Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Dizziness is a term patients use to describe a variety of symptoms including spinning or movement of the environment (vertigo), lightheadedness, presyncope, or imbalance. Patients may also use the term for other sensations such as visual distortion, internal spinning, nonspecific disorientation, and anxiety.
Patients may experience dizziness in isolation or with other symptoms. Neurological causes should be considered when other neurological signs and symptoms are present and also whenever specific peripheral vestibular or general medical disorders have not been identified. It is critical to ask the patient about associated symptoms, since they may be the key to diagnosis. Vertigo , a sensation of spinning of the environment, suggests a lesion within the vestibular pathways, either peripheral or central. Associated ear symptoms such as hearing loss and tinnitus can suggest a peripheral localization (i.e., inner ear, eighth nerve). Many different types of hearing loss occur with or without dizziness, and an understanding of common auditory disorders is important to the practicing neurologist. With an understanding of the neuro-otological bedside examination, specific findings can often be identified.
In this chapter, we provide background information regarding dizziness, vertigo, and hearing loss and the clinical information necessary for making specific diagnoses. We also include details on testing and management of these patients.
In 1861, Prosper Meniere was the first to recognize the association of vertigo with hearing loss and thus to localize the symptom to the inner ear ( ). Caloric testing, the most widely used test of the vestibulo-ocular reflex (VOR), was introduced by Robert Barany in 1906. He was later awarded the Nobel Prize for proposing the mechanism of caloric stimulation. Barany also provided the first clinical description of benign paroxysmal positional vertigo (BPPV) in 1921. Endolymphatic hydrops was identified in postmortem specimens of patients with Meniere disease in 1938. A method for measuring eye movements in response to caloric and rotational stimuli (electronystagmography [ENG]) was introduced in the 1930s, and in the 1970s digital computers began to be used to quantify eye movement responses.
Neuroimaging in the late 1970s and 1980s greatly expanded our understanding of causes of dizziness and vertigo. Prior to this time, stroke was considered an exceedingly rare cause of vertigo ( ). However, we now know that cerebellar and brainstem infarctions are identified in about 10%–15% of acute dizziness presentations that do not have obvious other central findings (Kerber, 2015). Imaging studies continue to lead to new discoveries of causes of vertigo, as demonstrated by the recently described disorder of superior canal dehiscence (SCD). But the most common causes of vertigo—Meniere disease, BPPV, and vestibular neuritis—still have no identifiable imaging characteristics.
Over the past 25 years, our understanding of the mechanisms for the common neuro-otological disorders has been greatly enhanced. BPPV can now be readily identified and cured at the bedside with a simple positional maneuver, and variants have also been described (Bhattacharyya, 2017). The head-thrust test can be used at the bedside to identify a vestibular nerve lesion, and because of this it has particular utility in helping distinguish vestibular neuritis from a posterior fossa stroke ( ). Controversies regarding Meniere disease have been clarified, and medical and surgical treatments have improved ( ). It is now clear that patients with recurrent episodes of vertigo without hearing loss, a condition once called vestibular Meniere disease , do not actually have Meniere disease.
Migraine is now recognized as an important cause of dizziness, even in patients without simultaneous headaches (Lempert, 2012). In fact, benign recurrent vertigo (BRV; patients with recurrent episodes of vertigo without accompanying auditory symptoms or other neurological features) is usually a migraine equivalent ( ). A more detailed description of the rotational vertebral artery syndrome has led to appreciation of the high metabolic demands of the inner ear and its susceptibility to ischemia ( ). Genetic research has identified ion channel dysfunction in disorders such as episodic ataxia (EA) and familial hemiplegic migraine, and patients with these disorders also commonly report vertigo ( ). It is hoped that identifying specific genes causing vertigo syndromes will lead to a better understanding of the mechanisms and also create the opportunity to develop specific treatments in the future.
Dizziness is a very common symptom. About 15%–20% of the adult US population reports problems with dizziness within a 12-month period (Kerber, 2017). Though most people report nonspecific types of dizziness, more than one-third report vertigo. Dizziness is more common among females and older people and has important healthcare utilization implications because up to 80% of patients with dizziness seek medical care at some point. In the United States, the National Centers for Health Statistics report 7.5 million annual ambulatory visits to physician offices, hospital outpatient departments, and emergency departments (EDs) for dizziness, making it one of the most common principal complaints ( ).
Hearing loss affects approximately 16% of adults (age >18 years) in the United States ( ). Men are more commonly affected than women, and the prevalence of hearing loss increases dramatically with age, so that by age 75, nearly 50% of the population reports hearing loss, which is a common cause of disability. The most common type of hearing loss is sensorineural, and both idiopathic presbycusis and noise-induced forms are common etiologies. Bothersome tinnitus is less frequent in the US population, with about 3% reporting it, although this increases to about 9% for subjects older than 65 ( ). The most common type of tinnitus is a high-pitched ringing in both ears.
The inner ear is composed of a fluid-filled sac enclosed by a bony capsule with an anterior cochlear part, central chamber (vestibule), and a posterior vestibular part ( Fig. 22.1 ). Endolymph fills up the fluid-filled sac and is separated by a membrane from the perilymph. These fluids primarily differ in their composition of potassium and sodium, with the endolymph resembling intracellular fluid with a high potassium and low sodium content, and perilymph resembling extracellular fluids with a low potassium and high sodium content. Perilymph communicates with the cerebrospinal fluid (CSF) through the cochlear aqueduct.
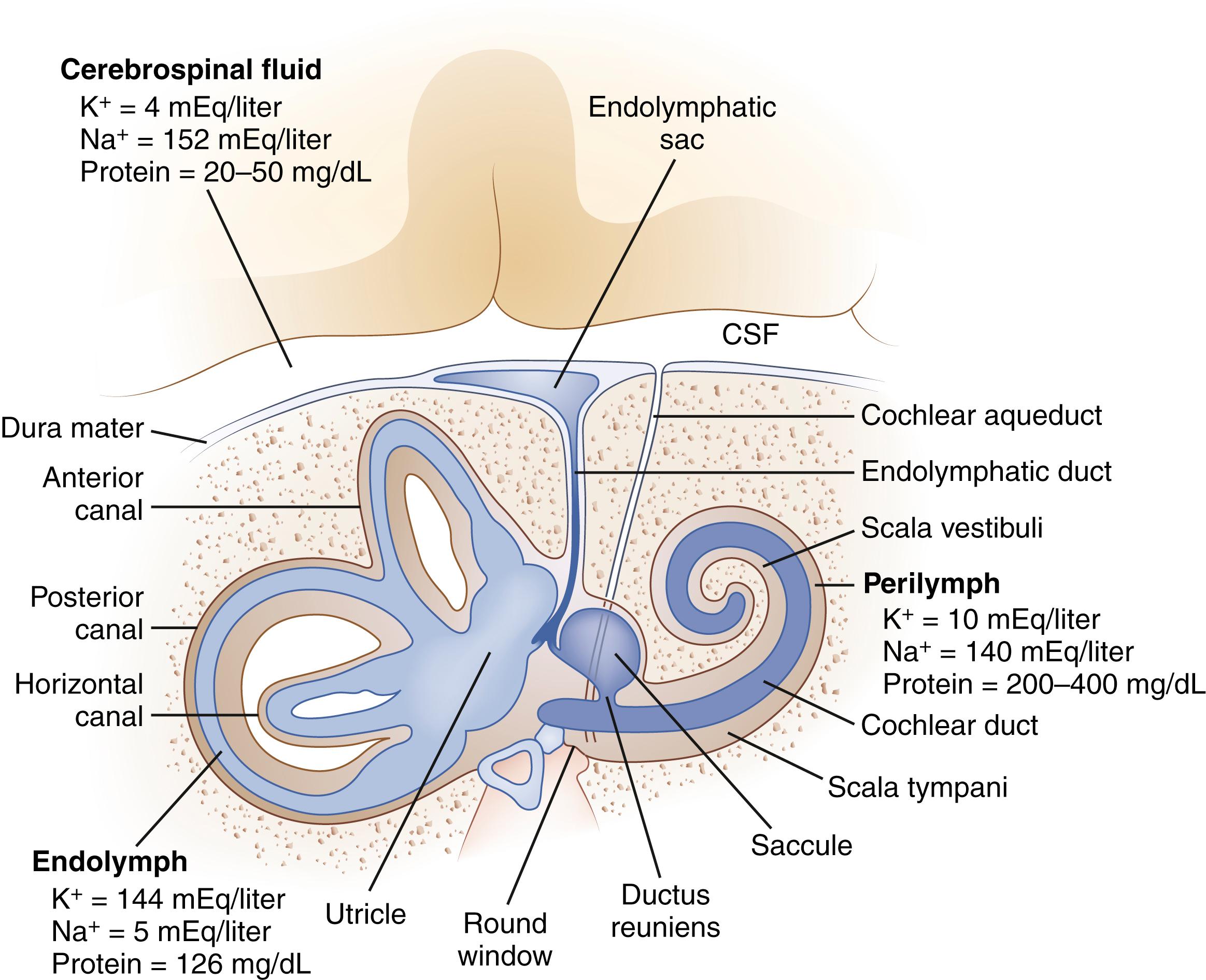
The cochlea senses sound waves after they travel through the external auditory canal and are amplified by the tympanic membrane and ossicles of the middle ear ( ). The stapes, the last of three ossicles in the middle ear, contacts the oval window, which directs the forces associated with sound waves along the basilar membrane of the cochlea. These forces stimulate the hair cells, which in turn generate neural signals in the auditory nerve. The auditory nerve enters the lateral brainstem at the pontomedullary junction and synapses in the cochlear nucleus. The trapezoid body is the major decussation of the auditory pathway, but many fibers do not cross to the contralateral side. Signals then travel to the superior olivary complex. Some projections travel from the superior olivary complex to the inferior colliculus through the lateral lemnisci, and others terminate in one of the nuclei of the lateral lemniscus. Next, fibers travel to the ipsilateral medial geniculate body, and then auditory radiations pass through the posterior limb of the internal capsule to reach the auditory cortex of the temporal lobe.
The peripheral vestibular system is composed of three semicircular canals, the utricle and saccule, and the vestibular component of the eighth cranial nerve ( ). Each semicircular canal has a sensory epithelium called the crista ; the sensory epithelium of the utricle and saccule is called the macule . The semicircular canals sense angular movements, and the utricle and saccule sense linear movements. Two of the semicircular canals (anterior and posterior) are oriented in the vertical plane nearly orthogonal to each other; the third canal is oriented in the horizontal plane (horizontal canal). The crista of each canal is activated by movement occurring in the plane of that canal. When the hair cells of these organs are stimulated, the signal is transferred to the vestibular nuclei via the vestibular portion of cranial nerve VIII. Signals originating from the horizontal semicircular canal then pass via the medial longitudinal fasciculus along the floor of the fourth ventricle to the abducens nuclei in the middle brainstem and the ocular motor complex in the rostral brainstem. The anterior (also referred to as the superior ) and posterior canal impulses pass from the vestibular nuclei to the ocular motor nucleus and trochlear nucleus, triggering eye movements roughly in the plane of each canal. A key feature is that once vestibular signals leave the vestibular nuclei they divide into vertical, horizontal, and torsional components. As a result, a lesion of central vestibular pathways can cause a pure vertical, pure torsional, or pure horizontal nystagmus.
The primary vestibular afferent nerve fibers maintain a constant baseline firing rate of action potentials (APs). When the baseline rate from each ear is symmetrical (or an asymmetry has been centrally compensated), the eyes remain stationary. With an uncompensated asymmetry in the firing rate, resulting from either increased or decreased activity on one side, slow ocular deviation results. By turning the head to the right, the baseline firing rate of the horizontal canal is physiologically altered, causing an increased firing rate on the right side and a decreased firing rate on the left side ( Fig. 22.2 ). The result is a slow deviation of the eyes to the left. In an alert subject, this slow deviation is regularly interrupted by quick movements in the opposite direction (nystagmus), so the eyes do not become pinned to one side. In a comatose patient, only the slow component is seen because the brain cannot generate the corrective fast components (“doll’s eyes”).
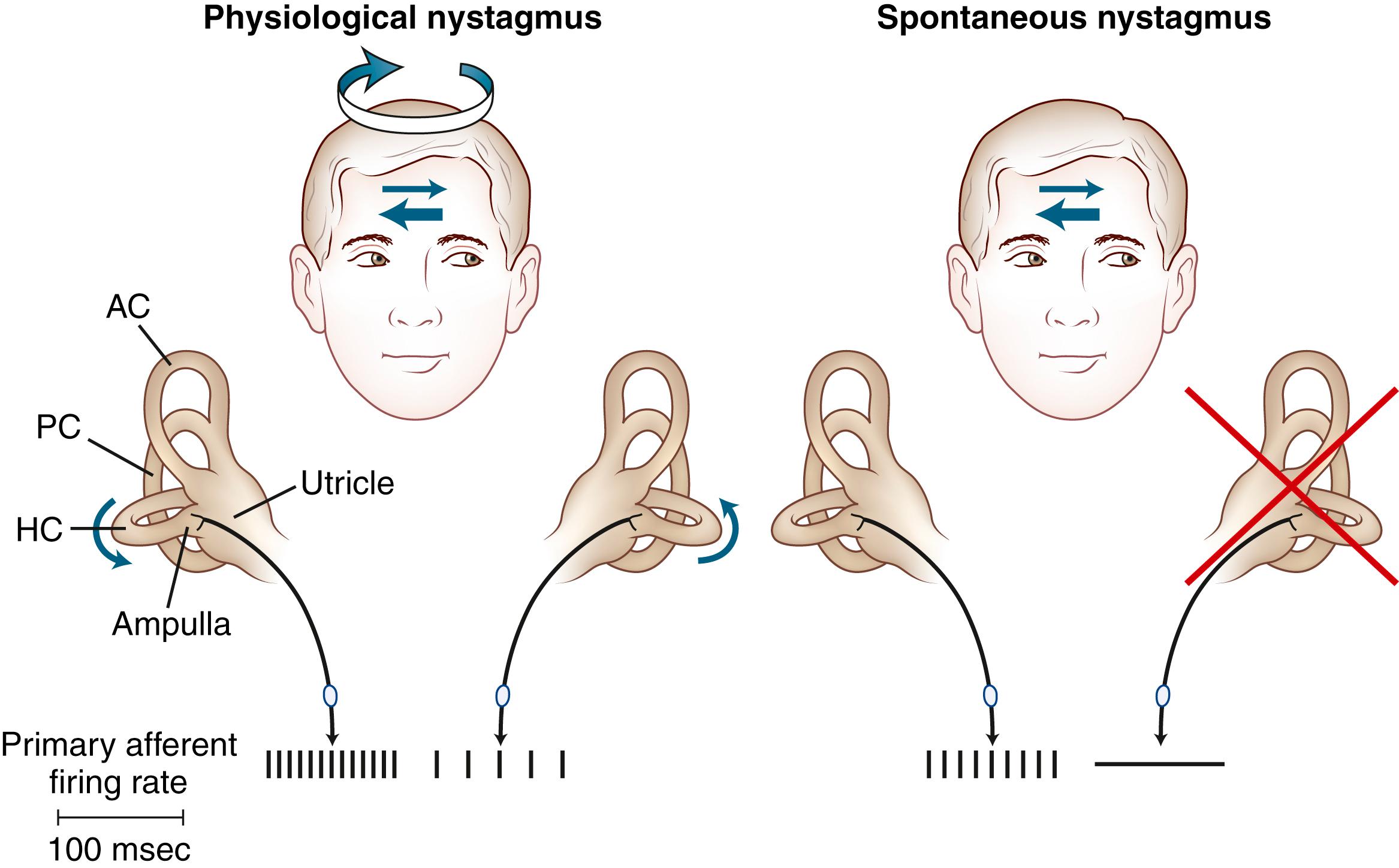
The plane in which the eyes deviate as a result of vestibular stimulation depends on the combination of canals that are stimulated ( Table 22.1 ). If only the posterior semicircular canal on one side is stimulated (as occurs with BPPV), a vertical-torsional deviation of the eyes can be observed, which is followed by a fast corrective response generated by the conscious brain in the opposite direction. However, if the horizontal canal is the source of stimulation (as occurs with the horizontal canal variant of BPPV [HC-BPPV]), a horizontal deviation with a slight torsional component (because this canal is slightly off the horizontal plane) results. If the vestibular nerve is lesioned (vestibular neuritis) or stimulated (vestibular paroxysmia), a horizontal greater than torsional nystagmus is seen that is the vector sum of all three canals—the two vertical canals on one side cancel each other out.
| Localization | Component(s) | Triggered Eye Movements | Common Clinical Conditions | Localizing Features |
|---|---|---|---|---|
| Semicircular Canals | ||||
| Posterior canal | PC | Vertical, torsional | BPPV-PC | Nystagmus |
| Anterior canal | AC | Vertical, torsional | BPPV-AC, SCD | Nystagmus, fistula test |
| Horizontal canal | HC | Horizontal ≫ torsional | BPPV-HC, fistula | Nystagmus |
| Vestibular Nerve | ||||
| Superior division | AC, HC, utricle | Horizontal > torsional | VN, ischemia | Nystagmus, head-thrust test |
| Inferior division | PC, saccule | Vertical, torsional | VN, ischemia | Nystagmus |
| Common trunk (cranial nerve 8) | AC, HC, PC, utricle, saccule | Horizontal > torsional | VN, VP, ischemia | Nystagmus, head-thrust test, auditory findings |
| Labyrinth | AC, HC, PC, utricle, saccule | Horizontal > torsional | EH, labyrinthitis | Nystagmus, auditory findings |
Over time, either an asymmetry in the baseline firing rates resolves (the stimulation has been removed) or the central nervous system (CNS) compensates for it. This explains why an entire unilateral peripheral vestibular system can be surgically destroyed and patients only experience vertigo for several days to weeks. It also explains why patients with slow-growing tumors affecting the vestibular nerve, such as an acoustic neuroma, generally do not experience vertigo or nystagmus.
The history and physical examination provide the most important information when evaluating patients complaining of dizziness ( ). Often, patients have difficulty describing the exact symptom experienced (Kerber, 2017), so the onus is on the clinician to elicit pertinent information. The first step is to define the symptom. No clinician should ever be satisfied to record the complaint simply as “dizziness.” For patients unable to provide a more detailed description of the symptom, the physician can ask the patient to place their symptom into one of the following categories: movement of the environment (vertigo), lightheadedness, or strictly imbalance without an abnormal head sensation. However, caution must be taken in placing too much emphasis on the type of dizziness because patient descriptions about dizziness can be unreliable, inconsistent, and overlap ( ). Most dizziness patients report more than one type of dizziness, and specific types of dizziness symptoms have a stronger correlation with each other than they do with disease-based constructs (Kerber, 2017). Therefore other details about the symptom (e.g., timing, triggers) need to be considered as well. Table 22.2 displays the key distinguishing features of common causes of dizziness. One key point is that any type of dizziness may worsen with position changes, but some disorders such as BPPV only occur after position change.
| Cause | History of Vertigo | Duration of Vertigo | Associated Symptoms | Physical Examination |
|---|---|---|---|---|
| Peripheral | ||||
| Vestibular neuritis | Single prolonged episode | Days to weeks | Nausea, imbalance | “Peripheral” nystagmus, positive head-thrust test, imbalance |
| BPPV | Positionally triggered episodes | <1 min | Nausea | Characteristic positionally triggered burst of nystagmus |
| Meniere disease | May be triggered by salty foods | Hours | Unilateral ear fullness, tinnitus, hearing loss, nausea | Unilateral low-frequency hearing loss |
| Vestibular paroxysmia | Abrupt onset; spontaneous or positionally triggered | Seconds | Tinnitus, hearing loss | Usually normal |
| Perilymph fistula | Triggered by sound or pressure changes | Seconds | Hearing loss, hyperacusis | Nystagmus triggered by loud sounds or pressure changes |
| Central | ||||
| Stroke/TIA | Abrupt onset; spontaneous | Stroke, >24 h; TIA, < 24 h | Brainstem, cerebellar | Spontaneous “central” nystagmus; gaze-evoked nystagmus; focal neurological signs; negative head-thrust test; skew deviation |
| MS | Subacute onset | Minutes to weeks | Unilateral visual loss, diplopia, incoordination, ataxia | “Central” types or rarely “peripheral” types of spontaneous or positional nystagmus; usually other focal neurological signs |
| Neurodegenerative disorders | May be spontaneous or positionally triggered | Minutes to hours | Ataxia | “Central” types of spontaneous or positional nystagmus; gaze-evoked nystagmus; impaired smooth pursuit; cerebellar, extrapyramidal and frontal signs |
| Migraine | Onset usually associated with typical migraine triggers | Seconds to days | Headache, visual aura, photo-/phonophobia | Normal interictal examination; ictal examination may show “peripheral” or “central” types of spontaneous or positional nystagmus |
| Familial ataxia syndromes | Acute-subacute onset; usually triggered by stress, exercise, or excitement | Hours | Ataxia | “Central” types of spontaneous or positional nystagmus Ictal, or even interictal, gaze-evoked nystagmus; ataxia; gait disorders |
A brief general medical examination is important. Identifying orthostatic drops in blood pressure can be diagnostic in the correct clinical setting. Orthostatic hypotension is probably the most common general medical cause of dizziness among patients referred to neurologists. Identifying an irregular heart rhythm may also be pertinent. Other general examination measures to consider in individual patients include a visual assessment (adequate vision is important for balance) and a musculoskeletal inspection (significant arthritis can impair gait).
The general neurological examination is very important in patients complaining of dizziness, because dizziness can be the earliest symptom of a neurodegenerative disorder ( ) and can also be an important symptom of stroke, tumor, demyelination, or other pathologies of the nervous system. One should ensure that the patient has full ocular ductions. A posterior fossa mass can impair facial sensation and the corneal reflex on one side. Assessing facial strength and symmetry is important because of the close anatomical relationship between the seventh and eighth cranial nerves. The lower cranial nerves should also be closely inspected by observing palatal elevation, tongue protrusion, and trapezius and sternocleidomastoid strength.
The general motor examination determines strength in each muscle group and also assesses bulk and tone. Increased tone or cogwheel rigidity could be the main finding in a patient with an early neurodegenerative disorder. The peripheral sensory examination is important because a peripheral neuropathy can cause a nonspecific dizziness or imbalance. Temperature, pain, vibration, and proprioception should be assessed. Reflexes should be tested for their presence and symmetry. One must take into consideration the normal decrease in vibratory sensation and absence of ankle jerks that can occur in elderly patients. Coordination is an important part of the neurological examination in patients with dizziness because disorders characterized by ataxia can present with the principal symptom of dizziness. Observing the patient’s ability to perform the finger-nose-finger test, the heel-knee-shin test, and rapid alternating movements adequately assesses extremity coordination ( ).
When the general neurological examination is not revealing, the neuro-otological exam can be the critical element. The neuro-otological examination is a specialty examination expanding upon certain aspects of the general neurological examination and also includes an audio-vestibular assessment.
The first step in assessing ocular motor function is to search for spontaneous involuntary movements of the eyes. The examiner asks the patient to look straight ahead while observing for nystagmus or saccadic intrusions. Nystagmus is characterized by a slow- and fast-phase component and is classified as spontaneous, gaze-evoked, or positional. The direction of nystagmus is conventionally described by the direction of the fast phase, which is the direction it appears to be “beating” toward. Recording whether the nystagmus is vertical, horizontal, torsional, or a mixture of these provides important localizing information. Spontaneous nystagmus can have either a peripheral or central pattern. Although central lesions can mimic a “peripheral” pattern of nystagmus ( ), unusual circumstances are required for peripheral lesions to cause “central” patterns of nystagmus. The peripheral pattern of spontaneous nystagmus is unidirectional: that is, the eyes beat only to one side ( ![]() ). Peripheral spontaneous nystagmus never changes direction. It is usually a horizontal greater than torsional pattern because of the physiology of the asymmetry in firing rates within the peripheral vestibular system whereby the vertical canals cancel each other out. The prominent horizontal component results from the unopposed horizontal canal asymmetry. Other characteristics of peripheral spontaneous nystagmus are suppression with visual fixation, increase in velocity with gaze in the direction of the fast phase, and decrease with gaze in the direction opposite of the fast phase. Some patients are able to suppress this nystagmus so well at the bedside, or have partially recovered from the initiating event, that spontaneous nystagmus may only appear by removing visual fixation. Several simple bedside techniques can be used to remove the patient’s ability to fixate. Frenzel glasses are designed to remove visual fixation by using +30 diopter lenses. An ophthalmoscope can be used to block fixation. While the fundus of one eye is being viewed, the patient is asked to cover the other eye. Probably the simplest technique involves holding a blank sheet of paper close to the patient’s face (so as to block visual fixation) and observing for spontaneous nystagmus from the side.
). Peripheral spontaneous nystagmus never changes direction. It is usually a horizontal greater than torsional pattern because of the physiology of the asymmetry in firing rates within the peripheral vestibular system whereby the vertical canals cancel each other out. The prominent horizontal component results from the unopposed horizontal canal asymmetry. Other characteristics of peripheral spontaneous nystagmus are suppression with visual fixation, increase in velocity with gaze in the direction of the fast phase, and decrease with gaze in the direction opposite of the fast phase. Some patients are able to suppress this nystagmus so well at the bedside, or have partially recovered from the initiating event, that spontaneous nystagmus may only appear by removing visual fixation. Several simple bedside techniques can be used to remove the patient’s ability to fixate. Frenzel glasses are designed to remove visual fixation by using +30 diopter lenses. An ophthalmoscope can be used to block fixation. While the fundus of one eye is being viewed, the patient is asked to cover the other eye. Probably the simplest technique involves holding a blank sheet of paper close to the patient’s face (so as to block visual fixation) and observing for spontaneous nystagmus from the side.
Acute Peripheral Vestibular Nystagmus.
Saccadic intrusions are spontaneous, involuntary saccadic movements of the eyes, without the rhythmic fast and slow phases characteristic of nystagmus. Saccades are fast movements of the eyes normally under voluntary control and used to shift gaze from one object to another. Square-wave jerks and saccadic oscillations are the most common types of saccadic intrusions. Square-wave jerks refer to small-amplitude, involuntary saccades that take the eyes off a target, followed after a normal intersaccadic delay (around 200 msec) by a corrective saccade to bring the eyes back to the target. Square-wave jerks can be seen in neurological disorders such as cerebellar ataxia, Huntington disease (HD), or progressive supranuclear palsy (PSP), but they also occur in normal individuals. If the square-wave jerks are persistent or of large amplitude (macro-square wave jerks), pathology is more likely.
Saccadic oscillations refer to back-to-back saccadic movements without the intersaccadic interval characteristic of square-wave jerks, so their appearance is that of an oscillation. When a burst occurs only in the horizontal plane, the term ocular flutter is used ( ![]() ). When vertical and/or torsional components are present, the term opsoclonus (or so-called dancing eyes) is used. The eyes make constant random conjugate saccades of unequal amplitude in all directions. Ocular flutter and opsoclonus are pathological findings typically seen in several different types of CNS diseases involving brainstem–cerebellar pathways. Paraneoplastic disorders should be considered in patients presenting with ocular flutter or opsoclonus.
). When vertical and/or torsional components are present, the term opsoclonus (or so-called dancing eyes) is used. The eyes make constant random conjugate saccades of unequal amplitude in all directions. Ocular flutter and opsoclonus are pathological findings typically seen in several different types of CNS diseases involving brainstem–cerebellar pathways. Paraneoplastic disorders should be considered in patients presenting with ocular flutter or opsoclonus.
Ocular Flutter.
The patient should be asked to look to the left, right, up, and down; the examiner looks for gaze-evoked nystagmus in each position ( ![]() ). A few beats of unsustained nystagmus with gaze greater than 30 degrees is called end-gaze nystagmus and variably occurs in normal subjects. Gaze-evoked downbeating nystagmus (
). A few beats of unsustained nystagmus with gaze greater than 30 degrees is called end-gaze nystagmus and variably occurs in normal subjects. Gaze-evoked downbeating nystagmus ( ![]() ), vertical nystagmus that increases on lateral gaze, localizes to the craniocervical junction and midline cerebellum. Gaze testing may also trigger saccadic oscillations (see , ).
), vertical nystagmus that increases on lateral gaze, localizes to the craniocervical junction and midline cerebellum. Gaze testing may also trigger saccadic oscillations (see , ).
Gaze-Evoked Nystagmus and Impaired Smooth Pursuit.
Gaze-Evoked Downbeating Nystagmus.
Smooth pursuit refers to the voluntary movement of the eyes used to track a target moving at a low velocity. It functions to keep the moving object on the fovea to maximize vision. Though characteristically a very smooth movement at low frequency and velocity testing, smooth pursuit inevitably breaks down when tested at high frequencies and velocities. Though smooth pursuit often becomes impaired with advanced age, a longitudinal study of healthy elderly individuals found no significant decline in smooth pursuit over 9 years of evaluation ( ). Patients with impaired smooth pursuit require frequent small saccades to keep up with the target; thus the term saccadic pursuit is used to describe this finding. Abnormalities of smooth pursuit occur as the result of disorders throughout the CNS and with tranquilizing medicines, alcohol, inadequate concentration or vision, and fatigue. However, in a cognitively intact individual presenting with dizziness or imbalance symptoms, bilaterally impaired smooth pursuit is highly localizing to the cerebellum. Patients with early or mild cerebellar degenerative disorders may have markedly impaired smooth pursuit with mild or minimal truncal ataxia as the only findings.
Saccades are fast eye movements (velocity of this eye movement can be as high as 600 degrees per second) used to quickly bring an object onto the fovea. Saccades are generated by the burst neurons of the pons (horizontal movements) and midbrain (vertical movements). Lesions or degeneration of these regions leads to slowing of saccades, which can also occur with lesions of the ocular motor neurons or extraocular muscles. Severe slowing can be readily appreciated at the bedside by instructing the patient to look back and forth from one object to another. The examiner observes both the velocity of the saccade and the accuracy. Overshooting saccades (missing the target and then needing to correct) indicates a lesion of the cerebellum ( ![]() ). Undershooting saccades are less specific and often occur in normal subjects.
). Undershooting saccades are less specific and often occur in normal subjects.
Hypermetric Saccades.
Optokinetic nystagmus (OKN) and fixation suppression of the VOR suppression can also be tested at the bedside. OKN is a combination of fast (saccadic) and slow (smooth pursuit) movements of eyes and can be observed in normal individuals when, for example, watching a moving train. OKN is maximally stimulated with both foveal and parafoveal stimulation, so the proper laboratory technique for measuring OKN uses a full-field stimulus by having the patient sit stationary while a large rotating pattern moves around them. This test can be approximated at the bedside by moving a striped cloth in front of the patient, though this technique only stimulates the fovea. Patients with disorders causing severe slowing of saccades will not be able to generate OKN, so their eyes will become pinned to one side. VOR suppression can be tested at the bedside using a swivel chair. The patient sits in the chair and extends his or her arm in the “thumbs-up” position out in front. The patient is instructed to focus on the thumb and to allow the extended arm to move with the body so the visual target of the thumb remains directly in front of the patient. The chair is then rotated from side to side. The patient’s eyes should remain locked on the thumb, demonstrating the ability to suppress the VOR stimulated by rotation of the chair. Nystagmus will be observed during the rotation movements in patients with impairment of VOR suppression, which is analogous to impairment of smooth pursuit. Both OKN and VOR suppression can also be helpful when examining patients having difficulty following the instructions for smooth pursuit or saccade testing.
Often omitted as part of the cranial nerve examination in general neurology texts, important localizing information can be obtained about the functioning of the vestibular nerve at the bedside. A unilateral or bilateral vestibulopathy can be identified using the head-thrust test ( ) ( Fig. 22.3 and ![]() ). To perform the head-thrust test, the physician stands directly in front of the patient, who is seated on the examination table. The patient’s head is held in the examiner’s hands, and the patient is instructed to focus on the examiner’s nose. The head is then quickly moved about 5–10 degrees to one side. In patients with normal vestibular function, the VOR results in movement of the eyes in the direction opposite the head movement. Therefore the patient’s eyes remain on the examiner’s nose after the sudden movement. The test is repeated in the opposite direction. If the examiner observes a corrective saccade bringing the patient’s eyes back to the examiner’s nose after the head thrust, impairment of the VOR in the direction of the head movement is identified. Rotating the head slowly back and forth (the doll’s eye test) also induces compensatory eye movements, but both the visual and vestibular systems are activated by this low-velocity test, so a patient with complete vestibular function loss and normal visual pursuit will have normal-appearing compensatory eye movements on the doll’s eye test. This slow rotation of the head, however, is helpful in a comatose patient who is not able to generate voluntary visual tracking eye movements. Slowly rotating the head can also be a helpful test in patients with impairment of the smooth-pursuit system, because smooth movements of the eyes during slow rotation of the head indicates an intact VOR, whereas continued saccadic movements during slow rotation indicates an accompanying deficit of the VOR ( ).
). To perform the head-thrust test, the physician stands directly in front of the patient, who is seated on the examination table. The patient’s head is held in the examiner’s hands, and the patient is instructed to focus on the examiner’s nose. The head is then quickly moved about 5–10 degrees to one side. In patients with normal vestibular function, the VOR results in movement of the eyes in the direction opposite the head movement. Therefore the patient’s eyes remain on the examiner’s nose after the sudden movement. The test is repeated in the opposite direction. If the examiner observes a corrective saccade bringing the patient’s eyes back to the examiner’s nose after the head thrust, impairment of the VOR in the direction of the head movement is identified. Rotating the head slowly back and forth (the doll’s eye test) also induces compensatory eye movements, but both the visual and vestibular systems are activated by this low-velocity test, so a patient with complete vestibular function loss and normal visual pursuit will have normal-appearing compensatory eye movements on the doll’s eye test. This slow rotation of the head, however, is helpful in a comatose patient who is not able to generate voluntary visual tracking eye movements. Slowly rotating the head can also be a helpful test in patients with impairment of the smooth-pursuit system, because smooth movements of the eyes during slow rotation of the head indicates an intact VOR, whereas continued saccadic movements during slow rotation indicates an accompanying deficit of the VOR ( ).
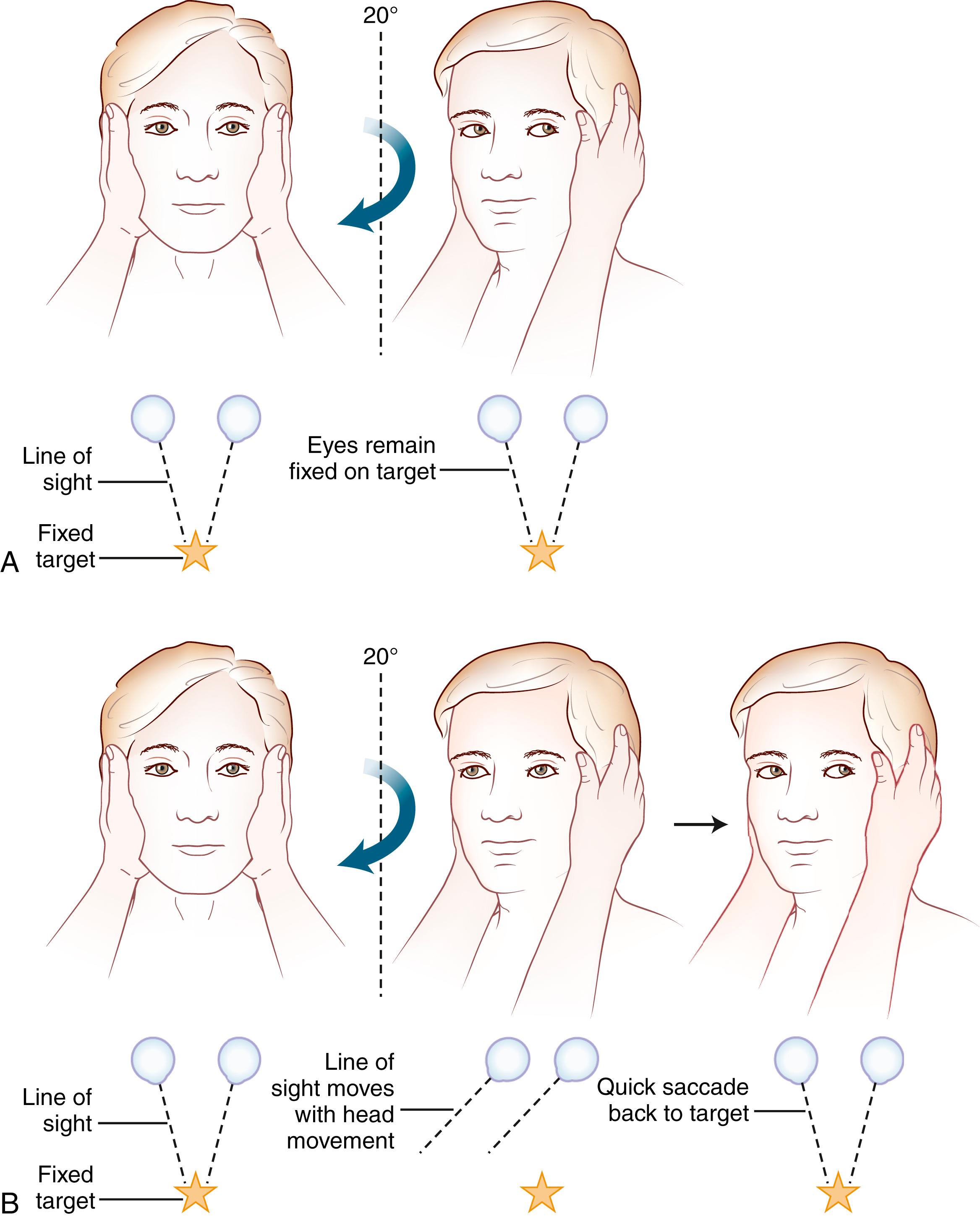
Head-Thrust Tests.
Positional testing can help identify peripheral or central causes of vertigo. The most common positional vertigo, BPPV, is caused by free-floating calcium carbonate debris, usually in the posterior semicircular canal, occasionally in the horizontal canal, or rarely in the anterior canal. The characteristic burst of upbeat torsional nystagmus is triggered in patients with BPPV by a rapid change from the sitting-up position to supine head-hanging left or head-hanging right (the Dix–Hallpike test; ![]() ). When present, the nystagmus is usually only triggered in one of these positions. A burst of nystagmus in the opposite direction (downbeat torsional) occurs when the patient resumes the sitting position since the debris moves in the opposite direction. A repositioning maneuver can be used to move the debris out of the canal. The modified Epley maneuver ( Fig. 22.4 and
). When present, the nystagmus is usually only triggered in one of these positions. A burst of nystagmus in the opposite direction (downbeat torsional) occurs when the patient resumes the sitting position since the debris moves in the opposite direction. A repositioning maneuver can be used to move the debris out of the canal. The modified Epley maneuver ( Fig. 22.4 and ![]() ) is more than 80% effective in treating patients with posterior canal BPPV, compared with 10% effectiveness of a sham procedure (Hilton, 2014). The key feature of this maneuver is the roll across in the plane of the posterior canal so that the debris rotates around the posterior canal and out into the utricle. Once the debris enters the utricle, it no longer disrupts semicircular canal function. Recurrences are common, however (see , ).
) is more than 80% effective in treating patients with posterior canal BPPV, compared with 10% effectiveness of a sham procedure (Hilton, 2014). The key feature of this maneuver is the roll across in the plane of the posterior canal so that the debris rotates around the posterior canal and out into the utricle. Once the debris enters the utricle, it no longer disrupts semicircular canal function. Recurrences are common, however (see , ).
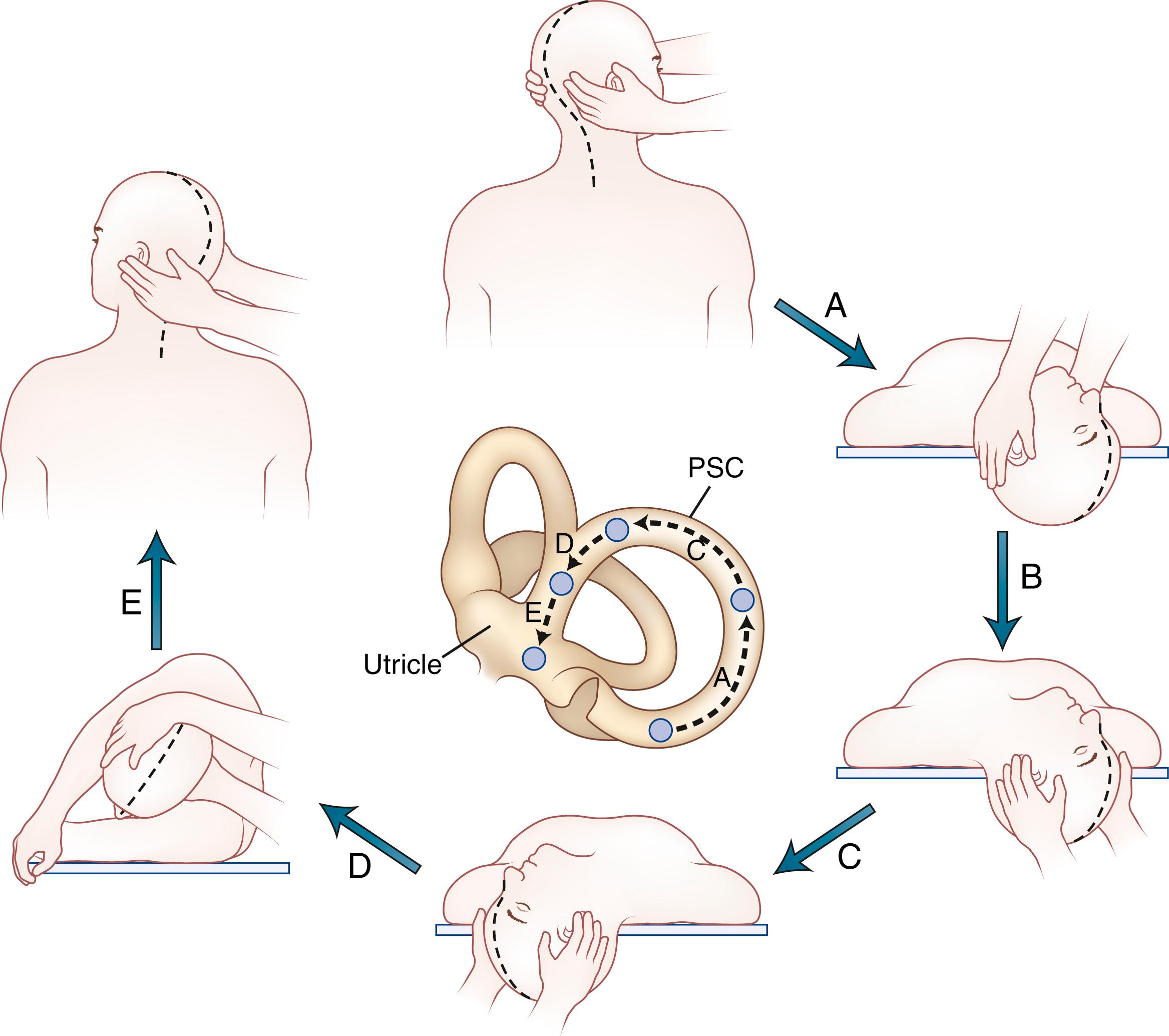
Benign Paroxysmal Positional Vertigo.
Epley Maneuver.
If the debris is in the horizontal canal, direction-changing horizontal nystagmus is seen. Patients are tested for HC-BPPV by turning the head to each side while lying in the supine position. The nystagmus can be either paroxysmal geotropic (beating toward the ground) or persistent apogeotropic nystagmus (beating away from the ground). In the case of geotropic nystagmus, the debris is in the posterior segment (or “long arm”) of the horizontal canal, whereas the debris is in the anterior segment (or “short arm”) when apogeotropic nystagmus is triggered. When geotropic nystagmus is triggered, the side with the stronger nystagmus is the involved side. However, when apogeotropic nystagmus is observed, the involved side is generally opposite the side of the stronger nystagmus. With the geotropic variant, class I evidence supports treatment with the barbecue maneuver or the Gufoni maneuver ( ). Another maneuver for HC-BPPV is the “forced prolonged position” ( ). In cases of the apogeotropic variant of HC-BPPV, a variation of the Gufoni maneuver or a head-shaking maneuver can effectively treat the condition, though patients may require a second maneuver to clear the debris from the long arm of the horizontal canal (the same maneuver to treat geotropic HC-BPPV; ).
Positional testing can also trigger central types of nystagmus (usually persistent downbeating), which may be the most prominent examination finding in patients with disorders like Chiari malformation or cerebellar ataxia ( ). Central positional nystagmus can also mimic the nystagmus of HC-BPPV. Positional nystagmus may also be prominent in patients with migraine-associated dizziness ( ).
In patients reporting sound- or pressure-induced dizziness, a defect of the bony capsule of the labyrinth can be tested for by pressing and releasing the tragus (small flap of cartilage that can be used to occlude the external ear canal) and observing the eyes for brief associated deviations. Pneumatoscopy (introducing air into the external auditory canal through an otoscope) or Valsalva against pitched nostrils or closed glottis can also trigger associated eye movements. The direction of the triggered nystagmus helps identify the location of the fistula.
Casual gait is examined for initiation, heel strike, stride length, and base width. Patients are then observed during tandem walking and while standing in the Romberg position (with eyes open and closed). A decreased heel strike, stride length, flexed posture, and decreased arm swing suggest Parkinson disease. A wide-based gait with inability to tandem walk is characteristic of truncal ataxia. Patients with acute vestibular loss will veer toward the side of the affected ear for several days after the event. Patients with peripheral neuropathy or bilateral vestibulopathy may be unable to stand in the Romberg position with eyes closed.
The bedside examination of the auditory system begins with otoscopy. The tympanic membrane is normally translucent; changes in color indicate middle ear disease or tympanosclerosis, a semicircular crescent or horseshoe-shaped white plaque within the tympanic membrane. Tympanosclerosis is rarely associated with hearing loss but is an important clue to past infections. The area just superior to the lateral process of the malleus should be carefully inspected for evidence of a retraction pocket or cholesteatoma. Findings on otoscopy are usually not associated with causes of dizziness because the visualized abnormalities typically do not involve the inner ear.
Finger rubs at different intensities and distances from the ear are a rapid, reliable, and valid screening test for hearing loss in the frequency range of speech ( ). If a patient can hear a faint finger rub stimulus at a distance of 70 cm (approximately one arm’s length) from one ear, then a hearing loss on that side—defined by a gold-standard audiogram threshold of greater than 25 dB at 1000, 2000, and 4000 Hz—is highly unlikely. On the other hand, if a patient cannot hear a strong finger rub stimulus at 70 cm, hearing loss on that side is highly likely. The whisper test can also be used to assess hearing at the bedside ( ). For this test, the examiner stands behind the patient to prevent lip reading and occludes and masks the nontest ear, using a finger to rub and close the external auditory canal. The examiner then whispers a set of three to six random numbers and letters. Overall, the patient is considered to have passed the screening test if they repeat at least 50% of the letters and numbers correctly. The Weber and Rinne tests are commonly used bedside tuning fork tests. To perform these, a tuning fork (256 Hz or 512 Hz) is gently struck on a hard rubber pad, the elbow, or the knee about two-thirds of the way along the tine. To conduct the Weber test, the base of the vibrating fork is placed on the vertex (top or crown of the head), bridge of the nose, upper incisors, or forehead. The patient is asked if the sound is heard and whether it is heard in the middle of the head or in both ears equally, toward the left, or toward the right. In a patient with normal hearing, the tone is heard centrally. In asymmetrical or a unilateral hearing impairment, the tone lateralizes to one side. Lateralization indicates an element of conductive impairment in the ear in which the sound localizes, a sensorineural impairment in the contralateral ear, or both. The Rinne test compares the patient’s hearing by air conduction with that by bone conduction. The fork is first held against the mastoid process until the sound fades. It is then placed 1 inch from the ear. Normal subjects can hear the fork about twice as long by air as by bone conduction. If bone is greater than air conduction, a conductive hearing loss is suggested.
Peripheral vestibular disorders are important for neurologists to understand because they are common, readily identified at the bedside, and often missed by frontline physicians (see Table 22.2 ).
A common presentation to the ED or outpatient clinic is the rapid onset of severe vertigo, nausea, vomiting, and imbalance. The symptoms gradually resolve over days to weeks, but about 20% of patients report some dizziness even 12 months later (Shupak, 2008). The typical etiology of this disorder is vestibular neuritis, which is presumed to be viral because the course is generally benign and self-limited, similar to Bell palsy. Small histopathological studies support the etiology of a viral cause. However, ischemia or demyelination can also cause an acute unilateral vestibulopathy. The key to identifying an acute unilateral vestibulopathy is recognizing the peripheral vestibular pattern of nystagmus and identifying a positive head-thrust test in the setting of a rapid onset of vertigo without other neurological symptoms. The course of vestibular neuritis is self-limited, and the mainstay of treatment is symptomatic. A course of corticosteroids might improve recovery of the caloric response but symptomatic and functional outcomes were not clinically different ( ). Vestibular physical therapy—delivered with in person sessions or even home training—can help patients compensate for the vestibular lesion (Hillier et al., 2011).
BPPV has a lifetime cumulative incidence of nearly 10% (von Brevern, 2005). Patients typically experience brief episodes of vertigo when getting in and out of bed, turning in bed, bending down and straightening up, or extending the head back to look up. As noted earlier, the condition is caused when calcium carbonate debris dislodged from the otoconial membrane inadvertently enters a semicircular canal. The debris can be free-floating within the affected canal (canalithiasis) or stuck against the cupula (cupulolithiasis). Though the positional attacks are the hallmark feature, some BPPV patients also report constant mild unsteadiness (Von Brevern, 2015). The gold standard test is the Dix-Hallpike test with the positive finding being the hallmark triggered and transient upbeat-torsional nystagmus. Repositioning maneuvers are highly effective in removing the debris from the canal, though recurrence is common (see Fig. 22.4 ; ). Once the debris is out of the canal, patients are instructed to avoid extreme head positions to prevent the debris from re-entering the canal. Patients can also be taught to perform a repositioning maneuver, should they have a recurrence of the positional vertigo.
Meniere disease is characterized by recurrent attacks of vertigo associated with auditory symptoms (hearing loss, tinnitus, aural fullness) during attacks. Over time, progressive hearing loss develops. Attacks are variable in duration, most lasting longer than 20 minutes, and are associated with severe nausea and vomiting. The course of the disorder is also highly variable. For some patients, the attacks are infrequent and decrease over time, but for others they can become debilitating. Occasionally, auditory symptoms are not appreciated by the patients or identified by interictal audiograms early in the disorder, but inevitably patients with Meniere disease develop these features, usually within the first year. Thus the term vestibular Meniere disease , previously used for patients with recurrent episodes of vertigo but no hearing loss, is no longer used. Though usually a disorder involving only one ear, Meniere disease becomes bilateral in about one-third of patients.
Endolymphatic hydrops, or expansion of the endolymph relative to the perilymph, is regarded as the etiology, though the underlying cause is unclear. In addition, the characteristic histopathological changes of endolymphatic hydrops have been identified in temporal bone specimens of patients with no clinical history of Meniere disease ( ). Some patients with well-documented Meniere disease experience abrupt episodes of falling to the ground, without loss of consciousness or associated neurological symptoms. Patients often report the sensation of being pushed or thrown to the ground. The falls are hard and often result in fractures or other injuries. These episodes have been called otolithic catastrophes of Tumarkin because of the suspicion that they represent acute stimulation of the otoliths. The bedside interictal examination of patients with Meniere disease may identify asymmetrical hearing, but the head-thrust test is usually normal. Treatment is initially directed toward an aggressive low-salt diet and diuretics, though the evidence for these treatments is poor. Intratympanic gentamicin injections can be effective and are minimally invasive. Sectioning of the vestibular nerve and destruction of the labyrinth are other procedures ( ). Autoimmune inner-ear disease presents as a fulminate variant of Meniere disease. Another variant is so-called delayed endolymphatic hydrops. Patients with this disorder report recurrent episodes of severe vertigo without auditory symptoms developing years after a severe unilateral hearing loss caused by a viral or bacterial infection.
Vestibular paroxysmia is characterized by brief (seconds to minutes) episodes of vertigo, occurring suddenly without any apparent trigger (Strupp, 2016). The disorder may be analogous to hemifacial spasm and trigeminal neuralgia, which are felt to be due to spontaneous discharges from a partially damaged nerve. In patients with vestibular paroxysmia, unilateral dysfunction can sometimes be identified on vestibular or auditory testing. Like the analogous disorders, it is conceivable that a normal vessel could be compressing the cranial nerve, and surgical removal of the vessel might seem to be a treatment option. However, many asymptomatic subjects have a normal vessel lying on the eighth nerve (usually the anterior inferior cerebellar artery), and most vestibular paroxysmia patients have a favorable course with conservative or medication management (Strupp, 2016), so the decision to operate in this delicate region is rarely indicated. Medications associated with a reduction in episodes include carbamazepine, oxcarbazepine, and gabapentin (Strupp, 2016).
Superior canal dehiscence was first described in 1998 ( ). As the name implies, dehiscence of the bone overlying the superior canal results in a fistula between the superior canal and the middle cranial fossa. Normally the semicircular canals are enclosed by the rigid bony capsule, so these vestibular structures are unaffected by sound pressure changes. The oval and round windows direct the forces associated with sound waves into the cochlea and along the spiral basilar membrane. A break in the bony capsule of the semicircular canals can redirect some of the sound or pressure to the semicircular canals, causing vestibular activation, a phenomenon known as Tullio phenomenon . Prior to the discovery of SCD, fistulas were known to occur with rupture of the oval or round window or erosion into the horizontal semicircular canal from chronic infection. Pressure changes generated by increasing intracranial pressure (ICP; Valsalva against closed glottis) or increasing middle ear pressure (Valsalva against pinched nostrils or compression of the tragus) triggers brief nystagmus in the plane of the affected canal. Surgically repairing the defect can be attempted if the patient is debilitated by the symptoms, but most patients do well with conservative management (Ward, 2017). A study looking at long-term outcome after SCD surgical treatment had a low follow-up rate (43%; 93/218) but reported trends in improved audio-vestibular symptoms that were greatest for autophony, pulsatile tinnitus, audible body sounds, and sensitivity to loud sounds, and least for dizziness (Alkhafaii, 2017). Patients with SCD may have hypersensitivity to bone-conducted sound and bone-conduction thresholds on the audiogram lower than the normal 0 dB hearing levels, even though air conduction thresholds remain normal ( ).
There are many other peripheral vestibular causes of vertigo, but most are uncommon. Vertigo often follows a blow to the head, even without a corresponding temporal bone fracture. This so-called labyrinthine concussion results from the susceptibility of the delicate structures of the inner ear to blunt trauma. Vestibular ototoxicity, usually from gentamicin, can cause a vestibulopathy that is usually bilateral but rarely can be unilateral ( ). A bilateral vestibulopathy can also occur from an immune-mediated disorder (e.g., autoimmune inner-ear disease, Cogan syndrome), infectious process (e.g., meningitis, syphilitic labyrinthitis), structural lesion (bilateral acoustic neuroma), or a genetic disorder (e.g., neurodegenerative or isolated vestibular). The bilateral vestibular loss often goes unrecognized because the vestibular symptoms can be overshadowed by auditory or other symptoms. Although the most prominent vestibular symptoms of bilateral vestibulopathy are oscillopsia and imbalance, some nonspecific dizziness and vertigo attacks may occur as well. Vestibular schwannomas typically present with slowly progressive unilateral hearing loss, but rarely vertigo can occur. Because the tumor growth is slow, the vestibulopathy is compensated by the CNS. Finally, any disorder affecting the skull base, such as sarcoidosis, lymphoma, bacterial and fungal infections, or carcinomatous meningitis, can cause either unilateral or bilateral peripheral vestibular symptoms.
The key to the diagnosis of CNS disorders in patients presenting with dizziness is the presence of other focal neurological symptoms or identifying central ocular motor abnormalities or ataxia. Because central disorders can mimic peripheral vestibular disorders, the most effective approach in patients with isolated dizziness is first to rule out common peripheral causes.
Ischemia affecting vestibular pathways within the brainstem or cerebellum often causes vertigo. Brainstem ischemia is normally accompanied by other neurological signs and symptoms, because motor and sensory pathways are in close proximity to vestibular pathways. Vertigo is the most common symptom with Wallenberg syndrome, infarction in the lateral medulla in the territory of the posterior inferior cerebellar artery (PICA), but other neurological symptoms and signs (e.g., diplopia, facial numbness, Horner syndrome) are typically present. Ischemia of the cerebellum can cause vertigo as the most prominent or only symptom, and a common dilemma is whether the patient with acute-onset vertigo needs a magnetic resonance imaging (MRA) scan to rule out cerebellar infarction. Computed tomography (CT) scans of the posterior fossa are not a sensitive test for acute ischemic stroke ( ). Abnormal ocular motor findings in patients with brainstem or cerebellar strokes include (1) spontaneous nystagmus that is purely vertical or torsional, (2) direction-changing gaze-evoked nystagmus (patient looks to the left and has left-beating nystagmus, looks to the right, and has right-beating nystagmus), (3) impairment of smooth pursuit, and (4) overshooting saccades. Central causes of nystagmus can sometimes closely mimic the peripheral vestibular pattern of spontaneous nystagmus ( ). In these cases, a negative head-thrust test (i.e., no corrective saccade) or a skew deviation could be the key indicators of a central rather than a peripheral vestibular lesion ( ). Cardiovascular risk factors are also independent predictors of stroke in dizziness patients (Kerber, 2015) and therefore should be considered in evaluation and management plans.
Dizziness is a common symptom in patients with multiple sclerosis (MS). Vertigo is the initial symptom in about 5% of patients with MS. A typical MS attack has a gradual onset, reaching its peak within a few days. Milder spontaneous episodes of vertigo, not characteristic of a new attack, and positional vertigo lasting seconds are also common in MS patients. Nearly all varieties of central spontaneous and positional nystagmus occur with MS, and occasionally patients show typical peripheral vestibular nystagmus when the lesion affects the root entry zone of the vestibular nerve. MRI of the brain identifies white matter lesions in about 95% of MS patients, although similar lesions are sometimes seen in patients without the clinical criteria for the diagnosis of MS.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here