Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Acquired immunodeficiency syndrome (AIDS) was first recognized in the United States in the summer of 1981 when unexplained occurrences of Pneumocystis jirovecii (formerly carinii ) pneumonia and Kaposi sarcoma were reported in cohorts of previously healthy homosexual men in Los Angeles, New York, and San Francisco. Within months, the disease became recognized in intravenous drug users (IDUs) and soon thereafter in recipients of blood transfusion and blood products, including hemophiliacs. As the epidemiological pattern unfolded, it became clear that a microbe transmitted by sexual (homosexual and heterosexual) contact and through blood and blood products was the etiological agent of the epidemic. In 1983, human immunodeficiency virus type-1 (and later type-2, henceforth called HIV) was isolated and confirmed the following year as the causal agent of AIDS. In 1985, a sensitive enzyme-linked immunosorbent assay (ELISA) test was developed, which led to the recognition of the scope of HIV infection among cohorts of individuals in the United States and elsewhere. As the disease spread, seroprevalence studies revealed the enormity of the global pandemic, with AIDS cases reported from virtually every country.
Now, after three and a half decades of AIDS, this disease has become one of the greatest public health challenges globally. The HIV pandemic has resulted in an estimated 77.3 million infections worldwide and it has claimed the lives of 35.4 million persons ( www.unaids.org ). Although the trend in AIDS-related deaths and new HIV infection has declined in recent years, during the year 2017 alone, 1.8 million (compared to peak of 3.4 million in 2004) persons died from AIDS and 940,000 (compared to 1.9 million in 2004 and 1.4 million in 2010) were newly infected with HIV. At the end of 2017, an estimated 36.9 million (31.5 million adults, 1.8 million children younger than 15) were living with HIV disease ( www.unaids.org ). The latest epidemiological data indicate that globally the spread of HIV had peaked in the late 1990s, when yearly an estimated 3.5 million new infections occurred. In 2017, the estimated number of new infections was approximately 51% lower in adults than it was in late 1990s. However, the population of people living with HIV continues to rise, chiefly from the combined effects of continued high rates of new infections and the survival benefits of antiretroviral therapy.
HIV continues to disproportionately affect certain geographic regions (e.g., sub-Saharan Africa, Caribbean nations) and certain subpopulations (homosexual men, women in sub-Saharan Africa, intravenous drug abusers, and sex workers). East and Southern Africa is home to 6.2% of the world’s population (440.2 million) but has 19.4 million people living with HIV, greater than 50% of the global total; Swaziland has the highest adult HIV prevalence in the world (27.2%), and HIV prevalence among sex workers varies between 50% and 70% in several countries in this region ( www.unaids.org ).
The staggering grim statistics of the worldwide HIV pandemic has been matched, however, by an explosion of information in the area of HIV virology, pathogenesis and treatment of HIV disease, treatment and prophylaxis of opportunistic diseases associated with HIV infection, and prevention of HIV infection. In the United States and other resource-rich countries, the incidence of AIDS and AIDS-related deaths has fallen by over 80% since 1995. This trend reflects reduced HIV infection rates since the mid-1980s and the use of highly effective combined active antiretroviral therapy (cART) early in the course of HIV infection. Perinatal HIV transmission from HIV-infected mothers has shown a dramatic decline (>95%) in the United States. This is attributable to several factors, including universal prenatal testing, antiretroviral prophylaxis, elective cesarean delivery, and avoidance of breastfeeding by infected mothers.
As the HIV epidemic has changed in recent years, new challenges have emerged. As more HIV-infected persons receive cART (50-fold increase in access to cART globally since 2001), the number of persons living with HIV is increasing, requiring that the prevention programs must be scaled up to prevent HIV transmission from those living with HIV infection to those at risk of infection. With no vaccine on the foreseeable horizon, stopping HIV transmission and halting AIDS-related deaths is the only practical strategy currently available to halt the AIDS epidemic.
HIV is an RNA virus belonging to the family of human retroviruses (Retroviridae) and the subfamily of lentiviruses. Electron microscopy shows that the HIV virion is an icosahedral structure. It contains numerous external spikes formed by the two major envelope proteins (gp120 and gp41) and one major core protein (p24). Like other retroviruses, HIV has genes that encode the structural and enzyme proteins of the virus: env encodes the surface glycoproteins, gag encodes the core protein, and pol and integrase encode for the enzymes responsible for reverse transcription and its integration into the nuclear genome. HIV also contains at least six other genes ( tat , rev , nef , vif , vpr , and vpu ), which encode for proteins involved in viral gene regulation and host cell modification to enhance viral growth. Several of these proteins play a role in the pathogenesis of HIV disease (see below). Flanking these genes are long terminal repeats that contain regulatory elements involved in gene expression. The major difference between the genomes of HIV-1 and HIV-2 is that HIV-2 lacks the vpu gene and has a vpx gene instead.
The replication cycle of HIV begins with high-affinity binding of the gp120 envelope protein to its receptor, the CD4 molecule, on the host cell surface ( ). The CD4 molecule is a 55-kDa protein found predominantly on CD4+ helper-inducer lymphocytes (CD4+ T cells), but also on the surface of monocytes-macrophages and dendritic-Langerhans cells. After gp120 binds to the CD4 molecule, it undergoes a conformational change that facilitates its binding to a co-receptor (CCR5, CXCR4) ( ). These co-receptors belong to the family of transmembrane G protein-coupled cellular receptors, and the use of one or other of these receptors by the virus for entry into the cell determines the cellular tropism of the virus strain. Following surface attachment and fusion, the HIV genomic RNA is uncoated and internalized into the host cell. The subsequent steps in viral replication include activation of reverse transcriptase enzyme, reverse transcription of the genomic RNA into double-stranded DNA, and integration of DNA into the host cell chromosome through the action of another virally encoded enzyme— integrase . HIV integration into the nuclear DNA is not entirely random but is preferential for transcriptionally active genes and for some hotspots. This integrated provirus may remain latent (transcriptionally inactive) or it may manifest varying levels of gene expression, up to active production of the whole virus. Progression through the viral replication cycle is profoundly influenced by a variety of viral regulatory gene products and host factors ( ).
Molecular analyses of various HIV isolates from a single person over time and from cohorts of patients reveal sequence variations over many parts of the viral genome. The degree of difference in coding sequences of the viral envelope genes, for example, can vary from a few percent to 50%. These sequence changes tend to cluster in hypervariable regions of the HIV genome. Single-base substitutions, insertions and deletions, recombinations, and gain or loss of glycosylation sites are involved in this phenomenon of genetic heterogeneity. The immune pressure and functional constraints on viral proteins (by antibodies, for example) and the use of retroviral therapy influence the level of variation in viral genes and protein products.
Detailed knowledge of the HIV agent is important for understanding the principles of HIV diagnostics and therapeutics. Diagnostic tests of HIV infection are based on host antibody response to viral proteins (ELISA), detection of major viral proteins (Western blot), or direct sequencing of viral genomic sites. The sensitivity and specificity of each of these tests differ. Likewise, each point in the replication cycle of HIV is a real or potential target for therapeutic intervention (see section on antiretroviral therapy). Study of viral genomic heterogeneity is also a powerful molecular tool to track viral trafficking worldwide and to understand drug resistance.
The hallmark of HIV disease is the profound immunodeficiency resulting from a progressive loss or dysfunction of the subset of T lymphocytes referred to as helper or inducer T cells in conjunction with polyclonal immune activation ( ). The CD4+ T lymphocytes are phenotypically defined by the presence on their surface of the CD4 molecules that serve as the primary cellular receptor for HIV. A co-receptor (CCR5 or CXCR4, see previous section) must also be present together with CD4 for efficient fusion and entry of HIV into the target cells. CD4+ T cells are the initial and crucial natural activators in immune defense mechanisms. It is thus ironic that these helper/inducer CD4+ T cells are the cells most likely to bind to the virus, be infected, and themselves be destroyed. Whether direct infection and destruction, or activation-induced cell death, or a combination of these mechanisms is primarily responsible for progressive dysfunction and depletion of CD4+ T cells in vivo is unclear. But when the number of CD4+ T cells declines below a certain level, the person is at high risk of developing a variety of opportunistic diseases: chiefly the AIDS-defining infections and neoplasms. Some features of AIDS, such as neurological abnormalities (encephalopathy, dementia, polyneuropathy), cannot be explained entirely by the immunosuppressive effect of HIV, since they can arise before the development of severe immunological impairment.
The current Centers for Disease Control and Prevention (CDC) classification system of HIV-infected persons categorizes patients on the basis of clinical conditions associated with HIV infection and CD4+ T cell counts ( Table 77.1 ). A confirmed HIV case can be classified in one of the five stages (0, 1, 2, 3, or unknown). Stage is 0 if the person was tested seronegative within previous 6 months of the first HIV infection diagnosis or until 6 months after diagnosis. Advanced HIV disease (AIDS) is classified as stage 3 if one or more specific opportunistic illness has been diagnosed. Otherwise, stage is determined by CD4+ T cell counts and immunological status. The stage is classified U (unknown) if none of these criteria apply (e.g., missing CD4+ T cell test results). The definition and staging criteria of AIDS are complex and comprehensive and were originally designed for surveillance purposes. The clinicians should not focus only on which stage is present, but instead should view HIV disease as a spectrum ranging from primary infection through asymptomatic stage to advanced disease.
| Age on Date of CD4+ T cell Count | ||||||
|---|---|---|---|---|---|---|
| ≤1 Year | 1–5 Years | 6 Years Through Adult | ||||
| Stage ∗ | Cells/μL | % | Cells/μL | % | Cells/μL | % |
| 1 | ≥1500 | ≥34 | ≥1000 | ≥30 | ≥500 | ≥26 |
| 2 | 750–1499 | 26–33 | 500–999 | 22–29 | 200–499 | 14–25 |
| 3 | <750 | <26 | <500 | <22 | <200 | <14 |
∗ The stage is based primarily on the CD4+ T cell count; the CD4+ T cell count takes precedence over the CD4+ T cell percentage, and the percentage is considered only if the count is missing.
The stage of systemic HIV infection influences both the risk and the nature of neurological disease as well as likely etiologies, and hence CD4+ T cell count provides critical information that helps guide the evaluation ( Fig. 77.1 ). In early infection (corresponding to CD4+ T cell counts >500/μL) and in a setting of polyclonal immune activation, autoimmune disorders, such as inflammatory demyelinating neuropathies, may develop. During midstage infection (CD4+ T cell counts of 200–500/μL), primary HIV-related disorders, such as low-grade encephalitis or HIV-associated neurocognitive dysfunctions (HANDs), may become symptomatic, as may such infections as varicella-zoster virus (VZV) radiculitis (shingles). In advanced HIV infection (CD4+ T cell count <200/μL), the risk of dementia, myelopathy, and polyneuropathy increases further, and patients become increasingly vulnerable to major opportunistic infections (OIs), such as cerebral toxoplasmosis, progressive multifocal leukoencephalopathy (PML), and cryptococcal meningitis, as well as to neoplasms, in particular, such as primary central nervous system lymphoma (PCNSL).
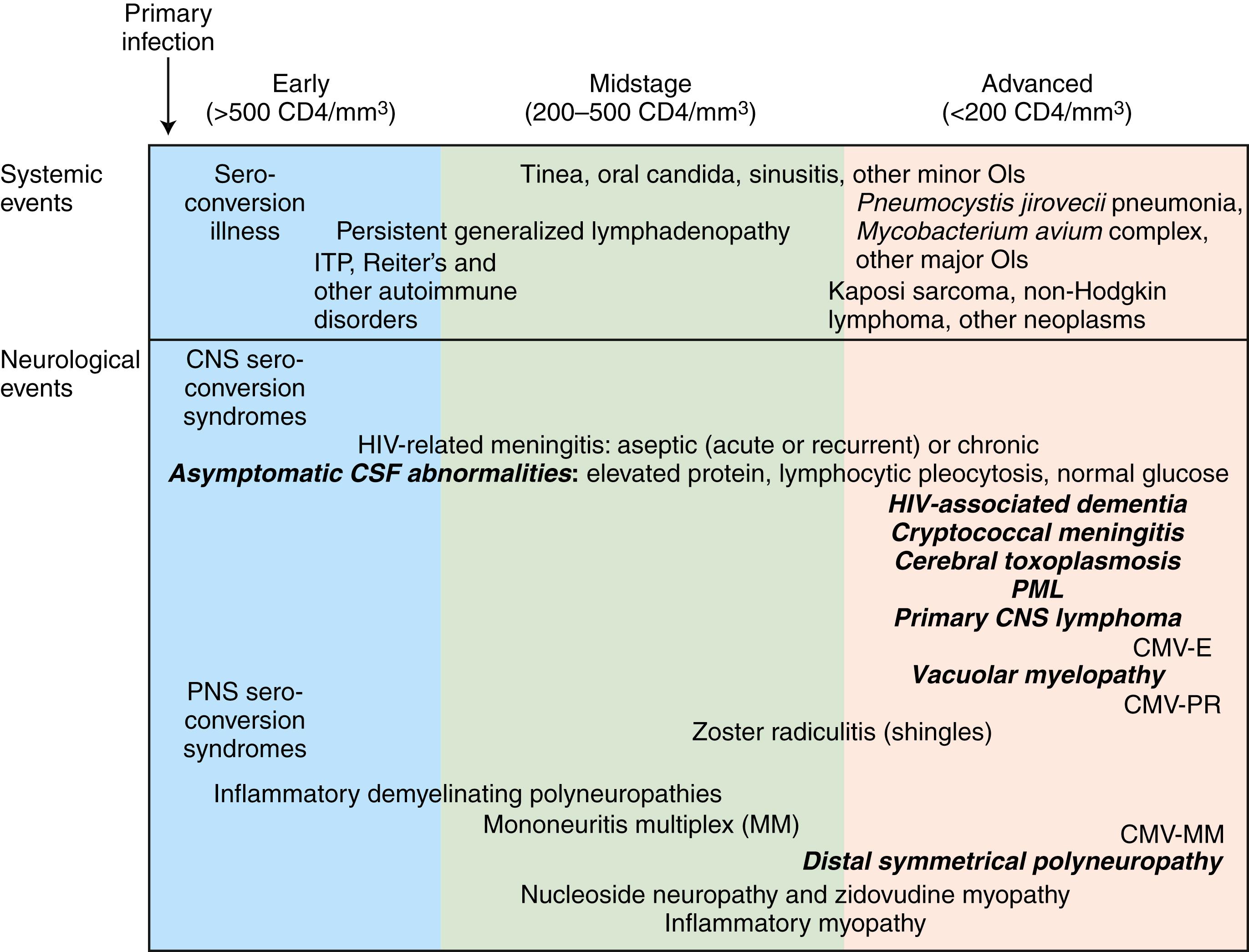
It is important to appreciate that the pathogenic mechanisms of neuro-HIV disease are complex and varied, they are multifactorial and multiphasic, and they are different at different stages of the disease ( ). Thus, it is essential to understand the typical clinical and immunological course of untreated HIV infection in order to fully appreciate the spectrum of HIV-associated neurological diseases.
There are a number of cardinal tenets of HIV-related neurological disease that need to be kept in mind in evaluating patients. First, neurological diseases are not infrequently the heralding manifestation of AIDS and may occur before HIV infection has been recognized. Second, HIV-related neurological disease may present in unusual fashions; common illnesses may present in atypical fashions. Third, despite upward of 36 years of experience with HIV/AIDS, new disorders attributed to the infection, including neurological illness, continue to be described ( ). Fourth, any part of the neuraxis may be affected by HIV infection. Fifth, the simultaneous presence of two or more HIV-related neurological disorders is frequently observed. Lastly, neurological disease occurring in the presence of HIV infection need not be related to the HIV infection or associated immunosuppression. Therefore, the clinician must have a high index of suspicion for HIV infection but not blindly attribute disease to the HIV infection.
HIV is transmitted by both homosexual and heterosexual contact; by sharing of contaminated needles in IDUs; by tainted blood and blood products; and from infected mothers to infants either intrapartum, perinatally, or via breast milk ( ). Virus that enters directly into the bloodstream via infected blood or blood products is trapped rapidly by the spleen and lymphoid tissue, whereas entry of virus through mucosal surface (sexual contact) requires mucosal dendritic cells or trafficking CD4+ T cells to first carry the virus to the regional lymphoid tissue. In either case, virus replication in CD4+ T cells intensifies (more rapidly with blood-borne infection) prior to development of the HIV-specific immune response, leading to a burst of viremia and rapid dissemination of the virus to other lymphoid organs, the brain, and other tissues. The initial viremic burst generally results in “acute HIV syndrome,” and these individuals often have a high level of viremia, measured in millions of HIV RNA virions per milliliter, that lasts for several weeks. Virtually all patients develop initial viremia during primary infection, even though they may remain asymptomatic or not recall any clinical event. HIV infection of the brain is believed to occur within 2 weeks of the infection.
It is estimated that 50%–70% of individuals with HIV infection experience an acute clinical syndrome 1–6 weeks after the primary infection. The syndrome is typical of an acute viral syndrome and has been likened to acute infectious mononucleosis. Fever, erythematous or maculopapular rash, headache, nausea, anorexia, lethargy, arthralgia, sore throat, and lymphadenopathy occur in different combinations in this syndrome.
Neurological manifestations occur in as many as 10% of cases at the time of initial HIV infection. The neurological presentation frequently involves multiple parts of the nervous system, although one part is usually dominant. Meningitis, meningoencephalitis of varying severity, seizures, myelopathy, and cranial and peripheral neuropathies have all been linked to the primary HIV infection. Laboratory analysis at this stage (with or without neurological disease) reveals cerebrospinal fluid (CSF) abnormalities with mild mononuclear pleocytosis and moderate rise in protein. Imaging of the brain is usually normal, whereas the electroencephalogram (EEG) may be diffusely or focally slow in brain-symptomatic cases. In most cases, primary infection with or without the acute HIV syndrome is followed by a prolonged period of clinical latency.
In most human viral infections, if the host survives, the virus is completely cleared from the body and a state of immunity against subsequent infection develops. HIV is very rarely fatal during the primary infection. Chronic infection subsequently develops with varying degrees of virus replication in a setting of polyclonal immune activation and progressive immunological impairment over a median of approximately 10 years before the infection becomes clinically evident. Establishment of chronic persistent infection is the biological hallmark of HIV disease and requires that the virus must evade control and elimination by the immune system. The virus accomplishes this via a number of mechanisms, including high rates of genomic mutation and molecular heterogeneity (see section on the HIV virus genome), sequestration of infected cells in immunologically privileged sites (e.g., brain), downregulation of human leukocyte antigen (HLA) class I molecules on the surface of HIV-infected cells by viral proteins (e.g., nef ), conformational masking of receptor-binding sites that fails to be neutralized by antibodies, and perhaps deletion of the initially expanded CD8+ T cell clones by the overwhelming initial burst of viremia and viral antigenemia. Thus, despite vigorous immune response and the marked downregulation of virus replication following primary HIV infection, HIV succeeds in establishing a state of chronic low-grade infection. Acute primary infection transitions to a relatively prolonged state of clinical latency. The level of steady-state plasma viremia (viral set point) in the latent phase probably determines the duration of this stage, or conversely, the rapidity of disease progression.
The half-life of a productively infected CD4+ T cell is approximately 1 day and that of a circulating virion is 30–60 minutes. Given the relatively steady level of plasma viremia and of infected cells in an individual, mathematical modeling indicates extremely large amounts of virus (over a billion or so copies) are produced and cleared from the circulation each day. Thus, clinical latency should not be confused with microbial latency. Vigorous virus replication, though with low-level viremia, is present during the period of clinical latency. Even the term “clinical latency” is misleading, because immunological progression of the HIV disease is generally relentless during this period.
Evidence indicates that the central nervous system (CNS) continues to harbor and mount a host reaction to the HIV throughout the asymptomatic or latent stage, yet without apparent immediate clinical sequelae ( ; Patel and Spudich, 2016). The CSF in patients with latent HIV infection generally shows abnormalities, including abnormal cell count, protein and immunoglobulin elevation, and local synthesis of anti-HIV antibodies within the CNS compartment; the intact virus can be recovered from the CSF. Pathological studies have shown evidence of inflammatory reactions in the CNS, with perivascular mononuclear cell infiltrations, although the HIV RNA burden, as measured by polymerase chain reaction (PCR) from CSF and brain samples, appears to be low at this stage. Neither overt nor subclinical cognitive or motor dysfunction appears to be common in the early latent stage. From a practical standpoint, the risk of cognitive decline in asymptomatic individuals is sufficiently small as to provide no basis for disability or disqualification from work based simply on HIV-positive status.
The length of the asymptomatic stage is determined by viral and host factors ( ). Some patients, who are termed “long-term nonprogressors or elite controllers” show little if any decline in CD4+ T cell counts over many years. These patients generally have extremely low levels of HIV RNA. Certain other patients remain entirely asymptomatic despite the fact that their CD4+ T cell counts show a progressive decline to extremely low levels. In these patients, the appearance of a systemic or CNS OI may be the first manifestation of HIV disease.
In untreated or inadequately treated patients, after a variable period of primary infection, usually measured in years, the CD4+ T cell counts fall below a critical level (<200/μL) and patients become susceptible to opportunistic diseases. The depletion of CD4+ T cells continues and may be profound. Yet these patients may survive for months or even years before, ultimately, succumbing to OIs or neoplasia.
HIV-associated neurological complications in late-stage HIV infection include primary manifestations, secondary complications related to the OIs and neoplasia, and complications arising from antiretroviral or prophylactic therapy. The primary neurological complications include HIV-associated dementia (HAD) or its less severe HAND forms—minor neurocognitive disorder (MND) and asymptomatic neurocognitive impairment (ANI), vacuolar myelopathy (VM), and HIV-associated distal sensory polyneuropathy (DSP). Generally, the nature and severity of illnesses (systemic and neurological) that occur change as the CD4+ count progressively declines. The frequent and life-threatening complications of HIV infection typically occur in patients with a significant decline in CD4+ T cell counts below 100/μL. Although the causative agents of secondary infections are characteristically opportunistic organisms such as Cryptococcus , cytomegalovirus (CMV), and other microorganisms that do not ordinarily cause disease in the absence of a compromised immune system, they also include common bacterial and mycobacterial pathogens. Secondary complications (systemic and neurological) are of great importance because of their frequency and high mortality. Approximately 80% of deaths among AIDS patients are a direct result of an infection other than HIV.
Neurons lack the conventional surface receptor for HIV binding and fusion, and therefore are not directly infected by the virus. The main cell types infected in the brain in vivo are those of the monocyte-macrophage lineage ( ). These include monocytes that have migrated into the brain from the peripheral blood, perivascular macrophages, and resident microglial cells. Although there have been reports of infrequent HIV infection of astrocytes, there is no convincing evidence that brain cells other than those of monocyte-macrophage lineage can be productively infected in vivo ( ). Nevertheless, in vivo infection of a neural cell line has been reported ( ), and it appears that galactosylceramide on neuronal surface is an essential component of the HIV gp120 receptor and that antibodies to galactosylceramide inhibit the entry of HIV into the neural cell lines. Some studies have demonstrated that viral entry is due, in part, to the ability of virus-infected and immune-activated macrophages to induce adhesion molecules such as vascular adhesion molecule-1 (VCAM-1) on brain endothelium. Other studies have demonstrated that HIV gp120 enhances the expression of intercellular adhesion molecule-1 (ICAM-1) in glial cells; these effects may facilitate entry of HIV-infected cells into the CNS and may promote syncytia formation ( Fig. 77.2 ).
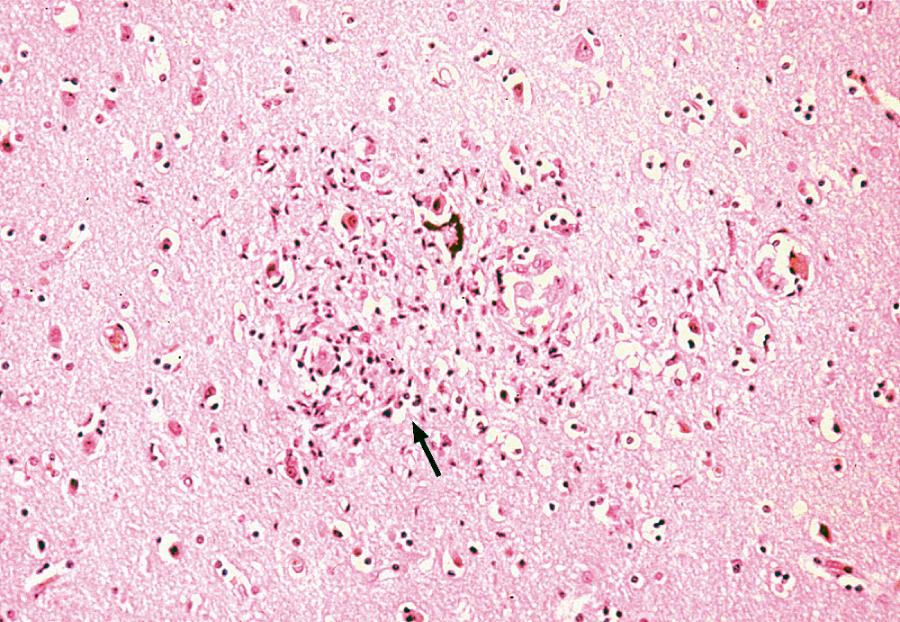
The R5 (CCR5 co-receptor) virus strains which are macrophage-tropic preferentially gain access into the brain rather than R4 (CXCR4 co-receptor) strains. HIV-infected individuals who are heterozygous for CCR5-del.32 (co-receptor gene with 32-base deletion) appear to be relatively protected against the development of HIV encephalopathy, as compared with persons with homozygous wild-type CCR5 ( ). Host HLA, chemokine receptor (CCR5, CXR4), and innate immunity loci and certain HIV envelope sequences are also linked with the clinical manifestation of HANDs ( ).
HIV-infected individuals may manifest white matter changes as well as neuronal loss ( ). Given the relative absence of evidence of HIV infection of neurons, it is unlikely that direct infection of these cells accounts for this cell loss. Rather, the HIV-mediated effects on brain tissue are thought to be due to a combination of indirect effects, either toxic or function-inhibitory of virus or viral antigens on neuronal cells and effects of a variety of neurotoxins released from the infiltrating monocytes, resident microglial cells, and astrocytes ( ). In this regard, it has been demonstrated that HIV-1 antigens—for example, nef and tat (viral core antigens)—can induce chemotaxis of leukocytes, including monocytes, into the CNS. Neurotoxins can be released from monocytes as a consequence of other HIV-associated infections or immune activation. Activated monocyte-derived neurotoxic factors have been reported to injure neurons via the N -methyl-d-aspartate (NMDA) receptor. Additionally, HIV gp120 shed by virus-infected monocytes, and a variety of cytokines, including tumor necrosis factor-alpha (TNF-α), interleukin (IL)-1, IL-6, interferon-α (IFN-α), and endothelin, can contribute directly or indirectly to the neurotoxic effects in HIV infection. Furthermore, infection or activation of monocyte lineage cells can result in increased production of eicosanoids, nitric oxide, and quinolinic acid, which may also contribute to the neurotoxicity.
Older HIV-infected individuals ( ) and individuals with the E4 allele for apolipoprotein E ( ) are reported to incur increased risk of HIV encephalitis and polyneuropathy. The fact that neuropsychiatric abnormalities may undergo remarkable and rapid improvement on the initiation of antiretroviral therapy, particularly in HIV-infected children, indicates that it is virus-driven, that the HIV or its products are involved in the neuropathogenesis of primary HIV neurological disorders, and that the dominant changes are either structural or functional at the synapses and dendritic spines, rather than indicative of neuronal death.
The dynamics of in vivo HIV production and turnover have been quantified using mathematical modeling in the setting of cART. Treatment with effective cART typically results in precipitous decline in the level of plasma viremia, often by well over 95% within a few weeks. The number of CD4+ T cells increases concurrently, suggesting that actively replicating virus is a major factor in CD4+ T cell depletion. Combination antiretroviral therapy therefore is the cornerstone of management of HIV infection. Suppression of HIV replication prolongs, as well as improves, the quality of life in patients with HIV infection.
Following the widespread use of cART in the United States from 1996, dramatic declines have been noted in the incidence of most AIDS-defining conditions, including neurological diseases (see earlier and following discussion). Successful cART with reconstitution of immune defense even has enabled some patients to discontinue secondary prophylaxis against CNS opportunistic pathogens, which had not been possible in the pre-cART era. It is recognized also that certain primary neurological diseases, in which the disease processes are driven by the HIV burden, can be prevented, delayed, or transformed into less severe forms by successful cART ( ), although the evolution of drug-resistant viral strains may eventually limit the sustained benefits of antiretroviral therapy. It also appears that in the future, as patients survive longer and develop increasing drug-resistant HIV mutations to newer antiretroviral therapy, the incidence of neuro-HIV diseases may begin to actually rise again.
Treatment decisions must take into account the fact that one is dealing with chronic infection that can only be controlled; eradication of HIV infection has not yet been possible. Unfortunately, some of the most important questions related to the treatment of HIV disease lack definitive or straightforward answers. Among them are the questions of when cART should be started in an individual, what the best initial regimen is, when a given regimen should be changed, and what changes should be made once the decision to change is made. Notwithstanding these difficulties, the physician and patient must come to a mutually agreeable plan based on the best available data. Given the complexity of this field, decisions regarding antiretroviral therapy are best made in consultation with experts.
At present, the US Department of Health and Human Services Guidelines panel recommends everyone with HIV infection be treated with cART and the therapy be initiated as soon as possible after diagnosis. Therapy has been associated with a decrease in disease progression in patients at all stages of HIV infection and it leads to a decrease in the risk of transmission of infection. All pregnant women regardless of their stages of HIV disease should receive cART. In addition, one may wish to administer therapy to uninfected individuals immediately following a high-risk exposure; a 6-week course of cART therapy decreases risk of infection. Additionally, pre-exposure prophylaxis in individuals at high risk of HIV infection should be considered. For patients diagnosed with an OI and HIV infection at the same time, one may consider a 2- to 3-week delay in the initiation of cART during which time treatment is focused on the OI. This delay may decrease the severity of any subsequent immune reconstitution inflammatory syndrome (IRIS) by lowering the antigenic burden of the OI. However, for patients with advanced HIV infection (CD4+ <50/μm), cART should be initiated as soon as possible.
Currently licensed drugs for the treatment of HIV infection are summarized in Table 77.2 . Fixed-dose combinations of many of these powerful drugs provide exciting new strategies for reducing pill burden, thus ensuring adherence and limiting the drug resistance (see Table 77.2 ).
| Drug | Dose | Toxicity | Remarks |
|---|---|---|---|
| NRTIs | |||
| Abacavir (Ziagen) | 300 mg bid | Rash, GI, LA | Abacavir-resistant HIV strains are typically also resistant to Lamivudine, Didanosine, and Zalcitabine |
| Emtricitabine (FTC, Emtriva) ∗ | 200 mg qd | Rash, GI | Generally well tolerated |
| Lamivudine (3Tc, Epivir) ∗ | 150 mg bid | HA, GI, P, LA | May cause hepatic steatosis |
| Zidovudine (Retrovir) ∗ | 300 mg bid | GI, HA, BM, rash | May cause myopathy, pancytopenia |
| NtRTIs | |||
| Tenofovir alafenamide (Vemlidy) | 25 mg qd | GI, renal | Commonly used in combination therapy |
| Tenofovir disoproxil fumarate (Viread) | 300 mg qd | Renal, osteomalacia | Commonly used in combination therapy |
| NNRTIs | |||
| Efavirenz (Sustiva) ∗ | 600 mg qhs | Rash, liver, CNS | — |
| Etravirine (Intelence) | 200 mg bid | Rash, GI | Taken with food |
| Nevirapine (Viramune) ∗ | 200 mg bid for 2 wk, then 200 mg bid or 400 mg extended release qd | Rash, liver | Close monitoring for initial 2 wk for rash and hepatitis |
| Rilpivirine (Edurant) | 25 mg qd | GI, liver, lipodystrophy | Mostly used in combination therapy |
| PIs | |||
| Atazanavir (Reyataz) | 400 mg qd or 300 mg qd + Ritonavir 100 qd when given with Efavirenz | GI, metabolic changes | Mostly used in combination therapy |
| Darunavir (Prezista) ∗ | 600 mg bid | Rash, liver, lipodystrophy | Mostly used in combination therapy |
| Ritonavir (Norvir) | 600 mg bid | GI, perioral paresthesia | Impairs metabolism of other PIs |
| Entry Inhibitors | |||
| Enfuvirtide (Fuzeon) ∗ | 90 mg SC bid | Local injection reaction, hypersensitivity | Increase incidence of bacterial pneumonia |
| Ibalizumab (Trogarzo) | Single-loading dose of 2000 mg followed by a maintenance dose of 800 mg q2 weekly | Rash, GI | Mostly used in combination therapy for multidrug-resistant HIV strains |
| Maraviroc (Selzentry) | 150–600 mg bid | Rash, fever, myalgia, cough | In combination therapy in adults infected with only CCR5-tropic HIV |
| Integrase Inhibitors | |||
| Bictegravir (available only in combination therapy, Biktarvy) ∗ | 25 mg qd | HA, GI | Available only in combination with Tenofovir alafenamide and Emtricitabine |
| Dolutegravir (Tivicay) | 50 mg qd in treatment-naïve and 50 mg bid in treatment-experienced cases | HA, hepatitis, CNS | Mostly used in combination therapy |
| Elvitegravir (available only in combination therapy, Stribild) ∗ | 1 tablet qd | GI, HA, upper respiratory infection | Available only in combination with Cobicistat ∗∗∗ , Tenofovir, and Emtricitabine |
| Raltegravir (Isentress) ∗ | 400 mg bid | HA, GI, myopathy | Mostly used in combination therapy |
| Combination Formulations | |||
| Atripla ∗∗ | Tenofovir disoproxil fumarate + Emtricitabine + Efavirenz | ||
| Biktarvy ∗∗ | Tenofovir alafenamide + Emtricitabine + Bictegravir | ||
| Combivir | Zidovudine + Lamivudine | ||
| Complera ∗∗ | Tenofovir disoproxil fumarate + Emtricitabine + Rilpivirine | ||
| Descovy | Tenofovir alafenamide + Emtricitabine | ||
| Epzicom | Abacavir + Lamivudine | ||
| Genvoya ∗∗ | Tenofovir alafenamide + Emtricitabine + Elvitegravir + Cobicistat ∗∗∗ | ||
| Kaletra | Lopinavir + Ritonavir | ||
| Odefsey ∗∗ | Tenofovir alafenamide + Emtricitabine + Rilpivirine | ||
| Prezcobix | Darunavir + Cobicistat ∗∗∗ | ||
| Stribild ∗∗ | Tenofovir disoproxil fumarate + Emtricitabine + Elvitegravir + Cobicistat ∗∗∗ | ||
| Symfi ∗∗ | Tenofovir disoproxil fumarate + Lamivudine + Efavirenz (600 mg) | ||
| Symfi Lo ∗∗ | Tenofovir disoproxil fumarate + Lamivudine + Efavirenz (400 mg) | ||
| Triumeq ∗∗ | Abacavir + Lamivudine + Dolutegravir | ||
| Trizivir | Zidovudine + Lamivudine + Abacavir | ||
| Truvada | Tenofovir disoproxil fumarate + Emtricitabine | ||
∗∗ Complete, once-daily, single tablet regimens.
∗∗∗ Inhibits cytochrome CYP3A enzyme and enhances the levels of antiretroviral agents that are metabolized by this enzyme.
Once the decision has been made to initiate therapy, the physician must decide which drugs to use as the first therapeutic regimen. Initial choice of drugs will determine the immediate response to therapy, and it will have implications regarding options for future therapeutic regimens. The goal of treatment is to achieve a viral load of less than 50 copies/μL within 4–6 months of initiation. The two options for initial therapy most commonly in use today involve a three- or four-drug regimen from two different antiretroviral classes (see Table 77.2 ). Recommendations regarding specific regimens are continuously evolving. In order to determine an optimal therapeutic regimen for initial therapy or for a patient on a failing regimen, one may attempt to measure antiretroviral drug susceptibility through genotyping or phenotyping of HIV quasispecies—to determine adequacy of dosing through measurement of drug levels. It is generally recommended that resistance testing be used in selecting initial therapy in settings where the risk of transmission of resistant virus is high and in determining new regimens for patients experiencing virological failure while on therapy.
For HIV-associated brain disease, drugs that exhibit good blood–brain barrier penetration should be preferred (see Table 77.2 ). Following the initiation of therapy, one should monitor virological (HIV RNA levels) and immunological (CD4+ T cell count) responses periodically. The HIV RNA levels in serum generally reflect viral levels in CSF, at least until late stages of the HIV disease. In terminal stages, the CNS compartment may harbor slightly different and divergent HIV strains, with different degrees of drug susceptibility ( ). Maximal suppression of viral replication is the goal of therapy, not just to prevent the disease progression but also to prevent the appearance of drug-resistant HIV quasispecies. The principles of current therapy for HIV infection are well articulated in publications of the US Department of Health and Human Services (see updates at www.hivatis.org ; www.nih.gov/phar/hiv-mgt ).
As cART has become widely available and has prolonged the life of patients with AIDS, an increasingly observed rare manifestation has been noted characterized by a paradoxical clinical deterioration following therapy. Referred to as “the immune reconstitution inflammatory syndrome,” it is defined as a paradoxical deterioration of the clinical status attributable to the recovery of the immune system. Although typically observed with OI, it may also occur as a response to noninfectious antigens. IRIS related to a known pre-existing infection or neoplasm is referred to as paradoxical IRIS , while IRIS associated with a previously undiagnosed condition is referred to as unmasked IRIS . IRIS has been described with a wide variety of systemic infections. In the CNS, IRIS has been well described with Cryptococcus neoformans infection, CMV retinitis, and PML, as well as other disorders. The term “immune reconstitution disease” is sometimes used to distinguish IRIS manifestations related to opportunistic diseases from IRIS manifestations related to autoimmune diseases. There is a suggestion that IRIS may attend primary HIV infection of the CNS. The symptoms mostly regress with continued cART, although a short course of high-dose parenteral corticosteroids has been suggested as effective in abrogating the response in some cases ( ).
The high prevalence and striking diversity of neurological disorders complicating AIDS were recognized early in the epidemic ( ). Clinical description of neurological OIs and malignancies predominated in early reports, but it also became clear that AIDS was associated with distinct neurological syndromes, such as dementia and painful neuropathy, that appeared to result from the HIV itself. It also became recognized that the risk of neurological complications increased with the progression of the HIV infection and decline of the CD4+ T cell counts. Clinically apparent neurological disease develops in approximately one-half of HIV-infected patients. Neuropathological abnormalities are nearly universal in patients dying with AIDS, suggesting subclinical disease, underdiagnosis, or both, in many cases. Neurological disorders cause significant morbidity and mortality, and they may be the AIDS-defining illnesses in previously asymptomatic HIV disease or, occasionally, herald unrecognized HIV infection. Nervous system complications may directly threaten life, as well as impair a patient’s ability to work or comply with complex cART regimens necessary to manage HIV disease optimally.
The neurological complications of HIV infection occur at all stages of the HIV disease ( ). Disorders of both the CNS and peripheral nervous system (PNS) can complicate HIV infection from the period after initial infection through the end stages of the severe immunosuppression. These neurological complications can be classified in a number of ways. A classification based on the underlying pathophysiology and HIV disease stages is summarized in Fig. 77.1 . Box 77.1 presents classification based on neuroanatomical localization, following the classic proven methods of the neurologist. Clinicians must be aware that more than one site of neural axis can be involved in the same HIV-infected patient at the same time. Furthermore, the clinical deficits from one site may be masked by another lesion higher in the neuraxis; appropriate investigations may be necessary to delineate the complete clinical diagnosis. The clinician must be vigilant also for other common conditions that are not necessarily associated with the HIV or AIDS.
Aseptic HIV meningitis
Cryptococcal meningitis
Tuberculous meningitis
Syphilitic meningitis
Listeria monocytogenes meningitis
Lymphomatous meningitis (metastatic)
HIV-associated neurocognitive disorders (HANDs)
HIV-associated dementia (HAD)
Mild neurocognitive disorder (MND)
Asymptomatic neurocognitive impairment (ANI)
Toxoplasmic encephalitis
Cytomegalovirus (CMV) encephalitis
Aspergillus encephalitis
Herpes encephalitis
Metabolic encephalopathy (alone or concomitantly)
Cerebral toxoplasmosis
Primary CNS lymphoma (PCNSL)
Progressive multifocal leukoencephalopathy (PML)
Cryptococcoma
Tuberculoma
Varicella-zoster virus (VZV) encephalitis
Stroke
Vacuolar myelopathy (VM)
CMV myeloradiculopathy
VZV myelitis
Spinal epidural or intradural lymphoma (metastatic)
Human T cell lymphocytotropic virus-1 (HTLV-1)-associated myelopathy
Acute inflammatory demyelinating polyradiculoneuropathy (AIDP)
Chronic inflammatory demyelinating polyradiculoneuropathy (CIDP)
Vasculitic neuropathy
Brachial plexopathy
Lumbosacral plexopathy
Cranial mononeuropathy
Multiple mononeuropathies
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here