Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Parathyroid adenoma
Parathyroid carcinoma
Secondary neoplasms
Definition: Benign neoplasm of the parathyroid parenchymal cells, including chief cells and/or oncocytic cells.
| Hyperplasia | Adenoma | Carcinoma | |
|---|---|---|---|
| Gender; age | Slight female predilection; most common in 5th-6th decades | More common in women; most common in 4th decade | Equal gender predilection; wide age range |
| Clinical | Asymptomatic or complaints of lethargy, weakness, polyuria, polydipsia, arthralgia, constipation, and depression | Similar to hyperplasia | Similar to hyperparathyroidism of benign cause but more severe due to the higher serum calcium levels; higher proportion of renal disease (nephrolithiasis) and bone disease; peptic ulcer disease; palpable neck mass more common than in adenoma |
| Serum calcium | 11.7 mg/dl (average) | 12.5-13.5 mg/dl | Often >14 mg/dl |
| Intraoperative findings | 2 or more glands enlarged, easily dissected; enlargement may be very asymmetric | 1 gland enlarged; easily dissected; more frequent in lower glands or ectopic sites | 1 gland enlarged; often adherent to surrounding tissues |
| Weight | Total gland weight usually <1 g, but may be up to 5 g | 0.3-1.0 g commonly, but may weigh several grams in patients with bone disease | >1.5 g (often much larger) |
| Capsule | Circumscribed by capsule of parathyroid gland, may be incomplete. No compressed rim of atrophic or normal parathyroid tissue | Thin tumor capsule, often surrounded by rim of uninvolved parathyroid, which may appear atrophic | Thickened capsule; rim of normal parathyroid rarely seen |
| Gross appearance | Gray-brown, soft. Cut surface may be homogeneous or nodular. Lacks fibrous bands | Red-brown, firm. Usually homogeneous, lacks fibrous bands | Gray-white, firm, often lobulated or irregular. Fibrous bands often produce coarse nodularity |
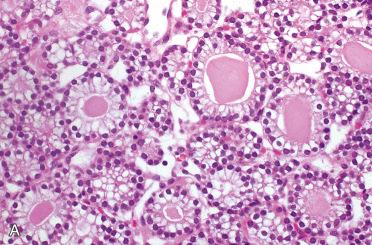
| Histologic pattern | Diffuse or nodular, sometimes pseudofollicular or acinar | Diffuse or nodular, frequently pseudofollicular or acinar | Diffuse, nodular, pseudofollicular, or acinar; often trabecular pattern with distinctive nuclear palisading predominates |
| Cytologic features | Chief cells predominate; transitional and oncocytic cells often present | Chief cells predominate, but mixture of chief, transitional, and oncocytic cells may be seen; rarely, purely oncocytic | Cells usually resemble chief cells, but variable cytoplasmic oncocytic change may be seen; cell borders often indistinct |
| Intracytoplasmic lipid | Decreased | Decreased in tumor; abundant in atrophic rim of parathyroid | Usually absent |
| Stromal fat cells | Scanty to absent | Usually absent in tumor; present rim of atrophic parathyroid | Absent |
| Nuclear morphology | Normal to slightly increased N-to-C ratio; usually without nuclear pleomorphism | Nuclei enlarged, with variability in size; scattered groups of large pleomorphic, hyperchromatic nuclei, or multinucleated cells | Increased N-to-C ratio; enlarged atypical nuclei; often with monotonous (bland appearing) nuclei |
| Nucleoli | Inconspicuous to small | Inconspicuous to small | Frequently prominent and enlarged |
| Mitoses | Common (60% of cases; most with <1 mitotic figure/10 HPF) | Common (70% of cases; most with <1 mitotic figure/10 HPF) | Common (80% of cases), may include atypical mitoses; may be numerous |
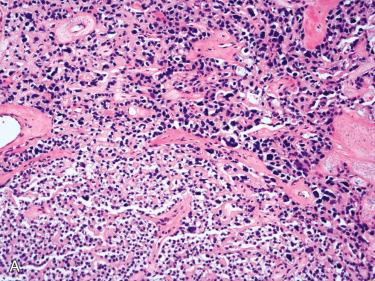
| Capsular and vascular invasion | Absent | Absent; entrapment of tumor cells may occur in capsule if degenerative changes present | Capsular invasion present in two thirds; may involve only capsule or extend into adjacent tissues Vascular invasion present in up to 15%; usually in capsular vessels |
| Remainder of gland | Entire gland is abnormal | Normal or atrophic | Normal |
| Degenerative changes | May be seen in very large glands Includes hemorrhage, areas of fibrosis, and cystic change |
Common, especially in larger adenomas; includes hemorrhage, fibrosis, and cystic change, sometimes calcification | Tumor cell necrosis; calcification and cystic changes may be present |
| Treatment | Subtotal parathyroidectomy with surgical removal of three glands, leaving a remnant of the 4th or total parathyroidectomy * with autotransplantation of parathyroid tissue in forearm | Surgical removal of the enlarged gland | En bloc resection, including ipsilateral thyroid lobe and adjacent soft tissues |
| Prognosis | Excellent | Excellent | Up to 50% of patients are cured by en bloc resection; considered an indolent malignancy even in presence of recurrence or metastasis with long survival even after recognition of tumor recurrence; morbidity and mortality correlate to complications of severe hypercalcemia |
| Recurrence and metastasis | Recurrence in approximately 16% of cases due to inadequate neck exploration and may not be evident for years | Absent | Recurrence in two thirds of patients usually within 3 years of the first surgery; metastasis is 35%, is a late event usually preceded by local recurrence; most commonly to lung, cervical lymph nodes, and liver |
| Familial and/or MEN association | Yes, in approximately 20% of cases | Uncommon | Rare |
More common in females (F : M = 3 to 4 : 1); occur over a broad age range but are most frequently discovered in the fourth and fifth decades.
Represents the major cause of primary hyperparathyroidism
Clinical findings are essentially the same as those associated with primary hyperparathyroidism due to hyperplasia (see Chapter 32 ).
As in primary hyperparathyroidism due to hyperplasia, the symptomatology in patients with parathyroid adenomas is changing as a result of routine biochemical screening and early detection:
Hypercalcemia may be incidentally discovered in asymptomatic patients, and many patients complain only of fatigue, weakness, or depression.
Nephrolithiasis is documented in 69% of men and in 36% of women with adenomas overall, but the incidence has been decreasing in recent years to between 5% and 20%.
Severe bone disease, once a common complication, is now rare; however, osteopenia is often present, and joint disease similar to that found in patients with parathyroid hyperplasia occurs.
Serum calcium levels are generally higher than in patients with primary chief cell hyperplasia but not usually as high as in parathyroid carcinoma.
Hypophosphatemia, hyperphosphaturia, and elevated serum parathormone levels are found.
Rarely parathyroid adenomas present as a palpable mass.
Radiology:
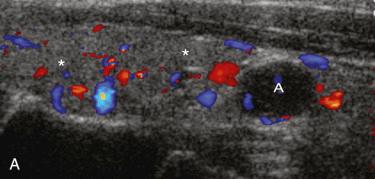
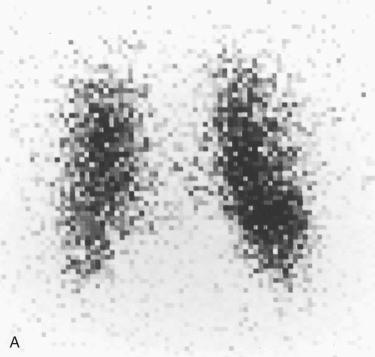
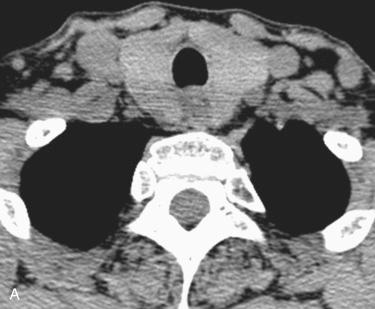
Several imaging methods have been used for localization of hyperfunctioning parathyroid tissue, including retrograde phlebotomy for determination of serum parathormone levels, CT scanning, ultrasonography, magnetic resonance imaging (MRI), thallium subtraction scanning, and technetium-99m sestamibi imaging.
Technetium-99m sestamibi imaging appears to be the most useful, with localization of more than 90% of adenomas, and has been most widely used in patients with anatomic distortion due to previous surgery and in patient who are high surgical risks; however, more routine utilization has gained support.
Multidimensional CT (referred to as 4D-CT) emerging technique used in detection when a lesion is not identified by more conventional techniques (e.g., ultrasonography, sestamibi imaging)
May be associated with hyperparathyroidism-jaw tumor syndrome (HPT-JT):
Autosomal-dominant disorder with germline mutation in HRPT-2 gene on chromosome 1q25-31
Characterized by:
Parathyroid adenoma or carcinoma
Fibro-osseous lesions of the jaw (e.g., ossifying fibroma of mandible or maxilla): 30% of cases
Renal cyst, hamartoma, carcinoma: 20% of cases
Approximately 80% of patients develop hyperparathyroidism:
Usually presents late in adolescence
Hypercalcemia tends to be severe.
Higher incidence of parathyroid carcinoma in comparison with patients with MEN-1 and MEN-2A
Renal lesions may include:
Renal cysts, polycystic renal disease, renal hamartoma
Papillary renal cell carcinoma, renal cortical adenomas, Wilms tumor
90% of adenomas are found in parathyroid glands in their usual locations:
Lower glands are more commonly involved.
Ectopic parathyroid adenomas:
Parathyroid adenomas may occur in any location in which parathyroid tissue may be found, including ectopic sites such as the mediastinum, retroesophageal soft tissue, within the thyroid gland, or in thymic tissue
Ectopic adenomas, especially intrathyroidal ones, may be missed by clinical evaluation and/or surgical exploration:
Intrathyroidal parathyroid adenomas may be misinterpreted by pathologic evaluation as a thyroid follicular lesion or medullary thyroid carcinoma.
Reports of adenomas occurring in supernumerary glands include tumors arising in the vagus nerve, pericardium, or other soft tissue sites in the neck.
Occasionally enlarged parathyroid glands, either hyperplastic or, more commonly, adenomatous, have been serendipitously subjected to fine-needle aspiration as a clinically suspected “solitary thyroid nodule”; reports of ultrasonically guided fine-needle aspiration for localization and confirmation of parathyroid proliferative disease have appeared.
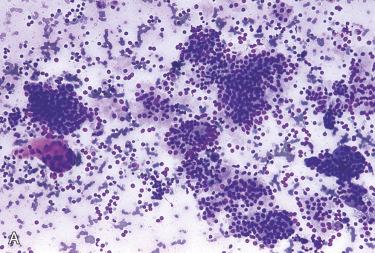
An awareness of the typical cytologic appearance of parathyroid tissue can be helpful during intraoperative examination of biopsies during a neck exploration for hyperparathyroidism, as examination of touch preparations provides a rapid means of confirming the presence of parathyroid tissue.
Aspirates of parathyroid tissue typically contain numerous naked nuclei, as well as small sheets of cells, sometimes forming acinar or follicular structures; small aggregates of dense colloid-like material may be seen but are not numerous.
The cells are generally small, with predominantly round nuclei; anisonucleosis in scattered cells and occasional large, atypical, naked nuclei are common.
The cytoplasm is granular and may exhibit scattered large metachromatic granules with a May-Grünwald-Giemsa or Romanowsky stain; Papanicolaou-stained cells have clear to finely granular cytoplasm.
The nuclei are generally hyperchromatic with coarse chromatin typical of neuroendocrine cells.
The distinction from follicular epithelium of the thyroid may be difficult, although the cells are usually smaller than those of the thyroid:
Immunohistochemical staining for PTH, parafibromin, chromogranin, thyroglobulin, and TTF1 may be helpful in this differential diagnosis.
Adenomas are almost always solitary (see below for Double Adenoma).
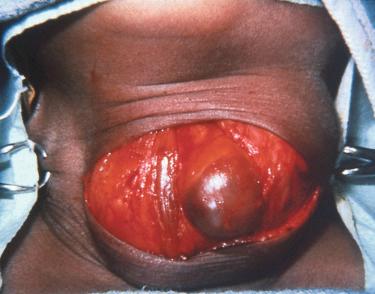
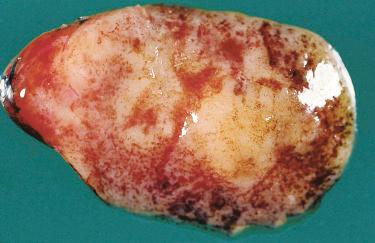
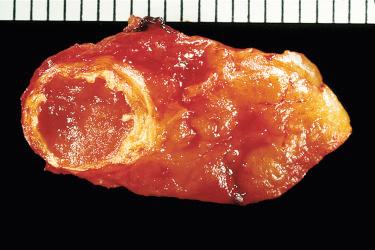
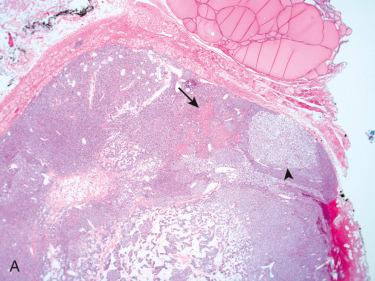
Adenomas have rounded borders, are firm, brown to tan, and are contained within a delicate capsule; they may be ovoid or lobulated.
A remnant of uninvolved parathyroid tissue at the periphery of the tumor may be visible.
Cystic change may be present, and when marked may mask the neoplastic nature of the proliferation; marked cystic degeneration is frequently associated with scarring and calcification.
There is significant variation in weight, with most between 0.3 and 1.0 g.
Well circumscribed and uncommonly surrounded by a thin fibrous capsule:
Complete encapsulation not commonly seen
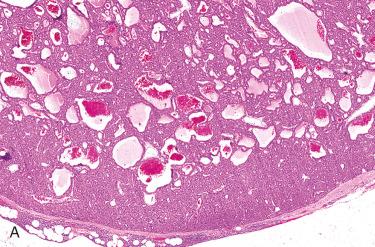
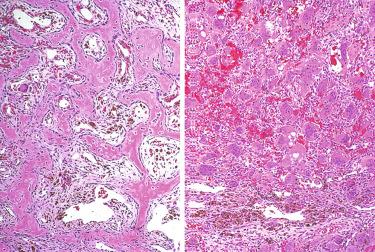
Hypercellular proliferation lacking intraparenchymal fat cells with diffuse growth; vague nodularity may be present and rarely may be multinodular.
A rim of non-neoplastic parathyroid tissue found in association with only about half of the adenomas:
Very helpful finding, if present, in making the distinction between adenoma and hyperplasia
“Rim” generally contains abundant stromal fat cells, in contrast to the very cellular adenoma.
Nuclei of residual normal or atrophic parathyroid gland typically smaller than the nuclei of the adenoma.
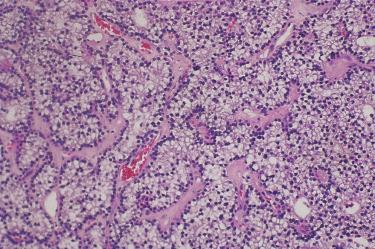
Growth patterns include solid (diffuse) sheets, cords, nests, acini, follicles, and microcysts:
Follicle formation may contain eosinophilic “colloid-like” material.
Distinct trabecular pattern is uncommon.
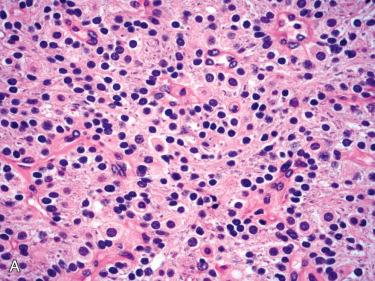
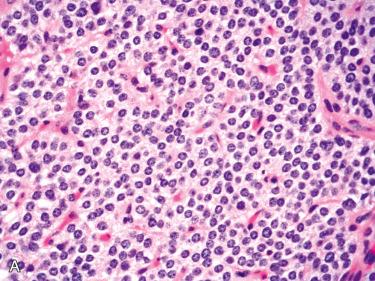
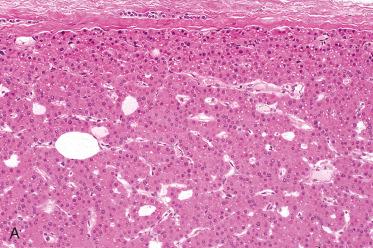
Predominantly composed of chief cells:
Nuclei round and regular with central to slightly basal location within the cell and have inconspicuous nucleoli; usually lack significant pleomorphism:
Cells with hyperchromatic enlarged nuclei as well as multinucleated cells are common and can be found scattered throughout the tumor or may be clustered in small foci; these scattered atypical nuclei are not an indicator of malignancy in the absence of other evidence of a malignancy (see section on parathyroid carcinoma ).
Cytoplasm varies, including slightly eosinophilic, amphophilic, clear, or oxyphilic.
Chief cells of an adenoma frequently larger than the non-neoplastic chief cells in the uninvolved rim of parathyroid tissue, if one is present
Oncocytic cells may be seen in variable numbers, focally admixed with chief cells or as nodular aggregates.
Mitotic figures are identifiable in many adenomas, but usually number fewer than 1 per 10 high-power fields; mitotic rates as high as 4 mitoses per 10 high-power fields have been described in occasional cases.
Delicate vascular network composed of thin fibrovascular stroma, sinusoid-like blood vessels or capillaries traverse the neoplasm.
Intraparenchymal fat cells absent but may focally be seen as single cells or groups in the peripheral aspect of the neoplasm:
Neoplastic cells usually have less intracellular fat than do the cells in the uninvolved (or suppressed) parathyroid tissue, either in other glands or in a rim of non-neoplastic parathyroid tissue in an adenomatous gland.
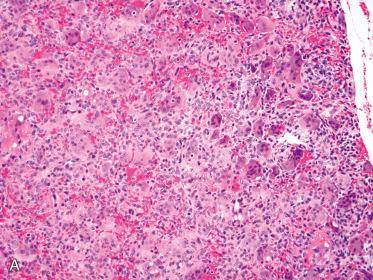
Cells with markedly enlarged and hyperchromatic pleomorphic or bizarre-appearing nuclei may be present and when present:
Appear admixed with bland-appearing nuclei.
Tend to be focally and not diffusely identified.
Occur in the absence of increased mitotic activity and/or other features that may be associated with parathyroid carcinoma.
Reactive and degenerative changes may be present, including cyst formation, edema, fibrosis, hemorrhage (fresh or in the form of hemosiderin deposition), or infarction:
May occur spontaneously or may occur following a traumatic event such as prior surgery to the neck or fine-needle aspiration biopsy
Presence of reactive and degenerative changes, especially fibrosis, may cause adherence to adjacent structures, suggesting invasive growth and a possible diagnosis of parathyroid carcinoma.
Infarcted tumor may retain antigenicity for parathyroid hormone.
Rarely, an associated mature lymphocytic cell infiltrate may be present.
Uninvolved parathyroid parenchymal cells in patients with adenomas are typically smaller and often have more stromal fat cells than the glands in patients without hyperparathyroidism; they also have more cytoplasmic fat, often found as large droplets, than normally functioning parathyroid glands.
Histochemical stains:
Colloid-like material in follicular structures are PAS positive.
Considerable variation in the literature regarding the utility of fat stains in the diagnosis of parathyroid proliferative diseases
Generally, hyperfunctioning cells have a significantly decreased amount of intracellular fat (using Sudan black or oil red O) compared with normal or suppressed parenchymal cells; there is, however, variability in this finding.
Fat stains, when used with adequate clinical information, intraoperative findings, and histologic examination, are useful if their limitations are kept in mind.
Immunohistochemistry:
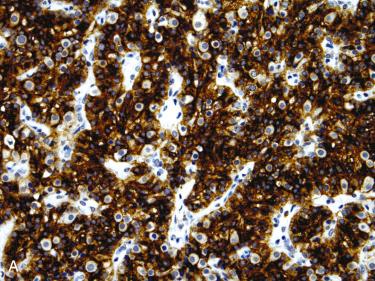
Positive for parathyroid hormone and parafibromin (nuclear staining):
Majority of parathyroid adenomas express parafibromin
Loss of parafibromin expression may be seen in patients with hyperparathyroidism-jaw tumor syndrome indicative of gene inactivation through mutation of the HRPT-2 gene.
Cytokeratin, chromogranin A positive
Calcitonin and synaptophysin typically negative but in small percentage of cases may be focally positive
PAX8 (nuclear) reactivity present in approximately 40% of adenomas and hyperplasia
Galectin 3 rarely positive (< 5%).
Ki67 (MIB1) proliferative index is low:
An index greater than 5% should raise suspicion for carcinoma, but the diagnosis of carcinoma requires confirmatory diagnostic findings.
Proliferative indices in differentiating adenoma from carcinoma are of limited utility given overlapping findings in these lesions.
Cyclin D1 staining in 39% of cases
Negative for thyroglobulin and TTF-1
Electron microscopy:
Adenomas associated with very high serum calcium levels may have a large number of microvilli, which are thought to reflect a higher level of endocrine activity
Adenomas often have more abundant rough endoplasmic reticulum and more prominent Golgi apparatus than non-neoplastic cells.
Annulate lamellae may be seen.
Cytogenetic and molecular findings:
Approximately 5% show pericentric inversion of chromosome 11, causing translocation of cyclin D1 ( CCND-1/PRAD-1 ) gene with parathyroid hormone gene, resulting in overexpression of cyclin D1.
Somatic mutation in MEN-1 gene at 11q13 in 40% of cases
Absence of RET mutation
5% have somatic mutation in CDKN1B gene (p27Kip1)
Most “multiple adenomas” represent cases of asymmetric or nodular hyperplasia:
Distinction between hyperplasia and adenoma may be extremely difficult and requires the pathologic examination of more than a single gland.
Diagnostic criteria for double adenomas include:
2 enlarged, hypercellular parathyroid glands
Intraoperative confirmation that remaining parathyroid glands are normal and/or biopsy proven histologically normal parathyroid glands
Absence of family history of MEN or familial hyperparathyroidism
Permanent cure of hypercalcemia following excision of enlarged glands:
Arguably the most definitive criterion
Requires years of follow-up to include monitoring of serum calcium and parathyroid hormone levels
If the above criteria are fulfilled, then a diagnosis of double adenomas can be confirmed.
True double parathyroid adenomas are rare:
Majority (greater than 70%) are bilateral
Predilects to superior glands
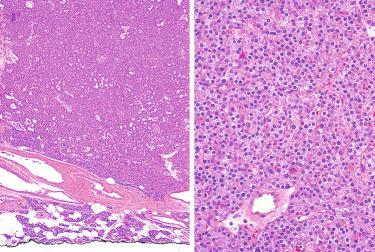
Oncocytic (oxyphilic) adenoma (see Fig. 33-18 ):
Exclusively composed of oncocytic cells with prominent eosinophilic granular cytoplasm
Demographic features are similar to those of the more common adenomas composed of chief cells
Thought to be nonfunctional; usually associated with lesser degree of hypercalcemia; however, several reports document an association with primary hyperparathyroidism
Composed of large cells with abundant eosinophilic granular cytoplasm and hyperchromatic nuclei:
Scattered large atypical nuclei or multinucleated cells may be seen.
Cytoplasm is stuffed with mitochondria on electron microscopy.
An important differential consideration is the frequent presence of nodular oncocytic cell change seen in normal glands with increasing age.
Intrathyroidal localization may suggest a diagnosis of an oncocytic thyroid follicular (so-called Hürthle cell) neoplasm:
Parathyroid oncocytic adenomas have more distinct cell membranes.
Presence of thyroglobulin and TTF1 support lesion of thyroid origin.
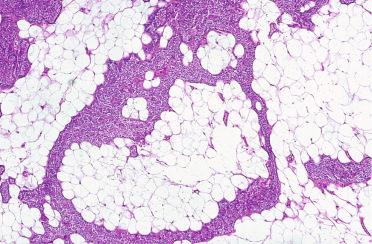
Lipoadenoma (see Fig. 33-19 )
Also referred to as parathyroid hamartoma
Rare benign neoplasm characterized by proliferation of chief and oxyphilic cells forming cords, islands, and follicles admixed with mature adipocytes:
Mature adipocytes make up from 20% to 90% of the neoplasm.
Circumscription and/or encapsulation as well as large size (1 to 15 cm) support adenomatous nature:
May be associated with a compressed rim of “normal” gland
May be difficult to recognize as “abnormal” parathyroid tissue in small biopsies, when they are easily mistaken for normal parathyroid tissue because of the abundance of stromal fat
Stromal fat often contains areas of fibrosis or myxoid alteration.
Most are associated with hyperparathyroidism.
Other rare variants of parathyroid adenoma include:
Papillary variant characterized by prominent papillary architecture:
May not be a “true” variant but papillary architecture, especially in association with fibrosis and hemorrhage (recent and remote), likely is a reactive/degenerative phenomenon
Follicular variant characterized by prominent follicular (acinar) architecture
Water-clear variant characterized by presence of polygonal cells with clear cytoplasm and distinct cell membranes.
Atypical parathyroid adenoma:
Definition: parathyroid tumor showing features worrisome for parathyroid carcinoma but lacking absolute diagnostic features for parathyroid carcinoma
Synonyms include atypical parathyroid neoplasm, parathyroid neoplasm of uncertain biologic significance, or parathyroid neoplasm inconclusive for malignancy
Atypical histologic features suggesting carcinoma but falling short for this diagnosis may include:
Capsular irregularities or invasion without infiltration into adjacent soft tissues
Increased mitotic activity (>5 per 10 high-power fields)
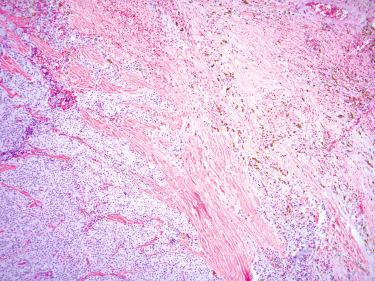
Intralesional fibrosis characterized by broad fibrous bands coursing through the lesion
Coagulative necrosis
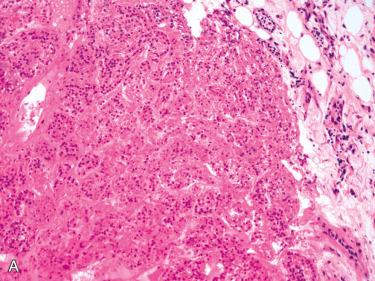
Diffuse cellular atypia
Diffuse sheet-like growth of monotonous cells with increased nuclear-to-cytoplasmic ratio
Macronucleoli in many cells
Atypical parathyroid adenomas lack conclusive features diagnostic for carcinoma including:
Invasion of surrounding soft tissues
Invasion of surrounding structures including thyroid gland, larynx, trachea, pharynx, esophagus, carotid artery, recurrent laryngeal nerves
Angioinvasion
Perineural invasion
Metastasis
Most atypical parathyroid adenomas prove to be benign in long-term follow-up, but owing to the uncertainty in their malignant potential, they have been termed atypical.
Treatment is similar to a typical parathyroid adenoma, but patients should be followed for potential recurrent hyperparathyroidism, local recurrence of tumor, and/or evidence of aggressive behavior (e.g., metastasis).
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here