Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
The first renal neoplasm was reported nearly 200 years ago. In 1953, about eight renal tumors were described in a well-known textbook on surgical pathology. As a consequence of the contributions of numerous investigators over many decades, the most recent World Health Organization (WHO) classification of renal neoplasms includes about 50 well-defined and distinctive renal tumors, as well as various miscellaneous and metastatic tumors. In addition, a number of emerging or provisional new entities are under active investigation and may be included in future classifications. In this chapter, we examine these diverse and fascinating neoplasms.
Excluding cancer of the renal pelvis and ureter, kidney cancer is the seventh most common malignancy worldwide, accounting for 3.3% of all newly diagnosed cancers in 2012. In 2018, nearly 65,340 new cases were reported, with ~ 14,970 related deaths in the United States. At diagnosis, 25% to 30% of patients had metastatic cancer. Highest incidences were in the Czech Republic, Lithuania, Slovakia, and Northern America, and lowest rates were in Middle, Western, and Sub-Saharan Africa. Mortality rates were higher in more developed regions than in less developed regions.
Renal cell carcinoma (RCC) afflicts males approximately twice as often as females, and is most commonly diagnosed in patients in their early sixties. The risk for development of RCC is two to three times higher in individuals who have a first-degree relative with RCC. Approximately 2% to 4% of cases of RCC are associated with several distinctive hereditary cancer syndromes that will be discussed in this chapter under the appropriate headings; the remainder are considered sporadic.
Recognized risk factors for renal cancer include cigarette smoking, obesity, and hypertension. Cigarette smoking has consistently been shown to be a causal risk factor for RCC. The risk increases with consumption level and gradually decreases after years of cessation. There is strong epidemiologic support for a causal association between trichloroethylene exposure and kidney cancer. Overweight and obesity are estimated to contribute to the development of more than 40% of renal cancers in the United States and more than 30% in Europe, possibly mediated by insulin resistance and chronic hyperinsulinemia, increased bioavailability of steroid hormones, and localized inflammation. Although the role of hypertension in the development of RCC is difficult to assess, the bulk of epidemiologic evidence implicates it as a causal risk factor. Long-term renal dialysis exposes patients to a considerably higher average annual incidence of RCC than the general population. The major risk factor in such patients appears to be the presence of acquired cystic kidney disease (ACKD).
Hereditary renal tumors comprise single neoplasms or a constellation of tumor types in more than one first-degree or second-degree family member that are inherited through the passage of germline mutations, each of which is specific for a hereditary renal tumor syndrome. Bilaterality, multifocality, and early age of onset are typical of hereditary renal tumors. It is estimated that between 2% and 4% of renal cell neoplasms arise based on an underlying hereditary abnormality. Well-characterized examples include the following occurrences: clear cell renal cell carcinoma (CCRCC) in von Hippel–Lindau (VHL) syndrome and constitutional chromosome 3 translocations syndrome; papillary renal cell carcinoma (PRCC) in hereditary PRCC syndrome, hereditary leiomyomatosis and renal cell carcinoma (HLRCC) syndrome (non-PRCC1 only), familial papillary thyroid carcinoma syndrome, hyperthyroidism-jaw tumor syndrome, and Birt-Hogg-Dubé syndrome; oncocytoma in hyperthyroidism-jaw tumor syndrome; and chromophobe RCC and hybrid chromophobe oncocytoma in Birt-Hogg-Dubé syndrome. A meta-analysis of reported cases in 1998 failed to establish a link between tuberous sclerosis complex (TSC) and renal cell neoplasms, but more recent reports indicate that the incidence of RCC in patients with TSC is 2% to 4% higher than its incidence in the general population. Although patients with autosomal dominant polycystic kidney disease appear to have a higher incidence of renal cortical adenoma than the general population, there is no convincing evidence that patients with this condition have a higher risk for development of RCC than the general population.
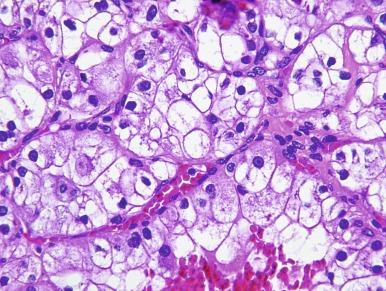
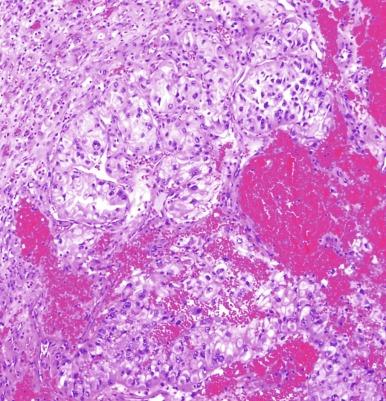
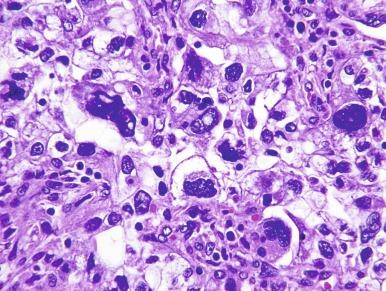
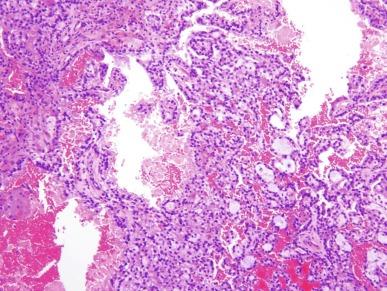
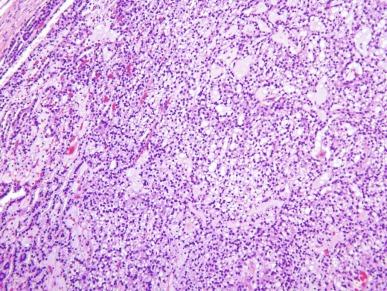
It has been a long-held tenet that cytologic grading is important in predicting the biologic behavior of RCC. Several grading systems proposed in the last 50 years have fallen into disuse because they lacked sufficient validation or applicability to the currently accepted classification of RCC. In an ideal grading system, significant outcome differences should be demonstrable between patients with different tumor grades, both univariate and after adjusting for important clinical and pathologic features. Until recently, the most widely used grading system was the one proposed by Fuhrman et al. The validation, reproducibility, and interpretation of the Fuhrman system drew substantial criticism, and consequently the four-tiered WHO/International Society of Urologic Pathology (ISUP) grading system has been recommended for use in grading CCRCC and PRCC, cancers for which there is sufficient outcome data to allow the system to be used for predicting prognosis. Nucleolar prominence defines grades 1 to 3 ( Fig. 2.1 through 2.3 ). Pronounced nuclear pleomorphism, tumor giant cells, and rhabdoid or sarcomatoid changes are all features of grade 4 tumors ( Fig. 2.4 through 2.6 ). These grades are assigned based on these criteria within the single high-power field (hpf) showing the highest nucleolar grade or greatest degree of nuclear pleomorphism ( Table 2.1 ). The WHO/ISUP grading system is not applicable for predicting outcome for other types of RCC due to a current paucity of outcome data for those tumors.
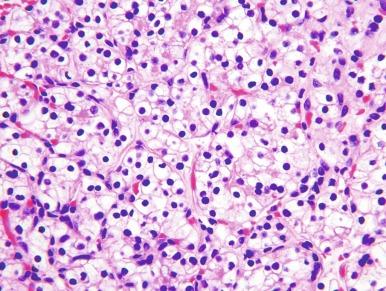
Nucleoli are inconspicuous or absent at 400 × magnification.
Nucleoli are distinctly visible at 400 × magnification, but inconspicuous or invisible at 100 × magnification.
Nucleoli are distinctly visible at 100 × magnification.
Tumor exhibits the presence of tumor giant cells and/or extreme nuclear pleomorphism and/or sarcomatoid differentiation and/or rhabdoid differentiation.
Analysis of the utility of the WHO/ISUP system continues. One group of investigators did not find a statistically significant difference in outcome between patients with ISUP grade 1 or 2 and ISUP grade 3 tumors, perhaps, in part, because their dataset did not include cases of multilocular cystic renal neoplasm of low malignant potential (MCRNLMP) or cases of clear cell PRCC (CCPRCC). In contrast, others have found clear separation of cancer-free survival curves between the four grades of the WHO/ISUP system, and once again found evidence of its superiority over the now-abandoned Fuhrman grading system.
There has been some interest in further refining the accepted WHO/ISUP grading system by amalgamating defined grades with the presence or absence of tumor necrosis (defined as homogeneous clusters and sheets of dead cells, or coalescing groups of cells forming a coagulum, containing nuclear and cytoplasmic debris). Two studies furnished evidence that combining these parameters for cases of CCRCC provides additional prognostic information compared with the use of WHO/ISUP nucleolar grade alone.
Tumor stage reflects the extent of anatomic spread and involvement of disease and is considered to be the most important factor in predicting the clinical behavior and outcome of RCC. Staging systems for RCC have been in use for nearly 50 years and have been continually updated and improved. Since undergoing simplifications and refinements in 1992, the tumor, nodes, and metastasis (TNM) staging system has become the predominant staging system for RCC. In 2017, the eighth edition of the AJCC Cancer Staging Manual was published. Staging parameters for carcinomas arising in the kidney are provided, and definitions for primary tumor (T), regional lymph nodes (N), and distant metastasis (M) are given ( Table 2.2 ). T1 and T2 tumors are localized to the kidney and are defined by their greatest dimensions. T1 tumors are ≤ 7 cm and are subclassified as T1a (≤ 4 cm) or T1b (> 4 cm but ≤ 7 cm). T2 tumors are more than 7 cm and are subclassified as T2a (> 7 cm but ≤ 10 cm) or T2b (> 10 cm). T3 tumors extend beyond the renal parenchyma to involve major veins (renal vein or segmental branches, or vena cava), the pelvicaliceal system ( Fig. 2.7 ), or perinephric tissues, but do not involve the ipsilateral adrenal and do not extend beyond Gerota fascia. T3a tumors conform to this description but do not extend into the vena cava. T3b tumors extend into vena cava below the diaphragm ( Fig. 2.8 ), and T3c tumors extend into vena cava above the diaphragm. T4 tumors invade the ipsilateral adrenal gland directly or invade beyond Gerota fascia ( Fig. 2.9 ). Lymph nodes submitted and uninvolved by tumor define N0; node involvement is staged N1. NX denotes absence of lymph nodes in the surgical specimen. Stage M1 denotes known distant metastases ( Figs. 2.10 and 2.11 ); M0 denotes absence of known distant metastases.
Definition of primary tumor (T)
|
|||
Definition of regional lymph node (N)
|
|||
Definition of distant metastasis (M)
|
|||
| American Joint Committee on Cancer (AJCC) Prognostic Stage Groups | |||
| When T is… | And N is… | And M is… | Stage group is: |
| T1 | N0 | M0 | I |
| T1 | N1 | M0 | III |
| T2 | N0 | M0 | II |
| T2 | N1 | M0 | III |
| T3 | N0 | M0 | III |
| T3 | N1 | M0 | III |
| T4 | Any N | M0 | IV |
| Any T | Any N | M1 | IV |
CCRCC accounts for 60% to 70% of all cases of RCC. It is believed to arise in epithelial cells lining the proximal tubule. Although patients in all age groups are at risk for its occurrence, the great majority develop in patients older than 40 years, and the majority are males, with a ratio of approximately 1.5:1.
More than 90% of cases of CCRCC have characteristic cytogenetic abnormalities that involve loss of genetic material from the short arm of chromosome 3 (3p) and mutations in the VHL gene. The VHL gene, which is located at 3p25–26 and functions as a tumor suppressor gene, was identified through studies of patients with VHL disease, and subsequent investigations implied the presence of one or more additional genes near this site. The VHL gene in CCRCC is typically found to be inactivated by a combination of allelic deletion and either mutation or, less often, hypermethylation. Loss of the VHL protein function contributes to tumor initiation, progression, and metastasis. It is suspected that other genes on 3p may act as tumor suppressors andmay be involved in the development of CCRCC, particularly 3p14.2 deletions, possibly resulting in inactivation of the FHIT gene, as well as another tumor suppressor gene at 3p12. A continuous deletion from 3p14.2–p25, including the FHIT and VHL genes, can be identified in up to 96% of cases. The 3p locus harbors at least four other CCRCC tumor suppressor genes: KD-M6A, KDM5C, SETD2, and PBRM1. After the initiating event involving the 3p gene, additional genetic alterations occur in clonal tumor cell populations as tumor progression occurs and metastatic capability increases. Consequently, these additional genetic abnormalities, when detectable, are often associated with higher histologic grade, higher pathologic stage, and an adverse prognosis. The genetic abnormalities associated with these effects are losses on chromosome 14q, loss of 4p, loss of 9p, and loss-of-function mutations in the BAP1 gene.
There are several familial settings in which CCRCC may arise, the commonest of which is VHL disease. This condition affects 1 in every 36,000 births. It is an autosomal dominant disease associated with the development of retinal angioma, cerebellar and spinal angioma and hemangioblastoma, bilateral multifocal pheochromocytoma, papillary cystadenoma of the epididymis, pancreatic cyst and malignant neuroendocrine tumor, inner ear endolymphatic sac tumor, and renal cyst. In addition, 35% to 45% of affected patients with VHL experience development of bilateral multifocal CCRCC. An average VHL kidney may harbor as many as 1100 cysts and 600 microscopic CCRCCs. Onset of renal carcinoma in patients with VHL is often early; clinically evident renal cancer has been reported in adolescence, and the mean age at diagnosis is 39 years. Historically, without treatment, up to 40% of patients with VHL died of advanced renal carcinoma. In VHL disease, patients are born with a germline defect in one of the two alleles of the VHL gene, located on chromosome 3p925–26, which functions as a tumor suppressor. Loss of the second allele results in clinical disease expression. Additional heritable settings in which CCRCC may develop include Cowden syndrome, Birt-Hogg-Dubé syndrome, families segregating constitutional chromosome 3 translocations, tuberous sclerosis, and families with succinate dehydrogenase B ( SDHB ) deficiency germline mutations.
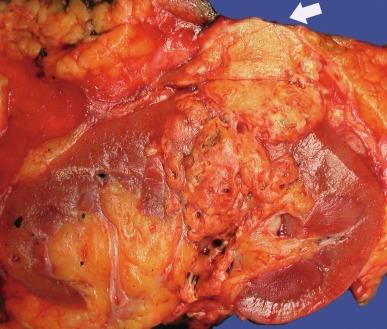
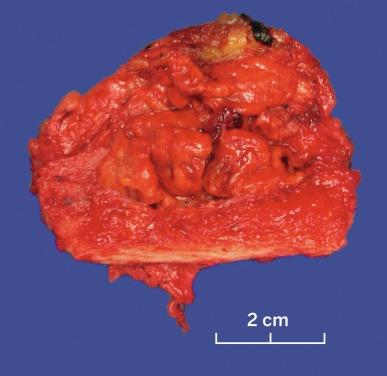
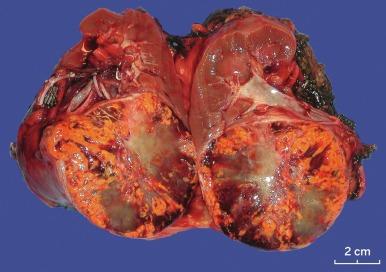
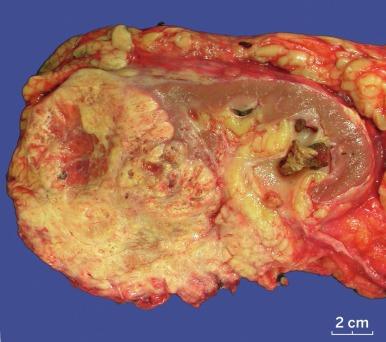
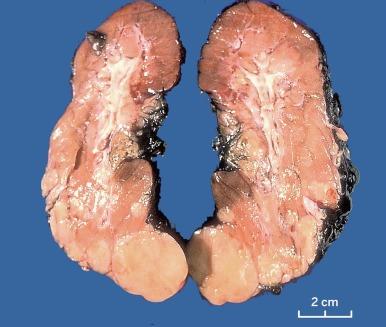
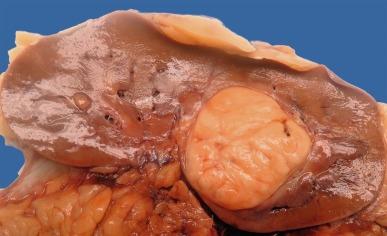
Grossly, CCRCC ranges from a few millimeters to very large, weighing several kilograms; the average size is about 7 cm. It is usually unilateral and unicentric; bilaterality or multicentricity are features of hereditary CCRCC. It commonly forms a bosselated mass that protrudes from the cortical surface ( Fig. 2.12 ). The cut surface is at least in part a characteristic bright golden yellow color, owing to the abundance of cholesterol, phospholipids, and neutral lipids within the tumor cells. The cut surface is typically variegated with areas of gray-white fibrosis and recent or old hemorrhage. It usually has an expansile pushing growth pattern and is well demarcated from the adjacent normal kidney by a fibrous pseudocapsule of varying thickness; in some cases it may appear to infiltrate the adjacent renal parenchyma ( Fig. 2.13 ). Areas of cystic change and calcification are commonly found, particularly within areas of necrosis.
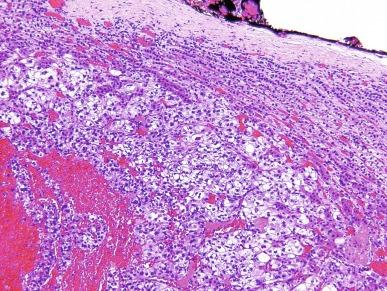
Microscopically, CCRCC displays a variety of architectural patterns. Tumor cells are arranged most often in sheets, compact nests, alveolar, acinar, and microcystic or macrocystic structures, separated by an abundance of thin-walled blood vessels ( Fig. 2.14 ). Tubular structures are variable in size. Microcysts contain extravasated red blood cells or eosinophilic fluid. Infrequently, small papillary structures lined by clear cells may be present focally but account for an insignificant proportion of the overall architecture.
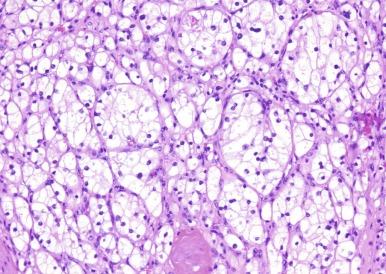
The classic cell of CCRCC has distinct cell membranes and optically clear cytoplasm due to loss of cytoplasmic lipids and glycogen during histologic processing. Some cases of CCRCC display varying numbers of cells with granular eosinophilic cytoplasm; such cells are more often seen in high-grade cancer or near areas of hemorrhage or necrosis. The nuclei of CCRCC show considerable variation in size, shape, and nucleolar prominence, as discussed in the earlier Grading Renal Cell Carcinoma section, and these features are assessed when assigning a nuclear grade to an individual tumor ( Figs. 2.1 through 2.6 ).
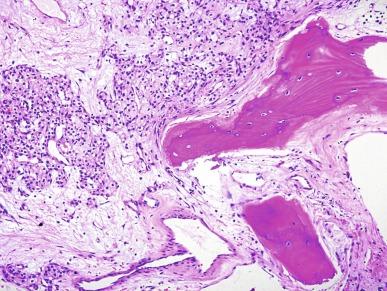
Numerous uncommon histologic variations have been described in CCRCC including heterotopic bone formation ( Fig. 2.15 ), intracellular and extracellular hyaline globules, basophilic cytoplasmic inclusions, abundant multinucleated giant cells, sarcoid-like granulomas, myospherulosis, and lymphomatoid features. The significance of these rare findings is unknown.
Immunohistochemically, CCRCC typically shows positive immunostaining for vimentin, CAM5.2, AE1/AE3, RCC-Ma, CD10, PAX2, PAX8, and carbonic anhydrase-IX (CA-IX) ( Table 2.3 ). Immunostains for 34E1β2, cytokeratin 7 (CK7), CK20, mucin 1 (MUC1), parvalbumin, α-methylacyl–coenzyme A racemase (AMACR), kidney-specific cadherin, and CD117 are negative in most cases.
| PAX8 | CD10 | CA-IX | RCC-Ma | Melanocytic markers a | Vimentin | CK7 | CK20 | P63 | HMWCK | CD117 (c-kit) | AMACR | GATA3 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Clear cell RCC | + | + | + | + | - | + | - | - | - | - | - | -/+ | - |
| Papillary RCC | + | + | +/- | + | - | + | + b | - | - | - | - | + | - |
| Clear cell papillary RCC | + | - c | + (cuplike) | +/- | - | + | + | - | - | +/- | - | - | - |
| Chromophobe RCC | + | -/+ | - | +/- | - | - | + | - | - | - | + | - | +/- |
| Oncocytoma | + | -/+ | - | - | - | - | Focal d | - | - | - | + | -/+ | - |
| Angiomyolipoma | - | - | -/+ | - | + | -/+ | - | - | - | - | - | - | - |
| ESC RCC | + | -/+ | -/+ | NA | - | + | -/+ | + | - | - | - | -/+ | - |
| Collecting duct carcinoma | + | - | -/+ | - | - | + | +/- | - | -/+ | + | - | - | - |
| Tubulocystic carcinoma | + | + | +/- | + | - | +/- | -/+ | - | - | - | - | +/- | - |
| Translocation RCC | + | +/- | -/+ | +/- | +/- | + | - | - | - | - | - | +/- | - |
| MTSCC | + | -/+ | -/+ | +/- | - | + | + | - | - | -/+ | - | +/- | - |
| UCUUT | -/+ e | -/+ | -/+ | - | - | +/- | + | + | + | + | -/+ | -/+ | + |
a Melanocytic makers include HMB45, Melan-A/Mart-1, tyrosinase A, and microphthalmia-associated transcription factor ( MITF ).
b CK7 may be negative in type II papillary RCC.
c CD10 can be focally positive in the cyst lining cells of clear cell papillary RCC.
d In contrast with diffuse and strong immunoreactivity for CK7 in chromophobe renal cell carcinoma, oncocytoma typically shows scattered focal positivity for CK7(< 5% of tumor cells staining).
e Approximately 20% to 30% of UCUUTs showed positive staining for PAX8.
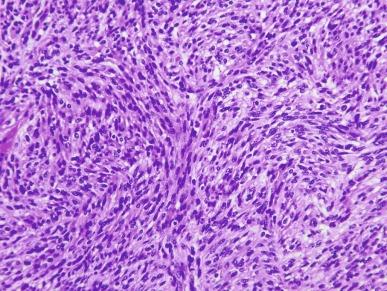
Pathologic stage most accurately predicts the prognosis for patients with CCRCC. Prognosis for tumors within the same stage is further definable by tumor grade, the presence or absence of tumor necrosis, and the presence or absence of sarcomatoid or rhabdoid differentiation. Grade and tumor necrosis were discussed in the earlier section Grading Renal Cell Carcinoma. Progressively higher tumor grades are associated with progressively worsening prognosis. Tumor necrosis accounting for more than 10% of the total tumor volume is associated with a less favorable outcome. Sarcomatoid differentiation is seen in 5% to 8% of cases of CCRCC. Rhabdoid differentiation is seen in 3% to 7% of RCC overall and is most frequently associated with CCRCC. The presence of either sarcomatoid or rhabdoid differentiation worsens the prognosis. Patients whose tumors exhibit sarcomatoid change have a 5-year survival rate of 15% to 22%; for those whose tumors exhibit rhabdoid differentiation, the median survival is 8 to 31 months.
MCRNLMP is an uncommon renal tumor that was described as an entity distinguishable from conventional CCRCC in 1991. By definition it has a fibrous pseudocapsule, and is composed entirely of cysts and septa with no expansile solid nodules; the septa should contain aggregates of epithelial cells with clear cytoplasm that are indistinguishable from the tumor cells that compose grade 1 RCC. It accounts for less than 1% of all renal tumors and has a male-to-female ratio of approximately 2:1. Age range is 20 to 76 years; most patients are in their fifties or sixties, and females tend to present at a younger age than males.
Although most patients are asymptomatic, and their tumors are discovered incidentally, a minority have either a palpable mass, gross hematuria, abdominal or back discomfort, or rarely, systemic symptoms. Laboratory findings may include anemia and microhematuria, but most patients have no laboratory abnormalities. Imaging studies usually reveal a complex cystic mass that may have focal calcification.
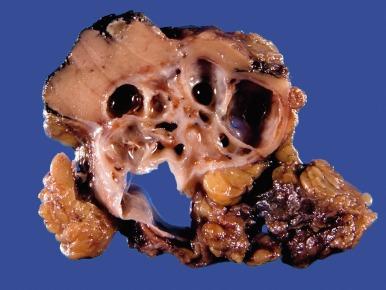
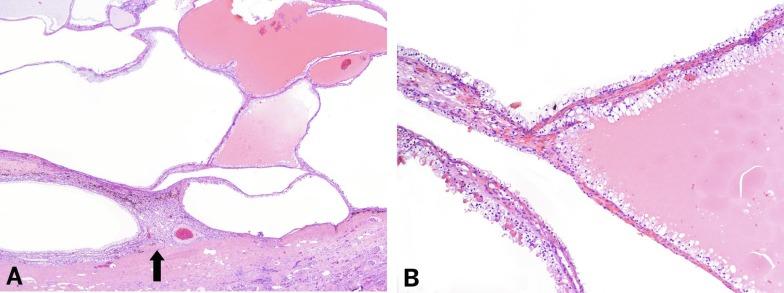
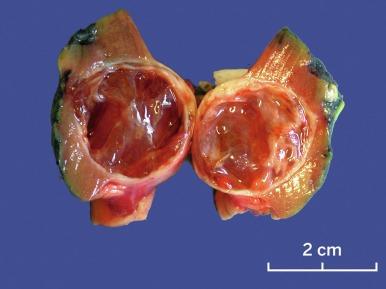
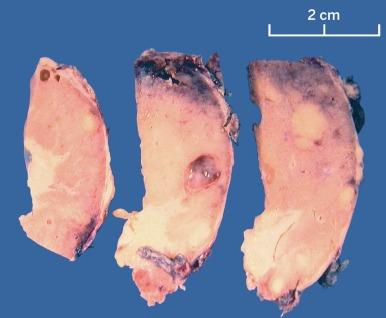
Grossly, MCRNLMP ranges from 0.5 to 13.0 cm in greatest dimension. It is typically a unilateral and solitary well-circumscribed mass composed entirely of cysts, separated from adjacent renal parenchyma by a fibrous wall; however, it can be multifocal, as well as bilateral ( Fig. 2.16 ). The cysts are of variable size and contain clear or hemorrhagic fluid. The septa between cysts are thin. Necrosis is absent, and there are no grossly visible nodules expanding the septa, a feature that differentiates this tumor from extensively cystic CCRCC.
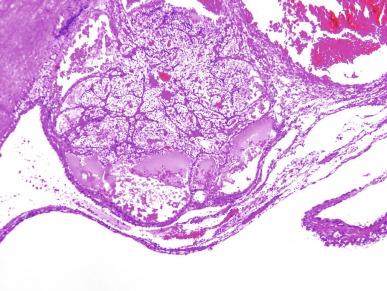
Microscopically, the cysts are lined by epithelial cells, usually as a single layer, but occasionally multilayered, and occasionally forming minute papillary structures; some cysts lack any lining cells ( Fig. 2.17 ). The lining cells have variable amounts of cytoplasm that may be clear or lightly eosinophilic; however, the cytologic characteristics of the lining epithelial cells do not constitute part of the criteria for defining MCRNLMP, because renal cystic lesions of many types may sometimes be lined by epithelium with clear cells. The septa consist of fibrous tissue. More than 20% of tumors show calcifications within the septa, and metaplastic bone formation is sometimes observed. Within the septa in all cases there are clusters of epithelial cells with clear cytoplasm ( Fig. 2.18 ). These cells resemble the cyst lining cells, usually have small, dark nuclei, and do not form expansile nodules; this is a critical feature that distinguishes MCRNLMP from extensively cystic CCRCC ( Figs. 2.19 and 2.20 ). They are often difficult to distinguish from histiocytes or from lymphocytes with surrounding retraction artifact. In difficult cases, the epithelial nature of the tumor cell clusters can be confirmed by their immunoreactivity to antibodies against CK and epithelial membrane antigen (EMA); immunostains for histiocytic markers are negative.
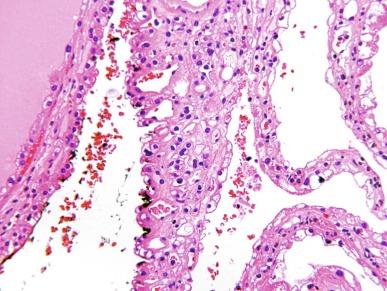
Ploidy studies reveal diploid DNA content in more than 90% of cases. Electron microscopy in one case revealed apical microvilli and other ultrastructural features in the tumor cells that were similar to those seen in conventional CCRCC of the kidney. VHL gene mutations are identifiable in about 25% in MCRNLMP, and there is no difference in the status of chromosome 3p deletion between low-grade CCRCC and MCRNLMP, supporting the concept that MCRNLMP is a subtype of CCRCC. Tumor cells of MCRNLMP are strongly immunoreactive for PAX8 and CA-IX.
Only rare cases have shown extension beyond the kidney into the perirenal fat. The proper diagnosis hinges on strict adherence to the diagnostic criteria for this entity. There are no reports of recurrence or metastasis of MCRNLMP. Surgical resection is indicated to exclude extensively cystic RCC.
PRCC, the second most common type of RCC, accounts for up to 18.5% of all renal epithelial neoplasms. Most cases occur sporadically, but some develop in members of families with hereditary PRCC, an inherited renal cancer characterized by mutations in the MET oncogene at 7q31 and by a predisposition to development of multiple bilateral papillary renal tumors.
The mean age of patients with PRCC ranges from 52 to 66 years, and males are affected slightly more than twice as often as females (2.4:1 male-to-female ratio). Currently only 5% to 10% of patients diagnosed with PRCC present with flank pain, hematuria, or abdominal mass; discovery is predominantly incidental by various radiologic studies. About 30% of PRCC contain foci of calcification; furthermore, PRCC tends to appear hypovascular on radiologic studies because of spontaneous ischemic necrosis. PRCC is also more likely to be multifocal (8% of cases) and necrotic (46%) than other common RCC subtypes, as well as being overrepresented in end-stage renal disease. When multiple papillary tumors are found in patients without a family history of renal tumors, each papillary tumor arises independently; multifocality is not the result of intrarenal metastasis. In patients with hereditary papillary renal cancer, it has been estimated that each kidney may harbor as many as 3400 separate microscopic papillary tumors.
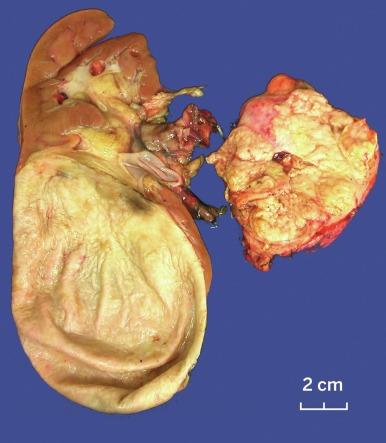
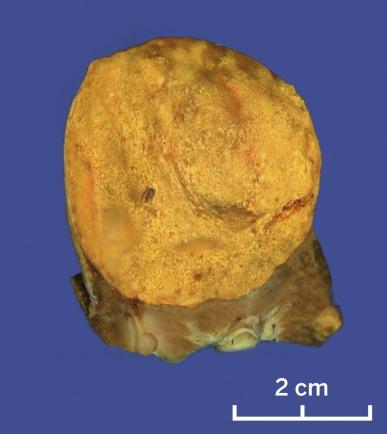
Grossly, PRCC is typically well circumscribed, and up to 90% of cases are confined to the renal parenchyma. Multifocality may be grossly evident, particularly in cases of hereditary PRCC ( Figs. 2.21 and 2.22 ). A thick, fibrous pseudocapsule is present in up to two-thirds of cases ( Fig. 2.23 ). The degree of pseudocapsule formation often parallels the extent of hemorrhage and necrosis in the tumor. The cut surface varies from light gray-tan to golden yellow to red-brown, depending on the preponderance of macrophages in the stroma and the degree of remote hemorrhage and hemosiderin accumulation ( Figs. 2.24 and 2.25 ).
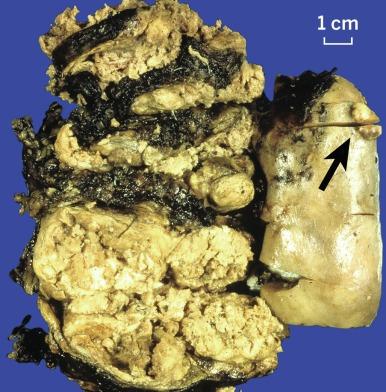
Microscopically, PRCC is composed of varying proportions of papillary and tubular structures and, in some instances, contains cysts with papillary excrescences or with tumor infiltrating the cyst wall ( Fig. 2.26 ). Papillae are lined by a single layer of tumor cells that may sometimes appear pseudostratified. The stalks of the papillary structures contain fibrovascular cores and are commonly infiltrated by variable numbers of macrophages; the prevalence of macrophages has no apparent correlation with the extent of accompanying hemorrhage or necrosis. The architectural arrangement of the papillary stalks varies. They may form exquisite and readily recognizable papillae. Compact and tight packing in some tumors results in a solid appearance ( Fig. 2.27 ). In some tumors, the papillae are arranged in long parallel arrays, creating a trabecular appearance ( Fig. 2.28 ).
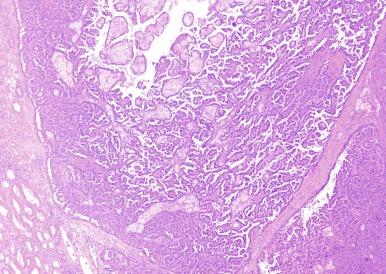
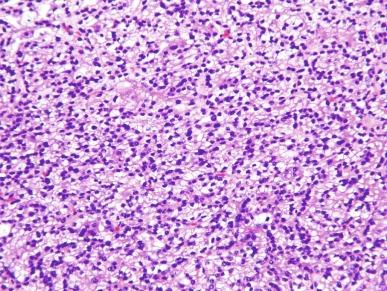
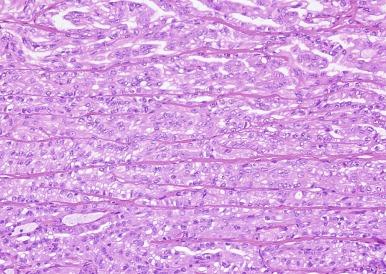
Infrequently, PRCC exhibits solid areas composed of spindle cells with low-grade nuclear features, admixed with variable amounts of tumor with tubular or papillary architecture. In one series of such cases, all were confirmed to be PRCC by molecular studies. It is important to distinguish these tumors from mucinous tubular and spindle cell carcinoma, and to recognize that the low-grade spindle cell proliferations do not represent sarcomatoid change.
For more than two decades, morphologic heterogeneity in histologic grade and cytoplasmic quality (eosinophilic or basophilic) has been recognized in PRCC. Based on studies of morphology, immunohistochemical staining, growth kinetics, clinicopathologic staging parameters, and patient survival data, it was proposed that PRCC could be subclassified into two morphologic variants, which have been designated types 1 and 2 (PRCC1 and PRCC2, respectively). Notably, both types exhibit tumor-associated acute and chronic inflammation, extensive necrosis, evidence of recent hemorrhage, psammoma bodies, cholesterol clefts, foreign-body-type giant cells, and areas of dystrophic calcification. However, beyond that, there are a number of substantial differences.
PRCC1 is composed of papillae covered by a single or double layer of small epithelial cells of low nuclear grade, bearing small round to ovoid nuclei with inconspicuous nucleoli, and possessing minimal pale or clear cytoplasm ( Fig. 2.29 ). Tubular structures in these neoplasms have similar lining cells. The papillae of PRCC1 are usually thin, delicate, and often short, and frequently show expansion by edema fluid. The short complex papillae may impart a glomeruloid appearance. Aggregates of foamy macrophages are commonly noted within the papillary cores or within sheets of tumor cells. PRCC1 tumor morphology is characteristic of papillary neoplasms that occur in hereditary PRCC ( Fig. 2.30 ).
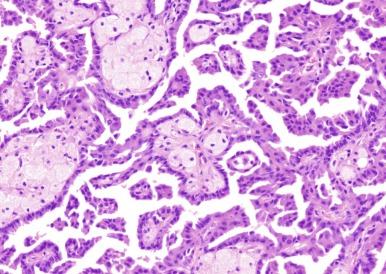
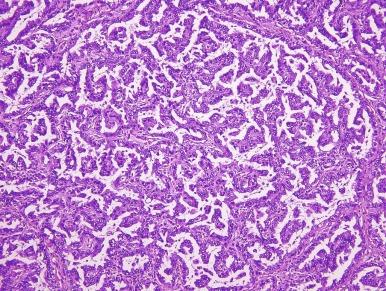
PRCC2 tends to be larger than PRCC1 (mean diameters, 6 cm for PRCC1 and 9.5 cm for PRCC2) and is of significantly higher nucleolar grade. Tumor cells in PRCC2 exhibit large and spherical nuclei, prominent nucleoli, and varying degrees of nuclear pseudostratification. The cytoplasm is abundant and typically eosinophilic ( Fig. 2.31 ). The fibrovascular cores of the majority of PRCC2 tumors tend to be dense and fibrous rather than thin and delicate, and edema and glomeruloid bodies are less prevalent than in PRCC1 tumors. Macrophages, rather than populating the papillae, are more likely to be found near areas of necrosis.
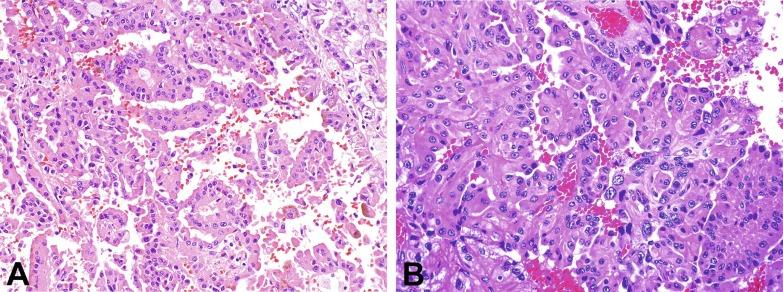
The histologic distinctions noted earlier have been augmented by molecular analysis. PRCC1 exhibits loss of Y chromosome and gains in chromosomes 7, 17, 16, and 20. Activation of the MET pathway is a feature of up to 80% of PRCC1 tumors. PRCC2 tumors exhibit a heterogeneous array of chromosomal gains and losses, involving chromosomes 1, 3, 4, 5, 6, 8q, 9, 14, and 15.
PRCC2 tumors are more often associated with aggressive clinicopathologic parameters than PRCC1 tumors, including higher TNM stage, larger tumor size, and an overall worse prognosis. The worsened prognosis for PRCC2 tumors was evident in some studies on univariate analysis, but in other studies the statistical significance of specific tumor subtype was not maintained on multivariate survival analysis when adjusting for grade and stage.
Immunohistochemically, PRCCs often expresses AE1/AE3, CK7, CAM5.2, EMA, 34βE12, AMACR, CA-IX, RCC marker (RCC-Ma), CD10, and vimentin ( Table 2.3 ). PRCC1 tumors are more likely than PRCC2 to express CK7 ( Fig. 2.32 ).

The practicality of subtyping PRCC is complicated by the fact that a clearcut distinction may be difficult in some cases. In various studies, the proportion of PRCC1 tumors varies between 32% and 70%, and conversely the percentage of PRCC2 tumors ranges from 30% to 68%. Specifically some papillary tumors exhibit nuclear features typical of PRCC1 but cytoplasmic features typical of PRCC2, and some are composed of mixtures of cells of generally low nuclear grade but with substantial variations in cytoplasmic characteristics. In short, it seems clear that a number of PRCC cases do not meet the well-accepted histologic criteria for either PRCC1 or PRCC2, and such tumors have been variously designated as mixed, unclassified, overlapping, or not otherwise specified. Strict histologic criteria for assignment to the PRCC, not otherwise specified, category have been developed, indicating that 45% to 47% of PRCCs fall into this category. Furthermore, an oncocytic low-grade variant of PRCC has been identified, composed predominantly of tumor cells with oncocytic cytoplasm and round, nonoverlapping low-grade nuclei with inconspicuous nucleoli, and a linear arrangement toward cell apices ( Fig. 2.33 ). These tumors resemble PRCC1 molecularly, with similar gains of chromosomes 7 and 17, and clinically, exhibiting indolent behavior, and they have a good prognosis.
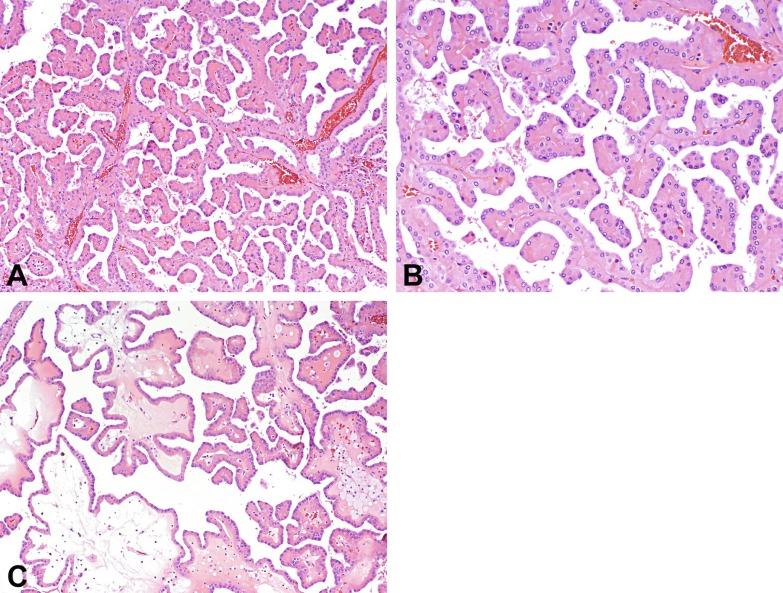
Alternative classifications and suggested classifications of PRCC have been published recently. Based on comprehensive molecular characterization of 161 PRCCs, the Cancer Genome Atlas (TCGA) Research Network showed that PRCC1 and PRCC2 are different types of renal cancer. PRCC1 is associated with MET alterations. Multiple molecular clusters were described within the PRCC2 group. PRCC2 tumors were characterized by CDKN2A silencing, SETD2 mutations, transcription factor E3 (TFE3) fusions, and increased expression of the NRF2-antioxidant response element pathway. A CpG island methylator phenotype was observed in a distinct subgroup of PRCC2 that was characterized by poor survival and mutation of the gene-encoding fumarate hydratase (FH). Disregarding the fact that the TCGA study included tumors that were not conventional PRCCs (specifically translocation carcinomas and FH-deficient carcinomas were included in the study and the report), it seems clear that PRCC2, rather than being homogeneous, represents a heterogeneous group of tumors that requires further study.
A recent study using several immunohistochemical stains (CA9, GATA3, and ABCC2), microRNA (miRNA) expression, and copy number variation analysis provided evidence in support of subclassifying PRCC into four subtypes, designated PRCC1, PRCC2, and two new subtypes, PRCC3 and PRCC4/OLG (the latter representing the oncocytic low-grade variant of PRCC noted earlier). PRCC1, PRCC2, and PRCC3 each have unique staining signatures. The PRCC subtypes have different clinical characteristics, justifying their proper stratification.
Compared with other common types of RCC, the cancer-specific survival rates at 5 years after surgery for a large series of patients with CCRCC, PRCC, and chromophobe RCC were 72%, 91%, and 88%, respectively. PRCC has a more favorable outcome than collecting duct carcinoma and HLRCC-RCC. Patients with PRCC1 tumors have significantly longer survival than those with PRCC2, but the difference is better related to the stage and grade at presentation than the actual tumor type. ISUP nucleolar grading has been validated for use in pathologic descriptions of PRCC. Sarcomatoid and rhabdoid change are included in ISUP grading as grade 4; grade 4 tumors are associated with decreased survival.
Germline mutations of the FH gene at chromosome 1q42 is the underlying genetic abnormality in an inherited autosomal dominant disorder known as HLRCC syndrome. Biallelic inactivation of the gene in FH-mutated cells results in accumulation of fumarate, which reacts spontaneously, via protein succination, with the cysteine sulfhydryl group of proteins to form S -2(2-succino)-cysteine (2SC). These accumulations modulate the activity of various transcription factors, notably HIF1A, NRF2, and AMPK, resulting in increased proliferation and resistance to apoptosis.
Cutaneous leiomyomas occur in 76% of family members overall ( Fig. 2.34 ), and uterine leiomyomas arise in 100% of female members. In addition, about 20% to 30% of family members develop a distinctive type of aggressive RCC that is already metastatic in 50% of cases when first diagnosed.
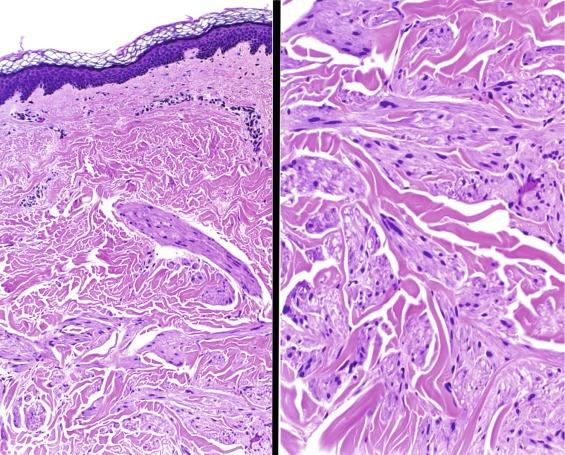
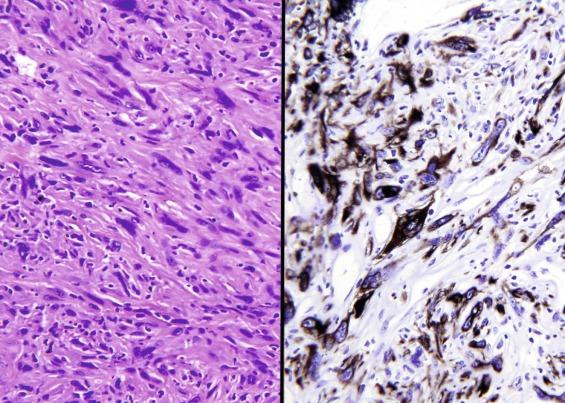
Renal tumors are, with rare exceptions, unilateral and solitary, rather than multiple and bilateral, as observed in other familial RCC syndromes, and typically there is little in the history of patients with these seemingly sporadic tumors to suggest a familial tendency. Patients with HLRCC renal tumors tend to present at a relatively young age (mean, 36 years), but 25% of patients are older than 50 years at diagnosis. Tumors range in size from 2.3 to 20 cm. Some are cortical, some are medullary, and some involve both regions. The degree of tumor circumscription is widely variable. Cystic or multicystic change is noted infrequently. Histologically, tumors exhibit a wide range of architectural patterns. Intracystic papillary, tubulopapillary, and tubulocystic patterns are most prevalent and characteristic of FH-deficient RCC. Other architectural patterns include papillary, tubular, solid, and cystic elements; collecting duct carcinoma-like areas with infiltrating tubules, nests, and single cells surrounded by desmoplastic stroma; and sarcomatoid change. The morphologic hallmark of these tumors is the presence of a characteristic large nucleus with a very prominent inclusion-like orangiophilic or eosinophilic nucleolus, surrounded by a clear halo ( Fig. 2.35 ). However, this feature is often not uniformly present throughout the tumor.
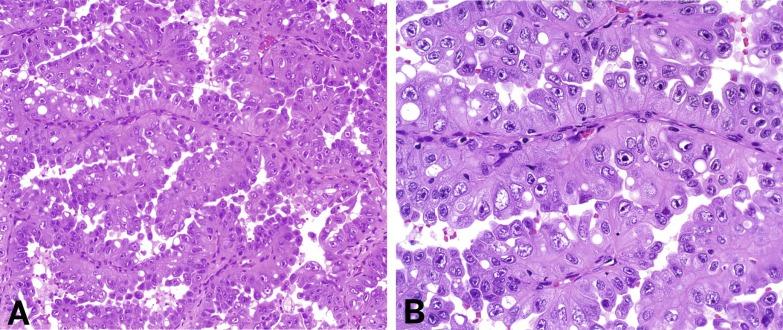
An early study of 2SC immunostaining detected positivity for 2SC in various HLRCC-related tumors, but not in normal tissues or a variety of non-HLRCC-related tumors, and predicted genetic alterations of the FH gene in patients referred for genetic testing. Subsequently, it was found that all confirmed HLRCC tumors demonstrate diffuse and strong nuclear and cytoplasmic 2SC immunostaining, a finding not present in a wide range of other renal carcinoma types. In addition, positive nuclear and cytoplasmic 2SC staining correlated well with the presence of FH germline mutation. Somatic inactivation of the remaining FH allele is found in HLRCC renal cancers, consistent with the role of FH as a tumor-suppressor gene. Its loss of function in the tumor cells corresponded to strong 2SC staining, whereas adjacent renal parenchyma maintained a wild-type allele and did not show 2SC immunoreactivity.
After reports of loss of FH immunostaining in HLRCC-associated tumors, a study was conducted to ascertain the status of 2SC and FH immunostaining in many high-grade unclassified RCC, unclassified RCC with papillary pattern, and PRCC2. One-fifth of renal cancers initially diagnosed as “unclassified RCC, high grade” or “unclassified RCC with papillary pattern” were found to have deficient FH immunostaining, almost invariably accompanied by FH mutations, as well as strong 2SC immunoreactivity. A small minority (0.5%) of tumors previously diagnosed as PRCC and 3% of tumors previously diagnosed as PRCC2 showed FH deficiency by immunostaining and FH mutations. Furthermore, the FH-deficient RCC exhibited considerable clinicopathologic similarities with well-documented cases of HLRCC-associated RCC. Further testing confirmed an association with HLRCC syndrome in about one-third of patients with FH-deficient RCC in this study. Evaluation of both FH and 2SC immunostaining status appears to be warranted in cases exhibiting the morphologic and clinical features noted earlier.
As noted later in the Tubulocystic Renal Cell Carcinoma section, about 50% of poorly differentiated renal carcinomas with areas resembling classic tubulocystic carcinoma exhibit loss of FH immunoreactivity and positive 2SC immunostaining. The term FH-deficient RCC has been proposed provisionally for tumors with a combination of suggestive morphology and immunophenotype but where genetic confirmation is unavailable at the time of diagnosis.
HLRCC-RCC is a lethal disease with rapid aggressive growth and early metastasis even when the tumor is small. The majority of patients present with advanced stage disease and succumb to their cancers. Consequently, recognition of the characteristic features of this tumor are important not only in managing the affected patient but in counseling and follow-up of family members. Aggressive excision of even small tumors in syndromic patients is warranted.
Chromophobe cells were first described in chemically induced renal tumors in rats. Chromophobe RCC was first reported in 1985; its name was derived from the morphologic similarity between the predominant tumor cells in the human tumor to those comprising the experimentally produced rat kidney tumor. Subsequently, the eosinophilic variant was described in 1988.
Genomic studies indicate that chromophobe RCC arises from cells of the distal nephron. Most cases arise sporadically. Some are associated with an folliculin (FLCN) germline mutation in the autosomal dominant cancer predisposition Birt-Hogg-Dubé syndrome, characterized by hair follicle fibrofolliculomas (mainly on the face and neck) and a predisposition to development of bilateral multifocal renal tumors, as well as pulmonary cysts, that can give rise to spontaneous pneumothorax. Chromophobe RCC comprises approximately 34% of the renal neoplasms that occur in patients with this syndrome; the remaining neoplasms include oncocytoma (7%), CCRCC (9%), and tumors with features that resemble a mixture of oncocytoma and chromophobe carcinoma (50%). Another hereditary predisposition to development of chromophobe RCC results from a germline mutation of PTEN in Cowden syndrome.
Chromophobe RCC accounts for 4.9% of surgically excised renal epithelial neoplasms. It comprises a higher proportion of renal cancers in the Middle East. Patients range in age from childhood to extreme old age, with a slight male preponderance. The majority of cases are found incidentally. Symptomatic patients typically report flank pain or gross hematuria; rarely a mass is palpable, or weight loss has occurred. Paraneoplastic symptoms are generally absent. Radiologically, there are no features that reliably distinguish chromophobe RCC from other renal epithelial neoplasms (see later Oncocytoma section with respect to [99m] Tc-sestamibi single-photon emission computed tomography/computed tomography [SPECT/CT] scanning).
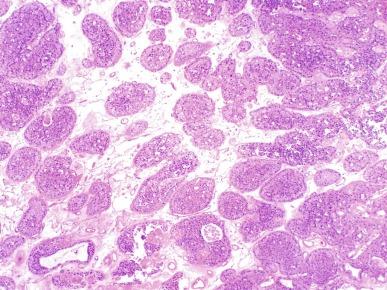
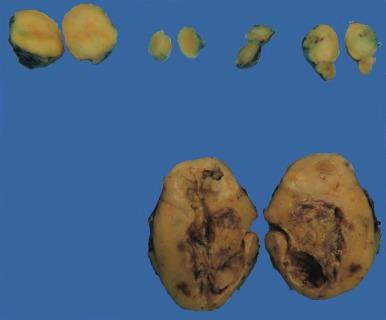
Sporadic chromophobe RCC is typically solitary, with rare exceptions. Tumors are well circumscribed and vary widely in size, from 1.5 to 25 cm in diameter; mean diameter ranges from 6.9 to 8.5 cm. The cut surface is usually solid, homogeneous, and light brown or beige; less often the tumor is mahogany brown, gray-tan, light pink, or yellow-white ( Figs. 2.36 and 2.37 ). A minority show hemorrhage or necrosis, and these features, when present, are limited in extent. An area of central scarring is infrequently present.
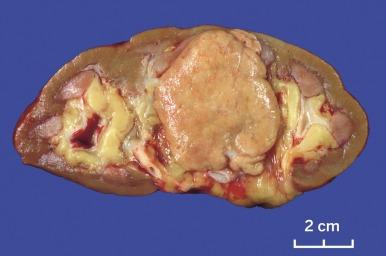
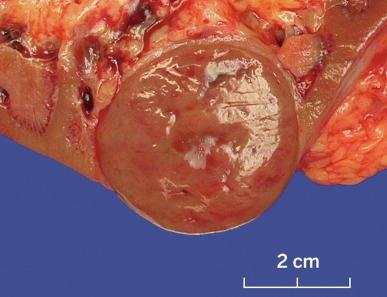
Microscopically, the tumor cells of chromophobe RCC are typically arranged in solid sheets; in some cases, areas of tubulocystic architecture are present. Tumors are intersected randomly by delicate to broad fibrous septa and blood vessels that are predominantly of medium caliber, in contrast with the small sinusoidal vessels that are seen in CCRCC ( Fig. 2.38 ).
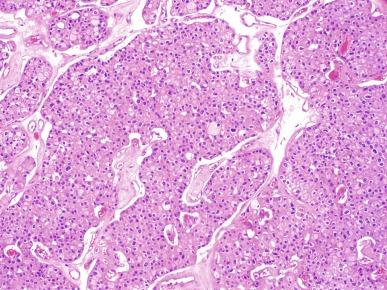
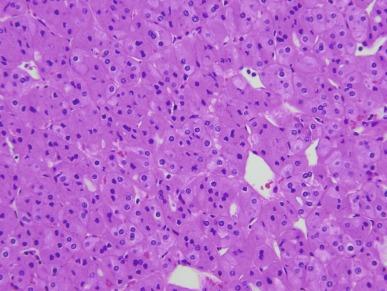
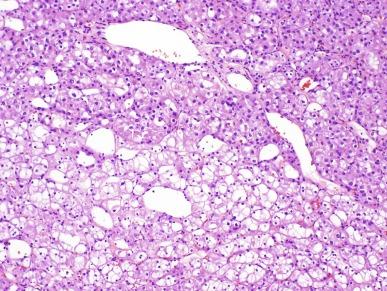
Two types of tumor cells may be present in varying proportions. One type, the chromophobe cell, is a large polygonal cell with abundant, almost transparent and slightly flocculent cytoplasm and prominent, often “plantlike” cell membranes ( Fig. 2.39 ). The chromophobe cells are frequently found adjacent to vascular channels. Usually they are admixed with a second population of smaller cells with less abundant cytoplasm that is granular and eosinophilic ( Fig. 2.40 ). This combination of cell types characterizes the “typical” variant of chromophobe RCC. The nuclei of both cell types are typically hyperchromatic with irregular wrinkled nuclear contours; binucleation is common. Perinuclear halos are frequently present in the more eosinophilic cells, and this feature can be of considerable diagnostic importance.
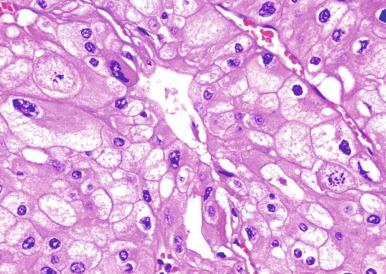
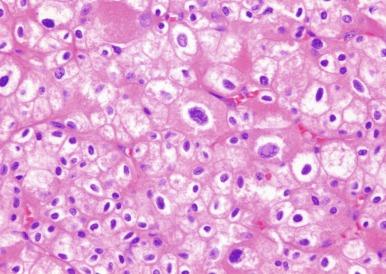
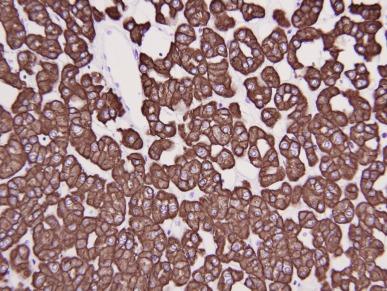
A variant of chromophobe RCC that is virtually entirely composed of intensively eosinophilic cells with prominent cell membranes has been designated as the eosinophilic variant of this neoplasm ( Figs. 2.41 and 2.42 ). In some cases of this variant the problem of making a distinction from oncocytoma is compounded by areas of tumor architecture that entirely mimic that entity ( Fig. 2.43 ). It may be difficult to distinguish chromophobe RCC, especially the so-called eosinophilic variant of chromophobe RCC, from oncocytoma. Strong diffusely positive tumor cell immunoreactivity for CK7 is supportive of a diagnosis of eosinophilic variant of chromophobe RCC (see later Oncocytoma section) ( Fig. 2.44 ). Hale’s colloidal iron stainings are positive in chromophobe RCC and negative in oncocytoma.
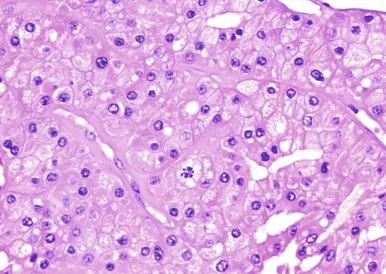
Sarcomatoid transformation, a feature common to other types of RCC, may be found in up to 9% of cases, and in certain geographic locations, it may be unusually prevalent ( Fig. 2.45 ). Rare cases with osteosarcoma-like differentiation, rhabdoid differentiation, or extensive calcification and ossification have been reported. The ISUP has recommended that chromophobe RCC should not be graded.
Some renal neoplasms exhibit mixed morphologies, with elements suggestive of oncocytoma and chromophobe RCC in the same tumor, and have been referred to as hybrid oncocytic/chromophobe tumors. Such tumors arise in several distinct settings: as tumors present in cases of renal oncocytosis, as tumors arising in patients with Birt-Hogg-Dubé syndrome ( Fig. 2.46 ), and as sporadic tumors. Descriptions of hybrid oncocytic/chromophobe tumors vary somewhat with the setting in which they arise, but a common theme is the admixture of tumor cells resembling typical oncocytoma with tumor cells resembling chromophobe RCC, either gradually transitioning from one morphology to another, or as fairly distinctly separate tumor cell clusters adjacent to one another, or intimately admixed with one another ( Fig. 2.47 ). Remarkably, despite somewhat overlapping morphologies, tumors from all three groups have different molecular genetic makeups, and their molecular signatures are different from those of oncocytoma and chromophobe RCC. The precise nature of these tumors awaits further elucidation. Available data on follow-up of patients with hybrid oncocytic/chromophobe tumors indicate that tumors in all three groups exhibit indolent behavior.
Ultrastructurally, chromophobe cells contain numerous round, ovoid, or elongate microvesicles ranging from 150 to 300 nm in diameter, scant numbers of small mitochondria with tubulovesicular cristae, and rare glycogen particles. The nature and origin of the microvesicles are unknown; it is postulated that they are derived from mitochondria. Eosinophilic cells have fewer microvesicles and more abundant mitochondria with tubulovesicular or lamellar cristae.
The genetic abnormality most consistently observed in chromophobe RCC has been loss of one copy of the entire chromosome for most or all of the chromosomes 1, 2, 6, 10, 13, 17, and 21 (in 86% of cases), as well as losses of various other chromosomes (at frequencies varying from 12% to 58% of cases). Chromosomal losses result in a hypodiploid DNA index. Sarcomatoid chromophobe RCC frequently has multiple gains (polysomy) of chromosomes 1, 2, 6, 10, and 17; distant metastases in cases of metastatic chromophobe RCC show the same genetic patterns, usually chromosomal losses (monosomy), as are found in the primary tumors. About one-half of cases of the eosinophilic variant of chromophobe RCC do not share the same chromosomal abnormalities as the classic type.
Chromophobe RCC virtually always shows positive immunohistochemical staining for PAX8, CD117 (ckit), pancytokeratin, EMA, and parvalbumin, and negative immunostaining for vimentin, CK20, and AMACR ( Table 2.3 ). Hale colloidal iron stainings are positive in chromophobe RCC. The use of immunostaining in distinguishing chromophobe RCC from oncocytoma is discussed in the Oncocytoma section. The most useful staining is CK7. CK7 are typically diffusely positive in chromophobe RCC, in contrast with oncocytomas, which usually show focal scattered stainings (< 5% of tumor areas). Oncocytoma is negative for Hale colloidal iron stainings.
Genetic analysis allows separation of these entities based on the finding that oncocytomas do not demonstrate combined losses of heterozygosity at chromosomes 1, 2, 6, 10, 13, 17, and 21, characteristic of chromophobe RCC. Fluorescence in situ hybridization (FISH) studies indicate that detection of losses of chromosomes 2, 6, 10, or 17 effectively excludes the diagnosis of oncocytoma and supports the diagnosis of chromophobe RCC.
A more recent study demonstrated that approximately 70% of chromophobe RCCs carry either a hemizygous deletion of RB1 or ERBB4, and no oncocytomas showed the deletions of these genes. The combined use of RB1 and ERBB4 FISH to detect deletion of these genes may offer a highly sensitive and specific assay to distinguish chromophobe RCC from oncocytoma.
The prognosis for chromophobe RCC has been shown in a number of large series to be significantly better than for CCRCC, and there is no significant difference in outcome between the typical and the eosinophilic variants of chromophobe RCC. Stage at presentation with chromophobe RCC is usually significantly lower than with CCRCC. Only 4% of patients with chromophobe RCC present with metastases, compared with 27% of patients with CCRCC. The cancer-specific survival rate for chromophobe RCC at 5 years after nephrectomy is about 90%. Adverse risk factors for survival include histologic tumor necrosis and sarcomatoid change. Metastatic chromophobe RCCs have been found to be enriched for TP53 mutations, PTEN mutations, and imbalanced chromosome duplications (duplication of three or more chromosomes). Median cancer-specific survival for patients with metastatic chromophobe RCC is 0.6 year, similar to the prognosis for patients with other types of metastatic RCC.
A formal description of collecting duct carcinoma was first published in 1986. This neoplasm arises in the principal cells of the collecting ducts of Bellini. It accounts for less than 1% of renal malignancies. Patients range in age from 13 to 87 years of age, with a mean age of 55 years. Collecting duct carcinoma occurs more commonly in males, with a male-to-female ratio of at least 2:1. Although up to 25% of cases are discovered incidentally, the majority of patients present with symptoms, including abdominal or flank pain and hematuria. Less common symptoms include weight loss, fatigue, fever, musculoskeletal pain, anorexia, and gastrointestinal disturbances, and rarely the presenting symptoms are those of metastatic cancer. Imaging studies usually demonstrate a predominantly solid mass consistent with renal carcinoma. Urine cytology is positive for malignant cells in a small percentage of cases. Bone metastases, which may be present at the time of diagnosis, are often osteoblastic.
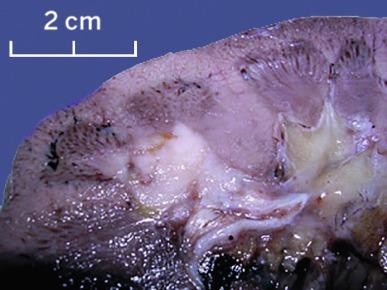
Small collecting duct carcinomas are usually located centrally and may be localized to a medullary pyramid. However, collecting ducts extend from the medulla to the cortex, and therefore collecting duct carcinoma can originate anywhere in the parenchyma, and identification of the site of origin may not be possible, particularly in large tumors. Tumors range from 1 to 16 cm in greatest dimension. The cut surface of collecting duct carcinoma is usually gray-white and firm, and areas of necrosis may be evident ( Fig. 2.48 ). Tumor borders are irregular and indistinct. Intrarenal metastases may be evident as satellite tumor nodules. Tumor involvement of the adrenal gland, perirenal fat, renal sinus fat, renal pelvis, Gerota fascia, renal vein, or regional lymph nodes is grossly evident in most cases.
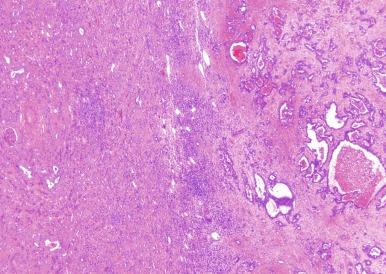
Microscopically, collecting duct carcinoma has an ill-defined border, with extensive infiltration of adjacent parenchyma and frequently an interstitial growth pattern with preservation of glomeruli. Usually there is an acute and chronic inflammatory cell infiltrate at the interface between tumor and normal parenchyma ( Figs. 2.49 and 2.50 ). Pronounced stromal desmoplasia is a constant feature ( Fig. 2.50 ). A variety of growth patterns may coexist within a given tumor. Tumors may grow in solid sheets/cords/nests, form tubulopapillary structures, or infiltrate desmoplastic stroma as small to medium-sized elongated tubules, but do not exhibit the type of tubulocystic architecture that typifies tubulocystic carcinoma. The presence of multiple nodules of desmoplastic stroma infiltrated by papillary tumor is a feature somewhat distinctive for collecting duct carcinoma ( Fig. 2.51 ). Some tumors have microcysts with intracystic papillary proliferations of high-grade carcinoma. Some tumors show intratubular extension with microscopic subcapsular deposits distant from the main tumor. The epithelium lining native ducts adjacent to or distant from the main tumor mass may show marked cytologic atypia or frank carcinoma in situ. Tumor cells are almost always of high nuclear grade, with nuclear pleomorphism and prominent nucleoli and varying amounts of eosinophilic cytoplasm ( Fig. 2.52 ). Cells lining luminal structures may display a hobnail appearance, a finding that is not typical of other types of RCC. Small vessel invasion is common, and renal vein involvement is evident in about 20% to 44% of cases. The incidence of sarcomatoid change varies from 16% to 36%.
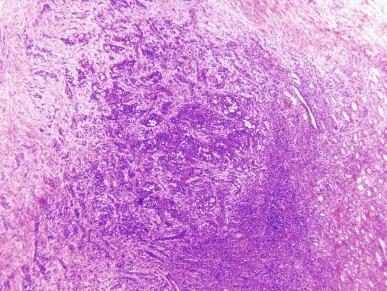
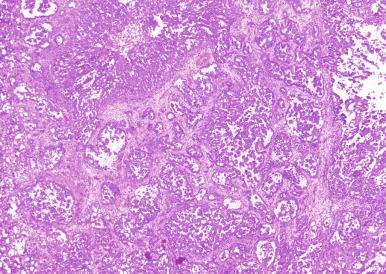
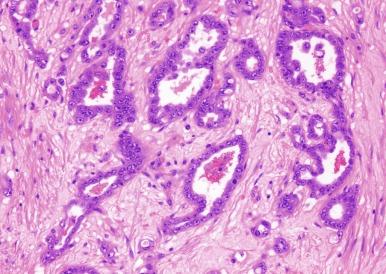
Mucin is often demonstrable by mucicarmine, Alcian blue, or undigested periodic acid–Schiff stains, within the tumor cell cytoplasm, at the luminal borders, or extracellularly ( Fig. 2.53 ).
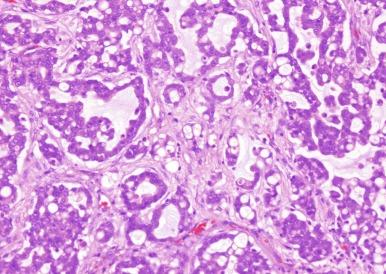
A diagnosis of collecting duct carcinoma is challenging, in that several other entities enter the differential ( Table 2.4 ). Metastatic adenocarcinoma must be excluded on clinical grounds. High-grade urothelial carcinoma must be excluded by generous sampling of the pelvicaliceal system. An immunohistochemical panel of PAX8, p63, and GATA3 may assist: tumors that are GATA3 + or p63 + and PAX8 - are more likely urothelial, whereas collecting duct carcinoma is likely to be PAX8 + , p63 - , and GATA3 - ( Table 2.3 ). Unfortunately, PAX8 has also been noted in urothelial carcinomas of the renal pelvis. Tumors expressing both PAX8 and S100 protein are highly likely to be of renal origin, and use of this panel may assist in excluding urothelial carcinoma. In confirming a diagnosis of collecting duct carcinoma, renal medullary carcinoma (RMC) must be considered and excluded. This issue is addressed later in the Renal Medullary Carcinoma section. Finally, recent experience with aggressive high-grade renal cancers has shown that as many as 25% previously diagnosed as collecting duct carcinoma are in fact examples of FH-deficient RCC. These tumors are addressed more fully in the Hereditary Leiomyomatosis and Renal Cell Carcinoma–Associated Renal Cell Carcinoma section. Consequently, immunostains for FH and 2SC should be performed on tumors suspected of being collecting duct carcinoma, complemented as needed by molecular genetic testing for FH mutations. In short, a diagnosis of collecting duct carcinoma is most confidently rendered after exclusion of metastatic adenocarcinoma, urothelial carcinoma of the pelvicaliceal system, RMC, and FH-deficient RCC.
Medullary involvement
Predominantly tubular morphology
Desmoplastic stromal reaction
Cytologically high grade
Infiltrative growth pattern
Absence of other renal cell carcinoma subtypes or urothelial carcinoma
The reported molecular changes in collecting duct carcinoma are markedly variable and limited, and no distinctive molecular mechanism or pathway has been proposed for collecting duct carcinoma.
The prognosis for collecting duct carcinoma is poor. Tumor stage is often advanced at the time of presentation; in one series 91% were pT3 or pT4 when initially diagnosed. Nearly 50% of patients have nodal or distant metastases at the time of initial diagnosis. Lung, bone, and brain are common sites of metastasis. Approximately two-thirds of patients with collecting duct carcinoma die of cancer within 2 years, and mortality rates as high as 83% have been reported by some investigators.
First reported in 1995, RMC is an uncommon, highly aggressive renal malignancy. It is believed to arise in the terminal collecting ducts and their adjacent papillary epithelium. Its occurrence is very strongly associated with sickle cell hemoglobinopathies, so it has been postulated that the earlier-noted epithelium suffers chronic ischemic damage related to sickling erythrocytes, and that RMC originates in a setting of chronic regenerative proliferation of this damaged epithelium. The majority of affected patients have been African Americans with sickle cell trait (HbAS) or hemoglobin SC disease (HbSC), but it has also been reported in a patient with sickle cell disease (HbSS).
Most reported patients with RMC are in their second and third decades of life (range, 5 to 58 years); mean age at diagnosis is approximately 20 years. The male-to-female ratio is at least 2:1, and for reasons that are inapparent, about 75% of tumors occur on the right and 25% occur on the left. The patients are most commonly of African ancestry, but individuals of Central and South American and Mediterranean ancestry are also at risk for RMC. In reported cases in which sickle cell status was known, 87% had sickle cell trait (HbAS), 9% had hemoglobin SC disease (HbSC), and 4% had sickle cell disease (HbSS).
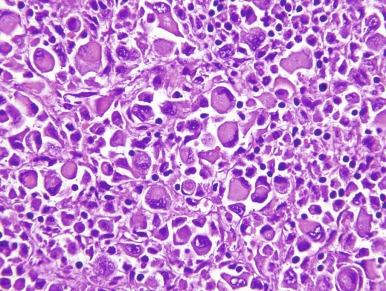
Abdominal or flank pain (52%), gross hematuria (52%), and weight loss (23%) are the most common presenting symptoms. Fever, nausea, and vomiting are also common, and about 10% of patients have a palpable abdominal mass. Duration of symptoms before diagnosis ranges from 2 days to 60 weeks, but the median is relatively short (8 weeks).
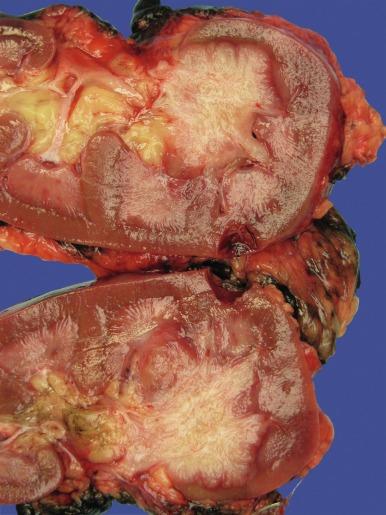
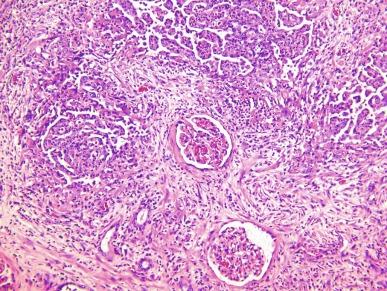
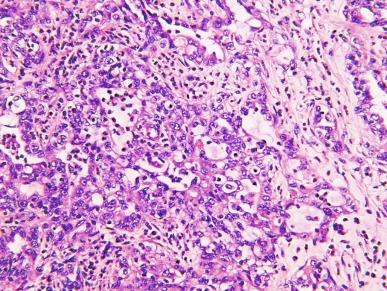
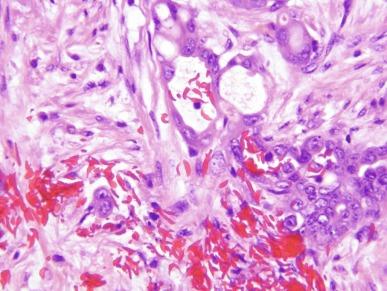
Grossly, these tumors typically occupy the renal medulla and are poorly circumscribed, lobulated, firm to rubbery, tan to gray, with varying degrees of hemorrhage and necrosis ( Fig. 2.54 ). They range in size from 1.8 to 13 cm in greatest dimension, with a mean of 7 cm. Satellite nodules are commonly present, as is extension into the perinephric and sinus fat. Microscopically, they demonstrate a variety of morphologic patterns. The most characteristic finding is a reticular or microcystic growth pattern that resembles yolk sac tumor of the testis. Sievelike/cribriform growth patterns are also commonly noted. Other common patterns include tubule formation and growth in solid sheets, cords, or nests. Tumor cells are typically pleomorphic, with large nuclei, prominent nucleoli, and varying amounts of eosinophilic cytoplasm. Tumor cells may have a squamoid or rhabdoid appearance, particularly in areas of sheetlike growth. Abundant neutrophils may be noted within the tumor, and there is often an intense inflammatory response at the interface between tumor and the adjacent renal parenchyma ( Fig. 2.55 ). The tumor incites a prominent desmoplastic stromal reaction. Varying degrees of mucin production are evident in most RMCs ( Fig. 2.56 ). In most histologic sections, sickled erythrocytes are identified ( Fig. 2.57 ).
Tumor cells in all cases show positive immunostaining for PAX8. Loss of expression of SMARCB1 (INI1), a nuclear transcription regulator encoded on chromosome 22, is considered to be a necessary criterion for a diagnosis of RMC ( Fig. 2.58 ). This is due to loss of heterozygosity or hemizygous deletions at the SMARCB1 locus, and in rare instances, loss of chromosome 22. In one study, inactivation of the SMARCB1 gene was shown to be due to interchromosomal balanced translocations. In the same study, whole exome sequencing showed that RMC has no other recurrent genetic alterations and an overall stable genome, underscoring the oncogenic potency of SMARCB1 inactivation. Between 40% and 70% of RMCs show positive immunostaining for OCT3/4 (also known as POU5F1). Variable degrees of positive immunostaining are noted for AE1/AE3 and CAM5.2, CK7, CK20, polyclonal carcinoembryonic antigen (CEA), Ulex europaeus agglutinin (UEA-1), and EMA.
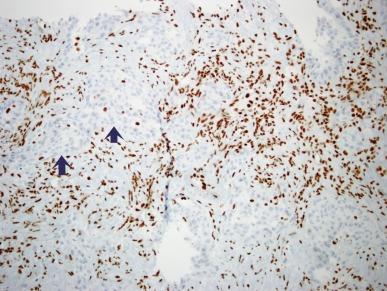
In view of the fact that 95% of patients with RMC have metastatic cancer at the time of diagnosis or shortly thereafter, it is not surprising that the prognosis for this neoplasm is poor. Common sites of metastasis are lymph nodes, lung, liver, and adrenals. Long-term disease-free survival is rare; survival ranges from 2 to 68 weeks, with a mean survival duration of 19 weeks. Survival is somewhat longer overall in patients given adjuvant therapy, but is usually limited in duration, and even in cases of prolonged remission, recurrence and death are the usual outcome.
There is some overlap in the clinical and pathologic features of collecting duct carcinoma and RMC, prompting speculation that RMC is a subtype of collecting duct carcinoma. Some investigators have adopted the stance that loss of SMARCB1 immunostaining and documentation of hemoglobinopathy by history and appropriate laboratory studies, and evidence of diffuse sickling within the tumor stroma as well as blood vessels, are of paramount importance in making a diagnosis of RMC. The rare tumors that exhibit RMC-like histology, SMARCB1-deficient immunophenotype, and aggressive clinical features but arising in patients in whom sickle cell trait or disease has been rigorously excluded are designated as RCC, unclassified, with medullary phenotype.
Tumors in this class of RCCs are characterized by gene fusions involving TFE3 or TFEB , two members of the MiT family of transcription factors. Translocation renal cell carcinomas (TRCCs) that are associated with Xp11 translocations harbor gene fusions involving TFE3 , and those with t(6;11) translocation harbor a MALAT1-TFEB gene fusion. The t(6;11) TRCCs are less common than Xp11 TRCCs.
Although the majority of patients with TRCC are younger than 50 years, patients up to 79 years of age have been reported. Translocation carcinoma occurs with approximately equal frequency in males and females, and in some patients its development has been associated with prior chemotherapy treatment. About one-half of patients present with abdominal or flank pain or hematuria; some tumors are detected as a palpable mass, and some are found incidentally.
Renal carcinoma accounts for less than 5% of pediatric renal neoplasms. Half or more of pediatric RCCs are translocation RCCs. Translocation carcinoma accounts for between 1% and 4% of adult RCCs.
TFE3 rearranged tumors involve the Xp11.2 gene locus and fall into one of three general categories: Xp11 TRCCs, melanocytic Xp11 TRCCs, and Xp11 PEComas.
Xp11 TRCCs are defined by a variety of chromosome translocations, all of which involve a breakpoint at Xp11.2, and all of which result in the fusion of any one of multiple translocation partners with the TFE3 transcription factor gene at this locus, triggering oncogenic activation of the TFE3 transcription factor. Known TFE3 fusion partners include ASPSCR1 (ASPL) , PRCC , SFPQ1 (PSF) , NONO , CLTC , PARP14 , LUC7L3 , DVL2 , and KHSRP. Of these the most common are the ASPSCR1-TFE3 (also known as ASPL-TFE3 ) TRCC that results from a t(X;17)(p11;q25) translocation and the PRCC-TFE3 TRCC, resulting from a t(X;1)(p11;q21) translocation.
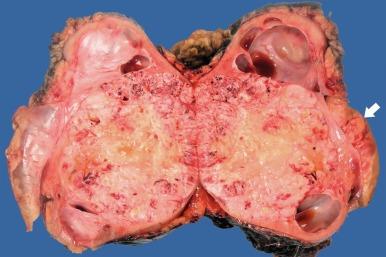
PRCC-TFE3 TRCC is typically yellow or gray, ranges in size from 3 to 14 cm in diameter, and has a fibrous pseudocapsule that has varying degrees of calcification in the majority of cases, sometimes substantial enough to impart an egg shell–like consistency ( Fig. 2.59 ). It is composed of tumor cells in papillary formations and in nests surrounded by thin-walled capillary vessels ( Figs. 2.60 and 2.61 ). The tumor cell nests are usually solid, but may be centrally discohesive, imparting an alveolar appearance, and some nests have central lumina; fresh blood in these lumina imparts a “bloody-gland” appearance typical of conventional CCRCC. The extent of the papillary component is variable; it may be the predominant pattern, or it may account for less than 10% of the tumor. Foam cells are evident in the minority of papillae. Psammoma bodies are often present and, although usually rare and isolated, may be abundant in some tumors. Transitions between areas of nested and papillary architecture are usually sharply demarcated. Tumor cells are uniformly polygonal, with sharply defined cell borders and with abundant cytoplasm that varies from clear and finely granular to distinctly eosinophilic. Proportions of clear and eosinophilic cells vary from tumor to tumor. Nuclei appear rounded at low power, but appear wrinkled and irregular at high power; nuclear grade is 2 or 3 in most cases. Areas of necrosis are seen in the majority of cases, but mitotic activity is very limited.
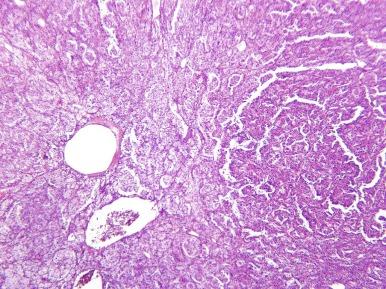
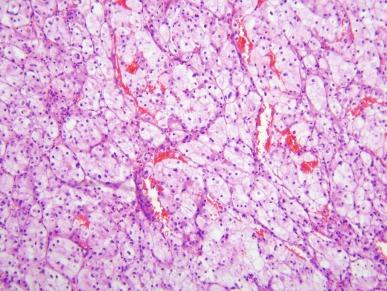
ASPSCR1-TFE3 TRCC is usually well circumscribed but unencapsulated. It displays nested and pseudopapillary growth patterns and psammomatous calcifications ( Fig. 2.62 ). Papillary structures lack foam cells and intracytoplasmic hemosiderin. Tumor cells are epithelioid, with well-defined cell borders and voluminous cytoplasm that is predominantly clear, but may also be finely granular, eosinophilic, or densely granular ( Fig. 2.63 ). Nuclei have vesicular chromatin and a single prominent nucleolus.
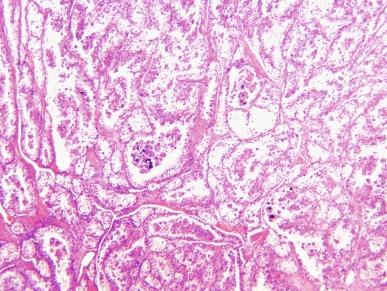
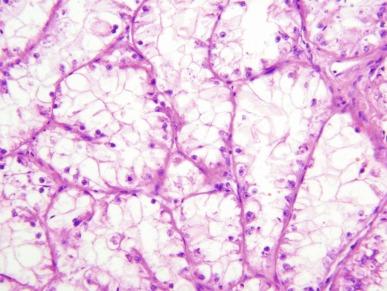
Rare examples of melanotic Xp11 translocation renal cancers have been reported in recent years. These tumors are composed of solid sheets and nests of polygonal epithelioid cells with clear to finely granular cytoplasm, containing variable amounts of finely brown pigment confirmed to be melanin by histochemical stains ( Fig. 2.64 ). Tumor cells show patchy immunoreactivity for melanoma antigens but negative immunostaining for keratins, PAX-8, and PAX-2.
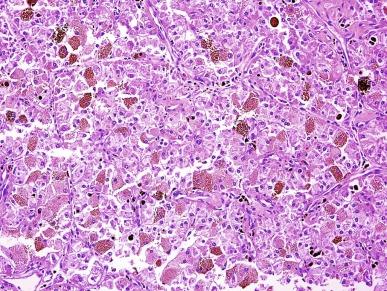
Immunohistochemically, MiT family RCCs consistently express PAX-8, but they underexpress epithelial immunohistochemical markers. Only about one-half of these tumors show positive staining for various cytokeratins or EMA, and the staining is often weak and/or focal. Only about 60% of Xp11 TRCCs express cathepsin K, and they only infrequently express melanocytic markers.
Although TFE3 immunostaining to detect the C-terminal portion of TFE3 is the most distinctive, sensitive, and specific immunohistochemical marker for Xp11 TRCCs, reliability of this stain has proven to be challenging because of fixation issues. A TFE3 break-apart FISH probe can be used to confirm the diagnosis ( Fig. 2.65 ).
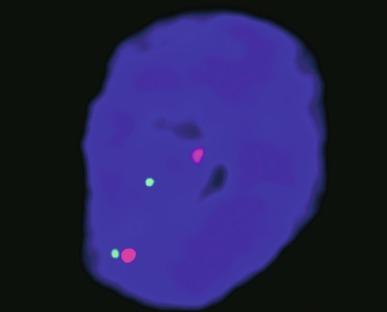
The number of patients with documented follow-up after treatment for the various types of translocation carcinoma is limited. Regarding TFE3 TRCCs, there are notable clinical differences between PRCC-TFE3 TRCC and ASPSCR1-TFE3 TRCC. TFE3 TRCC patients who present with distant metastases usually have ASPSCR1-TFE3 TRCC and usually die of cancer or exhibit disease progression. More than two-thirds of ASPSCR1-TFE3 TRCCs have metastatic cancer in regional lymph nodes; despite this, in the absence of concurrent distant metastases, the great majority of these patients achieve long-term freedom from disease. Only about one-third of PRCC-TFE3 TRCCs have positive nodes at presentation. However, PRCC-TFE3 TRCC occasionally recurs as late as 20 to 30 years after surgery, requiring long-term follow-up. The specific type of TFE3 TRCC appears to have no significant impact on outcome; only older age or advanced stage at presentation are predictive of death from cancer. Overall, cancer-specific survival for patients with TFE3-rearranged RCC is similar to the survival of patients with conventional CCRCC, but worse than the survival of patients with PRCC.
Only about 50 cases of t(6;11) TRCC have been reported. It is characterized by a t(6,11)(p21;q12) translocation that results in fusion of the 5′ portion of the Alpha gene with the transcription factor gene TFEB at 6p21.
Patients with t(6;11) TRCC range in age from 3 to 68 years, with a mean and median age of about 33 to 34 years. About one-half of patients report abdominal pain or hematuria, and the rest are asymptomatic.
Macroscopically, tumors are solid but often contain cysts, which rarely may be very large. Tumors are usually confined within a pseudocapsule, and range from 2 to 12 cm in greatest dimension (mean diameter, 7 cm). They typically display a homogeneous tan-yellow-brown color, without evidence of necrosis.
Microscopically, tumors demonstrate a solid, nested pattern of growth, characteristically with entrapment of single native renal tubules at their periphery, and distinctively biphasic cell populations. The predominant cell type is epithelioid, with well-defined borders, abundant clear, finely granular, or eosinophilic cytoplasm, and rounded or vesicular nuclei that have prominent nucleoli evident at low magnification and mild nuclear membrane irregularities at high power.
In the majority of cases, there is a second cell population comprising 5% to 30% of the tumor and consisting of smaller cells with denser chromatin, usually clustered around nodules of hyaline basement membrane material; these nodules have been described by some as “pseudorosettes” ( Fig. 2.66 ). Less commonly, tumors contain areas resembling epithelioid angiomyolipoma, chromophobe carcinoma, or CCRCC with cystic change. Mitotic figures are inapparent. Abortive papillae, brown pigmentations, psammomatous calcifications, and well-formed tubules are seen in a minority of cases.
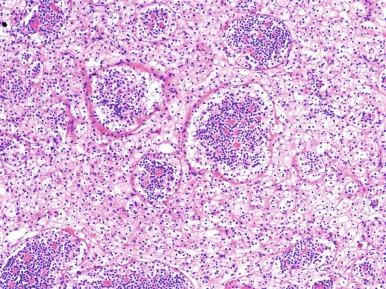
The t(6;11) TRCCs consistently express the cysteine protease cathepsin K and melanocytic markers Melan A and HMB45. They also express the KP1 clone of CD68, but do not express the PG-M1 clone of CD68. This can be used to distinguish them from pure epithelioid angiomyolipomas (AML), which express both the KP1 and the PG-M1 clones of CD68, as well as cathepsin and melanocytic markers. Although positive nuclear immunostaining is highly specific for t(6;11) TRCCs, a transcription factor EB (TFEB) break-apart FISH assay in formalin-fixed paraffin-embedded tissue is preferable as a diagnostic marker.
The t(6;11) TRCCs are generally confined within an intact pseudocapsule and usually present at stage pT1 or pT2. Of about 50 reported t(6;11) TRCCs, 11 patients were known to have metastatic cancer and of these, 4 were known to have died.
A subset of tumors occurs in the setting of germline mutations of SDHA, SDHB, SDHC, SDHD, and SDHAF2. These include pheochromocytomas/paragangliomas, gastrointestinal stromal tumors, RCC, and pituitary adenomas. Tumors associated with germline mutations of SDHB, SDHC, SDHD, and SDHAF2 show loss of immunostaining for SDHB, but retain positive staining for SDHA, whereas tumors associated with SDHA mutation show loss of immunostaining for SDHB, but also show loss of staining for SDHA. In 2010, a morphologically unique renal tumor occurring in a patient with a germline mutation and somatic loss of the wild-type SDHB allele was reported. In 2011, it was reported that renal carcinomas related to SDH mutation could be identified by loss of immunohistochemical staining for SDHB, and later that year it was reported that SDH-deficient RCC had distinctive morphologic features that could prompt immunohistochemical staining for SDHB, with the premise that loss of staining could prompt definitive genetic testing. In recent years, more than 60 renal neoplasms arising in the setting of germline SDH mutation have been reported, with mutation frequencies approximately as follows: SDHB, 81%; SDHC, 9%; SDHD, 5%; and SDHA, 5%. The molecular abnormality that defines SDH-deficient RCC is double-hit inactivation of one of the SDH genes, and this is an event that occurs only rarely, if ever, in the absence of a germline mutation. With this in mind, all patients with SDH-deficient RCC should be offered genetic testing, and surveillance for other SDH-deficient neoplasms (e.g., paraganglioma, pituitary adenoma, SDH-deficient gastrointestinal stromal tumor) should be initiated.
SDH-deficient RCC is estimated to account for about 0.05% to 0.2% of renal neoplasms. Vague flank pain is noted in some instances, but many are found incidentally. Mean age at presentation is 37 years (range, 14 to 76 years), and there is a male predominance of nearly 2:1. The tumors are typically well circumscribed, 2 to 20 cm in diameter, with a tan to red-brown cut surface, and sometimes with a cystic component. Tumors are expansile with a lobulated or pushing border and infrequently are separated from the adjacent normal kidney by a pseudocapsule. At the interface between tumor and normal kidney, tumors may surround benign elements from the adjacent kidney. Microcysts and macrocysts are commonly noted, usually containing pale eosinophilic fluid ( Fig. 2.67 ). Stromal myxoid change or hyalinization is seen in some tumors. Tumors are composed of cuboidal to ovoid cells with round nuclei. Nucleolar prominence consistent with ISUP nucleolar grade 2 is seen in the majority, but some exhibit grade 3 nucleoli, and sarcomatoid change consistent with ISUP grade 4 is noted in some cases. Cell borders may be indistinct ( Fig. 2.68 ). The cytoplasm is eosinophilic or flocculent and may appear bubbly, but not truly oncocytic. A distinctive finding is the presence of cytoplasmic vacuoles and inclusion-like spaces, containing either pale eosinophilic fluid or flocculent material. True coagulative necrosis is seen in some tumors, usually in nucleolar/nuclear grade 3 or 4 tumors. Tumor cells show positive immunostaining for PAX8 and kidney-specific cadherin, but absence of immunostaining for SDHB. Other immunostains are not helpful in establishing the diagnosis. Notably, immunostains for CD117, RCC-Ma, and CA-IX are negative, and keratin markers are frequently negative.
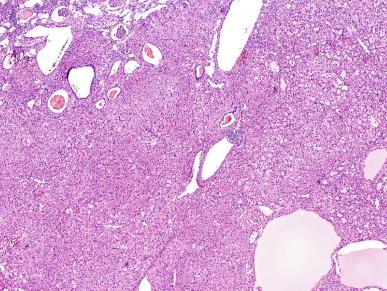
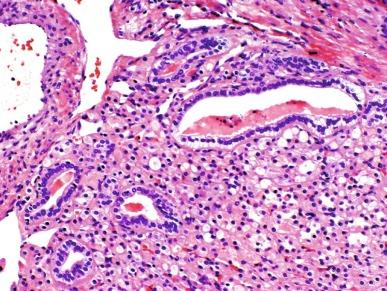
Patients whose SDH-deficient RCCs are uniformly low grade and lack coagulative necrosis have about a 90% likelihood of a good outcome. Tumors that exhibit coagulative necrosis, grade 3 nucleoli, or sarcomatoid change have a guarded prognosis, with metastasis in up to 70% of cases. Cases in which metastatic cancer appeared 16 to 30 years after tumor excision may represent metastasis of an unsampled metachronous tumor in the opposite kidney.
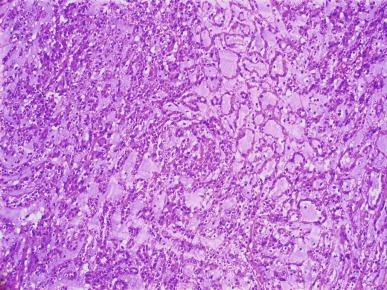
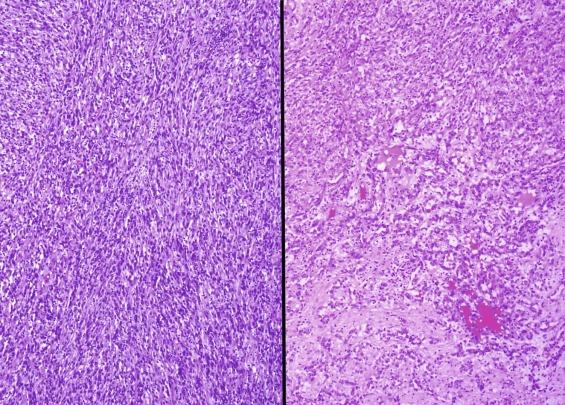
Five examples of mucinous tubular and spindle cell carcinoma were first reported in 1997 by MacLennan and colleagues under the name of “low-grade collecting duct carcinoma.” Four similar cases were reported in 2001, described as “low-grade myxoid renal epithelial neoplasms,” and five more patients with similar tumors in 2002 as “low-grade tubular-mucinous renal neoplasms.” The tumor occurs predominantly in females, with a 1:3 male-to-female ratio. Patients range in age from 13 to 82 years, with a mean age of 58 years. Although some tumors are symptomatic, the majority are discovered incidentally.
Tumors range in diameter from 2.2 to 12 cm, averaging 6 to 7 cm. Tumors are sharply circumscribed, gray-white, tan, or yellow, sometimes with minimal hemorrhage or necrosis ( Fig. 2.69 ). Classic tumors are composed of tightly packed, small elongated tubules separated by abundant basophilic extracellular mucin, sometimes with a “bubbly” myxoid consistency ( Figs. 2.70 and 2.71 ). Focally, aggregates of spindled cells may be present ( Fig. 2.72 ). The mucin stains strongly with Alcian blue at pH 2.5 ( Fig. 2.73 ). Tubules are lined by uniform low cuboidal cells with scant cytoplasm and round nuclei of low nuclear grade with absent or inconspicuous nucleoli. Mitotic figures are rare.
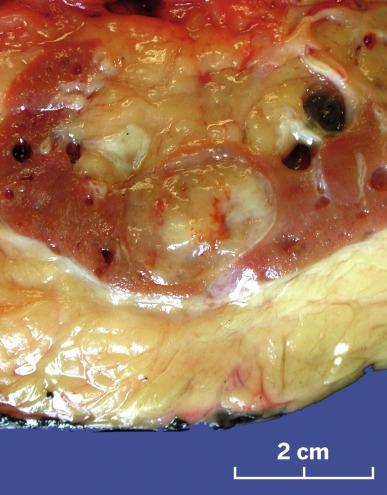
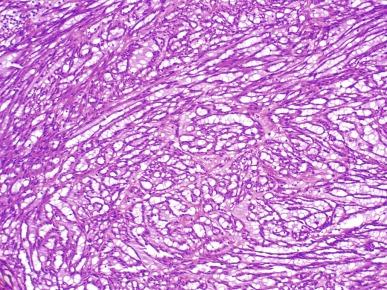
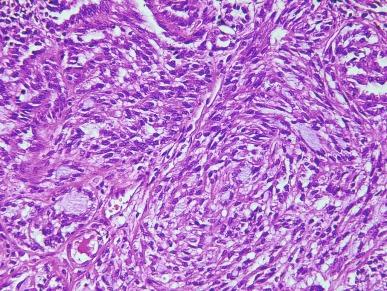
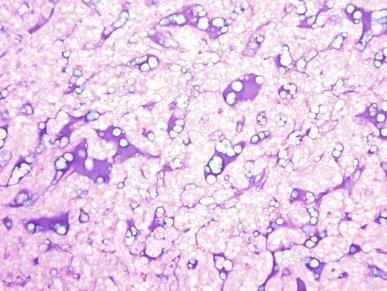
The spectrum of acceptable histologic findings in mucinous tubular and spindle cell carcinoma has expanded with increasing recognition of this tumor, to include cases with relative paucity of mucinous matrix, aggregates of foamy macrophages, papillations or small components of well-formed papillae, focal clear cell change in tubular cells, focal necrosis, oncocytic tubules, numerous small vacuoles, psammomatous calcification, or heterotopic bone formation ( Figs. 2.74 and 2.75 ). Rarely the spindle cell component rivals or even exceeds the tubular component ( Figs. 2.76 through 2.79 ). Furthermore, there is increasing recognition that high nuclear grade, areas of coagulative tumor necrosis, sarcomatoid change, and aggressive biologic behavior can be encountered in a small subset of these tumors. Tumor cells have a complex and markedly variable immunophenotype, but are typically immunoreactive for CK7, AMACR, and PAX8 ( Table 2.3 ).
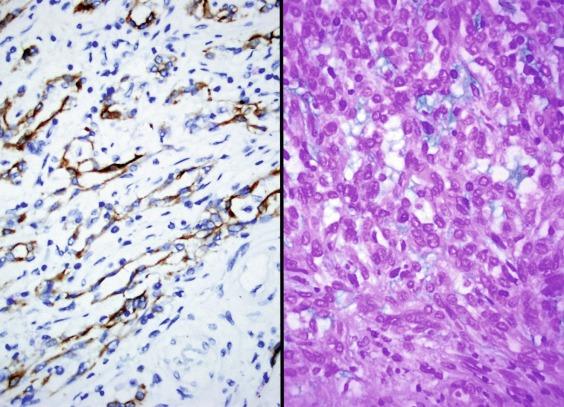
Despite morphologic similarities to PRCC in some cases, the gains of chromosomes 7 and 17 and loss of Y chromosome that are characteristic of PRCC are not seen in mucinous tubular and spindle cell carcinoma. Alterations in the Hippo pathway are present in mucinous tubular and spindle cell carcinoma but are not seen in PRCC. Cytogenetic analyses and comparative genomic hybridization studies have revealed multiple genetic alterations that include losses of chromosome 1, 4, 6, 8, 9, 13, 14, 15, 18, and 22.
These tumors are generally of low pathologic stage at the time of excision, and most tumors with classic histologic findings behave in an indolent fashion. However, high nuclear grade and sarcomatoid change in isolated cases have been associated with fatal outcomes.
Tubulocystic renal cell carcinoma is a rare distinctive neoplasm; examples of it were presented at a meeting of the United States and Canadian Academy of Pathology and described in abstract form in 1994, and several illustrations of the gross and microscopic appearance of these neoplasms were presented in the AFIP Atlas of Tumor Pathology, Series III, in 1994. Details of the clinical and pathologic details of eight examples of this neoplasm were first reported in 1997 by MacLennan and colleagues. It was emphasized that although the neoplasm had some features suggestive of collecting duct origin, it was distinctly different from classic collecting duct carcinoma in many ways. Because these tumors were clearly composed of tubules and ductlike structures, showed areas of hobnail change, and had immunohistochemical characteristics similar to those of classic collecting duct carcinoma (i.e., positivity for 34βE12 and UEA-1), it was hypothesized that they were of collecting duct origin and that they represented the low-grade end of a spectrum of findings in collecting duct carcinoma, hence the term low-grade collecting duct carcinoma. Subsequently, the clinical and pathologic features of additional examples of similar tumors were reported, and the name of the lesion was changed to tubulocystic carcinoma.
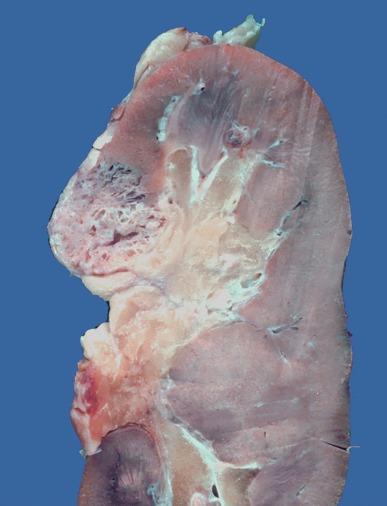
Close to 100 cases of this neoplasm have been reported, with a strong male preponderance (≥ 7:1). Tubulocystic carcinoma of the kidney is a well-circumscribed tumor that ranges in size from 0.7 to 17 cm, is often grossly cystic, and lacks hemorrhage or necrosis; some show a “bubble-wrap” appearance of the cut surface ( Fig. 2.80 ). The microscopic appearance at very low power is reminiscent of a spiderweb or lace doily ( Fig. 2.81 ). The tumor is composed of tubules and cystic structures of markedly variable size, separated by septa that are commonly delicate and “spiderweb-like,” or by fibrotic septa of variable thickness ( Fig. 2.82 ). Tubules are lined by a single layer of low cuboidal epithelial cells with modest to abundant amounts of eosinophilic cytoplasm, commonly with areas of hobnail appearance ( Fig. 2.83 ). Nuclei are round, with evenly dispersed chromatin, and have readily evident nucleoli in some cases. Nuclear grade is 1 or 2 in all instances. No necrosis is present, and mitotic activity is extremely limited.
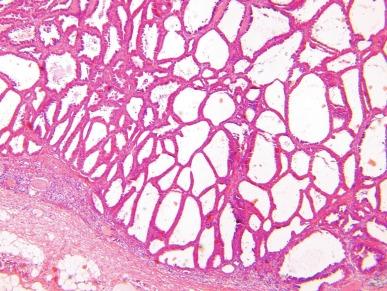
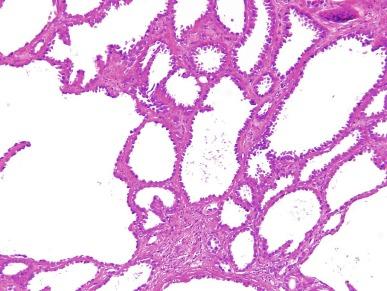
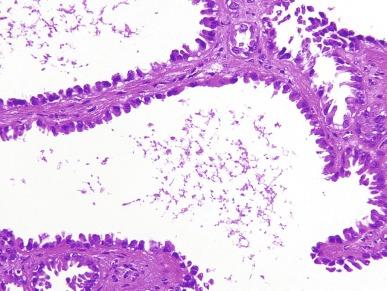
Immunohistochemically, tumor cells in classic cases of pure tubulocystic carcinoma show strong and diffuse labeling for PAX8, with frequent AMACR expression ( Table 2.3 ). The minority show focal immunoreactivity for CA-IX and patchy reactivity with antibody against CK7 (in 30% to 40% of cells).
Molecular studies in classic cases of pure tubulocystic carcinoma show no gains of chromosomes 7 or 17, and no TFE3 translocation by FISH. Other investigators have noted disomy of chromosome 7 in the majority, loss of Y chromosome in one case, and polysomy or gain of chromosome 17 in 50% of their classic cases. Gene expression profiling demonstrates that the molecular signature of tubulocystic carcinoma is distinct from those of papillary, clear cell, and chromophobe carcinomas. Tubulocystic carcinoma and collecting duct carcinoma are unrelated from a molecular perspective.
Stage and nuclear grade are typically low, and biologic activity is indolent, with a potential for metastasis that is less than 10%. Staging data and available follow-up in currently reported patients showed that one patient had lymph node metastases when first diagnosed, but was alive without disease at follow-up, another had a local recurrence, one patient experienced bone metastases, and a fourth developed both bone and liver metastases and died of cancer.
The diagnosis of tubulocystic carcinoma should be restricted to tumors with the classic histologic features. Some investigators have proposed a close relationship between tubulocystic carcinoma and PRCC, based on evaluation of tumors with papillary components admixed with areas resembling tubulocystic architecture. Some have noted morphologic similarities between tubulocystic carcinoma and t(6,11) TRCC, and others have reported tumors with poorly differentiated solid and papillary components admixed with areas resembling tubulocystic architecture. Poorly differentiated high-grade renal carcinomas that have areas closely resembling tubulocystic carcinoma have been shown in many instances to be examples of FH-deficient RCC. Cases of tubulocystic carcinoma with purely classic morphology are distinctive and lack the full spectrum of genetic and immunohistochemical features that are classically seen in PRCC.
The incidence of ACKD in the dialysis population is proportional to dialysis interval, with close to 100% of patients affected by 10 years of dialysis. Patients with ACKD are especially prone to the development of carcinoma, with an incidence rate of 3% to 7% and a risk that increases with the duration of dialysis, approaching 100 times that of the general population. The biologic basis for increased renal carcinogenesis in end-stage renal disease is undefined. Depressed immunity, excessive free radical production related to inflammation, impaired antioxidant defenses, and deposition of oxalate crystals have been postulated.
The RCCs that arise most commonly in a setting of ACKD are, in descending order of frequency, acquired cystic disease–associated RCC (ACD-RCC) (36%), CCPRCC (23%), CCRCC (18%), PRCC (15%), and chromophobe RCC (5%). Of these tumors, only ACD-RCC, by definition, occurs only in patients with ACKD. The earliest report of RCC with abundant oxalate crystals appeared in 1998. The characteristics of ACD-RCC were subsequently described more fully in large series of cases in 2005 and 2006. Patients with ACD-RCC tend to be relatively young and predominantly male. Tumors are identified primarily through routine screening protocols, although some present with microscopic hematuria or flank pain.
Grossly, ACD-RCCs range from 1 to 8 cm, and are often multifocal and bilateral ( Fig. 2.84 ). More than 50% of cases are multifocal, and more than 20% are bilateral. Tumors are generally well circumscribed. Foci of hemorrhage and necrosis are common, and some have focally calcified capsules. Many appear to have arisen in cysts. Histologically the tumor architecture demonstrates various combinations of solid sheetlike, papillary, acinar, and cribriform and tubulocystic patterns. The tumor cells are typically large with abundant granular eosinophilic cytoplasm and large round to oval, mildly irregular nuclei with prominent nucleoli. About 80% of these tumors show abundant intratumoral calcium oxalate crystals within luminal structures and in the stroma ( Figs. 2.85 and 2.86 ). The crystals are not associated with fibrosis, necrosis, increased mitoses, or inflammation. Intratumoral calcium oxalate deposition is a feature that appears to be restricted to tumors arising in a background of ACKD.
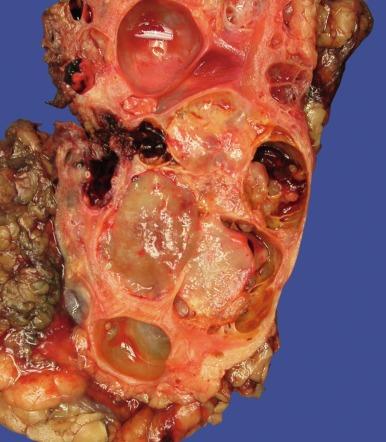
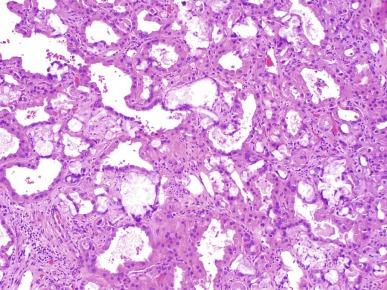
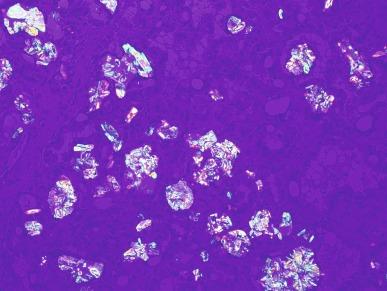
Studies indicate that ACD-RCC arises from cells of the proximal nephron. Tumor cells show positive immunostaining for CD10, RCC-Ma, and often for AMACR. Tumor cells typically show no immunostaining for CK7. Gains in chromosomes 3, 7, 16, and 17 and the sex chromosomes are noted with high frequency in ACD-RCC.
The prognosis for ACD-RCC is generally favorable, because it tends to have an indolent behavior, and constant medical surveillance of dialysis patients facilitates diagnosis at an early stage. However, when sarcomatoid or rhabdoid morphology is identified, it often portends a poor outcome ( Figs. 2.87 through 2.89 ).
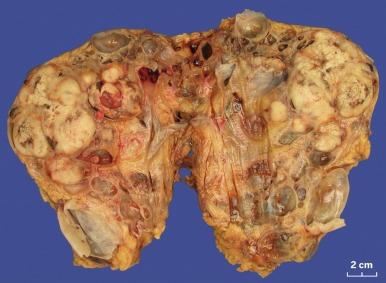
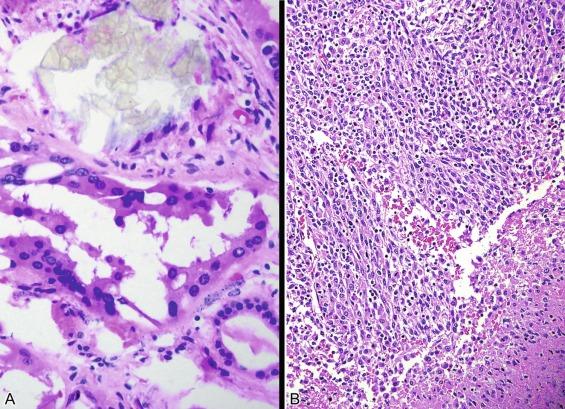
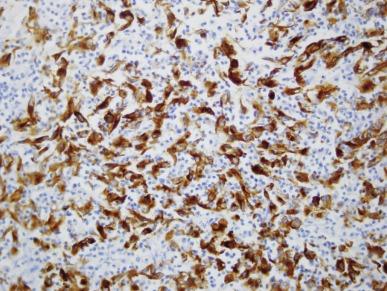
Although originally described as one of two unique tumors typically arising in end-stage renal disease, with and without acquired cystic change, it is now known that the majority of CCPRCCs arise sporadically, accounting for up to 4.3% of resected kidney tumors, occurring about equally in males and females, and comprising the fourth most common renal tumor. Patients range in age from 18 to 88 years, with a mean age of ~ 60 years. In one series, there was a propensity for occurrence in African American patients.
Grossly, CCPRCC is typically small, with a mean diameter of 2.0 cm, but varies in size between 0.2 and 9 cm. Tumors are well circumscribed, often cystic, with a thin, fibrous capsule and without hemorrhage or necrosis ( Fig. 2.90 ), and may be multiple or bilateral. Necrosis and hemorrhage are absent. Cyst formation is nearly universal and is extensive in about one-third of tumors. Solid areas appear white-tan to pale yellow.

Histologically, papillary architecture is usually limited to focal branching or small blunt papillae; extensively branched papillae are uncommon ( Fig. 2.91 ). Most tumors exhibit areas of a dense, compact arrangement of tubular structures, similar to CCRCC. A distinctive finding is the presence of cells with nuclei aligned at the apical end of the cells, similar to early secretory phase endometrium ( Fig. 2.92 ). Most cells lining the papillary and tubular structures exhibit clear cytoplasm and a low nucleolar grade. Necrosis is absent.
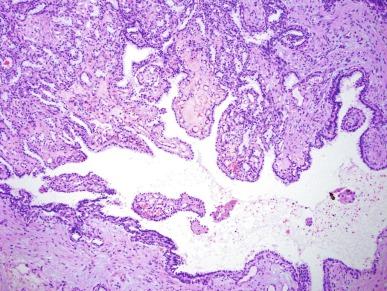
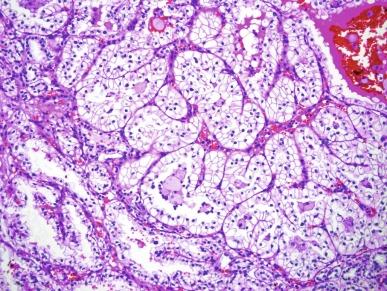
A small subset of tumors (previously designated renal angiomyoadenomatous tumors) show similar cytologic findings but with “collapsed” acini, variable tubular/acinar architecture, and prominent components of fibrous or smooth muscle stroma.
Immunohistochemically, tumor cells show positive immunostaining for CK7 and CA-IX ( Table 2.3 ) and negative immunostaining for AMACR and CD10. Positive immunostaining for 34βE12 is noted in more than 90% of cases.
In contrast with conventional CCRCC and PRCC, CCPRCC lacks the VHL 3p25 deletion and also lacks VHL gene mutations, VHL promoter hypermethylation, trisomies of chromosomes 7 and 17, and loss Y chromosome, although low copy number gains of chromosomes 7 and 17 have been reported in a small number of cases. No mutations of KRAS , NRAS , BRAF , PIK3CA , ALK , ERBB2 , DDR2 , MAP2KI , RET , or EGFR genes were detected in a recent study. Currently no specific characteristic genetic abnormality has been found.
Although the differential diagnosis for CCPRCC is broad, the morphologic distinction between CCPRCC and conventional CCRCC presents the greatest challenge. Immunopositivity for CK7 does not exclude CCRCC. Discordant immunoprofiles, such as immunopositivity for CD10 or AMACR, or an immunoprofile that fits neither CCPRCC nor CCRCC, are best regarded as a reason to avoid making a diagnosis of CCPRCC. Similarly, tumors with solid growth or large nested areas resembling CCRCC are best not diagnosed as CCPRCC, nor may it be prudent to make a definitive diagnosis of CCPRCC on limited material.
Rare cases of CCPRCC have been reported to arise in patients with VHL disease. Studies of a number of CCPRCC-like tumors occurring in patients with VHL disease showed that they usually lack the characteristic immunohistochemical profile of sporadic CCPRCC, and FISH analysis of chromosomes 3p, 7, and 17 supported a molecular pathogenesis more similar to that of CCRCC than of CCPRCC.
More than 90% of CCPRCCs are stage pT1 at presentation. A single patient with sarcomatoid change in a CCPRCC had a fatal outcome. Of several hundred reported cases of conventional CCPRCC, no patient has reportedly died of or suffered recurrence of this neoplasm, leading some to propose that CCPRCC be renamed “clear cell papillary neoplasm of low malignant potential.”
Renal cell carcinoma, unclassified is a diagnostic category for the designation of RCCs that have histologic features that do not resemble those of any of the well-characterized RCC subtypes, accounting for 0.7% to 5.7% of RCC cases. Examples of tumors that are appropriately assigned to this category are tumors that are composites of recognized types, tumors composed of unrecognizable cell types, tumors with mucin production, and renal carcinomas with entirely sarcomatoid morphology, lacking recognizable epithelial elements ( Fig. 2.93 ). Low- or high-grade unclassifiable oncocytic neoplasms were also included in this category.
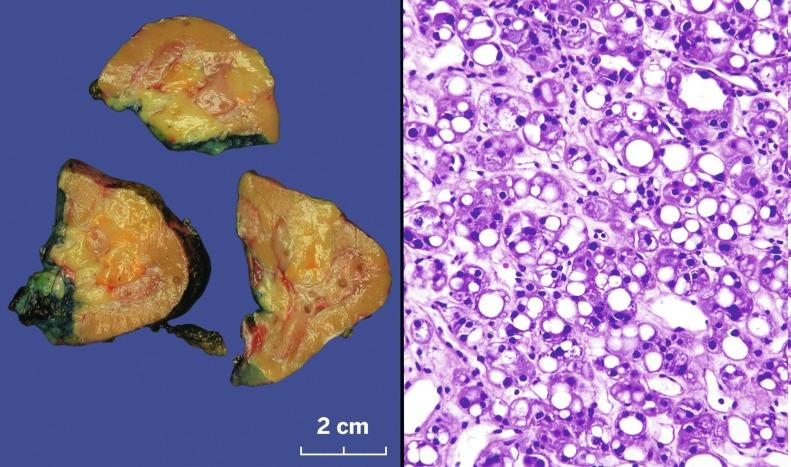
Reported age at presentation ranges from 21 to 91 years. There is a general tendency for such tumors to be large, at an advanced stage at presentation, and to exhibit histologic features that correlate with poor outcome, such as high nuclear grade, tumor necrosis, and microvascular invasion. The limited data available suggest that unclassified RCC have marked genetic instability; in one study, 40 mutated genes were identified in the tissues of unclassified RCC compared with a number of normal renal tissue samples. In another study of 62 unclassified RCC, molecular analysis revealed that about 75% of cases fell into one of several subsets of abnormalities of variable prognostic significance, raising the possibility that molecular analysis of these enigmatic tumors may have diagnostic and therapeutic implications.
Unclassified RCC is a histologically and clinically heterogeneous category of tumors whose prognosis seems to be linked to the same clinical and pathologic findings that influence outcome in conventional renal cancers. One study compared unclassified RCC with a 4:1 matched group of patients with CCRCC, with respect to year of surgery, symptoms at presentation, tumor size, stage, regional lymph node involvement, metastases, grade, coagulative tumor necrosis, and sarcomatoid change, and found no statistically significant differences in outcome after adjusting for these features in a matched analysis. Other investigators have reported that unclassified RCC tend to behave more aggressively than conventional renal carcinomas of the same stage and grade.
Secondary malignant neoplasms develop in approximately 13% of children treated for cancer. More than 30 cases of RCC arising in children who survived neuroblastoma in childhood have been reported in the last 40 years. Although the great majority were exposed to chemotherapy or radiation therapy, at least two never had either exposure, raising the possibility that the cause of such cancers may lie in an underlying genetic relationship or susceptibility. The tumors reported include CCRCC, PRCC, MiT family RCC, hybrid oncocytic-chromophobe tumor, and SDHB-deficient RCC, as well as a unique oncocytic RCC that is rare but is included in the category of emerging/provisional RCC.
The interval between a diagnosis of neuroblastoma and of oncocytic RCC postneuroblastoma ranges from 3 to 38 years, and age at diagnosis of oncocytic RCC postneuroblastoma ranges from 8 to 40 years. Tumors form expansile masses, composed microscopically of cells arranged in solid sheets intersected by cystic areas, nests, papillary structures, or forming tubules ( Fig. 2.94 ). Occasional psammoma bodies are present. Tumor cells have sharply defined cell membranes and abundant eosinophilic granular cytoplasm, giving them an “oncocytoid” appearance. Cells with abundant reticular cytoplasm may be present, with some morphologic resemblance to chromophobe cells ( Fig. 2.94 ). Tumor cell nuclei are medium sized and irregular, and many bear prominent nucleoli. Only occasional mitotic figures are observed. The tubular and solid architecture with varying cyst formation, cyst lining cells with a hobnail appearance and eosinophilic to flocculent cytoplasm, and variably sized cytoplasmic granules are similar to the findings in a recently described but not fully characterized renal carcinoma, eosinophilic solid and cystic RCC. There are insufficient data regarding molecular features and outcome for these tumors.
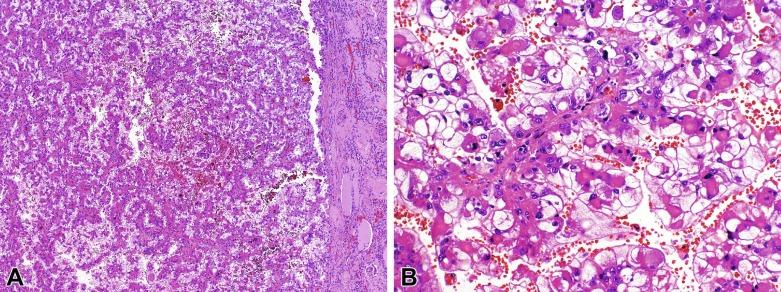
More than 50 cases of this rare tumor have been well studied and reported. Although it was initially identified in patients with TSC, it is now known to occur sporadically as well ( Fig. 2.95 ). The earliest reported patients were all women with a mean age of about 55 years (range, 31 to 79), but in a more recently reported series of cases, this tumor has been identified in males as well as females, and in younger patients as well (median age, 27 years; range, 14 to 35 years). The majority of eosinophilic solid and cystic renal cell carcinomas are single, but multifocality and bilaterality have been reported. Grossly they are tan or yellow-gray and most often solid and cystic grossly, although about 25% are entirely solid. They range from 1.5 to 13.5 cm in greatest dimension (median, 3.8 cm). With rare exceptions, they present at stage pT1 or pT2.
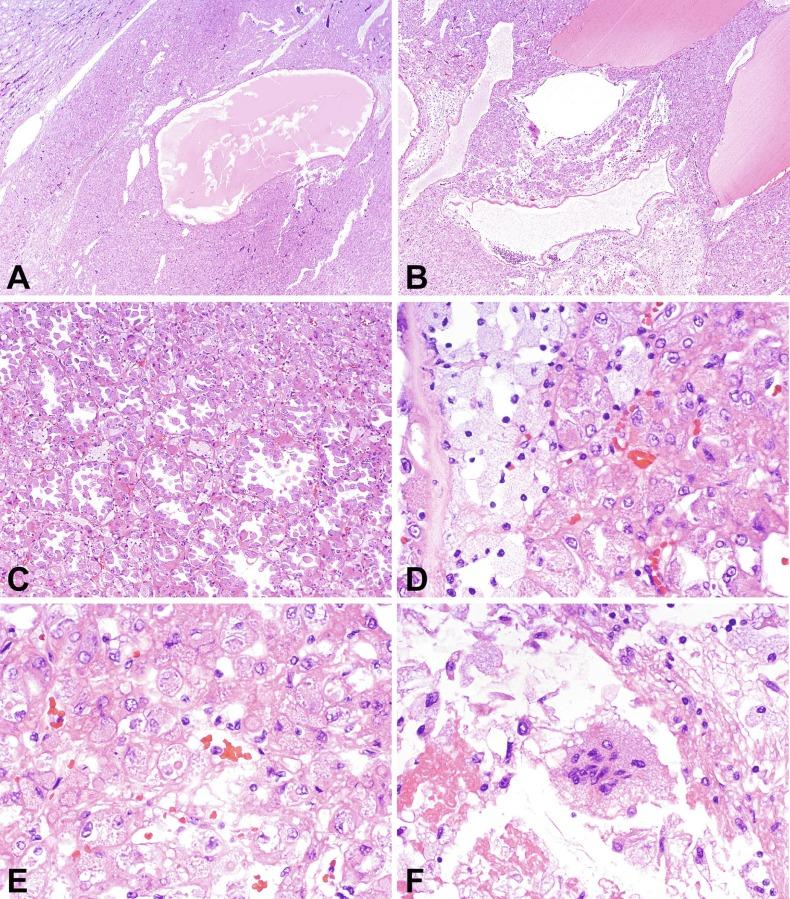
Microscopically, they are arranged in solid nests or sheets, always with interspersed aggregates of lymphocytes and histiocytes, and with psammoma bodies in about 50% of tumors ( Fig. 2.95 ). All tumors harbor variably sized macrocysts and microcysts; cells lining the cysts often exhibit a prominent hobnail appearance. Tumors are composed of cells with voluminous eosinophilic cytoplasm and round to oval nuclei, with focally prominent nucleoli. Multinucleated tumor cells are common. Intracytoplasmic vacuoles may be present. A unique and striking feature is the presence of cytoplasmic “stippling” by eosinophilic or basophilic amorphous granular structures of variable size and prominence.
Immunohistochemically, tumor cells are always PAX-8 + and CK8/18 + . Distinctively, about 75% of these tumors express CK20, and 75% do not express CK7, so the majority have a CK20 + /CK7 − phenotype. About 75% of tumors express keratin AE1/AE3, AMACR (patchy), and CD10 (patchy). Importantly, these tumors express the following markers only rarely (CD117; CK5/6) or never (melanocytic markers). SDHA and SDHB staining, as well as FH staining, is retained, and MiT family translocations are not present.
A recurring set of genomic alterations has been identified: copy number gains in 16p, 7p, 13q, and 19p; copy number losses in Xp11.2 and 22q; and a variety of losses of heterozygosity.
Initially it was believed that eosinophilic solid and cystic RCC was an indolent tumor. With increasing awareness of this tumor, currently two patients are known to have developed metastases (an incidence rate of about 3% in known cases).
The first reports of a primary renal neoplasm bearing a striking resemblance to follicular carcinoma of the thyroid appeared in 2006 and 2009. To date 39 cases have been reported. In all cases, no evidence of thyroid cancer was discovered, concurrently or subsequently. About 70% are found incidentally; the remainder experience hematuria or flank or abdominal pain. Patients range in age from 19 to 83 years (median, 35 years; mean, 43 years), and male-to-female ratio is nearly 1:2. Tumors are brown, tan-yellow, or gray; range in size from 1.1 to 1.3 cm; and are typically well circumscribed, often with a pseudocapsule. Cysts and areas of hemorrhage or necrosis are noted in about 50% of cases. Gross or microscopic extension into perinephric tissues is noted in 13% of cases, but extension into renal pelvis or renal vein has not been noted. Microscopically, tumors exhibit a striking resemblance to thyroid tissue or thyroid neoplasia with follicular architecture, being composed of variably sized follicles with inspissated colloid-like material in the follicular lumina ( Fig. 2.96 ). The follicles are lined by cuboidal or flattened epithelial cells with uniform nuclei of modest size. About 20% of cases have microcalcifications. Exclusion of metastatic thyroid carcinoma or ovarian struma ovarii is necessary. In this regard immunostains for thyroglobulin and TTF1 have been uniformly negative in reported cases. About 10% of patients have metastases at presentation or have them later; lymph nodes, lung, and skull are favored sites. The carcinoma has not resulted in patient death in any case.
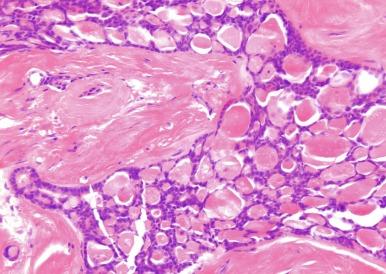
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here