Physical Address
304 North Cardinal St.
Dorchester Center, MA 02124
Myofascial pain is a common source of pain in the neck, low back, and other areas of the body. The term “myofascial pain” encompasses many different painful conditions, including muscle strain, myofascial trigger points (TrPs). It also includes specific myofascial pain syndromes, including piriformis syndrome, iliopsoas-related pain, and pain related to compression of the brachial plexus by the scalene muscles (neurogenic thoracic outlet syndrome). Skeletal muscle consists of muscle fibers under somatic nervous control. Each nerve root innervates a muscle or group of muscles known as a myotome. The muscle belly of skeletal muscles is connected by tendons to the bone. TrPs occur in the body of the muscle, most often located in the center of the muscle.
While much remains to be discovered about the pathophysiology of myofascial pain, several theories have been advanced in recent years. Underlying biomechanical and postural factors may interact with neurologic factors, psychological elements including depression and anxiety, and hormonal and nutritional imbalances. These factors (in total or in part) may create peripheral sensitization, autonomic dysregulation, and ultimately, central spinal cord sensitization, which can amplify the symptoms experienced by patients with myofascial pain. Vasoactive mediators, pro-nociceptive neurotransmitters, and inflammatory mediators including bradykinin, norepinephrine, serotonin, calcitonin gene-related peptide, substance P, tumor necrosis factor-a, and interleukin 1-B have been identified in the hypersensitive loci of TrPs. These substances are pro-nociceptive and sensitize peripheral nociceptors. They are responsible for the sensory experience of myofascial pain, including referred pain and the local twitch response.
The motor phenomena seen in myofascial pain have been hypothesized to be caused by excessive acetylcholine (ACh) leakage, which creates dysfunctional endplates responsible for taut muscle band formation. Excessive ACh release leads to sustained muscle contraction by increased depolarization of the postjunctional endplate. Evidence of maximal sarcomere shortening in TrPs has been found in animal and human subjects. A positive feedback cycle may be created by the interaction of increased ACh release, sarcomere shortening, and the release of sensitizing substances. The hypothalamus-pituitary-adrenocortical and sympathetic-adrenal-medullary system responses to experimentally induced stress in patients with myofascial pain revealed that plasma concentrations of cortisol, epinephrine, and norepinephrine were found to be significantly elevated in myofascial pain patients than in healthy controls.
The taut muscle band present in myofascial pain syndrome has a higher resting tension and contains hypercontracted muscle fibers. Chronicity of this hypercontraction can increase local energy consumption and may cause areas of tissue hypoperfusion and ischemia. Vasoactive mediators are released in the setting of muscle ischemia, leading to increased ACh release, exacerbation of local ischemia, and sensitization of peripheral nociceptors, thereby causing pain. Vasoactive mediators such as those released in the taut bands of MP have been known to sensitize peripheral nociceptive nerve fibers such as those found in skeletal muscle. Nociceptors spontaneously discharge with a lower threshold to painful stimulation and exhibit discharge to non-painful stimuli in a sensitized state. Over time, this heightened abnormal peripheral sensory input creates a state of central neuronal sensitization.
The clinical manifestation of abnormal electrical activity in the TrP is called a local twitch response (LTR) and is believed to be mediated by a segmental spinal reflex. LTR is elicited by deep palpation or needling of the TrP and leads to a brisk muscle contraction in the taut band. The location of the LTR is called the “sensory locus,” which has been correlated histologically with sensory nerve receptors. The “active locus” is the site where spontaneous electrical activity is recorded, the waveforms of which correspond to published reports of motor endplate noise. According to this model, the sensory locus and the active locus act as the nociceptor and the motor endplate and are distributed throughout muscle. Where these align and are highly concentrated, we observe myofascial TrPs. Abnormal spontaneous electrical activity is present at the site of TrPs, with excessive ACh release creating endplate noise seen on electrophysiologic studies at the neuromuscular junction. Spontaneous electrical activity occurs in TrPs more frequently than in normal tissue and displays aberrant patterns in TrPs. Therefore this spontaneous electrical activity is distinct from normal endplate potentials. This abnormal electrical activity detected in TrPs is thought to be directly connected to excessive ACh release.
A careful history and physical exam remain the keystone of diagnosis. The most common presentation of myofascial pain syndrome includes the following diagnostic criteria: regional body pain and stiffness, limited range of motion of the affected muscle, twitch response produced from a taut band, referred pain from a TrP to a zone of reference, and resolution of the symptoms with local anesthesia applied to the TrP. As discussed previously, TrPs are localized painful areas of skeletal muscle containing taut bands that can be exquisitely sensitive to digital pressure. TrPs may be active or latent. Active TrPs are present in patients with painful regional conditions. Latent TrPs are asymptomatic but may be revealed by deep palpation on physical examination. Latent TrPs have been found in the shoulder girdle muscles of 45%–55% of healthy young adults. myofascial pain may occur after an injury, with chronic strain from repetitive microtrauma, or without any clear precipitating event. Aberrant body mechanics or postural abnormality may initiate or further maintain the problem. The quality of pain tends to be a deep “aching” of variable intensity, and the pain is confined to a specific anatomic region. Characteristic referred pain patterns are associated with specific muscles, although these referral patterns are often unreliable. The muscles commonly involved are the trapezius, splenii, cervical and lumbar paraspinal, and quadratus lumborum.
It is essential to have hands on formal training in the physical examination of myofascial pain and TrPs to achieve a reliable result. Musculoskeletal examination should be performed with the objective of identifying potential orthopedic or neurologic dysfunction that could have a role in generating secondary myofascial pain and dysfunction. Although there are no universally accepted diagnostic criteria for myofascial pain, physical findings may help establish a diagnosis. A distinct pattern of TrP findings may reveal itself in myofascial pain syndrome after a given insult. These painful TrPs limit the full range of passive motion in the afflicted muscle group. While these findings have been suggested as diagnostic criteria, investigators have found it problematic to demonstrate consistent agreement in the presence or absence of TrPs among examiners in blinded studies with control groups. Discrepancies in diagnosis may be attributed in part to a lack of a standardized examination technique and variability in the interpretation of examination findings. Variations in muscle anatomy, physical conditioning, and deconditioning can pose obstacles to proper diagnosis as well. The most reproducible diagnostic findings on physical examination include identification of a TrP in an affected muscle, referral of pain to a zone of reference, and reproduction of the patient’s regular pain on physical exam.
Differential diagnosis of myofascial pain should include (1) musculoskeletal and neuropathic disorders such as arthritis, degenerative disk disease, radiculopathy, bursitis, and tendonitis; (2) autoimmune or infectious etiologies; (3) metabolic and endocrine dysfunction including hypothyroidism; (4) psychiatric disorders including depression and anxiety; and (5) fibromyalgia or diffuse amplified musculoskeletal pain.
Imaging studies have only recently demonstrated anatomic changes associated with trigger points. Ultrasound examination combined with Doppler blood flow has been reported to allow visualization of trigger points, and ultrasound imaging can help direct muscle injection techniques (see later). Recently developed techniques using magnetic resonance and ultrasound elastography purport to reveal changes in intramuscular signal consistent with trigger points, but this technology has not yet been validated. ,
Treatment of trigger points commonly includes physical therapy (manual release techniques, stretching and strengthening, conditioning, therapeutic modalities), trigger point injections (TPIs), dry needling and acupuncture, and transcutaneous electrical nerve stimulation. A broad range of treatment modalities have been reported, but weak research methodology continues to limit interpretation of the results. Regarding medications, tizanidine, but not cyclobenzaprine, was noted to be efficacious for acute skeletal muscle spasms. Diazepam, amitriptyline, and lidocaine patch were found to be effective, but literature support for the use of anti-convulsants in treating myofascial pain syndrome is limited. Although clonazepam appears to be effective, its use is limited by the side effects of depression, liver dysfunction, and difficulty weaning the patient from this drug. Nonsteroidal anti-inflammatory drugs (NSAIDs), specifically ibuprofen, appear to be effective when given with other agents, but not as monotherapy.
TPI is a widely used invasive therapy wherein a needle is guided directly into a TrP that has been previously identified on physical examination. TPI is best utilized in a series of injections and as part of a comprehensive treatment plan that includes guided, structured physical therapy. This strategy can be particularly beneficial when TPI is initially employed to reduce pain in patients otherwise intolerant of physical therapy or stretching, allowing the physical modalities to be more effective. The following conditions are contraindications to TPI: (1) infection, systemic or localized; (2) coagulopathy; (3) distorted or complicated anatomy; and (4) patient refusal.
Saline, corticosteroids, various local anesthetics including lidocaine and bupivacaine, botulinum toxin serotype A (BoNT-A), and dry needling have all been used and studied. Stimulation of the LTR in direct needling of the TrP is valuable in achieving immediate effect. There is good evidence to suggest that there is no advantage of one injection therapy over another or of any drug injectate over dry needling. In a systemic review of 23 RCTs, Cummings and White concluded that any effect derived from TPI is likely derived from the needle itself rather than any specific substance injected, as there was no difference in the therapeutic benefit of “wet” needling versus “dry” needling. Their review also suggested that pain reduction with saline TPI is equal to pain reduction with local anesthetic TPI, both significant. Although adding corticosteroid preparation to local anesthetic is common, it has not been reliably shown to reduce pain more than TPI with local anesthetic alone. Despite the widespread practice of TPI for MP, there is no consensus regarding the number of injection points, frequency of administration, and volume or type of injectate. Controlled studies are needed to evaluate the comparative efficacy of TPIs and their potential benefits in long term pain reduction if any.
Botulinum toxin serotype A (BoNT-A) produces sustained and prolonged relaxation of muscles by inhibiting the release of ACh at the motor endplate and is itself an analgesic inhibiting central sensitization. Commercially prepared BoNT-A is expensive and should be employed with care by a well-trained physician. Although this therapy is promising, the results of RCTs have been mixed, and multiple systematic reviews have concluded that the current clinical evidence to support the use of BoNT in the treatment of MPS is inconclusive. Ferrante et al. found no statistically significant improvement compared to placebo with BoNT-A injection when injected directly into painful TrPs for cervicothoracic MP. They concluded that although it is intuitive for the clinician to consider therapeutic injection of BoNT-A as a treatment for MP (given its a priori similarity to TPI), peculiarities inherent to the use of toxin instead of dry needling or local anesthetic must be accounted for (i.e. toxin spread through fascial planes), including the effects of dosing of toxin, volume of injectate, muscles chosen to inject, postural relations and abnormalities, and injection technique. Harden et al. were able to identify a short term (12-week) reduction in MP of chronic tension-type headache with BoNT-A injection compared to placebo. Graboski et al. found no significant difference in BoNT-A versus 0.5% bupivacaine injected into TPs of patients with MPS, though both were effective in reducing pain below the baseline level. Venancio et al. studied 45 MP patients assigned randomly to one of three groups: dry needling, 0.25% lidocaine TPI, and BoNT-A TPI, and assessed over a 12-week period. While all three groups showed favorable response to treatment, the BoNT-A group demonstrated less use of rescue medication and less post-injection local sensitivity. In another study, Nicol et al. reported that BoNT-A injected directly into painful muscle groups using a “follow the pain” and pattern injection technique instead of trigger points led to reduced average numerical pain scores, reduced number of headaches per week, and improvement in general activity and sleep quality of life measures. Similar positive findings were seen in the studies by Benecke et al. and Miller et al., wherein patients with cervical myofascial pain received BoNT-A using a fixed-location injection technique.
After informed consent, the muscle is palpated, and the trigger points are identified and marked. The area is prepared in a sterile fashion, and a 25- to 27-gauge, 1.5 inch needle is inserted into the trigger point. A local twitch may be elicited at this time, or the patient verbally identifies the painful area. After negative aspiration, 0.25% bupivacaine, 0.2% ropivacaine, or 1% lidocaine is injected with or without steroid, either dexamethasone (4 mg in a 30 mL bottle of local anesthetic) or a low dose of particulate steroid (e.g. 20–40 mg of methylprednisolone or triamcinolone diluted in the local anesthetic solution).
Possible complications include bleeding, hematoma, nerve block, and infection. The patient should be monitored closely for bleeding, development of neurologic symptoms (numbness or weakness, urinary or bowel incontinence), or signs of infection. Depending on the location of the injection, the patient should be instructed on the signs and symptoms of pneumothorax (from neck, shoulder, thoracic, and anterior chest wall injections) or local nerve blockade (for example, inadvertent median nerve block with injection of the flexor carpi ulnaris).
The success of the procedure is dependent on the diagnosis and localization of the trigger point. Patients with widespread chronic pain or psychological disorders are less likely to respond to TPI only. Patients with focal muscle pain and the characteristics of myofascial pain can achieve significant relief for days to months with a well-performed TPI. The success of TPI is often dependent on subsequent stretching and strengthening and neuromuscular education of the muscle. The most effective treatment of myofascial pain syndrome is often a multi-disciplinary approach tailored to the individual needs of the patient that incorporates TPI with physical therapy and medication, as noted earlier. Practitioners are counseled to provide appropriate follow up after TPI and encourage patients to address other factors that perpetuate chronic pain (coping, employment, decreased social activity). ,
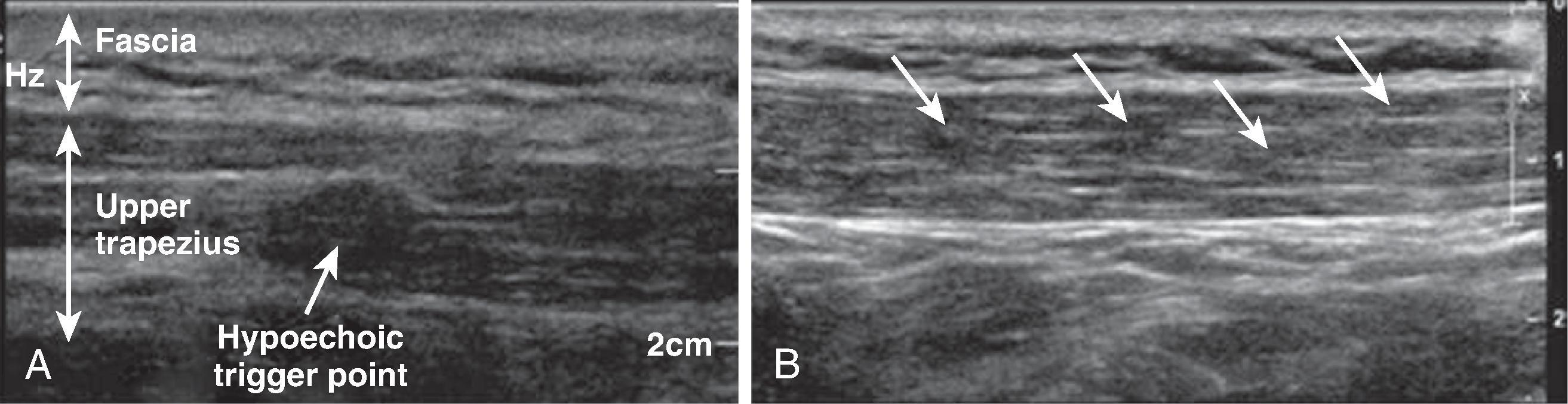
Ultrasound guidance provides several theoretical advantages over blind needle insertion. As stated, it allows visualization of the trigger point ( Fig. 68.1 ). The practitioner is ensured that the needle is penetrating muscle tissue, which may be of use in patients with considerable adipose tissue, and can avoid local anatomic structures that are sensitive to needle penetration or local anesthetic infiltration (i.e. neurovascular structures or viscera). Ultrasound imaging also allows visualization of the muscle twitch response with needle penetration, especially in muscles that are deep or small. An additional use of the ultrasound is to confirm local anesthetic infiltration between fascial planes. It also aids in blocking the spinal accessory nerve for diagnosis of trapezius muscle-related myofascial pain.
Piriformis syndrome is described in detail in Chapter 30 of this text but generally consists of pain in the buttock with or without radiation in the distribution of the ipsilateral sciatic nerve. It is considered the principal pain generator in approximately 8% of patients presenting with the buttock as the origin of pain. The syndrome can be a consequence of an abnormal relationship between the sciatic nerve and the piriformis muscle that results in irritation of the sciatic nerve. A hypertrophic muscle, infection, or invasion of the muscle by a tumor can cause pressure or irritation on the nerve. , In 78%–84% of the population, the sciatic nerve passes in front of the muscle. In 12%–21% of individuals, the divided nerve passes through or posterior to the piriformis and is exposed to muscle contractions, which trigger sciatic symptoms. Piriformis syndrome is a diagnosis of exclusion since there are no standardized diagnostic criteria for it. The syndrome should be considered in patients with buttock pain, tenderness to palpation over the piriformis muscle, and a positive response to provocative maneuvers, including:
Pace’s sign: pain and weakness with seated abduction of the hip against resistance.
Lasègue’s sign (also known as the straight-leg raise test): pain with unresisted flexion, adduction, and internal rotation of the flexed hip.
Freiberg’s sign: pain with forced (i.e. against resistance) internal rotation of the extended hip.
The piriformis muscle is usually injected under imaging guidance because of its small size compared to the gluteus muscles, its proximity to neurovascular structures, and its deep location. Studies have universally shown that the injections of local anesthetics and steroids can significantly reduce pain from piriformis syndrome, which holds true for the most common imaging techniques: fluoroscopic, , ultrasound guided, and computed tomography (CT)-guided. Other imaging guidance techniques include nerve stimulators and electromyography, which were used more often prior to the increased availability of fluoroscopy and consistently improving the image quality of ultrasound. Contraindications are the same as those noted in the section on TPI.
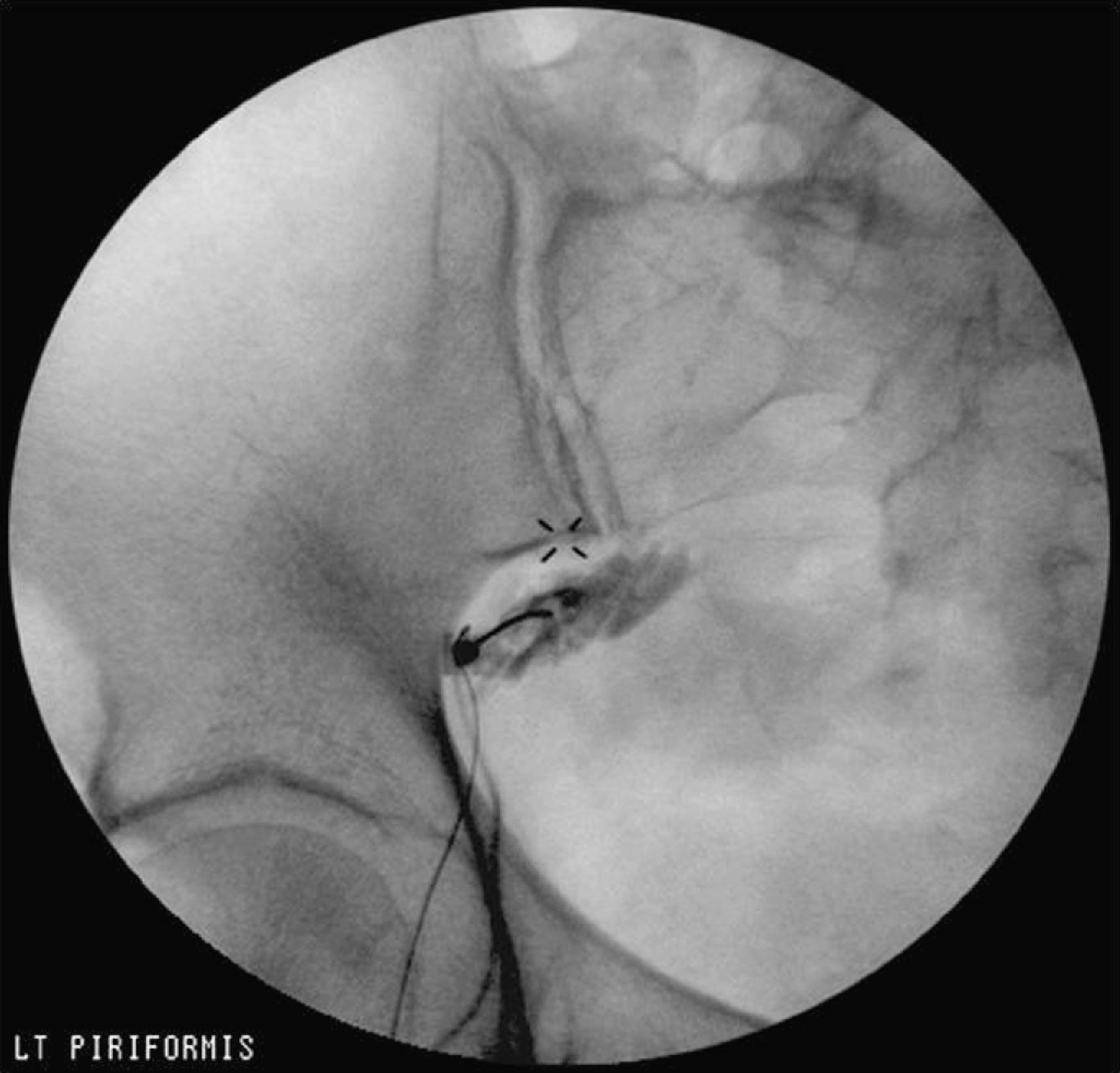
The patient is placed prone on a fluoroscopy table, and the inferior margin of the sacroiliac joint is imaged and marked. The needle insertion site is 1–2 cm caudal and 1–2 cm lateral to the inferior margin of the sacroiliac joint. After sterile preparation and infiltration of local anesthetic, a 7–10 cm insulated needle is inserted and advanced with the nerve stimulator turned on (1 mA, 2 Hz, 0.1 ms) until an evoked motor response of the sciatic nerve is achieved (dorsiflexion, plantar flexion, eversion, inversion) at 0.4–0.6 mA. The needle is then withdrawn slightly until the sciatic stimulation disappears to avoid intraneural injection. Steroid (40 mg of either methylprednisolone or triamcinolone) plus 5 mL of saline is injected around the sciatic nerve—this is helpful, especially in patients with signs of sciatic nerve irritation. The needle is then pulled back 1 cm into the belly of the piriformis muscle, and 1–2 mL of contrast agent is injected. The contrast agent should outline the piriformis muscle belly with no sign of spillage ( Fig. 68.2 ). After the characteristic spread of dye is achieved—outline of the piriformis muscle with clean margins—the local anesthetic solution with steroid is administered.
The ultrasound guided technique permits a direct view of the piriformis muscle and allows examination of the anatomic variation in the relationship between the piriformis and the sciatic nerve. The patient is placed in the prone position with the ultrasound machine on the side opposite the operator. A curved, low-frequency ultrasound probe (2–6 MHz) is used to scan a wider and deeper area. The ultrasound machine should have Doppler to identify the inferior gluteal artery medial to the sciatic nerve and anterior to the piriformis. A 20- to 22-gauge, 10–12 cm long needle is recommended.
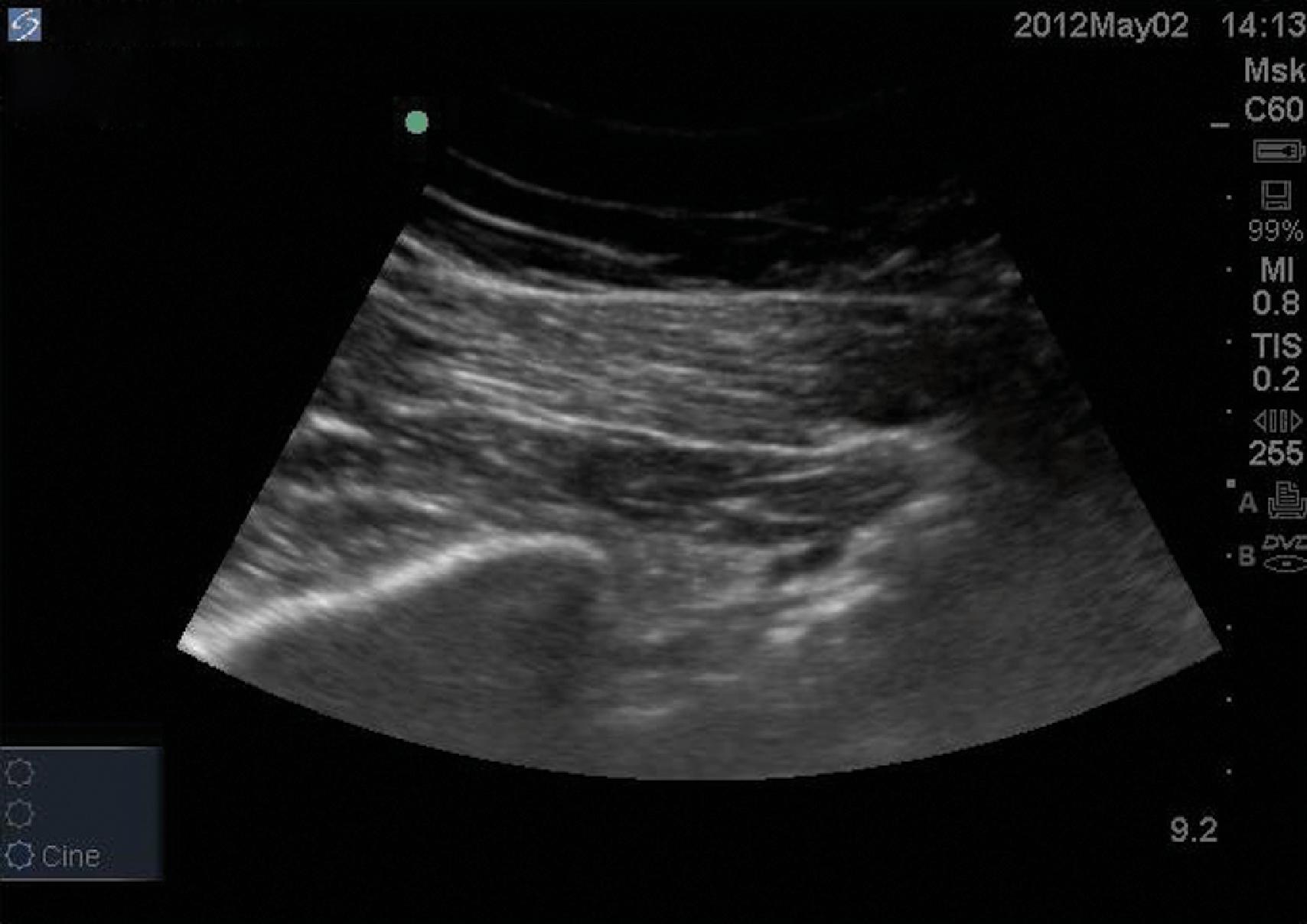
One technique involves positioning the transducer in the short axis (transverse) over the sacroiliac joint, where medially the sacrum will be visible, and laterally the ilium/gluteus maximus muscle complex will be observed. While keeping the sacroiliac joint in the center of the screen, the transducer is moved caudally until the lateral view of the ilium is lost, which indicates that the transducer is over the greater sciatic notch. In this position, the operator will see the hyperechoic lateral portion of the sacrum medially. In the center of the image, the following are visualized: skin and fat in the near field and then the gluteus maximus muscle. Deeper to the gluteus maximus and originating from the anterior and lateral sacral edge is the piriformis muscle with its typical longitudinal fibers ( Fig. 68.3 ). By moving the transducer slightly lower and mildly rotating the left gluteal area clockwise (counterclockwise on the right side), the sciatic nerve becomes visible deeper in the medial aspect of the piriformis. The ischium initially appears as a curved hyperechoic line (posterior acetabulum); more caudally, it becomes a flat line deeper to the piriformis. Another way to find the piriformis is to place the transducer over the line between the greater trochanter and ischial tuberosity. Once the sciatic nerve is identified, it is followed cephalad until the piriformis and gluteus maximus muscles are visualized overlying the sciatic nerve.
To confirm the view of the piriformis muscle, flex the patient’s knee 90 degrees and rotate the hip internally and externally. During this maneuver, the piriformis will slide over the ischium while the position of the gluteus maximus remains stable. While observing the sciatic notch, it is useful to identify the ischial spine and differentiate other muscles close to the piriformis that insert in the area (i.e. the gemelli and obturator muscles).
The piriformis runs in a nearly axial plane between the sacrum and the femur. A lateral-to-medial, in-plane approach is ideal to maximize visualization of the needle; the needle enters the skin 3–4 cm lateral to the lateral edge of the transducer. The needle should cross the skin and fat in a lateral-to-medial, posterior-to-anterior direction and enter the gluteus maximus and then the piriformis in its medial half. Hydrodissection with normal saline (or a nonelectrolyte solution such as 5% dextrose if using stimulation) may help confirm the position of the tip of the needle before administering injectate. After injecting the piriformis muscle, to decrease sciatic nerve irritation, the needle can be advanced and positioned between the piriformis and the sciatic nerve to deposit injectate in that plane.
The most common combination for piriformis injectate is a corticosteroid (CS) plus local anesthetic (LA), often 5 mL of 0.25% bupivacaine plus 40 mg of methylprednisolone or triamcinolone. A retrospective study noted that 76% of patients treated with physical therapy and CS/LA injection had greater than 50% improvement at an average 10.5 month follow up. A prospective study noted that patients had improved outcomes with CS/LA injection for a full year after treatment than with medication and physical therapy alone.
Although steroid injection is common, a 2015 randomized controlled trial (RCT) reported no difference in CS plus LA injection to the piriformis than LA alone. That said, both injection groups were clinically effective in treating pain. It is not uncommon for weakness or numbness to develop in the distribution of the sciatic nerve for the expected duration of the LA.
Botulinum toxin is an increasingly common injection when the duration of effect from LA and CS injections is insufficient. A typical dose would be 100 mouse units in a 2 mL volume. Due to its paralytic effect on the muscle, it causes atrophy and fatty degeneration of the muscle over time, as evidenced by magnetic resonance imaging (MRI). This reduction in muscular volume would decrease pressure on the sciatic nerve and is the mechanism of analgesia, , , but is a more profoundly effective treatment when combined with physical therapy. , , Several uncontrolled studies have evaluated the use of botulinum toxin, often in combination with physical therapy, and reported high rates of success that lasted for months ( Table 68.1 ). , , A controlled study demonstrated the superiority of botulinum toxin over placebo injection for ten weeks. Other controlled studies have reported that the efficacy of botulinum toxin is superior to that of local anesthetic/steroid or normal saline injections for the treatment of piriformis syndrome. , ,
| Study | Muscle Injected | Type of Study | Groups Compared | Results |
|---|---|---|---|---|
| Porta | Piriformis | Randomized, controlled | Botox A (100 units) vs. steroid | Pain scores were significantly lower in the Botox group 60 days after injection |
| Lang | Piriformis | Open label | Botox B (5000 units) | Significant reduction in buttock and hip pain up to 16 weeks |
| Fishman et al. | Piriformis | Prospective, dose ranging | Botox B (5000 7500, 10,000, 12,500 units)* | 12,500 units superior to 10,000 units 12 weeks after injection |
| Yoon et al. | Piriformis | Prospective, open label | Botox A (150 units) vs. lidocaine/dexamethasone (5 mg) | Improvement in SF-36 subscales (pain, physical and social functioning, vitality, general health) at four weeks |
| Childers et al. | Piriformis | Double-blind, crossover | Botox A (100 units) vs. vehicle | Results better with Botox injection |
| Porta | Iliopsoas | Randomized, controlled | Botox A (150 units) vs. steroid | Pain scores were significantly lower in the Botox group 60 days after injection |
| De Andrés et al. | Iliopsoas | Randomized, controlled | Botox A (50 units) vs. 0.25% bupivacaine or NaCl (injected into the contralateral side) | No improvement with either treatment; no difference between groups; trend toward a decrease in pain scores with Botox |
| Jordan et al. | Scalene | Prospective, open label | Botox (12–15 units per muscle) | 64% of patients had >50% pain reduction for at least one month |
| Jordan et al. | Scalene | Retrospective | Botox A (12–15 units per anterior/middle scalene); fluoroscopy/EMG guidance compared to ultrasound/EMG guidance | Results comparable: 91% good results with ultrasound and 81% with fluoroscopy |
| Christo et al. | Scalene | Prospective | Botox (20 units) into the scalene muscle | Significant relief of pain for three months |
When comparing clonidine versus normal saline as an additive to bupivacaine, a prospective study found a significantly lower pain score at six months in the clonidine group.
Injection of the piriformis may improve pain related outcomes for several months. However, the published studies are limited by poor study methodology and reporting, inconsistent outcome selection, small sample sizes, heterogeneous patient populations, and the absence of standardized diagnostic criteria. The combination of a sciatic nerve conduction study with the flexion, abduction, internal rotation test may predict those who will respond to physical therapy. More research into this topic comparing different treatment combinations would be welcome, but at present, it does appear that injection into the piriformis is an effective treatment modality for a patient that has failed conservative therapy.
Become a Clinical Tree membership for Full access and enjoy Unlimited articles
If you are a member. Log in here