Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Neurosurgery is defined as surgery of the brain, spinal cord, peripheral nerves, and their supporting structures, including the blood supply, protective elements, spinal fluid spaces, bony cranium, and spine. This chapter is intended for nonneurosurgeons who want to initiate a framework on which to add further knowledge and experience. It is hoped that it will also help personnel in a community hospital emergency department, residents, advanced practice providers, and medical students communicate effectively and efficiently with neurosurgeons about urgent, emergent, and elective patient care issues. The chapter is divided into sections and includes a discussion of the following: cerebrovascular disorders, central nervous system (CNS) tumors, traumatic brain injury, degenerative diseases of the spine, epilepsy and functional neurosurgery, hydrocephalus, pediatric neurosurgery, and neurosurgical management of CNS infections. The field of neurosurgery is simply too broad to make a detailed encyclopedic overview realistic, but it is hoped that an introduction to the specialty will be useful to the reader.
It is essential at the outset to understand some basic principles concerning intracranial dynamics, cerebrospinal fluid (CSF), cerebral blood flow (CBF), and intracranial pressure (ICP), and these are summarized here for review.
The first principle has been recognized for decades. The cranial cavity has a fixed volume composed of brain tissue (parenchyma), CSF, and blood vessels with their intravascular blood. According to the Monro-Kellie doctrine, the sum of these components within the fixed volume of the cranial cavity is constant, and an increase in one component must be accompanied by an equal and opposite decrease in one or both of the remaining components if ICP is to remain constant. If this does not occur, the ICP will rise, and, at some point, the increase in pressure per unit increase in volume becomes asymptotic, with the increased ICP causing a decrease in blood flow and oxygen delivery. As a consequence, if there is an elevation in the volume of any one compartment, there is a stage of compensation in which the volume of one or more of the other compartments can be reduced to avoid elevations in the ICP. Table 68.1 summarizes and simplifies some of the excess volume syndromes and specific treatment for each.
| Component | Excess Volume Syndrome | Specific Treatment |
|---|---|---|
| Brain tissue | Edema: cytotoxic, vasogenic, perineoplastic, inflammatory | Diuretics: mannitol, furosemide, hypertonic saline; steroids for perineoplastic and inflammatory vasogenic edema |
| Vascular | Elevated P co 2 : hyperperfusion state with loss of autoregulation as in severe hypertension, after trauma or AVM removal; relative venous obstruction | Increased ventilation; diuretics (in hyperperfusion state, avoid mannitol), barbiturates; clear venous obstruction; elevate head of bed (to reduce venous volume) |
| Cerebrospinal fluid | Impaired absorption with congenital, posthemorrhagic, or postinfectious hydrocephalus, communicating or obstructive; loculations; arachnoid or periventricular cysts; rare increased production of CSF with choroid plexus papilloma | Ventricular external drainage (or lumbar drainage only if no threat of herniation) or shunt; with flocculation, or with some types of obstructive hydrocephalus, endoscopic fenestration or third ventriculostomy may be possible; acetazolamide and steroids may temporarily decrease CSF production |
| Mass lesion | Tumor, cyst, abscess, hematoma, radiation necrosis, or cerebral infarction necrosis | Remove, fenestrate, aspirate lesion (often with stereotactic guidance); less commonly, it might be useful to enlarge intracranial volume by decompression |
The second principle is that spinal fluid is produced at a constant rate (≈15–20 mL/h) by the choroid plexus of the ventricles. It is essential to understand that production of CSF is little affected by increased ICP. Therefore, any derangement in CSF absorption or flow may increase the ICP effects of diseases affecting the CNS. Thus, CSF production continues unabated, even to lethal elevations of ICP. In the following discussions on tumors, infection, intracranial hemorrhage, and trauma, many examples will become apparent whereby impaired CSF absorption contributes to the pathologic condition. The only exceptions to the almost constant CSF production are the excess production associated with the rare choroid plexus papilloma and the occasional decreased CSF production seen with ventriculitis.
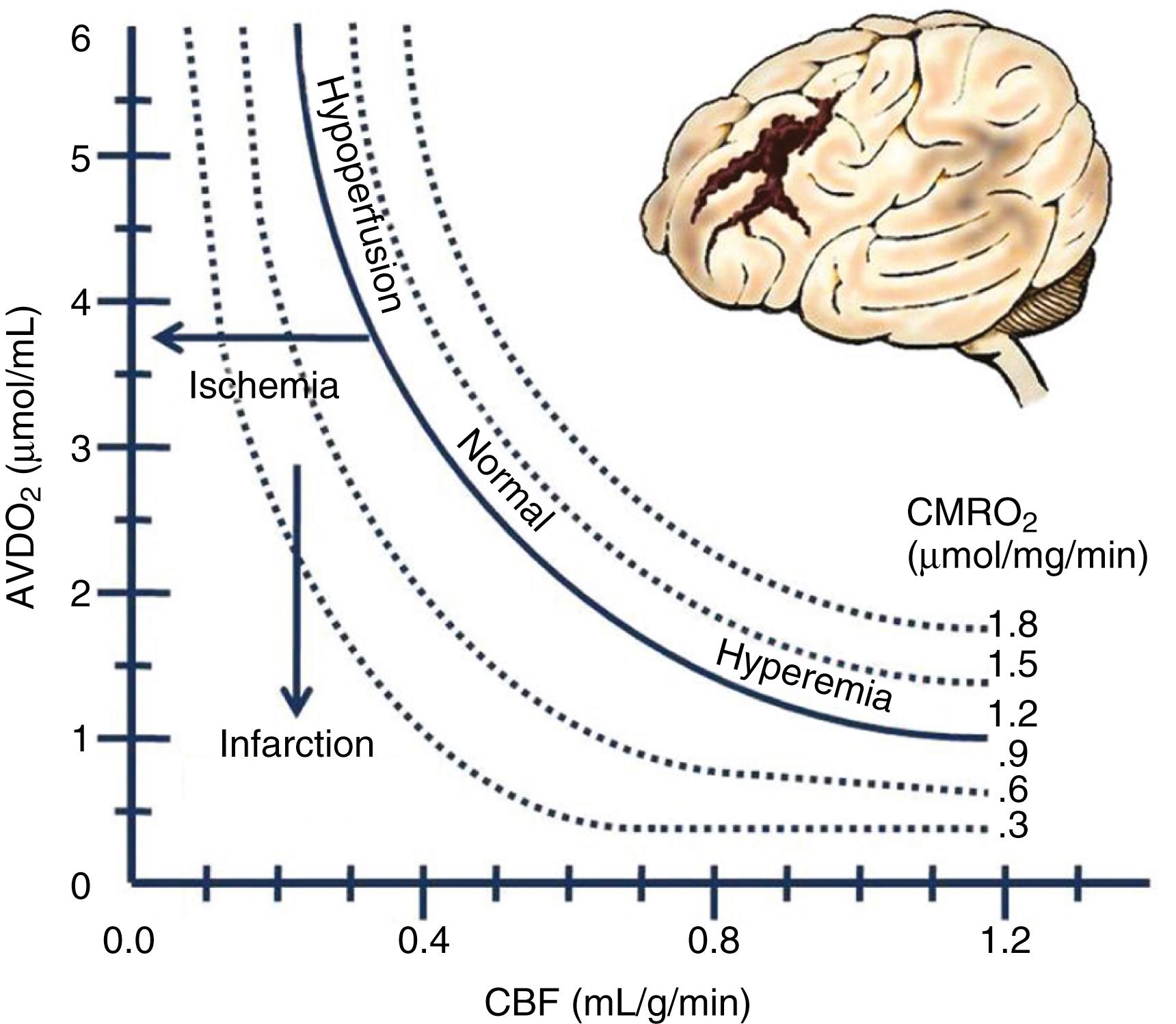
The third basic principle is that the CBF normally varies over a wide range (30–100 mL/100 g of brain tissue per minute), depending on metabolic demand from neuronal activity within a particular area of the brain. The blood flow to any brain area is generally abundant, exceeding demand by a wide margin, so that oxygen extraction ratios are often low. The brain vasculature matches the blood flow to tissue metabolic demand, and the CBF generally maintains what is needed, despite wide variations in systemic blood pressure, by a phenomenon known as autoregulation. Factors such as an elevated or decreased arterial P co 2 shift the curve as indicated. In the setting of traumatic brain injury, the curve becomes more pronounced (i.e., smaller changes in blood pressure or P co 2 ) and affects the CBF dramatically ( Fig. 68.1 ). If tissue demand exceeds autoregulation, or if CBF declines for pathologic reasons, the first defense is that the oxygen extraction will increase (i.e., arteriovenous oxygen difference). The tissue begins to experience dysfunction at levels below 0.25 mL per gram of brain tissue per minute. With levels between 0.15 and 0.20, the brain tissue may undergo reversible ischemia; however, infarction will occur when levels range between 0.10 and 0.15 ( Fig. 68.2 ). The metabolic consumption of oxygen in the brain is decreased after traumatic brain injury to levels between 0.6 and 1.2 μmol/mg/min. Complete loss of blood flow to any brain area results in infarction (irreversible damage) within a few minutes. Swelling of the infarcted tissue takes days to peak and weeks to resolve.
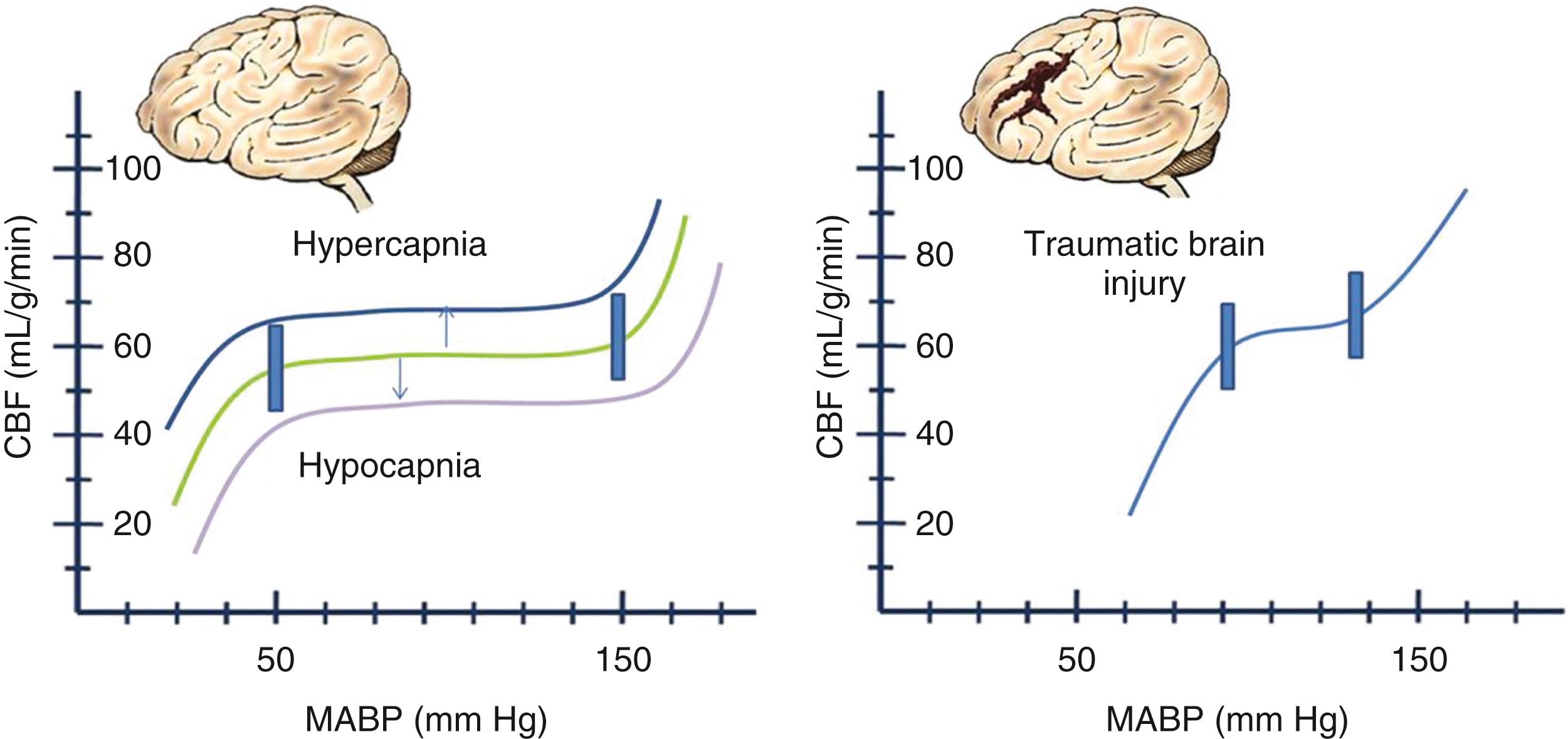
A fourth principle derives from the other three and the fact that as injured tissue swells, it may create a vicious cycle and cause a cascading injury ( Fig. 68.3 ). If ICP is elevated high enough by some mechanism so that cerebral perfusion pressure (CPP) declines, CBF can decline to levels at which tissue injury occurs.
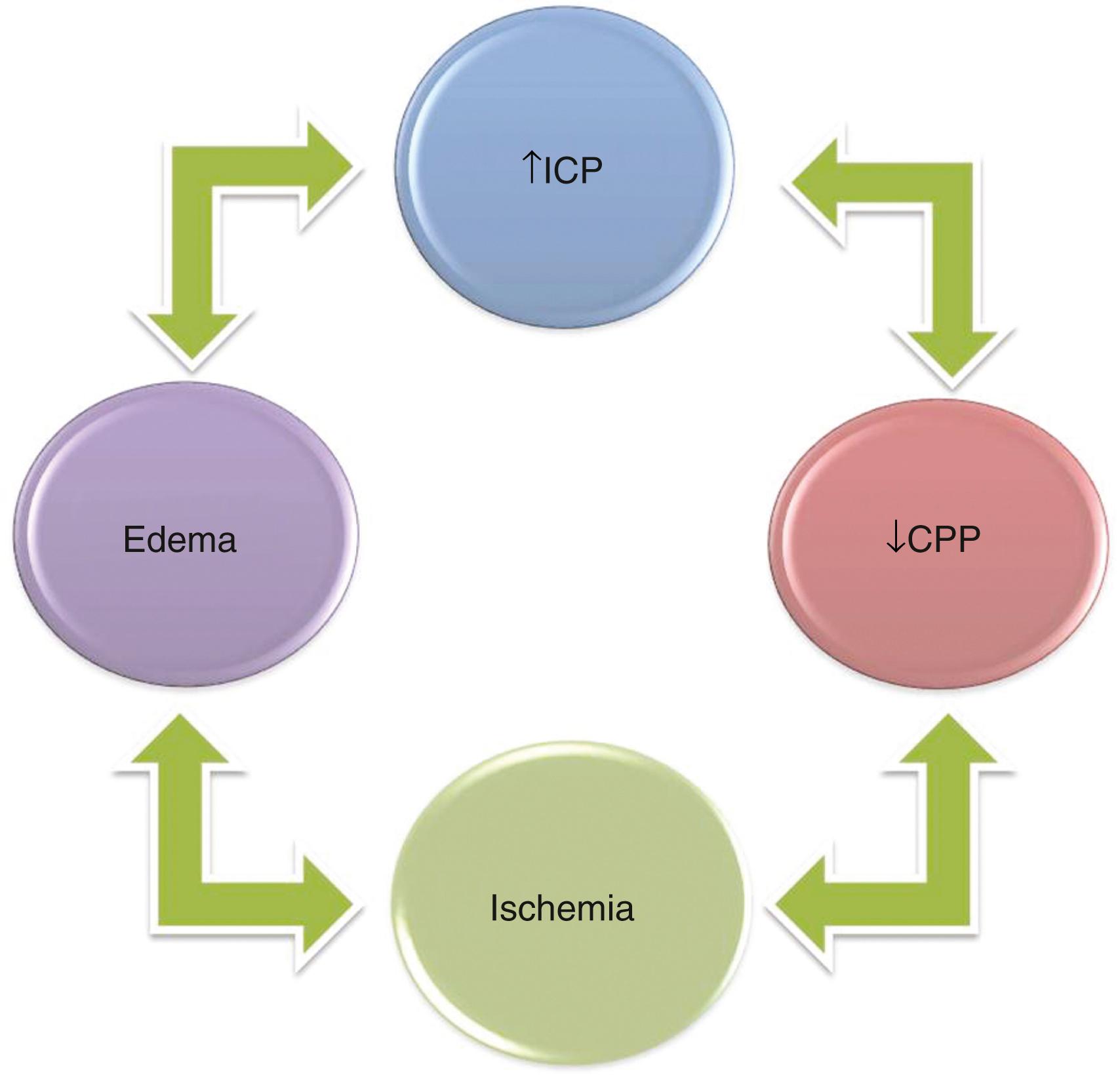
Brain edema (swelling) within the closed cranium will lead to further increases in ICP with even further decreases in CPP in a stage of decompensation. When the capacity for autoregulation is exceeded or damaged so that it can no longer play a role, CBF is linked directly to the CPP.
In the management of intracranial disease, ICP and CPP are easy to measure continuously and thus serve as highly practical surrogates for the more fundamental but much more difficult to measure CBF. However, these are not equivalent, and the limitations of these parameters for guiding therapy need to be remembered. Regardless of causation, when concern arises about the possibility of cascading injury, every effort is made to keep the CPP in the range of 60 to 70 mm Hg and ICP below 22 mm Hg if possible. Routinely using pressors and volume expansion to maintain CPP higher than 70 mm Hg is not supported on the basis of systemic complications.
A fifth principle concerns focal mass effect and its progression in regard to the complex anatomy of the cranial cavity. The cranial cavity contains several knifelike projections of folded dura, the falx and tentorium, which divide the cavity into a right and left supratentorial compartment and an infratentorial compartment (the posterior fossa). The sphenoid wing is a prominent, mostly bony ridge that separates the anterior fossa containing the frontal lobe from the middle fossa containing the temporal lobe. A narrow opening, the incisura, edged by the tentorium, surrounds the midbrain and is the only passage between the supratentorial and infratentorial compartments. Apart from the small openings for the cranial nerves and arteries, the foramen magnum is the only sizable opening in the cranial cavity.
The condition that classically illustrates the expanding mass lesion is the acute epidural hematoma, seen after trauma with skull fracture. Regardless of the source, however, the progression can be similar and has been termed rostrocaudal decay to reflect the early and late stages, as listed in order here:
Focal distortion only
Effacement of gyri and sulci
Compression of the lateral (or other) ventricle
Midline shift
Subfalcine herniation
Temporal lobe tentorial herniation
Third nerve compression (unilateral dilated pupil)
Obliteration of basal cisterns
Midbrain compression
Midbrain infarction, Duret hemorrhages (both pupils dilate, with irreversible damage to midbrain)
Further brainstem compression
Loss of brainstem reflexes: progression from flexor posturing to extensor posturing; vestibuloocular and oculocephalic reflexes; corneal reflexes
Medullary compression syndrome: respiratory reflexes; vasomotor reflexes, Cushing reflex with elevation of the systolic blood pressure, widening of the pulse pressure, bradycardia
Foramen magnum herniation
At stages beyond tentorial herniation, it is unusual for focal mass effects not to be accompanied by an overall increase in ICP. The point at which focal mass effect evolves to include a rise in overall ICP depends largely on the compliance within the cranial cavity. Young patients with so-called tight brains can develop raised ICP, even with relatively small volumes of mass that produce only effacement of the cortical gyri. On the other hand, patients with advanced cerebral atrophy can, for example, tolerate large frontal intracerebral hematomas or chronic subdural hematomas with compression of the lateral ventricle and midline shift while maintaining a tolerable ICP and a surprising degree of near- normal neurologic function.
Cerebrovascular disorders encompass a host of disorders: congenital, acquired, and idiopathic ( Box 68.1 ).
Arteriovenous malformation and fistula
Cavernous malformation
Telangiectasis
Venous anomaly (angioma)
Traumatic
Some arteriovenous fistulas (type I carotid-cavernous fistula)
Traumatic aneurysm
Degenerative
Atherosclerotic, occlusive disease
Most cerebral (berry) aneurysms
Some arterial dissections
Spontaneous intracerebral hemorrhage
Infectious
Mycotic aneurysms
Moyamoya
Some arteriovenous fistulas: dural AVM–like or type II carotid-cavernous fistulas
An arteriovenous malformation (AVM) is an abnormal collection of blood vessels wherein arterial blood flows directly into draining veins without the normal interposed capillary beds. AVMs are congenital lesions that can enlarge somewhat with age, recruiting new vascular supply, and often progress from low-flow lesions at birth to higher flow lesions in adulthood. They present with hemorrhage, ischemia of the brain parenchyma around the lesion due to a vascular “steal” phenomenon, or seizures. They have a prevalence of 15 to 18/100,000 and typically are manifested before the age of 40 years. The risk of hemorrhage rate is up to 4% per year, and once they bleed, they may be even more prone to hemorrhage. They are typically composed of one or more feeding arteries; a nidus of varying size, shape, and compactness that is composed of abnormal vessels; and draining veins. They are sometimes associated with high-flow related aneurysms of the feeding arteries.
Patients typically present with headaches, neurologic deficit, seizures, or varying combinations of the three. Workup generally includes computed tomography (CT) and magnetic resonance imaging (MRI), demonstrating the lesion ( Fig. 68.4 ). Catheter angiography is then performed to define the vascular anatomy of the lesion and is used for treatment planning.

Treatment options include craniotomy with surgical resection of the lesion, embolization, and stereotactic radiosurgery (SRS). Commonly, more than one modality is used. SRS is usually reserved for compact lesions less the 2.5 cm in diameter. It can take up to 3 years for irradiated
AVMs to shut down after SRS. The Spetzler-Martin grading system was developed over 30 years ago and continues to be used to help make treatment decisions ( Table 68.2 ).
| Feature | Points |
|---|---|
| Nidus size (cm) | |
| Small (<3) | 1 |
| Medium (3-6) | 2 |
| Large (>6) | 3 |
| Eloquence of adjacent brain | |
| Noneloquent | 0 |
| Eloquent (sensorimotor, language, visual, thalamus, hypothalamus, internal capsule, brainstem, cerebellar peduncles, deep cerebellar nuclei) | 1 |
| Pattern of venous drainage | |
| Superficially only | 0 |
| Deep | 1 |
A cavernous malformation is a well-circumscribed, benign vascular lesion consisting of irregular thin-walled sinusoidal vascular channels located within the brain but lacking intervening neural parenchyma, large feeding arteries, or large draining veins. Characterized by McCormick in an autopsy series, these lesions have a prevalence of approximately 0.5%. There are three familial forms described. Because these are low-pressure, low-flow lesions, hemorrhage is typically not catastrophic unless it is in a highly eloquent area of the brain.
Patients usually present with hemorrhage or headaches with or without a history of new-onset seizures. Although it is often evident on CT, MRI is the imaging modality of choice and demonstrates a characteristic dark hemosiderin ring. Treatment for symptomatic lesions includes seizure control and surgical excision. Radiosurgery, while not necessarily curative, has been shown to decrease the rate of bleeding in lesions not amenable to surgical resection.
This lesion is composed of vascular channels with extremely thin walls similar to those of dilated capillaries. These are usually grouped in small clusters, generally with prominent intervening brain tissue. They are often clinically silent and generally do not appear on imaging studies. They are not evident on conventional catheter angiography unless they are large, and then only in the capillary venous phase. They clearly differ from an AVM in that flow through the lesion is not fast enough to demonstrate arteries and veins in the same conventional angiographic image. These lesions are typically not treated surgically.
These lesions are composed of an abnormally configured venous drainage system converging on a single, enlarged venous outflow channel. The typical appearance is that of a hydra, with radially converging veins. A characteristic feature of this lesion appears to be that the abnormal venous bed is poorly collateralized. The abnormal venous drainage may or may not be fully adequate to the needs of the brain tissue supplied. Slowly evolving degenerative changes in the brain tissue supplied can occur as a result, but, unfortunately, this is not helped by any known intervention. However inadequate, the venous anomaly represents the only venous drainage available to that area of brain, and therefore removal of the venous anomaly is not recommended. Doing so could lead to a venous infarction with swelling and hemorrhage, the consequences of which are particularly dangerous in the posterior fossa.
Both the internal carotid artery and vertebral artery enter the cranial cavity immediately after passing through a venous network. The internal carotid artery passes through the cavernous sinus, which communicates with the superior ophthalmic vein, petrosal sinus, and sphenoparietal sinus. The vertebral artery passes through a venous plexus at the occipital-C1 epidural space, which communicates with the jugular vein, epidural venous plexus, and paraspinal venous plexus. Trauma leading to a tear in the carotid or vertebral artery at its tether point passing through the skull base can lead to fistula with the surrounding venous plexus. The consequences may vary in severity and suddenness but typically include periorbital swelling, with proptosis and scleral edema in the case of the carotid-cavernous fistula (CCF) and prominent pulsatile bruit in the case of the vertebral-jugular fistula. Intraocular pressure measurement by tonometry can guide the urgency in treating CCF. On radiologic examination, dilation of the superior ophthalmic vein is characteristic ( Fig. 68.5 ). These lesions are usually treated by endovascular techniques. A catheter is advanced through the tear in the artery into the venous side of the fistula. The high flow and large fistulous channel facilitate this process. Embolic material, a coil, or a detachable balloon is then used to occlude the venous side of the fistula. When conventional transvenous routes fail, a direct approach through transorbital puncture may be required to provide endovascular therapy.
Aneurysms are an excessive localized enlargement of a vessel due to a weakening and subsequent defect in the wall of the artery. There is an adult prevalence of 2% (this varies according to the study) with an annual incidence of aneurysmal subarachnoid hemorrhage of 6 to 8/100,000 with peak age at 50 years. Modifiable risk factors for subarachnoid hemorrhage include hypertension, smoking, and excessive alcohol. Aneurysms may be further divided into saccular, fusiform, dissecting, infectious, and traumatic aneurysms.
As the name implies, these aneurysms, also referred to as berry aneurysms, are usually saccular in form and come off the vessel wall or at a bifurcation. Many of these are found incidentally, given the frequency of neuroimaging, but many present with hemorrhage. The classic presentation of subarachnoid hemorrhage due to a cerebral aneurysm is that of sudden onset of a headache, described as the “worst headache of my life.” Workup typically includes a CT scan, demonstrating a typical distribution of blood ( Fig. 68.6 ). Between 10% and 15% of subarachnoid hemorrhages from saccular aneurysms are fatal before the hospital is even reached. Of those individuals who reach a medical facility, one third do not survive (usually because of rebleeding), one third will survive with varying degrees of neurologic disability, and one third return to their baseline level.
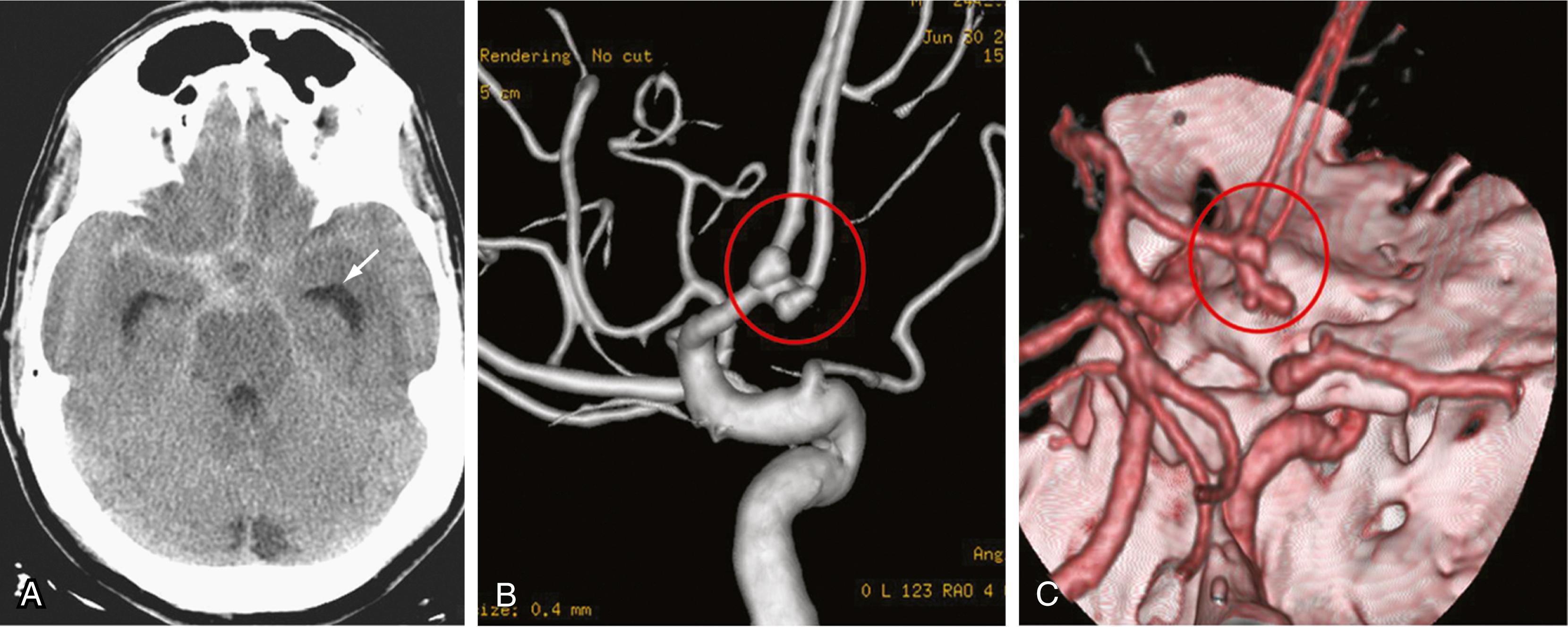
The major complications of subarachnoid hemorrhage include rebleeding, hydrocephalus (which is observed in around 15% to 20% of cases), cardiac events in around 50% of patients, vasospasm, hyponatremia, and seizures. Rebleeding is a major cause of death in patients who reach medical care after their initial episode of bleeding. Hydrocephalus is caused by disruption in the function of the arachnoid granulations and is commonly treated with an external ventricular drain. Vasospasm is a narrowing of the cerebral arteries thought to be caused by smooth muscle dysfunction related to blood breakdown products in the spinal fluid. When present, it can contribute to significant ischemia of the brain and, as such, be a significant cause of morbidity. Treatment of symptomatic vasospasm is typically multimodality and includes the use of hypervolemia and induced hypertension, endovascular procedures, and a variety of pharmacologic agents ( Box 68.2 ).
Subarachnoid blood removal
Prevention of dehydration and hypotension
Calcium channel blockers (nimodipine)
Intra arterial calcium channel blockers
Transluminal balloon angioplasty
Hypertension, hypervolemia, hemodilution
A classic study of cerebral aneurysms and their treatment documents the risk for rupture according to size and location as demonstrated in Table 68.3 . As a means of predicting possible vasospasm and overall outcomes, the Hunt and Hess clinical grading scale and the World Federation of Neurological Surgeons clinical grading scale were developed ( Tables 68.4 and 68.5 ). Another method of classifying subarachnoid hemorrhage is described by Fisher and colleagues and is based on CT scan imaging ( Table 68.6 ).
| Type Of Aneurysm | <7 mm and No Prior Sah | <7 mm and Prior Sah | 7–12 mm | 13–24 mm | >24 mm |
|---|---|---|---|---|---|
| Carotid-cavernous | 0 | 0 | 0 | 3.0 | 6.4 |
| Anterior circulation | 0 | 1.5 | 2.6 | 14.5 | 40.0 |
| Posterior circulation | 2.5 | 3.4 | 14.5 | 18.4 | 50.0 |
| Description | Grade | Good Outcome |
|---|---|---|
| Asymptomatic or minimal headache and slight nuchal rigidity | 1 | ≈70 |
| Moderate to severe headache, nuchal rigidity, ± cranial nerve palsy only | 2 | ≈70 |
| Drowsy, confusion, or mild focal deficit | 3 | ≈15 |
| Stupor, moderate to severe hemiparesis, possibly early decerebrate rigidity | 4 | ≈15 |
| Deep coma, decerebrate rigidity, moribund appearance | 5 | ≈0 |
| Grade | GCS Score | Motor Deficit |
|---|---|---|
| I | 15 | No |
| II | 13–14 | No |
| III | 13–14 | Yes |
| IV | 7–12 | Yes or no |
| V | 3–6 | Yes or no |
| Grade | CT Findings |
|---|---|
| I | No hemorrhage evident |
| II | Diffuse SAH with vertical layers <1 mm thick |
| III | Localized clots and/or vertical layers of SAH >1 mm thick |
| IV | Diffuse or no SAH but with intracerebral or intraventricular hemorrhage |
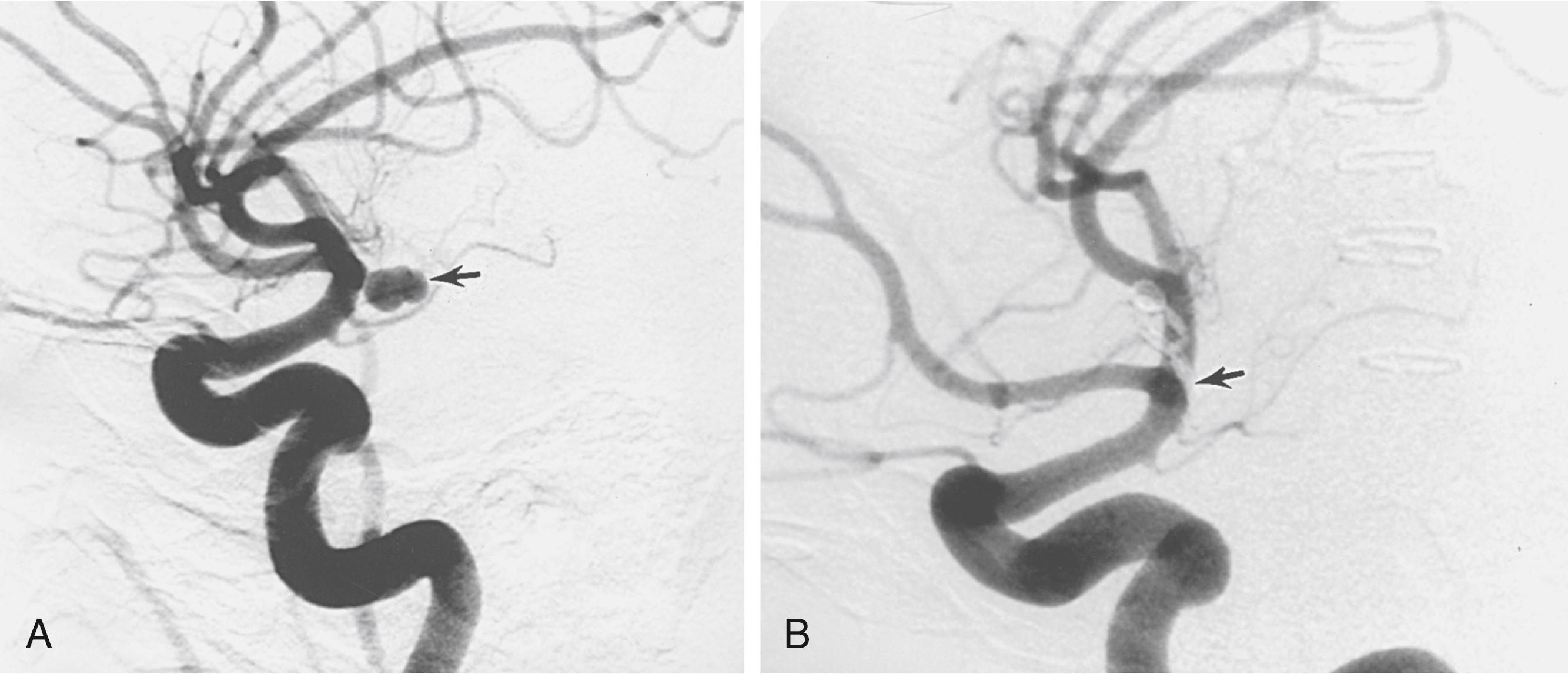
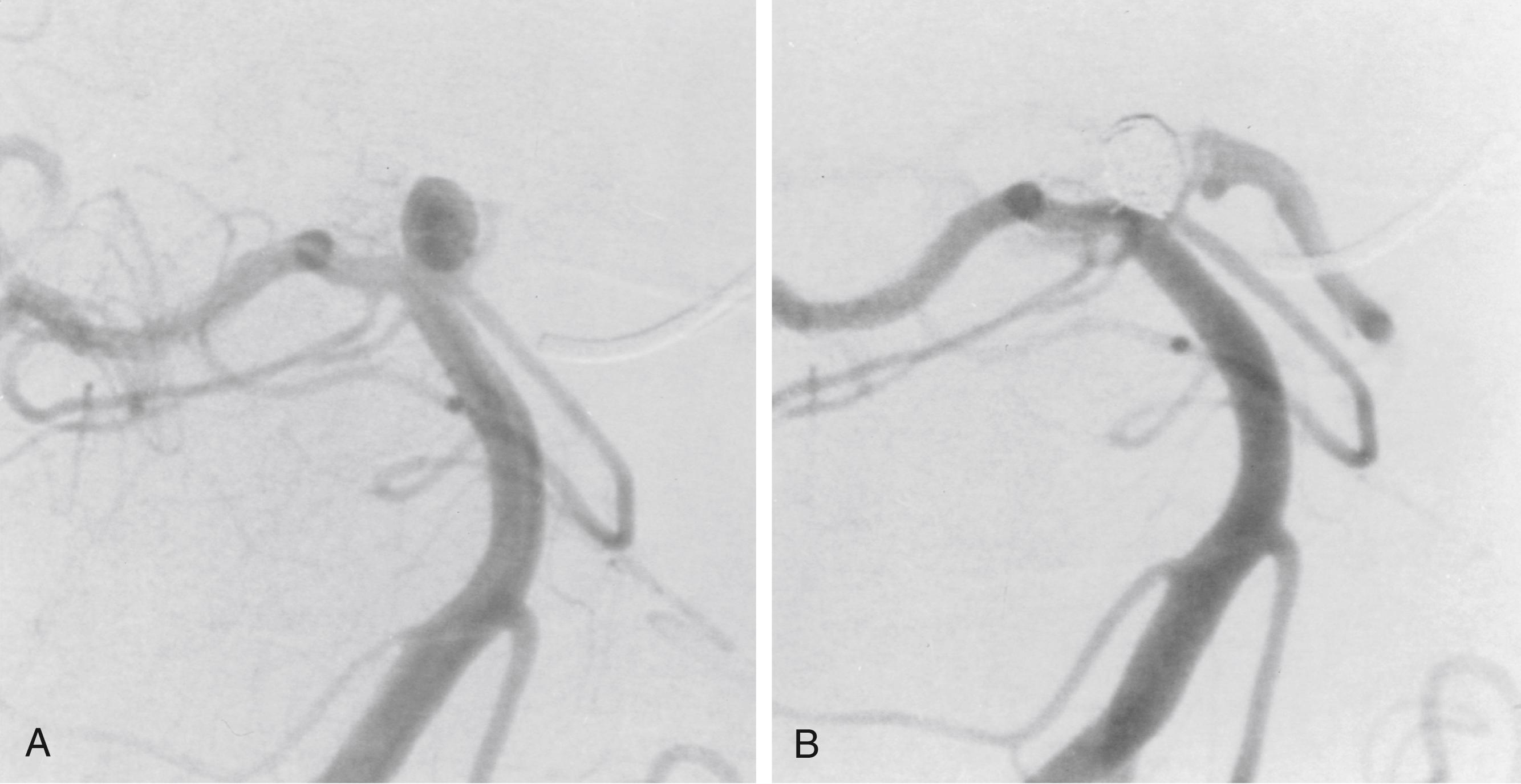
Treatment of saccular aneurysms consists of coiling, coiling through a stent, flow diversion stent , or surgical clipping ( Figs. 68.7 and 68.8 ) with subsequent intensive critical care unit management of potential vasospasm and comorbidities. Once the standard of care in the treatment of cerebral aneurysms, open craniotomy and clipping is now typically reserved for those lesions believed not to be amenable to endovascular techniques. , , It is generally believed that treatment of ruptured aneurysms is best accomplished as soon as possible after the initial hemorrhage.
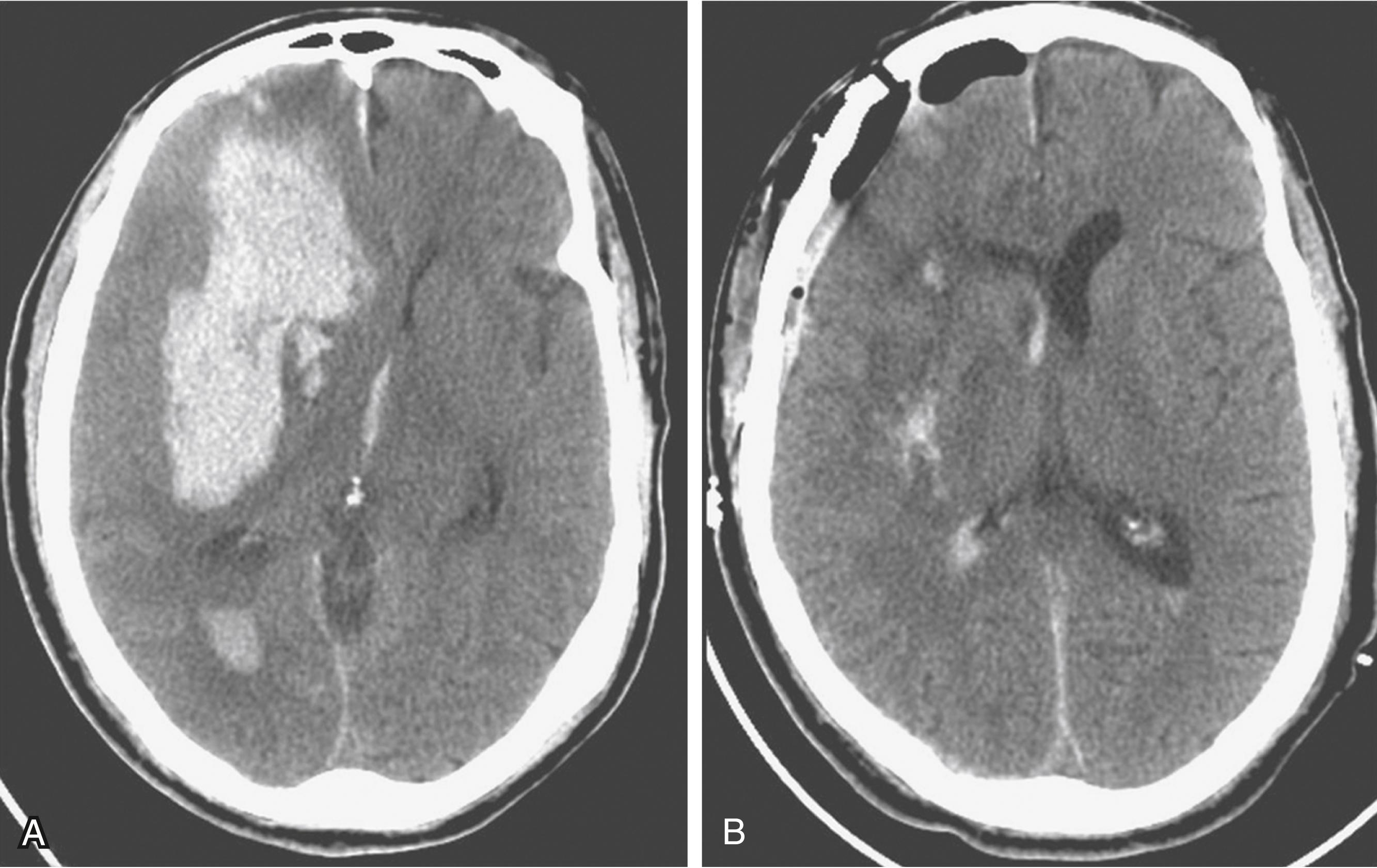
Spontaneous intracerebral hemorrhages into the brain parenchyma are common, accounting for approximately 10% of all strokes. They generally occur in older patients, usually because of degenerative changes in the cerebral vessels that are often associated with chronic hypertension ( Box 68.3 ). In younger patients, they are more likely related to drug abuse or vascular malformation. They can occur anywhere in the cerebral circulation or brainstem but are classically described in association with small degenerative aneurysms (Charcot-Bouchard aneurysms) of the perforating vessels and larger vessels at the skull base. They are typically located on the perforating lenticulostriate vessels, leading to hemorrhage in the basal ganglia. The clinical presentation of a stroke, with sudden-onset neurologic signs and symptoms corresponding to the area of brain affected. Symptoms are more likely to include headache than ischemic stroke. The diagnosis is with CT, usually done in an emergency department setting. The size and location of the acute hematoma are well visualized with CT, as is any associated brain shift or hydrocephalus ( Figs. 68.9A and 68.10 ). Older patients with a known history of hypertension present with a hematoma in the putamen, thalamus, cerebellum, or pons. Blood pressure control is essential in order to prevent rehemorrhage. Further investigation might be warranted with an atypical hematoma location or appearance, especially if there is any component of subarachnoid blood. Also, investigation is usually recommended for younger patients without known hypertension and those with a potential underlying cause for hemorrhage (e.g., history of neoplasm, blood dyscrasias, bacterial endocarditis).
Hypertension
Vascular anomaly
Cerebral aneurysm
Arteriovenous malformation
Cavernous malformation
Cerebral infarction (stroke) transformation
Cerebral amyloid angiopathy
Coagulopathy
Tumors
Drug abuse
Other
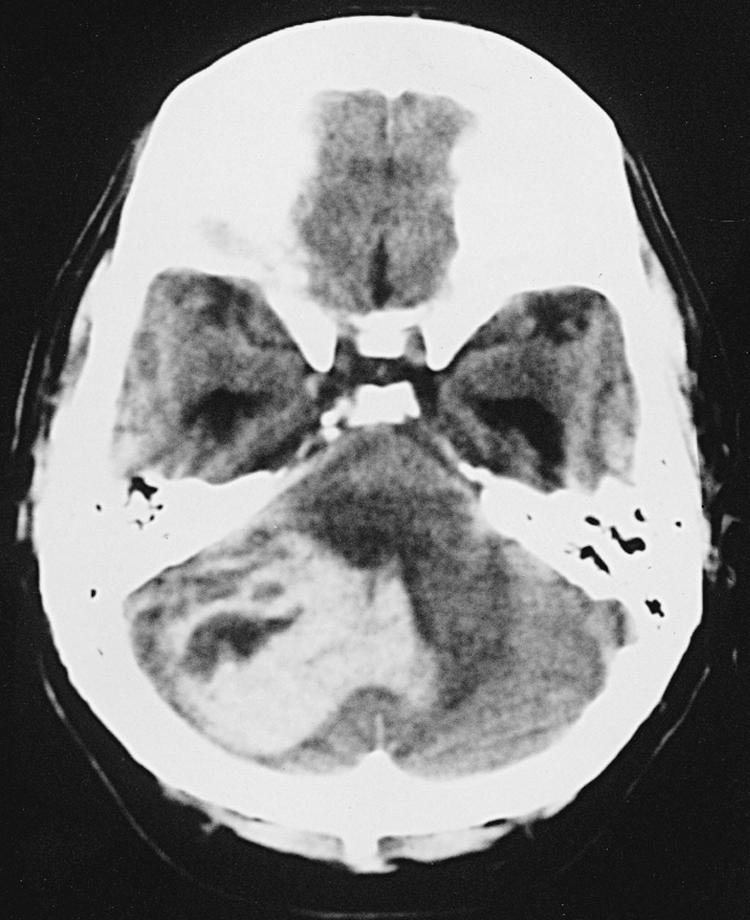
Further investigation is generally done with contrast MRI or magnetic resonance angiography. Any suggestion of aneurysm or AVM is followed by conventional catheter angiography. In older patients with a history of early dementia and multiple episodes of more peripherally located intracerebral hematomas, the diagnosis of amyloid angiopathy needs to be considered.
Most cases of spontaneous intracerebral hemorrhage do not require surgical intervention. Many hemorrhages are small and well tolerated. Patients who obey commands and can be monitored by changes in their neurologic examination can generally be managed conservatively with hospital observation for at least 5 to 7 days. Peak swelling and decompensation are probably most likely to occur within that time frame. Surgery for evacuation of the hematoma may be appropriate in a small group of patients with intermediate-sized hemorrhages in accessible locations who appear to tolerate the hematoma initially but then deteriorate in a delayed fashion with edema, despite medical therapy. Steroids have not demonstrated benefit. Attempts to predict which patients will deteriorate solely on the basis of hematoma volume have been frustrated by the broad spectrum of intracranial compliance exhibited by different patients. In general, younger patients with smaller ventricles and small subarachnoid spaces have a lower compliance, with lower tolerance, than older patients with cerebral atrophy and generous ventricles and subarachnoid spaces.
The Surgical Trial in Intracerebral Hemorrhage has noted a lack of clinical outcome difference in comparing early surgery with conservative management. If indicated, surgical evacuation is usually done by craniotomy over the most accessible part of the hematoma ( Fig. 68.9B ). Intraoperative ultrasound is often helpful in finding hematomas that do not quite come to the cortical surface and in monitoring the progress of the evacuation. The goal of surgery is decompression rather than complete removal, but it is generally done as far as safely practical. Minimally invasive techniques continue to evolve. Stereotactic aspiration and methods with fibrinolytic agents are being developed and may be a consideration for patients with hematomas in deep locations that are otherwise difficult to access.
A special situation to consider is the patient with cerebellar hemorrhage ( Fig. 68.10 ). Surgery is offered more readily in these cases because the danger of sudden deterioration from brainstem compression is more of a concern and because even extensive damage to the cerebellum itself is generally survivable with good functional outcome. Patients with fourth ventricular obstruction and hydrocephalus from cerebellar hemorrhage can sometimes be treated with ventricular drainage alone but are usually offered surgical evacuation of the hematoma by suboccipital craniotomy because of the risk for brainstem compression.
These aneurysms are associated with a systemic infection capable of showering small particles of bacteria-infected material into the cerebral vascular bed. Subacute bacterial endocarditis and some pulmonary infections can do this. A distinguishing feature of these aneurysms is that they are generally found more distal in the cerebral vascular bed, as opposed to berry aneurysms, which are usually found on larger vessels near the circle of Willis. There can also be many of them. When the bacterial emboli lodge in distal cerebral arterial branches, they can erode through the wall of these smaller vessels, often creating a hemorrhage contained by the perivascular tissue. Maximal antibiotic treatment is essential at the outset. The presence of an intracerebral hematoma may force immediate craniotomy for evacuation. Operation on the aneurysm at this early stage often reveals a component of subarachnoid hemorrhage and an early inflammatory reaction in the subarachnoid space, with only a blood collection covering the erosion defect in the wall of the small artery. Attempts to dissect and to define a neck are frustrated by a lack of developed fibrous tissues, and intraoperative hemorrhage is then common. Typically, the diseased arterial segment must be occluded and resected when it is operated on in this early stage. The need for arterial bypass to maintain blood flow to critical cerebral areas should be anticipated, but this is not always possible.
If the mycotic aneurysms are discovered or treated at some later stage, a fibrous wall to the aneurysm may have had time to develop, and clipping can then be a possibility. However, the surgeon needs to be forewarned that it may be difficult to find the aneurysm in a distal location, often buried deep in a cerebral sulcus thickened with reactive fibrous scar tissue.
Moyamoya disease is a cerebrovascular disorder that is characterized by an idiopathic nonatherosclerotic narrowing or occlusion of major intracranial blood vessels with the development of a conspicuous compensatory collateral rete vessel network, which allows continued cerebral perfusion around the occluded or severely narrowed segment. The disorder is usually bilateral, although not necessarily exactly symmetrical. Although generally rare, the disease is more common in persons of Asian ancestry and was first recognized from cases studied with angiography in Japan before the advent of CT and MRI. The term moyamoya comes from the Japanese word for “puff of smoke” or mist. The actual disease is sometimes confused with the less conspicuous collateral vascular networks seen around severe narrowing of common atherosclerotic origin in persons of Western origin. In the juvenile form, moyamoya typically is manifested as cognitive decline, with deteriorating school performance and evidence of multiple infarcts. Angiography reveals the internal carotid artery, proximal middle cerebral artery, or proximal anterior cerebral artery with severe narrowing or occlusion and, generally, multiple clusters of fine collateral vessels. In the adult form of moyamoya disease, the rete vessels cause subarachnoid or basal ganglia hemorrhage, the most common presentations. The hemorrhage can usually be treated conservatively. Some form of extracranial to intracranial bypass is generally attempted to take the load off the collateral vascular network. Surgical treatment consists of extracranial intracranial bypass (typically superficial temporal to middle cerebral artery), onlay synangiosis, or a combination of the two. EC-IC bypass is typically preferred in the adult form of the disease, while children tend to respond better to synangiosis.
Dural AVMs consist of the abnormal communication between dural arteries and the cerebral venous system. The lesions seem to occur only in adults and are probably acquired lesions that follow a dural sinus thrombosis, usually of the cavernous sinus or sigmoid-transverse sinus junction area. With subsequent healing, the thrombosed segment triggers a neovascular response that evolves to an AVM configuration with fistulous channels that can gradually enlarge. Lesions demonstrating communication with cortical veins pose a risk of hemorrhage and require treatment. This is typically done with endovascular techniques, although open surgical resection is sometimes necessary.
The past decade has witnessed an impressive body of basic science and clinical research focused on ischemic stroke. The continued prevalence, morbidity, disability, and subsequent cost to society has prompted the creation of primary and comprehensive stroke centers. Today, neurosurgeons play a critical role in the care and management of patients suffering from ischemic stroke. This involvement consists of (1) endovascular treatment of ischemic stroke and (2) decompressive craniectomy to decrease ICP and decrease morbidity and mortality in patients with brain infarction.
Endovascular treatment of acute stroke consists of catheter angiography and clot retrieval, intraarterial clot lysis, and stent placement. Numerous recent clinical trials have demonstrated to clinical utility of aggressive early intervention in the treatment of acute stroke. ,
When patients do not make it to care within the allotted time window, or when catheter intervention is unsuccessful in reestablishing perfusion, they often go on the brain infarction which is often followed by subsequent swelling. In older patients with brain atrophy, this is often well tolerated. In younger patients or those with full brains, this brain swelling can be life threatening. Many of these patients may be candidates for decompressive craniectomy, wherein a large portion of the bone over the affected side is removed and placed in the freezer or implanted in the abdominal fat. The dura is opened and the brain allowed to swell out, decreasing ICP. Once the swelling goes down, the bone flap can be replaced in a second procedure. Guidelines regarding the use and utility of decompressive craniectomy in the setting of fulminant brain swelling in the setting of infarction continue to evolve.
Intracranial tumors can be classified as primary versus secondary, as pediatric versus adult, by cell of origin, or by location in the nervous system. Primary tumors arise from tissues in the nervous system, whereas secondary tumors originate from tissues outside the nervous system and metastasize secondarily to the brain. They may represent local extension of regional tumors, such as chordoma or scalp cancer, but usually reach the nervous system through the hematogenous route.
According to the Central Brain Tumor Registry of the United States (CBTRUS), the overall incidence of primary brain tumors was 23.03/100,000 between 2011 and 2015. Of the 1.7 million cases of new cancers diagnosed in a given year, between 100,000 and 240,000 are expected to eventually metastasize to the brain.
The clinical manifestations of various brain tumors can be divided into those caused by focal compression and dysfunction caused by the tumor itself and those attributed to secondary consequences, namely, increased ICP, peritumoral edema, and hydrocephalus. Usually, symptoms are caused by a combination of these factors.
The clinical presentation does not differ much by tumor histology; rather, rate of growth and location of the tumor contribute to the clinical features. A meningioma peripherally located in a relatively silent area of the brain, with a slow rate of growth, may enlarge to a significant size in a neurologically intact patient because the brain can accommodate to a slowly growing lesion. On the other hand, a very small metastatic lesion in the sensorimotor strip can present early with seizures.
Headache occurs in 50% to 60% of primary brain tumors and in 35% to 50% of metastatic tumors. It is classically described as being worse in the morning, probably because of hypoventilation during sleep, with consequent elevation of the P co 2 and cerebrovascular dilation. Seizures may be the first symptom of a brain tumor. Patients older than 20 years presenting with a new-onset seizure are aggressively investigated for a brain tumor.
Infratentorial lesions may be manifested with headache, nausea and vomiting, cerebellar signs and symptoms, vertigo, and cranial nerve deficits. Supratentorial lesions may be manifested with different symptoms, depending on the location. Frontal lobe lesions present with personality changes, dementia, hemiparesis, or dysphasia. Temporal lobe lesions may be manifested with memory changes, auditory or olfactory hallucinations, or contralateral quadrantanopia. Patients with parietal lobe lesions may develop contralateral motor or sensory impairment, apraxia, and homonymous hemianopia, whereas those with occipital lobe lesions may show contralateral visual field deficits and alexia.
The initial workup generally involves a CT scan of the brain. CT provides a rapid means of evaluating changes in brain density, calcifications, acute hemorrhage (<48 hours old), and skull lesions. MRI of the brain is the modality of choice for diagnosis, presurgical planning, and posttherapeutic monitoring of brain tumors. Gadolinium contrast enhancement with MRI is more sensitive in demonstrating defects in the blood-brain barrier and localizing small metastases (up to 5 mm). Advances in MRI techniques have evolved from strictly morphology-based imaging to a modality that encompasses function, physiology, and anatomy. Diffusion-weighted imaging can help distinguish between gliomas and abscesses, and perfusion-weighted imaging can predict response to radiotherapy in low-grade gliomas. Functional MRI can be used in planning of surgery for tumors in eloquent areas of the brain to enable radical resection with less morbidity. Diffusion tensor imaging can demonstrate the effect of a tumor on white matter tracts. Magnetic resonance angiography is used more routinely as a noninvasive modality to evaluate the vascularity of a tumor or anatomic relationship of a tumor to normal cerebral vasculature.
Dexamethasone is recommended for the management of edema associated with tumors, given its propensity to reduce peritumoral edema by stabilizing the cell membrane. An antiepileptic drug is also recommended for patients presenting with seizures or in those with tumors close to the sensorimotor strip.
Technical advances have made tumor surgery safer and more effective. The intraoperative microscope provides superior illumination and magnification, thereby allowing the surgeon to resect tumors from critical areas through small cranial openings. The cavitational ultrasonic surgical aspirator simultaneously breaks up and sucks away firm tumors while protecting vital neural and vascular structures. Intraoperative ultrasonography provides real-time imaging of tumors and cysts in subcortical and deep areas of the brain. Intraoperative CT or MRI is standard practice in some centers, enabling on-table imaging of the extent of resection ( Fig. 68.11 A ). CT and MRI also allow real-time visualization of a biopsy needle within a target. Image-guided (CT or MRI) frameless surgical navigation allows instant and accurate localization of the tip of a probe during a craniotomy by displaying that point on a preoperative CT or MRI scan ( Fig. 68.11 B ).
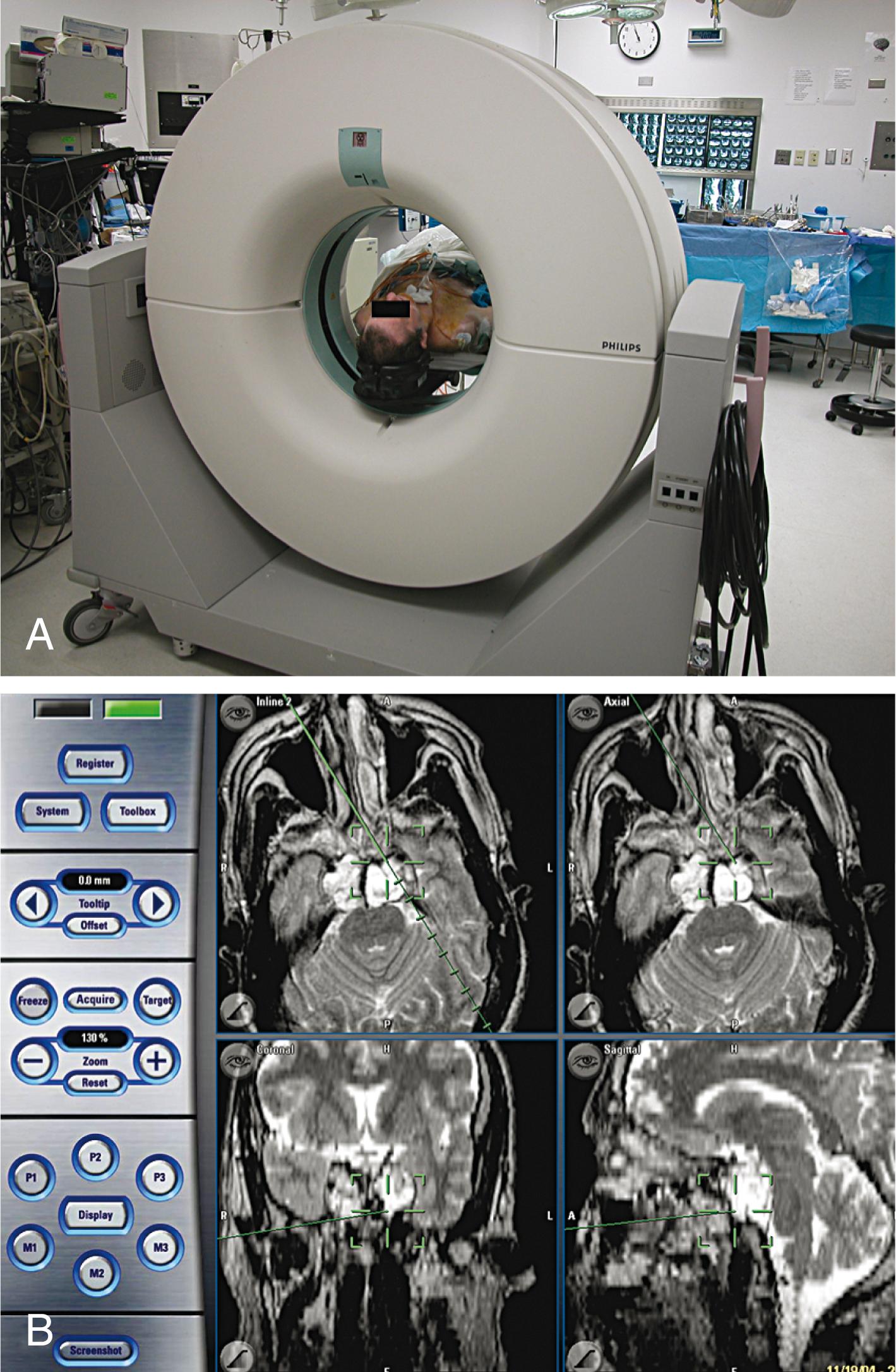
The primary goals of operation include histologic diagnosis and reduction of mass effect by removal of as much tumor as is safely possible to preserve neurologic function. The decision between a needle biopsy and more radical surgical resection depends on the location and size of the tumor, its sensitivity to radiation or chemotherapy, the preoperative Karnofsky performance score of the patient, and the systemic status of the primary cancer in case of metastatic brain lesions.
Primary tumors of the brain are divided into intraaxial (those arising from within the brain parenchyma) and extraaxial (those arising from outside the brain parenchyma).
Most primary intraaxial brain tumors develop from the glial cells, or supportive structures, of the neurons and are collectively called gliomas. Total surgical resection of gliomas is extremely rare because of their ability to infiltrate widely along the white matter tracts and to cross the corpus callosum into the contralateral hemisphere. Radiation therapy and chemotherapy options vary according to the histology of the brain tumor. Biologic therapies and the molecular biology of gliomas are an area under intensive laboratory and clinical research. An ideal therapy will target rapidly, growing malignant glioma cells along with infiltrating tumor cells with minimal toxicity to normal cells. This requires that the therapeutic vehicle of choice have access to all cells in the brain and be able to distinguish invasive or quiescent tumor cells from normal cells.
The current histopathologic classification of brain tumors was recently updated by the World Health Organization (WHO). WHO classifies intraaxial brain tumors by cell type and grades them on a scale of I to IV based on light microscopy characteristics that include the degree of cellularity, pleomorphism, mitotic figures, endothelial proliferation, and necrosis. The higher the grade, the more aggressive and malignant the tumor.
An exhaustive review of neurooncology is beyond the scope of this chapter. What follows is an overview of the most commonly encountered and unique tumors of the central and peripheral nervous systems.
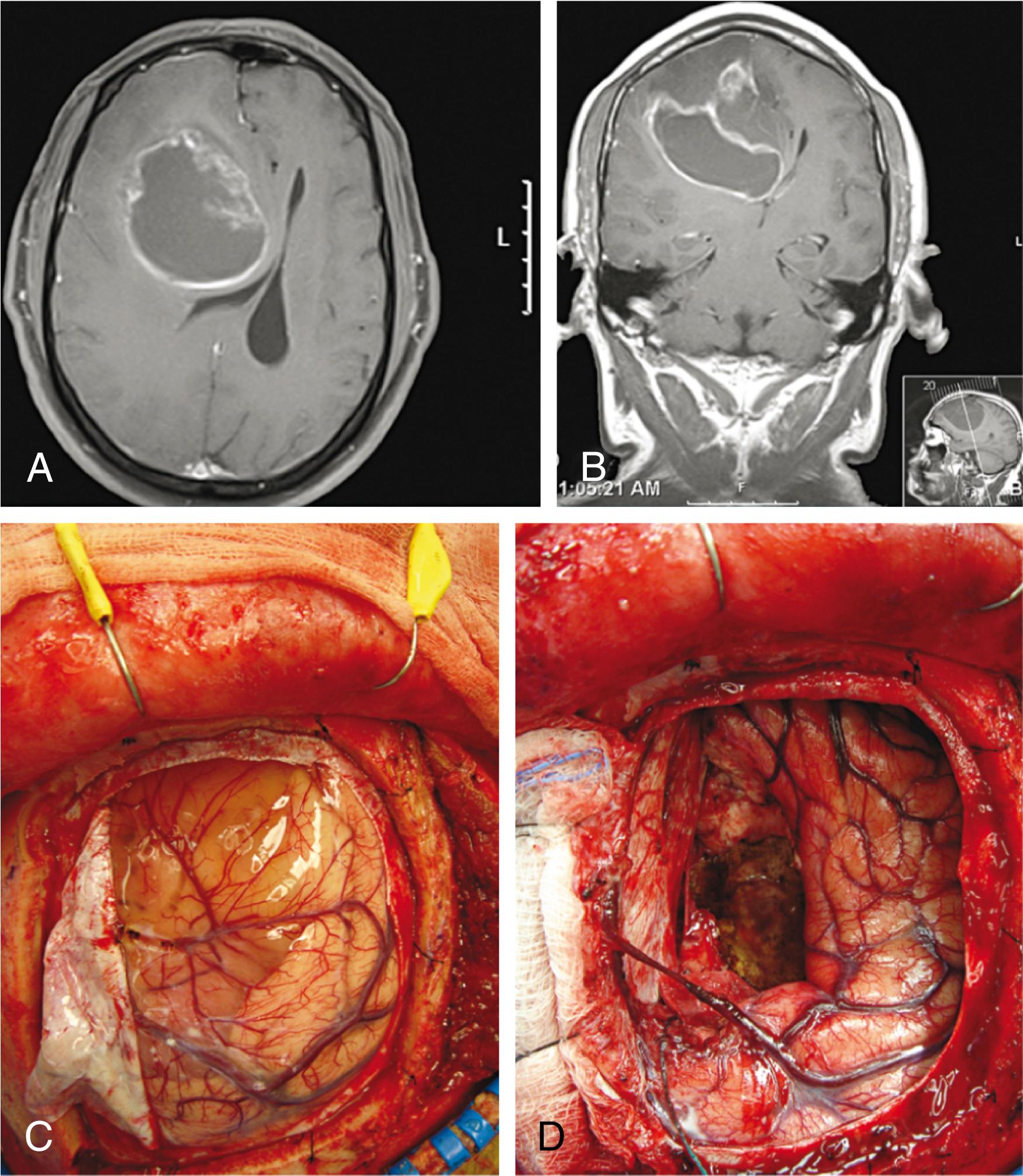
A WHO grade IV tumor, this is the most commonly encountered primary brain tumor in adults. Presentation can include headache, seizures, focal deficits, and personality changes. Imaging usually demonstrates a ring-enhancing lesion with surrounding edema and mass effect ( Fig. 68.12 ). Treatment of accessible lesions involves attempted gross total resection and postoperative radiation therapy and chemotherapy. Temozolomide is the current drug of choice and has been shown to improve both survival and quality of life. Recurrence is common, and repeated surgical resection is often deemed reasonable.
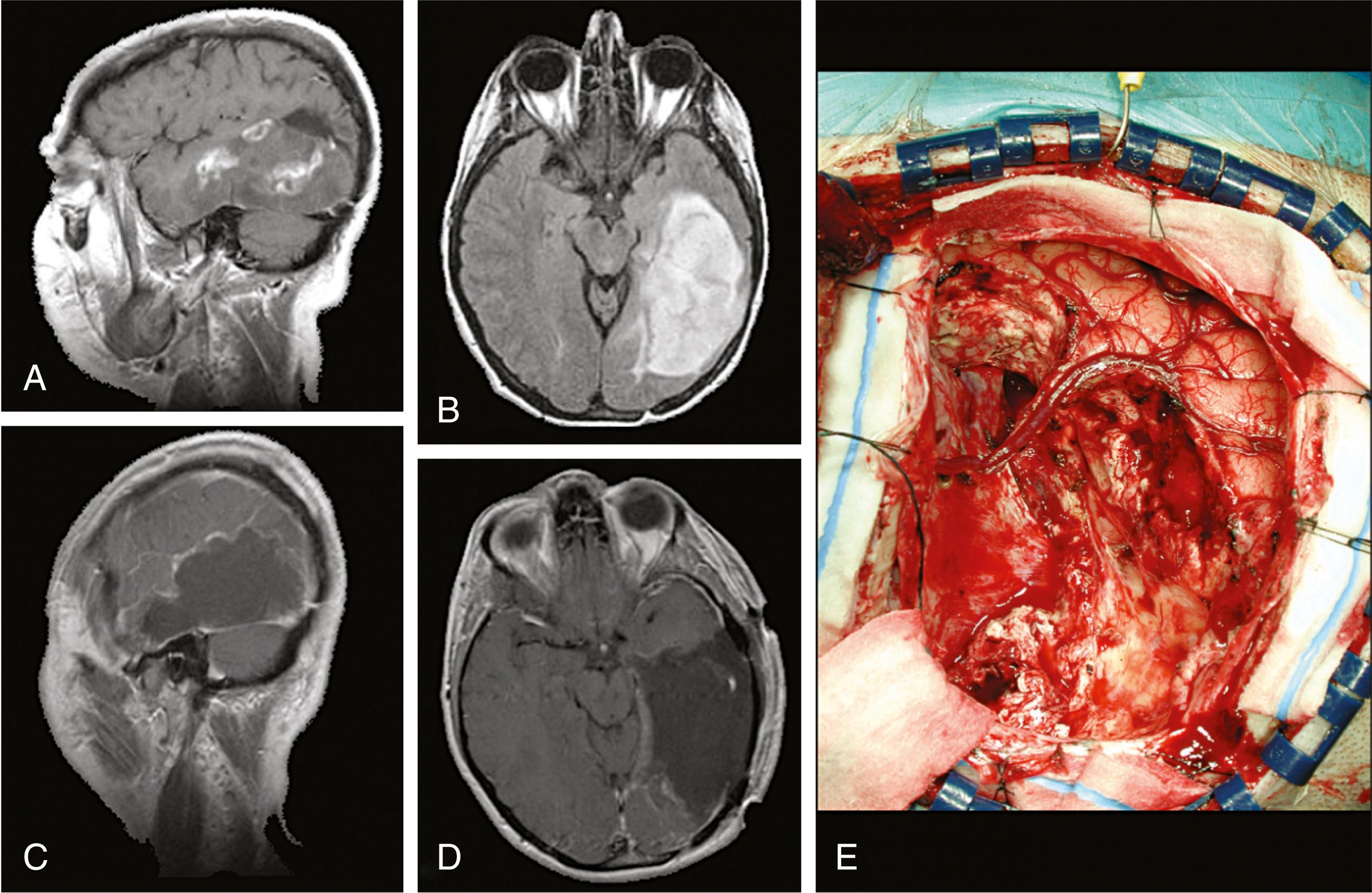
A WHO grade III tumor, this presents much the same as glioblastoma multiforme, with similar treatment but slightly better long-term prognosis ( Fig. 68.13 ).
Pilocytic astrocytomas are typically classified as WHO grade I gliomas. When arising in the posterior fossa, they can cause obstructive hydrocephalus and cerebellar signs on examination. Surgical resection is the treatment of choice for these posterior fossa lesions. However, for lesions in the hypothalamus or optic tract, biopsy and chemotherapy or radiation therapy should be considered.
These tumors originate from oligodendroglial cells and represent 25% of all glial tumors; they occur with a male to female predominance of 3:2, observed at an average age of 40 years. They often present clinically with seizures or hemorrhage and nonspecific mass effect. A 5-year survival rate can be observed between 40% and 70%, depending on the grade, with an overall median survival of 3 years. Another form known as oligoastrocytoma behaves like oligodendroglioma, and both have aggressive anaplastic forms.
Treatment consists of surgical resection followed by chemotherapy. A particularly favorable response rate is associated with tumors that show allelic losses of chromosomes 1p and 19q. Radiation therapy is considered for tumors with anaplastic transformation.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here