Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Although weighing only about 19 grams, the testis is responsible for a complex array of neoplasms. The rapidly proliferating spermatogenic cells give rise to the majority of testicular tumors, 95% of which are of germ cell derivation. Most are malignant and usually occur in young men, but they can be cured by current therapies; therefore accurate diagnosis is essential. The supporting cells and interstitial cells of the testis are responsible for the less common sex cord–stromal tumors that comprise a disproportionate number of diagnostic problems. Some of these are associated with clinical syndromes that may be suspected based on the testicular pathology. A number of tumors of soft tissue origin may be identified in the paratestis, and secondary tumors are relatively frequent in both the testis and paratestis. The spectrum of lesions and the capacity of many tumors to mimic others make testicular neoplasia a continuing challenge to surgical pathologists, and this topic has been the subject of several recent reviews.
The currently recommended staging system for testicular cancer is the eighth edition of the Cancer Staging Manual of the American Joint Committee on Cancer (AJCC), published in 2017 ( Table 13.1 ), which replaces the seventh edition, published in 2010. Interpretation of two difficult areas of staging using 2010 AJCC criteria (tunica vaginalis invasion and hilar fat invasion without vascular invasion) was identified as the most significant area of variation in practice with regard to handling and reporting testicular cancer specimens in a recent survey. These points have been clarified in the 2017 AJCC staging manual. In the eighth edition, changes include subdivision of the T1 category, for seminoma only, into T1a (tumor less than 3 cm) and T1b (tumor 3 cm or larger), assignment of stage T2 for epididymal or hilar soft tissue invasion independent of vascular invasion, and classification of discontinuous involvement of the spermatic cord by vascular-lymphatic invasion as M1 disease. Serum marker studies play a key role in the evaluation of patients with testicular germ cell tumors, so the values of serum α-fetoprotein (AFP), human chorionic gonadotropin (hCG), and lactate dehydrogenase (LDH) are incorporated into the determination of the stage groupings.
| pT Category | Criteria |
|---|---|
| pTX | Unknown status of testis |
| pT0 | No apparent primary (includes scars) |
| pTis | Germ cell neoplasia in situ |
| pT1a | Tumor limited to testis (including rete testis invasion) without lymphovascular invasion, tumor smaller than 3 cm a |
| pT1b | Tumor limited to testis (including rete testis invasion) without lymphovascular invasion, tumor 3 cm or larger a |
| pT2 | Tumor either: limited to testis (including rete testis invasion) with lymphovascular invasion or involving hilar soft tissue or epididymis or penetrating visceral mesothelium of tunica albuginea with or without lymphovascular invasion |
| pT3 | Tumor invading spermatic cord (although discontinuous involvement of the spermatic cord soft tissue via a vascular thrombus is regarded as pM1) |
| pT4 | Tumor invading scrotum |
| pN Category | Criteria |
|---|---|
| pNX | Unknown nodal status |
| pN0 | No regional node involvement |
| pN1 | Node mass or single nodes ≤ 2 cm; ≤ 5 nodes involved; no node > 2 cm |
| pN2 | Node mass > 2 but < 5 cm; or > 5 nodes involved; none > 5 cm; or extranodal tumor |
| pN3 | Node mass > 5 cm |
| pM Category | Criteria |
|---|---|
| pM0 | No distant metastases |
| pM1a | Nonregional nodal or lung metastases |
| pM1b | Distant metastasis other than nonregional nodal or lung |
| S Category | Criteria |
|---|---|
| SX | No marker studies available |
| S0 | All marker studies normal |
| S1 | LDH < 1.5 × upper limit of normal and hCG < 5000 mIU/mL and AFP < 1000 ng/mL |
| S2 | LDH 1.5-10 × upper limit of normal or hCG 5000-50,000 mIU/mL or AFP 1000-10,000 ng/mL |
| S3 | LDH > 10 × upper limit of normal or hCG > 50,000 mIU/mL or AFP > 10,000 ng/mL |
| Stage Grouping | |
|---|---|
| 0 | pTis, N0, M0, S0 |
| I | pT1-4, N0, M0, SX |
| IA | pT1, N0, M0, S0 |
| IB | pT2-4, N0, M0, S0 |
| IS | Any pT, N0, M0, S1-3 |
| II | Any pT, N1-3, M0, SX |
| IIA | Any pT, N1, M0, S0-1 |
| IIB | Any pT, N2, M0, S0-1 |
| IIC | Any pT, N3, M0, S0-1 |
| III | Any pT, any N, M1, SX |
| IIIA | Any pT, any N, M1a, S0-1 |
| IIIB | Any pT, N1-3, M0, S2 or Any pT, any N, M1a, S2 |
| IIIC | Any pT, N1-3, M0, S3 or Any PT, any N, M1a, S3 or Any pT, any N, M1b, any S |
a Subclassification based on size criterion applies only to seminomas.
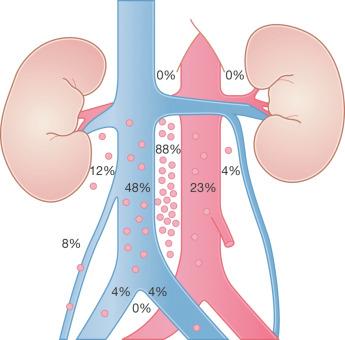
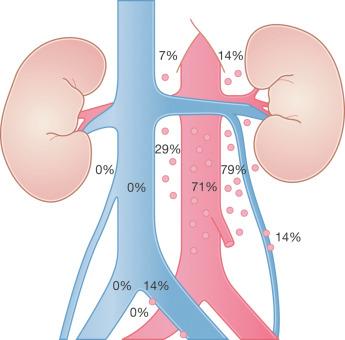
Testicular neoplasms usually first metastasize to retroperitoneal lymph nodes. There tends to be selective lymph node involvement with early-stage tumors, which depends on whether the right or left testis is involved. For right-sided tumors, the interaortocaval nodes at about the level of the second lumbar vertebra are usually first involved, although right paracaval and precaval involvement may also occur. In early stage involvement from right-sided tumors, there is absence of both suprahilar nodal involvement and involvement of the left paraaortic nodes below the inferior mesenteric artery ( Fig. 13.1 ). For left-sided tumors, the left paraaortic nodes, in an area bounded by the left ureter, left renal vein, aorta, and origin of the inferior mesenteric artery, are first involved. Suprahilar nodal metastases may be seen in early-stage disease from left-sided testicular tumors, in contrast with right-sided lesions ( Fig. 13.2 ). As metastases become more widespread, right-sided lesions develop suprahilar and contralateral spread and left-sided tumors develop interaortocaval and precaval involvement, as well as a greater frequency of suprahilar involvement. As the volume of retroperitoneal disease increases, retrograde involvement of iliac and inguinal nodes may be seen. Inguinal lymph node involvement may also be seen when the primary tumor has extended to the scrotal skin or a transcrotal approach was used for the primary resection. The extension of the primary tumor to the epididymis also correlates with the development of external iliac nodal spread. Eventually, supradiaphragmatic spread occurs to the mediastinum and supraclavicular and cervical lymph nodes, tending to involve the left supraclavicular nodes much more commonly than the right.
Seminoma tends to metastasize in an orderly pattern through lymphatics, whereas choriocarcinoma more frequently spreads by hematogenous routes. The other germ cell tumors, such as embryonal carcinoma, tend to have a lymphatic pattern of spread, although hematogenous spread can also be seen. Hematogenous spread is most commonly reflected by lung, liver, central nervous system, and bone involvement. Brain involvement is most common with choriocarcinoma and, perhaps unexpectedly, bone involvement with seminoma.
Gross examination and proper handling of the orchiectomy specimen are often neglected, and many diagnostic problems at the microscope can be traced to suboptimal processing of the gross specimen. Under the best circumstances, the testis and accompanying tunics and spermatic cord should be received fresh, dissected, and allowed to thoroughly fix before tissue blocks are submitted. What often happens, however, is that the urologist places the radical orchiectomy specimen intact into fixative, and only hours later is the specimen dissected. The testicular tunics are resistant to penetration of fixative, so this approach results in autolytic changes. It is preferable for the urologist to make a single, nearly through-and-through incision in the specimen before placing it into fixative if it is not feasible to send it to the laboratory immediately in the fresh state.
A radical orchiectomy specimen consists of the testis, tunica vaginalis, and a portion of spermatic cord. The specimen should be weighed, measured in three dimensions, and the length of the cord noted. We recommend examination of the spermatic cord next, before incision of the testis, to avoid the common contamination by “buttered” tumor of the cord, with submission of the cord resection margin and a cross-section adjacent to the testis, just superior to the head of the epididymis. The tunica vaginalis should then be incised, any abnormalities described, the quantity and nature of any intratunical fluid recorded, and the tunica albuginea carefully inspected and palpated for penetration by neoplasm. The testis should then be bisected in the plane of its long axis, through the testicular hilum, by a long, sharp knife. Fresh tissue may then be harvested for special studies such as cytogenetics, flow cytometry, electron microscopy, and molecular studies, although these are not routinely needed for diagnosis. Photographs may be obtained, and then multiple, serial, parallel cuts at 3-mm intervals should be made, leaving the tunica albuginea intact posteriorly to keep the specimen together. The specimen should then be placed in a generous volume of 10% neutral buffered formalin and allowed to thoroughly fix before further processing. After fixation, the neoplasm should be described and measured, with attention paid to the relationships to the tunica albuginea and the testicular hilum. Most examples of extratesticular spread occur by extension through the hilum. Multiple blocks of neoplasm should be submitted, because many tumors are quite heterogeneous. Blocks of all the different-appearing areas should be made, including hemorrhagic and necrotic areas, with attention to the testis/parenchymal interface, where lymphovascular invasion is often most apparent. A minimum of one block of neoplasm for every centimeter of maximum tumor dimension or a total of 10 blocks, whichever is greater, is a general rule of thumb. It is prudent to submit blocks quite generously if the gross appearance suggests seminoma, because the discovery of nonseminomatous elements may change therapy. Hence, small seminomas should be totally submitted, and at least 10 blocks of larger tumors (or one block for every centimeter of maximum tumor dimension) should be submitted. The nonneoplastic testis should also be sampled, as well as a block to include the testicular hilum. The epididymis should be incised by multiple, parallel cuts perpendicular to its long axis, any abnormalities noted, and the appropriate blocks submitted.
About 95% of testicular neoplasms are of germ cell origin. The 2016 World Health Organization (WHO) classification of testicular germ cell tumors ( Table 13.2 ) incorporates recent advances in the understanding of the pathogenesis of these tumors, specifically separating the germ cell neoplasia in situ–related and germ cell neoplasia in situ–unrelated tumors. This classification replaces previous classifications including the British Testicular Tumor Panel (BTTP) classification that was sometimes used in Europe.
Germ cell tumors
Germ cell neoplasia in situ
Germ cell tumors derived from germ cell neoplasia in situ
Tumors of a single histologic type
Seminoma (including seminoma with syncytiotrophoblast cells)
Nonseminomatous germ cell tumors
Embryonal carcinoma
Yolk sac tumor, postpubertal type
Trophoblastic tumors
Choriocarcinoma
Nonchoriocarcinomatous trophoblastic tumors
Teratoma, postpubertal type
Teratoma with somatic-type malignancy
Nonseminomatous germ cell tumors of more than one histologic type
Mixed germ cell tumors
Germ cell tumors of unknown type
Regressed germ cell tumors
Germ cell tumors unrelated to germ cell neoplasia in situ
Spermatocytic tumor
Teratoma, prepubertal type
Mixed teratoma and yolk sac tumor, prepubertal type
Yolk sac tumor, prepubertal type
Pure tumors
Leydig cell tumor
Sertoli cell tumor, not otherwise specified
Large cell calcifying Sertoli cell tumor
Intratubular large cell hyalinizing Sertoli cell neoplasia
Granulosa cell tumor
Adult granulosa cell tumor
Juvenile granulosa cell tumor
Tumors in the fibroma-thecoma group
Myoid gonadal stromal tumor
Mixed and unclassified sex cord–stromal tumors
Gonadoblastoma
Ovarian-type epithelial tumors
Juvenile xanthogranuloma
Hemangioma and other stromal tumors
Hematolymphoid tumors
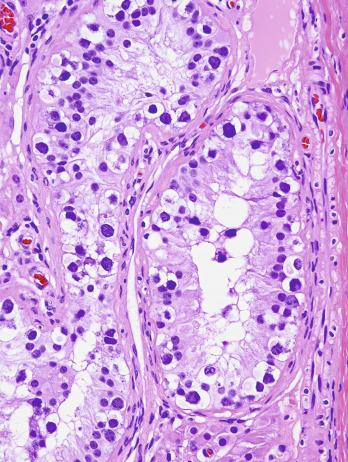
The histogenesis of testicular germ cell tumors has been clarified over the past several decades by several clinical, morphologic, and immunohistochemical observations. Gene expression profiling and other molecular techniques have allowed a great increase in understanding of the molecular changes in the most common testicular cancers, the germ cell neoplasia in situ–associated tumors, which have also been described as type II germ cell tumors. Perhaps foremost in importance is the recognition that all of the adult germ cell tumors, with the exceptions of spermatocytic tumor, rare benign teratomas (a category now including epidermoid and dermoid cysts), and even more rare yolk sac tumors, are derived from a common precursor that Skakkebaek originally recognized and described as “carcinoma in situ” of the testis. Intratubular germ cell neoplasia of the unclassified type (IGCNU) had later been considered the appropriate term for this lesion, given the nonepithelial nature of the constituent cells, but the preferred term in the recent WHO classification is germ cell neoplasia in situ (GCNIS). GCNIS consists of a basilar proliferation of seminoma-like cells (occupying the “spermatogonial niche”) with clear cytoplasm and enlarged, hyperchromatic nuclei having one or two prominent nucleoli ( Fig. 13.3 ). Additionally it has many features, apart from its light microscopic appearance, that it shares with seminoma, including ultrastructure, immunohistochemical reactions with various antibodies (M 2 A/podoplanin/D2-40, TRA-1-60, placental-like alkaline phosphatase [PLAP], glutathione-S-transferase [isoenzyme π], the 14–3–3 beta protein, OCT3/4, NANOG, stem cell factor, TSPY protein, SOX17 +/SOX2– immunophenotype), its DNA content, the number of nucleolar organizer regions, and lectin-binding patterns.
The strong similarities between GCNIS and seminoma imply that seminoma is also a precursor for other germ cell tumors. This interpretation is supported by several morphologic, immunohistochemical, and molecular observations. These include autopsy studies showing nonseminomatous elements in patients who died of metastatic germ cell neoplasm after orchiectomy of pure testicular seminoma. Additionally, seminoma may undergo subtle transition to either embryonal carcinoma or yolk sac tumor. Also, 10% to 20% of seminomas contain syncytiotrophoblast cells, and some trophoblastic hormone–containing cells in seminoma are not easily distinguished histologically from the surrounding seminoma cells. Ultrastructural studies of seminoma have demonstrated evidence of epithelial differentiation ( seminoma with early carcinomatous features ) in some light microscopically typical cases. Furthermore, the DNA content of seminoma is consistently higher than in nonseminomatous germ cell tumors, suggesting that nonseminomatous tumors evolve from seminoma as a consequence of gene loss, perhaps caused by loss of cancer suppressor genes. Karyotypic analyses have shown a striking tendency for certain chromosomes to be in parallel excess or deficiency in seminoma and the nonseminomatous germ cell tumors, and loss of heterozygosity studies also show similar patterns of allelic imbalance between coexisting seminoma and nonseminoma in the testis. These data indicate that seminomas transform to nonseminomatous tumors.
Genetic changes precede the development of an invasive germ cell tumor from GCNIS. Although overrepresentation of gene sequences from the short arm of chromosome 12, mostly in the form of an isochromosome [i(12p)], is consistent in invasive germ cell tumors of adult patients, such overrepresentation is not found in the associated GCNIS. It is believed that additional 12p sequences are essential for invasive growth by inhibiting apoptosis and thereby permitting survival of invasive tumor cells outside of the microenvironment of the seminiferous tubules. There are marked similarities between many of the genetic changes in GCNIS and the associated invasive tumor, in support of the precursor role of the former. Immunohistochemical study has shown that loss of the cell cycle–dependent kinase inhibitors, p18 and p21, accompanies invasive growth, as does gain of the ubiquitin ligase murine double minute-2 (mdm-2) and increased production of cyclin E.
Transformation of GCNIS to nonseminomatous tumor may apparently occur at the time of invasion, because pure embryonal carcinoma, yolk sac tumor, or choriocarcinoma are associated with GCNIS. Alternatively, these elements may overgrow a small focus of seminoma from which they arose. The common occurrence of seminoma with nonseminomatous elements also supports transformation from invasive seminoma. Allelotyping analysis supports this concept, as loss of heterozygosity is seen at progressively more loci from GCNIS to seminoma and then to embryonal carcinoma. Loss of heterozygosity at various loci was frequently shared among the seminoma and embryonal carcinoma components, and heterozygosity was retained in GCNIS, further supporting that the embryonal carcinoma component arose from the seminoma.
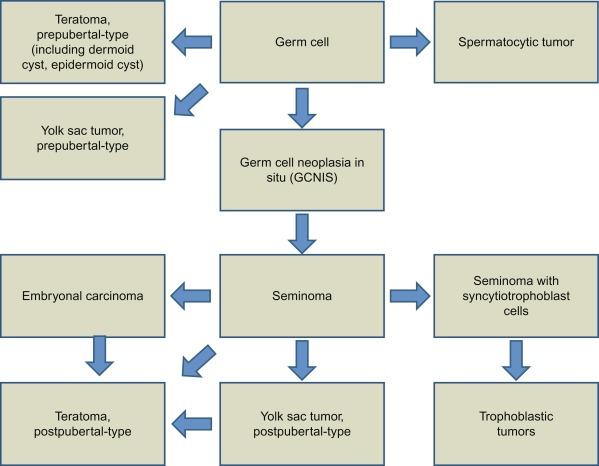
Additional data show that GCNIS adjacent to seminoma or nonseminomatous tumors shares chromosomal abnormalities with the invasive tumor that differ depending on whether the adjacent tumor is seminomatous or nonseminomatous, an observation that supports genetic transformation within the tubules before morphologic change. Collectively, these observations led to a revised model of testicular germ cell tumor histogenesis based on the tetrahedron model proposed by Srigley et al. ( Fig. 13.4 ).
The histogenesis of testicular germ cell tumors in children (excluding those with disorders of sex development) is different from that of postpubertal patients. This is reflected in epidemiologic studies that documented a progressive increase in the incidence of testicular germ cell tumors throughout the twentieth century in postpubertal patients but not in the prepubertal group. Unlike the postpubertal tumors, the examples in children (confined to two types of neoplasm, yolk sac tumor and teratoma) lack association with GCNIS. Furthermore, the teratomas have a diploid DNA content and a normal karyotype, unlike those of postpubertal patients, and the yolk sac tumors lack consistent 12p abnormalities. The pediatric germ cell tumors therefore have a fundamentally different pathogenesis. These observations and others have led one group to propose that there are five fundamentally different forms of germ cell tumor: type I represented by the pediatric types, type II consisting of the usual postpubertal germ cell tumors, type III consisting only of spermatocytic tumor, type IV represented by ovarian dermoid cyst, and type V consisting of gestational choriocarcinoma. Each has its own unique pattern of gene activation and genomic imprinting.
Germ cell tumors of the testis (with the notable exception of spermatocytic tumor) occur mostly in young men, with the incidence accelerating rapidly after puberty and peaking close to 30 years of age ( Fig. 13.5 ). There is a small peak in early childhood, but many cases of “testicular cancer” in elderly men correspond to lymphoma or secondary tumors rather than to germ cell tumors ( Fig. 13.5 ). Whites have a much higher frequency of testicular germ cell tumors than do non-whites, with the exception of the Maori of New Zealand who have an incidence comparable to white populations. Native Hawaiians, Native Alaskans, and Native Americans are also at higher risk than other non-whites. The rates of increase of testicular cancer in black men (+ 1.67%/year), Hispanic men (+ 2.94%/year), and American Indian/Alaska Native men (+ 2.96%/year) in the United States are, however, increasing faster than that of white men (+ 1.23%/year) in recent decades. Denmark and Switzerland have the highest rates of testicular cancer, about 9 cases per 100,000 males per year, compared with the rate in the US white population of 5 to 6 per 100,000 males. The rates in Africans and Asians are generally about 1 per 100,000 males. In a study of over 1 million Israeli males, risk was strongly associated with birthplace, with odds ratios of 0.10 for those born in North Africa and 0.35 for those born in Asia compared with those born in Israel.
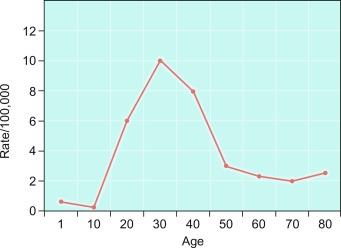
The incidence of most testicular germ cell tumors increased steadily in the United States during the twentieth century, and recent National Cancer Institute Surveillance, Epidemiology, and End Results Program (SEER) data indicate an increase in the rate of testis cancer in 15- to 39-year-old males in the United States at a time when the incidences of most other cancers in this age group are decreasing. Increasing testicular cancer rates have been noted in several other countries, including Denmark, Norway, England, Germany, Scotland, New Zealand, Australia, Canada, Iceland, and Japan, although, as noted, the rate remained steady in children. Eastern Europe, which previously had a lower rate, has had the most rapid increase in testicular cancer incidence in recent years and now has a rate similar to Northern and Western Europe.
Numerous studies have demonstrated a higher frequency of testicular germ cell tumors in professional workers or those of higher socioeconomic class compared with laborers or those of lower socioeconomic status. Higher rates have also been observed in those with occupational exposures to fertilizers, phenols, heat, smoke, or fumes; farm workers; draftsmen; those involved in food manufacture and preparation; leather workers; pesticide applicators; those exposed to insect repellants; metalworkers; police exposed to handheld radar; aircraft repairmen; motor vehicle mechanics; fishermen; electrical workers, paper and printing workers, and foresters; men of tall stature; and physicians. Other studies have implicated in utero exposure to high estrogen levels, dietary iron, testicular trauma, various HLA-haplotypes, a family history of breast cancer, early puberty, early birth order, dizygotic twinship, ichthyosis, Marfan syndrome, the Li-Fraumeni syndrome, the dysplastic nevus syndrome, and HIV infection. One study has shown a correlation between testicular cancer and a variant allele of the glutathione-S-transferase π gene. Another study demonstrated increased risk in the sons of fathers who were wood processors, metalworkers, or employed in the food product industry. Recently, baldness and a history of severe acne, both associated with high androgen levels at puberty, have been associated with testicular germ cell tumors. Marijuana use has been associated with a 1.7 to 2.2-fold increased risk for testicular nonseminomatous germ cell tumors. Most of these associations, however, are weak and fail to account for the general increase in testicular germ cell tumors. It is hypothesized that important causative factors in testicular cancer occur in the antenatal period, with a protective effect in European countries for men born during the World War II era. This protective effect leads to the hypothesis that testicular cancer is causally related to prosperity, probably secondary to in utero effects. Environmental endocrine-disrupting chemicals have attracted interest as a possible causative factor for germ cell tumors (and the other components of the proposed testicular dysgenesis syndrome). Phthalates, chemicals present in plastics, have attracted interest as an environmental endocrine disruptor, but a causative role remains unproven. A potential unifying thread to several of the most compelling observations is that maternal obesity (leading to elevated estrogens) and/or maternal exposure to estrogen simulators or antiandrogens lead to testicular maldevelopment with impaired Sertoli cell function that is key to germ cell neoplasia. The use of alcohol and tobacco, prior vasectomy, radiation exposure, and maternal and paternal occupational pesticide exposure have not been associated with testicular germ cell tumors.
Despite the weak correlation of most etiologic factors with testicular germ cell tumors, four contributing factors are proven: cryptorchidism, prior testicular germ cell tumor, family history of testicular germ cell tumors, and certain disorders of sex development. An estimate of the increased risks associated with these disorders is provided in Table 13.3 .
| Condition | Estimated Increased Risk | References |
|---|---|---|
| Cryptorchidism | 3.5-5X | Pottern, 1985 Giwercman, 1987 |
| Prior testicular germ cell tumor | 5-10X | Giwercman, 1991 Dieckmann, 1999 Giwercman, 1987 Loy, 1993 |
| Family history (first-degree male relative) | 3-10X | Dieckmann, 1997 Fuller, 1986 Tollerud, 1985 Forman, 1992 |
| Gonadal dysgenesis with a Y-chromosome | 50X a | Giwercman, 1991 Rutgers, 1987 Hughesdon, 1970 |
| Androgen insensitivity syndrome | 15X | Giwercman, 1991 Manuel, 1976 Morris, 1953 Rutgers, 1991 Rutgers, 1991 |
An increased frequency of cryptorchidism, varying from 6.5% to 14.5%, has been found in patients with testicular germ cell tumors, which has led to increased risk calculations of 2.5 to 35 times higher among cryptorchid patients. Such risk does not manifest before 20 years of age and is probably most accurately assessed as 3.5 to 5.0 times increased over a control population. If cryptorchidism is unilateral, the noncryptorchid testis is also at increased risk for a testicular germ cell tumor, although at a lower rate than the cryptorchid testis. Ectopia alone cannot explain the association of cryptorchidism and testicular germ cell tumor, a fact reinforced by the failure of orchiopexy to reduce the risk to that of the general population (although there is probably insufficient experience with orchidopexy in the very young to rule out an ameliorating effect). Early surgical correction, at 6 to 18 months of age, has been recommended as treatment for cryptorchidism, as the risk reduction is related to age, with a relative risk for developing cancer of 2.2 if orchiopexy is performed before 13 years of age compared with 5.4 if performed after 13 years of age.
It seems likely that cryptorchidism is a marker of patients with a general defect in testicular and genital development and that cryptorchid testes are “dysgenetic,” as supported by abnormalities of the external genitalia or sex chromosomes in some cryptorchid patients with germ cell tumors. This has been conceptualized as the “testicular dysgenesis syndrome,” a collection of findings including cryptorchidism, infertility, disorders of sex development, GCNIS, and germ cell tumors that may share common etiologic factors. A distinctive array of testicular lesions may be seen in the parenchyma adjacent to germ cell tumors, including Leydig cell hyperplasia, microlithiasis, angiopathy, Sertoli cell nodules, tubular atrophy, and multinucleated spermatogonia, supporting an underlying developmental problem. Cryptorchidism may disproportionately predispose to seminoma compared with nonseminomatous tumors. GCNIS has been estimated to be present in about 2% to 4% of patients with cryptorchidism. At least 50% of such patients develop germ cell tumor within 5 years. Thus bilateral testicular biopsy at 18 to 20 years of age has been recommended for cryptorchid patients. A negative result is good evidence of no increased risk, although there are infrequent false-negative results. A positive biopsy should prompt orchiectomy of the affected testis. In testes with extreme atrophy, the biopsy should be directed to sample the region near the rete testis. Apart from germ cell tumors, cryptorchid testes have an increased frequency of nodules composed of small tubules lined by immature Sertoli cells, often with central deposits of basement membrane. These Sertoli cell nodules have been termed Pick adenoma, which is a misnomer because they are not true neoplasms.
A second germ cell tumor occurs in the remaining testis of 1% to 5% of patients with a previous germ cell tumor. The risk for a second germ cell tumor is higher in patients with seminoma, especially in men 30 years of age or younger. Similarly, there is a 3.2% to 6.6% frequency of GCNIS in the opposite testis of patients with a germ cell tumor. There is a low risk in the absence of GCNIS on biopsy because of occasional false-negative biopsies. In one study, 5 of 1859 patients (0.3%) who had negative testicular biopsies opposite a germ cell tumor developed a second tumor on follow-up. Sensitivity is improved if three biopsies of the contralateral testis are performed. If the residual testis is either atrophic or cryptorchid, the risk is greater, with a 23% frequency of GCNIS. Unfortunately, almost 50% of cases of contralateral GCNIS would be missed if contralateral biopsies were restricted to patients with atrophy or cryptorchidism. One study suggested that atrophy rather than maldescent is the important predictor of contralateral GCNIS. Young (but postpubertal) age at onset of the first tumor and bilateral cryptorchidism are also associated with an increased risk for bilateral occurrence. It is estimated that biopsy of an atrophic testis opposite a germ cell tumor in a patient younger than 31 years of age will detect GCNIS in one-third. Prepubertal testicular germ cell tumors, in contrast, do not appear to be associated with an elevated risk for second gonadal cancers.
About 50% of second primary tumors of the testis occur within 3 to 5 years after diagnosis of the initial germ cell tumor, with a mean of 5.6 to 6.5 years, but intervals of more than a decade can occur. Concordant or discordant neoplastic types may occur, with some tendency for concordance of pure seminoma. The risk for bilateral tumors is increased about fourfold with a positive family history. Chemotherapy administered for the first tumor has been observed to decrease the risk for a contralateral tumor, but not in all studies. Contralateral tumors may occur even in the absence of GCNIS in biopsies of the opposite testis performed at the time of the first germ cell tumor diagnosis. An increased frequency of rare alleles of the HRAS gene is seen in patients with testicular germ cell tumors, and such alleles are associated with bilaterality and early age of onset. Androgen receptor polymorphisms (with high or low numbers of CAG repeats) have been associated with testicular cancer risk. In one study, 64% of bilateral germ cell tumors had KIT mutations compared with 6% of unilateral tumors, suggesting a causative role for KIT mutations occurring early in embryonal development in patients with bilateral tumors. Genetic associations with germ cell tumor risk have been reviewed, and the association with KIT signaling has been confirmed with genome-wide association studies that have found associations with five molecular pathways: KIT signaling, other pathways of male germ cell development, telomerase function, microtubule assembly, and DNA damage repair.
First-degree male relatives of patients with germ cell tumor of the testis have a 3 to 10 times greater risk for a testicular germ cell tumor than the general population. The risk is highest for brothers (10 times), intermediate for sons (6 times), and lowest for fathers (4 times). Also, a family history of testicular germ cell tumor is associated with an 8% to 14% frequency of bilaterality, compared with the 1% to 5% frequency in the general population of patients with testicular germ cell tumors. The occurrence of an unexpected number of testicular germ cell tumors in the relatives of children with soft tissue sarcomas has raised the question of whether testicular germ cell tumors may represent part of the spectrum of the Li-Fraumeni cancer syndrome. It appears, however, that neither germline nor somatic TP53 mutations occur in these cases. Immunohistochemical demonstration of p53 protein therefore indicates overexpression of nonmutated protein. Segregation analysis of data regarding familial cases has suggested the presence of a major gene that conveys risk in a recessive model. Genetic linkage analysis has implicated a susceptibility gene localized to Xq27 in one study, but this was not confirmed in a second. Single nucleotide polymorphisms in BAK1, DMRT1, KITLG, and TERT-CLPTM1L genes have been associated with testis cancer risk odds ratios of 1.6 to 2.3. In a study including 147 families with either two or more cases of testis cancer or one case of bilateral testis cancer, no rare, highly penetrant susceptibility gene was identified. It is believed that, instead, familial testis cancer most likely results from multiple, common, low-penetrance alleles in a manner more similar to type 2 diabetes or familial prostate cancer than to the more well-known adult cancer susceptibility disorders with high-penetrance genes associated with breast, ovarian, and colorectal carcinoma. No increase in overall risk for nontesticular cancers was seen in relatives of familial testicular tumor patients in a recent study of 1041 first-degree relatives from 66 multiple case testicular germ cell tumor families, suggesting that tumors other than testis cancer are not part of the association.
Patients with some disorders of sex development are at increased risk for germ cell tumors. Patients with gonadal dysgenesis in the presence of a Y-chromosome, including patients with pure 46,XY gonadal dysgenesis (Swyer syndrome) and mixed gonadal dysgenesis, commonly develop gonadal germ cell tumors. About 25% to 30% of such patients develop gonadoblastoma, and this may serve as the precursor lesion for the development of an invasive germ cell tumor (discussed later in this chapter). GCNIS is also present in about 8% of children and adolescents with gonadal dysgenesis. Because an invasive tumor may develop in childhood, gonadectomy is indicated as soon as the diagnosis is established. Patients with androgen insensitivity syndrome, caused by various mutations in the androgen receptor gene, develop a malignant germ cell tumor in 5% to 10% of cases overall. The tumor usually develops after puberty, and this may permit delayed gonadectomy until full feminization has occurred, although this remains controversial because of an occasional case of invasive germ cell tumor developing at an early age. Delay of prophylactic gonadectomy beyond the early postpubescent period in patients with androgen insensitivity syndrome (AIS) is risky, with a 22% frequency of malignant germ cell tumor in such patients beyond 30 years of age. Not all testicular masses in patients with the AIS are germ cell tumors; these patients commonly develop hamartomatous nodules composed of Sertoli cell–lined tubules with intervening clusters of Leydig cells in the interstitium, as well as pure Sertoli cell adenomas and occasional juvenile granulosa cell tumors. In addition to patients with mutations in the androgen receptor gene, there is a single report of GCNIS in a phenotypic female patient with an XY karyotype and a nonsense mutation in the gene for steroidogenic acute regulatory protein.
Patients with infertility have an increased but less than 1% frequency of testicular germ cell tumor. The relative risk for germ cell tumor in patients with infertility is much lower than that associated with cryptorchidism or a history of contralateral germ cell tumor. It is not clear, however, if infertility is a risk factor for germ cell tumor that is independent of cryptorchidism or gonadal dysgenesis. Impaired semen quality, hypospadias, cryptorchidism, and germ cell tumors are the components of the testicular dysgenesis syndrome theoretical construct and are each related to the other. Semen quality may be a highly sensitive marker for environmental exposures, which are also related to germ cell tumor incidence.
Testicular microlithiasis, seen ultrasonographically as hyperechogenic foci without shadowing, is observed in about 13% of patients evaluated by this method. Rare cases of germ cell tumor have been reported in patients, including teenage boys, being followed for testicular microlithiasis. It is unusual for a patient with testicular microlithiasis but without other risk factors to have GCNIS on biopsy, so biopsy is only recommended if other risk factors, such as infertility, cryptorchidism, or contralateral tumor, exist.
Inguinal hernia and genital malformations other than cryptorchidism including hypospadias have also been associated with testicular germ cell tumor risk, although not as strongly as cryptorchidism.
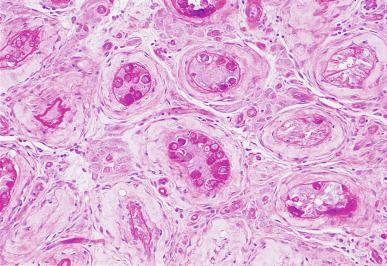
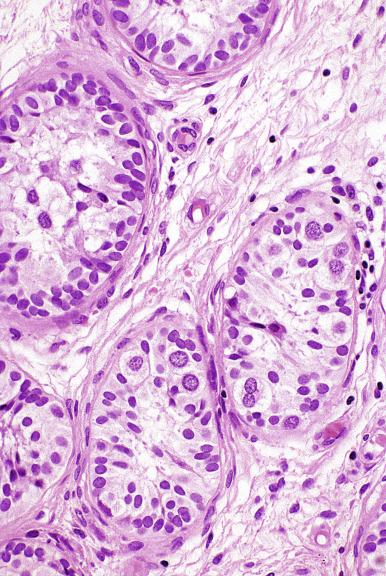
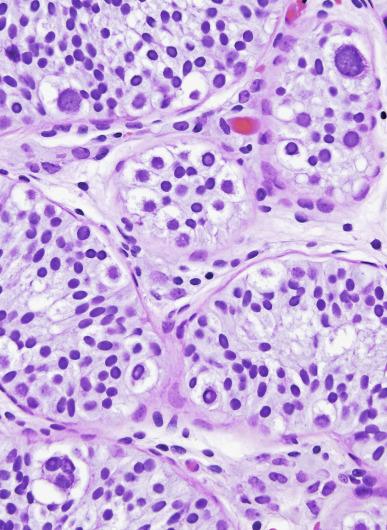
Grossly, the testis with GCNIS may be unremarkable or appear atrophic and fibrotic. Microscopically, intratubular germ cell neoplasia consists of a proliferation of undifferentiated germ cells resembling primitive gonocytes or, less frequently, may be composed of cells of a specific neoplastic type, such as intratubular embryonal carcinoma. The primitive gonocyte-like form of intratubular germ cell neoplasia is confined to the basilar aspect of the seminiferous tubules (in the so-called “spermatogonial niche”) and is associated with all types of the postpubertal germ cell tumors, except for spermatocytic tumor and the rare benign teratomas (including dermoid/epidermoid cyst) and prepubertal-type yolk sac tumors. GCNIS cells have enlarged, hyperchromatic nuclei, often with one or two prominent nucleoli, thickened nuclear membranes, and clear cytoplasm ( Fig. 13.3 ). The median nuclear diameter is 9.7 μm, compared with a median nuclear diameter of spermatogonia of 6.5 μm. Spermatogenesis in the affected tubules is usually decreased or absent, and the tubules may have a thickened peritubular basement membrane. Sertoli cells are often displaced luminally ( Fig. 13.3 ). The distribution of GCNIS is characteristically patchy, and adjacent profiles of seminiferous tubules may appear unremarkable with intact spermatogenesis ( Fig. 13.6 ). The patchy distribution has implications for biopsy diagnosis, as a small volume sample may result in a false-negative diagnosis. Leydig cell hyperplasia may occur in the interstitium. GCNIS often spreads into the rete testis in a “Pagetoid” fashion, intermixing with nonneoplastic epithelium, which is displaced toward the lumen ( Fig. 13.7 ). The strong similarity of the cells of seminoma and GCNIS suggests that GCNIS could be termed intratubular seminoma; however, this term may falsely suggest that GCNIS is the precursor lesion only for seminoma rather than for virtually all postpubertal germ cell tumors. The term intratubular seminoma has been applied to those proliferations of GCNIS-like cells that fill and distend seminiferous tubules ( Fig. 13.8 ), although such lesions may simply be a more advanced stage of GCNIS.
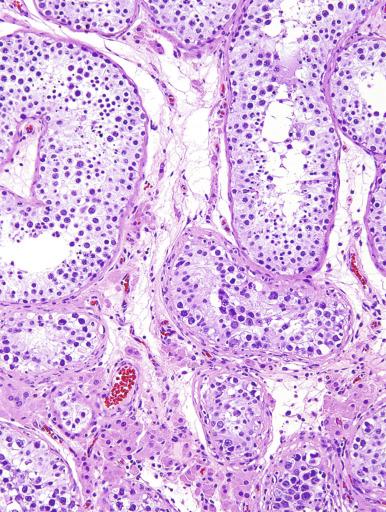
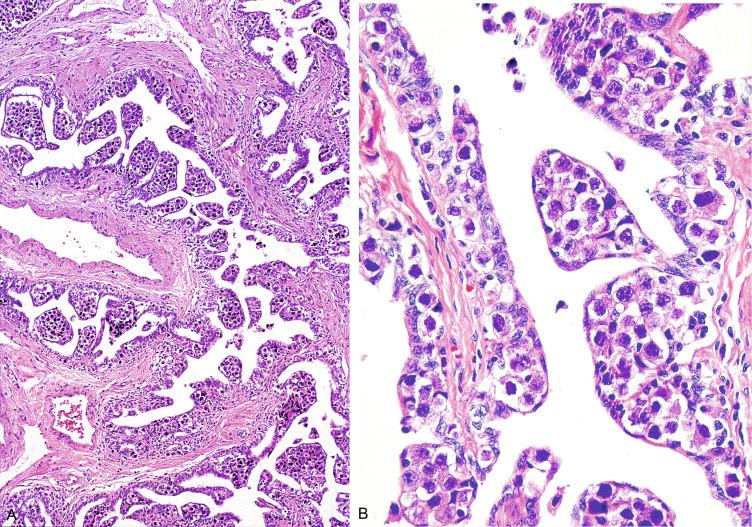
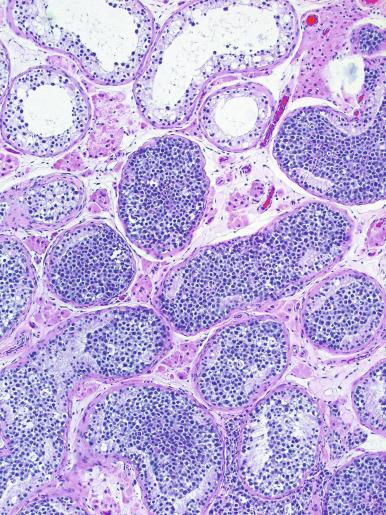
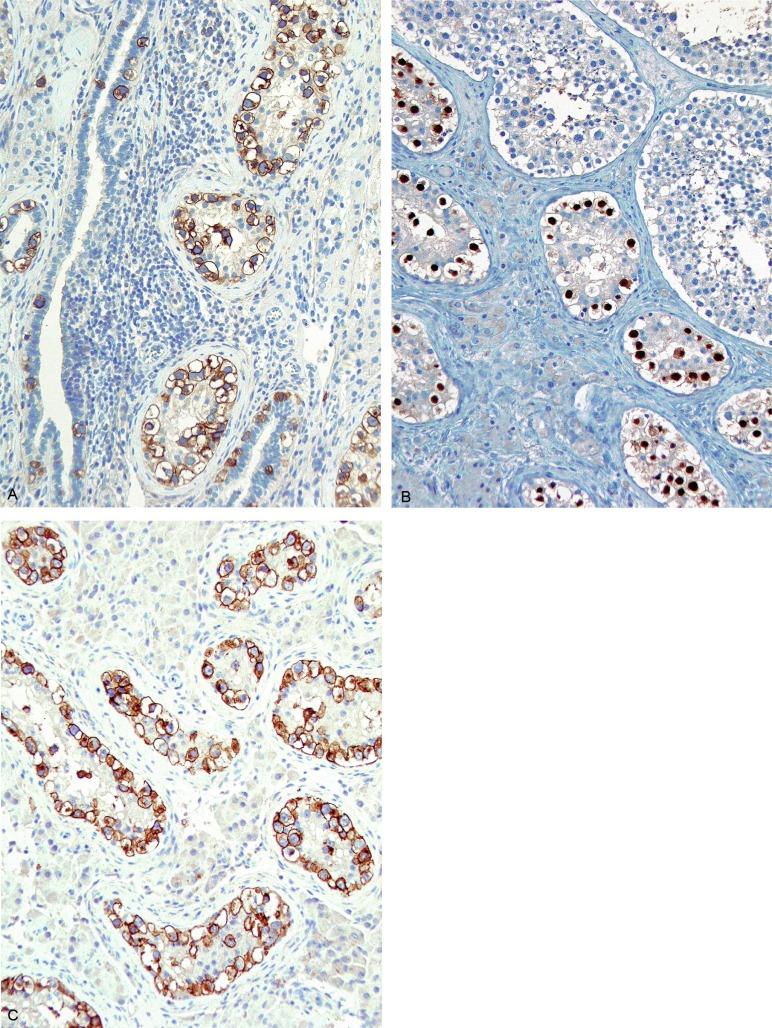
Glycogen is present in the cytoplasm of 98% of cases of GCNIS ( Fig. 13.9 ), and its demonstration is diagnostically helpful but nonspecific because nonneoplastic spermatogonia and Sertoli cells may also contain glycogen. The current best immunohistochemical marker for GCNIS is OCT3/4, a nuclear transcription factor with a key role in maintaining pluripotency in embryonic stem cells. OCT3/4 is uniformly sensitive for GCNIS ( Fig. 13.10 ). It, as well as other GCNIS markers, is also seen in germ cells with delayed maturation in gonadal dysgenesis, Down syndrome, and undervirilization syndromes that have different features from GCNIS. PLAP is also a good marker for GCNIS. It highlights the cytoplasmic membranes of most cases (reported sensitivity: 83%-100%; Fig. 13.10 A). Only rarely (less than 1%) are isolated nonneoplastic spermatocytes (which are unlikely to be confused with GCNIS) PLAP positive, with spermatogonia being PLAP negative. Immunohistochemical staining for NANOG, a regulatory factor upstream to OCT3/4, similarly marks GCNIS. GCNIS has also been found to react with monoclonal antibodies M 2 A (D2-40/gp36/podoplanin), 43-9F, TRA-1-60, HB5, HF2, HE11, and with antibodies directed against glutathione-S-transferase, isoenzyme π, CD117 ( Fig. 13.10 C), angiotensin-converting enzyme, p53, growth differentiation factor 3 (GDF3), stem cell factor, and Krüppel-like factor 4. It is negative for the RNA-binding motif protein, in contrast with nonneoplastic germ cells. GCNIS is SOX17 positive and SOX2 negative, but SOX17 also marks nonneoplastic germ cells, a problem we have also encountered with CD117. In contrast, intratubular embryonal carcinoma is SOX17 negative and SOX2 positive. The positive reactions for PLAP, OCT3/4, CD117, and TRA-1-60 support the idea that GCNIS resembles fetal gonocytes. There is some heterogeneity in expression of germ cell markers in cells in GCNIS tubules, and it is the OCT3/4 + /MAGEA4 – population of cells that has the higher proliferative activity (relative to OCT3/4 + /MAGEA4 + cells) and is thought to progress to invasive tumor. By electron microscopy, GCNIS has evenly dispersed chromatin, intricate nucleoli, and sparse cytoplasmic organelles with prominent glycogen deposits. Occasional rudimentary intercellular junctions may be identified. These features are essentially the same as those of seminoma. The DNA content of GCNIS is similar to that of seminoma, usually in the triploid and hypotetraploid range. Some of the genetic features of GCNIS have been previously mentioned.
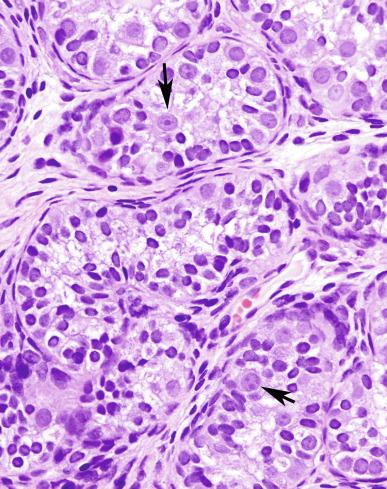
GCNIS should be distinguished from specific forms of intratubular germ cell neoplasia. Intratubular seminoma fills and distends the tubules, whereas GCNIS is restricted to the basilar area, although the cells are histologically identical. Atypical germ cells that do not resemble GCNIS cells may also occur in seminiferous tubules. These cells may have large nuclei or be multinucleated. They lack the cytoplasmic clarity and nucleolar prominence of GCNIS cells and do not stain for the usual GCNIS markers. Although they may indicate a perturbation in testicular development, as evidence by increased frequency in cryptorchid boys and adjacent to testicular germ cell tumors, their significance is not clear, unlike GCNIS.
Despite some controversy, GCNIS does not occur in prepubertal patients except for those with disorders of sex development. Many purported cases represent germ cells with delayed maturation ( Fig. 13.11 ). These cells may occur in young children who have conditions that place them at increased risk for germ cell tumors, including cryptorchidism, gonadal dysgenesis, Down syndrome, and undervirilization syndromes. They, however, are distinguished from GCNIS by their nonbasilar location in the tubules ( Fig. 13.11 ) and tendency to cluster in areas of the parenchyma. More recently, “pre-GCNIS” has been put forth as a lesion intermediate between maturation delay and GCNIS. It differs from them by having the lesional cells in both basilar and nonbasilar loci ( Fig. 13.12 ) and, unlike maturation delay, is associated with expression of stem cell factor in the affected tubules, a feature shared by GCNIS. The full clinical significance of maturation delay is not clear, meaning it is not known how frequently it progresses to an invasive germ cell tumor. It is important to distinguish such cells from GCNIS because it is known that GCNIS will progress in virtually all cases. If GCNIS or pre-GCNIS is found in a prepubertal patient, it is in the context of a disorder of sex development. Pre-GCNIS in patients with a disorder of sex development has also been termed infantile GCNIS .
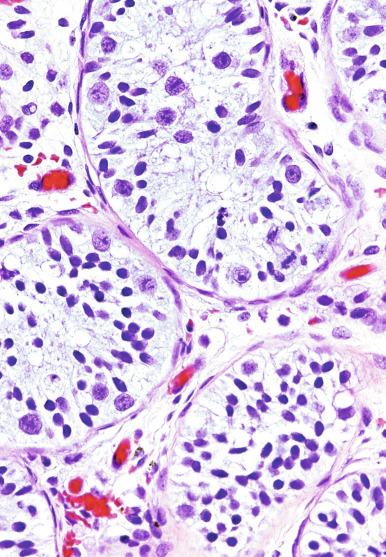
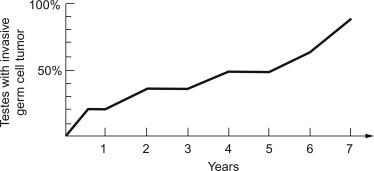
The practical importance of GCNIS is its progression to an invasive germ cell tumor (either seminomatous or nonseminomatous) in about 50% of cases within 5 years after identification. Only a small fraction of patients remain free of an invasive tumor by 7 or 8 years of follow-up ( Fig. 13.13 ), although some patients may not develop a tumor for more than 15 years. Furthermore, there is no documented case of spontaneous regression of typical GCNIS. GCNIS is identified with increased frequency in patients with cryptorchidism, a previous history of a testicular germ cell tumor, gonadal dysgenesis, androgen insensitivity syndrome, and infertility. GCNIS is also identified in the residual seminiferous tubules of virtually every postpubertal patient with a testicular germ cell tumor, with the exceptions of spermatocytic tumor, prepubertal-type teratoma/dermoid cyst/epidermoid cyst, and prepubertal-type yolk sac tumor. The prepubertal-type germ cell tumors, as mentioned previously, lack association with GCNIS. In our opinion, many reported cases in children ( Figs. 13.14 and 13.15 ) represent reactive enlargement of nonneoplastic germ cells induced by a mass lesion, as described by Hawkins and Hicks, atypical but nonneoplastic germ cells of unknown significance, or examples of delayed maturation of germ cells.
Testicular biopsies are a sensitive method for detecting GCNIS. Fixatives such as Bouin, B-5, and Stieve enhance cytologic detail and may permit easier detection of GCNIS compared with formalin fixation. Formalin, Bouin, and Stieve fixatives permit immunohistochemical detection of placental alkaline phosphatase, whereas Cleland fluid yields inconsistent results. Nonformalin fixation may result in weaker immunohistochemical staining, so formalin may be preferred if immunohistochemical staining is expected. Berthelsen and Skakkebaek concluded that one or two 3-mm biopsies of a testis harboring GCNIS will detect virtually every case, although rare false-negative results do occur (0.3% in one study of 1859 biopsies that were initially interpreted as negative). Two site biopsies have a 17% improvement in sensitivity over one site and have been recommended as standard. Another study supported three biopsies, each 5 mm in length, as optimal. In cases of severe atrophy, with obliteration of many tubules, it may be necessary to sample the region near the hilum, where GCNIS is more frequently preserved within the epithelium of the rete testis. Recently, some authors have considered OCT3/4 immunohistochemistry mandatory for the evaluation of testis biopsies, as it identifies GCNIS in approximately 20% more cases than hematoxylin and eosin staining alone. It should be noted that positivity for immunohistochemical markers, including OCT3/4 and D2-40, may be seen before 2 years of age in the absence of GCNIS, so, in younger patients, positive immunohistochemical staining alone does not indicate a positive diagnosis. Potential indications for screening biopsies include patients with cryptorchidism (2% to 4% positivity for GCNIS), a prior testicular germ cell tumor (4% to 5% positivity), somatosexual ambiguity (25% positivity), and, less strongly, oligospermic infertility (0% to 1% positivity). A recent study of 4130 germ cell tumor patients found a rate of metachronous germ cell tumor of 1.9% in the cohort who had prior screening biopsies (and treatment if positive) and 3.1% in the unscreened cohort, but this did not reach statistical significance. Sperm concentration, contralateral testis volume, and ultrasound echo pattern have been proposed to estimate the risk for contralateral GCNIS in germ cell tumor patients and to select patients for biopsy.
GCNIS in a biopsy from a patient with retroperitoneal germ cell tumor probably indicates the regression of a prior invasive tumor that metastasized to the retroperitoneum. Therefore, a patient with presumed primary germ cell tumor of the retroperitoneum may benefit from testicular biopsy.
Because of the high rate of progression of GCNIS to invasive testicular germ cell tumor, these patients should receive appropriate ablative treatment, either orchiectomy or low-dose external beam radiation. Unilateral GCNIS is usually managed by orchiectomy and bilateral GCNIS with radiation. Because GCNIS is very radiosensitive, low doses are used, permitting preservation of androgen function. Chemotherapy, often given to patients with metastatic disease from a contralateral testicular tumor, may ablate GCNIS in the remaining testis but is not a consistently effective therapy. In a German cooperative group study, radiation treatment was highly effective in curing GCNIS, although failures occur in about 2% of patients. Chemotherapy reduced the incidence of subsequent germ cell neoplasia, but was much less effective than radiotherapy.
Seminoma is the most common pure testicular germ cell tumor, and pure seminoma accounts for about 50% of all cases, although its relative proportion may be declining. In one study, the percentage of seminomas decreased from 52% to 43% over the 20-year study period. It occurs in patients at an average age of 40 years, which is about 10 years older than those with nonseminomatous germ cell tumor. African Americans may have an earlier age of onset. Most patients present with a painless testicular mass, but there may be a dull, aching sensation. Up to 11% have normal-sized or atrophic testes. Occasional patients (2% to 3%) present with symptoms of metastases, usually back pain caused by retroperitoneal involvement, but gastrointestinal bleeding, bone pain, central nervous system dysfunction, dyspnea and cough, and other symptoms may rarely be presenting complaints. Gynecomastia may occur as a result of elevation of serum hCG caused by intermingled syncytiotrophoblast elements in seminoma and is rarely a presenting feature. Very rarely, a paraendocrine form of exophthalmos can be a presenting complaint, as can paraneoplastic hypercalcemia, hemolytic anemia, and limbic encephalopathy. About 30% of seminoma patients have metastases at the time of diagnosis, and this is probably decreasing as a result of an overall trend for earlier detection of germ cell tumors.
Patients with seminoma usually lack serum AFP and hCG elevations that occur commonly in patients with nonseminomatous germ cell tumors. AFP levels should be normal, although concomitant liver disease (including seminomatous metastases to the liver) may cause modest AFP elevation, and “borderline” elevated AFP levels have been seen in some cases without evidence of nonseminomatous elements. Most oncologists regard significant AFP elevation in a patient with apparently pure testicular seminoma as evidence of nonseminomatous elements and treat accordingly. About 10% to 20% of patients with clinical stage I “pure” testicular seminoma have elevated serum hCG, and 25% or more with advanced seminoma have hCG elevations. At initial diagnosis, 7% to 25% of patients with seminoma have elevated hCG. If blood is sampled from the testicular vein, 80% to 85% of patients have elevated hCG. Such elevation reflects the presence of intermingled syncytiotrophoblast elements in these tumors, and the elevations are generally modest. Although rare, large seminomas with abundant syncytiotrophoblast cells may produce elevations of over 4000 IU/L. Elevations of serum hCG exceeding 40 IU/L have been correlated with a poorer prognosis, although this is controversial. Peripheral venous elevations of hCG have been correlated with larger tumors, perhaps explaining the relationship of elevated hCG with adverse prognostic features. Elevation of serum levels of lactate dehydrogenase, PLAP, and neuron-specific enolase may also occur in patients with seminoma. Such elevations, however, are neither specific nor especially sensitive, which limits their clinical utility. MicroRNA-based evaluation for miR-371-3 and miR-367 as a potentially more sensitive marker of tumor recurrence is currently being investigated.
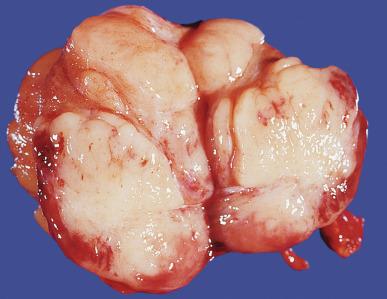
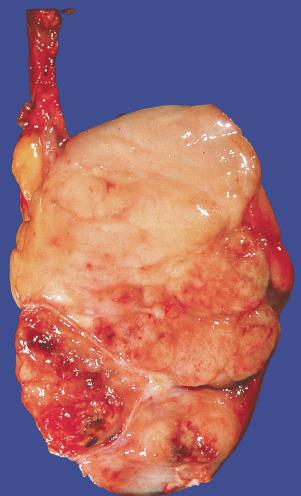
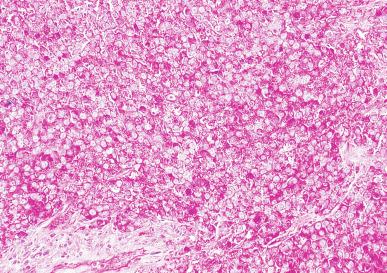
Grossly, seminoma is usually cream to tan to pale pink and often multinodular ( Fig. 13.16 ), with occasional yellow foci of necrosis. Infrequently, necrosis is extensive. In some cases, the tumor is diffuse, fleshy, and encephaloid, similar to testicular lymphoma ( Fig. 13.17 ). In contrast with lymphoma, however, only about 10% of seminomas extend into paratesticular structures. Intraparenchymal hemorrhage may cause red discoloration. The cut surface usually bulges from the surrounding parenchyma ( Fig. 13.16 ). Punctate foci of hemorrhage often correspond to intermingled foci of syncytiotrophoblast elements. A fibrous consistency is uncommon but results when prominent fibrous septa develop in the tumor or if there is extensive intratumoral sclerosis.
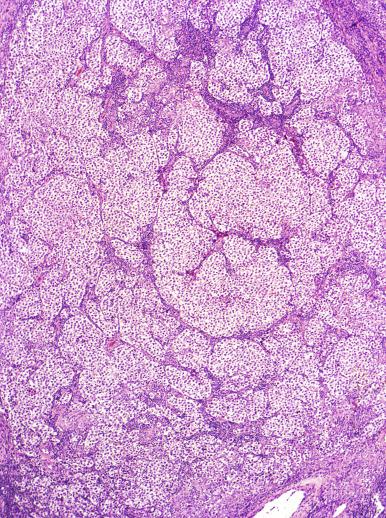
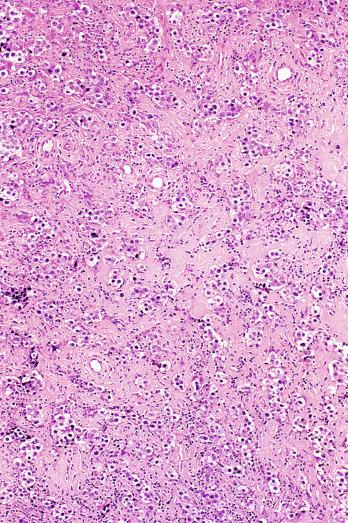
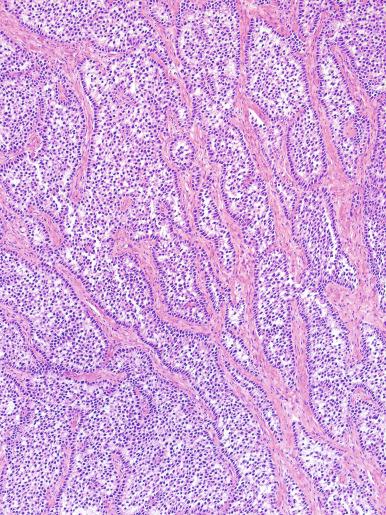
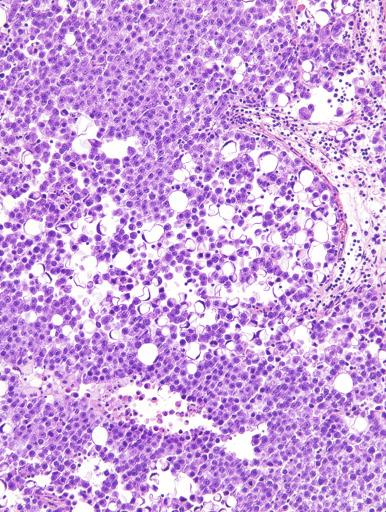
Microscopically, seminoma is usually arranged in a diffuse, sheetlike pattern interrupted by branching, fibrous septa containing an inflammatory infiltrate ( Fig. 13.18 ) consisting chiefly of lymphocytes but often containing plasma cells and sometimes eosinophils. Distinct nodules may be apparent, sometimes with confluent growth imparting a lobulated pattern. In some cases, a prominent cordlike arrangement of cells is present ( Fig. 13.19 ), often at the periphery of nodules showing a sheetlike pattern. Foci of intertubular growth may be seen, with preservation of seminiferous tubules. This pattern is usually most apparent at the periphery of neoplastic nodules and may be associated with rete testis invasion and aggressive behavior. In rare seminomas, an intertubular pattern may predominate ( Fig. 13.20 ), with well-preserved seminiferous tubules even in the central portions of the neoplasm. Such cases are prone to being overlooked because they do not destroy the seminiferous tubules, and they often do not form a discrete gross mass. The presence of a lymphocytic infiltrate may be a clue to intertubular seminoma ( Fig. 13.20 ). This pattern is much more common, however, in testicular lymphoma. With time, many seminomas develop foci of scarring. Hyalinized deposits of collagen may separate the neoplastic cells into small nests resembling solid pseudotubules ( Fig. 13.21 ). Extensive collagen deposits may result in broad scars with only a few scattered neoplastic cells. Calcification and even ossification may rarely occur in hyalinized fibrous trabeculae of seminoma. Rarely, seminoma may show a distinctly tubular pattern in which a palisade-like arrangement of neoplastic cells occurs at the periphery of tubule-like structures that may contain loosely cohesive neoplastic cells in their “lumens” ( Fig. 13.22 ). Seminoma may also develop intercellular edema with separation of neoplastic cells and formation of microcystic spaces ( Figs. 13.23 and 13.24 ). These spaces are generally, but not always, irregular in outline and frequently contain visible edema fluid and intracystic exfoliated neoplastic cells that contrast with the “cleaner,” more round and regular microcystic spaces commonly identified in yolk sac tumor. Foci of coagulative necrosis are present in about one-half of seminomas and may be extensive in a minority of cases.
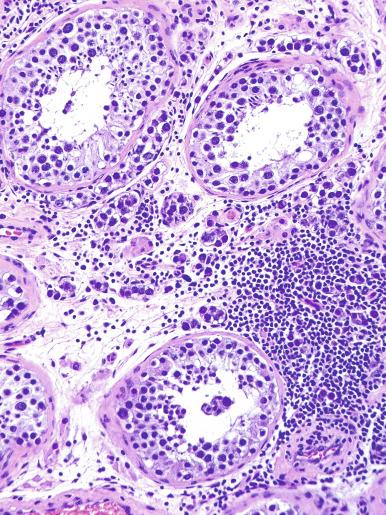
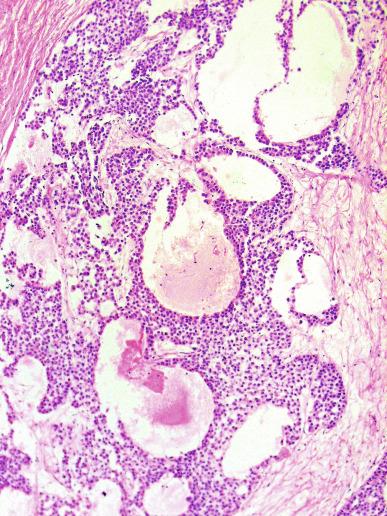
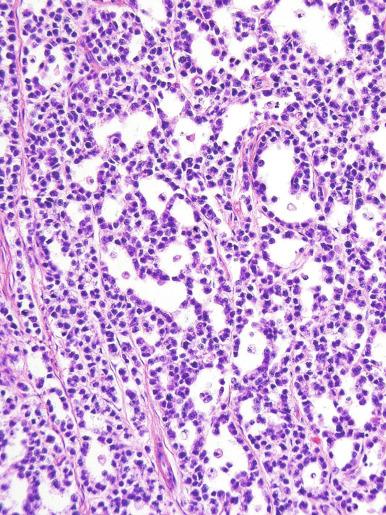
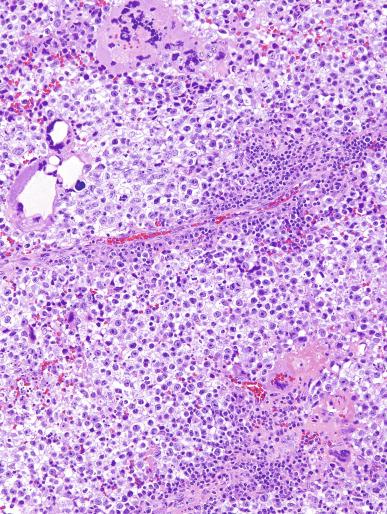
A lymphoid infiltrate is a virtually constant feature of seminoma and is usually most evident in perivascular areas and around fibrous trabeculae, which also contain many capillaries ( Fig. 13.25 ). Lymphocytes are also frequently intermingled with the seminoma cells elsewhere. A florid lymphoid reaction, with formation of germinal centers, occurs in a minority of cases, but most of the lymphocytes in seminomas are T cells. Ultrastructural studies have demonstrated a cytolytic effect of the lymphocytes on the seminoma cells, correlating with the observation of some investigators of a better prognosis in cases associated with a prominent lymphocytic reaction.
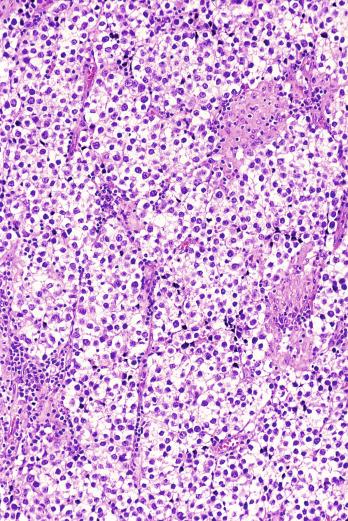
A variable granulomatous reaction occurs in up to 50% of seminomas. In most cases, it consists of small clusters of epithelioid histiocytes scattered among neoplastic cells ( Fig. 13.25 ). Langhans-type giant cells and other multinucleated giant cells may be present. Intratubular collections of epithelioid histiocytes may also be seen. Rarely, the granulomatous reaction can be extensive and virtually efface the neoplasm ( Fig. 13.26 ); in such cases, it may be difficult to distinguish a florid granulomatous reaction in a seminoma from granulomatous orchitis, and careful search for residual seminoma and GCNIS is indicated. Intravascular granulomas have been reported in 15% of seminomas but were not a significant predictor of tumor recurrence.
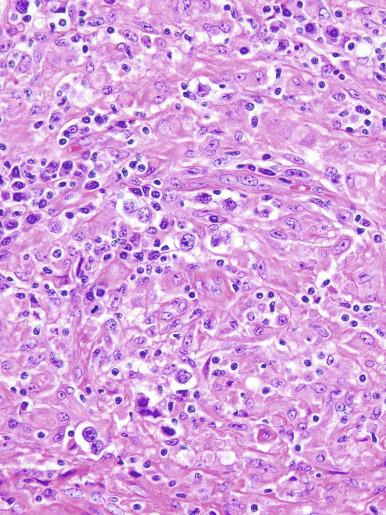
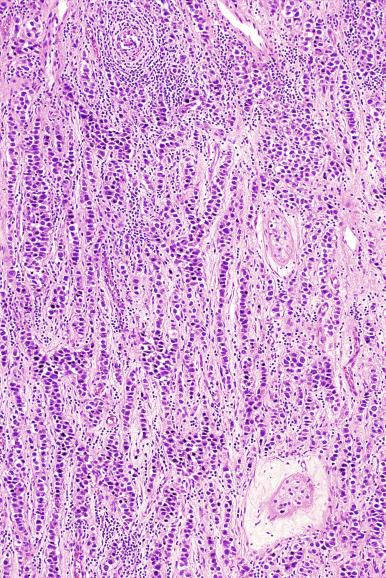
The cells of seminoma have generally clear to lightly eosinophilic cytoplasm, measuring 15 to 25 μm in diameter. The nuclei are relatively uniform, polygonal, usually centrally or slightly eccentrically placed, with finely granular chromatin and one or two prominent nucleoli ( Fig. 13.27 ). Many nuclei have a relatively flat edge that has been described as “squared-off,” lending the nuclei a “boxy” appearance. The nuclear membranes are irregularly thickened. The cell borders are typically well defined in adequately fixed specimens ( Fig. 13.27 ). Abundant cytoplasm separates the nuclei so that overlapping nuclei are usually not seen. Occasionally, seminoma displays foci of increased cellular atypia with less well-defined cytoplasmic membranes, darker cytoplasm, and enlarged, crowded nuclei ( Fig. 13.28 ). These changes may impart a plasmacytoid appearance to the tumor cells ( Fig. 13.29 ). Such changes may be seen in association with early necrosis and contain pyknotic nuclear fragments. Such isolated foci, in the absence of distinct epithelial differentiation, do not exclude a diagnosis of seminoma. Rarely seminoma cells develop intracytoplasmic vacuoles that cause a signet ring cell morphology ( Fig. 13.30 ). This may be a focal finding in an otherwise usual-appearing tumor. We have also seen prominent aggregates of glycogen create a signet ring cell appearance ( Fig. 13.31 ).
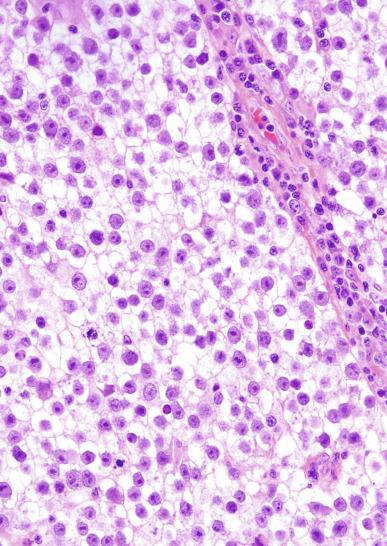
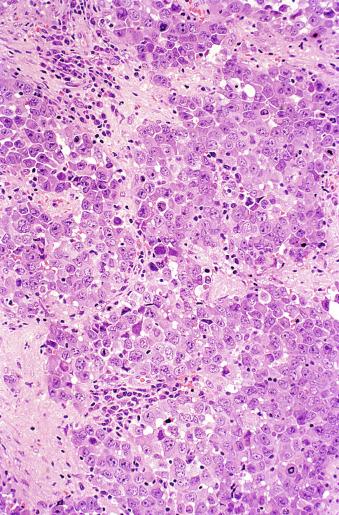
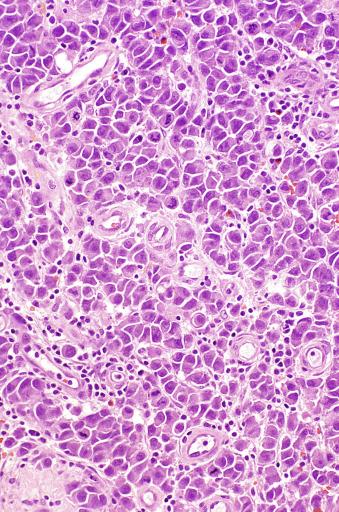
Mitotic figures in seminoma are prominent, and in the past, when present in sufficient numbers, were considered as evidence of “anaplastic seminoma.” Now it is clear that mitotic rate in seminoma does not correlate with prognosis. Furthermore, there is no immunohistochemical difference between typical and “anaplastic” seminomas. Use of the term anaplastic seminoma is therefore discouraged. Although some seminomas behave more aggressively than most, it remains unclear if such cases can be identified prospectively. Tickoo et al. described cases of “seminoma with atypia” based on nuclear pleomorphism and crowding, paucity of lymphocytes, and darker-staining cytoplasm. In their experience, such tumors were more likely to present at advanced clinical stage, to express CD30, and to lose KIT expression. It remains unclear, however, if such cases should receive different treatment.
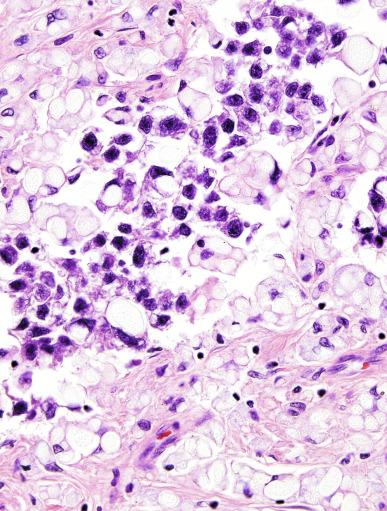
Syncytiotrophoblast cells are present in 10% to 20% of seminomas. The morphology of these cells is variable, ranging from typical syncytiotrophoblast cells with cytoplasmic lacunae and multinucleation ( Fig. 13.32 ) to large mononucleated or binucleated cells that may not be easily distinguished from the background seminoma cells but that are highlighted by immunostains against hCG. Intermediate between these extremes are cells containing multiple nuclei in a “mulberry” pattern. Syncytiotrophoblast cells are often located close to capillaries, and microhemorrhages may be seen in these foci. Unlike choriocarcinoma, the syncytiotrophoblast cells are not intermingled with a mononucleated trophoblast cell component but are randomly admixed as single cells or small clusters with seminoma cells. With hCG immunohistochemistry, intratubular trophoblasts can be detected in a substantial fraction of seminoma cases, indicating that differentiation toward trophoblastic elements may occur before invasion.
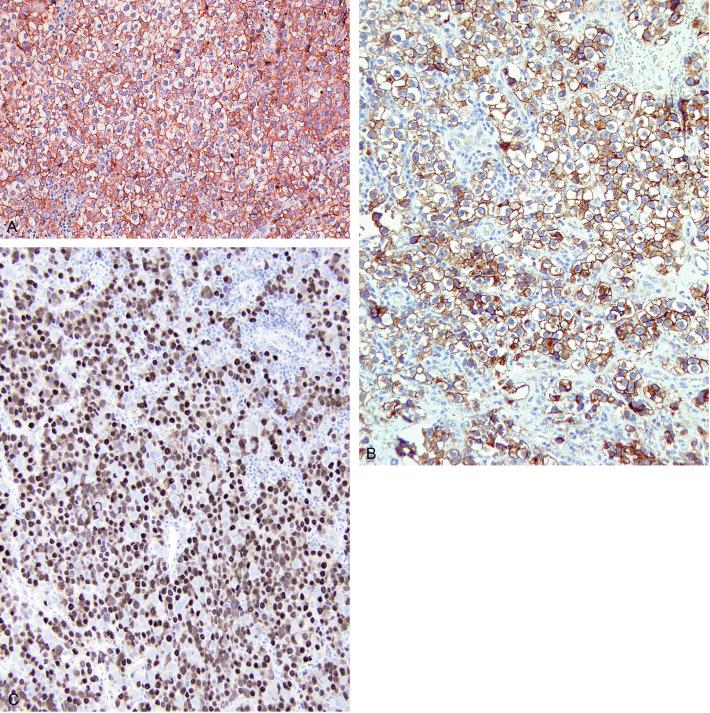
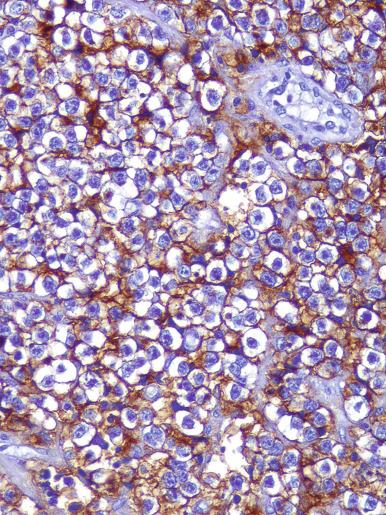
Glycogen is usually prominent in seminoma ( Fig. 13.33 ), and most cases show immunoreactivity for PLAP, generally in a peripheral, “membranous” staining pattern ( Fig. 13.34 A ); cytoplasmic staining may also be identified. Seminomas are uniformly reactive for OCT3/4 ( Fig. 13.34 C), a nuclear transcription factor identified in nonneoplastic stem cells and embryonic cells that plays an important role in the maintenance of a pluripotential capacity. OCT3/4 staining is also seen in embryonal carcinoma but not in other testicular tumors. Angiotensin-converting enzyme was demonstrated in 100% of 91 seminomas in one study. Reactivity for CD117 (c-kit protein) occurs in most seminomas, with a cytoplasmic membrane pattern similar to PLAP ( Fig. 13.34 B). Podoplanin (D2-40) is positive ( Fig. 13.35 ), as is SALL4, a general germ cell tumor marker. The NANOG protein, a product of a gene located at 12p13, a region frequently amplified in testicular germ cell tumors, is similarly detectable by immunohistochemistry in GCNIS, seminoma, and embryonal carcinoma, but not in teratoma or yolk sac tumor. SOX17, a nuclear transcription factor, was identified in 95% of seminomas but not in embryonal carcinomas. Many seminomas also express limited degrees of cytokeratin immunoreactivity, although such staining may only be demonstrable using frozen sections, with frequent negativity in routinely processed tissue. The most common cytokeratins in seminoma are cytokeratins 7, 8, and 18, although others, including cytokeratins 4, 17, and 19, can be identified occasionally. Cytokeratin 20 is negative. Epithelial membrane antigen (EMA) is only rarely expressed in seminoma, and the combination of positivity for PLAP and negativity for EMA and cytokeratin AE1/AE3 in formalin-fixed, paraffin-embedded tissue is a relatively specific pattern for seminoma. Vimentin, LDH, and NSE may be present in seminoma but are not specific. A minority of seminomas may stain for Leu-7, α-1-antitrypsin, desmin, and neurofilament protein. Desmoplakins and desmoglein are usually present in seminoma. The syncytiotrophoblast cells that occur in some seminomas contain hCG, and some cells containing hCG may not have an overtly “syncytiotrophoblastic” appearance in routine sections. CD30 is usually negative in seminoma, which contrasts with its virtually uniform presence in embryonal carcinoma, except after chemotherapy, in which case loss of CD30 expression is frequently seen.
Ultrastructurally, seminoma has closely apposed cytoplasmic membranes that usually show only sparse, primitive, intercellular junctions. The cellular organelles consist of scattered mitochondria, occasional cisternae of smooth and rough endoplasmic reticulum, ribosomes and polyribosomes, occasional membrane-bound lysosome-like structures, and occasional Golgi bodies. Glycogen may be present in large quantities but may also be sparse. It is common for the cytoplasmic organelles to be polarized eccentrically in the cytoplasm. The nuclei are round and have evenly dispersed chromatin and intricate, large nucleoli. Occasional seminomas that are typical-appearing at the light microscopic level may show evidence of epithelial differentiation, with small, extracellular lumens, microvilli, and well-defined junctional complexes. Such cases, despite “transitional” morphology at the ultrastructural level, appear to behave as typical seminoma.
The DNA content of seminoma is generally in the triploid to hypotetraploid range and is greater than that of nonseminomatous tumors. The DNA content of seminoma with syncytiotrophoblast cells does not differ from seminoma lacking them, supporting classification as seminoma. Evolution of seminoma to other types of germ cell tumor may occur as a result of gene loss. As in many testicular germ cell tumors, seminoma frequently contains an isochromosome derived from the short arms of chromosome 12, i(12p). Numerous other cytogenetic abnormalities have also been described; certain chromosomes are commonly overrepresented (1q, 7, 8, 12, 14q, 15q, 17, 21q, 22q, and X) and others underrepresented (3, 4, 5, 10, 11, 12q, 13q, 16, 18q, and Y).
A number of molecular observations are reported in seminoma, including absence of Fas, loss of p18 INK4C , upregulation of cyclin E, cyclin D2 expression, and activation of p16 INK4 by hypermethylation. Activating exon 17 mutations of the KIT gene are seen in 12% to 13% of cases and about 20% have KIT gene amplification. Seminomas are reported to have loss of Notch 1, Jagged 2, and Fhit expression, show SMAD4 and ras mutations, and express DAZL1 protein, hst-1, N-myc, Krüppel-like factor 4, and MAGE genes. The MAGE gene products, along with NY-ESO-1, are members of a group known as “cancer testis antigens” which show variable, low-level expression in seminoma. These antigens are consistently strongly expressed in spermatocytic tumors and are infrequently expressed by nonseminomatous germ cell tumors and GCNIS. Gene expression studies have confirmed significant upregulation of genes previously known to be upregulated in seminoma such as POU5F1 ( OCT4 ), KIT , ALPL , and PROM1 and have also identified upregulation of a number of other genes including BOB1 , CD9 , THY1 , and PUM2 .
Seminoma can be misinterpreted as the solid pattern of embryonal carcinoma, especially in poorly fixed preparations. The formation of glands, true tubules, or papillae argues against the diagnosis of seminoma; the nuclei of seminoma are more uniform, less crowded, and more evenly spaced than those of embryonal carcinoma, which are pleomorphic, irregularly shaped, crowded, and appear to abut or even overlap. The cytoplasmic borders of seminoma are well defined and those of embryonal carcinoma are mostly poorly defined, with occasional exceptions in foci. The cytoplasm of embryonal carcinoma is typically denser. Embryonal carcinoma lacks the regular fibrous septa of many seminomas. Cytokeratin reactivity is usually weaker and PLAP reactivity stronger in seminoma than in embryonal carcinoma. CD30 reactivity is rare in seminoma and positive in embryonal carcinoma, and CD30 immunohistochemistry may be especially useful in combination with CD117 (c-kit) to distinguish seminoma (CD30–/CD117 +) from embryonal carcinoma (CD30 +/CD117–). Weak staining of embryonal carcinoma for c-kit has been seen in up to 32% of tumors in one study, however. Podoplanin (M2A, D2-40), similar to CD117, is typically diffusely positive in seminoma and usually negative or focally reactive in embryonal carcinoma. Monoclonal antibody 43-9F is reported to be strongly reactive in embryonal carcinoma and negative or only weakly positive in seminoma, but additional experience is necessary to confirm this finding. An antibody to SOX2, a nuclear transcription factor, appears to be a highly sensitive nuclear marker for embryonal carcinoma and is consistently negative in seminoma. A related protein, SOX17, in contrast, is positive in seminoma and negative in embryonal carcinoma. Immunohistochemical staining for AP-2γ, another nuclear transcription factor, marks a higher percentage of nuclei in embryonal carcinoma than seminoma, but is probably not of use in differentiating these two tumors, although it may, like OCT3/4, be a useful marker for the two as a group. AFP is occasionally positive in embryonal carcinoma and is negative in seminoma.
The distinction of seminoma from spermatocytic tumor is discussed in the Spermatocytic Tumor section later in this chapter.
Yolk sac tumor with a solid pattern may mimic seminoma. Such cases are usually distinguished from seminoma by the presence of typical patterns of yolk sac tumor, the tendency for microcyst formation in the solid areas, intercellular bandlike deposits of basement membrane, intracellular hyaline globules, nuclear pleomorphism, and absence of lymphocytes and fibrous septa. Edematous seminomas, however, may produce a microcystic pattern reminiscent of yolk sac tumor, but the cystic spaces are usually more irregular and contain edema fluid and exfoliated neoplastic cells, in contrast with those of yolk sac tumor, although there are exceptional seminomas with regular and uniform microcysts. The typical polygonal cells of seminoma in microcystic areas, however, contrast with the flattened cellular profiles and more variable nuclear appearance of yolk sac tumor cells lining microcystic spaces. AFP is negative in seminoma and usually positive in yolk sac tumor; glypican-3 has a similar pattern of reactivity but is more sensitive for yolk sac tumor. Cytokeratin is often negative or weak in seminoma (in routinely processed tissues), whereas it is almost always strongly positive in yolk sac tumor. OCT3/4 and podoplanin are strongly positive in seminoma but negative in yolk sac tumor. CD117 is not useful in this differential diagnosis because it is often positive in both.
Lymphoma must be distinguished from seminoma; most patients with testicular lymphoma are older than 50 years of age, whereas patients with seminoma are usually younger. Bilateral involvement is more likely in lymphoma than in seminoma. Lymphoma usually infiltrates the interstitium, preserving the seminiferous tubules, whereas most seminomas do not show as prominent a degree of intertubular growth, although there are exceptions. GCNIS is seen in seminoma but not in lymphoma. Lymphoma often is more pleomorphic than seminoma and may be composed of cells with cleaved and irregularly shaped nuclei that stand in contrast with the polygonal and relatively uniform nuclei of seminoma. The cytoplasm of lymphoma is usually amphophilic and less distinct than that of seminoma. Immunostains directed against PLAP and OCT3/4 are positive in seminoma and negative in lymphoma, whereas leukocyte common antigen (LCA) and other lymphoid markers show opposite results.
Rarely, seminoma with a tubular pattern may be confused with Sertoli cell tumor, a neoplasm that frequently has a tubular architecture. The cytoplasmic clarity in seminoma is caused by glycogen, whereas lipid is mainly responsible for this appearance in Sertoli cell tumor. Most Sertoli cell tumors have low-grade cytologic atypia and infrequent mitotic figures that contrast with the high-grade atypia and conspicuous mitotic activity of seminoma. GCNIS is present in almost all seminomas but is not associated with Sertoli cell tumor. Tubular patterns in seminoma are usually focal but may be widespread in Sertoli cell tumor. Immunostains for PLAP, OCT3/4, and SALL4 are positive in seminoma but negative in Sertoli cell tumor, whereas inhibin and nuclear β-catenin are positive in a substantial proportion (but not all) of Sertoli cell tumors but negative in seminomas. This differential diagnosis may be further complicated by those Sertoli cell tumors that show a mostly diffuse growth pattern, frequently associated with a lymphocytic infiltrate.
Although patients with early seminoma (clinical stage I or nonbulky stage II) have traditionally been treated with orchiectomy and radiation to the paraaortic and paracaval nodes, frequently with ipsilateral pelvic nodal radiation (so-called “dogleg” field) (although the pelvic field component may not be necessary for clinical stage I patients), current American and European treatment guidelines for clinical stage I seminoma consider surveillance an appropriate option, with adjuvant treatment also being discussed. The American National Comprehensive Cancer Network (NCCN) guidelines state that active surveillance is preferred, but they do not make recommendations between chemotherapy and radiation treatment. In contrast, the European Association of Urology Guidelines state that surveillance and carboplatin chemotherapy are recommended and that radiotherapy is not recommended. Stratification by risk factors has also been employed, with carboplatin recommended for patients with two risk factors (tumor size > 4 cm and rete testis involvement) and surveillance for those with zero or one of these risk factors, and use of a nomogram considering these two factors has been proposed, but others have found this model for risk stratification to be inadequately predictive and have found only tumor size to be associated with recurrence. Radiotherapy for seminoma is associated with a slight (hazard ratio 1.36) increased risk for second malignant neoplasms. Most recurrences arise outside of the radiated field, in the mediastinum, cervical lymph nodes, or lungs.
With the increasingly chosen option of surveillance for clinical stage I patients, 13% to 20% of patients will undergo relapse. Nevertheless, chemotherapy salvages virtually all relapses, and therefore 5-year disease-specific survival for surveillance management approaches 100%. About 92% of relapses are within the first 3 years, and 87% are detected by computed tomography (CT) (as opposed to tumor markers).
Chemotherapy is recommended for patients with bulky retroperitoneal involvement or in more advanced stages. (“Bulky” is defined variously in different studies as metastases greater than 5 cm, 6 cm, or 10 cm in diameter.) There is an 87% progression-free survival for advanced-stage seminoma patients who are initially treated with chemotherapy, whereas such patients who are initially irradiated and then treated with chemotherapy for persistent disease have a 3-year progression-free survival of 69%. A four-cycle etoposide and platinum regimen has been evaluated to attempt to limit bleomycin toxicity in patients with germ cell tumors and retroperitoneal disease, but these patients are four times more likely to have active cancer in the retroperitoneal specimen when compared with the standard three-cycle bleomycin, etoposide, and platinum treatment.
A prominent lymphocytic reaction has been associated with improved prognosis. Elevation of serum hCG levels may indicate a poorer prognosis, although there are contradictory results in the literature. In one study, primary tumor size (> 4 cm), testicular vascular invasion, rete testis invasion, tunica albuginea penetration, spermatic cord invasion, epididymis invasion, and vascular invasion of the cord were predictive of metastasis in univariate analysis, but only tumor size (> 6 cm) and rete testis invasion were independently predictive in multivariate analysis. Rete testis involvement was not independently predictive of relapse of stage in other studies. In a series of 1954 stage I seminoma patients in Denmark followed on a surveillance program, size, vascular invasion, and epididymis involvement were significant predictors of relapse. Lymphovascular invasion, however, is often overdiagnosed in seminoma, with the rate of apparent lymphovascular invasion dependent on the handling of the gross specimen. Immunohistochemical expression of p53, Ki67, CD30, and loss of CD117/c-kit expression in a group of 62 seminomas did not correlate with metastasis or other histopathologic risk factors for metastasis, unlike a different study. In a mixed group of testicular germ cell tumors, high-level microsatellite instability by molecular analysis was significantly predictive of poorer prognosis, and a similarly significant but lower-magnitude difference was observed with loss of mismatch repair protein (MLH1) expression by immunohistochemistry. In germ cell tumor patients undergoing postchemotherapy retroperitoneal lymph node dissection, the number of lymph nodes removed has prognostic significance with a calculated 2-year relapse-free probability of 90% when 10 lymph nodes are removed compared with 97% when 50 lymph nodes are removed. Patients with viable seminoma in postchemotherapy retroperitoneal lymph node dissection specimens have decreased 5-year survival, 54% in a recent series.
Prognostic factors in patients with clinical stage I seminoma have been reviewed. Rete testis invasion was associated with relapse in some studies, but lymphovascular invasion, young age, and preoperative hCG level were not associated with recurrence. It seems likely that the nonsignificance of lymphovascular invasion stems from the difficulty in judging true versus pseudoinvasion. It would be useful to identify a subset of seminoma patients with a poor prognosis at an early stage, because it is likely that initial treatment with chemotherapy would improve the outcome in such a group. It is likely that the poor prognosis relates to the tendency of such cases to transform to nonseminomatous tumors, given the high frequency of nonseminomatous tumors at autopsy in patients who died after resection of pure testicular seminoma.
Testis-sparing surgery, not typically considered appropriate for germ cell tumors, has been performed for small (20 mm or smaller), clinical stage I, synchronous or metachronous bilateral germ cell tumors to attempt to allow preservation of fertility. Although testosterone production is consistently maintained, almost one-third of patients develop local recurrence, and only one-sixth achieve conception. Frequent local recurrences are expected, as, in addition to the frequent presence of GCNIS in these patients, about one-fourth of germ cell tumors are multifocal, and multifocality can be present even in small tumors.
Although very common in mixed germ cell tumors (occurring in 87% of nonseminomatous germ cell tumors,) embryonal carcinoma is less frequent as a pure testicular tumor, comprising only 2.3% of cases in a referral practice and about 10% of testicular tumors in two general series. A decline in the reported proportion of pure embryonal carcinoma among testicular germ cell tumors is largely attributable to the recognition of foci of yolk sac tumor in such cases, leading to their categorization as mixed germ cell tumors. This finding reflects the capacity of embryonal carcinoma to differentiate into other forms of testicular neoplasia, as verified by experimental observations including tissue culture. Embryonal carcinoma expresses the stage-specific embryonic antigen (SSEA) indicative of a primitive, undifferentiated stage of development, SSEA-3, but not SSEA-1, which indicates a more mature phenotype.
The peak incidence occurs at about 30 years of age, and embryonal carcinoma is distinctly rare in prepubertal children and likely limited to those with a disorder of sexual development. It usually presents as a testicular mass, with gynecomastia or symptoms of metastases each occurring in about 10% of cases. Rare cases may present with sudden death caused by massive tumor thromboemboli to the lungs. However, metastases are clinically or radiographically evident in about 40% of patients at presentation, and two-thirds of patients who are pathologically staged have metastases. As in seminoma, limbic encephalopathy may be a rare form of presentation.
Serum AFP elevation in embryonal carcinoma is usually the result of misclassification of mixed germ cell tumor (embryonal carcinoma and yolk sac tumor), and it is uncommon for morphologically pure embryonal carcinoma to be associated with serum AFP elevation. Many embryonal carcinomas are associated with syncytiotrophoblast cells, accounting for serum hCG elevation in 60% of cases. PLAP, LDH, and CA19-9 levels may also be elevated.
Grossly, embryonal carcinoma is usually poorly circumscribed and gray-white to pale yellow or tan with prominent areas of hemorrhage and necrosis ( Fig. 13.36 ). Microscopically, there are three major patterns, solid, glandular, and papillary, all of which consist of cohesive groups of primitive, anaplastic epithelial cells. In the solid pattern, the cells are arranged in diffuse sheets ( Fig. 13.37 ). In the tubular or glandular pattern, well-defined, glandlike or tubule-like structures are formed by epithelium varying from cuboidal to columnar ( Fig. 13.38 ). Anastomosing ( Fig. 13.39 ) or sievelike ( Fig. 13.40 ) patterns may be seen. The luminal spaces are cleftlike or round. In the papillary pattern, the papillae may or may not have stromal cores ( Fig. 13.41 ). When vascular stromal cores are cut transversely, a radial arrangement of tumor cells at their periphery resembles the endodermal sinus-like structures of yolk sac tumor (“pseudoendodermal sinuses”; Figs. 13.42 and 13.43 ). Uncommon patterns include nested, micropapillary, pseudopapillary, and blastocyst-like ( Fig. 13.44 ). A “double-layered” pattern of embryonalcarcinoma has also been described in which a papillary arrangement of embryonal carcinoma is accompanied by a parallel layer of flattened neoplastic epithelium ( Fig. 13.45 ). This pattern, however, is more accurately classified as embryonal carcinoma with yolk sac tumor and should therefore be regarded as a mixed germ cell tumor.
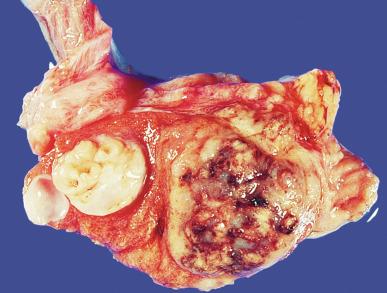
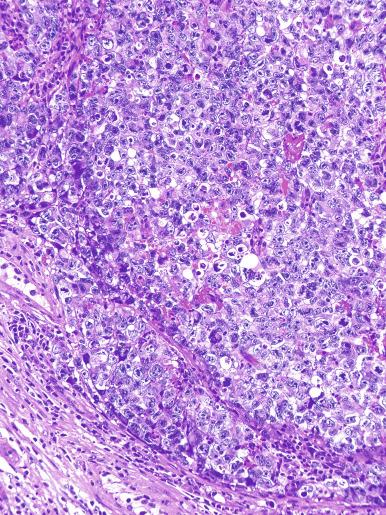
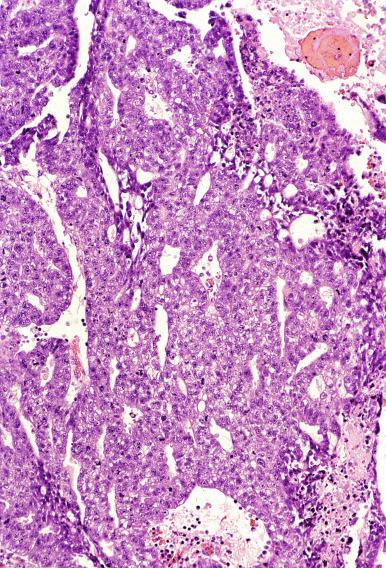
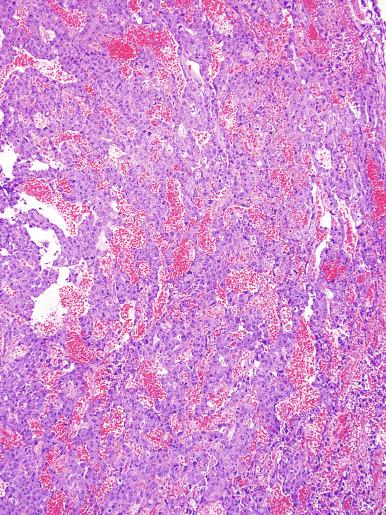
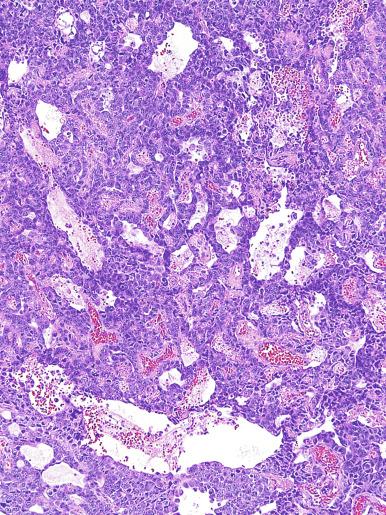
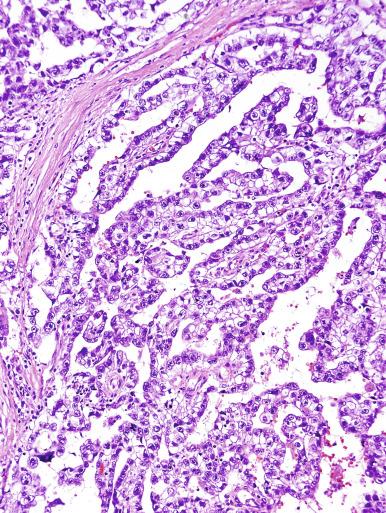
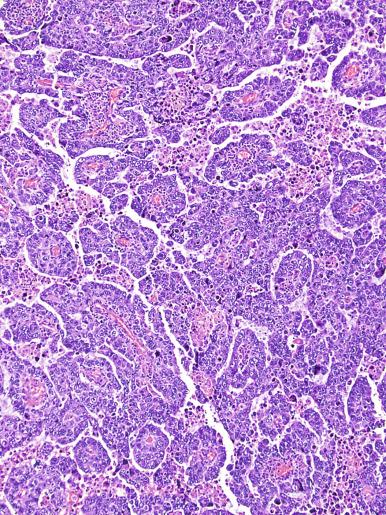
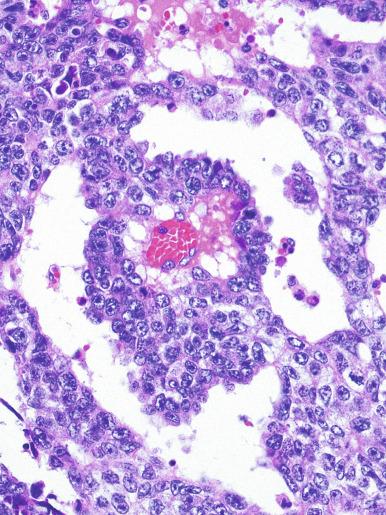
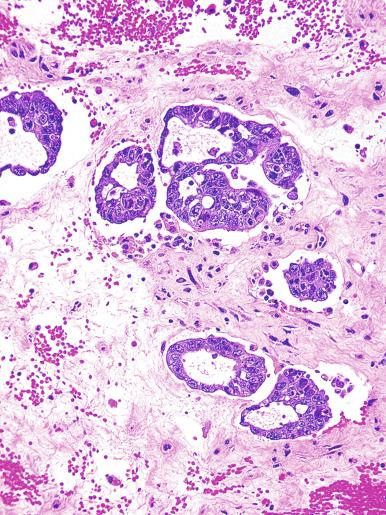
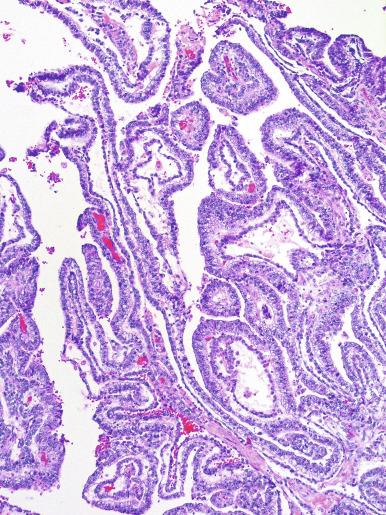
At high magnification, the cells have variable staining, abundant cytoplasm, and large, vesicular, irregular nuclei with prominent macronucleoli ( Fig. 13.46 ). The cell borders are characteristically ill defined, unlike seminoma, and the nuclei are often crowded, appearing to abut or overlap. Karyorrhectic fragments are frequent in the background, and the mitotic rate is high. Potentially deceptive histologic features occur in some cases, including foci of cells with clear cytoplasm and well-defined cytoplasmic borders, possibly causing concern for seminoma ( Fig. 13.47 ). Glands lined by tall, columnar cells may be mistaken for teratoma ( Fig. 13.48 ). In both circumstances, the pleomorphic nature of the nuclei supports embryonal carcinoma. Occasionally, the tumor cells may have clear cytoplasmic vacuoles giving the tumor a “lipoid” appearance ( Fig. 13.49 ).
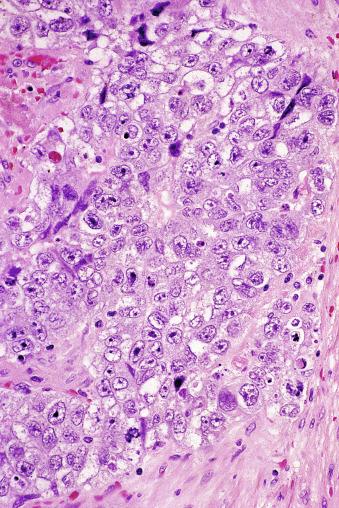
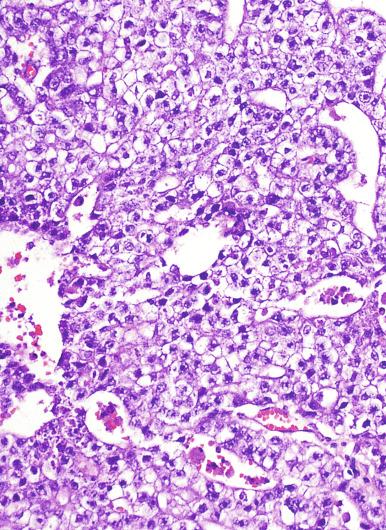
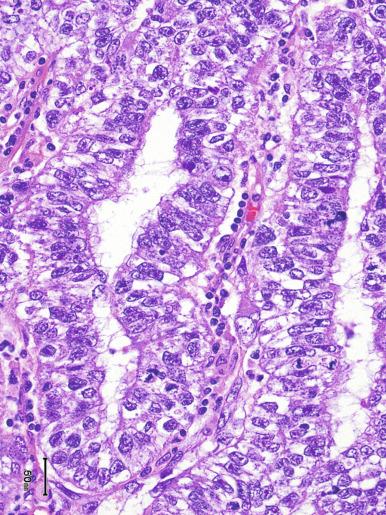
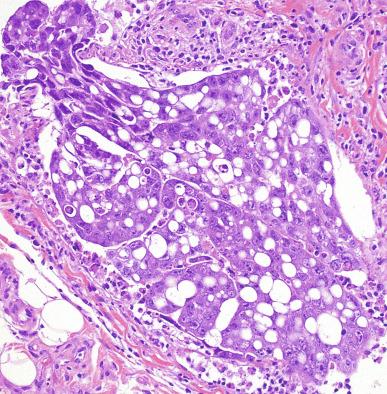
Prominent foci of eosinophilic, coagulative necrosis are common in all forms of embryonal carcinoma. Cells with smudged, hyperchromatic nuclei are distinctive but nonspecific ( Fig. 13.50 ). These commonly appear “applied” to the periphery of tumor nests (“appliqué” appearance). Such cells are considered to be degenerate embryonal carcinoma cells, but they may be misinterpreted as syncytiotrophoblast cells, thereby leading to a misdiagnosis of choriocarcinoma. Unlike syncytiotrophoblast cells, however, these degenerate embryonal carcinoma cells lack hCG and are not usually associated with hemorrhage.
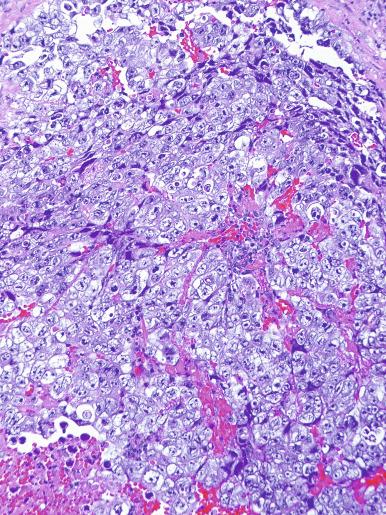
The capacity of embryonal carcinoma to generate a minor amount of undifferentiated neoplastic stroma ( Fig. 13.51 ) is widely recognized. The stroma may either be reactive, fibrous, and nonneoplastic or a neoplastic derivative of the tumor with atypia and mitotic activity. Although some regard neoplastic stroma within the spectrum of embryonal carcinoma, rationalizing that it is part and parcel of a primitive neoplasm recapitulating an early phase of embryonic development, we believe that neoplastic stroma should be considered a teratoma component. Based on the experience of the BTTP, no significant prognostic difference was noted in embryonal carcinoma with or without a stromal component, but these data were obtained in an era before effective chemotherapy. Because teratoma components in the testis are associated with an increased risk for persistent tumor in metastatic sites after chemotherapy, those BTTP data should have no bearing on current classification.
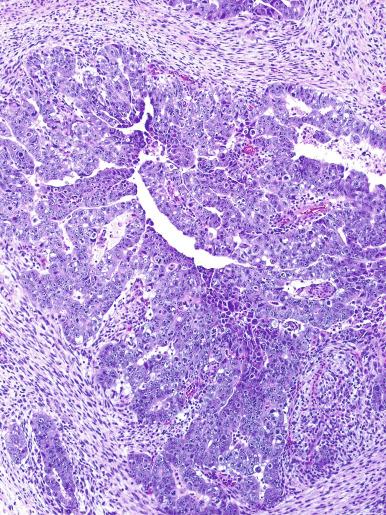
In about one-fourth of cases, embryonal carcinoma is associated with an intratubular embryonal carcinoma component, which is typically extensively necrotic, having a comedocarcinoma-like appearance ( Figs. 13.52 and 13.53 ). Such necrotic foci may undergo dystrophic calcification leading to the formation of so-called “hematoxylin-staining bodies.” Such coarse intratubular calcifications in scarred areas are excellent evidence of a regressed germ cell tumor.
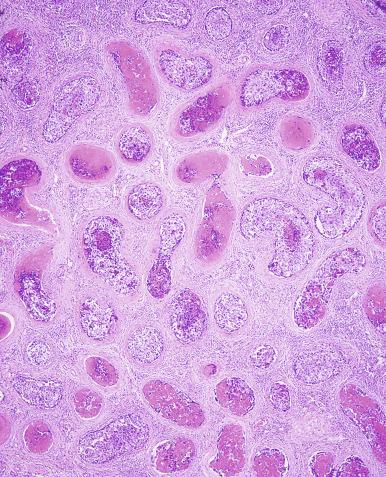
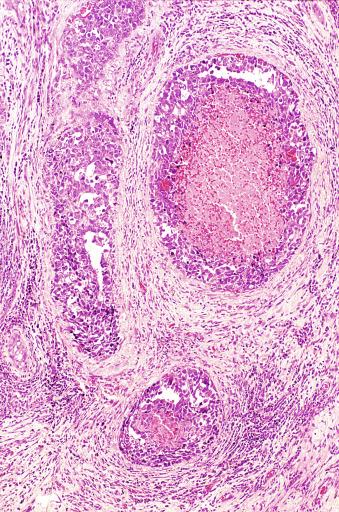
The identification of vascular invasion in nonseminomatous germ cell tumors (including embryonal carcinoma) may be helpful in deciding whether patients with clinical stage I tumors are appropriate candidates for “surveillance only” management and is a key feature in tumor staging. Embryonal carcinoma is the angioinvasive element in most cases when such invasion is present. There are some pitfalls in deciding if vascular invasion is present. First, intratubular neoplasm may closely resemble intravascular neoplasm. The presence of residual Sertoli cells in a possible “vessel” is good evidence that it is a tubule ( Fig. 13.54 ). Additionally, intratubular tumors are often extensively necrotic, and the tubules are nonbranching and of relatively similar caliber, whereas intravascular tumors do not typically show extensive necrosis and may occur in branched vessels of different sizes ( Fig. 13.55 ). Second, stringent criteria must be applied such that invaded tissue spaces are clearly lined by endothelial cells before regarding them as vessels. Third, germ cell tumors, and especially embryonal carcinoma and seminoma, are quite cellular and friable, leading to artifactual, knife-implantation of tumor cells into vascular spaces. Such implants, however, are loosely cohesive and unassociated with vascular thrombosis ( Fig. 13.56 ), whereas legitimate vascular invasion is characterized by neoplasm conforming to the shape of the vessel, which may also show evidence of thrombosis. Commonly, artifactual vascular implants are also associated with implants on the surfaces of tissues. It is usually easiest to appreciate vascular invasion a short distance away from the periphery of the neoplasm.
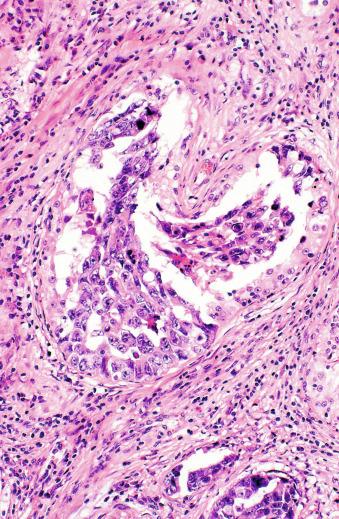
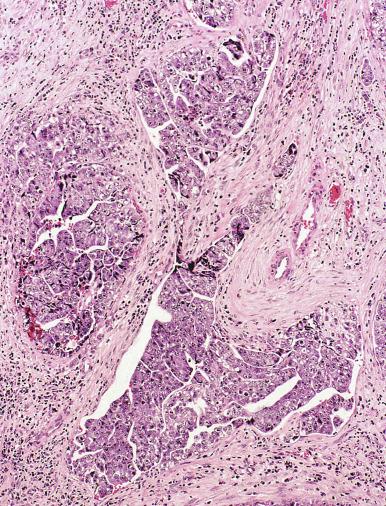
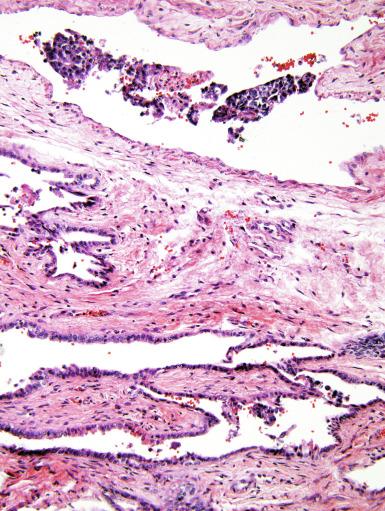
Only a small percentage of cases of pure embryonal carcinoma demonstrate AFP immunoreactivity, but such positivity is more common in the embryonal carcinoma component of a mixed germ cell tumor, a fact that likely represents early biochemical transformation to a yolk sac tumor component in cases of mixed germ cell tumor before morphologic differentiation. PLAP positivity occurs in 86% to 97% of cases, but is usually patchy and weaker than in seminoma. Several different cytokeratin classes are present in embryonal carcinoma, most prominently cytokeratins 8 and 18, but also cytokeratin 19, and occasionally cytokeratins 4 and 17. Most authors therefore report strong and diffuse positivity for cytokeratins in the majority of embryonal carcinomas, including routinely processed cases. Immunostains for CD30 (Ber-H2, Ki-1) are positive in 84% of cases ( Fig. 13.57 ), whereas EMA is negative in almost all embryonal carcinomas. CD117 and podoplanin (D2-40), in contrast with seminoma, are negative or only focally positive in embryonal carcinoma. OCT3/4, a nuclear transcription factor, is highly sensitive and mostly specific for embryonal carcinoma ( Fig. 13.58 ), seminoma, and GCNIS, with no staining observed in other germ cell tumor types or sex cord–stromal tumors, and only very rare positivity seen in lung and renal cell carcinomas and in large B-cell lymphomas. More recently, SOX2 has emerged as a valuable marker for embryonal carcinoma, with negativity in the other germ cell tumors except for immature teratoma. GATA3 is negative or only very focally positive (as opposed to consistent staining in choriocarcinoma). There is therefore a characteristic immunohistochemical profile: positivity for cytokeratin, PLAP, OCT3/4, SOX2, and CD30 and negativity for EMA, CD117, and podoplanin. This profile can be of great value in distinguishing embryonal carcinoma in an extragonadal site (either a metastasis or an extratesticular primary) from a poorly differentiated carcinoma of nongerm cell origin (typically, cytokeratin and EMA positive, PLAP variable [but most commonly negative], and negative for CD30 and OCT3/4). The presence of CD30 in embryonal carcinoma indicates the necessity for caution and additional supportive evidence before accepting a CD30-positive, poorly differentiated malignant neoplasm as an anaplastic large cell lymphoma. Occasionally, embryonal carcinoma will also stain for α-1-antitrypsin, Leu-7, vimentin, LDH, human placental lactogen (hPL), and ferritin. The product of the TP53 tumor suppressor gene is often identifiable in embryonal carcinoma, although the absence of mutations with molecular biologic techniques support that this is the result of nonmutated overexpression.
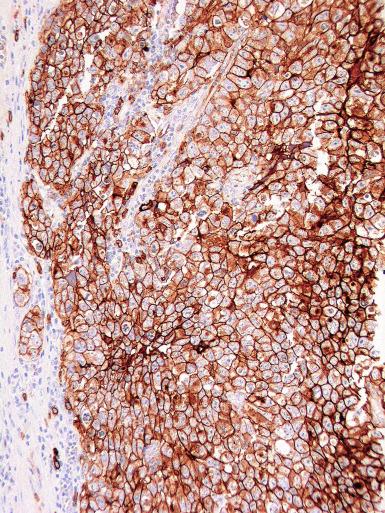
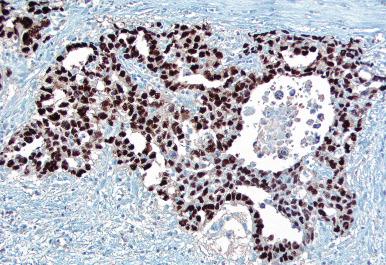
Ultrastructurally, embryonal carcinoma usually resembles a poorly differentiated, primitive adenocarcinoma with ill-defined lumens in solid areas and well-defined, large lumens in glandular areas. The lumens are bordered by cells having well-defined junctional complexes with characteristically long tight junctions. Short microvilli project into the luminal spaces, and the cytoplasm contains ribosomes, a prominent Golgi body, rough endoplasmic reticulum, teleolysosomes, mitochondria, glycogen, and occasional lipid droplets. The nuclei are large, deeply indented, often contain cytoplasmic inclusions, and have large nucleoli with complex nucleolonema.
Embryonal carcinoma has a DNA index ranging from 1.4 to 1.6 times normal, which is significantly less than that of seminoma. Embryonal carcinoma often contains isochromosome 12p, and increased copy numbers of i(12p) have correlated with a more aggressive clinical course. The presence of i(12p) or other 12p amplifications in a poorly differentiated carcinoma, either metastatic or a primary neoplasm of an extragonadal site, may be a useful means of separating embryonal carcinoma from other poorly differentiated neoplasms. The detection of 12p abnormalities can be accomplished in interphase cells obtained from fresh biopsy samples or in paraffin-embedded tumor by the fluorescence in situ hybridization (FISH) technique.
The distinction of embryonal carcinoma from seminoma was discussed earlier in this chapter. The distinction of embryonal carcinoma from yolk sac tumor depends on the presence, in yolk sac tumor, of one of several distinctive patterns, and the larger and more pleomorphic nature of the neoplastic cells in embryonal carcinoma. Yolk sac tumor often contains hyaline globules and intercellular basement membrane, which are usually lacking in embryonal carcinoma, and AFP is much more likely to be present in yolk sac tumor than in embryonal carcinoma. CD30 positivity is characteristic of embryonal carcinoma and is not usually present in yolk sac tumor. OCT3/4 and SOX2 are positive in embryonal carcinoma and negative in yolk sac tumor. Glypican-3, an immunohistochemical marker for yolk sac tumor, is negative in seminoma and positive in 0% to 8% of embryonal carcinomas. Because embryonal carcinoma may transform into yolk sac tumor, there are foci where the distinction is arbitrary. Such cases, however, invariably show areas of both neoplastic types. The smudged, degenerate cells common in embryonal carcinoma may be misinterpreted as syncytiotrophoblast cells, causing a misdiagnosis of choriocarcinoma. These cells, however, lack hCG, and the background is usually not hemorrhagic, unlike true choriocarcinoma. Large cell lymphoma usually occurs in older patients, lacks the epithelial patterns usually identified in embryonal carcinoma, has an interstitial growth pattern, is not associated with GCNIS, is PLAP negative, only rarely OCT3/4 positive, cytokeratin negative, and LCA positive, features contrasting with those of embryonal carcinoma. The differential with “anaplastic” spermatocytic tumor is discussed on the Spermatocytic Tumor section later in this chapter.
The treatment of nonseminomatous germ cell tumors, including embryonal carcinoma, depends on the clinical stage of the patient. Most patients with clinical stage I tumor receive surveillance management after orchiectomy, although patients at risk for occult metastases of chemoresistant elements (somatic-type malignancies, see the Teratoma with Somatic-Type Malignancy section later in this chapter) are often recommended for nerve-sparing retroperitoneal lymph node dissection (RPLND) tailored to excise the commonly involved nodal groups ipsilateral to the affected testis. From 20% to 30% of clinical stage I patients with nonseminomatous germ cell tumors relapse on surveillance management protocols. These are almost entirely caused by nondetected retroperitoneal involvement. As evidence for this conclusion, in one series of clinical stage I patients, RPLND confirmed pathologic stage II disease in 28%. Adjuvant chemotherapy (two courses of cisplatin, etoposide, and bleomycin) without RPLND for clinical stage I patients has also been evaluated in a prospective trial. A recent study of 382 patients with clinical stage I nonseminomatous germ cell tumor randomized to RPLND or one course of chemotherapy (bleomycin, etoposide, and cisplatin) found equivalent quality of life outcomes.
With surveillance management, relapse can often be detected early by serum marker elevation, allowing almost all relapsing patients to be salvaged by standard chemotherapy. Surveillance thus avoids unnecessary RPLND in the 69% to 81% of clinical stage I patients who are cured by orchiectomy alone. A review of 560 clinical stage I patients managed by “surveillance only” found that 97% were tumor-free and that 72% required no therapy after orchiectomy. About 90% of patients who relapse do so within 2 years. Only 1.6% of patients who relapse do so after 5 years or more. Relapse is detected by abdominal CT scan or by serum tumor markers in an approximately equal number of cases. With treatment, patients have an excellent prognosis even with relapse, with the 5-year disease-specific survival approaching 100% for the entire group of surveillance-managed clinical stage I nonseminoma patients.
The proportion of clinical stage IA (clinical T1, N0, M0) patients managed by surveillance increased from 65% between 2004 and 2005 to 74% between 2012 and 2013. Some clinical stage I patients at high risk for relapse may be less suitable candidates for “surveillance only” and may be identified by a careful, multifactorial analysis of the orchiectomy specimen. Factors that correlate with relapse or occult retroperitoneal metastases include: lymphovascular invasion, large proportion or volume of embryonal carcinoma, pure embryonal carcinoma, absence of a yolk sac tumor component, embryonal carcinoma in the absence of teratoma, less than 50% teratoma, the presence of choriocarcinoma, rete testis invasion, high S or G 2 M + S phase values as determined by flow cytometry, a high proportion of proliferating tumor cells, and highly aneuploid tumor stemlines. In a recent study of nonseminomatous germ cell tumors, only vascular invasion, rete testis invasion, and hilar soft tissue invasion were predictive of advanced clinical stage in multivariate analysis. Embryonal carcinomas with a high Ki-67 labeling index, low apoptosis, and low p53 expression have a better overall survival. A pre-orchiectomy AFP value exceeding 80 ng/mL and an abnormally slow decline in AFP values after orchiectomy have also correlated with relapse. In Europe, lymphovascular invasion has been used as a sole risk factor to recommend one cycle of chemotherapy (if lymphovascular invasion is present) or the choice between chemotherapy and surveillance (if absent).
Patients having clinically apparent, nonbulky retroperitoneal involvement (clinical stage II) may be managed by RPLND with either close follow-up or a limited course of adjuvant therapy or with initial chemotherapy. Survival in excess of 95% is expected. For patients with bulky clinical stage II or more advanced tumor, treatment is combination chemotherapy followed by surgical resection of residual masses if the serum markers have normalized. Survival of 70% to 80% is expected. Specimens resected after chemotherapy must be carefully evaluated pathologically to determine the need for additional chemotherapy. Total lymph node count of 28 or more is associated with a slightly decreased risk for relapse after RPLND compared with patients with fewer lymph nodes identified. It should be noted that even in the absence of viable tumor in a postchemotherapy RPLND specimen, recurrence may be seen in approximately 18% of patients.
Several different studies have been conducted to identify prognostic factors in patients with nonseminomatous testicular germ cell tumors. The most important factors are tumor stage, the extent of serum marker elevation, the age of the patient (older being worse), the presence of choriocarcinoma, and the proliferative fraction by flow cytometry. HER2 overexpression was prognostically significant for mixed germ cell tumors in one study but not in another. Epidermal growth factor receptor (EGFR) immunohistochemical overexpression has been reported in 43% of cases of chemorefractory metastatic embryonal carcinoma, suggesting that anti-EGFR targeted therapy could be a possibility for this group. Most of these tumors (71%) have polysomy for chromosome 7 rather than amplification of the EGFR gene. Treatment-resistant germ cell tumors are also more likely to have BRAF mutations, lack expression of the mismatch repair proteins MLH1 or MSH6, and demonstrate microsatellite instability on molecular analysis. The association with BRAF mutation in particular is of interest because of the possibility of tyrosine kinase inhibitor treatment in patients with relapse after initial chemotherapy.
A consensus classification of germ cell tumors into good, intermediate, and poor prognosis categories was published in 2013 and reproduced in the recent WHO Classification. Nonsemionmatous germ cell tumors of gonadal or retroperitoneal location, low serum tumor markers (AFP less than 1000 ng/mL, hCG less than 500 U/L, LDH less than 1.5 times the upper limit of normal), and no extrapulmonary visceral metastases are in the favorable prognosis category. Those with intermediate-level serum tumor marker elevation are in the intermediate prognosis category. Those with high-level tumor marker elevation (AFP over 10,000 ng/mL, hCG over 50,000 U/L, LDH greater than 10 times the upper limit of normal) or extrapulmonary visceral metastases are in the poor prognosis category.
Late recurrences are seen in a small percentage of patients (1% to 6%), can occur 30 years or more after initial treatment, and are likely to be treatment resistant. Somatic-type malignancies are seen in approximately 23% of late recurrences. Up-front surgical resection should be considered for late recurrences.
Yolk sac tumor is the most recently recognized among the five major categories of testicular germ cell tumor. For years, ovarian yolk sac tumor was misclassified, along with ovarian clear cell carcinoma, as “mesonephroma.” Teilum recognized the identity between testicular and ovarian yolk sac tumor and the frequent admixture of the testicular lesion with other forms of germ cell tumor. This observation permitted the classification of yolk sac tumor as a form of germ cell neoplasm and removal of the ovarian lesion from the “mesonephroma” category. Subsequently, Teilum recognized the resemblance of the mesenchyme of yolk sac tumor to the extraembryonic mesenchyme of development and its glomeruloid structures to the endodermal sinuses of the rat placenta.
Postpubertal patients with yolk sac tumor fall within the usual age spectrum for nonseminomatous testicular germ cell tumor, ranging from 15 to 45 years and averaging 25 to 30 years; rare cases have been reported in elderly patients. Prospective studies of nonseminomatous germ cell tumors have shown yolk sac tumor elements in 44% of cases.
Adults with yolk sac tumor in a mixed germ cell tumor are more likely to have lower-stage disease than those without a yolk sac tumor component, and the absence of a yolk sac tumor component in a mixed germ cell tumor may be a positive predictor of occult metastases in clinical stage I tumors. Almost all patients with yolk sac tumor have significant elevation of serum AFP, typically ranging from hundreds to thousands of ng/mL. Embryonal carcinoma or enteric elements of teratoma may cause minor elevation of serum AFP, but this is unusual. ,
The appearance is usually heterogeneous, with frequent areas of hemorrhage, necrosis, and cystic change ( Fig. 13.59 ). Numerous microscopic patterns are seen in yolk sac tumor and commonly include hybrid, incomplete, and transitional forms. The patterns, modified from Talerman, include: (1) microcystic (honeycomb, reticular, vacuolated); (2) endodermal sinus (perivascular); (3) papillary; (4) solid; (5) glandular/alveolar; (6) myxomatous; (7) sarcomatoid; (8) macrocystic; (9) polyvesicular vitelline; (10) hepatoid; and (11) parietal.
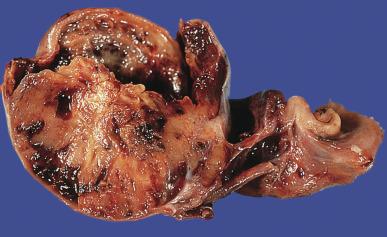
The microcystic pattern is most common and is characterized by intracellular vacuoles creating attenuated lengths of cytoplasm connected in a spiderweb-like array ( Fig. 13.60 ). The cells often resemble lipoblasts, with vacuoles compressing the nuclei, although they do not contain lipid. In some cases, the cells are arranged in cords and surround extracellular spaces, creating a reticular arrangement ( Fig. 13.61 ). The microcystic pattern is often seen with a myxoid stroma and blends with the myxomatous pattern ( Fig. 13.62 ). The solid pattern is also commonly intermingled with the microcystic ( Fig. 13.63 ).
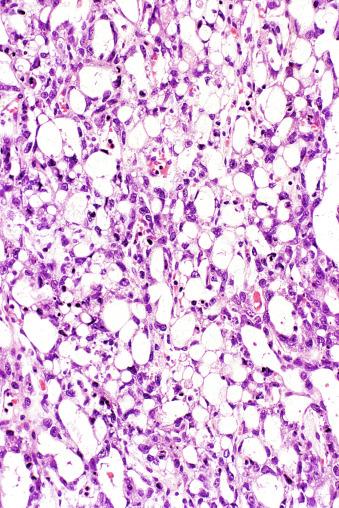
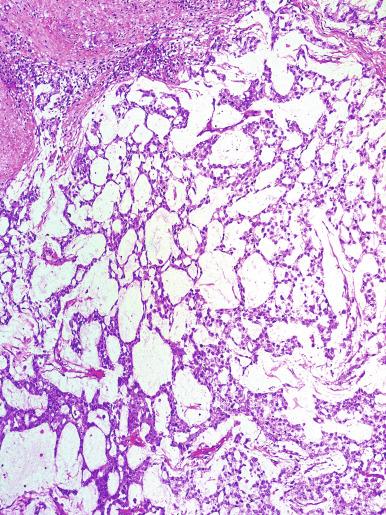
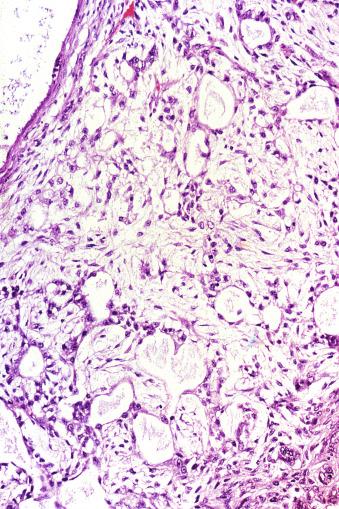
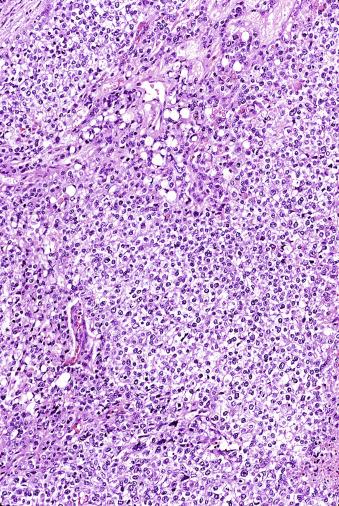
The endodermal sinus pattern consists of a central vessel rimmed by fibrous tissue that is surrounded by malignant epithelium. This structure is set in a cystic space that is often lined by flattened tumor cells ( Figs. 13.64 and 13.65 ). Oblique cuts of these structures result in fibrovascular cores of tissue that are “draped” or “festooned” by malignant cells with an accompanying complex (“labyrinthine”) arrangement of anastomosing extracellular spaces ( Fig. 13.66 ). This pattern is also designated as a “perivascular” or “festoon” pattern, and the endodermal sinus-like structure is termed a glomeruloid or Schiller-Duval body .
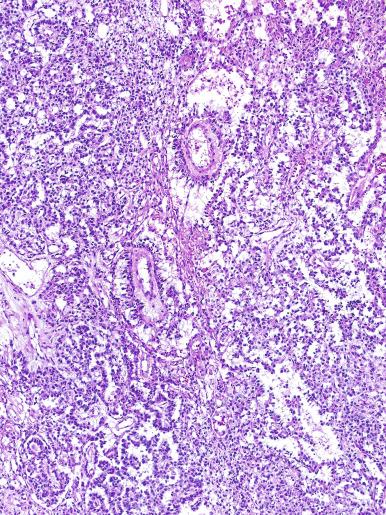
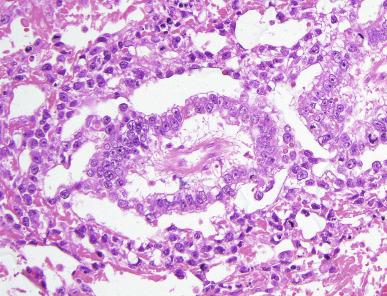
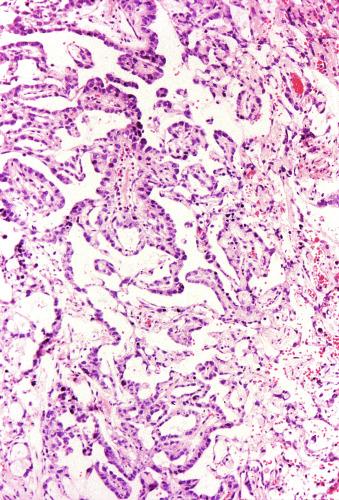
The papillary pattern has papillae, with or without fibrovascular cores, which project into cystic spaces ( Fig. 13.67 ). The cells are often cuboidal to low columnar and may have a “hobnail” configuration secondary to apical protrusion of the nucleus. Exfoliated clusters of neoplastic cells may be present in the cystic spaces. The papillary pattern may blend with the endodermal sinus pattern.
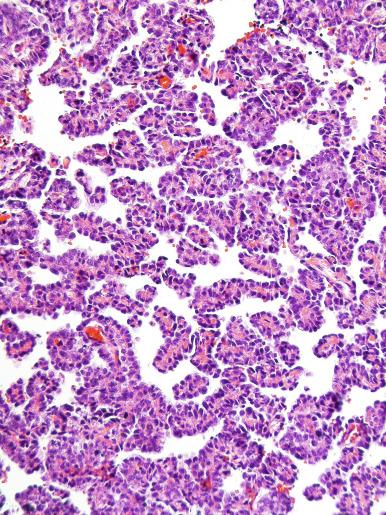
The solid pattern is quite common and may resemble seminoma, consisting of sheets of relatively uniform cells with lightly staining to clear cytoplasm and well-defined borders ( Fig. 13.68 ). However, the lymphoid component and fibrous septa of seminoma are usually absent, and the cells are more pleomorphic than those of seminoma. Some areas of solid pattern tumor have prominent thin-walled blood vessels, and focal microcysts may also be seen in an otherwise solid pattern ( Fig. 13.63 ). In some cases, the solid pattern has small cells with scant cytoplasm resembling blastema ( Fig. 13.69 ); such foci are intimately intermingled with classic patterns of yolk sac tumor ( Fig. 13.70 ).
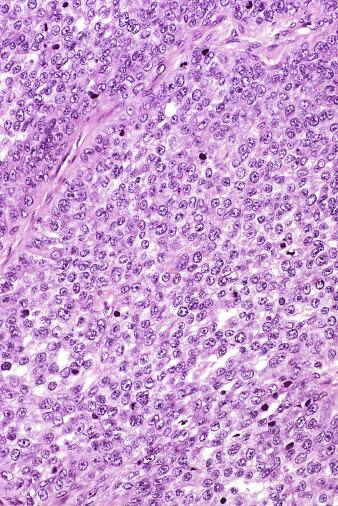
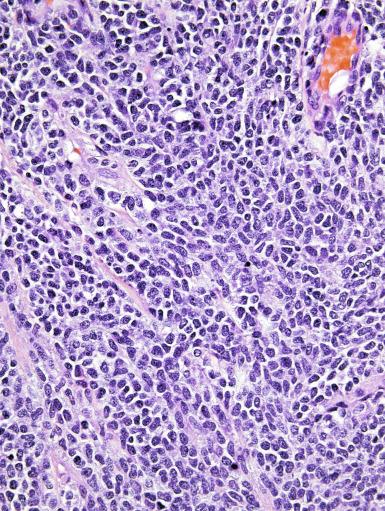
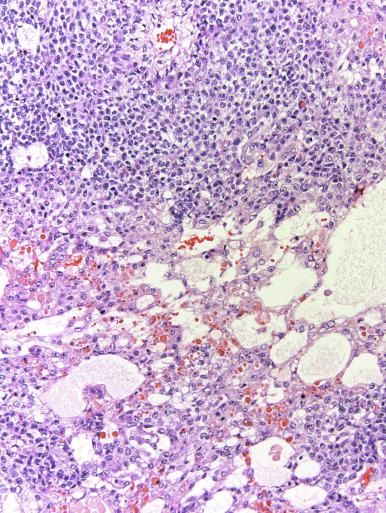
Well-defined glands, often with enteric features, are common in yolk sac tumor ( Fig. 13.71 ), present in 34% of cases in one series. The glands may be contiguous with vesicles typical of the polyvesicular vitelline pattern, or may appear in a background of myxomatous, microcystic, or solid patterns. Usually the glands are simple, round, and tubular, but may show an elaborate branching pattern or become quite intricate and complex ( Fig. 13.72 ). Unlike the glands of teratoma, those of yolk sac tumor are not associated with other teratomatous components and lack circumferential smooth muscle that is common but not invariable in teratoma. The nuclei of the glands are often more bland than those of the surrounding yolk sac tumor and may show subnuclear vacuolation, reminiscent of early secretory pattern endometrium. As a consequence, predominantly glandular ovarian yolk sac tumor has been termed endometrioid-like. Purely or predominantly glandular testicular yolk sac tumor is more rare than in the ovary, but may be associated with a high serum AFP. Purely glandular yolk sac tumor is more common after chemotherapy and is therefore usually found in metastases; it is particularly more frequent in late recurrences.
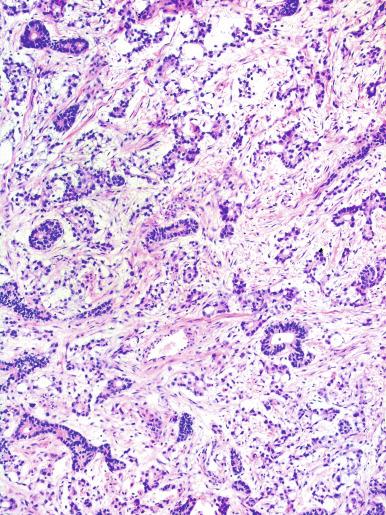
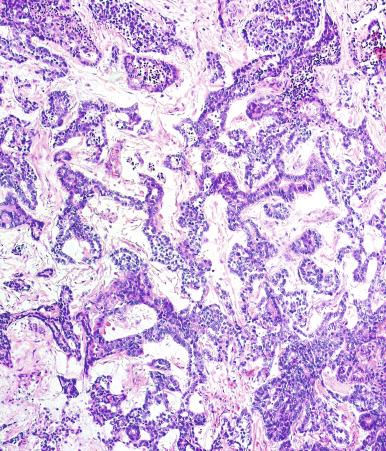
The myxomatous pattern is common, consisting of neoplastic epithelioid to spindle cells dispersed in a stroma that is rich in mucopolysaccharide, staining only lightly with hematoxylin and eosin ( Fig. 13.73 ). A prominent vascular network is common, and Teilum described this pattern as “angioblastic mesenchyme” that he felt was homologous with the extraembryonic mesenchyme (the “magma reticulare”) of development. Myxomatous foci commonly merge with other patterns, and hybrids of microcystic and myxomatous patterns are more the rule than the exception ( Fig. 13.62 ). By light microscopy, the spindle cells appear to arise from solid or microcystic foci by budding from them and blending into the surrounding myxoid stroma ( Fig. 13.74 ). Intense cytokeratin immunoreactivity within these cells supports derivation from the epithelial component of yolk sac tumor. These cells are, in fact, pluripotential cells with the capacity to form differentiated mesenchymal tissue such as skeletal muscle, cartilage, and bone, thus blurring the distinction between yolk sac tumor and teratoma ( Fig. 13.74 ). Classification of such elements as yolk sac tumor is justified by the recognition that the surrounding tissues are typical yolk sac tumor.
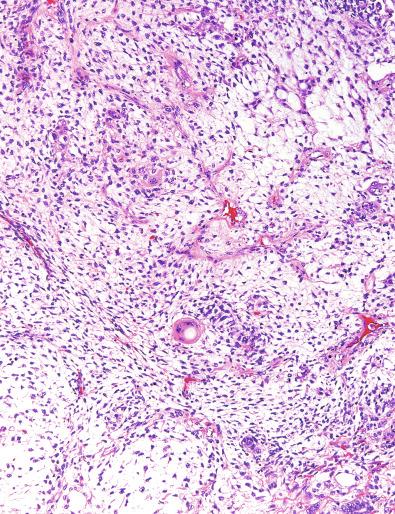
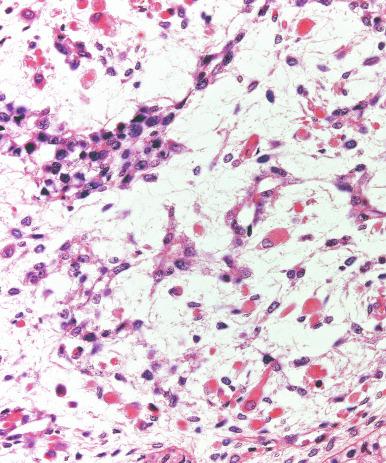
The uncommon sarcomatoid pattern consists of a cellular proliferation of spindle, and usually also epithelioid, cells ( Fig. 13.75 ) in continuity with other yolk sac tumor patterns, most commonly microcystic. It is distinguished from the solid pattern by the spindle cell nature of the component cells and the myxomatous pattern by its greater size as a single pattern. Despite the sarcomatoid appearance, the spindle cells express cytokeratin and glypican-3 and may also react for SALL4 but not AFP. It is likely that some embryonal rhabdomyosarcomas arising in testicular germ cell tumors derive from differentiation of sarcomatoid spindle cells to rhabdomyoblastic cells. The occasional intimate admixture of embryonal rhabdomyosarcoma with yolk sac tumor supports this hypothesis. Spindle cell patterns in yolk sac tumor have also been reported in primary mediastinal examples.
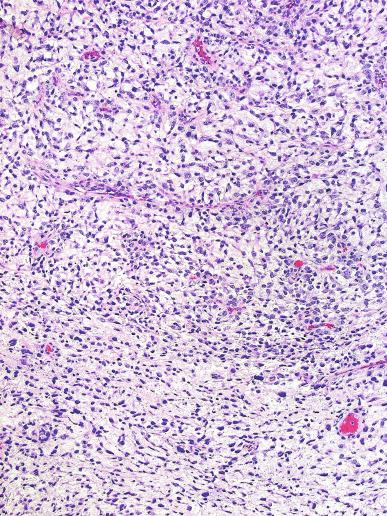
The macrocystic pattern appears to arise from coalescence of microcystic spaces to form large, round to irregular cysts ( Fig. 13.76 ), and the surrounding pattern is often microcystic.
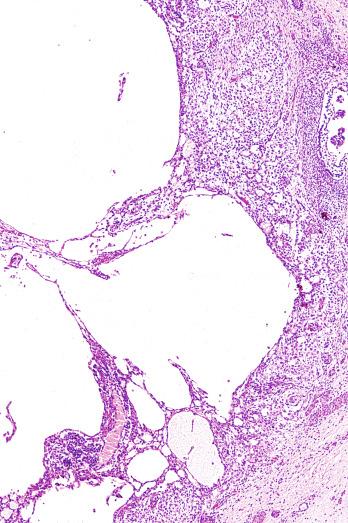
In the uncommon polyvesicular vitelline pattern, vesicle-like structures are lined by flattened, innocuous-appearing epithelium, in a myxoid to fibrous stroma ( Fig. 13.77 ). Sometimes the vesicles have a central constriction, resembling a dumbbell or a figure 8. Teilum compared these vesicles to the embryonic subdivision of the primary yolk sac into the secondary yolk sac. At the point of constriction, the epithelium may change from flattened to cuboidal or columnar; the latter often has enteric features, including an apical brush border. AFP is often present in the epithelium of the vesicles, and hyaline globules are occasionally seen within them. In some cases, a transition from a microcystic to polyvesicular vitelline pattern can be identified. The bland cytologic appearance of the polyvesicular vitelline pattern may falsely suggest a benign neoplasm, but the presence of other patterns should prevent this pitfall. The polyvesicular pattern is less frequent in testicular yolk sac tumor than in its ovarian counterpart, where it may rarely occur in pure form.
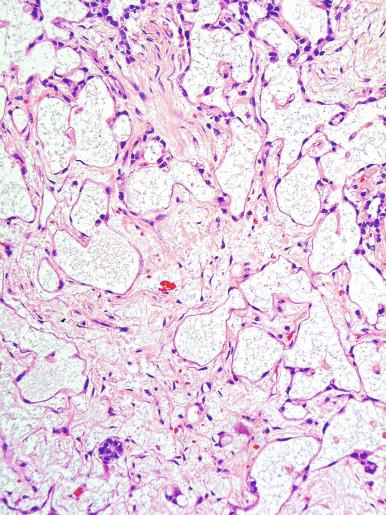
A hepatoid pattern also occurs in about 20% of yolk sac tumors and consists of small clusters of polygonal, eosinophilic cells arranged in sheets, nests, or trabeculae ( Fig. 13.78 ). The cells have round, vesicular nuclei with prominent nucleoli and contain abundant AFP; hyaline globules are common in hepatoid foci as are bile canaliculi, although bile is a rare finding. Hepatoid foci are scattered randomly in yolk sac tumor and usually are minor components; rarely, a more diffuse hepatoid pattern may be seen, although a prominent hepatoid pattern is more common in ovarian tumors and in late recurrences of testicular tumors.
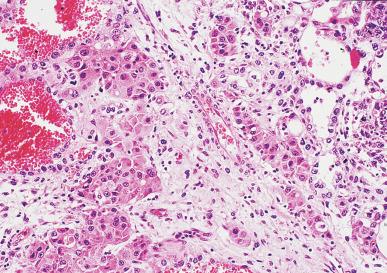
The parietal pattern has extensive deposits of extracellular basement membrane, with only scattered neoplastic cells in an abundant, eosinophilic matrix ( Fig. 13.79 ). It is considered the extreme end of parietal differentiation (see later) in which basement membrane is deposited in the extracellular space in a variety of yolk sac tumor patterns. In a true parietal pattern yolk sac tumor, the basement membrane deposits efface the underlying yolk sac tumor pattern. This is a rare pattern, most often seen after chemotherapy, particularly in late recurrences. There is some overlap with the sarcomatoid pattern when the tumor cells are spindled, and we have seen some cases that, in postchemotherapy resections, mimicked osteosarcoma.
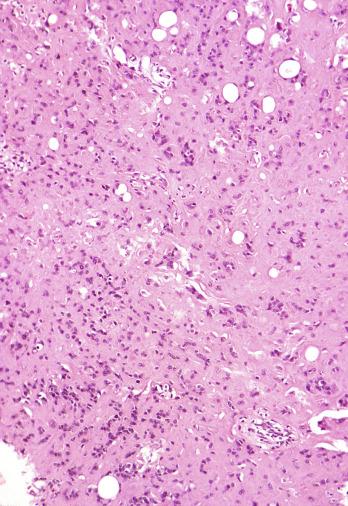
The frequency of the different patterns of yolk sac tumor is difficult to determine because of lack of uniformity in classification. The microcystic, solid, and myxomatous patterns are most common, with glandular, macrocystic, endodermal sinus, hepatoid, and papillary patterns also occurring frequently. The polyvesicular vitelline pattern is less common, and sarcomatoid and parietal patterns are unusual. If only four patterns are employed for classification, the frequency of the patterns diminishes from reticular to solid to endodermal sinus to polyvesicular vitelline. Jacobsen noted a “vacuolated network” in 91% of yolk sac tumors, microcystic pattern in 67%, myxomatous pattern in 51%, macrocystic pattern in 44%, solid pattern in 27%, hepatoid areas in 23%, labyrinthine formations in 17%, and endodermal sinus-like structures in 9%.
A common feature of yolk sac tumor is the deposition of extracellular basement membrane, identified in 92% of cases. These deposits generally are irregularly shaped, eosinophilic bands between the neoplastic cells ( Fig. 13.80 ) and have been referred to as parietal differentiation because of their homology to the parietal layer of the embryonic yolk sac of the rodent, which rests on a thick basement membrane (known as Reichert membrane). Such intercellular basement membrane, although not specific for yolk sac tumor, is characteristic and can be helpful in diagnosis, particularly in small biopsy samples taken from extratesticular tumors. Another characteristic but nonspecific feature in most yolk sac tumors is the presence of intracellular, round, hyaline globules of variable size (from 1 to more than 50 μm in diameter) ( Fig. 13.81 ). These globules are PAS-positive and diastase-resistant and may be present in the extracellular space after cell necrosis. Occasionally these globules may stain positively for AFP, but most do not; hence the staining is considered nonspecific. The hyaline globules and basement membrane deposits of yolk sac tumor are separate and distinct findings, although they have sometimes been confused in the literature.
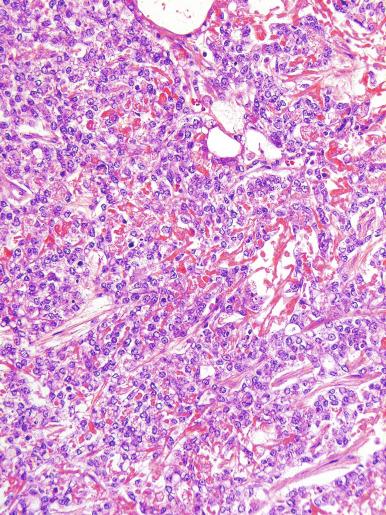
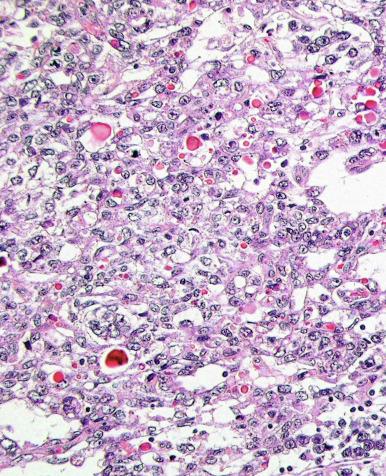
Hematopoietic elements, usually erythroblasts, are present in a minority of testicular yolk sac tumors, usually in vascular spaces or myxomatous tissues.
Most yolk sac tumors show cytoplasmic AFP positivity on immunostaining; the frequency varies from 50% to 100% depending on the technique employed and the number of blocks examined. Positivity is characteristically patchy ( Fig. 13.82 ); intense staining is usually present in hepatoid foci. Postpubertal-type yolk sac tumor stains more frequently for AFP than prepubertal-type. Glypican-3, a proteoglycan that plays a role in embryonic growth, is positive in yolk sac tumor and is much less commonly expressed in most other forms of testicular germ cell tumor ( Fig. 13.83 ). Glypican-3 is more sensitive for yolk sac tumor than AFP, staining 100% of cases in one study compared with 58% for AFP. Staining was seen in all patterns of all cases evaluated, except the solid pattern of a single case. Glypican-3 staining has been reported in some tumors other than yolk sac tumor and hepatocellular carcinoma, however, including cholangiocarcinoma, hepatoblastoma, immature teratoma, melanoma, and pulmonary squamous cell carcinoma, so caution is necessary if these tumors are in the differential diagnosis. Among other primary testicular tumors, focal staining may be seen in some embryonal carcinomas, teratomas, and spermatocytic tumors. Positivity for α-1-antitrypsin occurs in about 50% of yolk sac tumors, and the enteric glands may stain for carcinoembryonic antigen and villin, which also highlight the purely glandular tumors.
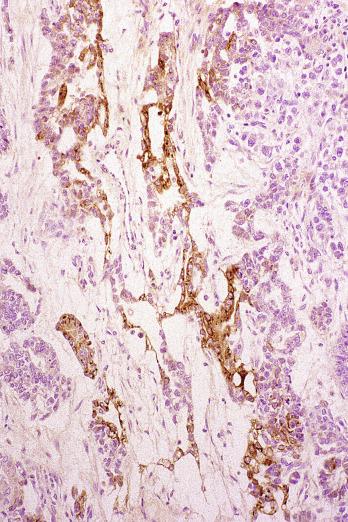
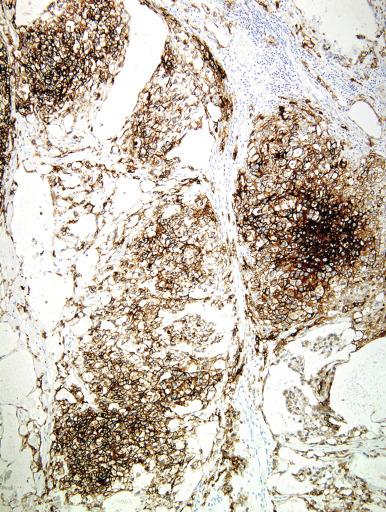
SALL4, a regulator of OCT3/4, is a nuclear marker that is expressed in essentially all cases of GCNIS, seminoma, spermatocytic tumor, embryonal carcinoma, and pre- and postpubertal type yolk sac tumors. This marker is consistently negative in testicular tumors other than germ cell tumors (including stromal tumors), so it is useful to identify germ cell tumors as a group. It is also useful to identify metastatic germ cell tumors, such as in the setting of a metastatic yolk sac tumor with glandular or solid pattern and absent AFP expression, although occasional somatic malignancies including esophageal, gastric, and colonic adenocarcinomas may show weak staining. Among somatic malignancies, ovarian serous carcinoma has the highest (29%) frequency of SALL4 staining, but urothelial and gastric carcinomas stain in almost one-fourth of cases, and other tumors stain in single-digit percentages. SALL4 tends to be more diffusely positive in yolk sac tumor ( Fig. 13.84 ) than glypican-3. Cytokeratin is present in virtually all cases, although cytokeratin 7 is characteristically absent. Positivity with HEA 125, an epithelial marker, occurs in most cases. Albumin, ferritin, neuron-specific enolase, and Leu-7 are present in a variable number of cases. Chromogranin reactivity is unusual. From 39% to 85% of yolk sac tumors are reported as positive for PLAP, and EMA is usually negative, as is CD99. p53 may be identified in yolk sac tumor, and laminin and type IV collagen are present in areas of parietal differentiation. We have noted reactivity of hepatoid foci for hepatocyte-specific antigen (HepPar1; clone OCH1E5) (unpublished observations, 2001) and others have reported HepPar1 positivity in occasional yolk sac tumors in the absence of light microscopically evident hepatoid features. In addition to HepPar1, other endodermal lineage markers such as TTF1 (2 of 15 cases) and CDX2 (6 of 15 cases) may show intermediate- to high-level expression in some testicular yolk sac tumors. The growth differentiation factor 3 (GDF3) antibody is immunoreactive with yolk sac tumors and a GDF3 +/OCT3/4–/CD30– immunophenotype is supportive of the diagnosis in morphologically difficult cases. GATA3 is expressed by yolk sac tumor, but choriocarcinomas are also positive; focal staining may be seen in some embryonal carcinomas, and staining is seen in carcinomas of many somatic sites, limiting its usefulness.
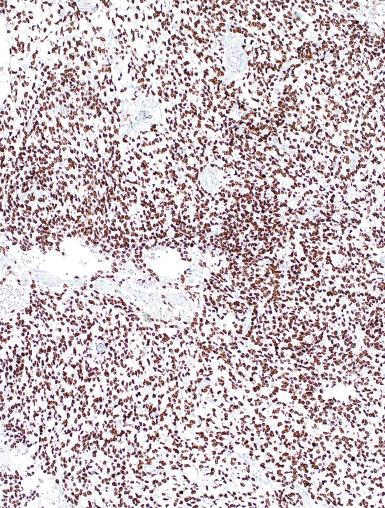
Ultrastructurally, yolk sac tumor shows clusters of epithelial cells joined by junctional complexes. Glands have microvilli with glycocalyceal bodies and long anchoring rootlets. Basement membrane material can be identified in the extracellular space, and flocculent material is present within dilated cisternae of endoplasmic reticulum, often with a central lucent zone. Cytoplasmic glycogen may be conspicuous. The nuclei are usually irregular, with complex nucleolonema. Densely osmiophilic, cytoplasmic, nonmembrane bound, round bodies correspond to the hyaline globules observed at the light microscopic level.
Solid pattern yolk sac tumor must be distinguished from seminoma, an issue that has been addressed in the Seminoma section earlier in this chapter. The distinction of yolk sac tumor from embryonal carcinoma has less clinical significance but is based on the distinctive patterns of yolk sac tumor and the less pleomorphic, less atypical nature of the neoplastic cells. CD30, OCT3/4, AFP, and glypican-3 stains are helpful; the first two are positive in embryonal carcinoma but not yolk sac tumor, and the latter two are positive in many yolk sac tumors and usually negative in embryonal carcinomas. Embryonal carcinoma probably transforms to yolk sac tumor, so there are transitional forms that are difficult to categorize. A hyperplastic reaction of the rete testis with hyaline globules may be induced by invasion of the rete by a neoplasm, thus simulating yolk sac tumor ( Fig. 13.85 ). The arborizing pattern of the rete testis and the bland nature of the hyperplastic cells should prevent this misinterpretation, although immunostains can also be used if doubt persists.
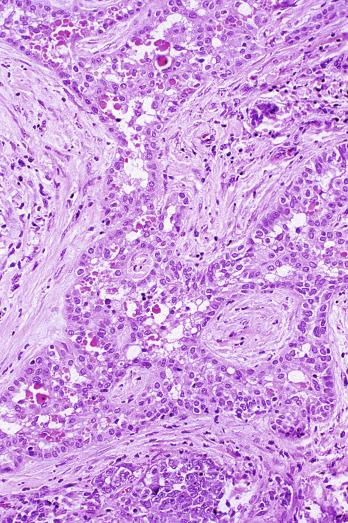
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here