Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
This chapter is dedicated to the memory of Graham Lister, who passed away a few months before this book went to press. Professor Lister was the original author of this chapter in the first edition in 1988 and continued as the single author through the fourth edition when I came on board as his coauthor. Dr. Lister’s original paragraphs on how to raise a flap continue to be, in my mind, the best written description of this exacting surgical technique. He was an extraordinary teacher and a mentor to hand surgeons worldwide. His clear thinking and ability to explain difficult concepts will be missed by all those he influenced.— W.C.P.
Acknowledgment: This section on skin grafts was originally a chapter written by Dr. Earl Browne, and much of his writing remains.
The skin is the largest organ of the body and serves a number of vital functions. It functions as a semipermeable membrane and a barrier to toxic material and microbes, but it also contributes to homeostasis through temperature regulation and sensibility. This latter function—perception of stimuli—is most important in the hand, especially on the palmar surfaces of the fingers.
All skin is composed of a thick layer of dermis covered by epidermis. The epidermis constitutes only about 5% of the thickness of the skin, and the thickness varies considerably depending on the area of the body. The skin on the palm and back is quite thick, whereas the skin of the inner arms and thighs is relatively thin. The quality of skin depends to a great extent on the epidermal appendages contained in the dermis, which also vary a great deal from area to area. This is especially true for the skin of the palmar and plantar areas, which contains no hair or sebaceous glands but is the almost exclusive domain of the Meissner and Vater-Pacini sensory end organs. It is reasonable to think of the epidermis as the barrier and the dermis as the functional portion of the skin.
One must also consider the hand as an organ and think of the skin as an envelope enclosing a multitude of tendons, nerves, vessels, bones, and joints. For the hand to function properly, this skin envelope must be elastic and nonadherent, contain as many of the appropriate appendages as possible, and be large enough to allow freedom of motion. The palm skin must also be thick enough to withstand the pressure and friction caused by grasp and pinch.
When considering the replacement of hand skin, as is done in skin grafting, one must keep these principles in mind. It is convenient to divide the hand into dorsal and volar surfaces, each having different basic requisites. Dorsal skin must be thinner, more elastic, and loose enough to not restrict flexion and must serve as a protective barrier to cover tendons and joints. Volar skin must be thicker and tougher while still being loose and elastic enough to allow motion, but above all it must retain its sensibility function. It is often possible to adequately replace dorsal skin by grafting, while this may not be possible with volar skin.
Figs. 44.1 and 44.2 depict the major differences between dorsal and palmar hand skin. Common to both is the irregularity of the border between the basal layer of the epidermis and the dermis, at the juncture of which are located the dermal papillae and the epidermal rete ridges. Both also contain intraepidermal nerve endings terminating in Merkel cell/neurite complexes and sweat ducts extending out from glands located in the base of the dermis and subcutaneous fat. A network of blood vessels and sensory and autonomic nerve fibers in the dermis is shared by all skin.
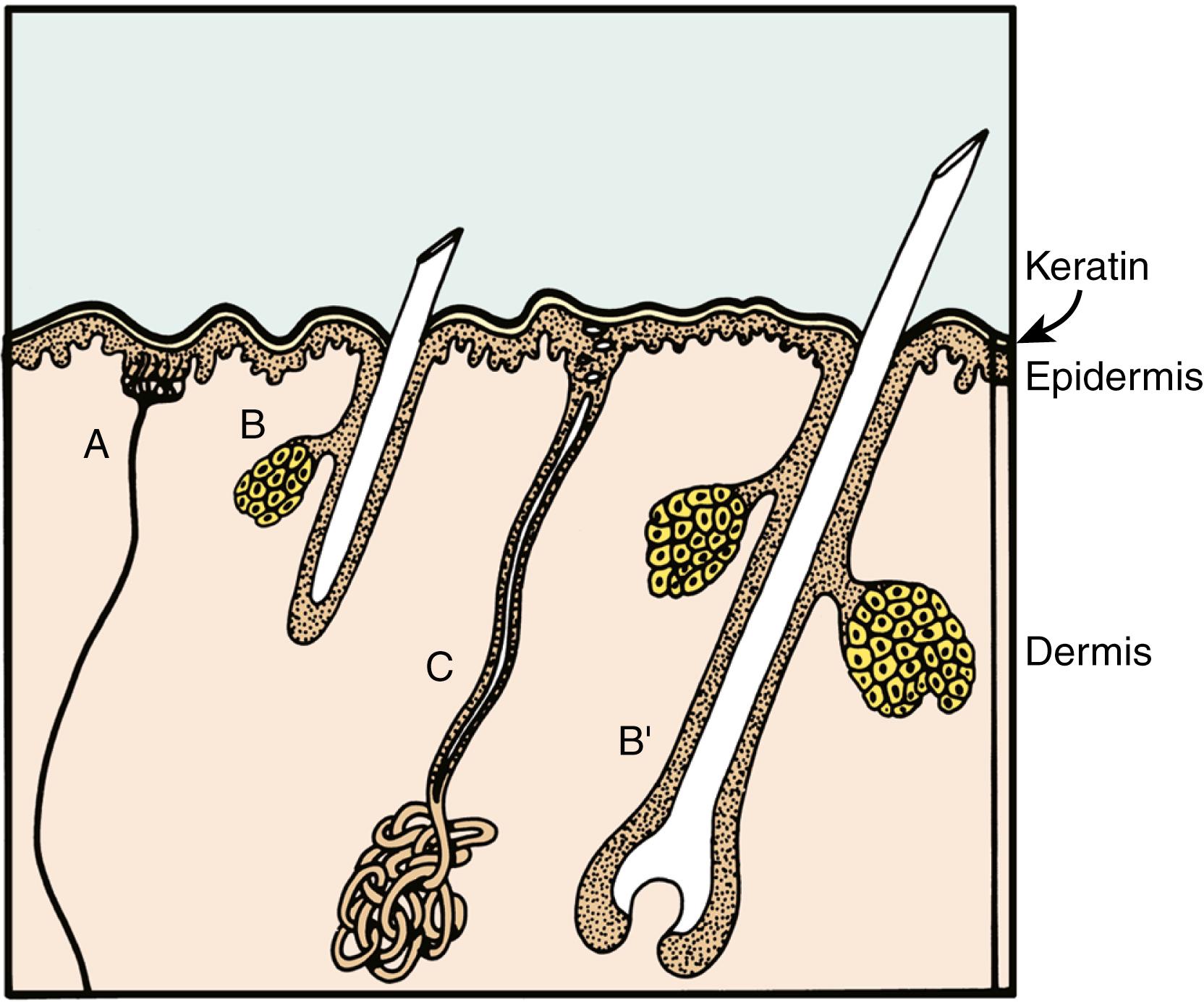
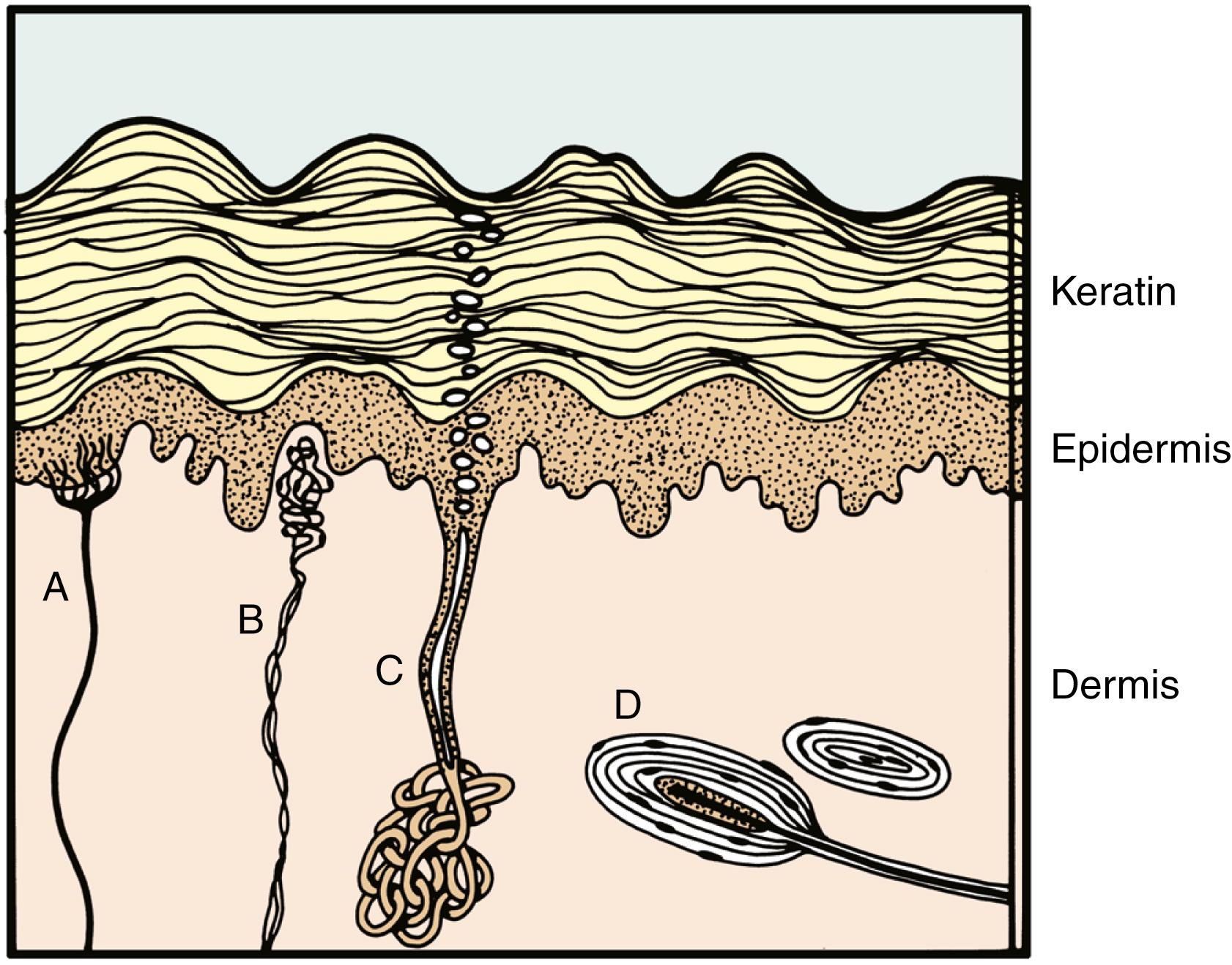
Fig. 44.1 depicts dorsal hand skin, which is similar to skin found anywhere else on the body. The basal layer of the epidermis is continuous with the hair sheaths and sebaceous glands. Hair follicles lie at different depths and are surrounded by a network of fine nerve endings.
Fig. 44.2 depicts the palmar hand skin. The papillae and ridges are deeper, and the keratin layer is considerably thicker than in dorsal hand skin. Most important, however, are the absence of pilosebaceous structures and the presence of specialized encapsulated nerve endings. Meissner corpuscles are present in dermal papillae, and Vater-Pacini corpuscles are present in the deep dermis. These latter structures are also found along nerve trunks, joints, and other areas but exist in the skin almost exclusively in digits and the genital area.
Because it is similar to the skin of most other areas, dorsal hand skin can usually be adequately replaced by a skin graft. No matter how thick the graft is, however, palmar skin cannot always be fully substituted by ordinary grafts because the special dermal neural mechanoreceptors will be absent. Only glabrous skin will provide these mechanoreceptors, and, unfortunately, not much of this type of skin is available for grafting.
When a split-thickness graft is removed, healing of the donor area occurs by epithelialization from hair follicles, and very little dermal regeneration occurs. The deeper the graft, the better the quality of the skin, but the less there is left behind at the donor site to aid in healing. Also, a deeper graft increases the chance of reinnervation of Merkel complexes and restoration of sensibility in the recipient site. Thick nonglabrous grafts will transfer hair follicles and result in unwanted hair growth if placed on palmar surfaces. In addition to poor return of sensibility, grafts on palmar areas do not provide the vascularized subcutaneous tissue necessary to cover tendons and nerves. In general, palmar grafts should be considered a compromise and are usually only indicated in release of flexion contractures in areas not requiring critical sensibility.
Any injury that results in the loss of a full-thickness segment of skin initiates the process of wound contraction. Although beneficial in helping to minimize defects in other parts of the body, this process of gradual shrinking of the wound edges can be disastrous in hand wounds ( Fig. 44.3 ). It appears that fibroblasts migrate into the base of the wound and differentiate into so-called myofibroblasts. These cells have the ability to contract, and the network of these cells and collagen fibers that forms in the base of the wound is responsible for pulling the wound edges together. It has been shown that contraction can be temporarily halted by treatment of the wound with smooth muscle relaxants. Once the contraction process has begun, its progression is not considerably altered by split-thickness skin grafting.

In a series of experiments by Stone and Madden, wounds in animals were treated with immediate grafting, delayed grafting, and immediate and delayed grafting with splinting. They concluded that immediate grafting with split-thickness skin did not significantly affect wound contraction. However, immediate grafting and splinting with a compressive dressing for 7 days did significantly inhibit wound contraction. On the other hand, delayed grafting had no effect on wound contraction no matter what adjunctive therapy was used. Splinting, although effective with immediate grafting, was ineffective in preventing contraction when used with delayed grafts.
It is generally believed that the condition of the wound is the main determinant in the result of skin grafting. Even full-thickness skin grafts can be found to contract if wound contraction has already begun. Rudolph has found that the application of split-thickness skin does not seem to have as much effect on the differentiation of fibroblasts into myofibroblasts as does a full-thickness graft. He has shown in animal experiments that wounds mature much more rapidly after the application of full-thickness grafts than after split-thickness grafts. The amount of actin and fibronectin found in rat wounds that were allowed to heal secondarily was compared in wounds with split-thickness grafts and those with full-thickness grafts. The thicker the graft, the less matrix protein present, which correlated well with the extent of wound contraction that occurred.
This is important in considering the source of a graft for palmar wounds. The Shriners Burn Institute has compared the results of split-thickness and full-thickness grafts for coverage of deep palmar burns. Normal range of motion was found in over half of the full-thickness group, and only one-fourth needed reconstructive procedures. In contrast, normal range of motion was present in only one-fourth of the split-thickness group, and two-thirds required reconstructive procedures.
A great deal of bacteria is normally present on the skin. It has been estimated that about 1000 organisms per gram of tissue are normally present in hair follicles, crevices, and recesses of the skin. This number of organisms does not seem to have a negative effect on wound healing. However, when contamination of the wound takes place, much higher levels are present. Surface bacteria, which can easily be removed, do not appear to be important in skin grafting if the wound is carefully prepared before surgery. On the other hand, penetration of the bed of the wound is very important. It has been established that 10,000 organisms per gram of tissue is a critical level of contamination. Wounds that contain fewer organisms than 10,000 organisms per gram of tissue do not commonly become infected. In a series of skin grafts applied to contaminated wounds, Krizek and colleagues have shown that regardless of the technique used to prepare the bed of the wound for grafting, a bacterial count of greater than 10,000 organisms resulted in a successful skin graft take of only 19%. If the count was less than 10,000 organisms, however, there was a 94% successful take.
After application of a skin graft to the wound, the graft first survives in a precarious fashion, apparently nourished by transudate from the wound. This has been referred to as “plasmatic circulation.” It is imperative that the graft become vascularized for ultimate survival, however, and anything that acts as a barrier to this process will cause loss of the graft. The most potent of all barriers is blood and a hematoma, which will result in a dead graft even in the absence of infection.
The actual process of vascularization is not altogether clear, but it is certain that this process must begin within a few days if the graft is to survive. It appears that the most important process is the ingrowth of capillary buds into the skin graft from the edges of the wound and, more importantly, from the bed of the wound. For this process to take place, there must be a vascularized bed containing the necessary fine network of capillaries from which budding will take place. Grafts will not take on denuded bone or tendon, and skin grafts are not satisfactory coverage for vital structures.
From the previous discussion, it is obvious that the ideal time for grafting the wound is as soon as it is clean enough and after the fine network of vessels has been established. Therefore wound contamination and the presence of a large milieu of myofibroblasts should be avoided. In general, immediate debridement and grafting are often possible, and no more than 2 or 3 days should be allowed to pass for the purpose of establishment of a good bed. The principle of allowing a healthy bed of granulation to develop is no longer considered correct, and it should be replaced with the concept of early grafting. The vacuum-assisted wound closure device may be helpful in promoting granulations in severe wounds and may aid in obtaining granulations over tendons and bone. The use of this device in the hand should be limited to avoid the marked scarring often caused by the device and increasing stiffness.
If it appears that the wound may be infected, determination of the bacterial content is necessary. Rather than rely on a wound swab to determine the presence of bacteria, which may yield a false-positive result, it is important to know whether the bed of the wound has become colonized with pathogenic bacteria. A useful method of determining this is by performing a wound biopsy. To do so, the surface of the wound is cleaned to prevent contamination, and a segment of tissue weighing about 1 g is removed. This can be done conveniently with a punch biopsy. A colony-count culture can be obtained from the biopsy material, or a more rapid determination can be made by crushing the tissue and examining it microscopically. It has been said that bacterial presence greater than 1 per high-power field roughly corresponds to a critical level of 10,000 organisms.
At times, the wound may not seem to be ideal to receive a graft because of infection, bleeding, or poor vascularity. It may be convenient to remove skin from the donor site at the time of surgery for wound preparation to avoid another round of anesthesia and operation. In this way, the skin can be stored while the wound is being prepared by dressing changes or periodic bedside debridement to reach an ideal state.
There are very convenient methods for storing such skin grafts after removal. One is simply to replace the skin on the donor site from which it was removed. The graft will begin to “take” and will remain well-nourished and viable. If this technique is used, it is necessary to remove the graft before considerable tensile strength has occurred, which usually occurs 5 or 6 days after harvest. Subsequent skin grafting can be done by injecting a solution of saline and local anesthetic between the graft and the donor site, and the graft can be placed on the recipient site at the bedside.
Another technique is to store the skin graft in tissue culture medium during this interval. A common practice is to wrap the skin graft in a saline-soaked sponge and store it in the refrigerator. This is effective for a few days, but seldom can the graft remain viable for more than a week. It has been shown that by storing the graft in tissue culture medium with various additives, the viability of the graft can be markedly prolonged. Hurst and associates have demonstrated that the use of McCoy 5-A medium containing amino acids and vitamins can allow skin grafts to be stored successfully for up to 30 days. The method of preparing the medium and storage of the grafts is described in detail in their article. This is an excellent method for 10 days to 2 weeks, but after that time the quality of the skin diminishes compared with a freshly harvested graft.
The two primary types of skin graft are split-thickness and full-thickness grafts. While other differences were discussed earlier, the main clinical difference in these grafts is in the method they are harvested. Split-thickness skin grafts are harvested with a knife or dermatome, and only the superficial layer of the skin is taken. The donor site of a split-thickness graft will heal on its own from propagation of epithelial cells from the deeper skin appendages (hair follicles and sweat glands). Full-thickness grafts are harvested with an incision with a knife and involve taking the entire layer of skin down to the subcutaneous fat. These donor sites must be closed as with an incision, otherwise the patient is left with a wound.
The standard thickness for harvesting split-thickness skin grafts is approximately 0.015-inch thick. Split-thickness skin grafts may be taken as either “thin” grafts (0.008 to 0.010 inch) or “thick” grafts (0.016 to 0.020 inch). Thicker split grafts have the theoretical advantage of potentially less contraction because they contain more dermis. The downside of these types of grafts is that the donor site may have difficulty healing, which can lead to worse scarring. One prospective study of the differences between standard and “thick” split grafts in the hand found no advantages in terms of range of motion, appearance, or patient satisfaction.
Although it is true that full-thickness grafts afford better protection, hold up better, establish better sensibility, contain more epidermal appendages, and contract less than split-thickness grafts, thick grafts are not always desirable. There are several disadvantages to thick grafts. Full-thickness grafts do not take as readily as split grafts. A full-thickness graft requires a much better recipient bed, and the graft is more prone to infection.
In addition, the availability of skin must be considered. It is not always possible to sacrifice enough skin to afford the luxury of full-thickness grafting, and split-thickness grafts must often be substituted. In general, full-thickness grafts should be used only in situations in which the quality of the skin and the tendency to less contracture are crucial. As a rule, these conditions are necessary only on palmar skin grafts, and the greater level of return of sensibility is an added bonus under these circumstances. Full-thickness grafts have recently been shown to be superior to split-thickness grafts in coverage of palmar wounds extending onto the digits in a large series of children with burns. Dorsal wounds can generally be treated adequately with intermediate split-thickness grafts.
There have been many attempts in recent years to develop skin substitutes. Autogenous epithelial cells have been cultured and placed on a variety of collagen matrices and used as skin grafts. These substitutes have been of great value in the treatment of massive burns, but the take of the graft is not equal to that of standard autografts, and the quality is not predictable. , Cultured epithelial autografts remain extremely expensive and afford very poor coverage, particularly in the hand.
Work continues on the goal of providing skin coverage without the need for a donor site, particularly in terms of reconstruction of the dermal layer. , Several products now exist that provide a collagen matrix to act as a layer of dermis when placed on a vascularized wound. , The primary problem with these dermal substitutes is that they generally have to be applied 1 or 2 weeks before coverage with a split-thickness graft to allow for revascularization of the collagen matrix, thus necessitating two operations. Some authors have noted no statistically significant long-term difference in elasticity, scar contracture, and patient satisfaction in patients treated with dermal substitutes plus split-thickness skin grafts versus split-thickness skin grafts alone in the management of donor sites from radial forearm free flaps. It should be noted that the time necessary for rehabilitation of the hand was significantly longer with the dermal substitutes, as the hand had to be immobilized while the dermal autograft became vascularized.
Recent reports have noted that dermal substitutes have few complications when utilized in the upper extremity and obviate the morbidity of a full-thickness skin graft donor site. One fairly large study found that placement of dermal substitute after contracture release was as effective as full-thickness grafting. Other reports of smaller series of patients have shown the utility of this approach to avoid the need for a full-thickness graft in burns and wounds of the upper extremity. , , Skin elasticity in patients managed with dermal substitutes and split-thickness skin grafts has been found to be close to normal in objective testing. The issue remains, however, that the application of dermal substitutes in the hand necessitates a period of immobilization while the dermis becomes revascularized, and then a potential second period of immobilization while the split graft heals. Coupled with the fact that the morbidity of most full-thickness skin graft donor sites is minimal, the utility of these materials in the hand remains in question in my mind.
When determining the thickness of a graft to be removed, the skin thickness of the donor area must be considered. Epidermis and dermis vary according to age and sex, as well as location. It is said that the epidermis ranges from 20 to 1400 μm in thickness and the dermis from 400 to 2500 μm. When considering donor sites, however, there is only a practical range of 25 to 80 μm of epidermis and 500 to 1800 μm of dermis. In relation to dermatome settings, these values translate to 0.001 to 0.003 inch of epidermis and 0.020 to 0.070 inch of dermis.
In general, the epidermis is quite thin in infants and reaches a maximum thickness at puberty; it then becomes thinner with age until it is almost as thin in old age as it was during childhood. There is very little difference in epidermal thickness between sexes. The dermis, however, remains relatively thin in youth; it reaches its maximum thickness at about the fourth decade and subsequently becomes thin again in old age. There is a marked difference between sexes, with male dermis being considerably thicker than female dermis.
The skin of the trunk and dorsal and lateral surfaces of the extremities is the thickest. Skin thickness may range from 0.020 to 0.025 inch in a small child to 0.100 inch on the anterior of the abdomen in an adult man. In general, most donor sites cannot be expected to be much thicker than about 0.060 inch in a man and about 0.040 inch in a woman.
Removal of a skin graft leaves some morbidity at the donor site. Although regeneration of the epidermis will occur, it appears that loss of the dermis is irretrievable. The thicker the skin graft, the poorer the quality of the donor site after healing. Therefore the thickness of the desired graft must, to a certain extent, determine the location of the donor site. The thicker portions of the body, such as the posterolateral aspects of the trunk and thighs, afford the best chance of good healing when a thick graft is desired. The thinner areas, such as the inner aspect of the thigh, are generally unsuitable for donor sites (unless they are absolutely necessary) because of the poor quality of healing of the donor site and the tendency of the skin graft to become hyperpigmented in its new area. So that morbidity and subsequent scarring are prevented, a reasonable guideline for graft thickness is offered in Table 44.1 .
| Patient | Thickness of Graft |
|---|---|
| Infants | Never over 0.008 inch |
| Prepubertal children | If >0.010 inch necessary, remove from lower abdomen or buttocks |
| Adult males | 0.015 inch from thighs, 0.018 inch from abdomen or buttocks |
| Adult females | Try never to use inner thigh; if >0.015 inch, use lower abdomen |
| Elderly adults | Treat like children’s skin |
The optimal thickness for split-thickness grafts is 0.015 inch in thickness. Thinner grafts of 0.010 to 0.012 inch may be best for wounds where graft survival is at risk. Split grafts greater than 0.018 inch are seldom indicated because of donor site morbidity. If thicker skin is desired, it is best to use a full-thickness graft.
If possible, a graft should be taken from an area accessible to care during the healing process. Patients despise lying or sitting on a donor site. Donor areas that cross intertriginous areas are very unsuitable because of the constant cracking and subsequent drainage with motion. In women and children, the desired donor site is from the lateral aspect of the upper thigh or buttocks or perhaps the lower portion of the abdomen. These donor sites can be covered by a bathing suit or some other article of clothing. It is important to remember that the thicker the skin graft, the larger the number of hair follicles that are transferred in the graft; thus whenever possible, a relatively hairless area should be selected as a donor site for the hand.
A very small piece of skin can be removed with a No. 10 knife blade by dissecting superficially just under the surface of the skin. It is convenient to perform an intradermal lidocaine injection and make a very shallow skin before beginning the tangential dissection. This technique allows proper selection of the appropriate depth and width of the graft. The blade is then placed into the depth of the incision, and with a back-and-forth sawing action, parallel to the skin, the desired size of graft can be removed. It is possible to remove a reasonably uniform piece of skin up to about the size of a dime with this technique, but the procedure is difficult for a surgeon without some experience.
A much more satisfactory method of taking a small skin graft is with the Weck-Goulian knife set. There are a number of depth gauges available that can be fitted over the blade ( Fig. 44.4 ). These gauges vary in thickness from 0.004 to 0.030 inch. By placing the gauge next to the skin, the appropriate depth and angle of cut are automatically established. Because this instrument tends to take a rather thick graft, I prefer to use the 0.010 guard, avoiding excessive thickness. A strip of skin 2 cm or so in width can easily be removed, and with practice, a longer strip can be harvested. This instrument has the advantage of removing a strip of skin with relatively uniform thickness.

Split-thickness skin graft harvest from the upper inner aspect of the forearm is enticing because it resides in the operative field. This donor selection is suboptimal as the forearm skin is relatively thin and heals with a hypertrophic scar. These scars are obvious to both patients and casual observers, and often the patient will complain more of the donor site scar than the recipient area scar. Donor harvest from an area of thicker skin that is better concealed is a better choice.
The Padgett dermatome is a motorized dermatome (via electricity or nitrogen turbine) and uses a disposable blade that cuts with a very rapid back-and-forth motion. There is a depth gauge on the side of this instrument to set the desired depth of the graft. Variable-sized plates determine the width of the graft taken ( Fig. 44.5 ). After determination of the desired width, the blade is fitted on the machine and the appropriate plate placed over the two screws and screwed down tightly. To put the blade on, it is necessary to first slip the base of the blade under the two projecting metal pieces and snap it down so that the hole in the blade fits onto the projection of the drive shaft. The appropriate graft thickness is selected by moving the blade up and down by using the depth gauge on the side of the machine. The depth of the graft taken is also determined by the pressure applied to the device on the skin and the angle of attack of the blade against the skin.

The skin is surgically prepped and cleansed with saline before taking the graft. The skin should be lubricated so that the dermatome will slide easily and not skip. Either mineral oil or a surgical soap solution can be used for this purpose and is placed both on the skin and on the flat surface of the instrument that is in contact with the skin. The dermatome is placed flat on the donor site, and traction is held behind the dermatome. It is not necessary to stretch the skin in the direction in which the dermatome will be advanced. It is easy to remove a piece of skin with this dermatome, and minimal pressure should be exerted on the donor area. A common mistake is to dig the dermatome edges into the skin by pressing too hard, which creates an improper cutting angle. As the dermatome is advanced, the skin may be lifted up to check the thickness. This skin lifting is unnecessary, however, as a uniform piece of skin is removed with the Padgett dermatome. The Padgett dermatome is probably the best instrument to use to harvest a skin graft, especially if one is not experienced using a knife.
A similar instrument has been produced by Zimmer and is used in the same fashion as the Padgett dermatome. A special disposable blade is made to fit the dermatome and snaps easily onto the reciprocating post. The width gauges are fitted in place and screwed down like a Padgett dermatome. The thickness of the graft is adjusted by a gauge on the side of the instrument similar to the Padgett dermatome, and the technique of skin harvest is identical. One must be careful in the setup of this instrument, however, because it has been reported (and I have personal experience) that this dermatome can cut down to muscle and cause serious injury if it has not been properly set up with the appropriate blade by the operating room personnel.
At times, the condition of the wound cannot be made optimal for grafting. The risk of infection or hematoma decreases chance of survival of a sheet of autograft. In these instances, it is prudent to use a piece of skin that has been “meshed” so that many perforations are present to allow blood or exudate to escape. An instrument has been devised that will automatically make these perforations. The skin can be expanded so that the interstices allow drainage and rapidly become epithelialized ( Fig. 44.6 ). This method of grafting has wide application in the treatment of burns and large wounds.
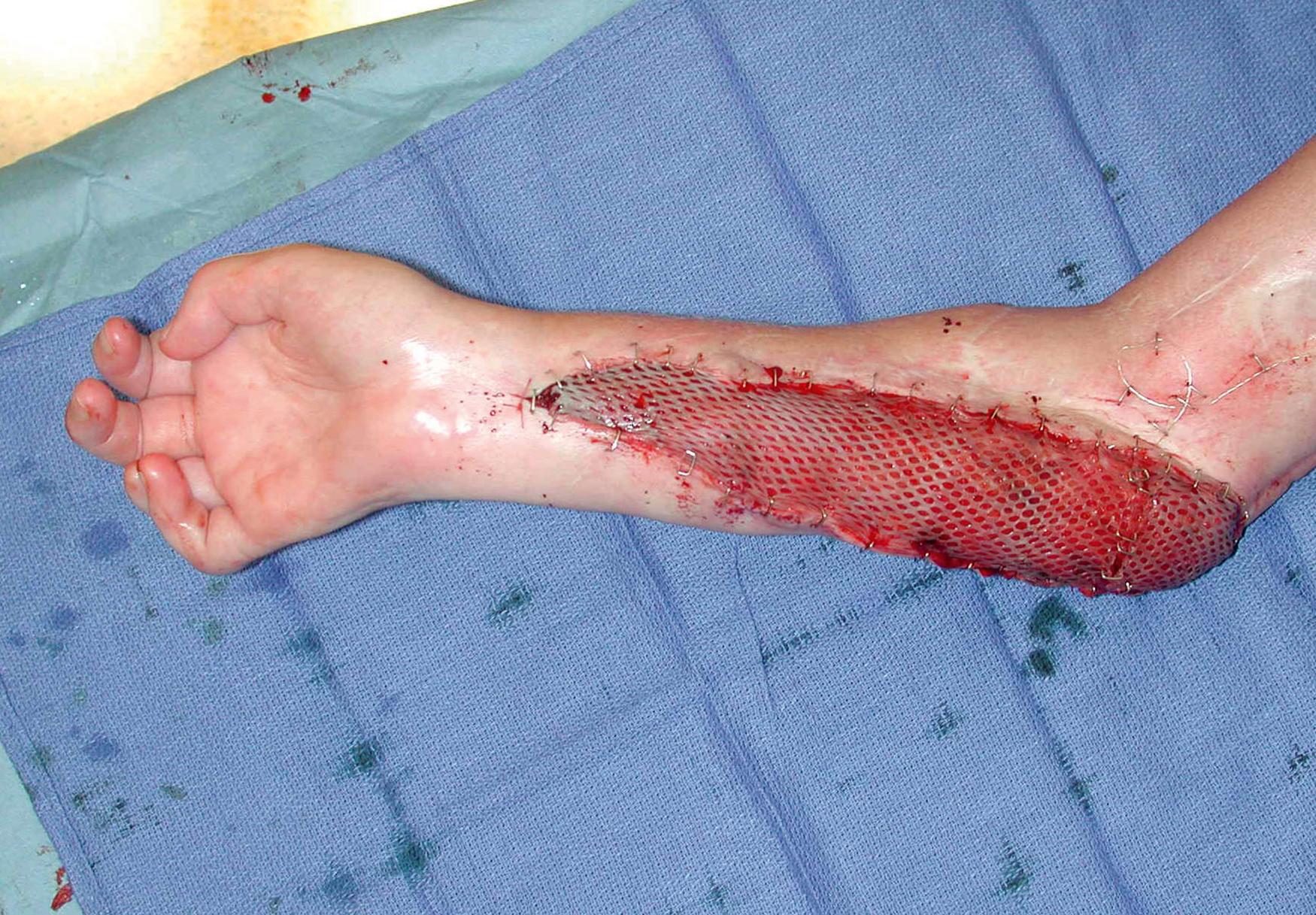
It must be remembered, however, that only the portion of the wound that has the skin directly applied is being grafted and the interstices of the mesh are left open and allowed to epithelialize. The quality of the graft will not be as good as that of a sheet of skin, and the tendency toward contraction and poor cosmetic results is greater. Meshed grafts placed over flexion creases are prone to joint contractures. These negative factors can be partially limited by compression. Therefore it is imperative to treat wounds that have undergone meshed grafting with long-term compression, such as can be obtained by a custom-fitted pressure glove.
Although they were not specifically evaluating hand grafts, el Hadidy and colleagues evaluated the results of meshed versus nonmeshed grafts in burns. As expected, the results of either graft following late excision were significantly worse than the results after early excision, but the meshed grafts contracted more in both groups. In addition, there was a difference in the amount of growth that occurred in the two groups. With early excision, nonmeshed grafts grew back to a size of 91% compared with 78.5% in the meshed group.
Simple wound with good underlying soft tissue bed
Marginal wound with plan for more complex reconstruction at a later time
Inspection of wound (for adequate vascularity); possibly, quantitative wound cultures
Avoid a graft that is too thick.
Small grafts may be taken with a freehand knife or Weck-Goulian dermatome.
Mesh or “pie-crust” graft to avoid fluid collection underneath
Recipient site should be very clean and debrided back to viable tissue everywhere.
Avoid air or fluid pockets underneath graft (mesh or pie-crust graft).
Graft must be secured in place to avoid moving against the wound bed.
Avoid removing the dressing for at least:
Three days if there is a question of wound cleanliness
Five to 7 days if the wound is very clean
Place a bolster for graft in a concave surface.
Early infection (usually Streptococcus or Pseudomonas ) that destroys the graft
Early motion that keeps the graft from adhering
Immobilize for at least 2 weeks.
Change the dressing if signs of drainage, and so on, are present.
Avoid any shearing force on the graft for a minimum of 3 weeks.
Skin graft will not match surrounding skin in terms of color and texture.
Split-thickness skin graft should afford stable coverage in most instances.
Donor site morbidity is minimal over the long term; however, donor site scarring can be conspicuous.
Meshed grafts are often placed on a wound without the graft “expanded,” which prevents collection of fluid under the graft but avoids the necessity for skin to grow over the open interstices. Whereas using a meshed graft in this way inevitably leads to more scarring than using a nonmeshed graft, the functional differences are usually minimal. The advantages of meshing a graft often outweigh the disadvantages. Nonetheless, split-thickness skin grafts are usually reserved for coverage of the dorsum of the hand, whether meshed or not. When the dorsum of the hand is covered with a muscular or fascial flap, it is certainly acceptable to place an expanded meshed split-thickness graft over the flap. It is sometimes convenient to use a mesh graft as a “temporary” biologic dressing until a later resurfacing procedure can be done as part of the formal reconstruction.
After harvest and preparation, the graft is spread out onto the surface of a plastic carrier, which is provided with the dermatome ( Fig. 44.7 ). The “dermacarrier” plastic board has angled troughs that dictate how much the graft will expand. The various boards are marked and allow expansion from 1.5:1 to 9:1, but sizes larger than 1.5:1 have very large holes that are not practical for hand surgery. Grafts thicker than 0.015 inch in thickness will not feed well into the meshing machine, and thus thicker grafts do not mesh well. After the graft has been spread onto the carrier, the construct is fed into the mesher and run through the machine by using the hand crank.

It is critical that the card be fed through the mesher with the grooved side up. If the skin is inadvertently placed on the wrong side of the mesh card and passed through the dermatome, the result will be multiple small strands of “linguini” that are unusable.
Full-thickness grafts transfer all the skin appendages and nerve endings except the sweat glands located in the subcutaneous tissue and some of the Vater-Pacini corpuscles of the palmar and plantar skin. This thickness is an advantage for the restoration of sensibility and quality of coverage. These grafts must always be taken from relatively hairless areas to minimize subsequent hair growth in the recipient area. Good donor sites for this type of skin are in the lower and lateral abdominal areas. These areas allow good-quality and relatively hairless skin. In addition, the area is loose enough to allow a large piece of skin harvest with no subsequent defect following closure. The widespread use of groin skin for grafting of the hand is only to be condemned. Skin from this area is always darker than the hand and carries a high potential to grow hair, which is extremely objectionable when placed on the fingers or palm of the hand. Smaller pieces of skin can be removed from the extremity undergoing surgery, which can be convenient as the area can be prepared within the operative field. The medial upper arm is a viable option for full-thickness grafts from the upper extremity and large pieces of skin may be harvested. If skin is harvested from the posteromedial aspect of the arm, the scar is usually not objectionable. The volar wrist crease and antecubital fossa are often described as good choices for full-thickness skin grafts, but these have the potential of an unsightly scar. Scars on the volar wrist crease can also be interpreted as a sign of a prior suicide attempt. For coverage of the palmar surface of the fingers, glabrous full-thickness grafts may be taken from the hypothenar eminence. A fairly large graft can be taken from this area with minimal scarring and morbidity. In instances that sensibility is critical, such as in the fingertips, a fairly large piece of “fingerprint” skin can be obtained from the hypothenar area ( Fig. 44.8 ). Small glabrous full-thickness grafts can also be taken from the lateral surface of the great toe. Glabrous full-thickness grafts have the added advantage of transferring skin of a more similar nature containing Meissner corpuscles, thereby potentially restoring better sensibility.
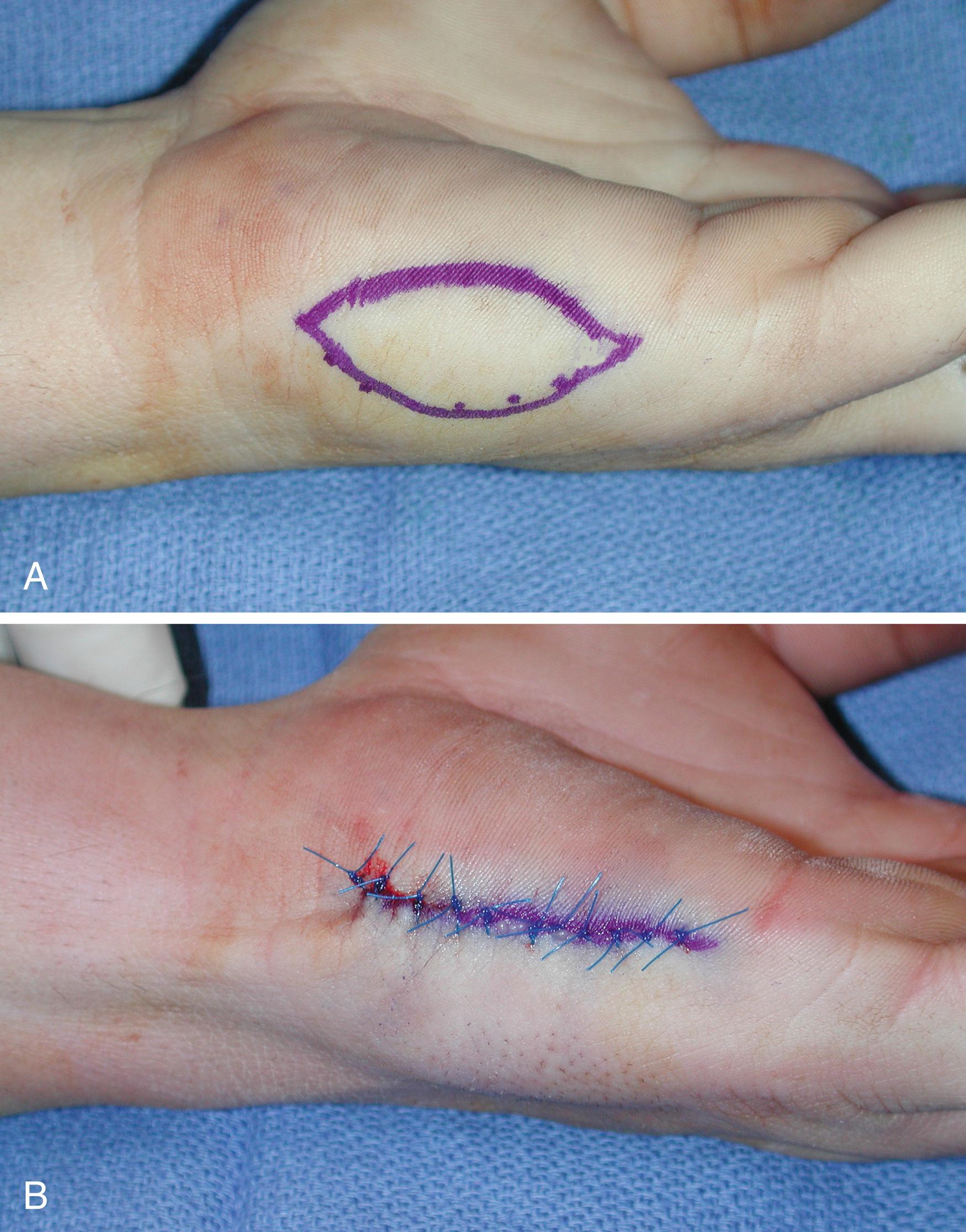
Most full-thickness grafts will be placed on the palmar aspect of the hand; however, the surgeon must remember that there is a marked difference in pigmentation of the palmar skin in dark-skinned patients, and nonglabrous donor sites should be avoided if possible. In dark-skinned patients, full-thickness grafts for placement on the palmar fingers or in the palm should be taken from the hypothenar area of the hand or from the previously mentioned foot donor sites.
After the appropriate size has been determined, an area for harvest of the full-thickness graft is marked. I prefer to infiltrate underneath this area with plain lidocaine or marcaine, which will elevate the skin somewhat and make harvesting easier (as well as provide some anesthesia in the postoperative period). Local anesthetic with epinephrine should be avoided as the epinephrine has been reported to cause capillary bed spasm and poor “take” of the graft. An ovoid of skin is excised from the predetermined area with the axis in the direction of minimal tension. The wound is closed in standard fashion. Most donor sites for hand coverage will be small, and primary closure can be performed easily without undermining of the edges. Because there will be a moderate amount of tension on the edges, closure is best with buried subdermal sutures and a subcuticular suture to avoid the cross-hatching with ordinary cutaneous sutures.
Graft preparation must remove all fat and subcutaneous tissue from the undersurface to avoid any barrier to vascularization. If desired, this can be accomplished during graft harvest with careful excision of only the skin, leaving the subcutaneous tissue behind using sharp dissection with a scalpel in the subdermal plane. The graft can be rolled over a finger as it is harvested and, with appropriate tension, most of the underlying subcutaneous fat can be removed. Once the graft is excised, any remaining subcutaneous tissue is removed. This is done by draping the graft around the surgeon’s nondominant index finger and holding tension along the ends of the graft with the other fingers. The remaining fatty tissue is removed with a small, curved pair of scissors. The normal tendency is to remove portions of the dermis as well, which can actually lead to creating a “thick” split-thickness graft and lead to more contracture. The final result in terms of quality of the graft will be better if the subdermal areolar tissue is left with the graft. The fat can also be removed by placing small mosquito hemostats on the tips at each end of the graft and draping the skin over a finger or the thumb while keeping tension on both hemostats. If some tension is maintained, it is easy to snip away the fat in this fashion.
As previously indicated, a thicker graft contracts less, holds up better, and regains better sensibility compared with a thin graft. In general, split-thickness grafts should not be used on palmar surfaces of the hand unless there is no other choice or the coverage is temporary with secondary resurfacing at a later date. If split-thickness grafts are to be used, they should be relatively thick, and long-term compression with an elastic glove should be used.
Full-thickness grafts are seldom indicated on the dorsal surface of the hand. The full-thickness requirements are not as critical on the dorsum, and split-thickness grafts are generally satisfactory. In addition, the convexity of the dorsum and the subsequent greater tension on the wound edges with motion allow split grafts to develop less contraction compared with the concave palmar aspect. A general rule is that a sheet of good-quality skin graft of approximately 0.015 inch thickness placed on a well-vascularized bed early in the evolution of the wound will heal so well that it is difficult for a casual observer to notice ( Fig. 44.9 ). If the bed is not ideal, regardless of the type of graft used, the result will always be unsatisfactory. Large areas of bare tendon, bone denuded of periosteum, and nerves and vessels bowstringing in the wound should never be covered with a graft. In such instances, coverage should be obtained by the use of a flap (see later in this chapter). Very small areas of exposed tendon can be successfully covered with a full-thickness graft only if the surrounding bed is good. This success is due to the presence of the capillary bed in a full-thickness graft, which allows vascularization of small areas of graft not located over areas with adequate vascularity (assuming that the rest of the graft becomes vascularized).
Negative pressure wound management (most usually the vacuum-assisted closure [VAC] device) has become a popular and effective
Wounds with very healthy bed in an area where better coverage is desired
Management of scar contraction in palmar surface
Wound bed vascularity
Lack of infection in wound bed
Avoid grafts from wrist crease and antecubital fossa.
Graft from medial upper arm or groin.
Do not “defat” graft so much that it becomes a split-thickness graft.
Place lidocaine (without epinephrine) under the skin to be taken first.
Defat the skin graft as you elevate it.
Take an ellipse so that the donor site can be closed primarily.
Precisely suture the graft into the defect, trimming as necessary.
Avoid straight suture lines along web spaces.
Use a “tie-over” bolster for most full-thickness grafts.
Leave bolster in place with the extremity immobilized for at least 2 weeks.
Infection under grafts
Motion of graft before revascularization
Overly aggressive defatting that turns a full-thickness graft into a split-thickness graft
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here