supranuclear vacuole accumulation seen in day 19 is followed by extrusion of secretory contents into the glandular lumen. Indeed, the amount of intraluminal secretions peaks at day 20 of the cycle ( Fig. 9.5A ), and the amount of intracellular secretions starts to decrease. At day 20 the stroma is still largely unchanged, but in subsequent days becomes increasingly edematous. Stromal edema is focal by day 21 ( Fig. 9.5B ) and more prominent by day 22 ( Fig. 9.5C ). Stromal nuclei become more dispersed which may simulate predecidual change; however, cytoplasm is scant (“naked nuclei” appearance), the nuclei are usually small and the chromatin condensed, and cell dispersion is at the expense of extracellular fluid accumulation. In day 22 the spiral arteries become more prominent and are surrounded by stromal cells with a “cuffing” pattern ( Fig. 9.5C ).
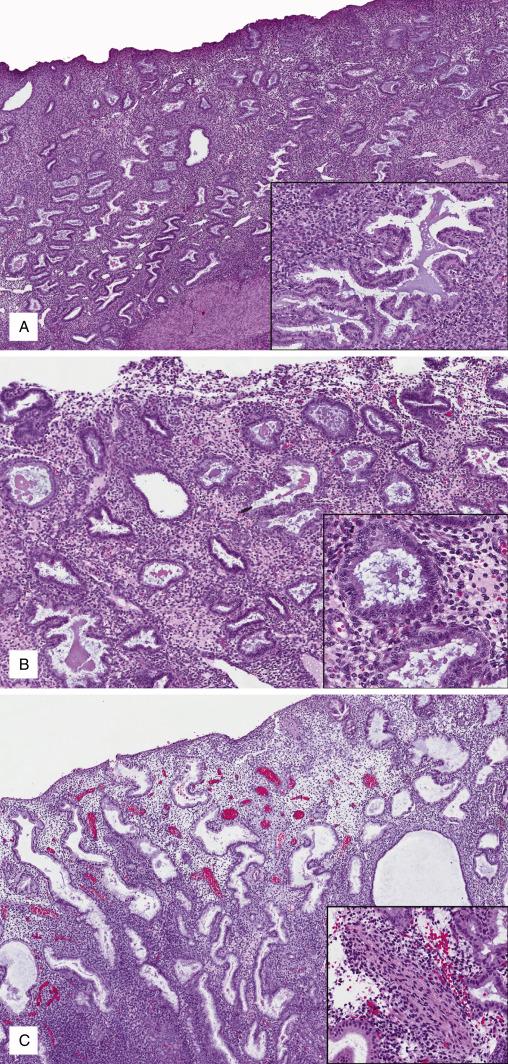
The hallmark of the late secretory phase is the appearance of stromal cytologic changes under the effect of rising circulating progesterone, which are described as “precidual.” The term “predecidua” is used since changes are akin to those seen in the endometrial stroma during gestation (decidua). Predecidualized stromal cells have a polygonal shape and distinct borders; cytoplasm is abundant and basophilic; nuclei are round with evenly dispersed, fine chromatin pattern. In day 23, predecidual change is limited to stromal cuffs surrounding spiral arterioles ( Fig. 9.6A ). By day 24, predecidua bridges multiple vessels and glands in the functionalis ( Fig. 9.6B ). On Day 25 thin aggregates of predecidua appear below the endometrial surface ( Fig. 9.6C ) which develop into a thick band of by day 26 ( Fig. 9.6D ). At this point, occasional granulocytes start to infiltrate the endometrium. Day 27 endometrium shows diffuse predecidual change expanding downward from the endometrial surface, associated with increasing numbers of granulocytes ( Fig. 9.6E ). During the late secretory phase, endometrial glands become progressively exhausted and display tortuous shapes, ruffled luminal borders (“saw tooth” pattern), and variable amounts of luminal secretions.
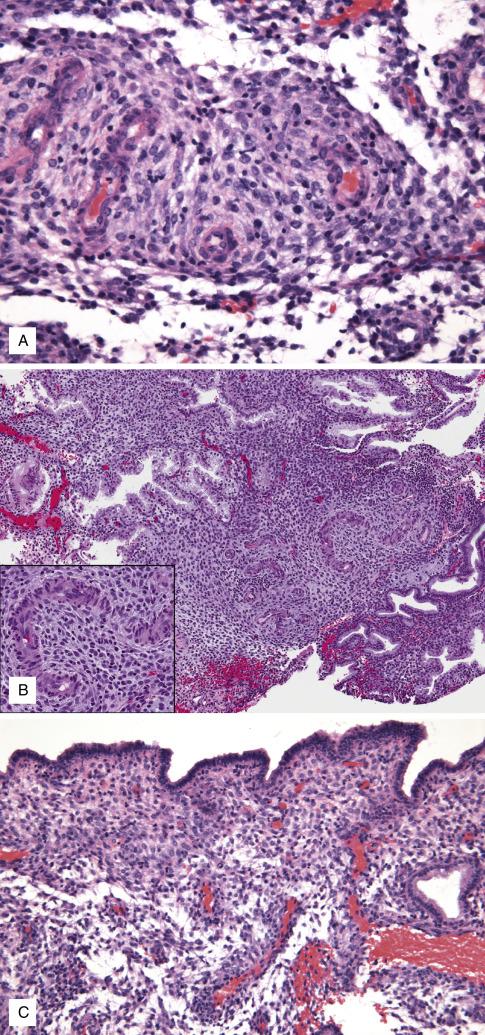
Menstruation ensues when progesterone levels decrease, causing collapse of the predecidualized secretory endometrium by apoptosis. The earliest feature of the menstrual phase is the breakdown of the predecidualized stroma, which histologically appears as dense round aggregates of degenerated stromal cells admixed with inflammation, and blood ( Fig. 9.7A ). As the stromal breakdown progresses, it is followed by collapse of the functionalis. Collapsed glands vary in size and shape and are lined by cells with variable amounts of apical cytoplasm, ranging from abundant apical to minimal (secretory exhaustion) ( Fig. 9.7 ).
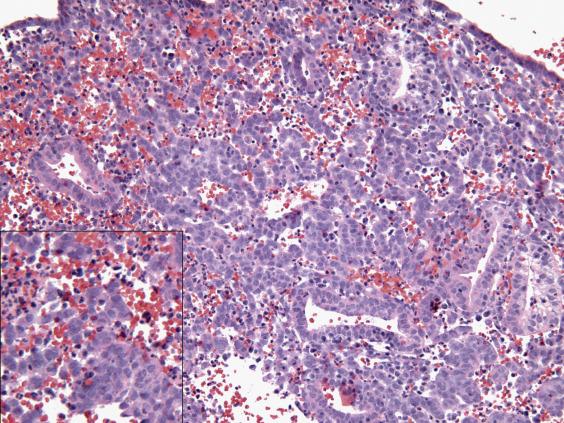
During pregnancy, the ongoing progesterone production by the surviving corpus luteum (and subsequently, the placenta) maintains the diffuse stromal predecidual changes seen in the late secretory phase (referred to as “decidua,” see Fig. 9.8A ). Glandular secretory differentiation is not only maintained but becomes more prominent with pronounced gland distension, intraluminal secretions, and ruffled borders due to cytoplasmic vacuolization. In addition, changes described as “Arias -Stella reaction” become evident: hypersecretory glands lined by vacuolated cells with enlarged, hyperchromatic nuclei, some of which protrude into the lumen (“hobnail”) ( Fig. 9.8B ). Of note, although Arias–Stella changes are highly suggestive of a concurrent/recent pregnancy, they can also be seen with exogenous hormones and gestational trophoblastic disease.
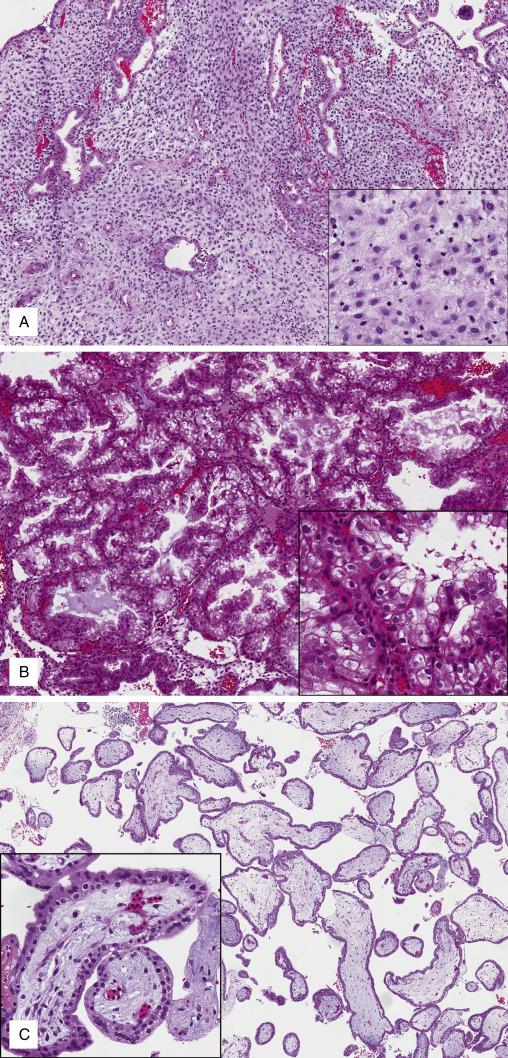
In routine practice, gestational-l type endometrium is mostly seen in the context of early pregnancy loss in which the examination of curetted tissues and confirmation of pregnancy is warranted. Endometrial sampling during the course of an early, unsuspected gestation is rare. In this setting, however, identification of gestational changes is critically important since it alerts one to the presence of an ongoing gestation (uterine or extrauterine). Once pregnancy is suspected, the pathologist should look for fetal and/or placental (chorionic villi, membranes, implantation site trophoblast) tissues in the sample: their presence will confirm an intrauterine gestation, whereas their absence will raise the possibility of an ectopic gestation and prompt clinical investigation.
Immature, first trimester chorionic villi have loose, edematous stroma with few visible capillaries. In normal gestations, the villous population shows only modest variation in size and has a uniform shape with smooth contours. The villous surface is lined by a double layer of trophoblast (inner layer of cytotrophoblast and outer layer of syncytiotrophoblast, Fig. 9.8C ). Fetal nucleated red blood cells are seen within villous capillaries between 4 and 12 weeks of gestational age. Frequently admixed with villi, fragments of the chorionic plate have a membranous appearance, variable edematous stroma, and a continuous layer of trophoblast (chorion). By comparison, free chorionic membranes are thinner, and the chorion is less prominent; the opposite site is lined by the amnion, which is usually inconspicuous. ( Fig. 9.8D ). Recent implantation site is characterized by the presence of intermediate trophoblast cells intermingled with decidua; both populations are admixed with fibrinoid material (Nitabuch fibrin, Fig. 9.8E ). The term “exaggerated placental site” is used to describe an exuberant implantation site intermediate trophoblast infiltrating myometrium; it contains multiple multinucleated cells but lacks proliferative activity ( Fig. 9.8 F ). Occasionally, placental site from remote pregnancies may be identified in endometrial samples, denominated “placental site nodule.” It appears as a nodular area of dense fibrosis with few entrapped decidualized stromal and quiescent trophoblast cells ( Fig. 9.8G ), described at length in Chapter 19 ).
Both intermediate trophoblast and decidual cells have similar size and polygonal shapes; however, they can be distinguished by the eosinophilic cytoplasm and hyperchromatic nucleus of intermediate trophoblast, and the more basophilic cytoplasm and pale round nucleus of the decidualized stroma. Implantation site trophoblast also contains large, lobated, or multinucleated cells with “smudgy” chromatin. Immunohistochemistry may help highlight the trophoblast, which is positive for cytokeratins, GATA-3, and human placental lactogen; decidua will be negative for these markers and positive for CD10.
The differential diagnosis of endometrium with gestational changes includes exogenous progestin therapy (discussed later in this chapter). In this situation, stroma is pseudodecidualized but the glandular hypersecretory changes are usually lacking. When florid, Arias–Stella reaction can mimic clear cell adenocarcinoma ; in the latter, however, cytologic atypia is diffuse rather than focal, and the tumor displays mitoses and high nuclear to cytoplasmic ratios. A potential pitfall to keep in mind is that both Arias–Stella reaction and clear cell carcinoma can be positive for Napsin A. Areas of fibrosis and scar in endometrial samples may simulate implantation site; clinical history (prior endometrial ablation, endometritis, Asherman syndrome), giant cell reaction, and absence of trophoblast will aid in the interpretation
Round to tubular glands with rare mitoses and subtle nuclear stratification
Residual patchy stromal breakdown and detachment of surface endometrium
Evenly spaced round to tubular glands with pseudostratified, elongated, and hyperchromatic nuclei
Surrounding stroma is dense to mildly edematous; cells are uniform, round to spindle
Gland to stroma ratio is <1:1 (glands represent less than 50% of surface area)
Abundant mitoses in glands and stroma
Mildly tortuous tubular glands with pseudostratified nuclei and scattered subnuclear (basal) vacuoles (in < 50% of cells in each gland)
Readily identifiable mitoses in glands and stroma
Key feature: Glandular vacuolization
Day 17: Diffuse subnuclear vacuolization in > 50% of cells in each gland (piano key pattern); mitoses are rare to absent
Day 18: Sub- and supranuclear vacuoles; subtle apical discharge, nuclei typically in midportion of cytoplasm
Day 19: Mostly supranuclear vacuoles and appearance of intraluminal secretions; basal nuclei; no mitotic activity
Key features: Glandular intraluminal secretion and stromal edema
Day 20: Peak in intraluminal secretions with increased gland tortuosity and uniform crowding; occasional subnuclear vacuoles
Day 21: Stroma displays focal edema; intraluminal secretions are still prominent
Day 22: Stroma displays diffuse edema with perivascular cuffing of naked nuclei (not predecidualized)
Key feature: Stromal predecidualization
Predecidual change: Visible basophilic cytoplasm with distinct cell borders; round nuclei with fine, even chromatin distribution
Day 23: Predecidual change limited to stroma around spiral arterioles (“cuffing”)
Day 24: Predecidual change bridging vascular elements and glands
Day 25: Thin patchy layer of predecidual change beneath surface
Day 26: Thick continuous layer of predecidual change beneath surface; rare neutrophils
Day 27: Diffuse predecidual change from surface to functionalis, numerous neutrophils
Collapsing stroma forming dense aggregates; viable areas with visible predecidual change can be seen
Exhausted secretory glands (irregular shape with collapsed lumens, mild stratification)
Prominent neutrophilic infiltrates
Endometrial tissue displays diffuse stromal decidualization (decidua) and prominent secretory glands.
Arias–Stella reaction: large hypersecretory glands with vacuolated cells containing hyperchromatic nuclei; hobnail pattern; no mitoses
Products of conception (in first-trimester samples)
Immature chorionic villi
Chorionic plate and membranes
Recent implantation site trophoblast (intermediate trophoblastic cells admixed with fibrinoid material at the interface with decidua), which includes exaggerated placental site (florid but nonconfluent implantation site trophoblast within myometrium)
Old placental site (placental site nodule)
Intrauterine vs. ectopic pregnancy
Exogenous progestin effect (vs. decidua)
Clear cell adenocarcinoma (vs. Arias-Stella reaction)
Scar from previous procedure/inflammation (vs. old implantation site)
( Fig. 9.8H ). Placental site nodule should not be confused with recent implantation site; the former will have larger amounts of collagenized stroma (usually nodular), and interspersed quiescent trophoblastic cells.
Inactive endometrium, not influenced by cyclic estrogen and progesterone levels, is usually thin and composed of evenly spaced simple glands lined by cuboidal to short columnar epithelium. Cells have basally oriented round nuclei with only subtle pseudostratification, homogeneous basophilia, and no mitotic activity . Surrounding stroma is dense and uniform ( Fig. 9.9A ). In addition to this morphology, endometrium after menopause undergoes additional “atrophic” changes which include: cystic distention of the glands with flattening of the epithelium, known as “cystic atrophy”; epithelial metaplasia (discussed later in this chapter); and collagen deposition in the stroma imparting a fibrotic and hypocellular appearance ( Fig. 9.9B ). Mitotic activity in the context of postmenopause is abnormal and may correlate with nonphysiologic sources of circulating estrogen (exogenous intake, hormone-producing neoplasms); thus, it should be reported as “proliferative endometrium” or “weakly proliferative endometrium” and correlated with the clinical history (e.g., a note stating “the findings may be secondary to endogenous or exogenous estrogen; clinical correlation is recommended”).
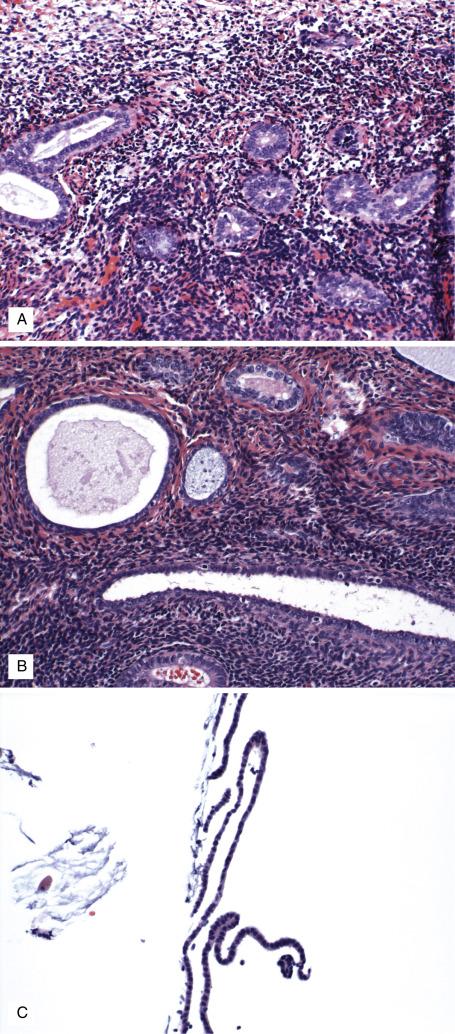
In biopsy and curettage specimens, inactive endometrial tissue is often scant and seen as thin strips of surface epithelium with minimal or absent stroma ( Fig. 9.9C ). In this context, assessing adequacy of the sample is paramount. In general, endometrial samples can be divided as inadequate (no endometrial tissue), adequate (intact endometrial tissue fragments with glands and stroma), and limited (detached strips of endometrial tissue, usually superficial). Adequacy of limited samples depends on the clinical scenario: it has been shown that samples with 10–30 strips of inactive endometrium are adequate in postmenopausal women, with a negative predictive value for endometrioid neoplasia of >99%. Limited samples with <10 strips in any patient, or with 10–30 strips in a patient who is premenopausal, has high body mass index, reports abnormal uterine bleeding or has thickened endometrium on imaging should be considered suboptimal and require consideration for repeated sampling.
Exogenous hormone administration in women is very common, with a wide variety of uses and types of preparations available. Oral contraceptives (OC) and hormone replacement therapy (HRT) are leading causes of abnormal bleeding, frequently occurring during the first 3 months of use. The pathologist should be familiar with the range of benign endometrial patterns secondary to hormone therapy. Progestin therapy produces classic changes in the endometrium, easily identifiable in most cases. Progesterone receptor modulators (PRM) also lead to a characteristic constellation of changes. Other agents induce less specific patterns, mostly irregular secretory endometrium and inactive endometrium . Since changes overlap, are frequently subtle, and vary over time, documentation of recent or ongoing therapy in the requisition form or medical records is extremely helpful in the interpretation.
Estrogen preparations are currently used to manage menopausal symptoms (for instance, topical control of vaginal dryness). In premenopausal women, unopposed exogenous estrogen will produce an abnormal proliferative pattern described as “anovulatory” or “disordered proliferative,” similar to instances of excess endogenous estrogen. In postmenopausal women, weak proliferative activity is seen in an otherwise inactive-appearing endometrium. Long-term exposure carries a risk for the development of hyperplasia, neoplasia, and adenocarcinoma.
Progestin-only preparations are used as contraceptive methods or to manage heavy menstrual bleeding and endometriosis. Progestin therapy does not affect ovulation; contraception is achieved by developing a thick cervical mucus barrier and an atrophic endometrium. Short-term preparations are usually oral and have low doses of progestin. Long-term preparations have high doses of progestin and include intramuscular injection (Depo-Provera), subcutaneous tubes (Norplant), and levonorgestrel-releasing intrauterine device (Mirena).
During the initial weeks of therapy, progestins induce secretory differentiation in both glands and stroma, highly resembling the physiologic secretory phase ( Fig. 9.10A ). With prolonged exposure, glands exhaust their secretions and become progressively small and atrophic, while the stroma remains diffusely pseudodecidualized (the so-called pill endometrium, Fig. 9.10B ). These long-term effects are attributable to the downregulation of hormone receptors in the endometrium by the persistent progestin levels. Stromal pseudodecidualization is particularly prominent in long-term methods such as the levonorgestrel-releasing intrauterine device ( Fig. 9.10C ). After months to years of continued therapy, stromal changes also fade and are followed by an inactive appearance akin to the postmenopausal endometrium.
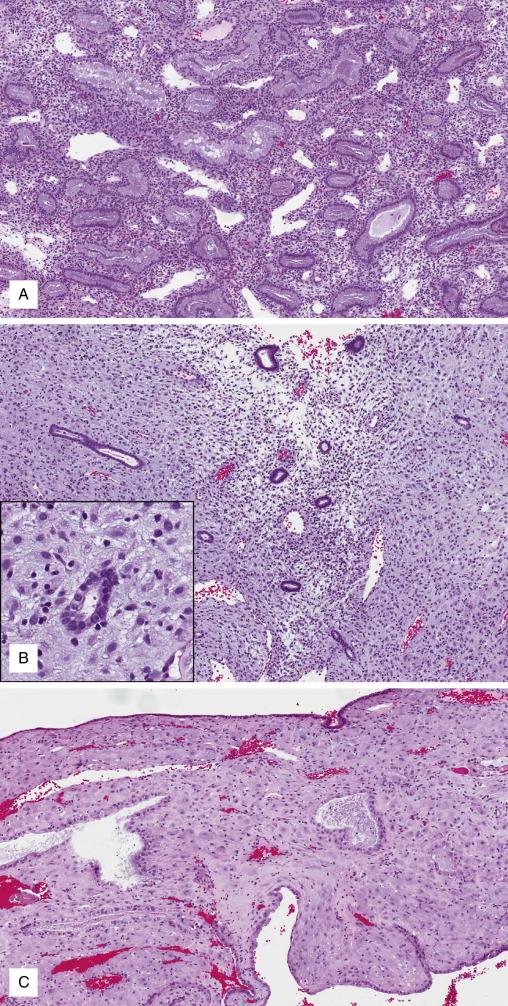
In addition to birth control, OC are used in the management of dysfunctional uterine bleeding and endometriosis. The most common forms of OC are cyclic combined estrogen/progestin and progestins only. Their use is preferred not only for their high contraceptive efficacy but also for their reduction in the risk of endometrial and ovarian carcinoma. With combined OC, the histologic appearance of the endometrium is variable depending on the number of cycles administered. During the first cycles, the endometrium has an “asynchronous” appearance: round and tubular glands reminiscent of proliferative phase but with vacuolization and absent to scant mitoses ( Fig. 9.11 ). With time, pseudodecidualization and more florid glandular secretory change develop. After numerous OC cycles, glands exhaust their secretions and become progressively small and atrophic, while the stroma remains diffusely pseudodecidualized (“pill endometrium”).
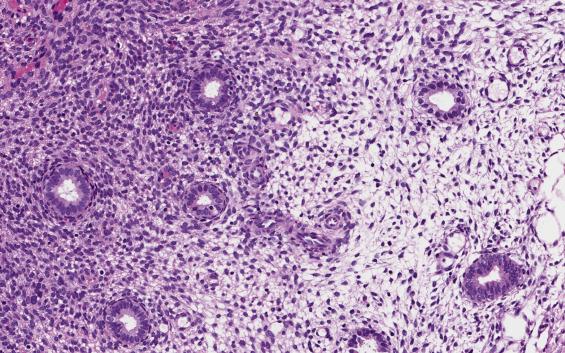
Current forms include estrogen and progesterone administration, either cyclic or combined. With cyclic HRT, the endometrial changes frequently resemble the physiologic cycle. During the estrogenic phase endometrium will show proliferative changes. During the progestin phase, however, patterns of secretory differentiation vary from the physiologic cycle, and include persisting vacuolization and patchy pseudodecidualization. An atrophic pattern is seen in a minority of patients. Combined HRT induces changes similar to combined OC (described earlier) or, more commonly, atrophy.
Estrogen receptor modulators have selective antagonist activity in reproductive organs. Given its antiestrogenic properties, tamoxifen is commonly used for treatment of hormone receptor-positive breast cancer. In the endometrium, tamoxifen is a competitive inhibitor of the estrogen receptor when levels of endogenous estrogen are high. However, when these are low (after menopause), tamoxifen has a weak agonist effect. Thus, while most women on tamoxifen have atrophic endometria, some develop changes associated with unopposed estrogen stimulation, ranging from disordered proliferative endometrium to adenocarcinoma (usually endometrioid). Endometrial polyps are also frequent. The risk of adverse effects increases with dosage and duration of therapy. Raloxifene, another type of selective modulator, does not elicit the estrogenic effects in the endometrium seen with tamoxifen. Endometrium under this agent looks atrophic; there is no reported risk for endometrial neoplasia.
Certain selective estrogen receptor modulators are used as inducers of ovulation. The most popular agent is clomiphene, which binds to the estrogen receptor with high avidity, competing with circulating estrogen. By blocking estrogen receptors in the hypothalamus, clomiphene interrupts the physiologic negative loop, promoting increase of gonadotropin-releasing hormone (GnRH) and subsequently follicle-stimulating hormone (FSH) and luteinizing hormone (LH) levels leading ultimately to ovulation. During the postovulatory period, levels of estrogen and progesterone are higher than in the physiologic state. The endometrium displays altered secretory differentiation with gland vacuolization that persists beyond the first three days of the secretory phase, decreased gland tortuosity and gland/stroma ratio, and less prominent stromal decidualization.
These agents are commonly used in the treatment of endometriosis and uterine leiomyomas. Currently used forms include ulipristal acetate, Asoprisnil, and mifepristone. The spectrum of morphologic findings is wide and is referred in the literature as Progesterone receptor modulator (PRM)-associated endometrial changes (PAEC) (progesterone receptor modulators). From the architectural standpoint, cystic dilation of the glands and gland tortuosity akin to the secretory phase are common. Glandular epithelium can be inactive or display occasional mitoses, apoptosis, and abortive subnuclear vacuolization ( Fig. 9.12A ). Stroma is variably cellular, contains ectatic capillaries, and lacks pseudodecidualization ( Figs. 9.12B and C ). Endometrial polyps can develop with ulipristal acetate. Importantly, endometrium returns to normal in most patients after months of treatment with this agent. Premalignant or malignant endometrial glandular lesions have not been reported as a side effect of PRM.
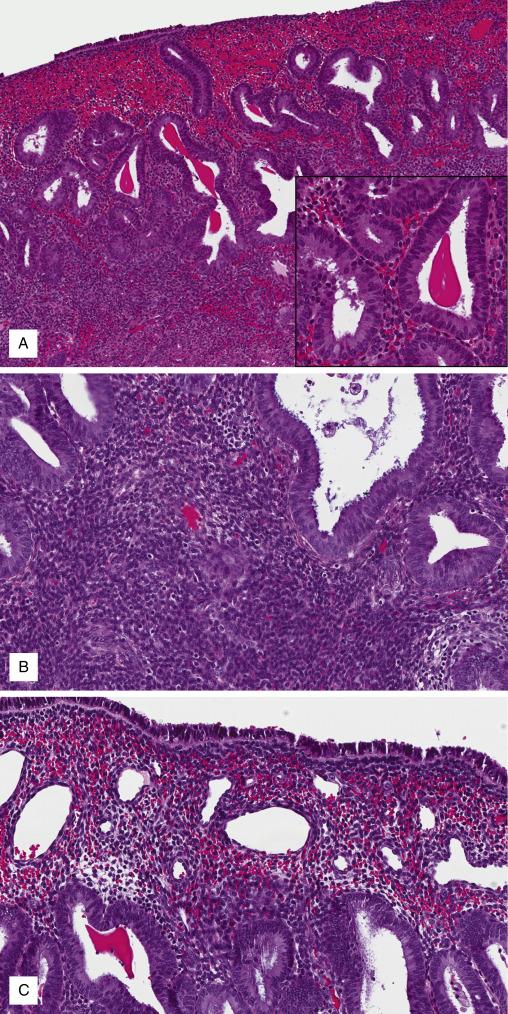
These agents, letrozole being the most common, inhibit the conversion of steroids to estradiol. They are used in the treatment of hormone receptor-positive breast cancer, endometriosis, leiomyomas, and infertility. Therapy results in markedly low levels of circulating estrogen, with consequent endometrial atrophy. Risk of endometrial glandular neoplasia is low.
These agents are used in the management of uterine leiomyomas and other mesenchymal neoplasms, endometriosis, and to suppress ovulation before oocyte harvest procedures. GnRH agonists produce steady high levels of the hormone, which in contrast to the physiologic pulsatile production, leads to an initial peak in FSH and LH secretion followed by suppression. This in turn manifests in reduced levels of estrogen and progesterone. The end-result is endometrial atrophy, comparable to the postmenopausal state. GnRH antagonists have similar effects without the initial FSH/LH peak.
Corticosteroid preparations have a weak progestational agonist effect. Danazol is a weak androgen agonist, which also has weak progestational effects. At high doses, endometrium in patients treated with these agents may display changes analogous to exogenous progestins. With prolonged exposure, endometrium becomes atrophic.
The entities discussed in this section are important causes of abnormal uterine bleeding. The diagnostic approach must take into consideration the patient’s age and the clinical presentation including colposcopy, hysteroscopy, and/or imaging findings. Importantly, hormonal contraception and HRT are the most common causes of uterine bleeding in premenopausal and postmenopausal women, respectively. This section discusses noniatrogenic pathologic conditions, which can be broadly defined as: 1) anatomic (endometrial polyp) which predominate in women during the fifth decade and older, or 2) functional (anovulation, other dysfunctional cycles), more common in reproductive age women (particularly third and fourth decades).
Proliferative changes; disordered proliferative patterns and risk of endometrial hyperplasia and neoplasia with prolonged exposure
During the initial weeks, glands and stroma display secretory differentiation (similar to the secretory phase)
With prolonged exposure, glands become small and inactive whereas stroma remains diffusely pseudodecidualized (“pill” endometrium)
After months/years of therapy, endometrium becomes atrophic (glands and stroma)
Combined OC: initial asynchronous patterns followed by secretory differentiation ultimately leading to “pill” endometrium morphology
Cyclic HRT produces proliferative and “delayed” secretory changes during estrogenic and progestational phases of the artificial cycle, respectively
Combined HRT produces atrophy or progestin-related changes
Tamoxifen induces atrophy or estrogenic changes due to its weak estrogen receptor agonist properties. Raloxifene induces atrophy
Ovulation induction therapy (clomiphene): altered secretory differentiation with persistent vacuolization, less gland tortuosity, and decreased predecidualization
Prominent glandular cystic dilation; gland shape and distribution resemble secretory architecture. Epithelium is inactive or displays only subtle proliferative or secretory changes. Stroma is variably cellular and lacks pseudodecidual change
Atrophic endometrium
Endometrial polyps are frequently encountered, accounting for 9%–24% of symptomatic patients undergoing endometrial sampling. The peak incidence is in the fifth decade; one-third of polyps occur in postmenopausal women. Hypertension, obesity, late menopause, and tamoxifen therapy are known risk factors. An association with endometriosis has also been documented. Lesions can be multiple (15%–26% of patients).
Transvaginal ultrasound and hysteroscopy have high sensitivity in the detection of polyps (89% and 96%, respectively). Both are valid methods, although hysteroscopy has superior specificity (75%, compared to 25% for ultrasound).
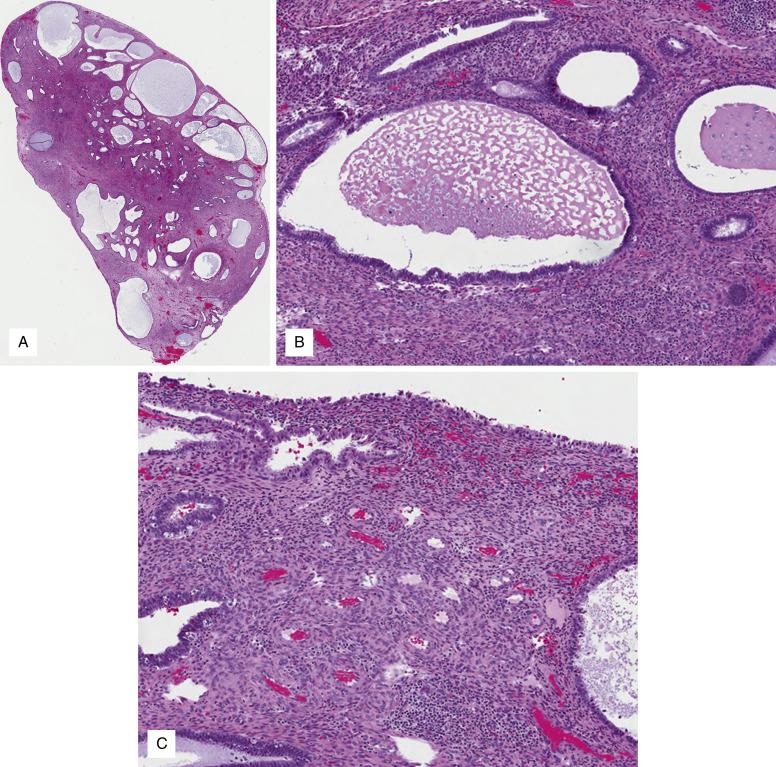
Polyps are easily identified in hysterectomy specimens as distinct exophytic growths in the uterine cavity with a narrow connecting stalk or a broad base (sessile lesions). Sometimes the lesion protrudes into the endocervical canal and through the cervical os clinically mimicking an endocervical polyp. Hysteroscopic resections commonly achieve resection of the polyp in a single fragment, which macroscopically appears as bulbous or elongated tan tissue with a base/stalk. The cut surface is firm and may be cystic.
Endometrial polyps are considered neoplastic proliferations of the endometrial stroma with an admixed nonneoplastic glandular component. In an intact polyp, histologic examination usually confirms the presence of surface epithelium surrounding the polyp and the presence of thick vessels in the stalk or base of the lesion. These features may be difficult to assess in blind biopsies and curettages, in which the lesion is fragmented. Additional features are helpful in this scenario, and the diagnosis of endometrial polyp can be rendered if two or three of the following criteria are met: 1) an irregular glandular component (variation in glandular size and shape with cystic dilation; Fig. 9.13A ), 2) altered stroma (fibrotic and collagenous; Fig. 9.13B ), and 3) thick-walled blood vessels (particularly if they are close to the surface epithelium; Fig. 9.13C ). In rare occasions in which the stroma contains significant smooth muscle differentiation, the term “adenomyomatous polyp” can be used.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here