Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Diagnostic dermatopathology of the vulva is a field that may be practiced on a multitude of levels. Patients may be seen by gynecologists, family practitioners, internists, or dermatologists. The biopsy may be read by a dermatopathologist, gynecologic pathologist, or general pathologist. Thus, any chapter addressing diagnostic vulvar dermatopathology must serve a multitude of audiences. This chapter is designed to provide an organized approach to inflammatory vulvar dermatoses and educate the reader on the potential pitfalls in the interpretation of and adherence to concepts that may hinder correct diagnosis and therapy. The reader who desires a more comprehensive discussion of these entities is referred to more extensive works.
For years, a diagnostic schism has existed between dermatopathologists and general surgical and gynecologic pathologists. Although there is virtually no form of dermatitis that has not been reported to involve the vulva, separate formal and informal classification schemes for inflammatory dermatoses of the vulva have been used for years. The classification of inflammatory dermatoses of the vulva using the same terminology applied to dermatoses affecting nonvulvar skin is advised for several reasons. In the past, there has been a tendency for general pathologists to lump groups of disorders into so-called wastebasket diagnoses, such as dystrophy or hyperplasia. The diagnostic term, squamous cell hyperplasia, appears in the classification scheme proposed by the International Society for the Study of Vulvovaginal Disease. Although squamous cell hyperplasia is appropriate as a histologic descriptor, it does not connote a specific entity and conceivably could be misused as a label for a more severe epithelial proliferation. The possibility of misclassification is real. One study that reviewed vulvar biopsies diagnosed by general pathologists found that a diagnosis of squamous cell hyperplasia had been applied to cases of lichen simplex chronicus, contact dermatitis, psoriasis, lichen planus, and hypertrophic lichen sclerosus. In this chapter, vulvar dermatoses are classified as for nonvulvar skin. Because a comprehensive review of dermatopathology is well beyond the scope of this chapter, the discussion here focuses on the most common and significant dermatoses that involve the vulva. The differential diagnosis of the more common entities is outlined in Table 2.1 .
| Diagnosis | Acan | PK | Spong | HK | LI | ID | IEN | Sclerosis | Atroph | TTR | Exclude |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Lichen simplex chronicus (LSC) | + | ± | + | ± | Candida | ||||||
| Lichen sclerosus (LS) | ± | ± | ± | ± | + | ± | dVIN | ||||
| Eczema | ± | + | ± | Seborrheic dermatitis | |||||||
| Lichen planus | ± | ± | + | + | Early LS | ||||||
| LSC + LS | + | + | + | + | dVIN | ||||||
| Eczema + LSC | + | + | + | Candida | |||||||
| Zoon | ± | + | ± | Syphilis | |||||||
| Psoriasis | + | + | ± | LSC, Candida |
Eczematous dermatitis (ED) is a commonly encountered group of conditions that frequently involves the vulva. A recent study from a single institution has found that 23% of vulvar biopsies over a 4-year period were diagnosed as eczematous dermatitis. The common histologic denominator of diseases classified under the rubric eczema is the presence of spongiosis (intercellular edema), which may be mediated by exogenous or endogenous factors. An inflammatory response, which is invariably present, is at least partially responsible for many of the patient's symptoms. Many clinical subtypes have been described, and those most frequently encountered in the vulva are discussed here. Such variants cannot be reliably separated on the basis of histology alone, and clinicopathologic correlation is essential. However, clinical distinction among the various causes of eczema is sometimes difficult or even impossible, especially when the vulva is predominantly affected.
To challenge recognition of these conditions further, ED evolves through stages, often rapidly, resulting in clinical and histologic patterns that can be strikingly similar, regardless of cause. Many patients are simply designated as having eczema. In some cases, only with specialized investigation, such as patch testing, is the disease precisely characterized. It is the pathologist's role to evaluate for the presence of spongiotic dermatitis and its stage of development, accepting the fact that in most cases, elucidation of the specific clinical subtype or cause based on histologic findings is impossible. In this section, the natural history of ED is discussed, followed by a discussion of the major clinical subtypes of ED affecting the vulva.
ED can be seen in those of all ages, including infants. The earliest detectable feature is erythema, usually associated with pruritus, dryness, or burning. The last two symptoms are usually reported with mucosal involvement. Pain on palpation and dyspareunia can be present. Less commonly, the condition may be asymptomatic.
Tiny delicate vesicles develop and, with time, may coalesce to form larger vesicles and even bullae. The epidermal edema often gives the skin surface a weepy, wet appearance, but this is not always a prominent feature. A superficial, pale, yellowish scale or crust is a frequent finding ( Fig. 2.1 ). Rubbing and scratching in response to pruritus leads to the development of superimposed changes of lichen simplex chronicus (LSC) and often to excoriation. LSC is characterized by a thickening of the skin, imparting a leathery quality, with accentuation of the skin markings (see below, “ Lichen Simplex Chronicus ”). Superinfection with fungal or bacterial organisms may occur, especially following vigorous scratching or topical steroid therapy. The chronic lesions of all forms of ED are strikingly similar in appearance.
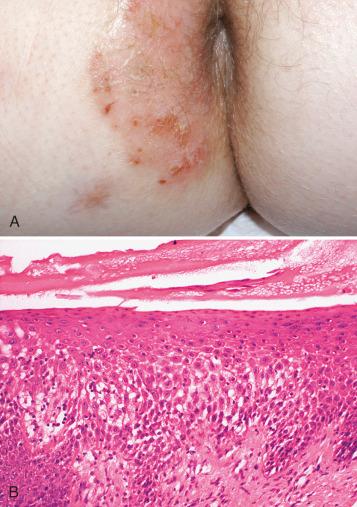
Exogenous dermatitis results from direct application of a substance to the skin. Two main types of exogenous dermatitis are seen in the vulva—irritant contact dermatitis and allergic contact dermatitis. The distinction between exogenous and endogenous dermatitis can be blurred because contact with irritants can promote or accentuate the latter condition.
Irritant contact dermatitis results from direct damage to the epithelium by exogenous agents. This variant is much more common than allergic contact dermatitis. The dermatitis is typically localized to the area where the offending substance has been applied, and the response is usually initiated within minutes to hours of exposure. A secondary inflammatory response develops in response to the injury. Visible vesicles are usually not a feature. Diaper rash is a common irritant contact dermatitis in infants or older adults, in which urine or stool combines with the friction of the diaper to produce eczematous dermatitis. Irritant contact dermatitis can be localized to the vulva when exposure is limited to this site or because the vulvar region is more sensitive to the offending substance compared with other areas. Common causes are urine, sweat, friction, topical products (e.g., lotions, soaps, perfumes, cleansers), and a variety of topical over-the-counter and prescription medications. Attempts at treatment may exacerbate the disease. In particular, the patient often perceives dermatitis in this area as a lack of cleanliness. This can incite an urge to cleanse the area, increasing the severity of the dermatitis. Secondary infection is often a problem. The cycle is further exacerbated by a reluctance to discuss symptoms with health care providers. Treatment mainly involves avoiding inciting and exacerbating substances; however, in some patients, topical steroids are also required.
Allergic contact dermatitis is less often encountered than irritant contact dermatitis and is distinctly uncommon in the very young and older adults. It represents a type IV hypersensitivity reaction to a specific antigen to which the patient has been previously exposed. Lesions are characterized by develop significant erythema, often with subsequent bulla formation. The condition usually arises within 48 hours after exposure in the sensitized patient and is generally confined to the area of exposure, but can become generalized. Lesions can persist for several weeks. This type of reaction is exemplified by the well-known response to poison ivy. The list of causes in the vulva is similar to those for contact dermatitis elsewhere and includes specific ingredients in substances such as soaps, lotions, perfumes, and cosmetics. Topical medication is also a common cause ( Fig. 2.2A ). Allergic dermatitis in response to semen and constituents of latex condoms has been described. The use of moist flushable wipes has been increasingly identified as a cause of allergic vulvar dermatitis due to the presence of methylchloroisothiazolinone.
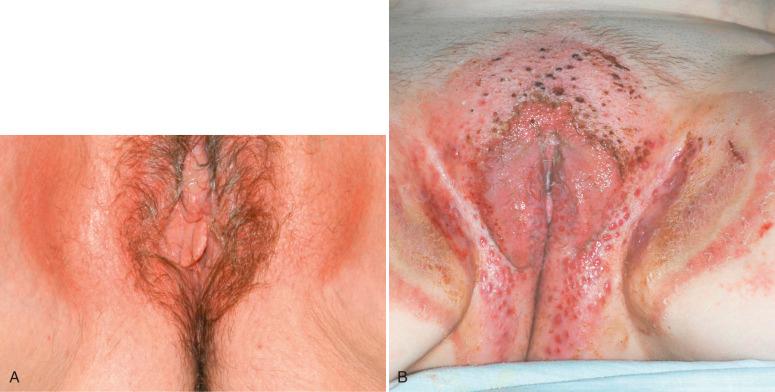
Allergic contact dermatitis is confirmed by skin patch testing, which involves placing small pieces of material containing the substance to be tested on the skin and measuring the clinical inflammatory response. Such methodology can identify the specific substance causing the dermatitis in a significant portion of appropriately selected patients. Allergic contact dermatitis is often seen in patients with a history of atopy (discussed next) and thus likely involves inheritance of a propensity for its later development. Complete avoidance of the contactant is recommended. The use of topical products should be minimized. If medications are required, an ointment base is recommended because creams contain many additives.
Atopic dermatitis (AD) is a hypersensitivity condition with familial clustering that involves skin and mucosal sites and is associated with respiratory conditions such as asthma and allergic rhinitis. It is seen in 10% to 20% of children, and its incidence may be increasing. The most common allergen inciting AD is from the common household dust mite, Dermatophagoides pteronyssinus . AD can be conceptualized as an exaggerated response to common cutaneous antigens. It usually involves the face, scalp, and extensor surfaces of the arms and legs. AD is believed to be an immunoglobulin E (IgE)–mediated immune reaction with pathogenic similarities to asthma; however, the precise mechanisms of the hypersensitivity are not well understood. Although AD often resolves by puberty, predisposition to ED with irritation of the skin from a variety of sources can endure for life. Some authorities believe this to be the most common form of ED involving the vulva. In patients with a history of atopic dermatitis, ED is often attributed to atopy when other causes are excluded. Treatment consists of eliminating inflammation and infection. Emollients and antipruritic agents are used to reduce the self-inflicted damage to the involved skin and control exacerbating factors. Most patients can be brought under adequate control in less than 3 weeks. Reasons for failure include poor patient compliance, the simultaneous occurrence of asthma or hay fever, inadequate sedation, and continued emotional stress. Allergic contact dermatitis is found to be more common in patients with a history of atopy.
Nummular dermatitis is generally seen more often in men than in women, with peak incidence in those older than 55 years. However, a predominantly female incidence is found from ages 15 to 25 years. The characteristic lesions start as erythema with tiny vesicles that quickly coalesce to form discrete, coin-shaped plaques that give rise to the alternative term, discoid eczematous dermatitis. These lesions can be single or multiple and are usually less than 10 cm in diameter. They are intensely pruritic and are commonly present in the upper extremities—dorsum of the hand is a classic site—and trunk, but they can be seen in a wider distribution. The vulva is rarely involved. With time, the lesions can show central clearing and may be mistaken for a fungal infection. The lesions can persist and will often recur in a previous site of involvement. Topical steroids are commonly used in the treatment of this condition.
Histologically, ED is defined by epidermal spongiosis—intercellular epidermal edema that splays keratinocytes apart, rendering their spinous processes more prominent (see Fig. 2.1B ). A spectrum of histologic changes corresponding to the duration of lesions is well recognized and has been traditionally divided into acute, subacute, and chronic phases. The composition and amount of inflammation are variable. Often, in the acute phase, there is a variable, superficial, papillary dermal inflammatory cell infiltrate consisting primarily of lymphocytes, sometimes with eosinophils. Scattered lymphocytes, eosinophils, and even neutrophils can be seen in the epidermis. With increasing spongiosis, the cell-cell junctions of the epidermal keratinocytes are compromised, and small intraepidermal vesicles form. These can eventually coalesce to form larger vesicles and even bullae. With severe edema, focal separations at the dermal-epidermal junction may also occur. With time, secondary keratinocyte necrosis may be seen in severe cases. The intraepidermal vesicles empty onto the epidermal surface and give rise to the weepy appearance and may dry to form a scale or crust. Incited by the pruritic nature of the vesicles, rubbing and scratching usually accelerate the uncapping of vesicles and produces erosions. Significant hyperkeratosis or epidermal acanthosis is not seen in the acute phase.
In the subacute phase, in addition to spongiosis, acanthosis or hyperplasia of the epidermis is seen. Often, the acanthosis occurs in a psoriasiform pattern (i.e., resembling psoriasis) and, at least in part, is a response to scratching or rubbing. A dermal inflammatory infiltrate of variable density and composition is seen. Mild hyperkeratosis and hypergranulosis are often present in subacute lesions.
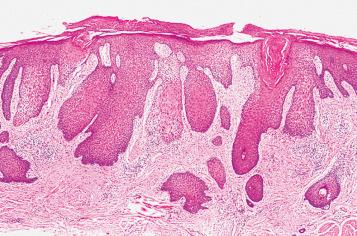
In the chronic phase, spongiosis may be more difficult to identify, and the epidermal hyperplasia becomes more pronounced. Marked hyperkeratosis and hypergranulosis are regular features. The changes come to resemble the microscopic appearance of acral skin, which is likewise determined by constant exposure to friction. Accordingly, in practice, these entities have been descriptively reported as acute, subacute, or chronic spongiotic dermatitis, with perivascular infiltrates of lymphocytes and eosinophils, rather than eczema or nummular dermatitis, depending on the stage of a particular lesion. When the features of epidermal acanthosis, hyperkeratosis, and hypergranulosis are sufficiently pronounced as a result of rubbing and scratching, the lesion is designated as chronic eczematous dermatitis with superimposed LSC ( Fig. 2.3 ). However, it should be understood that although ED is a common underlying cause of LSC, other associations are well documented. Therefore, the changes of LSC (without clear-cut associated spongiotic dermatitis) should not be viewed as synonymous with chronic ED (see later, “ Lichen Simplex Chronicus ”).
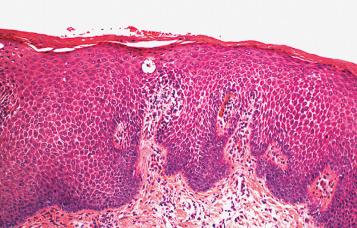
There is considerable histologic overlap among the various types and causes of ED in the vulva. The role of the pathologist is to recognize ED and be able to distinguish it from other dermatitides. Once a diagnosis of spongiotic dermatitis is reached, it is then the clinician's responsibility to search for the cause and classify the reaction precisely. It is especially important for the pathologist to be aware that all the histologic features of ED can be precisely mimicked by fungal infection. Therefore, a diagnosis of spongiotic dermatitis or ED should never be rendered until a periodic acid–Schiff (PAS) or silver staining procedure has been performed to exclude the possibility of a fungal infection. This is particularly important because a fungal infection misdiagnosed as ED and treated with topical steroids will result in exacerbation of the infection ( Fig. 2.4 ).
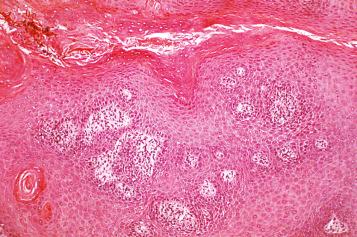
LSC is not a distinctive disease, but rather it denotes a characteristic clinical and pathologic response of skin to repeated physical trauma, such as rubbing or scratching, often superimposed on a pruritic condition such as eczematous dermatitis. LSC is commonly seen in patients who habitually scratch but have no identifiable underlying dermatoses. When no underlying cause is discovered, this process has sometimes been termed neurodermatitis . The vulva is a frequently involved site.
LSC presents as thickened skin that has erythema and scaling that is plaquelike. Physiologic skin markings are exaggerated, and visual excoriations are present ( Fig. 2.5A ). Hyperpigmentation may also be seen. Patients usually present with complaints of pruritus of weeks to months in duration, often worse at night. Scratching of the lesion can become habitual and subconscious and, if extensive, will lead to pronounced epidermal thickening (see Fig. 2.5B ). The lesion is usually solitary; the most common vulvar site is the labium majus. There is usually an erythematous hue to the skin. Postinflammatory hyperpigmentation may develop. Patches of hypopigmentation or depigmentation may also occur.
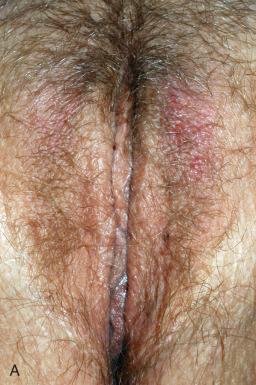
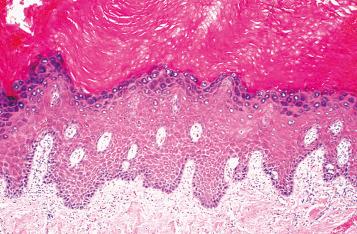
Biopsy is usually not necessary in LSC. However, when required, biopsy should not be attempted until the itch-scratch cycle is stopped. LSC is characterized by acanthosis of the skin. The epidermis shows hyperkeratosis and hypergranulosis ( Fig. 2.6 ). Superimposed excoriation and scale or crust may be seen following vigorous scratching. The rete ridges are elongated and thickened in an irregular psoriasiform pattern. In nonvulvar skin, vertically streaked coarse collagen and small vessels are often present, but these features are less prominent in the vulva. A sparse mononuclear infiltrate is often found in the dermis, and epidermal mononuclear cells can be seen. Neutrophils may be present in the epidermis if there is excoriation, with or without impetiginization.
Because LSC is often superimposed on another typically pruritic lesion, it is important not to diagnose LSC without pursuing the underlying cause. Conditions that can be confused with LSC include fungal infection, tinea cruris, chronic ED (see Fig. 2.4 ), and psoriasis. Fungal infections are often pruritic, and chronic infections may be associated with superimposed features of LSC (see Fig. 2.4 ). It is important to exclude fungi with PAS or silver staining, particularly if neutrophils are seen in the epidermis or stratum corneum. Chronic eczematous dermatitis is often associated with LSC (see “ Eczematous Dermatitis ”); it forms a spectrum with LSC, and its precise designation is somewhat arbitrary. However, if spongiosis is identified, it is important to indicate this in the diagnostic report. In our experience, ED is the most common specific dermatosis associated with LSC. To help differentiate chronic eczematous dermatitis from isolated LSC, it is necessary to identify epidermal spongiosis, which can be focal (see Fig. 2.3 ). Finally, psoriasis can be differentiated from LSC by virtue of its narrow rete pegs and more regular epidermal hyperplasia of the rete pegs; hypogranulosis and parakeratosis, often associated with neutrophils, are also characteristic.
When not associated with other epithelial changes, LSC is not considered to be a precursor to vulvar squamous carcinoma. However, certain patterns, such as verruciform LSC, verruciform LSC with altered squamous differentiation, LSC associated with lichen sclerosus, and LSC complicated by differentiated vulvar intraepithelial neoplasia (VIN), may increase the risk of squamous cell carcinoma of the vulva. These patterns are discussed in Chapter 6 .
For successful treatment, it is imperative to interrupt the itch-scratch cycle. A severe case of this condition is shown in Fig. 2.7 ; also see Fig. 2.5 . The labia are thickened and have many areas of tissue denudation from the constant scratching. Treatment involves topical steroids and wearing cotton gloves at night to prevent skin damage from scratching. Steroids are often needed to achieve acceptable relief and resolution. Because LSC is commonly superimposed on specific dermatoses, treatment of any associated disorder is also required.
Psoriasis is a chronic, relapsing hyperproliferative papulosquamous disorder that affects 1% to 2% of the white population. The mean age of onset is in the late 20s. The male-female ratio is equal in adults, but there is a female predominance in pediatric cases. The disease commonly affects the vulva (as many as one-third of cases), which can be involved at presentation. Isolated vulvar involvement is occasionally seen but is uncommon. The pathogenesis of psoriasis is complex but appears to involve immune dysfunction. Linkage to certain human leukocyte antigen (HLA) class I haplotypes is seen, which may help explain the familial clustering of psoriasis. Aberrant activities of CD4- and CD8-positive T lymphocytes, which appear to help initiate and sustain the lesions, are likely involved. The antigen or antigens inciting this response have not been elucidated. There are also epidermal abnormalities leading to hyperproliferation with abnormal maturation. As many as one-third of patients with psoriasis have a first-degree relative with psoriasis.
Psoriasis has a wide and variable spectrum of clinical presentations. Classic plaque (psoriasis vulgaris), inverse (or flexural), guttate, and pustular forms will be discussed in detail. These forms commonly involve the vulva.
Classic plaque-type psoriasis presents with symmetrically distributed erythematous, often indurated, plaques that are well circumscribed. This is the most common variant of psoriasis to involve the vulva ( Fig. 2.8A and B ). A silvery scale often covers the lesions, but this finding is less frequently seen on the vulva. When present, the scale tends to be present at the border of the plaque. Vulvar lesions often have a shiny erythematous appearance and are well demarcated. Removal of the scale may produce pinpoint bleeding due to trauma of the prominent superficial capillaries; this is referred to as the Auspitz sign. Although probably not diagnostic of psoriasis, it is a characteristic finding. The extensor surfaces of the upper and lower extremities are most often involved. The pretibial, umbilical, intragluteal, and lumbosacral regions may also be affected. Psoriasis is associated with the Koebner phenomenon, lesions arising in areas of prior trauma.
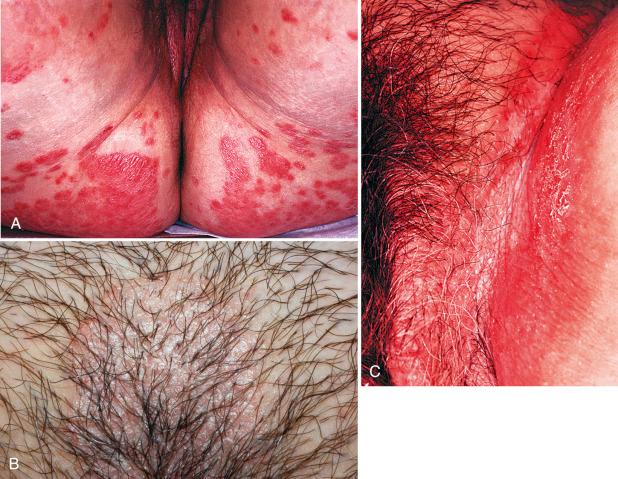
Inverse or flexural psoriasis shows an alternative distribution, in which moist flexural surfaces such as the inframammary, inguinal, and abdominal pannus areas are involved. This pattern is usually found in obese individuals. The involved areas are erythematous but generally do not show the silvery micaceous scale of classic plaque-type psoriasis. Given their location, these lesions may become macerated or fissured (see Fig. 2.8C ).
Guttate psoriasis preferentially affects children, adolescents, and young adults. The condition often arises after a beta-hemolytic streptococcal infection. Guttate lesions may herald the onset of plaque-type psoriasis. It presents as crops of small (<1 cm) pink to erythematous lesions, usually without prominent scale. The trunk, buttocks, and periphery of the extremities are usually affected. Vulvar involvement is rare, and lesions limited to the vulva would be exceptional.
The lesions of pustular psoriasis are typically red crusted plaques, with superimposed pustules. The crust is often more prominent than in other variants. Patients with pustular psoriasis usually show systemic symptoms such as fever, which may become severe enough to warrant hospitalization.
Psoriasis may be associated with a debilitating form of arthritis. Usually, psoriasis precedes the onset of arthritis by many years. The arthritis is most often asymmetric but can also be symmetric in distribution. Psoriatic spondylitis is also present in the occasional patient.
The classic lesion of plaque-type psoriasis is characterized by regular acanthosis or hyperplasia of the epidermis, with elongated rete pegs. This pattern of epidermal hyperplasia, termed psoriasiform hyperplasia, resembles test tubes in a test tube rack. The tips of the rete pegs may be widened or clubbed and can fuse in long-standing lesions ( Fig. 2.9 ). Parakeratosis is frequently associated with neutrophils, and intraepidermal pustules are often present. Munro microabscesses are intracorneal collections of neutrophils. The spinous layer can contain collections of neutrophils with spongiosis, referred to as spongiform pustules of Kogoj ( Fig. 2.10 ). The superficial dermal papillae contain ectatic tortuous vessels and marked attenuation of the suprapapillary plate. Trauma to the thinned epidermis above the expanded highly vascular papillae accounts for the Auspitz sign. A variable, superficial, interstitial mixed inflammatory infiltrate is usually present.
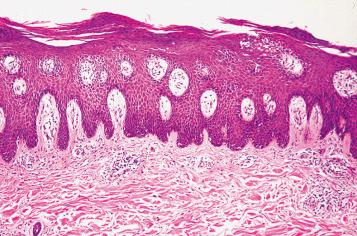
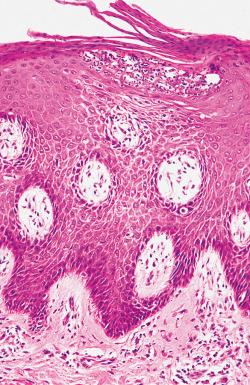
The lesions of psoriasis evolve over time. The earliest recognizable changes in psoriasis are dilation of the vessels of the superficial vascular plexus, with a mild lymphocytic perivascular and interstitial infiltrate. Over time, acanthosis and parakeratosis develop, with increased mitotic activity in the epidermis. Guttate lesions show a more subtle, irregular psoriasiform hyperplasia and may maintain a granular cell layer. Distinct foci of parakeratosis with associated neutrophils are typically seen.
Pustular psoriasis typically has more prominent Munro microabscesses, which often merge to form macropustules and conspicuous spongiform pustules of Kogoj.
The histologic features of psoriasis may be precisely mimicked by candidiasis. Thus, it is important to perform PAS staining to rule out fungal infection. Although focal spongiosis is not uncommon in psoriasis, eczematous lesions exhibit more diffuse spongiosis and are not associated with thinning of the suprapapillary plate, and parakeratosis, when present, is typically focal. Hypergranulosis and hyperkeratosis, in the setting of spongiosis, suggest a diagnosis of subacute to chronic eczematous dermatitis. Psoriasiform drug reactions are common and can closely mimic psoriasis. Significant numbers of eosinophils in the inflammatory cell infiltrate favors a psoriasiform drug reaction. Clinical history is paramount for differentiating these two possibilities. LSC shows marked acanthosis, often in an irregular pattern associated with hyperkeratosis and hypergranulosis. The lesions of Reiter syndrome can appear identical to those of psoriasis, particularly the pustular variant, and clinical information is required to make the distinction (see later, “ Reiter Syndrome ”).
The development or worsening of psoriasis during treatment with tumor necrosis factor (TNF) antagonists has been recognized. It may occur at any time after drug initiation, ranging from days to years. This reaction is associated with cytokine dysregulation following TNF inhibition, but the precise pathogenesis remains unknown. This phenomenon has been reported in almost all diseases treated with TNF antagonists, including rheumatoid arthritis, juvenile arthritis, ankylosing spondylitis, Behçet disease, inflammatory bowel disease and, paradoxically, psoriasis and psoriatic arthritis. Of note, biopsies of TNF antagonist–induced psoriatic skin lesions are histologically similar to those of patients with idiopathic psoriasis. As TNF antagonists have been increasingly used, a wide variety of unexpected and undesirable cutaneous reactions, other than psoriasis, is becoming evident.
The presence of psoriasiform hyperplasia and a lichenoid tissue reaction with epidermal infiltration by lymphocytes and sometimes neutrophils should alert the pathologist to the possibility of secondary syphilis (see Chapter 4 ).
Treatment for psoriasis depends on the severity of the disease; it includes topical steroids or intermittent tacrolimus ointment or pimecrolimus cream, antiproliferative agents, antibiotics to treat secondary infection, oral steroids, methotrexate, acitretin, systemic calcineurin inhibitors such as cyclosporine, and biologic agents (e.g., etanercept, infliximab, adalimumab, ustekinumab) in severe cases. Other immunosuppressive agents sometimes used in select cases of severe psoriasis include hydroxyurea, 6-thioguanine, and azathioprine. Another agent, apremilast, is used in the treatment of moderate to severe plaque psoriasis in patients who are not candidates for phototherapy or systemic therapy.
When considering the traditional therapeutic modalities for the management of psoriasis, be cautious to avoid irritating the sensitive genital skin. Treatment can lead to apparent remission of lesions, but the propensity to develop plaques remains; thus, psoriasis is usually a chronic and frustrating problem.
The three main features of Reiter syndrome are arthritis, genitourinary tract symptoms, and conjunctivitis. It is classified as a seronegative spondyloarthropathy with a clinical presentation of erythematous plaques, combined with urethritis or cervicitis that occurs following a bacterial infection. The cutaneous lesions of Reiter syndrome have been referred to as keratoderma blennorrhagicum; they begin as small pustules or erythematous macules that eventually coalesce to form erythematous plaques with a scale or crust. Clinically, these lesions are similar, if not identical to, those of pustular psoriasis, and usually involve the hands and feet. These cutaneous lesions occur as part of a postinfectious syndrome. Two broad types of infection predispose to Reiter syndrome and are designated as endemic or epidemic.
Endemic infections are often caused by Chlamydia trachomatis, commonly present as urethritis, and show a striking predilection for males (>80%). The epidemic form occurs in small subsets of people following outbreaks of an enteric infection. Gastroenteritis is usually due to Shigella dysenteriae, Salmonella typhimurium, or Yersinia enterocolitica. There is no gender predilection in epidemic outbreaks, but such outbreaks are a very rare cause of Reiter syndrome in Western countries.
The pathogenesis of Reiter syndrome is poorly understood. The major histocompatibility haplotype HLA-B27 is greatly overrepresented in patients with Reiter syndrome, making immune dysregulation a plausible explanation. Dysregulation of CD8-positive T cells has been proposed as a mechanism. Predisposition to the disease appears to be inherited and tends to run in families with other seronegative spondyloarthropathies and pustular psoriasis. These diseases occur in families and coexist in individuals with Reiter syndrome, indicating probable shared pathogenic mechanisms.
Vulvar involvement is rare, as exemplified by the paucity of reports in the English medical literature. Within approximately 1 month of infection, urethritis (or, more often, cervicitis in women) occurs. With time, arthritis, usually asymmetric and involving the large joints of the lower limbs, ensues. The cutaneous lesions can occur at any time during this course. Other symptoms include onychodystrophy, enthesopathy—tendon or ligament inflammation at its insertion; the Achilles' tendon is most commonly involved—mouth ulcers, iritis, and conjunctivitis. The syndrome is often chronic and relapsing and can be quite debilitating, even fatal. Among the most serious complications are heart block and mitral or aortic valve incompetence as a result of involvement of these sites. Reiter syndrome appears to be particularly serious in the setting of patients with acquired immunodeficiency syndrome (AIDS).
Vulvar disease appears strikingly similar to vulvar pustular psoriasis, with red crusted plaques involving the skin of the vulva and perineum. Well-defined ulcerations within areas of erythema may be seen. Small white papules on the mucosal surfaces of the vulva have been noted. Vaginal and cervical discharge can be present. The timing and severity of the symptoms vary greatly from case to case, making a high index of suspicion necessary to make this diagnosis, especially when it involves a less common site, such as the vulva.
The vulvar lesions of Reiter syndrome show all the features of pustular psoriasis, including uniform elongation of the rete pegs with thinning of the suprapapillary plate, epidermal hyperplasia, and parakeratosis. The granular cell layer is often greatly diminished or absent. Neutrophils can be found in the parakeratotic areas. The spinous layer often contains conspicuous collections of neutrophils, and microabscesses or macroabscesses can be seen in the corneal layer.
The major histologic differential diagnoses include psoriasis, mainly the pustular type, and infection. Histologically, psoriasis may show identical features. Clinical information is therefore required for discrimination. Nevertheless, there appears to be significant overlap among these conditions, and classification may be somewhat artificial. Clinical presentations that favor Reiter syndrome include cervicitis, iritis, conjunctivitis, mouth ulcers, and the presence of the HLA-B27 haplotype. Although arthritis can be a feature of either disease, asymmetric involvement of the larger joints of the lower extremities (rather than in the more distal joints of the hand, as seen in psoriasiform arthritis) favors Reiter syndrome. It should be kept in mind that Reiter syndrome uncommonly involves the vulva, whereas psoriasis is often seen at this site. As with psoriasis, PAS staining should be performed to exclude a fungal infection, which can mimic the histologic features of Reiter syndrome.
Although there is no cure for Reiter syndrome, many patients have resolution of symptoms for long periods. Antibiotics can be used to treat bacterial infection, and topical triamcinolone acetonide is often used for vulvar lesions. Oral acitretin, methotrexate, azathioprine, and cyclosporine have also been suggested. Tacrolimus ointment has been used, with limited benefit.
Seborrheic dermatitis (SD) is a chronic dermatosis of unknown cause. Some evidence has suggested an association with Malassezia (formerly Pityrosporum ) ovalis; however, the mechanism and significance of this association are controversial. SD is characterized by an ill-defined area of erythema that is pruritic and has associated light yellow to red-brown plaques with a dry to greasy scale. Mild involvement of the scalp is commonly referred to as dandruff. Intraepidermal edema and surface weeping are not prominent features. It is usually seen in areas with significant sebaceous activity, such as the upper trunk, scalp, face (especially the eyebrows and nasolabial folds), and ears (particularly the external auditory meatus).
The vulva is less often affected and is often accompanied by concurrent involvement of one of the more common sites. When the vulva is involved, it usually presents on the labia majora, with extension onto the mons pubis and crural folds. Mucosal involvement is not seen.
Mild SD is frequently evident in the general population (1% to 3%) but is present in more than 75% of those infected by human immunodeficiency virus (HIV). In immunocompetent patients, seborrheic dermatitis is limited to the areas mentioned previously. However, in patients with AIDS, the disease is often more severe and can have a wide distribution involving much of the skin surface. It can be seen early in HIV infection and is the most prevalent skin condition in this population. Seborrheic dermatitis has also been associated with Parkinson disease.
The biopsy findings in SD are often subtle and nonspecific. Usually, acanthosis of the epidermis is present, often in a psoriasiform pattern. Parakeratosis is typically present and may be accentuated around follicular ostia (so-called shoulder parakeratosis), in which small numbers of neutrophils are commonly present. Variable spongiosis and a dermal lymphocytic infiltrate may be seen.
Distinction from psoriasis is often difficult. Well-developed parakeratotic mounds associated with neutrophils and uniform test tube epidermal hyperplasia favor psoriasis. SD often shows significant spongiosis and is sometimes classified under the rubric of eczematous dermatitis. Distinction from other forms of eczema requires clinical correlation. It is important to exclude a fungal infection before diagnosing SD or other eczematous dermatitides. The clinician commonly does this with a potassium hydroxide preparation from the skin scale or an aerobic culture swab sent to microbiology for fungal culture, but it is also readily accomplished by diastase-PAS staining. SD is not commonly biopsied on the vulva because characteristic lesions are commonly seen elsewhere in the body, usually the scalp and face.
Topical steroids should be considered for symptomatic seborrheic dermatitis. Antifungals may also be required. Other agents used for this condition include topical calcineurin inhibitors.
Lichen sclerosus (LS) is a common chronic inflammatory dermatosis that frequently involves the vulva and can cause considerable tissue destruction. It is occasionally associated with vulvar intraepithelial neoplasia of the differentiated type (dVIN) and invasive squamous cell carcinoma (see Chapter 6 ). Although the cause of this condition is not known, it is seen in association with autoimmune diseases that also involve the skin (e.g., alopecia areata) and other organs (e.g., pernicious anemia, thyroid disorders). Certain HLA types are more common in patients with LS, underscoring its presumed autoimmune cause or predisposition. Women with LS have also been reported to have a deficiency of androgens. However, numerous studies have emphasized the lack of efficacy of testosterone and support the use of steroids. Infectious and environmental causes have also been considered. Rare familial cases have been reported.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here